Spatial and Temporal Characterization of Escherichia coli, Suspended Particulate Matter and Land Use Practice Relationships in a Mixed-Land Use Contemporary Watershed
Abstract
:1. Introduction
2. Methods
2.1. Study Site Description
2.2. Data Collection
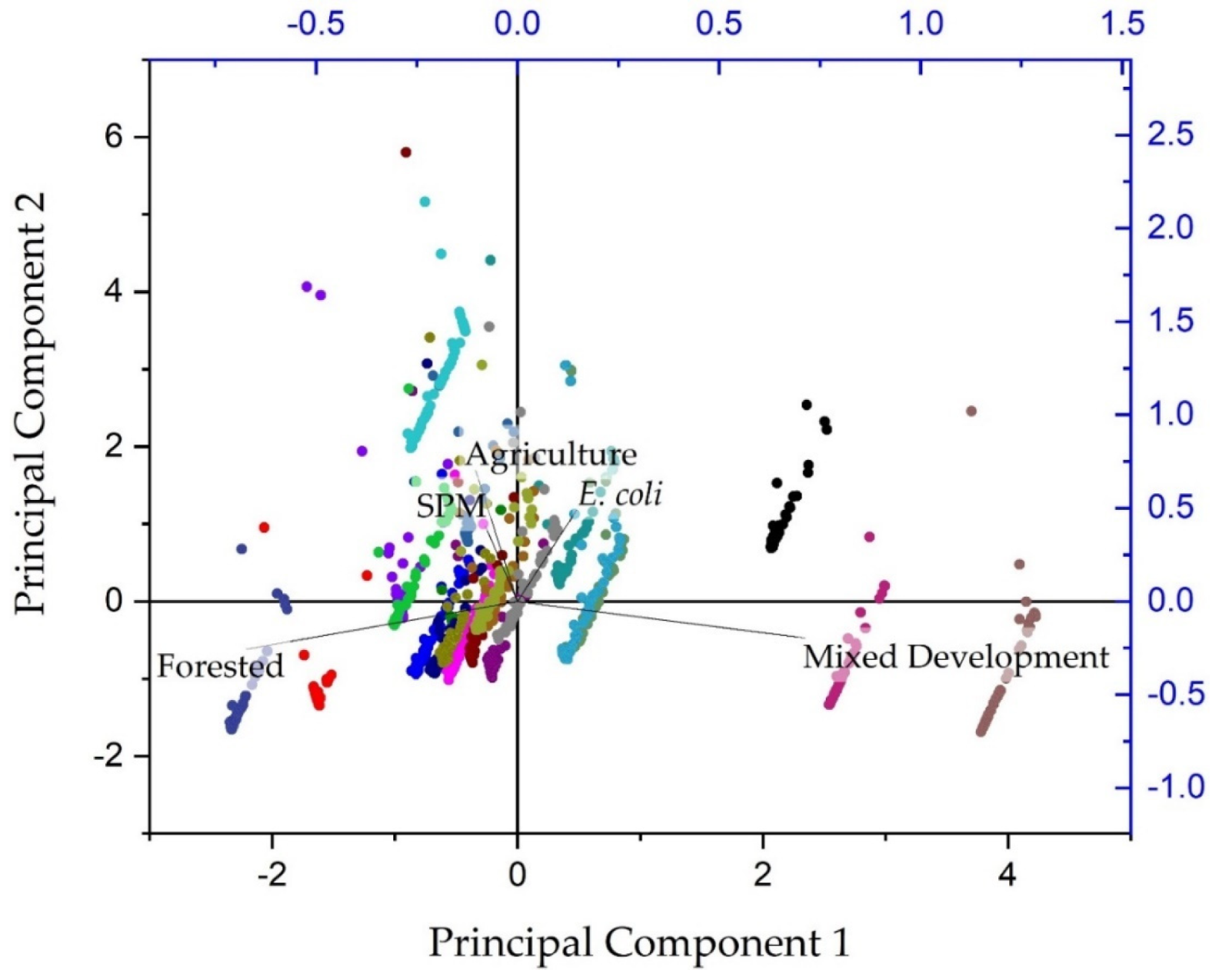
2.3. Data Analysis
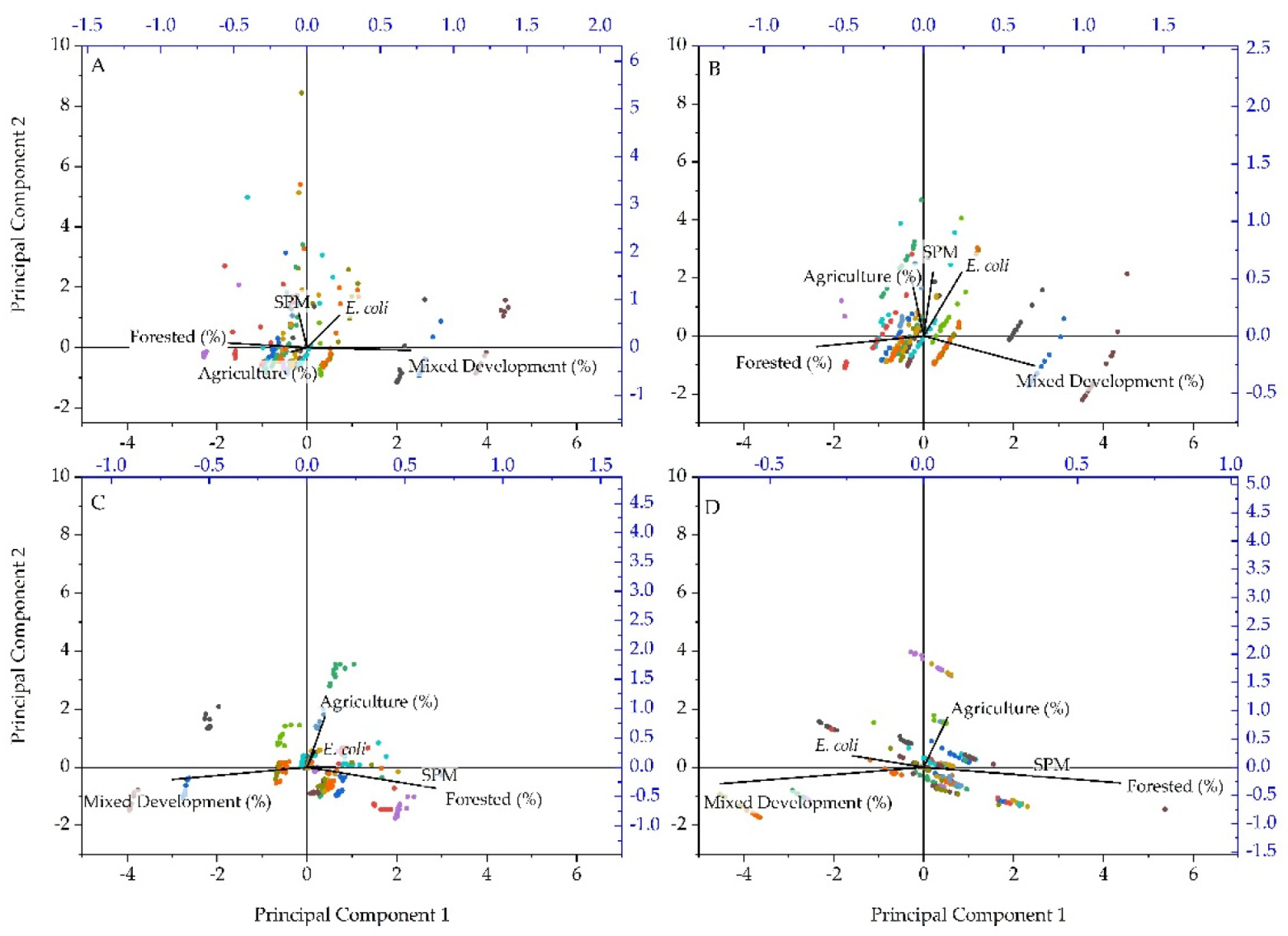
3. Results and Discussion
3.1. Climate during Study
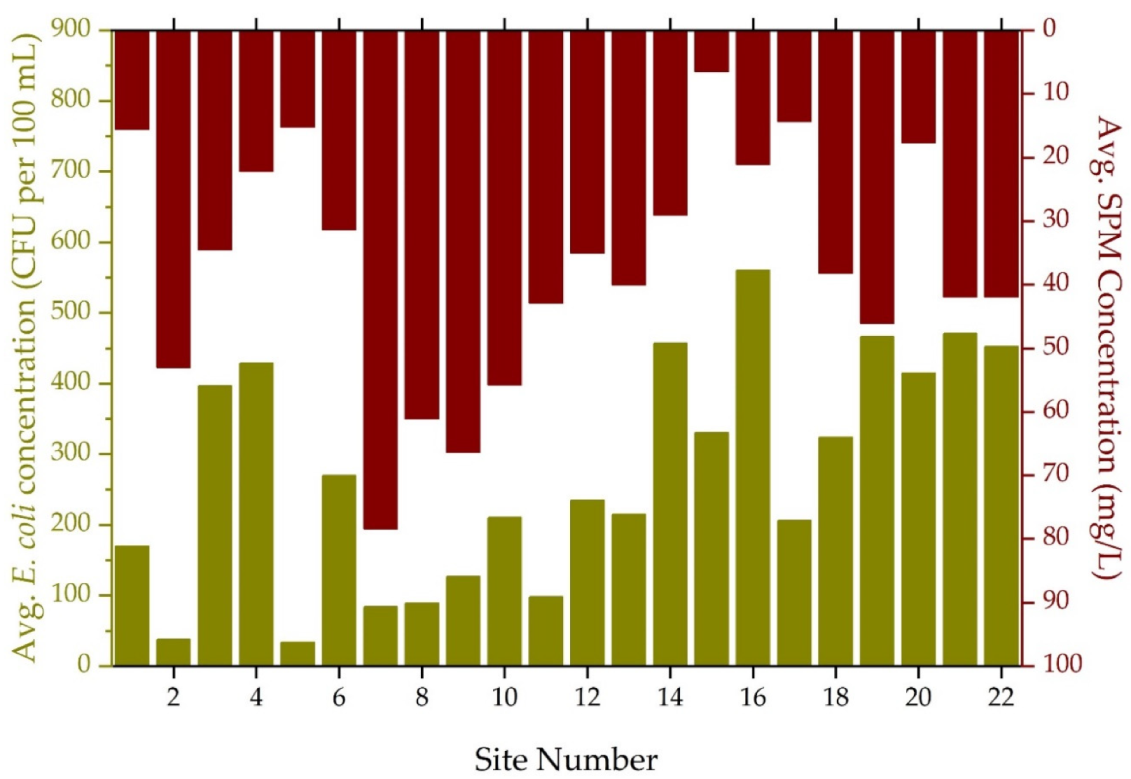
3.2. Annual Suspended Particulate Matter, E. coli Concentrations and Land Use Practices
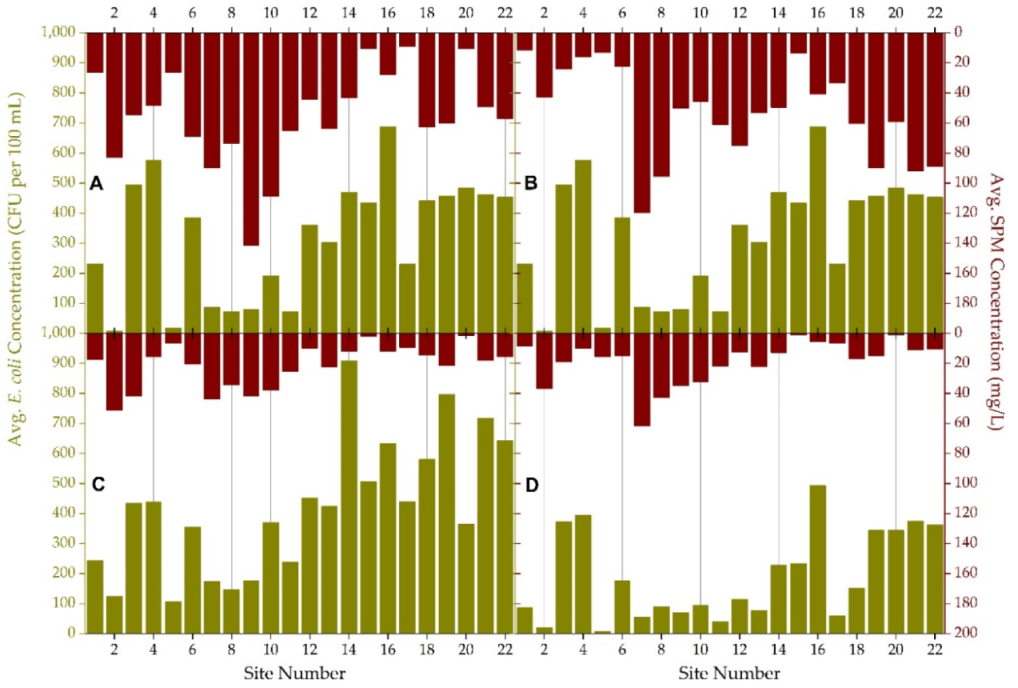
3.3. Quarterly Suspended Particulate Matter, E. coli Concentrations and Land Use Practices
3.4. Non-Parametric Statistical Results
3.5. Study Implications and Future Work
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. World Water Day Report. Available online: https://www.who.int/water_sanitation_health/takingcharge.html (accessed on 12 December 2019).
- CDC. E. coli (Escherichia coli). Available online: https://www.cdc.gov/ecoli/index.html (accessed on 8 January 2020).
- Madoux-Humery, A.-S.; Dorner, S.; Sauvé, S.; Aboulfadl, K.; Galarneau, M.; Servais, P.; Prévost, M. The effects of combined sewer overflow events on riverine sources of drinking water. Water Res. 2016, 92, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, R.; Joy, D.M.; Lee, H.; Kostaschuk, R.; Gordon, R. Transport and deposition of sediment-Associated Escherichia coli in natural streams. Water Res. 2005, 39, 2665–2675. [Google Scholar] [CrossRef] [PubMed]
- Amalfitano, S.; Corno, G.; Eckert, E.; Fazi, S.; Ninio, S.; Callieri, C.; Grossart, H.-P.; Eckert, W. Tracing particulate matter and associated microorganisms in freshwaters. Hydrobiologia 2017, 800, 145–154. [Google Scholar] [CrossRef]
- Bernard, J.M.; Steffen, L.L.; Iivari, T.A. Has the US sediment pollution problem been solved? In Proceedings of the Sixth Federal Interagency Sedimentation Conference, Sahara Hotel, Las Vegas, NV, USA, 10–14 March 1996; United States Geologic Survey. 1996; pp. 7–13. Available online: https://water.usgs.gov/osw/ressed/references/Bernard-Iivari-6Fisc2-8.pdf (accessed on 24 April 2020).
- Ongley, E.D. Control of Water Pollution from Agriculture; Food and Agriculture Organization of the United Nations: Rome, Italy, 1996; ISBN 92-5-103875-9. [Google Scholar]
- Hubbart, J.A.; Kellner, E.; Freeman, G. A case study considering the comparability of mass and volumetric suspended sediment data. Environ. Earth Sci. 2014, 71, 4051–4060. [Google Scholar] [CrossRef]
- Uri, N.D. The environmental implications of soil erosion in the United States. Environ. Monit. Assess. 2001, 66, 293–312. [Google Scholar] [CrossRef] [PubMed]
- Foster, I.D.L.; Charlesworth, S.M. Heavy metals in the hydrological cycle: Trends and explanation. Hydrol. Process. 1996, 10, 227–261. [Google Scholar] [CrossRef]
- Oschwald, W.R. Sediment-water interactions 1. J. Environ. Qual. 1972, 1, 360–366. [Google Scholar] [CrossRef]
- Russell, M.A.; Walling, D.E.; Webb, B.W.; Bearne, R. The composition of nutrient fluxes from contrasting UK river basins. Hydrol. Process. 1998, 12, 1461–1482. [Google Scholar] [CrossRef]
- Walling, D. The changing sediment loads of the world’s rivers. Ann. Wars. Univ. Life Sci. SGGW Land Reclam. 2008, 39, 3–20. [Google Scholar] [CrossRef]
- Bilotta, G.S.; Brazier, R.E. Understanding the influence of suspended solids on water quality and aquatic biota. Water Res. 2008, 42, 2849–2861. [Google Scholar] [CrossRef]
- Irvine, K.N.; Pettibone, G.W.; Droppo, I.G. Indicator Bacteria-Sediment Relationships: Implications for Water Quality Modeling and Monitoring. J. Water Manag. Model. 1995, 3, 205–230. [Google Scholar] [CrossRef] [Green Version]
- Abia, A.L.K.; Ubomba-Jaswa, E.; Genthe, B.; Momba, M.N.B. Quantitative microbial risk assessment (QMRA) shows increased public health risk associated with exposure to river water under conditions of riverbed sediment resuspension. Sci. Total Environ. 2016, 566–567, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.H.; Pachepsky, Y.A.; Oliver, D.M.; Muirhead, R.W.; Park, Y.; Quilliam, R.S.; Shelton, D.R. Modeling fate and transport of fecally-Derived microorganisms at the watershed scale: State of the science and future opportunities. Water Res. 2016, 100, 38–56. [Google Scholar] [CrossRef] [PubMed]
- Southard, J. Introduction to Fluid Motions, Sediment Transport, and Current-Generated Sedimentary Structures Course Textbook; Massachusetts Institute of Technology, MIT OpenCourseWare: Cambridge, MA, USA, 2006; pp. 1–536. [Google Scholar]
- Jeloudar, F.T.; Sepanlou, M.G.; Emadi, S.M. Impact of land use change on soil erodibility. Glob. J. Environ. Sci. Manag. 2018, 4, 59–70. [Google Scholar]
- Petersen, F.; Hubbart, J.A.; Kellner, E.; Kutta, E. Land-Use-Mediated Escherichia coli concentrations in a contemporary Appalachian watershed. Environ. Earth Sci. 2018, 77, 754. [Google Scholar] [CrossRef]
- AL-Kaisi, M. Frequent Tillage and its Impact on Soil Quality | Integrated Crop Management. 2020. Available online: https://crops.extension.iastate.edu/encyclopedia/frequent-tillage-and-its-impact-soil-quality (accessed on 14 February 2020).
- Petersen, F.; Hubbart, J.A. Quantifying Escherichia coli and suspended particulate matter concentrations in a mixed-Land use Appalachian watershed. Water 2020, 12, 532. [Google Scholar] [CrossRef] [Green Version]
- Rwego, I.B.; Gillspie, T.R.; Isabirye-Basuta, G.; Goldberg, T.L. High rates of Escherichia coli transmission between livestock and humans in rural Uganda. J. Clin. Microbiol. 2008, 46, 3187–3191. [Google Scholar] [CrossRef] [Green Version]
- Causse, J.; Billen, G.; Garnier, J.; Henri-des-Tureaux, T.; Olasa, X.; Thammahacksa, C.; Latsachak, K.O.; Soulileuth, B.; Sengtaheuanghoung, O.; Rochelle-Newall, E. Field and modelling studies of Escherichia coli loads in tropical streams of montane agro-Ecosystems. J. Hydro Environ. Res. 2015, 9, 496–507. [Google Scholar] [CrossRef]
- Jeng, H.C.; England, A.J.; Bradford, H.B. Indicator organisms associated with stormwater suspended particles and estuarine sediment. J. Environ. Sci. Health 2005, 40, 779–791. [Google Scholar] [CrossRef]
- Characklis, G.W.; Dilts, M.J.; Simmons, O.D., III; Likirdopulos, C.A.; Krometis, L.-A.H.; Sobsey, M.D. Microbial partitioning to settleable particles in stormwater. Water Res. 2005, 39, 1773–1782. [Google Scholar] [CrossRef]
- Oliver, D.M.; Clegg, C.D.; Heathwaite, A.L.; Haygarth, P.M. Preferential attachment of Escherichia coli to different particle size fractions of an agricultural grassland soil. Water Air Soil Pollut. 2007, 185, 369–375. [Google Scholar] [CrossRef] [Green Version]
- Kändler, M.; Blechinger, K.; Seidler, C.; Pavlů, V.; Šanda, M.; Dostál, T.; Krása, J.; Vitvar, T.; Štich, M. Impact of land use on water quality in the upper Nisa catchment in the Czech Republic and in Germany. Sci. Total Environ. 2017, 586, 1316–1325. [Google Scholar] [CrossRef] [PubMed]
- Thurston-Enriquez, J.A.; Gilley, J.E.; Eghball, B. Microbial quality of runoff following land application of cattle manure and swine slurry. J. Water Health 2005, 3, 157–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paster, E.; Ryu, W.S. The thermal impulse response of Escherichia coli. Proc. Natl. Acad. Sci. USA 2008, 105, 5373–5377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stein, E.D.; Tiefenthaler, L.; Schiff, K. Comparison of storm water pollutant loading by land use type. In Southern California Coastal Water Research Project 2008 Annual Report; 3535 Harbor Blvd STE 110: Costa Mesa, CA, USA, 2008; pp. 15–27. [Google Scholar]
- Rochelle-Newall, E.J.; Ribolzi, O.; Viguier, M.; Thammahacksa, C.; Silvera, N.; Latsachack, K.; Dinh, R.P.; Naporn, P.; Sy, H.T.; Soulileuth, B. Effect of land use and hydrological processes on Escherichia coli concentrations in streams of tropical, humid headwater catchments. Sci. Rep. 2016, 6, 32974. [Google Scholar] [CrossRef] [Green Version]
- Widgren, S.; Engblom, S.; Emanuelson, U.; Lindberg, A. Spatio-Temporal modelling of verotoxigenic Escherichia coli O157 in cattle in Sweden: Exploring options for control. Vet. Res. 2018, 49, 78. [Google Scholar] [CrossRef] [Green Version]
- Tetzlaff, D.; Carey, S.K.; McNamara, J.P.; Laudon, H.; Soulsby, C. The essential value of long-term experimental data for hydrology and water management. Water Resour. Res. 2017, 53, 2598–2604. [Google Scholar] [CrossRef] [Green Version]
- Zeiger, S.; Hubbart, J.A. Quantifying suspended sediment flux in a mixed-land-use urbanizing watershed using a nested-scale study design. Sci. Total Environ. 2016, 542, 315–323. [Google Scholar] [CrossRef]
- Leopold, L.B. Hydrologic research on instrumented watersheds. In International Symposium on the Results of Research on Representative and Experimental Basins; International Association of Scientific Hydrology: Wallingford, UK, 1970; pp. 135–150. [Google Scholar]
- Hewlett, J.D.; Lull, H.W.; Reinhart, K.G. In Defense of experimental watersheds. Water Resour. Res. 1969, 5, 306–316. [Google Scholar] [CrossRef]
- Bosch, J.M.; Hewlett, J.D. A review of catchment experiments to determine the effect of vegetation changes on water yield and evapotranspiration. J. Hydrol. 1982, 55, 3–23. [Google Scholar] [CrossRef]
- Nichols, J.; Hubbart, J.A. Using macroinvertebrate assemblages and multiple stressors to infer urban stream system condition: A case study in the central US. Urban Ecosyst. 2016, 19, 679–704. [Google Scholar] [CrossRef]
- Zeiger, S.; Hubbart, J.A.; Anderson, S.A.; Stambaugh, M.C. Quantifying and modelling urban stream temperature: A central US watershed study. Hydrol. Process. 2015, 30, 503–514. [Google Scholar] [CrossRef]
- Hubbart, J.A.; Kellner, E.; Zeiger, S. A case-study application of the experimental watershed study design to advance adaptive management of contemporary watersheds. Water 2019, 11, 2355. [Google Scholar] [CrossRef] [Green Version]
- Zeiger, S.J.; Hubbart, J.A. Nested-scale nutrient flux in a mixed-land-use urbanizing watershed. Hydrol. Process. 2016, 30, 1475–1490. [Google Scholar] [CrossRef]
- Kellner, E.; Hubbart, J. Advancing understanding of the surface water quality regime of contemporary mixed-land-use watersheds: An application of the experimental watershed method. Hydrology 2017, 4, 31. [Google Scholar] [CrossRef] [Green Version]
- Hubbart, J.A. Urban floodplain management: Understanding consumptive water-use potential in urban forested floodplains. Stormwater J. 2011, 12, 56–63. [Google Scholar]
- Cantor, J.; Krometis, L.-A.; Sarver, E.; Cook, N.; Badgley, B. Tracking the downstream impacts of inadequate sanitation in central Appalachia. J. Water Health 2017, 15, 580–590. [Google Scholar] [CrossRef] [Green Version]
- Dykeman, W. Appalachian Mountains. Available online: https://www.britannica.com/place/Appalachian-Mountains (accessed on 20 November 2019).
- Central Appalachian Broadleaf Forest—Coniferous Forest—Meadow Province. Available online: https://www.fs.fed.us/land/ecosysmgmt/colorimagemap/images/m221.html (accessed on 12 December 2019).
- Köppen, W. Das geographische System der Klimate. In Handb. Der Klimato; Gebruder Borntraeg: Berlin, Germany, 1936. [Google Scholar]
- Arcipowski, E.; Schwartz, J.; Davenport, L.; Hayes, M.; Nolan, T. Clean water, clean life: Promoting healthier, accessible water in rural Appalachia. J. Contemp. Water Res. Educ. 2017, 161, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. Discuss. 2007, 4, 439–473. [Google Scholar] [CrossRef] [Green Version]
- Arguez, A.; Durre, I.; Applequist, S.; Squires, M.; Vose, R.; Yin, X.; Bilotta, R. NOAA’s US climate normals (1981–2010). NOAA Natl. Cent. Environ. Inf. 2010, 10, V5PN93JP. [Google Scholar]
- Kellner, E.; Hubbart, J.; Stephan, K.; Morrissey, E.; Freedman, Z.; Kutta, E.; Kelly, C. Characterization of sub-watershed-scale stream chemistry regimes in an Appalachian mixed-land-use watershed. Environ. Monit. Assess. 2018, 190, 586. [Google Scholar] [CrossRef] [PubMed]
- The West Virginia Water Research Institute (WVWRI); The West Run Watershed Association. Watershed Based Plan for West Run of the Monongahela River; The West Virginia Water Research Institute, The West RunWatershed Association: Morgantown, WV, USA, 2008; pp. 1–59. [Google Scholar]
- Hubbart, J.A.; Muzika, R.-M.; Huang, D.; Robinson, A. Improving quantitative understanding of bottomland hardwood forest influence on soil water consumption in an urban floodplain. Watershed Sci. Bull. 2011, 3, 34–43. [Google Scholar]
- Hubbart, J.A. Measuring and Modeling Hydrologic Responses to Timber Harvest in a Continental/Maritime Mountainous Environment. Ph.D. Thesis, University of Idaho, Moscow, ID, USA, 2007. [Google Scholar]
- Wei, L.; Hubbart, J.A.; Zhou, H. Variable streamflow contributions in nested subwatersheds of a US Midwestern urban watershed. Water Resour. Manag. 2018, 32, 213–228. [Google Scholar] [CrossRef]
- Hubbart, J.A.; Kellner, E.; Hooper, L.W.; Zeiger, S. Quantifying loading, toxic concentrations, and systemic persistence of chloride in a contemporary mixed-land-use watershed using an experimental watershed approach. Sci. Total Environ. 2017, 581, 822–832. [Google Scholar] [CrossRef] [PubMed]
- Zeiger, S.J.; Hubbart, J.A. Quantifying flow interval–pollutant loading relationships in a rapidly urbanizing mixed-land-use watershed of the Central USA. Environ. Earth Sci. 2017, 76, 484. [Google Scholar] [CrossRef]
- Dusek, N.; Hewitt, A.J.; Schmidt, K.N.; Bergholz, P.W. Landscape-scale factors affecting the prevalence of Escherichia coli in surface soil include land cover type, edge interactions, and soil pH. Appl. Environ. Microbiol. 2018, 84, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Desai, M.A.; Rifai, H.S. Variability of Escherichia coli concentrations in an urban watershed in Texas. J. Environ. Eng. 2010, 136, 1347–1359. [Google Scholar] [CrossRef]
- American Society for Testing and Materials. Standard Methods for Determining Sediment Concentrations in Water Samples; American Society for Testing and Materials: West Conshohocken, PA, USA, 2007; pp. 1–6. [Google Scholar]
- Price, R.G.; Wildeboer, D. E. coli as an indicator of contamination and health risk in environmental waters. In Escherichia coli—Recent Advances on Physiology, Pathogenesis and Biotechnological Applications; Intech: London, UK, 2017. [Google Scholar] [CrossRef] [Green Version]
- IDEXX. Laboratories Colilert Procedure Manual. Available online: https://www.idexx.com/files/colilert-procedure-en.pdf (accessed on 4 April 2019).
- Cummings, D.; IDEXX. The Fecal Coliform Test Compared to Specific Tests for Escherichia Coli. Available online: https://www.idexx.com/resource-library/water/water-reg-article9B.pdf (accessed on 24 September 2019).
- Yazici, B.; Yolacan, S. A comparison of various tests of normality. J. Stat. Comput. Simul. 2007, 77, 175–183. [Google Scholar] [CrossRef]
- Fisher, R.A. Statistical Methods for Research Workers. In Breakthroughs in Statistics: Methodology and Distribution; Kotz, S., Johnson, N.L., Eds.; Springer Series in Statistics; Springer: New York, NY, USA, 1992; pp. 66–70. ISBN 978-1-4612-4380-9. [Google Scholar]
- United States Climate Data (USCD). Available online: https://www.usclimatedata.com/climate/morgantown/west-virginia/united-states/uswv0507/2012/7 (accessed on 28 September 2019).
- Chapman, D. Rivers. In Water Quality Assessments—A Guide to Use of Biota, Sediments and Water in Environmental Monitoring; UNESCO/WHO/UNEP: Paris, France, 1992; ISBN 0 419 21590 5. [Google Scholar]
- Lazar, J.A. Urban and Agricultural Land Cover Impacts on Storm Flow and Nutrient Concentrations in SW Ohio Streams. Master’s Thesis, Miami University, Oxford, OH, USA, 2018. [Google Scholar]
- Tong, S.T.; Chen, W. Modeling the relationship between land use and surface water quality. J. Environ. Manag. 2002, 66, 377–393. [Google Scholar] [CrossRef]
- RoyChowdhury, A.; Sarkar, D.; Datta, R. Remediation of acid mine drainage-impacted water. Curr. Pollut. Rep. 2015, 1, 131–141. [Google Scholar] [CrossRef]
- Brown, T.C.; Binkley, D.; Brown, D. Water Quality on Forest Lands | Rocky Mountain Research Station. Available online: /rmrs/projects/water-quality-forest-lands (accessed on 11 March 2020).
- Gotkowska-Plachta, A.; Golaś, I.; Korzeniewska, E.; Koc, J.; Rochwerger, A.; Solarski, K. Evaluation of the distribution of fecal indicator bacteria in a river system depending on different types of land use in the southern watershed of the Baltic Sea. Environ. Sci. Pollut. Res. 2016, 23, 4073–4085. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yunus, M.; Islam, M.S.; Emch, M. Influence of climate extremes and land use on fecal contamination of shallow tubewells in Bangladesh. Environ. Sci. Technol. 2016, 50, 2669–2676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Booth, D.B.; Roy, A.H.; Smith, B.; Capps, K.A. Global perspectives on the urban stream syndrome. Freshw. Sci. 2016, 35, 412–420. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.; Wang, G. Modeling seasonal vegetation variation and its validation against Moderate Resolution Imaging Spectroradiometer (MODIS) observations over North America. J. Geophys. Res. Atmos. 2005, 110, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Loch, R.J. Effects of vegetation cover on runoff and erosion under simulated rain and overland flow on a rehabilitated site on the Meandu Mine, Tarong, Queensland. Soil Res. 2000, 38, 299–312. [Google Scholar] [CrossRef]
- Chen, H.J.; Chang, H. Response of discharge, TSS, and E. coli to rainfall events in urban, suburban, and rural watersheds. Environ. Sci. Process. Impacts 2014, 16, 2313–2324. [Google Scholar] [CrossRef]
- Farewell, A.; Neidhardt, F.C. Effect of temperature on in vivo protein synthetic capacity in Escherichia coli. J. Bacteriol. 1998, 180, 4704–4710. [Google Scholar] [CrossRef] [Green Version]
- Mcmillan, S.K.; Wilson, H.F.; Tague, C.L.; Hanes, D.M.; Inamdar, S.; Karwan, D.L.; Loecke, T.; Morrison, J.; Murphy, S.F.; Vidon, P. Before the storm: Antecedent conditions as regulators of hydrologic and biogeochemical response to extreme climate events. Biogeochemistry 2018, 141, 487–501. [Google Scholar] [CrossRef]
- Multivariate Analysis: Principal Component Analysis: Biplots—9.3. Available online: http://support.sas.com/documentation/cdl/en/imlsug/64254/HTML/default/viewer.htm#imlsug_ugmultpca_sect003.htm (accessed on 23 December 2019).
- Jolliffe, I.T.; Cadima, J. Principal component analysis: A review and recent developments. Philos. Trans. A Math. Phys. Eng. Sci. 2016, 374, 1–18. [Google Scholar] [CrossRef]
- Bro, R.; Smilde, A.K. Principal component analysis. Anal. Methods 2014, 6, 2812–2831. [Google Scholar] [CrossRef] [Green Version]
- Verhougstraete, M.P.; Martin, S.L.; Kendall, A.D.; Hyndman, D.W.; Rose, J.B. Linking fecal bacteria in rivers to landscape, geochemical, and hydrologic factors and sources at the basin scale. Proc. Natl. Acad. Sci. USA 2015, 112, 10419–10424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong, H.; Qiu, J.; Liang, Y. Environmental factors influencing the distribution of total and fecal coliform bacteria in six water storage reservoirs in the Pearl River Delta Region, China. J. Environ. Sci. 2010, 22, 663–668. [Google Scholar] [CrossRef]
- Islam, M.M.M.; Hofstra, N.; Islam, M.A. The impact of environmental variables on faecal indicator bacteria in the Betna River Basin, Bangladesh. Environ. Process. 2017, 4, 319–332. [Google Scholar] [CrossRef]
- Biron, P.M.; Roy, A.G.; Courschesne, F.; Hendershot, W.H.; Côté, B.; Fyles, J. The effects of antecedent moisture conditions on the relationship of hydrology to hydrochemistry in a small forested watershed. Hydrol. Process. 1999, 13, 1541–1555. [Google Scholar] [CrossRef]
- Shi, P.; Zhang, Y.; Li, Z.; Li, P.; Xu, G. Influence of land use and land cover patterns on seasonal water quality at multi-spatial scales. Catena 2017, 151, 182–190. [Google Scholar] [CrossRef]
- Ali, S.; Ghosh, N.C.; SIngh, R. Rainfall–Runoff simulation using a normalized antecedent precipitation index. Hydrol. Sci. J. 2010, 55, 266–274. [Google Scholar] [CrossRef] [Green Version]
- Song, S.; Wang, W. Impacts of antecedent soil moisture on the rainfall-runoff transformation process based on high-resolution observations in soil. Water 2019, 11, 296. [Google Scholar] [CrossRef] [Green Version]
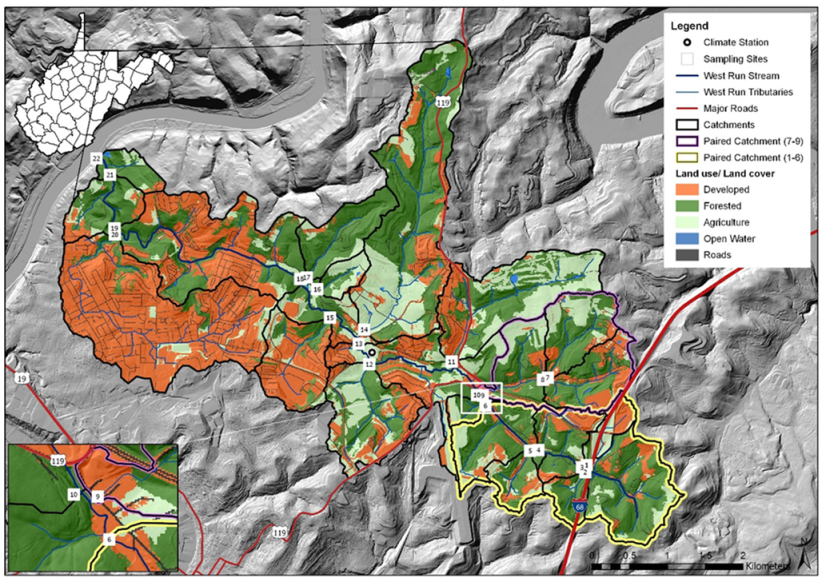
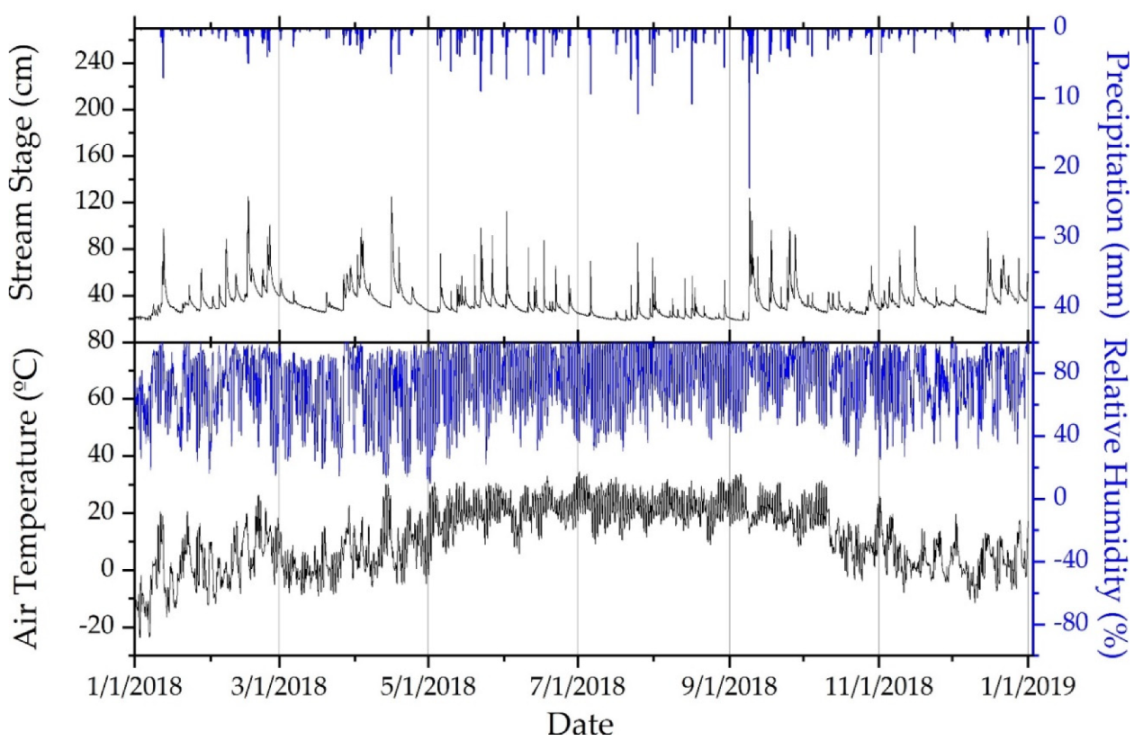
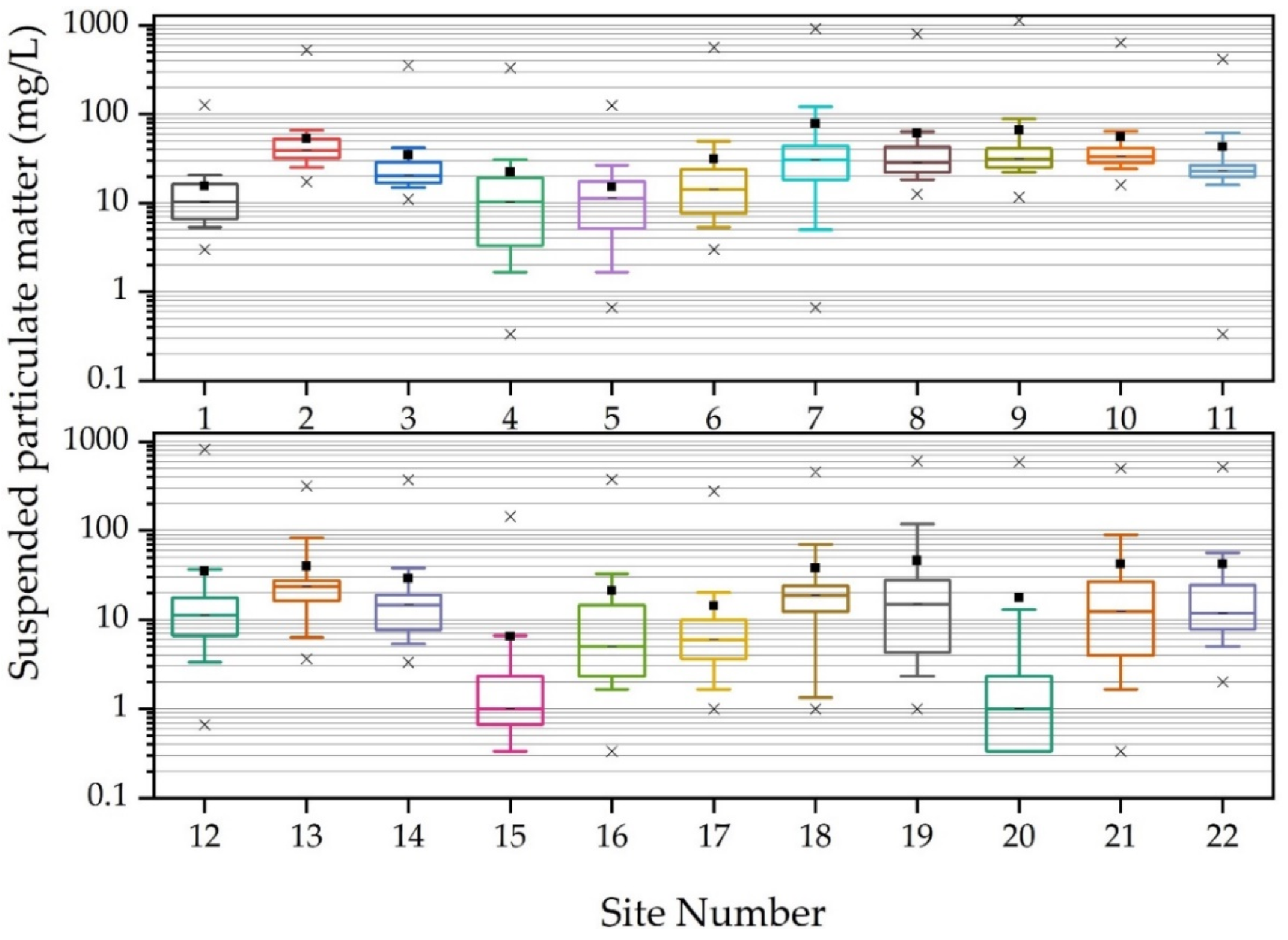
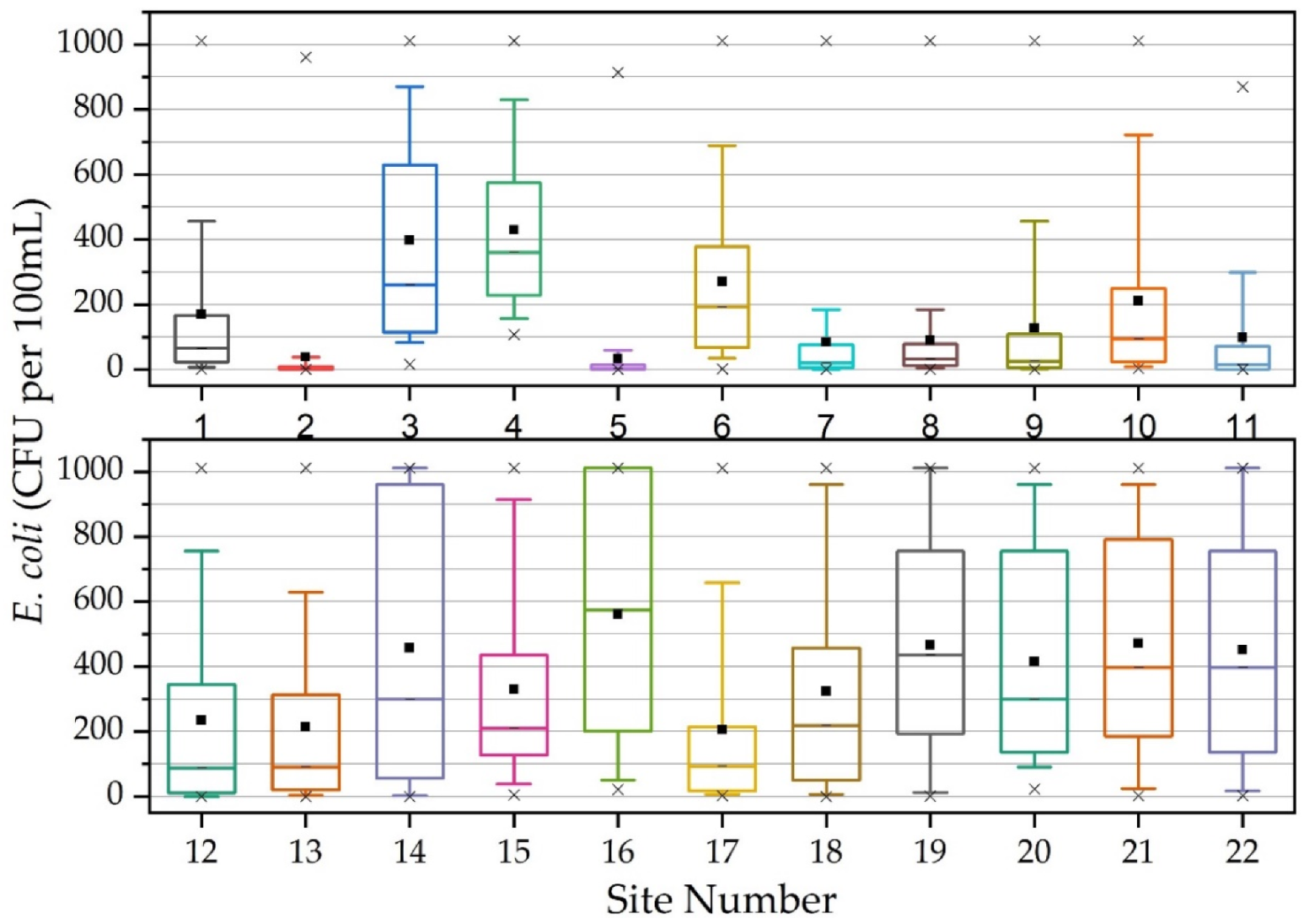
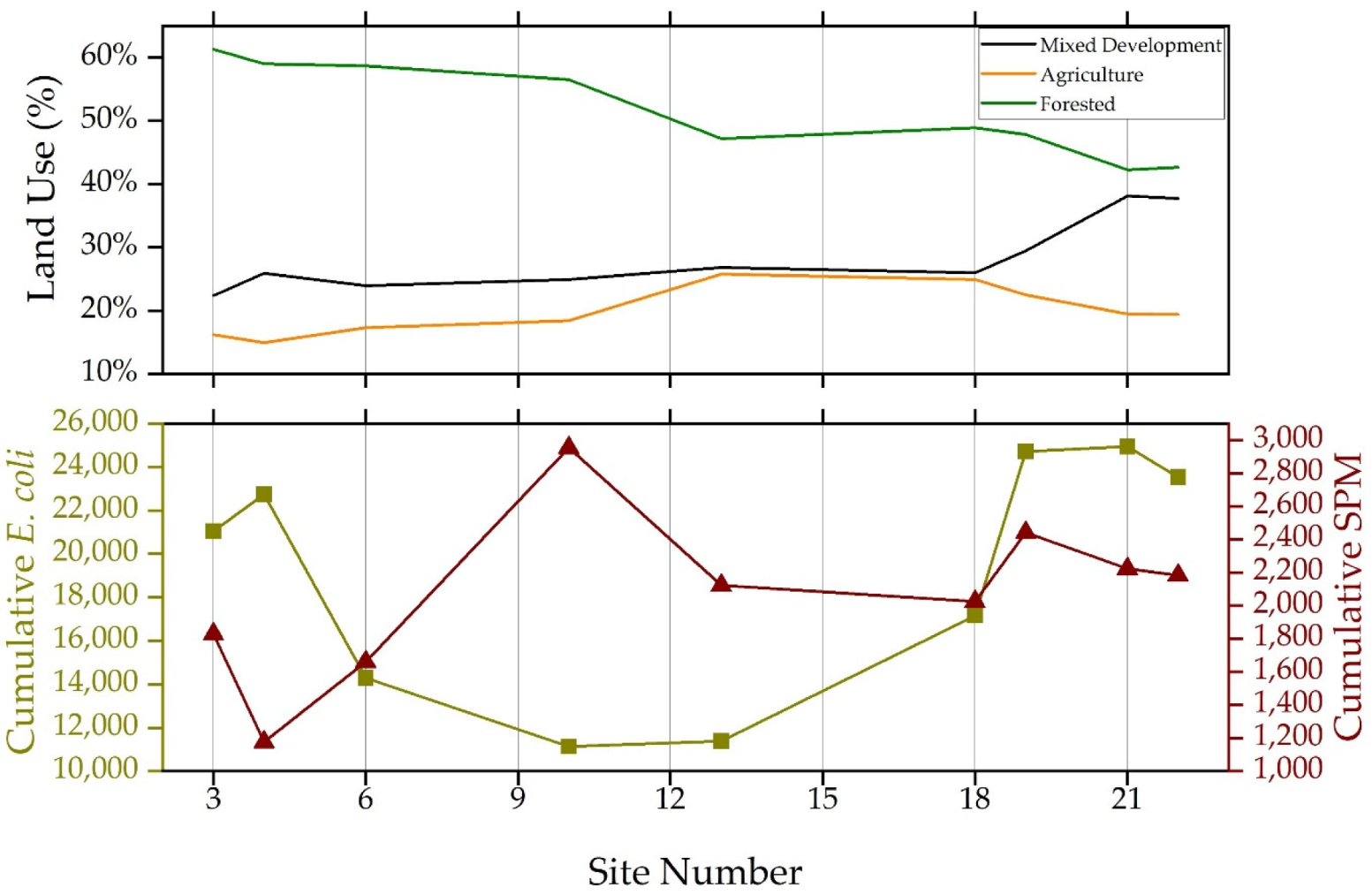
| Site | Mixed Development (%) | Agriculture (%) | Forested (%) | Drainage Area (km²) |
|---|---|---|---|---|
| 1 | 53.23% | 38.70% | 8.07% | 0.30 |
| 2 | 13.58% | 12.20% | 74.21% | 0.29 |
| 3 | 22.35% | 16.17% | 61.32% | 1.87 |
| 4 | 25.88% | 14.91% | 59.00% | 2.48 |
| 5 | 23.35% | 25.51% | 51.14% | 0.38 |
| 6 | 23.91% | 17.25% | 58.70% | 3.72 |
| 7 | 16.33% | 28.60% | 54.91% | 0.78 |
| 8 | 30.78% | 16.47% | 52.35% | 1.55 |
| 9 | 27.57% | 19.33% | 52.84% | 2.29 |
| 10 | 24.92% | 18.40% | 56.49% | 6.18 |
| 11 | 18.15% | 41.87% | 39.16% | 1.75 |
| 12 | 31.77% | 33.72% | 34.51% | 1.75 |
| 13 | 26.83% | 25.77% | 47.15% | 10.53 |
| 14 | 16.19% | 26.43% | 56.92% | 3.36 |
| 15 | 70.28% | 10.31% | 19.42% | 0.98 |
| 16 | 5.38% | 58.72% | 35.16% | 0.25 |
| 17 | 4.78% | 9.38% | 85.84% | 0.75 |
| 18 | 25.98% | 24.88% | 48.86% | 16.41 |
| 19 | 29.45% | 22.45% | 47.85% | 18.88 |
| 20 | 89.16% | 4.19% | 6.61% | 3.42 |
| 21 | 38.10% | 19.46% | 42.23% | 22.93 |
| 22 | 37.71% | 19.38% | 42.66% | 23.24 |
| Site Number | |||||||||||
| #1 | #2 | #3 | #4 | #5 | #6 | #7 | #8 | #9 | #10 | #11 | |
| Avg. | 15.6 | 53.0 | 34.5 | 22.2 | 15.2 | 31.3 | 78.4 | 61.1 | 66.4 | 55.7 | 42.9 |
| Med. | 10.3 | 39.3 | 20.3 | 10.3 | 11.3 | 14.3 | 30.7 | 28.7 | 31.7 | 33.3 | 23.0 |
| Min. | 3.0 | 17.3 | 11.0 | 0.3 | 0.7 | 3.0 | 0.7 | 12.7 | 11.7 | 16.0 | 0.3 |
| Max. | 126.7 | 528.7 | 357.0 | 332.3 | 125.3 | 569.7 | 928.3 | 803.3 | 1140.0 | 642.0 | 417.3 |
| Std. Dev. | 20.8 | 69.8 | 57.5 | 49.5 | 19.0 | 79.9 | 176.5 | 123.5 | 159.7 | 93.3 | 77.2 |
| Site Number | |||||||||||
| #12 | #13 | #14 | #15 | #16 | #17 | #18 | #19 | #20 | #21 | #22 | |
| Avg. | 35.0 | 40.1 | 29.1 | 6.5 | 21.1 | 14.4 | 38.2 | 46.1 | 17.7 | 41.9 | 42.0 |
| Med. | 11.3 | 23.3 | 14.7 | 1.0 | 5.0 | 6.0 | 18.7 | 15.0 | 1.0 | 12.3 | 11.8 |
| Min. | 0.7 | 3.7 | 3.3 | 0.0 | 0.3 | 1.0 | 1.0 | 1.0 | 0.0 | 0.3 | 2.0 |
| Max. | 819.3 | 316.0 | 370.7 | 144.7 | 376.0 | 277.3 | 456.7 | 603.3 | 590.0 | 502.0 | 518.0 |
| Std. Dev. | 116.4 | 62.0 | 59.7 | 21.5 | 59.3 | 38.6 | 75.3 | 103.7 | 83.3 | 101.1 | 99.1 |
| Site Number | |||||||||||
| #1 | #2 | #3 | #4 | #5 | #6 | #7 | #8 | #9 | #10 | #11 | |
| Avg. | 170 | 38 | 397 | 429 | 34 | 269 | 84 | 89 | 127 | 210 | 98 |
| Med. | 66 | 3 | 260 | 361 | 4 | 194 | 20 | 32 | 25 | 93 | 16 |
| Min. | 0 | 0 | 15 | 107 | 0 | 2 | 0 | 0 | 0 | 3 | 0 |
| Max. | 1011 | 961 | 1011 | 1011 | 914 | 1011 | 1011 | 1011 | 1011 | 1011 | 870 |
| Std. Dev. | 251 | 139 | 315 | 249 | 129 | 276 | 179 | 180 | 241 | 273 | 202 |
| Site Number | |||||||||||
| #12 | #13 | #14 | #15 | #16 | #17 | #18 | #19 | #20 | #21 | #22 | |
| Avg. | 234 | 215 | 457 | 330 | 560 | 206 | 324 | 466 | 415 | 471 | 452 |
| Med. | 88 | 91 | 299 | 211 | 575 | 93 | 218 | 436 | 299 | 397 | 397 |
| Min. | 0 | 0 | 0 | 5 | 22 | 3 | 0 | 1 | 23 | 2 | 3 |
| Max. | 1011 | 1011 | 1011 | 1011 | 1011 | 1011 | 1011 | 1011 | 1011 | 1011 | 1011 |
| Std. Dev. | 305 | 266 | 406 | 293 | 373 | 288 | 342 | 339 | 340 | 342 | 345 |
| Site Number | |||||||||||
| #1 | #2 | #3 | #4 | #5 | #6 | #7 | #8 | #9 | #10 | #11 | |
| SCC | 0.19 | 0.18 | 0.04 | 0.30 | −0.11 | 0.04 | 0.46 | 0.52 | 0.73 | 0.24 | 0.56 |
| p-value | 0.17 | 0.19 | 0.77 | 0.03 | 0.42 | 0.78 | <0.01 | <0.01 | <0.01 | 0.09 | <0.01 |
| Site Number | |||||||||||
| #12 | #13 | #14 | #15 | #16 | #17 | #18 | #19 | #20 | #21 | #22 | |
| SCC | 0.00 | 0.13 | −0.25 | 0.42 | 0.64 | 0.27 | 0.02 | −0.14 | 0.55 | 0.01 | 0.17 |
| p-value | 0.99 | 0.34 | 0.07 | <0.01 | <0.01 | 0.05 | 0.88 | 0.33 | <0.01 | 0.94 | 0.24 |
| Site Number | ||||||||||||
| #1 | #2 | #3 | #4 | #5 | #6 | #7 | #8 | #9 | #10 | #11 | ||
| Quarter 1 | SCC | 0.40 | 0.60 | 0.26 | 0.48 | 0.52 | 0.35 | 0.76 | 0.70 | 0.73 | 0.63 | 0.69 |
| p-value | 0.18 | 0.03 | 0.39 | 0.09 | 0.09 | 0.23 | <0.01 | 0.01 | <0.01 | 0.02 | 0.01 | |
| Quarter 2 | SCC | 0.19 | 0.18 | 0.03 | 0.26 | −0.04 | 0.07 | 0.78 | 0.88 | 0.78 | 0.51 | 0.93 |
| p-value | 0.53 | 0.56 | 0.91 | 0.38 | 0.89 | 0.81 | <0.01 | <0.01 | <0.01 | 0.08 | <0.01 | |
| Quarter 3 | SCC | −0.05 | −0.40 | 0.32 | 0.50 | 0.76 | 0.69 | −0.27 | 0.58 | 0.79 | 0.25 | 0.54 |
| p-value | 0.88 | 0.17 | 0.29 | 0.08 | <0.01 | 0.01 | 0.37 | 0.04 | <0.01 | 0.42 | 0.06 | |
| Quarter 4 | SCC | −0.30 | −0.32 | −0.39 | −0.04 | −0.02 | −0.01 | −0.08 | −0.09 | 0.62 | −0.22 | 0.51 |
| p-value | 0.31 | 0.27 | 0.17 | 0.89 | 0.95 | 0.98 | 0.78 | 0.75 | 0.02 | 0.45 | 0.06 | |
| Site Number | ||||||||||||
| #12 | #13 | #14 | #15 | #16 | #17 | #18 | #19 | #20 | #21 | #22 | ||
| Quarter 1 | SCC | 0.63 | 0.62 | −0.16 | 0.60 | 0.36 | 0.10 | 0.68 | 0.16 | 0.35 | 0.12 | 0.40 |
| p-value | 0.02 | 0.02 | 0.59 | 0.03 | 0.22 | 0.74 | 0.01 | 0.61 | 0.24 | 0.69 | 0.19 | |
| Quarter 2 | SCC | 0.17 | 0.44 | 0.58 | 0.71 | 0.91 | 0.42 | 0.54 | 0.24 | 0.75 | 0.39 | 0.37 |
| p-value | 0.57 | 0.13 | 0.04 | 0.01 | <0.01 | 0.15 | 0.06 | 0.43 | <0.01 | 0.19 | 0.22 | |
| Quarter 3 | SCC | 0.60 | 0.45 | 0.22 | 0.29 | 0.72 | 0.68 | 0.51 | 0.34 | 0.58 | 0.38 | 0.40 |
| p-value | 0.03 | 0.13 | 0.47 | 0.33 | 0.01 | 0.01 | 0.07 | 0.26 | 0.04 | 0.19 | 0.18 | |
| Quarter 4 | SCC | 0.09 | −0.16 | −0.39 | −0.28 | 0.56 | 0.07 | −0.31 | −0.18 | 0.38 | 0.19 | 0.28 |
| p-value | 0.75 | 0.58 | 0.17 | 0.33 | 0.04 | 0.82 | 0.27 | 0.53 | 0.19 | 0.53 | 0.34 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petersen, F.; Hubbart, J.A. Spatial and Temporal Characterization of Escherichia coli, Suspended Particulate Matter and Land Use Practice Relationships in a Mixed-Land Use Contemporary Watershed. Water 2020, 12, 1228. https://doi.org/10.3390/w12051228
Petersen F, Hubbart JA. Spatial and Temporal Characterization of Escherichia coli, Suspended Particulate Matter and Land Use Practice Relationships in a Mixed-Land Use Contemporary Watershed. Water. 2020; 12(5):1228. https://doi.org/10.3390/w12051228
Chicago/Turabian StylePetersen, Fritz, and Jason A. Hubbart. 2020. "Spatial and Temporal Characterization of Escherichia coli, Suspended Particulate Matter and Land Use Practice Relationships in a Mixed-Land Use Contemporary Watershed" Water 12, no. 5: 1228. https://doi.org/10.3390/w12051228
APA StylePetersen, F., & Hubbart, J. A. (2020). Spatial and Temporal Characterization of Escherichia coli, Suspended Particulate Matter and Land Use Practice Relationships in a Mixed-Land Use Contemporary Watershed. Water, 12(5), 1228. https://doi.org/10.3390/w12051228






