Springs and Springs-Dependent Taxa of the Colorado River Basin, Southwestern North America: Geography, Ecology and Human Impacts
Abstract
:1. Introduction
2. Methods
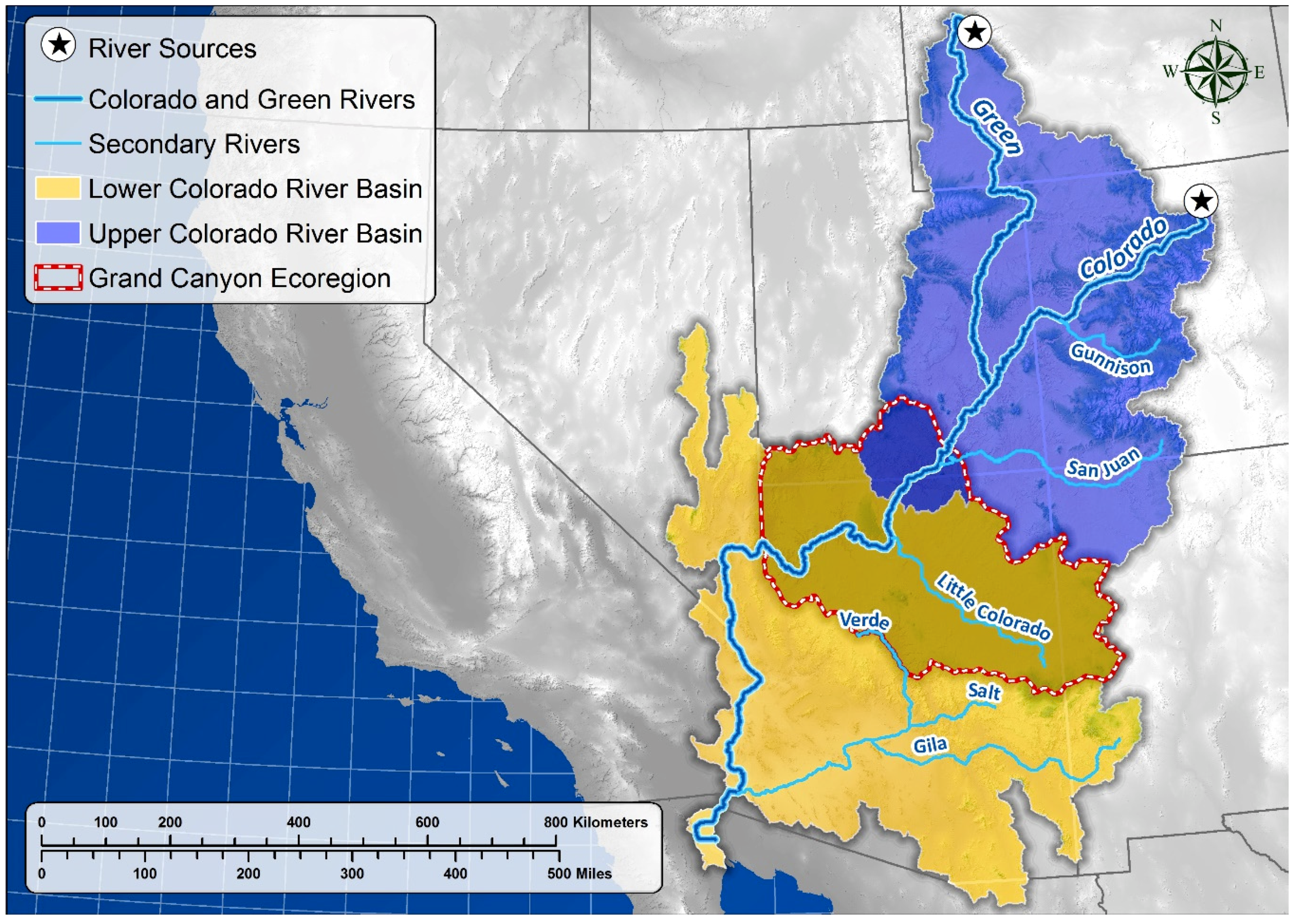
Study Area
3. Springs and Associated Species Data Sources
4. Human Demography Data and Analyses
5. Results and Discussion
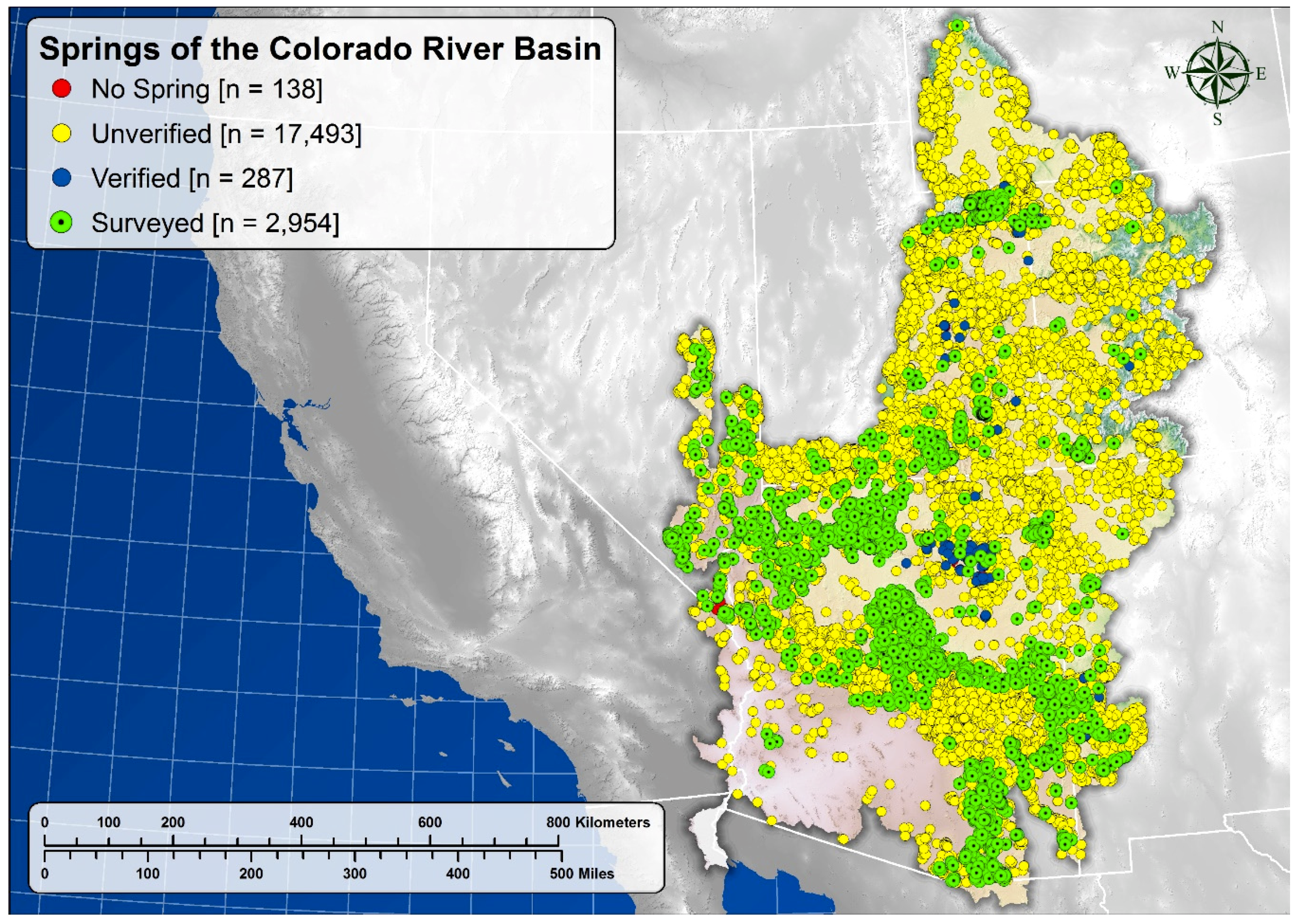
5.1. Springs Distribution
5.2. Habitat Area
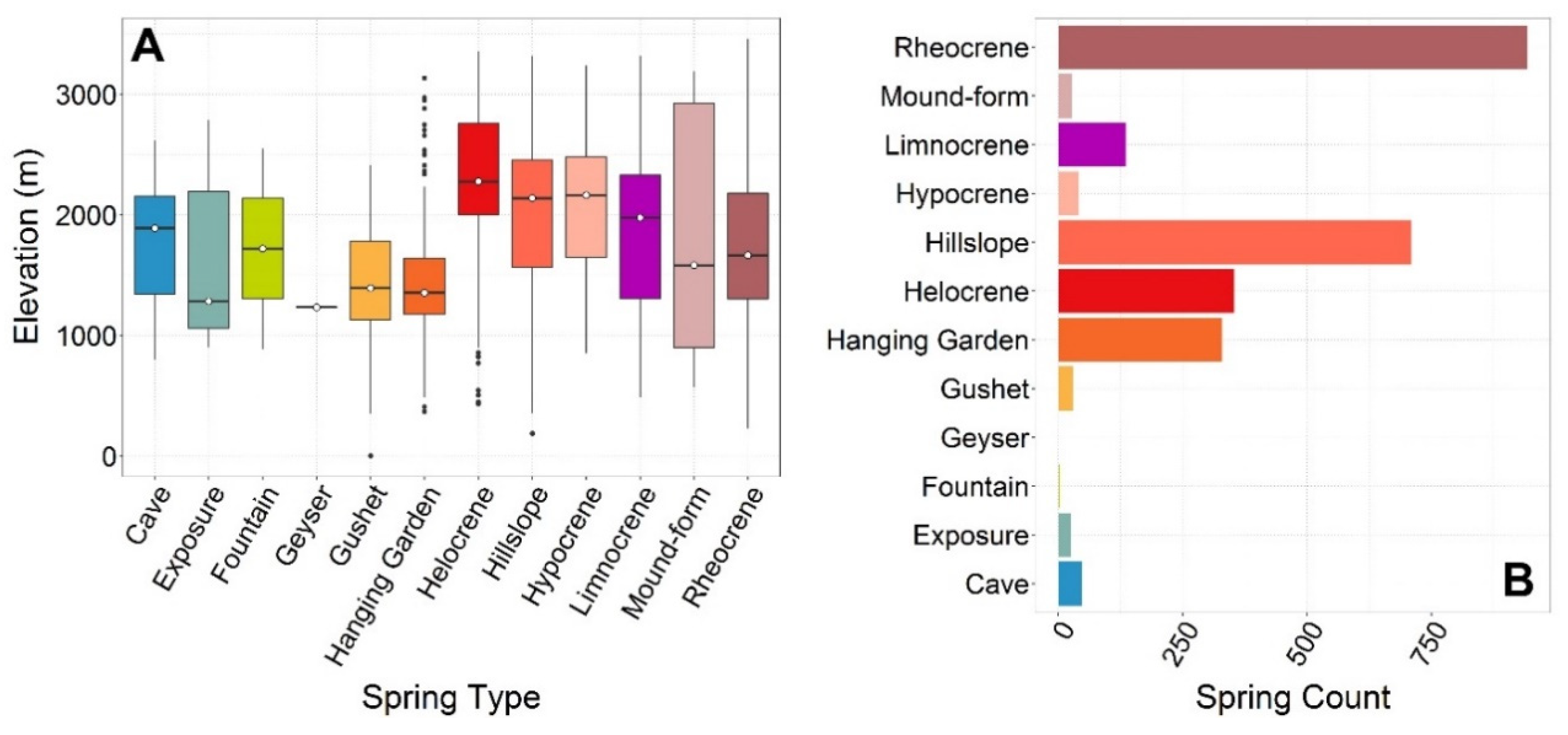
5.3. Springs Types
≈ Mound-form ≈ Exposure > Limnocrene > Fountain > Geyser.
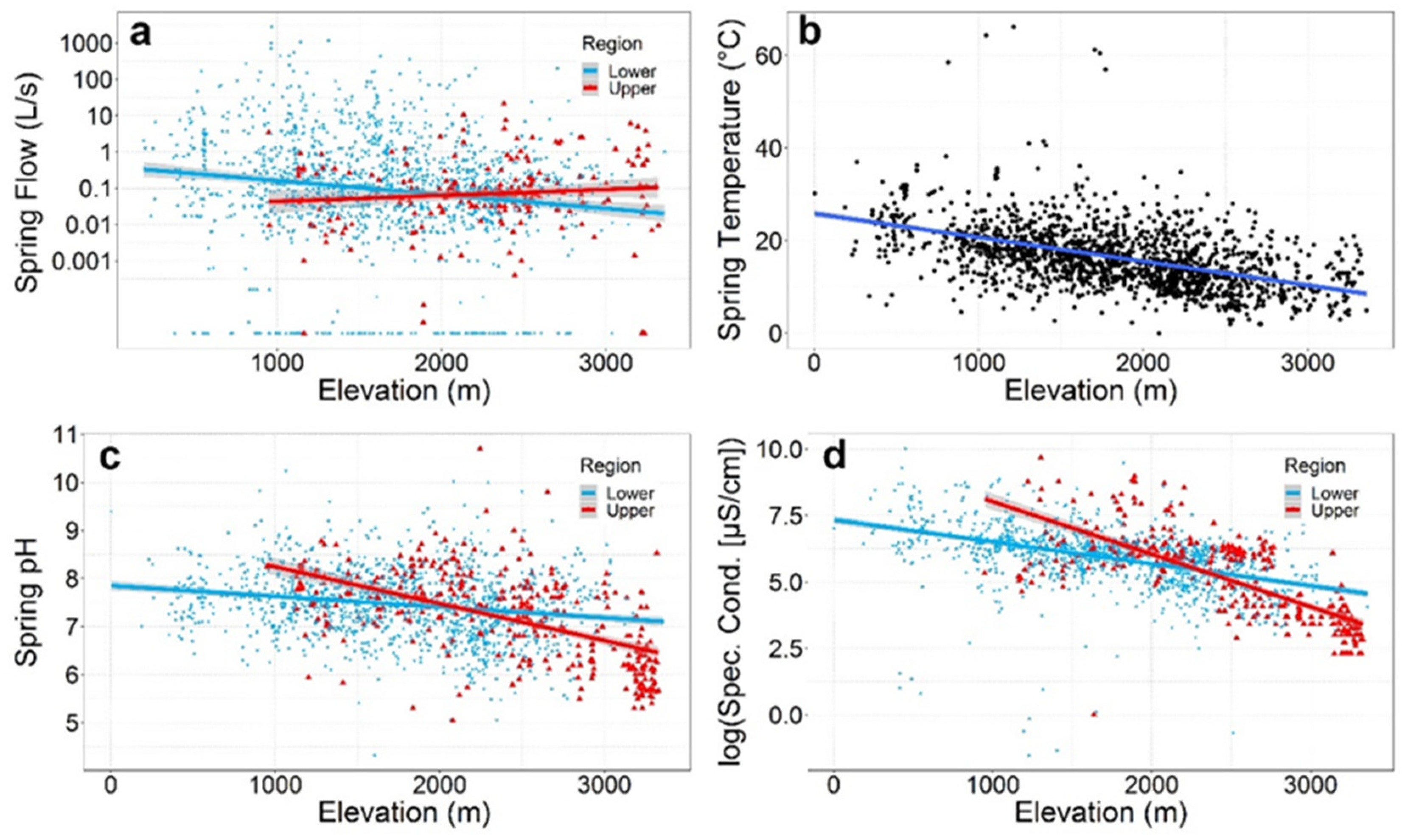
5.4. Flow
5.5. Water Temperature and Geochemistry
5.6. Springs-Dependent Taxa
5.7. SDT Patterns
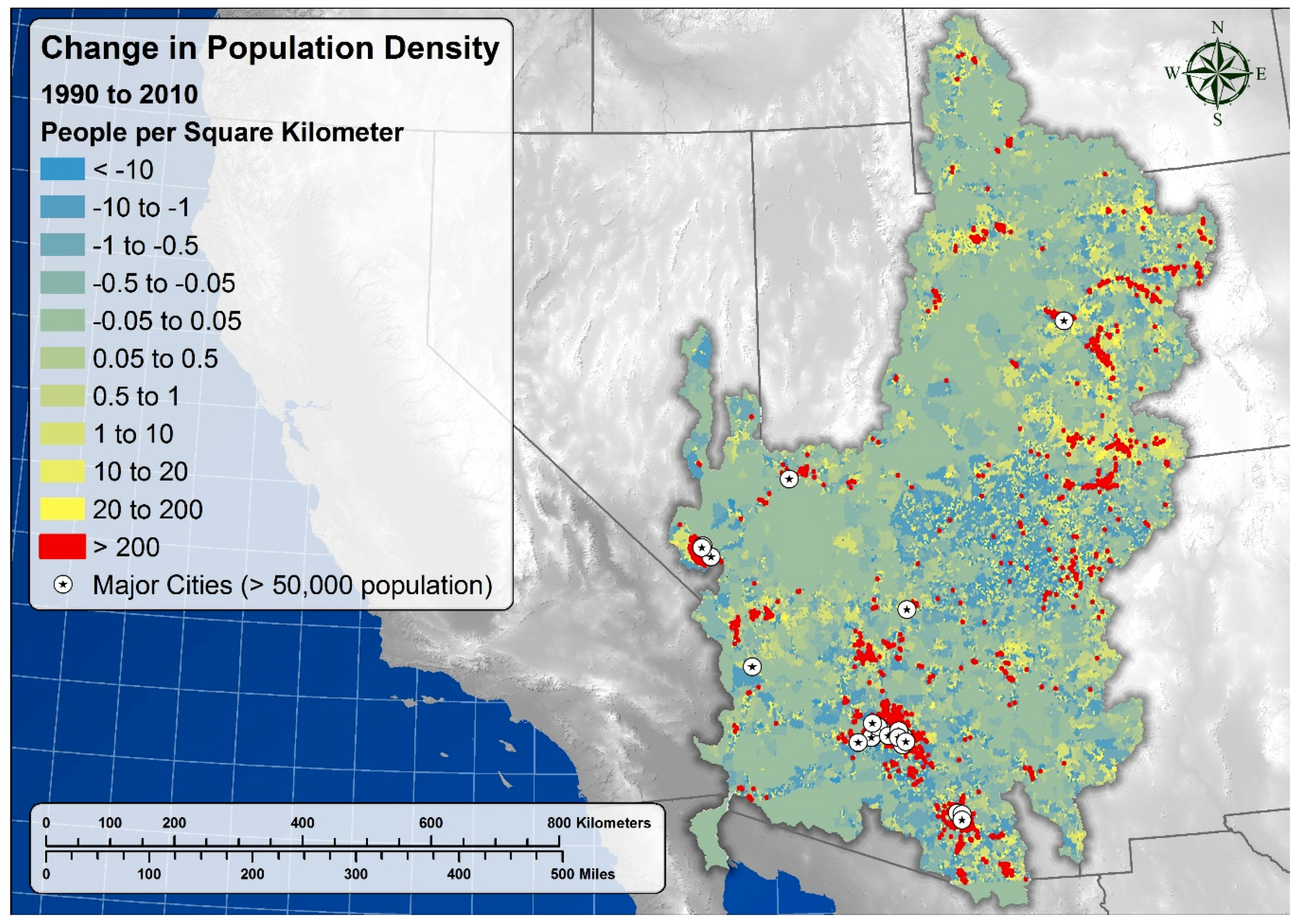
5.8. Socio-Cultural Significance and Demographic Impacts
6. CRB Springs Rehabilitation
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Perla, B.S.; Stevens, L.E. Biodiversity and productivity at an undisturbed spring in comparison with adjacent grazed riparian and upland habitats. In Aridland Springs in North America: Ecology and Conservation; Stevens, L.E., Meretsky, V.J., Eds.; Univ. Ariz. Press: Tucson, AZ, USA, 2008; pp. 230–243. [Google Scholar]
- Lindeman, R.L. The trophic-dynamic aspect of ecology. Ecology 1942, 23, 399–417. [Google Scholar] [CrossRef]
- Odum, H.T. Trophic structure and productivity of Silver Springs, Florida. Ecol. Monogr. 1957, 27, 55–112. [Google Scholar] [CrossRef]
- Stevens, L.E.; Meretsky, V.J. (Eds.) Aridland Springs in North America: Ecology and Conservation; Univ. Ariz. Press: Tucson, AZ, USA, 2008. [Google Scholar]
- Brown, J.; Bach, L.; Aldous, A.; Wyers, A.; Degagné, J. Groundwater-dependent ecosystems in Oregon: An assessment of their distribution and associated threats. Front. Ecol. Environ. 2010, 9, 97–102. [Google Scholar] [CrossRef]
- Knight, R.L. Silenced Springs: Moving from Tragedy to Hope, 1st ed.; Florida Springs Institute: High Springs, FL, USA, 2015; pp. 1–369. [Google Scholar]
- Abele, S.L. (Ed.) Nevada Springs Conservation Plan. In Springs Conservation Plan Working Group; The Nature Conservancy: Reno, NV, USA, 2011. [Google Scholar]
- Grand Canyon Wildlands Council, Inc. Inventory of 100 Arizona Strip Springs, Seeps and Natural Ponds; Arizona Water Protection Fund: Phoenix, AZ, USA, 2002.
- Utah Division of Wildlife Resources; Nevada Department of Wildlife; Nevada Natural Heritage Program; The Nature Conservancy; U.S. Fish and Wildlife Service, Region 8; U.S. Fish and Wildlife Service, Region 6; U.S. Bureau of Land Management (Utah); U.S. Bureau of Land Management (Nevada); U.S. Forest Service, Intermountain Region; U.S. Forest Service Dixie, Fishlake and Humboldt Toiyabe National Forests; et al. Conservation Agreement for Springsnails in Nevada and Utah; Nevada Department of Wildlife: Reno, NV, USA; Utah Division of Wildlife Resources: Salt Lake City, UT, USA, 2017.
- Stevens, L.E.; Johnson, R.R.; Estes, C. The watershed continuum: A conceptual fluvial-riparian ecosystem model. In Riparian Research and Management: Past, Present, Future, Volume 2; Johnson, R.R., Carothers, S.W., Finch, D.M., Kingsley, K.J., Stanley, J.T., Eds.; Gen. Tech. Rept RMRS-GTR-303; U.S. Department of Agriculture Forest Service, Rocky Mountain Research Station: Ft. Collins, CO, USA, in press.
- Miller, M.P.; Buto, S.; Susong, D.D.; Rumsey, C.A. The importance of base flow in sustaining surface water flow in the Upper Colorado River Basin. Water Resour. Res. 2016, 52, 3547–3562. [Google Scholar] [CrossRef]
- US Bureau of Reclamation. Colorado River Basin Water Supply and Demand Study. 2012. Available online: https://www.usbr.gov/lc/region/programs/crbstudy/finalreport/index.html (accessed on 2 October 2019).
- Patrick, R. Rivers of the United States, Volume V Part A: The Colorado River; John Wiley & Sons: New York, NY, USA, 2000. [Google Scholar]
- US Geological Survey. Surface-Water Annual Statistics for the Nation; Station 09419756; US Printing Office: Washington, DC, USA, 2019.
- National Research Council. Colorado River Basin Water Management; National Academies Press: Washington, DC, USA, 2007; Available online: https://www.nap.edu/catalog/11857/colorado-river-basin-water-management-evaluating-and-adjusting-to-hydroclimatic (accessed on 2 October 2019).
- Stevens, L.E. The biogeographic significance of a large, deep canyon: Grand Canyon of the Colorado River, southwestern USA. In Global Advances in Biogeography; Stevens, L.E., Ed.; InTech Publications: Rijeka, Croatia, 2012; pp. 169–208. ISBN 978-953-51-0454-4. [Google Scholar]
- US 67th Congress. Colorado River Compact. H.R. Volume 6877. 1922. Available online: https://www.usbr.gov/lc/region/pao/pdfiles/crcompct.pdf (accessed on 1 October 2019).
- Waring, G.L. A Natural History of the Intermountain West: Its Ecological and Evolutionary Story; Univ. of Utah Press: Salt Lake City, UT, USA, 2011. [Google Scholar]
- Rumsey, C.A.; Miller, M.P.; Susong, D.D.; Tillman, F.; Anning, D.W. Regional scale estimates of baseflow and factors influencing baseflow in the Upper Colorado River Basin. J. Hydrol. Reg. Stud. 2015, 4, 91–107. [Google Scholar] [CrossRef] [Green Version]
- US Department of Agriculture. 4FRI Rim Country Project draft Environmental Impact Statement, Volume 2: Apache-Sitgreaves, Coconino, and Tonto National Forests Coconino, Yavapai, Gila, and Navajo Counties, Arizona; US Dept. Agric. For. Serv. SW Region MB-R3-04-34; US Department of Agriculture: Washington, DC, USA, 2019.
- Junghans, K. Springs Ecosystem Distribution and Density for Improving Stewardship and Springs as Sources of Temperate and Tropical Perennial Streams. ProQuest Dissertations Publishing, 2016. Available online: http://search.proquest.com/docview/1808836106/ (accessed on 1 November 2019).
- Castle, S.L.; Thomas, B.; Reager, J.T.; Rodell, M.; Swenson, S.C.; Famiglietti, J. Groundwater depletion during drought threatens future water security of the Colorado River Basin. Geophys. Res. Lett. 2014, 41, 5904–5911. [Google Scholar] [CrossRef] [Green Version]
- Milly, P.C.; Dunne, K.A. Colorado River flow dwindles as warming-driven loss of reflective snow energizes evaporation. Science 2020, 367, 1252–1255. [Google Scholar] [CrossRef]
- Fassnacht, S.R.; Venable, N.; McGrath, D.; Patterson, G. Sub-Seasonal snowpack trends in the Rocky Mountain National Park area, Colorado, USA. Water 2018, 10, 562. [Google Scholar] [CrossRef] [Green Version]
- Springs Stewardship Institute. Springs Online. Springs and Springs-Dependent Species Online Database, 2020. Available online: http://springsdata.org/ (accessed on 17 October 2019).
- Christensen, N.S.; Wood, A.; Voisin, N.; Lettenmaier, D.P.; Palmer, R.N. The effects of climate change on the hydrology and water resources of the Colorado River Basin. Clim. Chang. 2004, 62, 337–363. [Google Scholar] [CrossRef]
- US Geological Survey. North America Rivers and Lakes. USGS ScienceBase-Catalog. 2019a. Available online: https://www.sciencebase.gov/catalog/item/4fb55df0e4b04cb937751e02 (accessed on 17 October 2019).
- Ranatunga, T.; Tong, S.T.; Sun, Y.; Yang, Y.J. A total water management analysis of the Las Vegas Wash watershed, Nevada. Phys. Geogr. 2014, 35, 220–244. [Google Scholar] [CrossRef]
- Dickinson, W.R. The Basin and Range Province as a composite extensional domain. Int. Geol. Rev. 2002, 44, 1–38. [Google Scholar] [CrossRef] [Green Version]
- Sheppard, P.R.; Comrie, A.; Packin, G.; Angersbach, K.; Hughes, M. The climate of the US Southwest. Clim. Res. 2002, 21, 219–238. [Google Scholar] [CrossRef] [Green Version]
- PRISM. PRISM Spatial Climate Datasets for the Conterminous United States. 2004. Available online: http://prism.oregonstate.edu/documents/PRISM_datasets.pdf (accessed on 1 November 2019).
- Timmons, J.M.; Karlstrom, K.E. Grand Canyon Geology: Two Billion Years of Earth’s History. Geol. Soc. Amer. Spec. Pap. 2012, 489, 1–156. [Google Scholar]
- Beck, D.A.; Wilson, J. Discharge and physical-property measurements from Virgin River Narrows, Arizona, to Lake Mead, Nevada, February 12, 2003. Sci. Investig. Rep. 2006. [Google Scholar] [CrossRef] [Green Version]
- Burke, K.J.; Harcksen, K.A.; Stevens, L.E.; Andress, R.J.; Johnson, R.J. Collaborative rehabilitation of Pakoon Springs in Grand Canyon–Parashant National Monument, Arizona. In The Colorado Plateau VI: Science and Management at the Landscape Scale; Huenneke, L.F., Van Riper, C., Hays-Gilpin, K.A., Eds.; Univ. Ariz. Press: Tucson, AZ, USA, 2015; pp. 312–328. [Google Scholar]
- Davis, C.J.; Springer, A.E.; Stevens, L.E. Have aridland springs restoration projects been effective in restoring hydrology, geomorphology, and invertebrate and plant species composition comparable to natural springs with minimal anthropogenic disturbance? Collab. Environ. Evid. Rev. 2011, 10-002 (SR87). Available online: http://www.environmentalevidence.org/Documents/Completed Reviews/SR87.pdf (accessed on 21 July 2018).
- Blasch, K.; Hoffmann, J.P.; Graser, L.F.; Bryson, J.R.; Flint, A.L. Hydrogeology of the upper and middle Verde River watersheds, central Arizona. Sci. Invest. Rep. 2006. [Google Scholar] [CrossRef] [Green Version]
- Haney, J.; Turner, D.S.; Springer, A.E.; Stromberg, J.C.; Stevens, L.E.; Pearthree, P.A.; Supplee, V. Ecological Implications of Verde River Flows; Arizona Water Institute, The Nature Conservancy, and the Verde River Basin Partnership: Tucson, AZ, USA, 2008; pp. 1–114. [Google Scholar]
- Verde River Basin Partnership. Verde River Basin water-resources primer. Friends of the Verde River, Cottonwood, Ariz. 2017. Available online: https://verderiver.org/wp-content/uploads/2017/11/verde-river-basin-water-resources-primer.pdf (accessed on 2 February 2019).
- Inkenbrandt, P.; Thomas, K.; Jordan, J.L. Regional groundwater flow and water quality in the Virgin River Basin and surrounding areas, Utah and Arizonz. Utah Geol. Surv. Rept. Investig. 2013, 272, 1–46. [Google Scholar]
- US 81st Congress. Upper Colorado River Basin Compact. S. Volume 790, 1949. Available online: https://www.loc.gov/law/help/statutes-at-large/81st-congress/session-1/c81s1ch48.pdf (accessed on 1 October 2019).
- Prudic, D.E.; Harrill, J.R.; Burbey, T.J. Conceptual evaluation of regional ground-water flow in the carbonate-rock province of the Great Basin, Nevada, Utah and adjacent states. US Geol. Surv. Prof. Pap. 1995, 1409–D, 1–102. [Google Scholar]
- Mueller, G.A.; Marsh, P.C. Lost, a Desert River and Its Native Fishes: A Historical Perspective of the Lower Colorado River; US Geol. Surv. Information and Tech. Rept. USFS/BRD/ITR-2002-0010; US Geological Survey: Reston, VA, USA, 2002.
- Ledbetter, J.D.; Stevens, L.E.; Hendrie, M.; Leonard, A. Ecological inventory and assessment of springs ecosystems in Kaibab National Forest, northern Arizona. In Proceedings of the 12th Biennial Conference of Research on the Colorado Plateau; Ralston, B.E., Ed.; US Geological Survey Scientific Investigations Report 2015–5180, U.S. Geological Survey: Reston, VA, USA, 2016; pp. 25–40. [Google Scholar]
- Grand Canyon Wildlands Council, Inc. An Inventory, Assessment, and Development of Recovery Priorities for Arizona Strip Springs, Seeps, and Natural Ponds: A Synthesis of Information; Arizona Water Protection Fund: Phoenix, AZ, USA, 2001.
- Stevens, L.E.; Springer, A.E.; Ledbetter, J.D. Springs Ecosystem Inventory and Assessment Protocols; Museum of Northern Arizona Springs Stewardship Institute: Flagstaff, AZ, USA, 2016. [Google Scholar]
- The PLANTS Database; National Plant Data Team: Greensboro, NC, USA, 2020. Available online: http://plants.usda.gov (accessed on 25 April 2020).
- Esri. ArcGIS Pro; Esri: Redlands, CA, USA, 2019. [Google Scholar]
- US Geological Survey; US Department of Agriculture Natural Resources Conservation Service. Federal Standards and Procedures for the National Watershed Boundary Dataset. Tech. Methods 2013, 11, 1–63. [Google Scholar]
- Gesch, D.; Oimoen, M.; Greenlee, S.; Nelson, C.; Steuck, M.; Tyler, D. The national elevation dataset. Photogram. Engin. Remote Sens. 2002, 68, 5–32. [Google Scholar]
- US Department of Agriculture Natural Resources Conservation Service. Watershed Boundary Dataset. 2014. Available online: http://datagateway.nrcs.usda.gov (accessed on 15 March 2019).
- Jenness, J. Tools for Graphics and Shapes; Jenness Enterprises: Flagstaff, AZ, USA, 2011; Available online: http://www.jennessent.com/arcgis/shapes_graphics.htm (accessed on 18 October 2018).
- Esri. ArcMap. ArcGIS for Desktop; Esri: Redlands, CA, USA, 2017. [Google Scholar]
- Simley, J.D.; Carswell, W.J., Jr. The National Map—Hydrography; U.S. Geological Survey Fact Sheet 2009-3054; U.S. Geological Survey: Reston, VA, USA, 2009.
- Jenness, J. Calculating landscape surface area from digital elevation models. Wildl. Soc. Bull. 2004, 32, 829–839. [Google Scholar] [CrossRef]
- Jenness, J. DEM Surface Tools: An ArcGIS Extension for Analyzing Raster Elevation Datasets; Jenness Enterprises: Flagstaff, AZ, USA, 2011; Available online: http://www.jennessent.com/arcgis/surface_area.htm (accessed on 27 June 2012).
- Springer, A.E.; Stevens, L.E. Spheres of discharge of springs. Hydrogeol. J. 2008, 17, 83–93. [Google Scholar] [CrossRef]
- Mundorff, J.C. Nonthermal Springs of Utah. Utah Geol. Mineral. Surv. Water-Resour. Bull. 1971, 16, 1–70. [Google Scholar]
- Mundorff, J.C. Major thermal springs of Utah. US Geol. Mineral. Surv. Water-Resour. Bull. 1970, 13, 1–87. [Google Scholar]
- Springer, A.E.; Boldt, E.M.; Junghans, K.M. Local vs. regional groundwater flow delineation from stable isotopes at western North American springs. Groundwater 2017, 56, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Kreamer, D.K.; Stevens, L.E.; Ledbetter, J.D. Groundwater Dependent Ecosystems–Science, Challenges, and Policy; Adelana, S.M., Ed.; Groundwater; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2015; pp. 205–230. [Google Scholar]
- Springer, A.E.; Stevens, L.E.; Harms, R. Inventory and Classification of Selected National Park Service Springs on the Colorado Plateau; NPS Cooperative Agreement Number CA 1200-99-009; National Park Service: Flagstaff, AZ, USA, 2006.
- US Bureau of the Census. 1990 Census Summary File 1 ASCII Files and CD-ROM Discs. 2013. Available online: http://www2.census.gov/census_1990/1990STF1.html (accessed on 28 July 2016).
- US Census Bureau. Census 2000 Summary File 1 Delivered via FTP, 2011a. Available online: ftp://ftp2.census.gov/census_2000/datasets/Summary_File_1/ (accessed on 28 July 2016).
- US Census Bureau. 2010 Census Summary File 1 Delivered via FTP. 2011b. Available online: http://www2.census.gov/census_2010/04-Summary_File_1/ (accessed on 28 July 2016).
- Sivinski, R.; Tonne, P. Survey and Assessment of Aridland Springs Ciénegas in the Southwest Region; NM Energy, Minerals and Natural Resources Department: Santa Fe, NM, USA, 2011.
- Sinclair, D. Geomorphology Influences Springs Ecosystem Physical and VegetationCharacteristics in the Grand Canyon Ecoregion. Master’s Thesis, Northern Arizona University, Flagstaff, AZ, USA, 2018. [Google Scholar]
- Tobin, B.W.; Springer, A.E.; Kreamer, D.K.; Schenk, E.R. Review: The distribution, flow, and quality of Grand Canyon Springs, Arizona (USA). Hydrogeol. J. 2017, 26, 721–732. [Google Scholar] [CrossRef]
- Spence, J.R. Spring-supported vegetation along the Colorado River on the Colorado Plateau: Floristics, vegetation structure, and environment. In Aridland Springs in North America: Ecology and Conservation; Stevens, L.E., Meretsky, V.J., Eds.; University of Arizona Press: Tucson, AZ, USA, 2008. [Google Scholar]
- North, E.G. Land Snail Diversity and Distribution in the Grand Canyon Ecoregion, Southwestern USA. Master’s Thesis, Northern Arizona University, Flagstaff, AZ, USA, 2017. [Google Scholar]
- Howitt, R.; Medellín, J.; MacEwan, D.; Lund, J.R.; Sumner, D. Economic Analysis of the 2014 Drought for California Agriculture; University of California Davis Center for Watershed Sciences: Davis, CA, USA, 2014; Available online: https://DroughtReport_23July2014.pdf (accessed on 1 November 2019).
- Monroe, S.A.; Antweiler, R.C.; Hart, R.J.; Taylor, H.E.; Truini, M.; Rihs, J.R.; Felger, T.J. Chemical characteristics of ground-water discharge along the south rim of Grand Canyon in Grand Canyon National Park, Arizona, 2000-2001. Sci. Investig. Report 2005, 2005, 2004–5146. [Google Scholar] [CrossRef]
- Crossey, L.; Fischer, T.P.; Patchett, P.J.; Karlstrom, K.E.; Hilton, D.R.; Newell, D.; Huntoon, P.; Reynolds, A.C.; De Leeuw, G.A. Dissected hydrologic system at the Grand Canyon: Interaction between deeply derived fluids and plateau aquifer waters in modern springs and travertine. Geology 2006, 34, 25. [Google Scholar] [CrossRef]
- Newell, D.; Crossey, L.; Karlstrom, K.E.; Fischer, T.P.; Hilton, D.R. Continental-scale links between the mantle and groundwater systems of the western United States: Evidence from travertine springs and regional He isotope data. GSA Today 2005, 15, 4. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Found. Stat. Comput.: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 5 March 2019).
- Chimner, R.A.; Lemly, J.M.; Cooper, D.J. Mountain fen distribution, types and restoration priorities, San Juan Mountains, Colorado, USA. Wetlands 2010, 30, 763–771. [Google Scholar] [CrossRef]
- Springer, A.E.; Stevens, L.E.; Ledbetter, J.D.; Schaller, E.M.; Gill, K.M.; Rood, S.B. Ecohydrology and stewardship of Alberta springs ecosystems. Ecohydrology 2015, 8, 896–910. [Google Scholar] [CrossRef]
- Stevens, L.E.; Schenk, E.R.; Springer, A.E. Springs ecosystem classification. Ecol. Appl. In press.
- Stevens, L.E. The springs biome, with an emphasis on arid regions. Ref. Modul. Earth Syst. Environ. Sci. 2020. [Google Scholar] [CrossRef]
- Han, W.S.; Lu, M.; McPherson, B.J.; Keating, E.H.; Moore, J.; Park, E.; Watson, Z.T.; Jung, N.-H. Characteristics of CO2-driven cold-water geyser, Crystal Geyser in Utah: Experimental observation and mechanism analyses. Geofluids 2012, 13, 283–297. [Google Scholar] [CrossRef]
- Hendrickson, D.A.; Minckley, W.L. Ciénegas vanishing climax communities of the American Southwest. Desert Plants 1985, 6, 131–175. [Google Scholar]
- Woodbury, A.M. Biotic relationships of Zion Canyon, Utah with special reference to succession. Ecol. Monogr. 1933, 3, 147–245. [Google Scholar] [CrossRef]
- Welsh, S.L. On the distribution of Utah’s hanging gardens. Great Basin Nat. 1989, 49, 1–30. [Google Scholar]
- Malonson, G.P. Habitat and plant distributions in hanging gardens of the Narrows, Zion National Park, Uta. Great Basin Nat. 1980, 40, 178–182. [Google Scholar]
- Weissinger, R.; Perkins, D.; Dinger, E. Biodiversity, water chemistry, physical characteristics, and anthropo-genic disturbance gradients of sand-stone springs on the Colorado Plateau. West. N. Am. Nat. 2012, 72, 393–406. [Google Scholar] [CrossRef] [Green Version]
- Springs Stewardship Institute; Sky Island Alliance. Arizona Springs Restoration Handbook. 2016. Available online: http://SpringStewardshipInstitute.org (accessed on 1 November 2019).
- Pool, D.R.; Blasch, K.W.; Callegary, J.B.; Leake, S.A.; Graser, L.F. Regional Groundwater-Flow Model of the Redwall-Muav, Coconino, and Alluvial Basin Aquifer Systems of Northern and Central Arizona; U.S. Geol. Surv. Sci. Investig. Rep. 2010–5189; US Geological Survey: Reston, VA, USA, 2011; pp. 1–101.
- Johnson, P.W.; Sanderson, R.B. Spring Flow into the Colorado River, Lees Ferry to Lake Mead, Arizona; Arizona State Land Department: Phoenix, AZ, USA, 1968.
- Huntoon, P.W. Variability of karstic permeability between unconfined and confined aquifers, Grand Canyon region, Arizona. Environ. Eng. Geosci. 2000, 6, 155–170. [Google Scholar] [CrossRef]
- Schenk, E.R.; O’Donnell, F.; Springer, A.E.; Stevens, L.E. The impacts of tree stand thinning on groundwater recharge in aridland forests. Ecol. Eng. 2020, 145, 105701. [Google Scholar] [CrossRef]
- Jones, N.A.; Hansen, J.; Springer, A.E.; Valle, C.; Tobin, B.W. Modeling intrinsic vulnerability of complex karst aquifers: Modifying the COP method to account for sinkhole density and fault location. Hydrogeol. J. 2019, 27, 2857–2868. [Google Scholar] [CrossRef]
- Johnson, R.H.; DeWitt, E.; Arnold, L.R. Using hydrogeology to identify the source of groundwater to Montezuma Well, a natural spring in Central Arizona, USA: Part 1. Environ. Earth Sci. 2012, 67, 1821–1835. [Google Scholar] [CrossRef]
- Winograd, I.; Thordarson, W. Hydrogeologic and hydrochemical framework, south-central Great Basin, Nevada-California, with special reference to the Nevada Test Site. Prof. Paper 1975, 712. [Google Scholar] [CrossRef] [Green Version]
- Drakos, P.G.; Riesterer, J.W.; Bemis, K. Recharge sources and characteristics of springs on the Zuni Reservation, New Mexico. In New Mexico Geological Society Guidebook: Geology of Route 66 Region; In Proceedings of the 64th Field Conference: Flagstaff to Grants; New Mexico Geological Society: Socorro, NM, USA, 2013; pp. 205–213. [Google Scholar]
- Breckenridge, R.M.; Hinckley, B.S. Thermal springs of Wyoming. Geol. Surv. Wyo. 1978, 60, 1–104. [Google Scholar]
- Beck, D.A.; Ryan, R.; Veley, R.J.; Harper, D.P.; Tanko, D.J. Water-surface elevations, discharge, and water-quality data for selected sites in the Warm Springs area near Moapa, Nevada. Open-File Rep. 2006, 1–230. [Google Scholar] [CrossRef] [Green Version]
- Moran, M.J.; Wilson, J.; Beard, L.S. Hydrogeology and sources of water to select springs in Black Canyon, south of Hoover Dam, Lake Mead National Recreation Area, Nevada and Arizona. Sci. Investig. Rep. 2015, 1–61. [Google Scholar] [CrossRef] [Green Version]
- Blinn, D. The extreme environment, trophic structure, and ecosystem dynamics of a large, fishless desert spring: Montezuma Well, Arizona. In Aridlands Springs in North America: Ecology and Conservation; Stevens, L.E., Meretsky, V.J., Eds.; Univ. Ariz. Press: Tucson, AZ, USA, 2008; pp. 98–126. [Google Scholar]
- Hershler, R.; Liu, H.-P. Annotated checklist of freshwater truncatelloidean gastropods of the western United States, with an illustrated key to the genera. U.S. Bur. Land Mgt. Tech. Note 2017, 449. [Google Scholar] [CrossRef] [Green Version]
- Scoppettone, G.G. Interactions between native and nonnative fishes of the Upper Muddy River, Nevada. Trans. Am. Fish. Soc. 1993, 122, 599–608. [Google Scholar] [CrossRef]
- Scoppettone, G.G.; Johnson, D.M.; Hereford, M.E.; Rissler, P.; Fabes, M.; Salgado, A.; Shea, S. Relative abundance and distribution of fishes and crayfish at Ash Meadows National Wildlife Refuge, Nye County, Nevada, 2010–2011. U.S. Geol. Surv. Open-File Rep. 2012, 2012, 1141. [Google Scholar]
- Bedford, B.L.; Godwin, K.S. Fens of the United States: Distribution, characteristics, and scientific connection versus legal isolation. Wetlands 2003, 23, 608–629. [Google Scholar] [CrossRef]
- Crews, S.C.; Stevens, L.E. Spiders of Ash Meadows National Wildlife Refuge, Nevada. Southwest. Nat. 2009, 54, 331–340. [Google Scholar] [CrossRef]
- Hershler, R.; Liu, H.-P.; Stevens, L.E.; Hershler, R. A new springsnail (Hydrobiidae: Pyrgulopsis) from the Lower Colorado River Basin, Northwestern Arizona. West. N. Am. Nat. 2016, 76, 72–81. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, T.M.; Schaechter, M. (Eds.) Topics in Ecological and Environmental Microbiology; Academic Press: Waltham, MA, USA, 2012. [Google Scholar]
- Potter-McIntyre, S.L.; Williams, J.; Phillips-Lander, C.; O’Connell, L. Taphonomy of microbial biosignatures in spring deposits: A comparison of modern, Quaternary, and Jurassic examples. Astrobiology 2017, 17, 216–230. [Google Scholar] [CrossRef] [PubMed]
- Marciano-Cabral, F. Biology of Naegleria spp. Microbiol. Rev. 1988, 52, 114–133. [Google Scholar] [CrossRef] [PubMed]
- Goveditch, F.R.; Bain, B.A. All about the leeches of Montezuma Well. 2005. Available online: http://www.nps.gov/moca/naturescience/upload/montezuma_well_leeches.pdf (accessed on 2 February 2020).
- Hershler, R.; Liu, H.-P.; Howard, J. Springsnails: A new conservation focus in western North America. Bioscience 2014, 64, 693–700. [Google Scholar] [CrossRef]
- Ng, D.; Barnes, J.R. Distributional study of the Zion Snail, Physa zionis, Zion National Park, Utah. Gr. Basin Nat. 1986, 46, 310–315. [Google Scholar]
- Spamer, E.E.; Bogan, W.E. Mollusca of the Grand Canyon and vicinity, Arizona: New and revised data on diversity and distributions, with notes on Pleistocene-Holocene mollusks of the Grand Canyon. Proc. Acad. Nat. Sci. Phila. 1993, 144, 21–68. [Google Scholar]
- Shelley, R.M.; Stevens, L.E. Discovery of the milliped, Tylobolus utahensis Chamberlin, in Arizona (Spirobolida: Spirobolidae). West. N. Am. Nat. 2004, 63, 541–542. Available online: https://ojs.lib.byu.edu/wnan/index.php/wnan/article/view/456/322 (accessed on 1 December 2019).
- Belk, D.; Fugate, M. Two new Branchinecta (Crustacea: Anostraca) from the Southwestern United States. Southwest. Nat. 2000, 45, 111. [Google Scholar] [CrossRef]
- Wang, D.; Holsinger, J.R. Systematics of the subterranean amphipod genus Stygobromus (Crangonyctidae) in western North America, with emphasis on species of the hubbsi group. Amphipacifica 2001, 3, 39–147. [Google Scholar]
- McCafferty, W.P.; Randolph, R.P.; Jacobus, L.M. Mayflies of the Intermountain West. Mem. Am. Entomol. Inst. 2012, 85, 1–317. [Google Scholar]
- McCafferty, W.P. Moribaetis mimbresaurus, new species (Ephemeroptera: Baetidae): First representation of the genus north of Mexico. Proc. Entomol. Soc. Wash. 2007, 109, 696–699. [Google Scholar]
- Stevens, L.E.; Bailowitz, R.A. Odonata Biogeography in the Grand Canyon Ecoregion, Southwestern USA. Ann. Èntomol. Soc. Am. 2009, 102, 261–274. [Google Scholar] [CrossRef]
- Stevens, L.E.; Polhemus, J.T. Biogeography of aquatic and semi-aquatic Heteroptera in the Grand Canyon ecoregion, southwestern USA. Monogr. West. N. Am. Nat. 2008, 4, 38–76. [Google Scholar]
- Baumann, R.W.; Gaufin, A.R.; Surdick, R.F. The stoneflies (Plecoptera) of the Rocky Mountain. Mem. Am. Entomol. Soc. 1977, 31, 1–208. [Google Scholar]
- Kondratieff, B.C.; Baumann, R.W. A review of the stoneflies of Colorado with description of a new species of Capnia (Plecoptera: Capniidae). Trans. Am. Entomol. Soc. 2002, 128, 385–401. [Google Scholar]
- Brown, W.S. The Aquatic Insects of Gunnison County, Colorado. 2005. Available online: www.gunisoninsects.org (accessed on 15 November 2019).
- Baumann, R.W.; Sheldon, A.L.; Bottorff, R.L. Stoneflies (Plecoptera) of Nevada. Monogr. West. N. Am. Nat. 2017, 10, 1–138. [Google Scholar] [CrossRef]
- Baumann, R.W.; Olson, C.A. Confirmation of the stonefly genus Anacroneuria (Plecoptera: Perlidae) from the Nearctic Region with the description of a new species from Arizona. Southwest. Nat. 1984, 29, 489. [Google Scholar] [CrossRef]
- Brown, H.P. A new species of Psephenus from Arizona (Coleoptera, Psephenidae). Coleop. Bull. 1970, 24, 34–38. [Google Scholar]
- Larson, D.; Alarie, Y.; Roughley, R. Predaceous Diving Beetles (Coleoptera: Dytiscidae) of the Nearctic Region, with Emphasis on the Fauna of Canada and Alaska; NRC Research Press: Ottawa, ON, Canada, 2011. [Google Scholar]
- Stevens, L.E.; Huber, R.L. Biogeography of tiger beetles (Cicindelidae) in the Grand Canyon Ecoregion, Arizona and Utah. Cicindela 2004, 5, 41–64. [Google Scholar]
- US Fish and Wildlife Service. Southern Arizona’s Stephan’s Riffle Beetle Believed Extinct: Species Withdrawn as Candidate for Endangered Species Act Protection. U.S. Fish and Wildlife Service Conserving the Nature of America. 2016. Available online: https://www.fws.gov/news/ShowNews.cfm?ref=southern-arizona%E2%80%99s-stephan%E2%80%99s-riffle-beetle-believed-extinct-&_ID=35838 (accessed on 1 November 2019).
- Sublette, J.E.; Stevens, L.E.; Shannon, J.P. Chironomidae (Diptera) of the Colorado River in Grand Canyon, Arizona, U.S.A., I: Taxonomy and ecology. Gr. Basin Nat. 1998, 58, 97–146. [Google Scholar]
- Stevens, L.E.; Ramberg, F.B.; Darsie, R.F. Biogeography of Culicidae (Diptera) in the Grand Canyon region, Southwestern, U.S.A. Pan-Pac. Èntomol. 2008, 84, 92–109. [Google Scholar] [CrossRef]
- Mathis, W.; Mathis, D. Checklist of shore flies (Diptera: Ephydridae) from Grand Staircase-Escalante National Monument. In Invertebrates, Arthropods (Primarily Insects) of the Grand Staircase-Escalante National Monument; Nelson, C.R., Baumann, R.W., Eds.; Appendix 3: 1–36; Grand Staircase-Escalante National Monument: Kanab, UT, USA, 2001. [Google Scholar]
- Blinn, D.W.; Ruiter, D.E. Tolerance values of stream caddisflies (Trichoptera) in Oak Creek, a desert perennial stream in Arizona. Southwest, Nat. 2006, 51, 326–337. [Google Scholar] [CrossRef]
- Blinn, D.W.; Ruiter, D.E. Caddisfly (Trichoptera) Assemblages along Major River Drainages in Arizona. West. N. Am. Nat. 2009, 69, 299–308. [Google Scholar] [CrossRef]
- Tuskes, P.M.; Mcgowan-Tuskes, A. Aquatic moths of the genus Petrophila and their biology in Oak Creek, Arizona (Crambidae). J. Lepidopterists’ Soc. 2019, 73, 43–53. [Google Scholar] [CrossRef]
- Miller, R.R.; Minckley, W.L. Fishes of Arizona. Copeia 1974, 1974, 1008. [Google Scholar] [CrossRef]
- US Fish and Wildlife Service. Desert Pupfish (Cyprinodon macularius) 5-Year Review: Summary and Evaluation; US Fish and Wildlife Service: Washington, DC, USA, 2010.
- Rinne, J.N.; Minckley, W.L. Native Fishes of Arid Lands: A Dwindling Resource of the Desert Southwest; US Department of Agriculture, Forest Service, Rocky Mountain Forest and Range Experiment Station: Fort Collins, CO, USA, 1991; Volume 206. [CrossRef] [Green Version]
- Brennan, T.C.; Holycross, A.T. A Field Guide to Amphibians and Reptiles in Arizona; Arizona Game and Fish Department: Phoenix, AZ, USA, 2006.
- Harper, K.T.; St. Clair, L.L.; Thorne, K.H.; Hess, W.M. (Eds.) Natural History of the Colorado Plateau and Great Basin; Univ. Colorado Press: Niwot, CO, USA, 1994; pp. 1–294. [Google Scholar]
- Arizona Game and Fish Department. Wildlife: Arizona’s Amphibians; Arizona Game and Fish Department: Phoenix, AZ, USA, 2020.
- Hoffmeister, D.F. Mammals of Arizona; University of Arizona Press: Tucson, AZ, USA, 1986. [Google Scholar]
- Himes, C.M.; Kenagy, G.J. Influence of montane isolation and refugia on population structure of Sorex palustris in western North America. J. Mammal. 2010, 91, 1000–1010. [Google Scholar] [CrossRef] [Green Version]
- Stevens, L.E.; Buck, K.A.; Brown, B.T.; Kline, N. Dam and geomorphic influences on Colorado River waterbird distribution, Grand Canyon, Arizona. Reg. Rivers Res. Manag. 1997, 13, 151–169. [Google Scholar] [CrossRef]
- Heidel, B. Wyoming Plant Species of Concern, March 2018; Wyoming Natural Diversity Database: Laramie, WY, USA, 2018. [Google Scholar]
- Phillips, B.G.; Phillips, A.M., III; Schmidt-Bernzott, M.A. Annotated checklist of vascular plants of Grand Canyon National Park; Grand Canyon Natural History Association Monograph No. 7; Grand Canyon Natural History Association: Grand Canyon, AZ, USA, 1987. [Google Scholar]
- SEINet. Online plant specimen database: Arizona-New Mexico Chapter. 2019. Available online: http://swbiodiversity.org/seinet/index.php (accessed on 1 November 2019).
- Arizona Rare Plant Commission. Arizona Rare Plant Field Guide; Arizona Game and Fish Department: Phoenix, AZ, USA, 2006.
- Licher, M.; Rink, G. Cyperaceae. Sedge family. Part 1: Family description, key to the genera, and Carex L. Canotia 2015, 11, 1–97. [Google Scholar]
- Willig, M.; Kaufman, D.; Stevens, R. Latitudinal gradients of biodiversity: Pattern, process, scale, and synthesis. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 273–309. [Google Scholar] [CrossRef]
- Spalink, D.; Pender, J.; Escudero, M.; Hipp, A.L.; Roalson, E.H.; Starr, J.R.; Waterway, M.; Bohs, L.; Sytsma, K.J. The spatial structure of phylogenetic and functional diversity in the United States and Canada: An example using the sedge family (Cyperaceae). J. Syst. Evol. 2018, 56, 449–465. [Google Scholar] [CrossRef] [Green Version]
- Felger, R.S. Flora of the Gran Desierto and Río Colorado of Northwest New Mexico; University of Arizona Press: Tucson, AZ, USA, 2000. [Google Scholar]
- Ezcurra, E.; Felger, R.S.; Russell, A.; Equihua, M. Freshwater islands in a desert sand sea: The hydrology, flora, and phytogeography of the Gran Desierto oases of northwestern Mexico. Desert Plants 1988, 9, 35–44, 55–63. [Google Scholar]
- Danks, H.; Williams, D.D. Arthropods of springs, with particular reference to Canada: Synthesis and needs for research. Memoirs Èntomol. Soc. Can. 1991, 123, 203–217. [Google Scholar] [CrossRef]
- De Granade, R.; Stevens, L.E. Desert oases: Aridland springs as diverse biocultural ecosystems. In Encyclopedia of the World’s Biomes; Elsevier: Cambridge, UK, in press.
- Gleick, P.H. Bottled and Saold: The Story Behind Our Obsession with Bottled Water; Island Press: Washington, DC, USA, 2010. [Google Scholar]
- US Census Bureau. United States census; US Census Bureau: Washington, DC, USA, 2020. Available online: Census.gov (accessed on 15 January 2020).
- Choy, J. 7 Lessons in Groundwater Management from the Grand Canyon State; Stanford Woods Institute for the Environment, Water in the West: Stanford, CA, USA, 2015; Available online: https://waterinthewest.stanford.edu/news-events/news-press-releases/7-lessons-groundwater-management-grand-canyon-state (accessed on 9 February 2020).
- Utah Department of Environmental Quality. Public Water Systems with Water Sources in Oil and Gas Fields: Map; Utah Department of Environmental Quality: Salt Lake City, UT, USA, 2018. Available online: https://Deq.utha.gov (accessed on 12 January 2020).
- Cartwright, J.M.; Dwire, K.A.; Freed, Z.; Hammer, S.J.; McLaughlin, B.; Misztal, L.W.; Schenk, E.R.; Spence, J.R.; Springer, A.E.; Stevens, L.E. Oases of the future? Evaluating springs as potential hydrologic refugia in drying climates. Front. Ecol. Environ. In press.
- Stevens, L.E.; Ayers, T.J. The biodiversity and distribution of alien vascular plant and animals in the Grand Canyon region. In Invasive Exotic Species in the Sonoran Region; Tellman, B., Ed.; University of Arizona Press: Tucson, AZ, USA, 2002; pp. 241–265. [Google Scholar]
- Garner, T.W.J.; Perkins, M.W.; Govindarajulu, P.; Seglie, D.; Walker, S.; Cunningham, A.A.; Fisher, M.C. The emerging amphibian pathogen Batrachochytrium dendrobatidis globally infects introduced populations of the North American bullfrog, Rana catesbeiana. Boil. Lett. 2006, 2, 455–459. [Google Scholar] [CrossRef] [Green Version]
- Scoppettone, G.G.; Rissler, P.H.; Gourley, C.; Martinez, C. Habitat restoration as a means of controlling non-native fish in a Mojave Desert oasis. Restor. Ecol. 2005, 13, 247–256. [Google Scholar] [CrossRef]
- Paffett, K.; Stevens, L.E.; Springer, A.E. Ecological assessment and rehabilitation prioritization for improving springs ecosystem stewardship. Wetl. Stream Rapid Assess. 2018, 475–487. [Google Scholar] [CrossRef]
- Gurrieri, J.T. Rangeland Water Developments at Springs: Best Practices for Design, Rehabilitation, and Restoration; US Dept. Agric. For. Serv. Rocky Mtn Res. Sta. Gen. Tech. Rep. RMRS-GTR-405; U.S. Department of Agriculture Forest Service, Rocky Mountain Research Station: Ft. Collins, CO, USA, 2020; pp. 1–21.
- Kodric-Brown, A.; Brown, J.H. Native fishes, exotic mammals, and the conservation of desert springs. Front. Ecol. Environ. 2007, 5, 549–553. [Google Scholar] [CrossRef]
- Relict Leopard Frog Conservation Team. Conservation agreement and conservation assessment and strategy for the Relict Leopard Frog (Rana onca [= Lithobates onca]); Nevada Department of Wildlife: Las Vegas, NV, USA, 2016.
- Congressional Research Service. Management of the Colorado River: Water Allocations, Drought, and the Federal Role; Congr. Res. Serv. Rep. R45546; US Printing Office: Washington, DC, USA, 2019. Available online: https://crsreports.congress.gov/product/pdf/R/R45546 (accessed on 2 April 2020).
- Schwinning, S.; Belnap, J.; Bowling, D.R.; Ehleringer, J.R. Sensitivity of the Colorado Plateau to change: Climate, ecosystems, and society. Ecol. Soc. 2008, 13. [Google Scholar] [CrossRef] [Green Version]
| Upper CRB Springs | Lower CRB Springs | Overall CRB Springs | ||||
|---|---|---|---|---|---|---|
| Variable | Descriptive Statistics | SLR Elev Model | Descriptive Statistics | SLR Elev Model | Descriptive Statistics | SLR Elev Model |
| Planview surface basin area (km2) | 283,384 | --- | 344,440 | --- | 627,823 | --- |
| Number of reported springs | 8052 | --- | 12,820 | --- | 20,872 | --- |
| Mean springs area (m2), estimated total springs habitat area ± 95% CI (km2) (% of total land area) * | 3700, 29.9 ± 9.8 (0.011%) | --- | 1200, 15.5 ± 3.76 (0.005%) | --- | 2200, 45.4 ± 4.4 (0.007%) | --- |
| Perennial stream length (km) | 25,821 | --- | 64,351 | --- | 90,172 | --- |
| Springs discharge + 95% CI L/s (median; N) * | 0.74 ± 0.44 (0.07; N = 211) | Y = −3.92 + 0.001X (F1205 = 6.86, R2 = 0.03, P = 0.009) | 0.10 ± 0.48 (0.11; N = 1476) | Y = −0.41 − 0.001X (F1,1362 = 46.6, R2 = 0.03, P < 0.0001) | 0.09 ± 0.04 (0.10; N = 1687) | Y = −0.61 − 0.001X (F1,1569 = 47.4, R2 = 0.03, P < 0.0001) |
| Mean water temperature ± 95% CI (°C) (median; N) * | 13.7 ± 0.77 (13.5; N = 281) | Y = 22.53 − 0.004X (F1279 = 54.8, R2 = 0.16, P < 0.0001) | 17.1 ± 0.4 (16.4; N = 1364) | Y = 26.28 − 0.005X (F1,1362 = 428, R2 = 0.24, P < 0.0001) | 16.5 ± 0.3 (15.9; N = 1645) | Y = 25.87 − 0.005X (F1,1643 = 557.0, R2 = 0.25, P < 0.0001) |
| Mean pH ± 95% CI (median; N) * | 7.19 ± 0.09 (7.35; N = 356) | Y = 9.01 − 0.001X (F1354 = 165, R2 = 0.32, P < 0.0001) | 7.47 ± 0.04 (7.50; N = 1335) | Y = 7.85 − 0.0002X (F1,1333 = 56.1, R2 = 0.04, P <0.0001) | 7.41 ± 0.04 (7.47; N = 1691) | Y = 8.08 − 0.0004X (F1,1689 = 205, R2 = 0.11, P < 0.0001) |
| Mean electrical conductivity (μS/cm) ± 95% CI (median; N) * | 616.1 ± 153.8 (263.5; N = 334) | Y = 10.0 − 0.002X (F1332 = 404, R2 = 0.55, P < 0.0001) | 642.7 ± 59.9 (414.56; N = 1292) | Y = 7.33 − 0.001 (F1,1290 = 360, R2 = 0.22, P < 0.0001) | 637.2 ± 57.0 (398; N = 1626) | Y = 7.72 − 0.001X (F1,1624 = 781, R2 = 0.32, P < 0.0001) |
| Taxon | Estimated Number of SDT | % of Total SDT |
|---|---|---|
| Plants | 62 | 18.8 |
| Mollusca | 21 | 6.4 |
| Non-Insecta Arthropoda | 7 | 2.1 |
| Insecta—Ephemeroptera | 21 | 6.4 |
| Insecta—Odonata | 37 | 11.2 |
| Insecta—Hemiptera | 22 | 6.7 |
| Insecta—Plecoptera | 30 | 9.1 |
| Insecta—Coleoptera | 22 | 6.7 |
| Insecta—Trichoptera | 47 | 14.2 |
| Insecta—Lepidoptera | 9 | 2.7 |
| Chordata—Fish | 33 | 10.0 |
| Chordata—Amphibia | 9 | 2.7 |
| Chordata—Repilomorphs | 4 | 1.2 |
| Chordata—Aves | 1 | 0.3 |
| Chordata—Mammalia | 5 | 1.5 |
| Plants | 62 | 18.8 |
| Invertebrates | 216 | 65.5 |
| Vertebrates | 52 | 15.8 |
| Total | 330 | 100.0 |
| Basin Name | Within Entire Basin | At Spring Locations | ||||
|---|---|---|---|---|---|---|
| Census Block 1990 Density | Census Block 2010 Density | Census Block Mean Annual Change in Density 1990–2010 | Spring Count | Mean Spring 2010 Density | Mean Annual Change in Spring Density 1990–2010 ± 95% CI | |
| Upper Basin | 2.02 | 3.07 | 0.05 | 8052 | 3.36 ± 1.89 | 0.11 ± 0.06 |
| Lower Basin | 11.36 | 22.92 | 0.58 | 12,820 | 6.7 ± 3.24 | 0.12 ± 0.11 |
| Full Basin | 7.26 | 14.00 | 0.34 | 20,872 | 5.41 ± 2.10 | 0.12 ± 0.07 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stevens, L.E.; Jenness, J.; Ledbetter, J.D. Springs and Springs-Dependent Taxa of the Colorado River Basin, Southwestern North America: Geography, Ecology and Human Impacts. Water 2020, 12, 1501. https://doi.org/10.3390/w12051501
Stevens LE, Jenness J, Ledbetter JD. Springs and Springs-Dependent Taxa of the Colorado River Basin, Southwestern North America: Geography, Ecology and Human Impacts. Water. 2020; 12(5):1501. https://doi.org/10.3390/w12051501
Chicago/Turabian StyleStevens, Lawrence E., Jeffrey Jenness, and Jeri D. Ledbetter. 2020. "Springs and Springs-Dependent Taxa of the Colorado River Basin, Southwestern North America: Geography, Ecology and Human Impacts" Water 12, no. 5: 1501. https://doi.org/10.3390/w12051501





