Optimization of Green Extraction and Purification of PHA Produced by Mixed Microbial Cultures from Sludge
Abstract
1. Introduction
2. Material and Methods
2.1. Fermentation Process for PHA Production
2.2. Homogenization of Biomass
2.3. Extraction Process
2.4. Purification Process
2.5. Analytical Methods
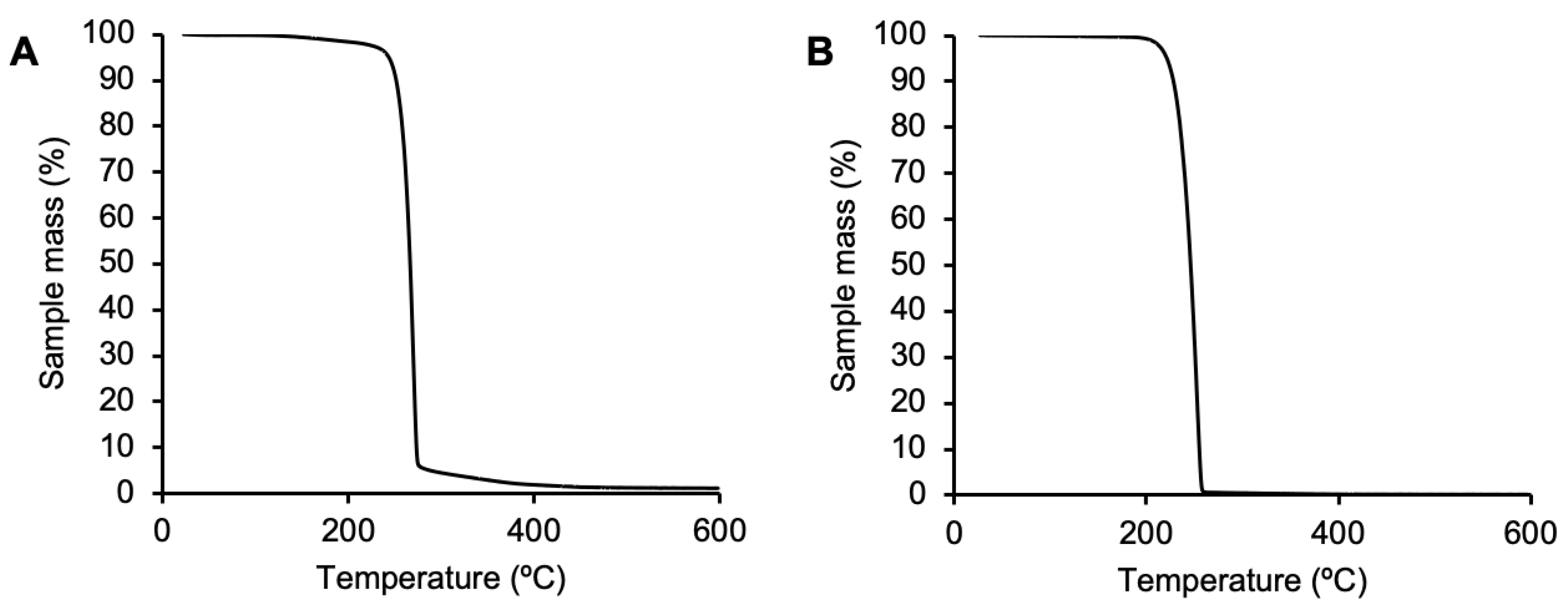
2.5.1. Thermal Gravimetric Analysis (TGA)
2.5.2. Gas Chromatography/Mass Spectrometry (GC-MS)
Sample Preparation
GC-MS Settings
3. Results and Discussion
3.1. PHA Accumulation
3.2. Extraction
3.3. Purification
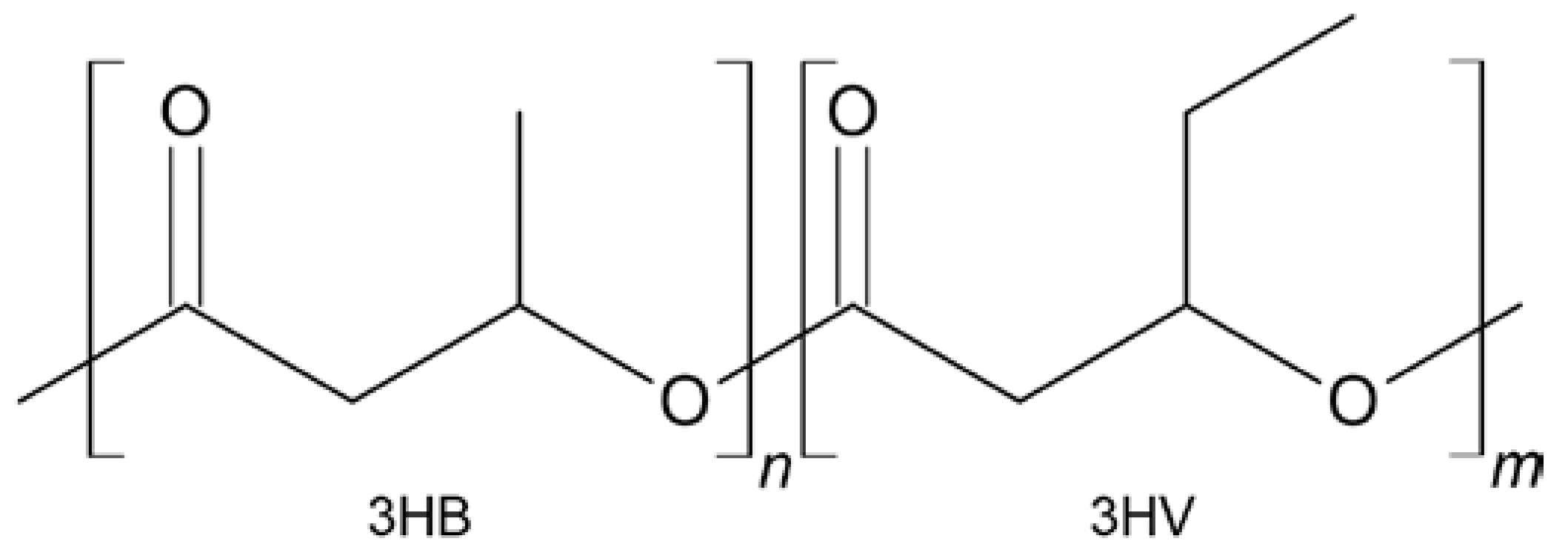
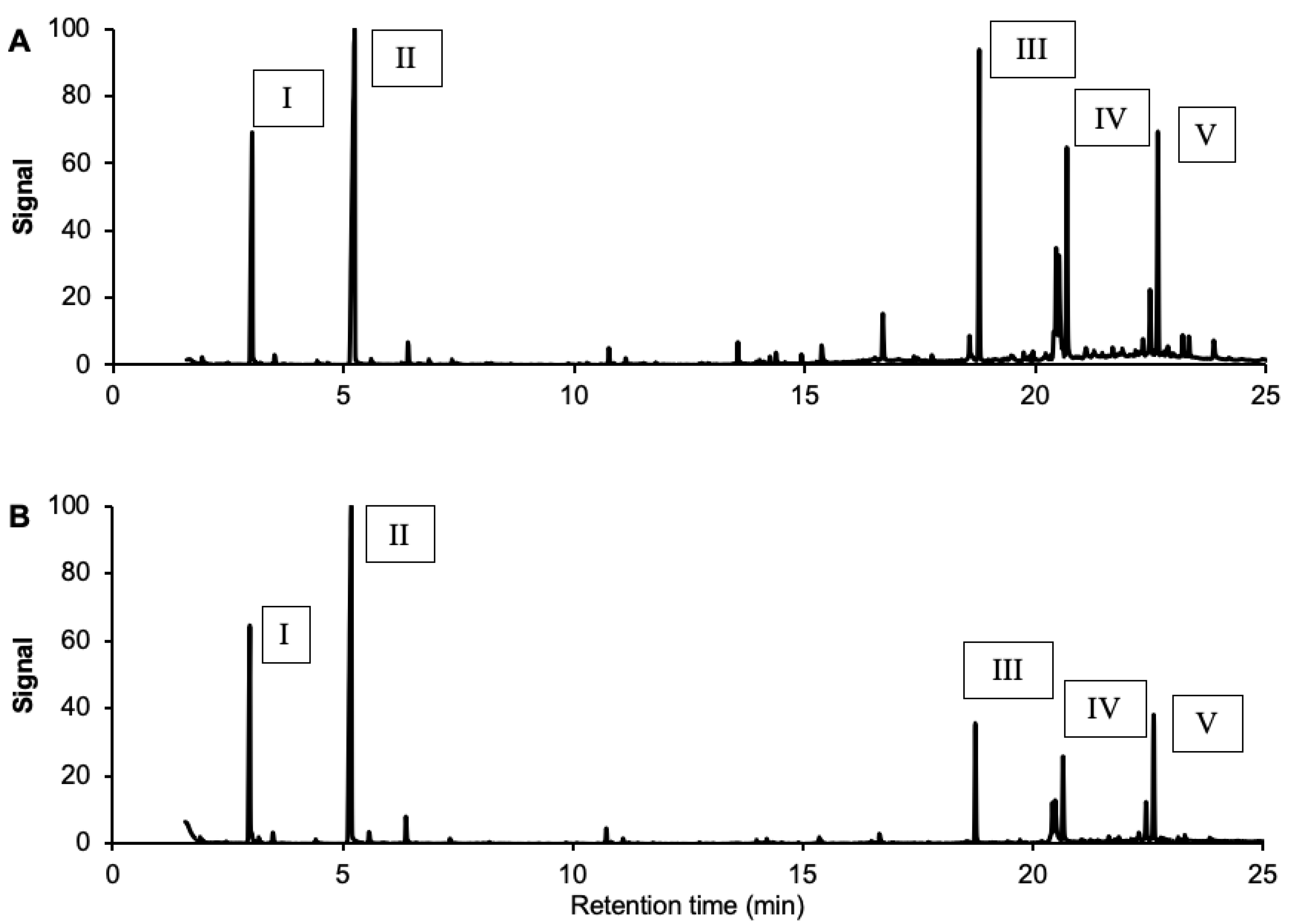
3.4. Analysis by Gas Chromatography-Mass Spectrometry (GC-MS)
3.5. Applicability
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 3HB | 3-hydroxybutyrate |
| 3HV | 3-hydroxyvalerate |
| COD | Chemical oxygen demand |
| DMC | Dimethyl carbonate |
| DO | Dissolved oxygen |
| GC-MS | Gas chromatography—mass spectrometry |
| MMC | Mixed Microbial Culture |
| PHAs | Polyhydroxyalkanoates |
| PHBV | Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) |
| TGA | Thermal gravimetric analysis |
| VFAs | Volatile fatty acids |
| VSS | Volatile suspended solids |
References
- Laycock, B.; Halley, P.; Pratt, S.; Werker, A.; Lant, P. The chemomechanical properties of microbial polyhydroxyalkanoates. Prog. Polym. Sci. 2014, 39, 397–442. [Google Scholar] [CrossRef]
- Shrivastav, A.; Kim, H.; Kim, Y. Advances in the Applications of Polyhydroxyalkanoate Nanoparticles for Novel Drug Delivery System. Biomed Res. Int. 2013, 2013, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-Q. Plastics from Bacteria: Natural Functions and Applications. Microbiol. Monogr. 2009, 14, 121–132. [Google Scholar]
- Elain, A.; Le Grand, A.; Corre, Y.-M.; Le Fellic, M.; Hachet, N.; Le Tilly, V.; Loulergue, P.; Audic, J.-L.; Bruzaud, S. Valorisation of local agro-industrial processing waters as growth media for polyhydroxyalkanoates (PHA) production. Ind. Crops Prod. 2016, 80, 1–5. [Google Scholar] [CrossRef]
- Koller, M.; Marsalek, L.; de Sousa Dias, M.M.; Braunegg, G. Producing microbial polyhydroxyalkanoate (PHA) biopolyesters in a sustainable manner. New Biotechnol. 2017, 37, 24–38. [Google Scholar] [CrossRef]
- Tamis, J.; Luzkov, K.; Jiang, Y.; van Loosdrecht, M.C.M.; Kleerebezem, R. Enrichment of Plasticicumulans acidivorans at pilot-scale for PHA production on industrial wastewater. J. Biotechnol. 2014, 192, 161–169. [Google Scholar] [CrossRef]
- Nikodinovic-Runic, J.; Guzik, M.; Kenny, S.T.; Babu, R.; Werker, A.; Connor, K.E. Carbon-Rich Wastes as Feedstocks for Biodegradable Polymer (Polyhydroxyalkanoate) Production Using Bacteria. Adv. Appl. Microbiol. 2013, 84, 139–200. [Google Scholar] [PubMed]
- Werker, A.G.; Johansson, P.S.T.; Magnusson, P.O.G.; Maurer, F.H.J.; Jannasch, P. Method for Recovery of Stabilized Polyhydroxyalkanoates from Biomass that Has been Used to Treat Organic Waste. US2013/0203954A1, 2013. Available online: https://patents.google.com/patent/US20130203954A1/fr (accessed on 2 March 2020).
- Bengtsson, S.; Werker, A.; Visser, C.; Korving, L. PHARIO—Stepping Stone for a Sustainable Value Chain for PHA Bioplastic Using Municipal Activated Sludge; Stowa: Amersfoort, The Netherlands, 2017; p. 15. [Google Scholar]
- Oliveira, C.S.S.; Silva, C.E.; Carvalho, G.; Reis, M.A.M. Strategies for efficiently selecting PHA producing mixed microbial cultures using complex feedstocks: Feast and famine regime and uncoupled carbon and nitrogen availabilities. New Biotechnol. 2017, 37, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, G.; Oehmen, A.; Albuquerque, M.G.E.; Reis, M.A.M. The relationship between mixed microbial culture composition and PHA production performance from fermented molasses. New Biotechnol. 2014, 31, 257–263. [Google Scholar] [CrossRef]
- Valentino, F.; Karabegovic, L.; Majone, M.; Morgan-Sagatsume, F.; Werker, A. Polyhydroxyalkanoate (PHA) storage within a mixed-culture biomass with simultaneous growth as a function of accumulation substrate nitrogen and phosphorus levels. Water Res. 2015, 77, 49–63. [Google Scholar] [CrossRef]
- Lemos, P.C.; Serafim, L.S.; Reis, M.A.M. Synthesis of polyhydroxyalkanoates from different short-chain fatty acids by mixed cultures submitted to aerobic dynamic feeding. J. Biotechnol. 2006, 122, 226–238. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, S. The utilization of glycogen accumulating organisms for mixed culture production of polyhydroxyalkanoates. Biotechnol. Bioeng. 2009, 104, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Dionisi, D.; Levantesi, C.; Renzi, V.; Tandoi, V.; Majone, M. PHA storage from several substrates by different morphological types in an anoxic/aerobic SBR. Water Sci. Technol. 2002, 46, 337–344. [Google Scholar] [CrossRef][Green Version]
- Kessler, B.; Witholt, B. Factors involved in the regulatory network of polyhydroxyalkanoate metabolism. J. Biotechnol. 2001, 86, 97–104. [Google Scholar] [CrossRef]
- Wang, J.; Yue, Z.-B.; Sheng, G.-P.; Yu, H.-Q. Kinetic analysis on the production of polyhydroxyalkanoates from volatile fatty acids by Cupriavidus necator with a consideration of substrate inhibition, cell growth, maintenance, and product formation. Biochem. Eng. J. 2010, 49, 422–428. [Google Scholar] [CrossRef]
- Pardelha, F.; Albuquerque, M.G.E.; Reis, M.A.M.; Dias, J.M.L.; Oliveira, R. Flux balance analysis of mixed microbial cultures: Application to the production of polyhydroxyalkanoates from complex mixtures of volatile fatty acids. J. Biotechnol. 2012, 162, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, M.G.E.; Martino, V.; Pollet, E.; Avérous, L.; Reis, M.A.M. Mixed culture polyhydroxyalkanoate (PHA) production from volatile fatty acid (VFA)-rich streams: Effect of substrate composition and feeding regime on PHA productivity, composition and properties. J. Biotechnol. 2011, 151, 66–76. [Google Scholar] [CrossRef]
- Hao, J.; Wang, H.; Wang, X. Selecting optimal feast-to-famine ratio for a new polyhydroxyalkanoate (PHA) production system fed by valerate-dominant sludge hydrolysate. Appl. Microbiol. Biotechnol. 2018, 102, 3133–3143. [Google Scholar] [CrossRef]
- Chen, G.Q.; Zhang, G.; Park, S.J.; Lee, S.Y. Industrial scale production of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate). Appl. Microbiol. Biotechnol. 2001, 57, 50–55. [Google Scholar]
- Manangan, T.; Shawaphun, S. Quantitative extraction and determination of polyhydroxyalkanoate accumulated in Alcaligenes latus dry cells. Scienceasia 2010, 36, 199–203. [Google Scholar] [CrossRef]
- Riedel, S.L.; Brigham, C.J.; Budde, C.F.; Bader, J.; Rha, C.K.; Stahl, U.; Sinskey, A.J. Recovery of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) from Ralstonia eutrophacultures with non-halogenated solvents. Biotechnol. Bioeng. 2012, 110, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Ramsay, J.A.; Ramsay, B.A. Acetone extraction of mcl-PHA from Pseudomonas putida KT2440. J. Microbiol. Methods 2006, 67, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Dacosta, C.; Posada, J.A.; Kleerebezem, R.; Cuellar, M.C.; Ramirez, A. Microbial community-based polyhydroxyalkanoates (PHAs) production from wastewater: Techno-economic analysis and ex-ante environmental assessment. Bioresour. Technol. 2015, 185, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Fei, T.; Cazeneuve, S.; Wen, Z.; Wu, L.; Wang, T. Effective Recovery of Poly-b-Hydroxybutyrate (PHB) Biopolymer from Cupriavidus necator Using a Novel and Environmentally Friendly Solvent System. Biotechnol. Prog. 2016, 32, 678–685. [Google Scholar] [CrossRef]
- Samorì, C.; Abbondanzi, F.; Galletti, P.; Giorgini, L.; Mazzocchetti, L.; Torri, C.; Tagliavini, E. Extraction of polyhydroxyalkanoates from mixed microbial cultures: Impact on polymer quality and recovery. Bioresour. Technol. 2015, 189, 195–202. [Google Scholar] [CrossRef]
- Samorì, C.; Basaglia, M.; Casella, S.; Favaro, L.; Galletti, P.; Giorgini, L.; Marchi, D.; Mazzocchetti, L.; Torri, C.; Tagliavini, E. Dimethyl carbonate and Switchable Anionic Surfactants: Two effective tools for the extraction of polyhydroxyalkanoates from microbial biomass. Green Chem. 2015, 17, 1047–1056. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. PubChem Database. Dimethyl Carbonate, CID:12021. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Dimethyl-carbonate (accessed on 2 March 2020).
- Hansch, C.; Leo, A.; Hoekman, D. Exploring QSAR—Hydrophobic, Electronic and Steric Constants; American Chemical Society: Washington, DC, USA, 1995; pp. 3–14. [Google Scholar]
- Werker, A.G.; Johansson, P.S.T.; Magnusson, P.O.G. Process for the Extraction of Polyhydroxyalkanoates from Biomass. US2015/0368393A1, 2015. Available online: https://patents.google.com/patent/US20150368393/en17 (accessed on 2 March 2020).
- Carrier, M.; Loppinet-Serani, A.; Denux, D.; Lasnier, J.-M.; Ham-Pichavant, F.; Cansell, F.; Aymonier, C. Thermogravimetric analysis as a new method to determine the lignocellulosic composition of biomass. Biomass Bioenergy 2011, 35, 298–307. [Google Scholar] [CrossRef]
- Lo, C.-W.; Wu, H.-S.; Wei, Y.-H. Optimizing acidic methanolysis of poly(3-hydroxyalkanoates) in gas chromatography analysis. Asia Pac. J. Chem. Eng. 2009, 4, 487–494. [Google Scholar] [CrossRef]
- Hahn, S.K.; Chang, Y.K. A thermogravimetric analysis for poly(3-hydroxybutyrate) quantification. Biotechnol. Tech. 1995, 9, 873–878. [Google Scholar] [CrossRef]
- Bengtsson, S.; Pisco, A.R.; Johansson, P.; Lemos, P.C.; Reis, M.A.M. Molecular weight and thermal properties of polyhydroxyalkanoates produced from fermented sugar molasses by open mixed cultures. J. Biotechnol. 2010, 147, 172–179. [Google Scholar] [CrossRef]
- Carrasco, F.; Dionisi, D.; Beccari, M.; Majone, M.; Martinelli, A. Thermal stability of polyhydroxyalkanotaes. J. Appl. Polym. Sci. 2006, 100, 2111–2121. [Google Scholar] [CrossRef]
- Keenan, T.M.; Tanenbaum, S.W.; Stipanovic, A.J.; Nakas, J.P. Production and characterization of poly-beta-hydroxyalkanoate copolymers from Burkholderia cepacia utilizing xylose and levulinic acid. Biotechnol. Prog. 2004, 20, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Chen, Y. The effects of the ratio of propionate to acetate on the transformation and composition of polyhydroxyalkanoates with enriched cultures of glycogen-accumulating organisms. Environ. Technol. 2009, 30, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Lambert, L.; Yuan, Z.; Keller, J. Characterisation of polyhydroxyalkanoate copolymers with controllable four-monomer composition. J. Biotechnol. 2008, 134, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Montano-Herrera, L.; Laycock, B.; Werker, A.; Pratt, S. The Evolution of Polymer Composition during PHA Accumulation: The Significance of Reducing Equivalents. Bioengineering 2017, 4, 20. [Google Scholar] [CrossRef]
- Castro, A.R.; Silva, P.T.S.; Castro, P.J.G.; Alves, E.; Domingues, M.R.M.; Pereira, M.A. Tuning culturing conditions towards the production of neutral lipids from lubricant-based wastewater in open mixed bacterial communities. Water Res. 2018, 144, 532–542. [Google Scholar] [CrossRef]

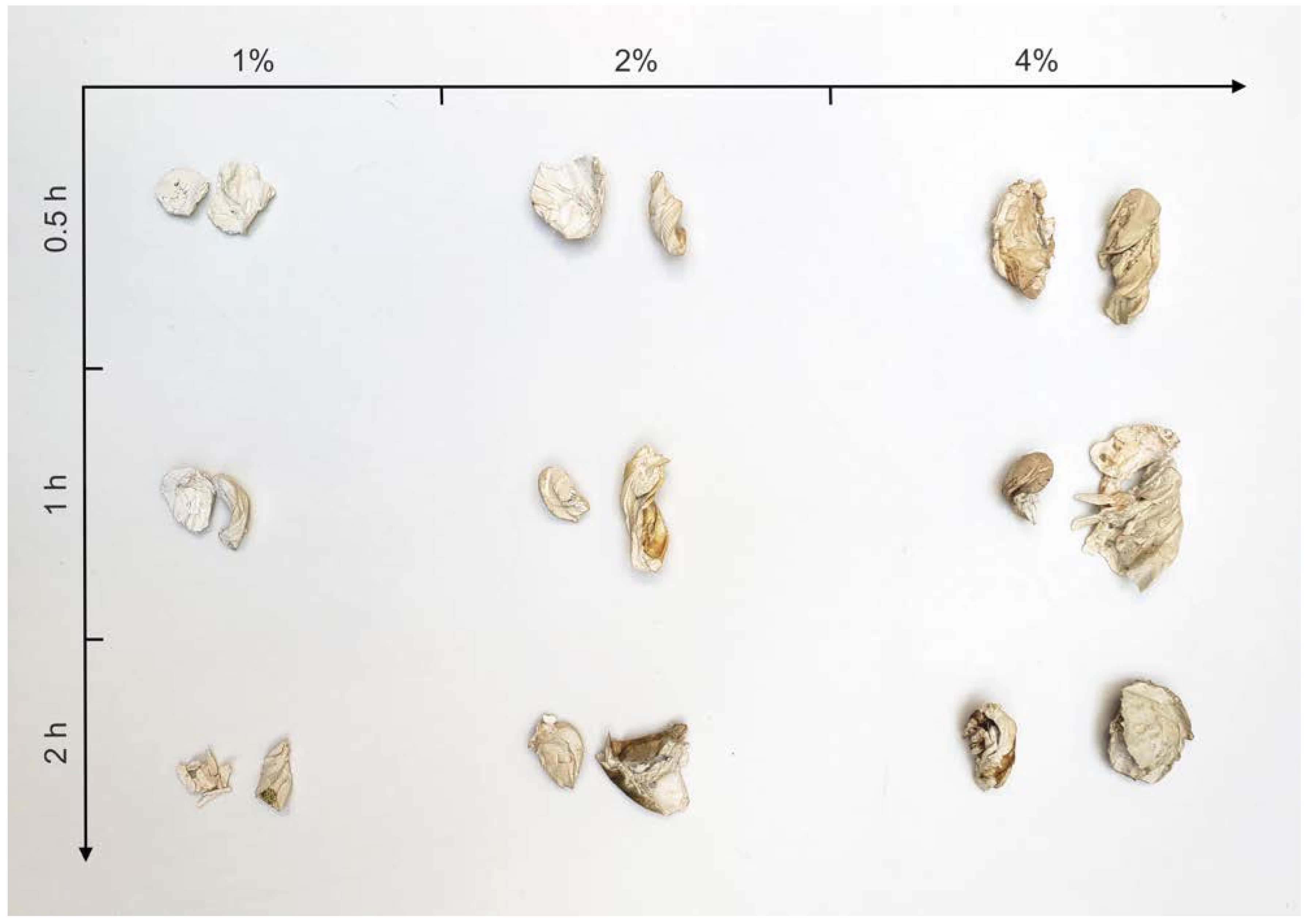
| Biomass to Solvent Ratio (%) | 0.25 h | 0.5 h | 1 h | 1.5 h | 2 h |
|---|---|---|---|---|---|
| 1% | 31.4 ± 1.0abc | 32.2 ± 0.1b | 31.7 ± 0.2bd | 32.9 ± 0.2b | |
| 2.5% | 30.6 ± 0.4cd | 30.4 ± 0.3cd | 31.5 ± 0.1ad | 32.3 ± 0.4bd | |
| 5% | 30.5 ± 0.1c | 30.0 ± 1.0abc | 30.8 ± 0.7abc | 30.4 ± 1.3abc | |
| 10% | 29.3 ± 1.1abc | 27.8 ± 0.4a | 28.2 ± 0.5ac |
| Solvent | g PHA/100 g Biomass |
|---|---|
| Dimethyl carbonate | 31.7 ± 0.2 |
| Chloroform | 37.5 ± 0.2 |
| Dichloromethane | 39.0 ± 0.2 |
| PHA to Solvent Ratio (%) | 0.5 h | 1 h | 2 h |
|---|---|---|---|
| 1% | 98.3 ± 0.1acd | 98.5 ± 0.1a | 98.2 ± 0.2acd |
| 2% | 98.4 ± 0.1bc | 98.9 ± 0.1ac | 97.9 ± 0.4acd |
| 4% | 98.3 ± 0.3acd | 98.2 ± 0.1bd | 98.4 ± 0.3acd |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Souza Reis, G.A.; Michels, M.H.A.; Fajardo, G.L.; Lamot, I.; de Best, J.H. Optimization of Green Extraction and Purification of PHA Produced by Mixed Microbial Cultures from Sludge. Water 2020, 12, 1185. https://doi.org/10.3390/w12041185
de Souza Reis GA, Michels MHA, Fajardo GL, Lamot I, de Best JH. Optimization of Green Extraction and Purification of PHA Produced by Mixed Microbial Cultures from Sludge. Water. 2020; 12(4):1185. https://doi.org/10.3390/w12041185
Chicago/Turabian Stylede Souza Reis, Guilherme A., Michiel H. A. Michels, Gabriela L. Fajardo, Ischa Lamot, and Jappe H. de Best. 2020. "Optimization of Green Extraction and Purification of PHA Produced by Mixed Microbial Cultures from Sludge" Water 12, no. 4: 1185. https://doi.org/10.3390/w12041185
APA Stylede Souza Reis, G. A., Michels, M. H. A., Fajardo, G. L., Lamot, I., & de Best, J. H. (2020). Optimization of Green Extraction and Purification of PHA Produced by Mixed Microbial Cultures from Sludge. Water, 12(4), 1185. https://doi.org/10.3390/w12041185





