Controlled Reservoir Drawdown—Challenges for Sediment Management and Integrative Monitoring: An Austrian Case Study—Part B: Local Scale
Abstract
:1. Introduction
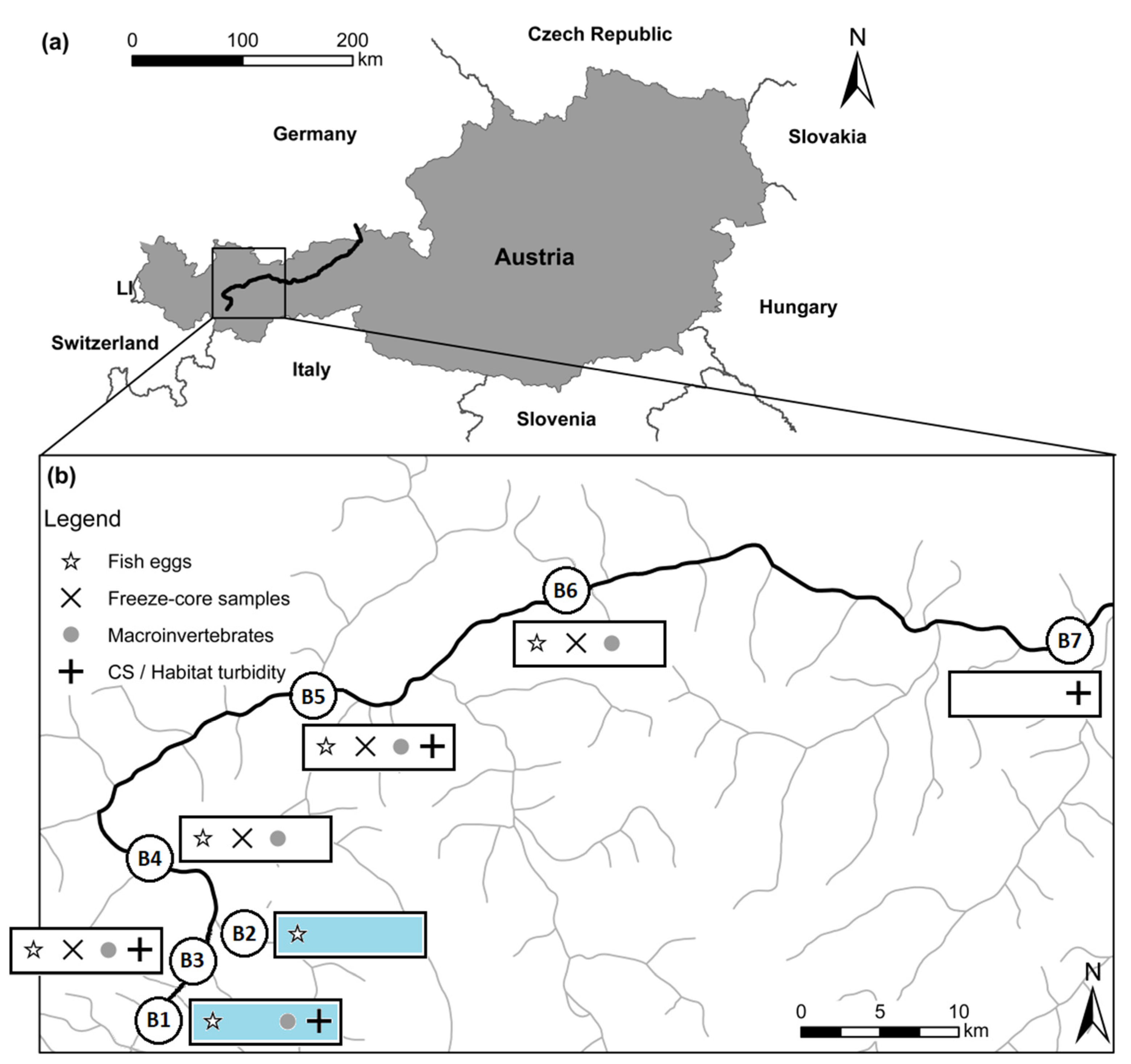
2. Materials and Methods
2.1. Cross section Variability and Habitat-Related Turbidity Measurements
2.2. Freeze-Core Sampling
2.3. Macroinvertebrates
2.4. Fish—Salmonid Incubation
3. Results
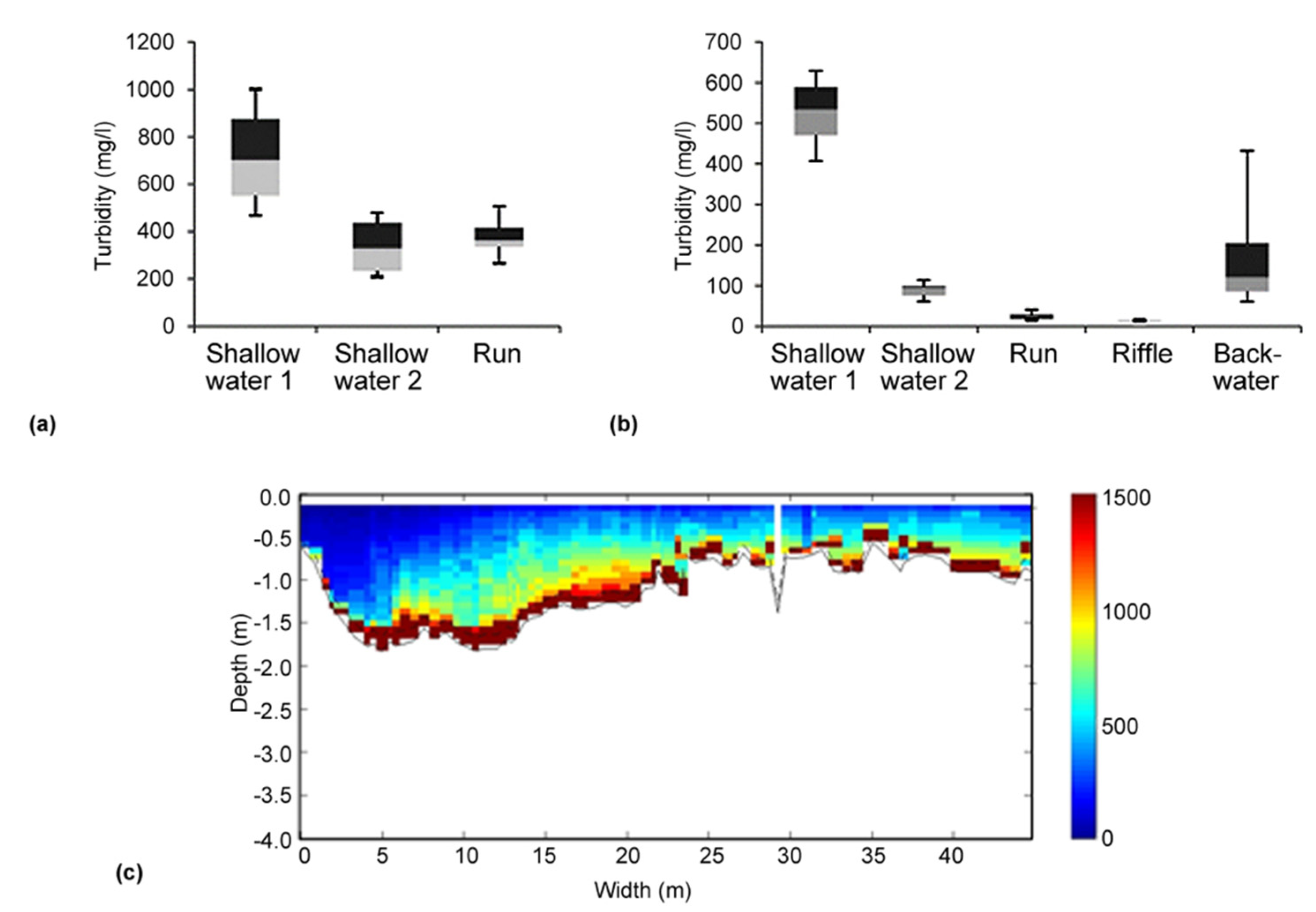
3.1. Cross Section Variability and Habitat-Related Turbidity Measurements
3.2. Freeze-Core Sampling
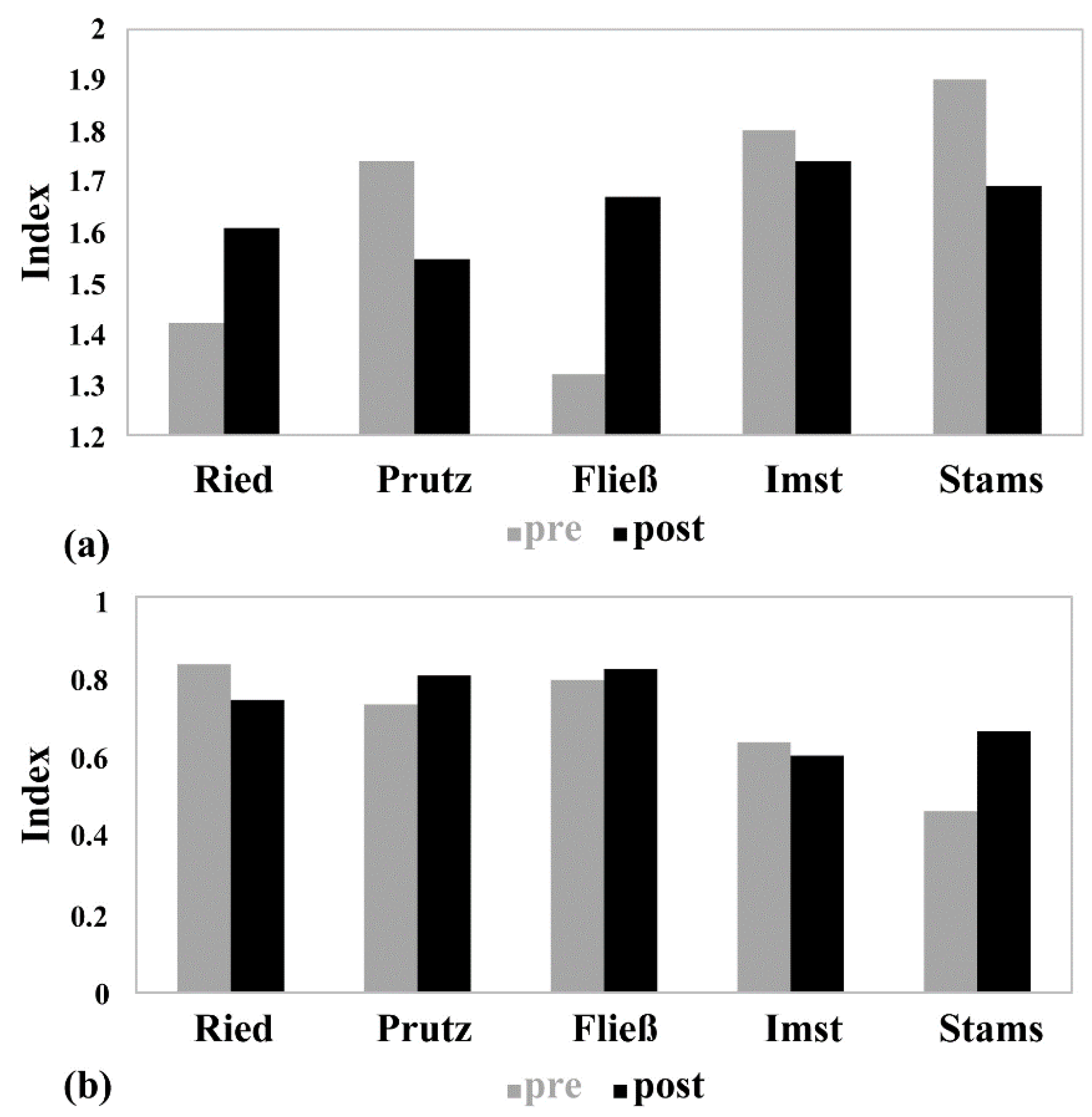
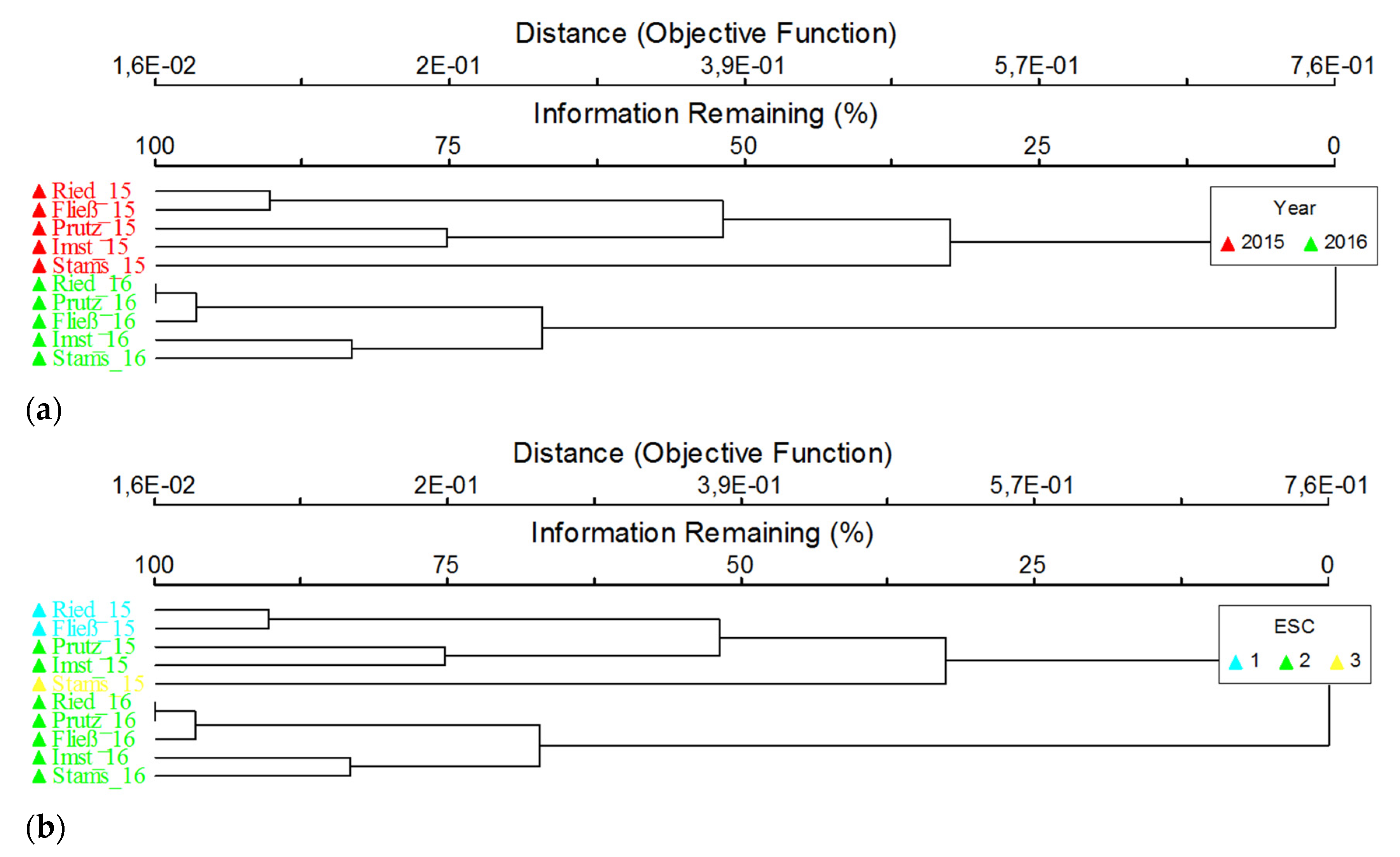
3.3. Macroinvertebrates
3.4. Fish: Salmonid Incubation
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zarfl, C.; Lumsdon, A.E.; Berlekamp, J.; Tydecks, L.; Tockner, K. A global boom in hydropower dam construction. Aquat. Sci. 2015, 77, 161–170. [Google Scholar] [CrossRef]
- Moog, O. Quantification of daily peak hydropower effects on aquatic fauna and management to minimize environmental impacts. Regul. Rivers Res. Manag. 1993, 8, 5–14. [Google Scholar] [CrossRef]
- Hauer, C.; Holzapfel, P.; Leitner, P.; Graf, W. Longitudinal assessment of hydropeaking impacts on various scales for an improved process understanding and the design of mitigation measures. Sci. Total Environ. 2017, 575, 1503–1514. [Google Scholar] [CrossRef]
- Dugan, P.J.; Barlow, C.; Agostinho, A.A.; Baran, E.; Cada, G.F.; Chen, D.; Marmulla, G. Fish migration, dams, and loss of ecosystem services in the Mekong basin. AMBIO 2010, 39, 344–348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, E.P.; Freeman, M.C.; Pringle, C.M. Ecological consequences of hydropower development in Central America: Impacts of small dams and water diversion on neotropical stream fish assemblages. River Res. Appl. 2006, 22, 397–411. [Google Scholar] [CrossRef]
- Ziv, G.; Baran, E.; Nam, S.; Rodríguez-Iturbe, I.; Levin, S.A. Trading-off fish biodiversity, food security, and hydropower in the Mekong River Basin. Proc. Natl. Acad. Sci. USA 2012, 109, 5609–5614. [Google Scholar] [CrossRef] [Green Version]
- Gray, L.J.; Ward, J.V. Effects of sediment releases from a reservoir on stream macroinvertebrates. Hydrobiologia 1982, 96, 177–184. [Google Scholar] [CrossRef]
- Kemp, P.; Sear, D.; Collins, A.; Naden, P.; Jones, I. The impacts of fine sediment on riverine fish. Hydrol. Process. 2011, 25, 1800–1821. [Google Scholar] [CrossRef]
- Crosa, G.; Castelli, E.; Gentili, G.; Espa, P. Effects of suspended sediments from reservoir flushing on fish and macroinvertebrates in an alpine stream. Aquat. Sci. 2010, 72, 85–95. [Google Scholar] [CrossRef]
- Hauer, C.; Holzapfel, P.; Tonolla, D.; Habersack, H.; Zolezzi, G. In situ measurements of fine sediment infiltration (FSI) in gravel-bed rivers with a hydropeaking flow regime. Earth Surf. Process. Landf. 2019, 44, 433–448. [Google Scholar] [CrossRef] [Green Version]
- Smith, E.P.; Orvos, B.W.; Cairns, J.J. Impact assessment using the Before-After-Control-Impact (BACI) model: Concerns and comments. Can. J. Fish. Aquat. Sci. 1993, 50, 627–637. [Google Scholar] [CrossRef]
- Smokorowski, K.E.; Bradford, M.J.; Clarke, K.D.; Clément, M.; Gregory, R.S.; Randall, R.G. Assessing the Effectiveness of Habitat Offset Activities in Canada: Monitoring Design and Metrics; Canada, 2015. Available online: https://waves-vagues.dfo-mpo.gc.ca/Library/347555.pdf (accessed on 1 March 2020).
- Seitz, L.; Noack, M.; Haun, S.; Reindl, R.; Senn, G.; Schletterer, M. Analysing sediment characteristics of the alpine river Brixentaler Ache (Austria) including in situ measurements of dissolved oxygen. In Proceedings of the 13th International Symposium on River Sedimentation, Stuttgart, Germany, 19–22 September 2016; pp. 915–921. [Google Scholar]
- Evans, E.; Wilcox, A.C. Fine sediment infiltration dynamics in a gravel-bed river following a sediment pulse. River Res. Appl. 2014, 30, 372–384. [Google Scholar] [CrossRef]
- Hauer, C.; Holzapfel, P.; Flödl, P.; Wagner, B.; Graf, W.; Leitner, P.; Holzer, G.; Haun, S.; Hammer, A.; Habersack, H.; et al. Controlled reservoir drawdown-challenges for sediment management and integrative monitoring: An Austrian case study—Part A: Reach Scale. Water 2020. [Google Scholar]
- McLeay, D.J.; Birtwell, I.K.; Hartman, G.H.; Ennis, G.L. Response of Arctic Grayling (Thymallusarcticus) to acute and prolonged exposure to Yukon placer mining sediment. Can. J. Fish. Aquat. Sci. 1987, 44, 658–673. [Google Scholar] [CrossRef]
- Doeg, T.J.; Milledge, G.A. Effect of experimentally increasing concentration of suspended sediment on macroinvertebrate drift. Mar. Freshw. Res. 1991, 42, 519–526. [Google Scholar] [CrossRef]
- Newcombe, C.P.; MacDonald, D.D. Effects of suspended sediments on aquatic ecosystems. North Am. J. Fish. Manag. 1991, 11, 72–82. [Google Scholar] [CrossRef]
- Auld, A.H.; Schubel, J.R. Effects of suspended sediment on fish eggs and larvae: A laboratory assessment. Estuar. Coast. Mar. Sci. 1978, 6, 153–164. [Google Scholar] [CrossRef]
- Newcombe, C.P.; Jensen, J.O. Channel suspended sediment and fisheries: A synthesis for quantitative assessment of risk and impact. North Am. J. Fish. Manag. 1996, 16, 693–727. [Google Scholar] [CrossRef]
- Walkotten, W.J. A Freezing Technique for Sampling Streambed Gravel; Northwest Forest and Range Experimental Station: Portland, OR, USA, 1976. [Google Scholar]
- Carling, P.A.; Reader, N.A. A freeze-sampling technique suitable for coarse river bed-material. Sediment. Geol. 1981, 29, 233–239. [Google Scholar] [CrossRef]
- Kondolf, G.M.; Lisle, T.E. Measuring bed sediment. In Tools in Fluvial Geomorphology; Kondolf, G.M., Piégay, H., Eds.; Wiley Blackwell: Hoboken, NJ, USA, 2016; pp. 278–305. [Google Scholar]
- Bunte, K.; Abt, S.R. Sampling Surface and Subsurface Particle-Size Distributions in Wadable Gravel- and Cobble-Bed Streams for Analyses in Sediment. Transport, Hydraulics and Streambed Monitoring; USDA: Washington, DC, USA, 2001.
- Tritthart, M.; Haimann, M.; Habersack, H.; Hauer, C. Spatio-temporal variability of suspended sediments in rivers and ecological implications of reservoir flushing operations. River Res. Appl. 2019, 35, 918–931. [Google Scholar] [CrossRef] [Green Version]
- Baran, E.; Nasielski, J. Reservoir Sediment Flushing and Fish Resources; Natural Heritage Institute: San Francisco, CA, USA, 2011. [Google Scholar]
- Scheurer, T.; Molinari, P. Experimental floods in the River Spöl, Swiss National Park: Framework, objectives and design. Aquat. Sci. 2003, 65, 183–190. [Google Scholar] [CrossRef]
- Wendelaar Bonga, S.E. The stress response in fish. Physiol. Rev. 1997, 77, 591–625. [Google Scholar] [CrossRef]
- Gorman, O.; Karr, J.R. Habitat Structure and Stream Fish Communities. Ecology 1978, 59, 507–515. [Google Scholar] [CrossRef]
- Glova, G.J.; McInerney, J.E. Critical swimming speeds of coho salmon (Oncorhynchus kisutch) fry to smolt stages in relation to salinity and temperature. J. Fish. Board Can. 1977, 34, 151–154. [Google Scholar] [CrossRef]
- Schletterer, M.; Hofer, B.; Obendorfer, R.; Hammer, A.; Hubmann, M.; Schwarzenberger, R.; Boschi, M.; Haun, S.; Haimann, M.; Holzapfel, P.; et al. Integrative monitoring approaches for the sediment management in Alpine reservoirs: Case study Gepatsch (HPP Kaunertal, Tyrol). In Proceedings of the 13th International Symposium on River Sedimentation, Stuttgart, Germany, 19–22 September 2016; pp. 1161–1169. [Google Scholar]
- Edwards, T.K.; Glysson, G.D. Field Methods for Measurement of Fluvial Sediment; U.S. Geological Survey, Techniques of Water-Resources Investigations, 1999. Available online: https://pubs.usgs.gov/twri/twri3-c2/ (accessed on 18 March 2020).
- Haun, S.; Rüther, N.; Baranya, S.; Guerrero, M. Comparison of real time suspended sediment transport measurements in river environment by LISST instruments in stationary and moving operation mode. Flow Meas. Instrum. 2015, 41, 10–17. [Google Scholar] [CrossRef]
- Haun, S.; Lizano, L. Sensitivity analysis of sediment flux derived by laser diffraction and acoustic backscatter within a reservoir. Int. J. Sediment. Res. 2018, 33, 18–26. [Google Scholar] [CrossRef]
- Christopher James, G. Potential of turbidity monitoring for measuring the transport of suspended solids in streams. Hydrol. Process. 1995, 9, 83–97. [Google Scholar]
- Hauer, C.; Unfer, G.; Tritthart, M.; Formann, E.; Habersack, H. Variability of mesohabitat characteristics in riffle-pool reaches: Testing an integrative evaluation concept (FGC) for MEM-application. River Res. Appl. 2011, 27, 403–430. [Google Scholar] [CrossRef]
- Hauer, C.; Mandlburger, G.; Habersack, H. Hydraulically related hydro-morphological units: Description based on a new conceptual mesohabitat evaluation model (MEM) using LiDAR data as geometric input. River Res. Appl. 2009, 25, 29–47. [Google Scholar] [CrossRef]
- Humpesch, U.H.; Niederreiter, R. Freeze-core method for sampling the vertical distribution of the macrozoobenthos in the main channel of a large deep river, the River Danube at kilometre 1889. Large Rivers 1993, 9, 87–90. [Google Scholar] [CrossRef]
- Lisle, T.E.; Eads, R.E. Methods to Measure Sedimentation of Spawning Gravels; Pacific Southwest Research Station, Forest Service, US Department of Agriculture: Berkeley, CA, USA, 1991.
- Zimmermann, A.E.; Lapointe, M. Intergranular flow velocity through salmonid redds: Sensitivity to fines infiltration from low intensity sediment transport events. River Res. Appl. 2005, 21, 865–881. [Google Scholar] [CrossRef]
- Ofenböck, T.; Moog, O.; Hartmann, A.; Stubauer, I. Leitfaden zur Erhebung der Biologischen Qualitätselemente, Teil A2—Makrozoobenthos; Umwelt und Wasserwirtschaft: Vienna, Austria, 2015; Available online: https://www.bmlrt.gv.at/wasser/wisa/fachinformation/ngp/ngp-2009/hintergrunddokumente/methodik/biologische_qe.html (accessed on 13 November 2019).
- Moog, D.B.; Whiting, P.J.; Thomas, R.B. Streamflow record extension using power transformations and application to sediment transport. Water Resour. Res. 1999, 35, 243–254. [Google Scholar] [CrossRef]
- Wien, Ö.N. ÖNORM M 6232—Richtlinie für die ökologische Untersuchung und Bewertung von Fließgewässern. 1997, p. 38. Available online: https://shop.austrian-standards.at/action/de/public/details/54417/OENORM_M_6232_1997_05_01 (accessed on 13 November 2019).
- Zelinka, M.; Marvan, P. Zur Präzisierung der biologischen Klassifikation der Reinheit fließender Gewässer. Arch. Hydrobiol. 1961, 57, 389–407. [Google Scholar]
- Moog, O.; Hartmann, A.; Schmidt-Kloiber, A.; Vogl, R.; Koller-Kreimel, V. ECOPROF—Version 4.0. Software zur Bewertung des Ökologischen Zustandes von Fliessgewässern nach WRRL. 2013. Available online: https://www.ecoprof.at/ (accessed on 13 November 2019).
- Eberstaller, J.; Köck, J.; Haunschmid, R.; Jagsch, A.; Ratschan, C.; Zauner, G. Leitfanden zur Bewertung Erheblich Veränderter Gewässer—Biologische Definition des Guten Ökologischen Potentials; IA des Bundesministeriums für Land-und Forstwirtschaft, Umwelt und Wasserwirtschaft: Vienna, Austria, 2015; Available online: https://www.bmlrt.gv.at/wasser/wasser-oesterreich/plan_gewaesser_ngp/nationaler_gewaesserbewirtschaftungsplan-ngp/hmwb_lf.html (accessed on 13 November 2019).
- Holzer, G.; Unfer, G.; Hinterhofer, M. Cocooning—eine alternative Methode zur fischereilichen Bewirtschaftung. Österreichs Fischerei 2011, 64, 16–27. [Google Scholar]
- Louhi, P.A.; Mäki-Petä, Y.S.; Erkinaro, J. Spawning habitat of Atlantic salmon and Brown trout: General criteria and intragravel factors. River Res. Appl. 2008, 24, 330–339. [Google Scholar] [CrossRef]
- Pulg, U.; Barlaup, B.T.; Sternecker, K.; Trepl, L.; Unfer, G. Restoration of spawning habitats of brown trout (Salmo trutta) in a regulated chalk stream. River Res. Appl. 2013, 29, 172–182. [Google Scholar] [CrossRef]
- Chapman, D.W. Critical review of variables used to define effects of fines in redds of large salmonids. Trans. Am. Fish. Soc. 1988, 117, 1–21. [Google Scholar] [CrossRef]
- Lisle, T.E. Sediment transport and resulting deposition in spawning gravels, North coastal California. Water Resour. Res. 1989, 25, 1303–1319. [Google Scholar] [CrossRef] [Green Version]
- Pauwels, S.J.; Haines, T.A. Survival, hatching, and emergence success of Atlantic salmon eggs planted in three Maine streams. North Am. J. Fish. Manag. 1994, 14, 125–130. [Google Scholar] [CrossRef] [Green Version]
- Sear, D.A. Fine sediment infiltration into gravel spawning beds within a regulated river experiencing floods: Ecological implications for salmonids. Regul. Rivers Res. Manag. 1993, 8, 373–390. [Google Scholar] [CrossRef]
- Seitz, L.; Christian, H.; Noack, M.; Wieprecht, S. From picture to porosity of river bed material using Structure-from-Motion with Multi-View-Stereo. Geomorphology 2018, 306, 80–89. [Google Scholar] [CrossRef]
- Tappel, P.D.; Bjornn, T.C. A new method of relating size of spawning gravel to salmonid embryo survival. North Am. J. Fish. Manag. 1983, 3, 123–135. [Google Scholar] [CrossRef] [Green Version]
- Olsson, T.I.; Näslund, I. Effects of mire drainage and peat extraction on benthic invertebrates and fish. In Proceedings of the International Peat Society Symposium; IPS: Hasselfors, Sweden, 1985; pp. 147–152. [Google Scholar]
- Kondolf, G.M. Assessing salmonid spawning gravel quality. Trans. Am. Fish. Soc. 2000, 129, 262–281. [Google Scholar] [CrossRef]
- Lapointe, M.F.; Bergeron, N.E.; Berube, F.; Pouliot, M.A.; Johnston, P. Interactive effects of substrate sand and silt contents, redd-scale hydraulic gradients, and interstitial velocities on egg-to-emergence survival of Atlantic salmon (Salmo salar). Can. J. Fish. Aquat. Sci. 2004, 61, 2271–2277. [Google Scholar] [CrossRef] [Green Version]
- Greig, S.M.; Sear, D.A.; Smallman, D.; Carling, P.A. Impact of clay particles on the cutaneous exchange of oxygen across the chorion of Atlantic salmon eggs. J. Fish. Biol. 2005, 66, 1681–1691. [Google Scholar] [CrossRef]
- Schoener, T.W. Resource partitioning in ecological communities. Science 1974, 185, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Hauer, C.; Unfer, G.; Holzapfel, P.; Haimann, M.; Habersack, H. Impact of channel bar form and grain size variability on estimated stranding risk of juvenile brown trout during hydropeaking. Earth Surf. Process. Landf. 2014, 39, 1622–1641. [Google Scholar] [CrossRef]
- Kondolf, G.M.; Williams, J.G.; Horner, T.C.; Milan, D. Assessing physical quality of spawning habitat. In Salmonid Spawning Habitat in Rivers; Sear, D.A., DeVries, P., Eds.; The American Fisheries Society, American Fisheries Society Symposium: Bethesda, MD, USA, 2008; p. 65. [Google Scholar]
- Adkins, S.C.; Winterbourn, M.J. Vertical distribution and abundance of invertebrates in two New Zealand stream beds: A freeze coring study. Hydrobiologia 1999, 400, 55–62. [Google Scholar] [CrossRef]
- Newbold, J.D.; Thomas, S.A.; Minshall, G.W.; Cushing, C.E.; Georgian, T. Deposition, benthic residence, and resuspension of fine organic particles in a mountain stream. Limnol. Oceanogr. 2005, 50, 1571–1580. [Google Scholar] [CrossRef]
- Weigelhofer, G.; Waringer, J. Vertical distribution of benthic macroinvertebrates in riffles versus deep runs with differing contents of fine sediments (Weidlingbach, Austria). Int. Rev. Hydrobiol. 2003, 88, 304–313. [Google Scholar] [CrossRef]
- Soszynska-Maj, A.; Paasivirta, L.; Giłka, W. Why on the snow? Winter emergence strategies of snow-active Chironomidae (Diptera) in Poland. Insect Sci. 2016, 23, 754–770. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.M.; Ferrington, L.C. Resistance and resilience of winter-emerging Chironomidae (Diptera) to a flood event: Implications for Minnesota trout streams. Hydrobiologia 2013, 707, 59–71. [Google Scholar] [CrossRef]
- Armitage, P.D.; Blackburn, J.H. Environmental stability and communities of Chironomidae (Diptera) in a regulated river. River Res. Appl. 1990, 5, 319–328. [Google Scholar] [CrossRef]
- Wood, P.J.; Armitage, P.D. Biological effects of fine sediment in the lotic environment. Environ. Manag. 1997, 21, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.T.; Uehlinger, U.; Monaghan, M.T. Stream ecosystem response to multiple experimental floods from a reservoir. River Res. Appl. 2004, 20, 359–377. [Google Scholar] [CrossRef]
- Robinson, C.T.; Uehlinger, U.; Monaghan, M.T. Effects of a multi-year experimental flood regime on macroinvertebrates downstream of a reservoir. Aquat. Sci. 2003, 65, 210–222. [Google Scholar] [CrossRef] [Green Version]
- Hering, D.; Carvalho, L.; Argillier, C.; Beklioglu, M.; Borja, A.; Cardoso, A.C.; Duel, H.; Ferreira, T.; Globevnik, L.; Hanganu, J.; et al. Managing aquatic ecosystems and water resources under multiple stress—An introduction to the MARS project. Sci. Total Environ. 2015, 503–504, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Statzner, B.; Higler, B. Stream Hydraulics as a Major Determinant of Benthic Invertebrate Zonation Patterns. Freshw. Biol. 1986, 16, 127–139. [Google Scholar] [CrossRef]
- Hynes, H.B.N. The Effects of Sediment on the Biota in Running water in Fluvial Processes and Sedimentation. In Proceedings of the Hydrology Symposium; National Research Council: Edmonton, AB, Canada, 1973; pp. 151–169. [Google Scholar]
- Richards, C.; Bacon, K.L. Influence of fine sediment on macroinvertebrate colonisation of surface and hyporheic stream sediments. Great Basin Nat. 1994, 54, 106–113. [Google Scholar]
- Descloux, S.; Datry, T.; Marmonier, P. Benthic and hyporheic invertebrate assemblages along a gradient of increasing streambed colmation by fine sediment. Aquat. Sci. 2013, 75, 493–507. [Google Scholar] [CrossRef]
- Holzer, G. Projekt Ilz: Teilprojekt Brutboxen (Cocooning) 2016. Available online: https://www.researchgate.net/publication/275016473_Cocooning_-_eine_alternative_Methode_zur_fischereilichen_Bewirtschaftung (accessed on 8 April 2020).
- Fernandes, J.N.; Boes, R.M.; Titzschkau, M.; Hammer, A.; Haun, S.; Schletterer, M. Sediment load, hydro-abrasive erosion and efficiency changes at high-head turbines during the drawdown of two Alpine reservoirs via the power waterway. In Proceedings of the HYDRO 2016—Hydropower and Dams, International Conference and Exhibition, Montreux, Switzerland, 10–12 October 2016. [Google Scholar]
- Espa, P.; Brignoli, M.L.; Crosa, G.; Gentili, G.; Quadroni, S. Controlled sediment flushing at the Cancano Reservoir (Italian Alps): Management of the operation and downstream environmental impact. J. Environ. Manag. 2016, 182, 1–12. [Google Scholar] [CrossRef] [PubMed]


| Ried | Prutz | Fließ | Imst | Stams | ||
|---|---|---|---|---|---|---|
| SI | 2015 | 1.42 (high) | 1.74 (good) | 1.32 (high) | 1.8 (good) | 1.9 (good) |
| 2016 | 1.61 (good) | 1.55 (good) | 1.67 (good) | 1.74 (good) | 1.69 (good) | |
| MMI | 2015 | 0.83 (high) | 0.73 (good) | 0.79 (good) | 0.63 (good) | 0.46 (moderate) |
| 2016 | 0.74 (good) | 0.8 (high) | 0.82 (high) | 0.6 (good) | 0.66 (good) | |
| status | 2015 | high | good | good | good | moderate |
| 2016 | good | good | good | good | good |
| Measuring Stations with Fish Breeding Boxes | Living Larvae (lL) | Dead Larvae (dL) | Dead Eggs (dE) | Eyed Eggs (eE) | Dissolved Eggs (Difference on 300) | Total Number of Added Eggs | Hatching Rate [(lL) + (dL)] | Survival Rate [(lL) + (eE)] |
|---|---|---|---|---|---|---|---|---|
| Station 1 | 134 | 38 | 21 | 9 | 98 | 300 | 57% | 48% |
| Station 2 | 254 | 1 | 13 | 13 | 19 | 300 | 85% | 85% |
| Station 3 | 0 | 23 | 234 | 0 | 43 | 300 | 8% | 0% |
| Station 4 | 238 | 11 | 26 | 0 | 25 | 300 | 83% | 79% |
| Station 5 | n/a | n/a | n/a | n/a | n/a | 300 | n/a | n/a |
| Station 6 | 218 | 7 | 24 | 0 | 51 | 300 | 75% | 73% |
| Station 1 | Station 2 | Station 3 | Station 4 | Station 5 * | Station 6 * | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Box 1 | Box 2 | Box 1 | Box 2 | Box 1 | Box 2 | Box 1 | Box 2 | Box 1 | Box 1 | |
| [mm] | {%] | {%] | {%] | {%] | {%] | {%] | {%] | {%] | {%] | {%] |
| 90.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| 63.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| 56.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| 31.50 | 12.74 | 16.17 | 20.87 | 21.08 | 21.84 | 18.86 | 16.34 | 15.35 | 18.93 | 11.52 |
| 22.40 | 28.31 | 27.64 | 29.73 | 31.82 | 25.45 | 23.08 | 41.60 | 36.86 | 27.47 | 27.02 |
| 16.00 | 29.24 | 28.09 | 25.06 | 26.95 | 27.56 | 28.90 | 23.45 | 32.44 | 26.26 | 31.77 |
| 11.20 | 12.79 | 7.54 | 8.80 | 7.92 | 7.49 | 9.15 | 8.80 | 7.82 | 8.53 | 8.01 |
| 8.00 | 3.18 | 1.74 | 1.48 | 1.19 | 0.78 | 1.53 | 1.73 | 0.70 | 1.93 | 1.42 |
| 4.00 | 3.84 | 2.48 | 2.00 | 0.37 | 0.15 | 0.56 | 0.38 | 0.19 | 3.05 | 2.63 |
| 2.00 | 8.15 | 13.99 | 7.99 | 6.35 | 5.90 | 4.55 | 2.01 | 1.69 | 10.21 | 14.01 |
| 1.00 | ||||||||||
| 0.50 | ||||||||||
| 0.25 | ||||||||||
| 0.125 | 1.76 | 2.35 | 4.07 | 4.33 | 10.81 | 13.36 | 5.69 | 4.95 | 3.61 | 3.62 |
| 0.063 | ||||||||||
| pan | ||||||||||
| u [i] | u [i] | χ² [i] | χ² [i] | ||
|---|---|---|---|---|---|
| Fish Breeding Boxes | Living Larvae | Dead Larvae | Living Larvae | Dead Larvae | |
| Station 1 | −2.29 | 2.68 | 5.27 | 7.19 | |
| Station 2 | 7.13 | −8.33 | 50.80 | 69.39 | |
| Station 3 | −13.16 | 15.38 | 173.20 | 236.58 | |
| Station 4 | 4.92 | −5.75 | 24.24 | 33.12 | |
| Station 5 | n/a | n/a | n/a | n/a | |
| Station 6 | 3.40 | −3.98 | 11.59 | 15.83 | |
| Total | 265.10 | 362.10 | χ² | ||
| 4 | 4 | df | |||
| 0.05 | 0.05 | p |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hauer, C.; Holzapfel, P.; Flödl, P.; Wagner, B.; Graf, W.; Leitner, P.; Haimann, M.; Holzer, G.; Haun, S.; Habersack, H.; et al. Controlled Reservoir Drawdown—Challenges for Sediment Management and Integrative Monitoring: An Austrian Case Study—Part B: Local Scale. Water 2020, 12, 1055. https://doi.org/10.3390/w12041055
Hauer C, Holzapfel P, Flödl P, Wagner B, Graf W, Leitner P, Haimann M, Holzer G, Haun S, Habersack H, et al. Controlled Reservoir Drawdown—Challenges for Sediment Management and Integrative Monitoring: An Austrian Case Study—Part B: Local Scale. Water. 2020; 12(4):1055. https://doi.org/10.3390/w12041055
Chicago/Turabian StyleHauer, Christoph, Patrick Holzapfel, Peter Flödl, Beatrice Wagner, Wolfram Graf, Patrick Leitner, Marlene Haimann, Georg Holzer, Stefan Haun, Helmut Habersack, and et al. 2020. "Controlled Reservoir Drawdown—Challenges for Sediment Management and Integrative Monitoring: An Austrian Case Study—Part B: Local Scale" Water 12, no. 4: 1055. https://doi.org/10.3390/w12041055
APA StyleHauer, C., Holzapfel, P., Flödl, P., Wagner, B., Graf, W., Leitner, P., Haimann, M., Holzer, G., Haun, S., Habersack, H., & Schletterer, M. (2020). Controlled Reservoir Drawdown—Challenges for Sediment Management and Integrative Monitoring: An Austrian Case Study—Part B: Local Scale. Water, 12(4), 1055. https://doi.org/10.3390/w12041055








