Identification of Aquifer Recharge Sources as the Origin of Emerging Contaminants in Intensive Agricultural Areas. La Plana de Castellón, Spain
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
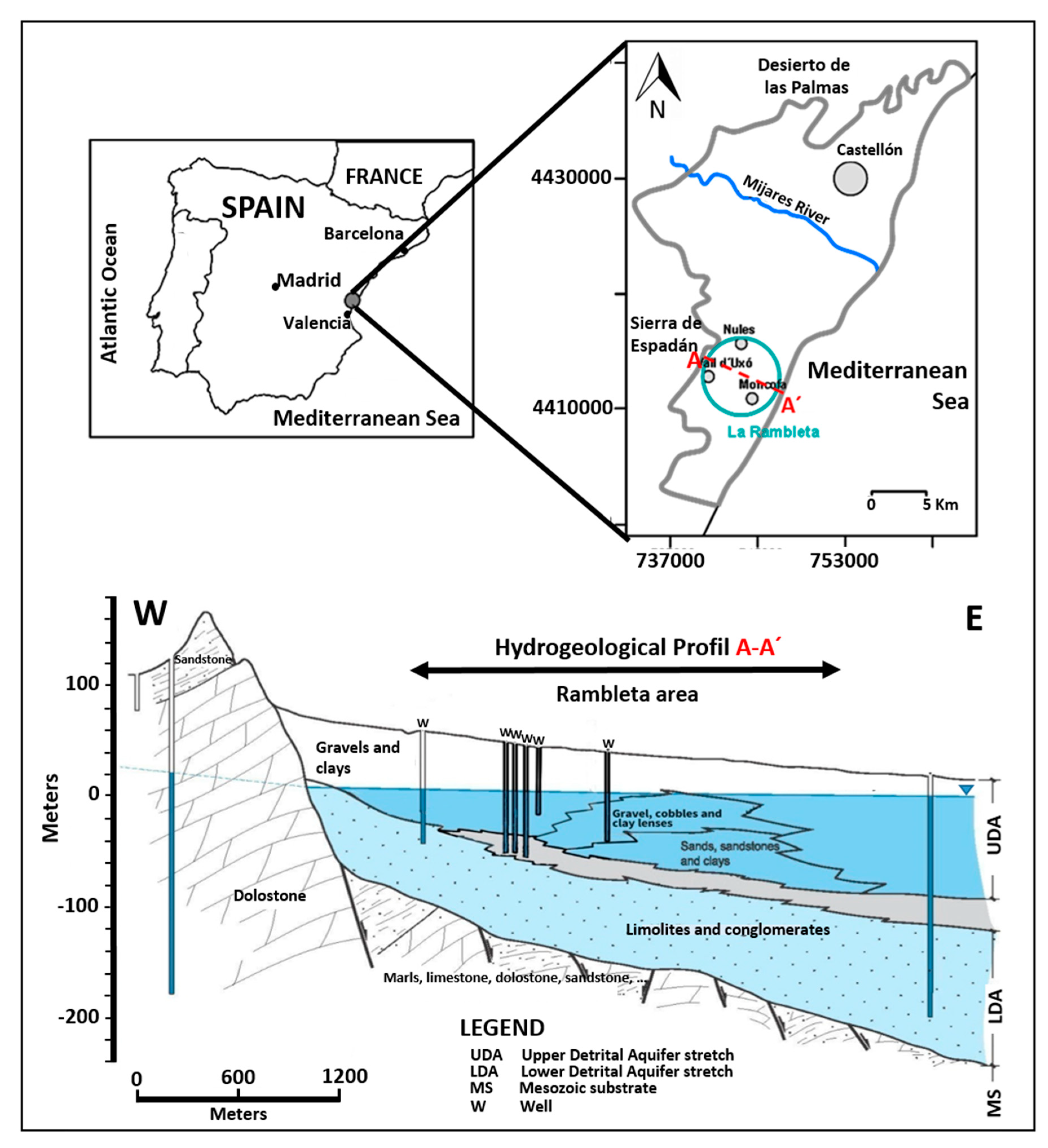
2.2. Hydrogeological Setting
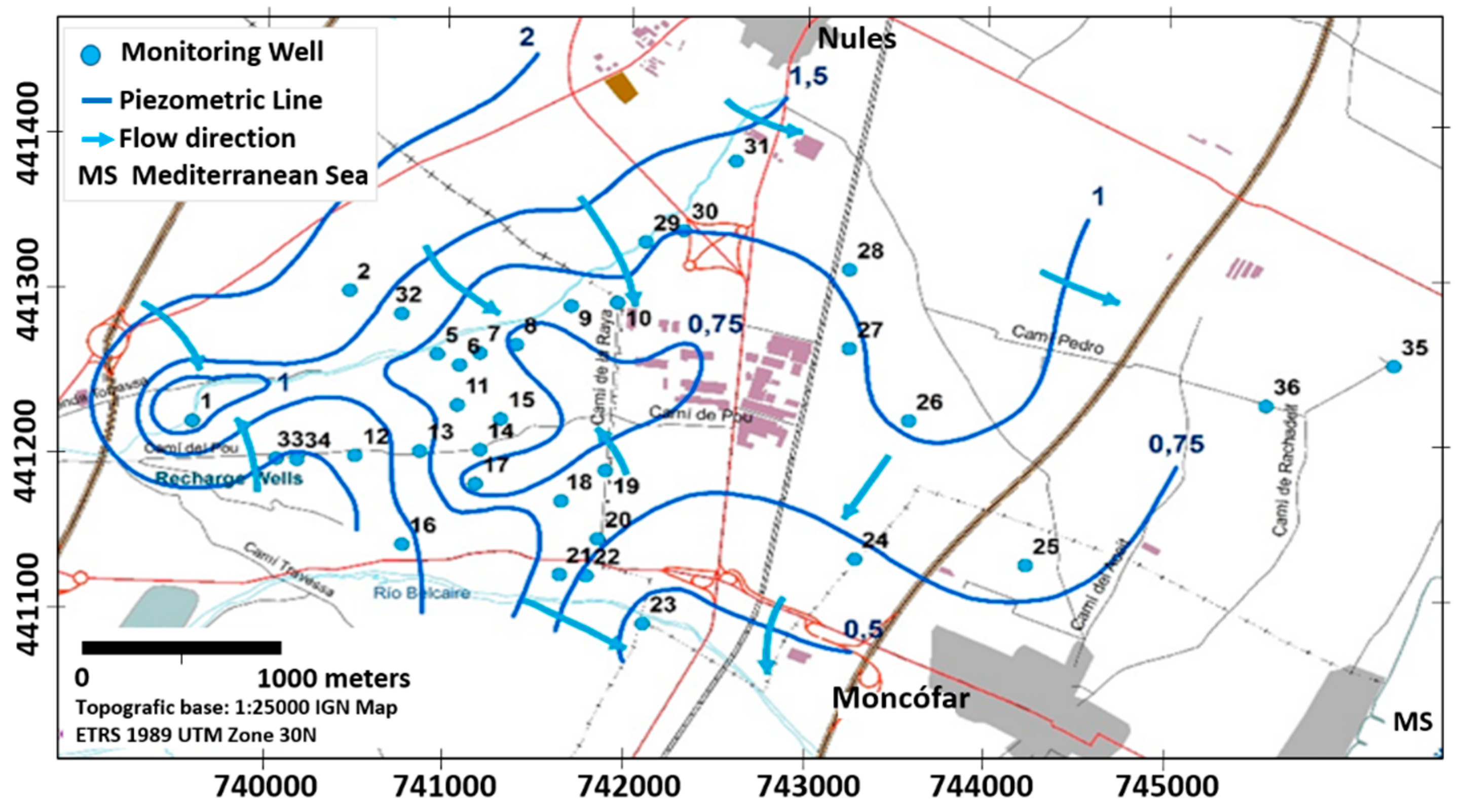
2.3. Monitoring Network and Sampling
2.4. Characteristics of the Wastewater Treatment Plant (WWTP) and Freshwater Dam (FW-D)
2.4.1. Wastewater Treatment Plant (WWTP)
2.4.2. Fresh Water Dam (FW-D)
2.5. Analytical Methods
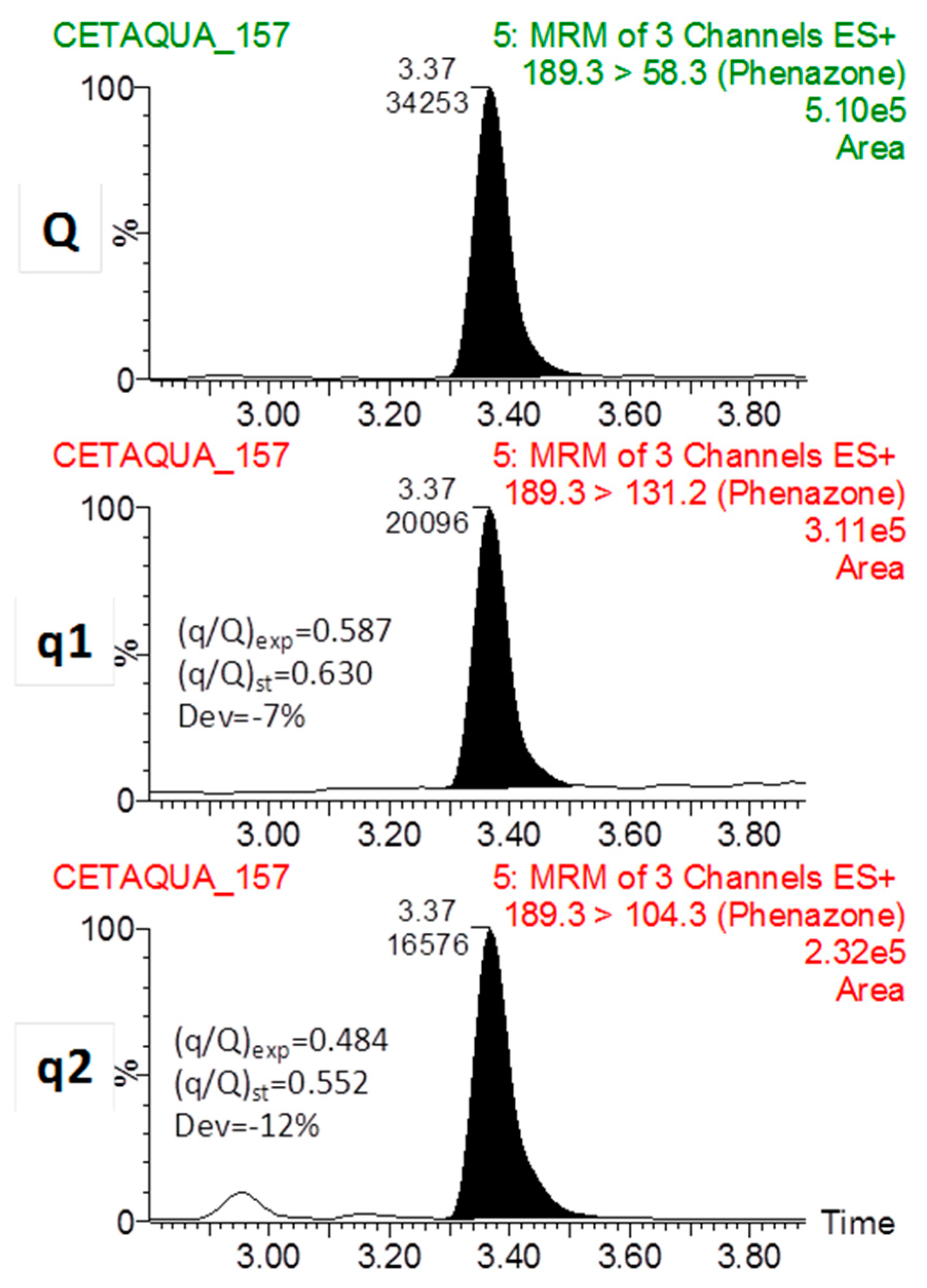
2.5.1. Instrumentation
2.5.2. Selection of Analytes
2.5.3. Sample Preparation
2.5.4. UHPLC-MS/MS Method
3. Results and Discussion
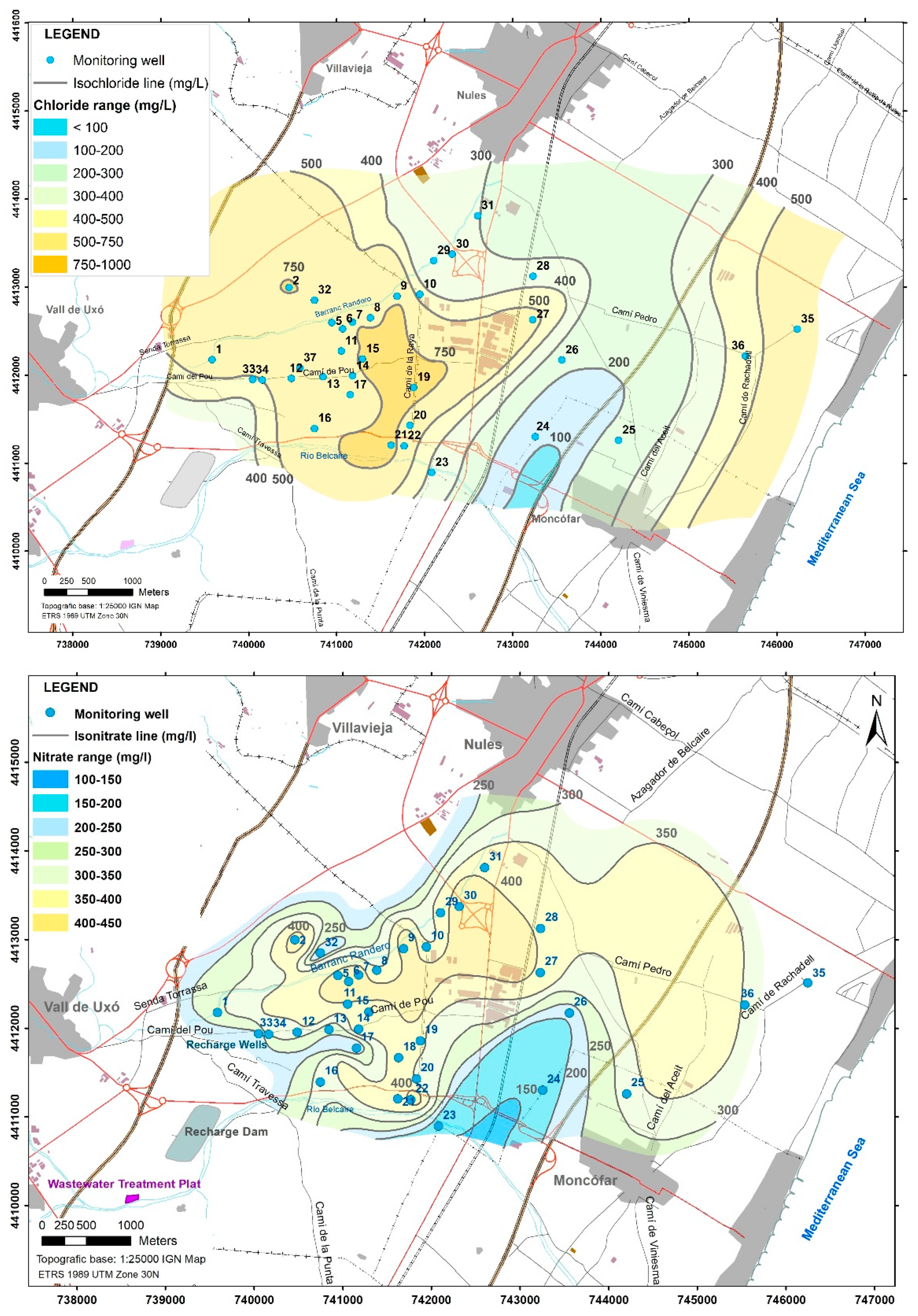
3.1. Assessment of the Occurrence, Spatial Distribution, and Concentration of Selected Typical ECs in Groundwater in a Common Agricultural Context
3.2. Identification of the Recharge (Pollution) Sources as an Origin of the ECs
3.3. ECs Suggested to be Indicators of Reclaimed Water Arrival in Detrital Heterogeneous Aquifers through the Vadose Zone
4. Conclusions
4.1. To Assess the Occurrence, Spatial Distribution, and Concentration of Selected Typical ECs in Groundwater in a Common Agricultural Context
4.2. Identification of the Recharge (Pollution) Sources as an Origin of ECs
4.3. Suggested Coes as Indicators of Reclaimed Water Arrival in Detrital Heterogeneous Aquifer
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barceló, D.; López de Alda, M.J. Contaminación Y Calidad Química Del Agua: El Problema de Los Contaminantes Emergentes. Instituto de Investigaciones Químicas y Ambientales-CSIC (Barcelona). 2008. Available online: https://fnca.eu/phocadownload/P.CIENTIFICO/inf_contaminacion.pdf (accessed on 1 August 2019).
- Daughton, C.G. Non-regulated water contaminants: Emerging research. Environ. Impact Assess. Rev. 2004, 24, 711–732. [Google Scholar] [CrossRef]
- Meffe, R.; de Bustamente, I. Emerging organic contaminants in surface water and groundwater: A first overview of the situation in Italy. Sci. Total Environ. 2014, 481, 280–295. [Google Scholar] [CrossRef]
- Pal, A.; Gin, K.Y.H.; Lin, A.Y.C.; Reinhard, M. Impacts of emerging organic contaminants on freshwater resources: Review of recent occurrences, sources, fate and effects. Sci. Total Environ. 2010, 408, 6062–6069. [Google Scholar] [CrossRef] [PubMed]
- Schriks, M.; Heringa, M.B.; van der Kooi, M.M.; de Voogt, P.; van Wezel, A.P. Toxicological relevance of emerging contaminants for drinking water quality. Water Res. 2010, 44, 461–476. [Google Scholar] [CrossRef] [PubMed]
- Tubau, I.; Vázquez-Suñé, E.; Carrera, J.; González, S.; Petrovic, M.; López de Alda, M.; Barceló, D. Occurrence and fate of alkylphenol polyethoxylate degradation products and linear alkylbenzene sulfonate surfactants in urban groundwater. Barcelona case study. J. Hydrol. 2010, 383, 102–110. [Google Scholar] [CrossRef]
- Hughes, S.R.; Kay, P.; Brown, L.E. Global synthesis and critical evaluation of pharmaceutical data sets collected from river systems. Environ. Sci. Technol. 2012, 47, 661–677. [Google Scholar] [CrossRef] [PubMed]
- Jurado, A.; Mastroianni, N.; Vazquez-Suñe, E.; Carrera, J.; Tubau, I.; Pujades, E.; Postigo, C.; Lopez de Alda, M.; Barceló, D. Drugs of abuse in urban groundwater. A case study: Barcelona. Sci. Total Environ. 2012, 424, 280–288. [Google Scholar] [CrossRef]
- Postigo, C.; Barceló, D. Synthetic organic compounds and their transformation products in groundwater: Occurrence, fate and mitigation. Sci. Total Environ. 2014. [Google Scholar] [CrossRef]
- Sorensen, J.P.R.; Lapworth, D.J.; Nkhuwa, D.C.W.; Stuart, M.E.; Gooddy, D.C.; Bell, R.A.; Chirwa, M.; Kabika, J.; Liemisa, M.; Chibesa, M.; et al. Emerging contaminants in urban groundwater sources in Africa. Water Res. 2015, 72, 51–56. [Google Scholar] [CrossRef]
- Gracia-Lor, E.; Sancho, J.V.; Hernández, F. Multi-class determination of around 50 pharmaceuticals, including 26 antibiotics, in environmental and wastewater samples by ultra-high performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2011, 1218, 2264–2275. [Google Scholar] [CrossRef]
- Bijlsma, L.; Beltrán, E.; Boix, C.; Sancho, J.V.; Hernández, F. Improvements in analytical methodology for the determination of frequently consumed illicit drugs in urban wastewater. Anal. Bioanal. Chem. 2014, 406, 4261–4272. [Google Scholar] [CrossRef] [PubMed]
- Hernández, F.; Ibáñez, M.; Portolés, T.; Cervera, I. Advancing towards universal screening for organic pollutants in Waters. J. Hazard. Mater. 2015, 282, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. Multiresidue methods for the analysis of pharmaceuticals, personal care products and illicit drugs in surface water and wastewater by solid-phase extraction and ultra performance liquid chromatography-electrospray tandem mass spectrometry. Anal. Bioanal. Chem. 2008, 391, 1293–1308. [Google Scholar] [CrossRef] [PubMed]
- Gracia-Lor, E.; Sancho, J.V.; Serrano, R.; Hernández, F. Occurrence and removal of pharmaceuticals in wastewater treatment plants at the Spanish Mediterranean area of Valencia. Chemosphere 2012, 87, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Gros, M.; Rodríguez-Mozaz, S.; Barcelo, D. Fast and comprehensive multi-residue analysis of a broad range of human and veterinary pharmaceuticals and some of their metabolites in surface and treated waters by ultra-high-performance liquid chromatography coupled to quadrupole-lineas ion trap tandem MS. J. Chromatogr. A 2012, 1248, 104–121. [Google Scholar] [PubMed]
- Loos, R.; Carvalho, R.; António, D.C.; Comero, S.; Locoro, G.; Tavazzi, S.; PAracchini, B.; Chiani, M.; Lettieri, T.; Blaha, L.; et al. EU-wide monitoring survey on emerging polar organic contaminants in wastewater treatment plant effluents. Water Res. 2013, 47, 6475–6487. [Google Scholar] [CrossRef]
- López-Serna, R.; Jurado, A.; Vàzquez-Suñé, E.; Carrera, J.; Petrovi, M.; Barceló, D. Occurrence of 95 pharmaceuticals and transformation products in urban groundwaters underlying the metropolis of Barcelona, Spain. Environ. Pollut. 2013, 174, 305–315. [Google Scholar] [CrossRef]
- López-Serna, R.; Kasprzyk-Hordern, B.; Petrovic, M.; Barceló, D. Multi-Residue enantiomeric analysis of pharmaceuticals and their active metabolites in the Guadalquivir River basin (South Spain) by chiral liquid chromatography couple with tandem mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 5859–5873. [Google Scholar] [CrossRef]
- Subedi, B.; Codru, N.; Dziewulski, D.V.; Wilson, L.R.; Xue, J.; Tun, S.; Braun-Howland, E.; Minihane, C.; Kannan, K. A pilot study on the assessment of trace organic contaminants including pharmaceuticals and personal care products from on-site wastewater treatment system along Skaneateles Lake in New York State, USA. Water Res. 2015, 72, 28–39. [Google Scholar] [CrossRef]
- Musolff, A.; Leschik, S.; Möder, M.; Strauch, G.; Reinstorf, F.; Schirmer, M. Temporal and spatial patterns of micropollutants in urban receiving waters. Environ. Pollut. 2009, 157, 3069–3077. [Google Scholar] [CrossRef]
- Vulliet, E.; Cren-Olivé, C. Screening of pharmaceuticals and hormones at the regional scale, in surface and groundwaters intended to human consumption. Environ. Pollut. 2011, 159, 2929–2934. [Google Scholar] [CrossRef] [PubMed]
- Jurado, A.; Vàzquez-Suñé, E.; Carrera, J.; de Alda, M.L.; Pujades, E.; Barceló, D. Emerging contaminants in groundwater in Spain: A review of sources, recent occurrence and fate in an European context. Sci. Total Environ. 2012, 440, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Stuart, M.; Lapworth, D.; Crane, E.; Hart, A. Review of risk from potential emerging contaminants in UK groundwater. Sci. Total Environ. 2012, 416, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Jurado, A.; Gago-Ferrero, P.V.; Àzquez-Suñé, E.; Carrera, J.; Pujades, E.; Díaz-Cruz, M.S.; Barceló, D. Urban groundwater contamination by residues of UV filters. J. Hazard. Mater. 2014, 271, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Jurado, A.; López-Serna, R.; Vàzquez-Suñé, E.; Carrera, J.; Pujades, E.; Petrovic, M.; Barceló, D. Occurrence of carbamazepine and five metabolites in an urban aquifer. Chemosphere 2014, 115, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lai, W.W.; Tung, H.; Lin, A.Y. Occurrence of pharmaceuticals, hormones and perfluorinated compounds in groundwaters in Taiwan. Environ. Monit. Assess. 2015, 187, 256. [Google Scholar] [CrossRef]
- Pitarch, E.; Cervera, M.I.; Portolés, T.; Ibáñez, M.; Barreda, M.; Renau-Pruñonosa, A.; Morell, I.; López, F.; Albarrán, F.; Hernández, F. Comprehensive monitoring of organic micro-pollutants in surface and groundwater in the surroundings of a solid-waste treatment plant of Castellón. Sci. Total Environ. 2016. [Google Scholar] [CrossRef]
- Tubau, I.; Vazquez-Suñe, E.; Carrera, J.; Valhondo, C.; Criollo, R. Quantification of groundwater recharge in urban environments. Sci. Total Environ. 2017, 592, 391–402. [Google Scholar] [CrossRef]
- Jones-Lepp, T.L.; Alvarez, D.A.; Petty, J.D.; Huckins, J.N. Polar organic chemical integrative sampling and liquid chromatography-electrospray/ion-trap mass spectrometry for assessing selected prescription and illicit drugs in treated sewage effluents. Arch. Environ. Contam. Toxicol. 2004, 47, 427–439. [Google Scholar] [CrossRef]
- Loch, J.P.; Verdam, B. Pesticide residues in groundwater in Netherlands: State of observations and future directions of research. Schr. Ver. Wasser Bodem Lufthgiene 1989, 79, 349–363. [Google Scholar]
- Jayachandran, K.; Steinheimer, T.O.R.; Somasundaram, L.; Moonman, T.B.; Kanwar, R.S.; Coats, J.R. Occurrence of atrazine and degradates as contaminants of subsurface drainage and shallow groundwater. J. Environ. Qual. 1994, 23, 311–319. [Google Scholar] [CrossRef]
- Ren, J.; Jiang, K. Atrazine and its degradation products in surface and ground waters in Zhangjiakou District, China. Chin. Sci. Bull. 2002, 47, 1612–1616. [Google Scholar] [CrossRef]
- Baran, N.; Mouvet, C.; Negrel, O. Hydrodynamic and geochemical constraints on pesticide concentrations in the groundwater of an agricultural catchment (Brévillers, France). Environ. Pollut. 2007, 148, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Ma, J.; Jia, R.; Xue, L.; Tao, C.; Li, C.; Lin, Y. Impact of long-term atrazine use on groundwater safety in Jilin Province, China. J. Integr. Agric. 2013, 12, 305–313. [Google Scholar] [CrossRef]
- Directive 2006/118/EC of the European Parliament and the Council of 12th of December 2006 on the protection of Groundwater Against Pollution and Deterioration. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:372:0019:0031:EN:PDF (accessed on 19 June 2019).
- Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 Amending DIRECTIVES 2000/60/EC and 2008/105/EC as Regards Priority Substances in the Field of Water Policy. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2013:226:0001:0017:EN:PDF (accessed on 14 June 2019).
- Valencian Institute of Agricultural Research (IVIA 2016). Available online: http://riegos.ivia.es/ (accessed on 23 July 2019).
- Renau-Llorens, E.A. Elementos Minoritarios Y Traza En La Masa De Agua Subterránea 080.021 (Plana De Castelló). Origen Y Procesos Asociados. Ph.D. Thesis, Universitat Jaume I, Castellón de la Plana, Spain, 2010. [Google Scholar]
- García-Menéndez, O.; Morell, I.; Ballesteros, B.J.; Renau-Pruñonosa, A.; Renau-Llorens, A.; Esteller, M.V. Spatial characterization of the seawater upconing process in coastal Mediterranean aquifer (Plana de Castellón, Spain): Evolution and controls. Environ. Earth Sci. 2016. [Google Scholar] [CrossRef]
- Canerot, J. Recherches Géologiques Aux Confins Des Chaines Ibériques Et Catalanes; Univ Paul Sabatier: Toulouse, Espagne, 1974. [Google Scholar]
- Giménez, E. Caracterización Hidroquímica De La Intrusión Marina En Acuíferos Detríticos Costeros. Aplicación a Los Acuíferos de Oropesa, Castellón y Gandía (Provincias de Castellón Y Valencia). Licenciatura Thesis, Universidad de Zaragoza, Zaragoza, Spain, 1988. [Google Scholar]
- Giménez, E.; Morell, I.; Esteller, M.V. La Sobreexplotación en Relación Con el Incremento Del Cultivo de Regadío en la Provincia de Castellón; Temas Geológicos-Mineros; Dirección General de Obras Hidráulicas: Madrid, Spain, 1989; pp. 357–370. ISBN 84-7840-031-1. [Google Scholar]
- Morell, I.; Giménez, E.; Esteller, M.V. Ejemplo de Utilización de Registros Verticales de Conductividad, Temperatura, pH Y Oxigeno Disuelto. Aplicación a Los Acuíferos de Oropesa-Torreblanca y Moncofar (Castellón); Hidrogeología y Recursos Hidráulicos; Manuel Regueiro: Madrid, Spain, 1987; pp. 171–182. ISBN 84-7840-471-6. [Google Scholar]
- Fidelibus, M.D.; Giménez, E.; Morell, I.; Tulipano, L. Salinization processes in the Castellón plain aquifer. In Study and Modelling of Salt Water Intrusion into Aquifers, Proceedings of the 12th Saltwater Intrusión Meeting, Barcelona, Spain, 1992; Custodio, E., Galofré, A., Eds.; The 12th Saltwater Intrusión Meeting: Barcelona, Spain, 1992; pp. 267–283. Available online: http://www.swim-site.nl/pdf/swim12/swim12_267_283.pdf (accessed on 1 June 2019).
- Gómez, J.D.; López-Geta, J.A.; Garrido, E. The State of Seawater Intrusion in Spain. Coastal Aquifers Intrusion Technology; IGME: Madrid, Spain, 2003; ISBN 84-7840-470-8. Available online: http://www.igme.es/actividadesIGME/lineas/HidroyCA/publica/libros2_TH/tiac-02/ESPA%C3%91A-I.pdf (accessed on 8 June 2019).
- Giménez, E.; Morell, I.; Renau, A. Geoquímica isotópica del boro en el acuífero de la Plana de Castellón. In Proceedings of the VIII Simposio de Hidrogeología. Hidrogeología y Recursos Hidráulicos, Zaragoza, Spain, 6 March 2003; pp. 137–146. [Google Scholar]
- López Gutiérrez, J.; García-Menéndez, O.; Ballesteros, B.J. The origins of mercury found in the coastal aquifer of la Plana de Castellón (Eastern Spain). Boletín Geológico Y Min. 2010, 121, 279–298. [Google Scholar]
- Giménez-Forcada, E.; Vega, M. Arsenic, barium, strontium and uranium geochemistry and their utility as tracers to characterize groundwaters from the Espadán-Calderona Triassic Domain, Spain. Sci. Total Environ. 2015, 599–612. [Google Scholar] [CrossRef]
- García-Menéndez, O.; Ballesteros, B.J.; Renau-Pruñonosa, A.; Morell, I.; Mochales, T.; Ibarra, P.I.; Rubio, F.M. Using electrical resistivity tomography to assess the effectiveness of managed aquifer recharge in a salinized coastal aquifer. Environ. Monit. Assess. 2018. [Google Scholar] [CrossRef]
- Morell, I.; Giménez, E.; Esteller, M.V. Application of principal components analysis to the study of salinization of the Castellon Plain (Spain). Sci. Total Environ. 1996, 177, 161–171. [Google Scholar] [CrossRef]
- Instituto Nacional de Estadística (INE, 2019). Available online: www.ine.es (accessed on 5 September 2019).
- Entidad Pública de Saneamiento de Aguas Residuales de la Comunidad Valencia (EPSAR, 2019). Available online: www.epsar.gva.es (accessed on 27 August 2019).
- Demonstration of Promising Technologies to Address Emerging Pollutants in Water and Waste Water (DEMEAU, 2017). Available online: https://demeau-fp7.eu/ (accessed on 10 December 2017).
- Boix, C.; Ibáñez, M.; Sancho, J.V.; Rambla, J.; Aranda, J.L.; Ballester, S.; Hernández, F. Fast determination of 40 drugs in water using large volume direct injection liquid chromatography-tandem mass spectrometry. Talanta 2015, 131, 719–727. [Google Scholar] [CrossRef]
- Benijts, T.; Lambert, W.; De Leenheer, A. Analysis of multiple endocrine disruptors in environmental waters via wide-spectrum solid-phase extraction and dual-polarity ionization LC-Ion Trap-MS/MS. Anal. Chem. 2004, 76, 704–711. [Google Scholar] [CrossRef] [PubMed]
- SANTE. Analytical quality control and method validation procedures for pesticide. Residues Anal. 2017, 1–46. [Google Scholar] [CrossRef]
- Siemens, J.; Huschek, G.; Siebe, C.; Kaupenjohann, M. Concentrations and mobility of human pharmaceuticals in the world’s largest wastewater irrigation system, Mexico City-Mezquital Valley. Water Res. 2008, 32, 2124–2134. [Google Scholar] [CrossRef] [PubMed]
- Chefetz, B.; Mualem, T.; Ben-Ain, J. Sorption and mobility of pharmaceutical compounds in soil irrigated with reclaimed wastewater. Chemosphere 2008, 73, 1335–1343. [Google Scholar] [CrossRef]
- Sacher, F.; Lange, F.T.; Brauch, H.; Blankenhorn, I. Pharmaceuticals in groundwaters. Analytical methods and results of monitoring program in Baden-Wüttemberf, Germany. J. Chromatogr. A 2001, 938, 199–210. [Google Scholar] [CrossRef]
- Fram, M.S.; Belitz, K. Occurrence and concentrations of pharmaceutical compounds in groundwater used for public drinking-water supply in California. Sci. Total Environ. 2011, 409, 3409–3417. [Google Scholar] [CrossRef]
- Candela, L.; Tamoh, K.; Vasdillo, I.; Valdes-Abellan, J. Monitoring of selected pharmaceuticals over 3 years in a detrital aquifer during artificial groundwater recharge. Environ. Earth Sci. 2016, 75, 244. [Google Scholar] [CrossRef]
- Boix, C.; Ibáñez, M.; Fabregat-Safont, D.; Morales, E.; Pastor, L.; Sancho, J.V.; Sánchez-Ramírez, J.E.; Hernández, F. Behaviour of emerging contaminants in sewage sludge after anaerobic digestion. Chemosphere 2016, 163, 296–304. [Google Scholar] [CrossRef]
- Lam, M.; Young, C.J.; Brain, R.A.; Johnson, D.J.; Hnason, M.A.; Wilson, C.J.; Richards, S.M.; Solomon, K.R.; Mabury, S.A. Aquatic persistence of eight pharmaceuticals in a microcosm study. Environ. Toxicol. Chem. 2004, 23. [Google Scholar] [CrossRef]
- Petrie, B.; Baden, R.; Kasprzyk-Hordern, B. A review on emerging contaminants in wastewaters and the environment: Current knowledge, understudied areas and recommendations for future monitoring. Water Res. 2015, 75, 3–27. [Google Scholar] [CrossRef]
- Stankiewicz, A.; Giebultowicz, J.; Stankiewicz, U.; Wroczynkki, P.; Nalecz-Jawecki, G. Determination of selected cardiovascular active compounds in environmental aquatic samples-Methods and results, a review of global publications from the last 10 years. Chemosphere 2015, 138, 642–656. [Google Scholar] [CrossRef]
- Papageorgiou, M.; Kosma, C.; Lambropoulou, D. Seasonal occurrence, removal, mass loading and environmental risk assessment of 55 pharmaceuticals and personal care products in a municipal wastewater treatment plant in Central Greece. Sci. Total Environ. 2016, 543, 547–569. [Google Scholar] [CrossRef]
- Pereira, A.M.P.T.; Silva, L.J.C.; Lino, C.M.; Meisel, L.M.; Pena, A. Assessing environmental risk of pharmaceuticals in Portugal: An approach for the selection of Portuguese monitoring stations in line with directive 2013/39/EU. Chemosphere 2016, 144, 2507–2515. [Google Scholar] [CrossRef]
- Watababe, Y.; Bach, L.T.; Dinh, P.V.; Prudente, M.; Aguja, S.; Phay, N.; Nakata, H. Ubiquitous detection of artificial sweeteners and iodinated X-ray contrast media in aquifer environmental and wastewater treatment plant samples from Vietnam, the Philippines and Myanmar. Arch. Environ. Contam. Toxicol. 2016, 70, 671–681. [Google Scholar] [CrossRef]
- Zeitzschgmann, F.; Stützer, C.; Jekel, M. Granular activated carbon adsorption of organic micro-pollutants in drinking water and treated wastewater-Aligning breakthrough curves and capacities. Water Res. 2016, 92, 180–187. [Google Scholar] [CrossRef]
- Hernández, F.; Marin, J.; Pozo, O.J.; Sancho, J.V.; López, F.J.; Morell, I. Pesticide residues and transformation products in groundwater from a Spanish agricultural region of the Mediterranean Coast. Int. J. Environ. Anal. Chem. 2008, 88, 409–424. [Google Scholar] [CrossRef]
- Hansen, A.M.; Treviño-Quintanilla, L.G.; Máequez-Pacheco, H.; Villada-Canela, M.; González-Márquez, L.C.; Guillén-Garcés, R.A.; Hernández-Antonio, A. Atrazina: Un herbicida polémico. Rev. Int. Contam. Ambient. 2013, 29, 65–84. [Google Scholar]
- Graymore, M.; Stagnitti, F.; Allison, G. Impacts of atrazine in aquatic ecosystem. Environ. Int. 2001, 26, 483–495. [Google Scholar] [CrossRef]
- Nödler, K.; Hillebrand, O.; Idzik, K.; Strathmann, M.; Schiperski, F.; Zirlewagen, J.; Licha, T. Occurrence and fate of the angiotensina II receptor antagonista transformation product valsartan acid in the water cucle—A comparative study with selected?-blockers and the persistent anthropogenic wastewater indicators carbamazepine and acesulfame. Water Res. 2013, 47, 6650–6659. [Google Scholar] [CrossRef]
- Tappe, W.; Groeneweg, J.; Jantsch, B. Diffuse atrazine pollution in German aquifers. Biodegradation 2002, 13, 3–10. [Google Scholar] [CrossRef]
- Tajla, K.M.; Kaukonen, S.; Kilpi-Koski, J.; Malin, I.; Kairesalo, T.; Romantschuk, M.; Tuominen, J.; Kontro, M.H. Atrazine and terbutryn degradation in deposits from groundwater environment with the Boreal region in Lathi, Finland. J. Agric. Food Chem. 2008, 56, 11962–11968. [Google Scholar]
- Thurman, E.M.; Meyer, M.T.; Mills, M.S.; Zimmermann, L.R.; Perry, C.A. Formation and transport of deethylatrazine and deisopropylatrazine in surface water. Environ. Sci. Technol. 1994, 28, 2267–2277. [Google Scholar] [CrossRef]
- Worrall, F.; Koplin, D.W. Aquifer vulnerability to pesticide pollution-Combining soil, land-use and aquifer properties with molecular descriptors. J. Hydrol. 2004, 293, 191–204. [Google Scholar] [CrossRef]
- Hernández, F.; Bakker, J.; Bijlsma, L.; de Boer, J.; Botero-Coy, A.M.; de Bruin, Y.B.; Fischer, S.; Hollender, J.; Kasprzyk-Hordern, B.; Lamoree, M.; et al. The role of analytical chemistry in exposure science: Focus on the aquatic environment. Chemosphere 2019, 222, 564–583. [Google Scholar] [CrossRef]
- Lesser, L.E.; Mora, A.; Moreau, C.; Mahlkbecht, J.; Hernández-ANtoio, A.; Ramírez, A.I.; Barrios-Piña, H. Survey of 218 organic contaminants in groundwater derived from theworld’s largest untreated wastewater irrigation system: Mezquital Valley, Mexico. Chemosphere 2018, 198, 510–521. [Google Scholar] [CrossRef]
- Drewes, J.E.; Heberer, T.; Reddersen, K. Fate of pharmaceuticals during indirect potable reuse. Water Sci. Technol. 2002, 46, 73–80. [Google Scholar] [CrossRef]



| Code | Type of Water | Total Depth (m) | Coordinate (X) ETRS 89 | Coordinate (Y) ETRS 89 | Observations |
|---|---|---|---|---|---|
| EWW | WWTP Effluent | - | 738,588 | 4,410,027 | 2 samples per survey (1 in weekend, 1 during the week) |
| FW-D | FW-D Effluent | - | 739,123 | 4,410,796 | Fresh water dam |
| GW-1 | Groundwater | 51 | 741,869 | 4,411,851 | Agricultural well. Abandoned |
| GW-2 | Groundwater | 100 | 739,579 | 4,412,167 | Agricultural well |
| GW-3 | Groundwater | 42 | 741,058 | 4,412,523 | Agricultural well |
| GW-4 | Groundwater | 100 | 740,485 | 4,411,955 | Agricultural well |
| GW-5 | Groundwater | NA | 740,583 | 4,412,081 | Piezometer. Abandoned |
| GW-6 | Groundwater | 100 | 740,054 | 4,412,034 | Piezometer |
| GW-7 | Groundwater | 100 | 740,235 | 4,411,950 | Piezometer |
| EWW | FW-D | |||
|---|---|---|---|---|
| Physicochemical Properties | April 2013 | April 2014 | April 2015 | April 2013 |
| CE (μS/cm) | 1145 | 1947 | 1023 | 311 |
| Na+ (mg/L) | 130 | 180 | 111 | 9 |
| K+ (mg/L) | 20 | 27 | 16 | 2 |
| Ca2+ (mg/L) | 85 | 101 | 75 | 32 |
| Mg2+ (mg/L) | 35 | 47 | 28 | 14 |
| Cl− (mg/L) | 118 | 180 | 107 | 14 |
| SO42− (mg/L) | 188 | 225 | 106 | 34 |
| HCO3− (mg/L) | 279 | 504 | 314 | 115 |
| CO32− (mg/L) | 0 | 0 | 0 | - |
| NO32− (mg/L) | 130 | 0 | 64 | 5 |
| NO2− (mg/L) | <0.05 | 0 | 0 | 0.66 |
| NH4+ (mg/L) | <0.05 | 0 | 0 | <0.05 |
| PO43− (mg/L) | <0.05 | 0 | 10.20 | |
| SiO2− (mg/L) | 11.1 | 12.7 | 10.6 | - |
| MnO4K oxidability (mg/L) | 29.7 | 19.9 | 23.2 | - |
| pH | 7.05 | 7.66 | 7.27 | 8.00 |
| Compounds | Concentration (ng/L) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EWW | GW | |||||||||||
| LOQ | (A-B) 1* | (A-B) 2* | LOQ | FW-D | GW-1 (1*2*) | GW-2 (1*2*) | GW-3 (1*2*) | GW-4 (1*2*) | GW-5 (1*2*) | GW-6 (1*2*) | GW-7 (1*2*) | |
| Pharmaceuticals | ||||||||||||
| Acetaminophen | 1.5 | 106–44 | 42–36 | 1.1 | <LOQ–10 | 4.8–<LOQ | 6.5–(–) | < LOQ | < LOQ–(–) | 2–< LOQ | 3.3–1.7 | 63–< LOQ |
| Bezafibrate | 2.1 | 1219–780 | 1095–1270 | 1.3 | 11–12 | 8.1–7.3 | 7–4.9 | 4.4–5 | 4.7–7.6 | 10–8.4 | 7.6–< LOQ | 5–2.5 |
| Carbamazepine | 1.1 | 84–97 | 73–86 | 0.2 | < LOQ | 0.5–1.2 | < LOQ | < LOQ | 1.2–1.5 | 0.9–1.3 | 1.1–1.9 | 1.2–1.9 |
| Diclofenac | 7.2 | 538–408 | 769–802 | 3 | – | 3.6–(–) | 3.1–(–) | 4.6–(–) | - | - | - | - |
| Gemfibrozil | 1.8 | 327–359 | 416–322 | 2.3 | 96–(–) | - | - | - | - | - | - | - |
| Irbesartan | 1 | 1547–1123 | 1147–1246 | 0.2 | < LOQ | < LOQ–0.3 | < LOQ | < LOQ | < LOQ–0.3 | < LOQ–0.3 | < LOQ–0.3 | < LOQ–0.3 |
| Metoprolol | 2.6 | 17–12 | 30–21 | 0.6 | - | 2.7–(–) | < LOQ–(–) | - | - | < LOQ–(–) | - | - |
| Pantoprazole | 0.8 | 7.9–4.9 | 6.8–8.5 | 0.1 | - | < LOQ | - | (–)–< LOQ | - | - | - | - |
| Phenazone | 8.2 | 612–145 | 563–607 | 2.1 | - | - | (–)–< LOQ | (–)–< LOQ | (–)–< LOQ | - | (–)–<LOQ | 4.2–(–) |
| Primidone | 4 | 151–57 | 37–28 | 1.1 | < LOQ | (–)–< LOQ | < LOQ–2.9 | < LOQ–– | 7.2–7.5 | 3.8–5.3 | 2.0–3.6 | 3.5–7.1 |
| Sulfamethoxazole | 0.8 | 140–115 | 153–158 | 0.5 | < LOQ | 4.1–< LOQ | < LOQ–4.4 | (–)–< LOQ | 6.1–4.7 | 3.4–2.8 | < LOQ–4.8 | < LOQ–3.6 |
| Trimethoprim | 2.3 | 103–81 | 145–102 | 1.8 | - | < LOQ–(–) | - | - | - | - | - | - |
| Valsartan | 4.2 | 23172–13509 | 23350–24283 | 3 | 9.2–18 | 3.1–< LOQ | < LOQ | - | - | - | - | 3.3–(–) |
| Venlafaxine | 1 | 962–619 | 831–801 | 0.2 | 1.1–< LOQ | 5.6–1.3 | 1.2–1.4 | < LOQ | < LOQ–1.2 | <LOQ–1.5 | < LOQ–1 | 5–1.2 |
| Contrast medium | ||||||||||||
| Iopromide | 21 | 1244–2362 | 2362–8616 | 7.6 | – | – | < LOQ | < LOQ–(–) | < LOQ | < LOQ–(–) | < LOQ | < LOQ–(–) |
| Drug of abuse | ||||||||||||
| Benzoylecgonine | 0.1 | 226–585 | 898–197 | 0.1 | - | 1.7–< LOQ | < LOQ–(–) | < LOQ | 0.2–< LOQ | 0.3–(–) | (–)–< LOQ | 0.9–0.3 |
| Pesticides | ||||||||||||
| Atrazine-desethyl (DEA) | 3.6 | 11–12 | - | 1.8 | - | 5–9 | 11–18 | 15–(–) | 19–19 | 11–11 | 9.2–9.0 | 14–18 |
| Atrazine-desisopropyl (DIA) | 3.4 | 16–19 | - | 1.7 | - | 5.2–6.1 | < LOQ–1.9 | 3.6–(–) | 4.5–4.1 | 4.0–4.6 | < LOQ–2 | < LOQ–2.9 |
| Simazine | 1.2 | 2.5–2.0 | 1.1–2.1 | 0.6 | - | 5.3–6.6 | (–)–1 | < LOQ–1.7 | < LOQ–18 | < LOQ–0.7 | 6.9–0.6 | 0.6 |
| Terbuthylazine | 1 | 15–8.5 | 23–70 | 0.5 | - | 5.4–4.7 | < LOQ–2.4 | 1.6–4.0 | 1–< LOQ | 6.5–8.7 | 4.2–3.7 | 0.5–1 |
| Compounds | Media EWW (ng/L) | Media GW (ng/L) | Degradation | Positive | Positive (%) | Quantified | Quantified (%) | Media EWW <150 (ng/L) | Positive (%) GW >85% | Degradation Compound <99% |
|---|---|---|---|---|---|---|---|---|---|---|
| Pharmaceuticals | ||||||||||
| Acetaminophen | 57 | 3.7 | 93.6 | 13 | 93 | 6 | 43 | x | x | x |
| Bezafibrate | 1091 | 5.5 | 99.9 | 14 | 100 | 13 | 114 | x | x | |
| Carbamazepine | 85 | 1.3 | 98.5 | 14 | 100 | 10 | 71 | x | x | x |
| Diclofenac | 629 | - | 100 | 3 | 21 | 3 | 21 | |||
| Gemfibrozil | 356 | - | 100 | 0 | 0 | 0 | 0 | x | ||
| Irbesartan | 1265 | 0.3 | 100 | 14 | 100 | 5 | 29 | x | ||
| Metoprolol | 20 | 2.7 | 86.5 | 3 | 21 | 1 | 7 | x | x | |
| Pantoprazole | 7 | - | 100 | 2 | 14 | 0 | 0 | x | ||
| Phenazone | 481 | 4.2 | 99.1 | 5 | 36 | 1 | 7 | x | ||
| Primidone | 68 | 4.8 | 93 | 12 | 86 | 8 | 57 | x | x | x |
| Sulfamethoxazole | 132 | 4.2 | 96.8 | 13 | 93 | 8 | 57 | x | x | x |
| Trimethoprim | 107 | - | 100 | 1 | 7 | 0 | 0 | x | ||
| Valsartan | 21078 | - | 100 | 5 | 36 | 2 | 14 | |||
| Venlafaxine | 803 | 2.2 | 99.7 | 14 | 100 | 9 | 64 | x | ||
| Contrast media | ||||||||||
| Iopromide | 4739 | - | 100 | 10 | 71 | 0 | 0 | |||
| Drugs of abuse | ||||||||||
| Benzoylecgonine | 476 | 0.7 | 99.8 | 11 | 79 | 5 | 35.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renau-Pruñonosa, A.; García-Menéndez, O.; Ibáñez, M.; Vázquez-Suñé, E.; Boix, C.; Ballesteros, B.B.; Hernández García, M.; Morell, I.; Hernández, F. Identification of Aquifer Recharge Sources as the Origin of Emerging Contaminants in Intensive Agricultural Areas. La Plana de Castellón, Spain. Water 2020, 12, 731. https://doi.org/10.3390/w12030731
Renau-Pruñonosa A, García-Menéndez O, Ibáñez M, Vázquez-Suñé E, Boix C, Ballesteros BB, Hernández García M, Morell I, Hernández F. Identification of Aquifer Recharge Sources as the Origin of Emerging Contaminants in Intensive Agricultural Areas. La Plana de Castellón, Spain. Water. 2020; 12(3):731. https://doi.org/10.3390/w12030731
Chicago/Turabian StyleRenau-Pruñonosa, Arianna, Olga García-Menéndez, María Ibáñez, Enric Vázquez-Suñé, Clara Boix, Bruno B. Ballesteros, Marta Hernández García, Ignacio Morell, and Félix Hernández. 2020. "Identification of Aquifer Recharge Sources as the Origin of Emerging Contaminants in Intensive Agricultural Areas. La Plana de Castellón, Spain" Water 12, no. 3: 731. https://doi.org/10.3390/w12030731
APA StyleRenau-Pruñonosa, A., García-Menéndez, O., Ibáñez, M., Vázquez-Suñé, E., Boix, C., Ballesteros, B. B., Hernández García, M., Morell, I., & Hernández, F. (2020). Identification of Aquifer Recharge Sources as the Origin of Emerging Contaminants in Intensive Agricultural Areas. La Plana de Castellón, Spain. Water, 12(3), 731. https://doi.org/10.3390/w12030731





