Effects of Irrigation with Desalinated Seawater and Hydroponic System on Tomato Quality
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Irrigation Treatments
2.2. Physicochemical Quality of Tomatoes
2.2.1. Dry Matter Content, Soluble Solids, pH, and Titratable Acidity
2.2.2. Firmness
2.2.3. Color
2.3. Nutritional and Bioactive Quality of Tomatoes
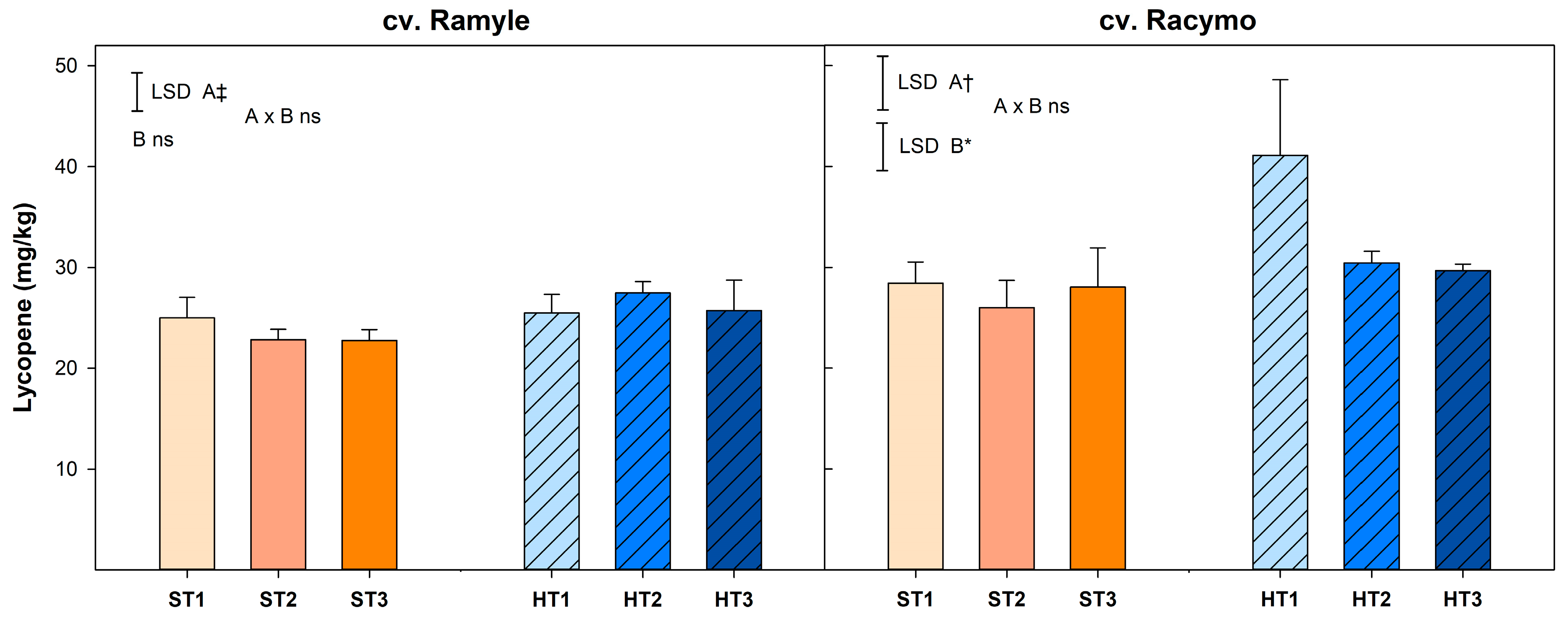
2.3.1. Lycopene
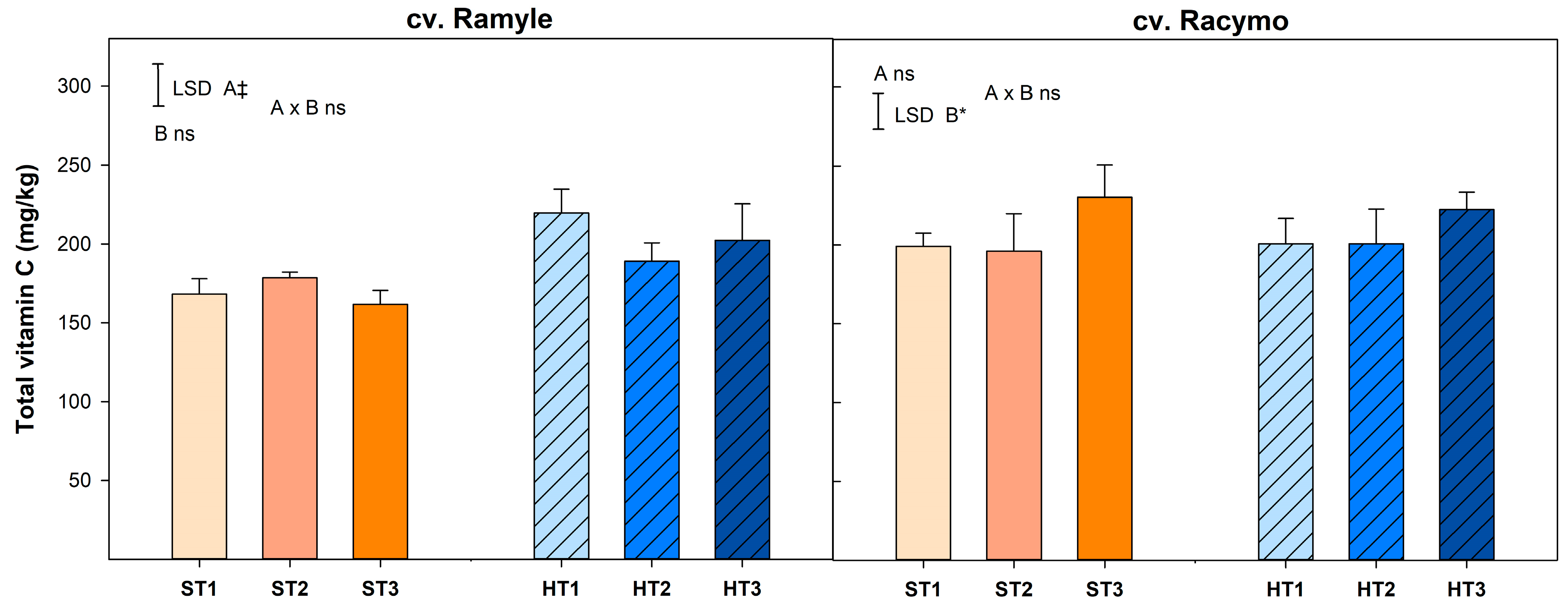
2.3.2. Total Vitamin C
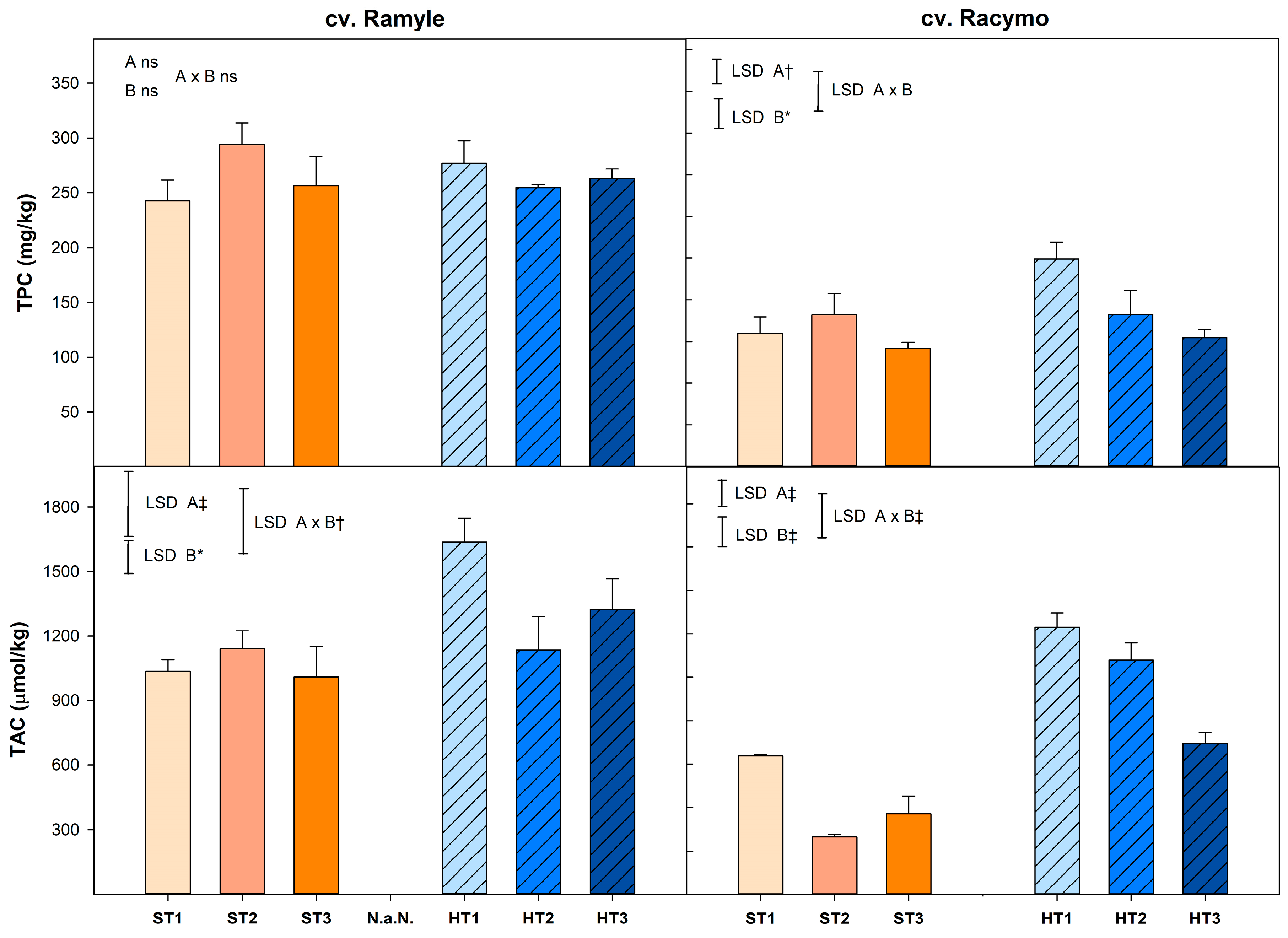
2.3.3. Total Phenolic Content
2.3.4. Total Antioxidant Capacity
2.4. Statistical Analyses
3. Results and Discussion
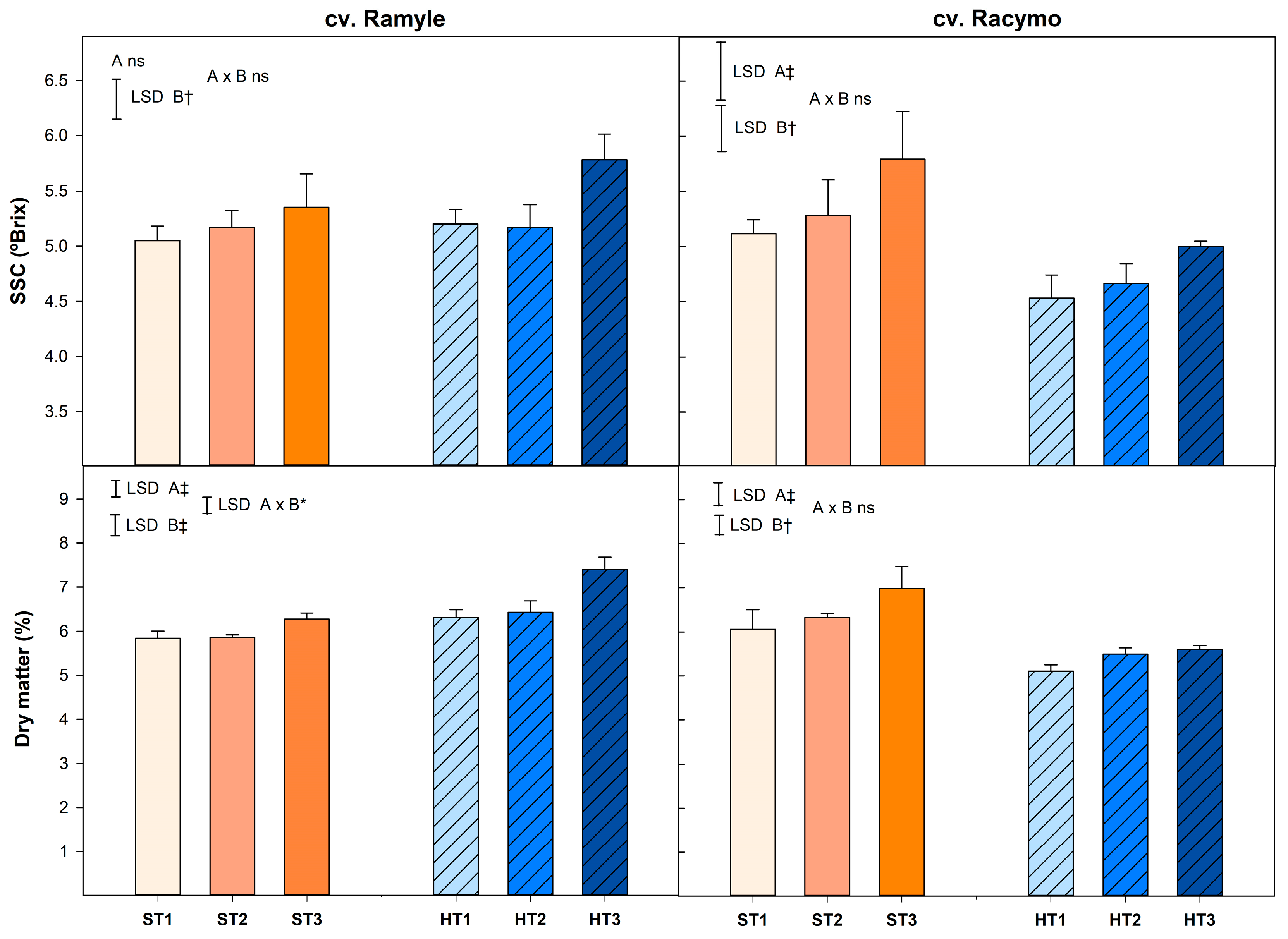
3.1. Soluble Solids Content and Dry Matter
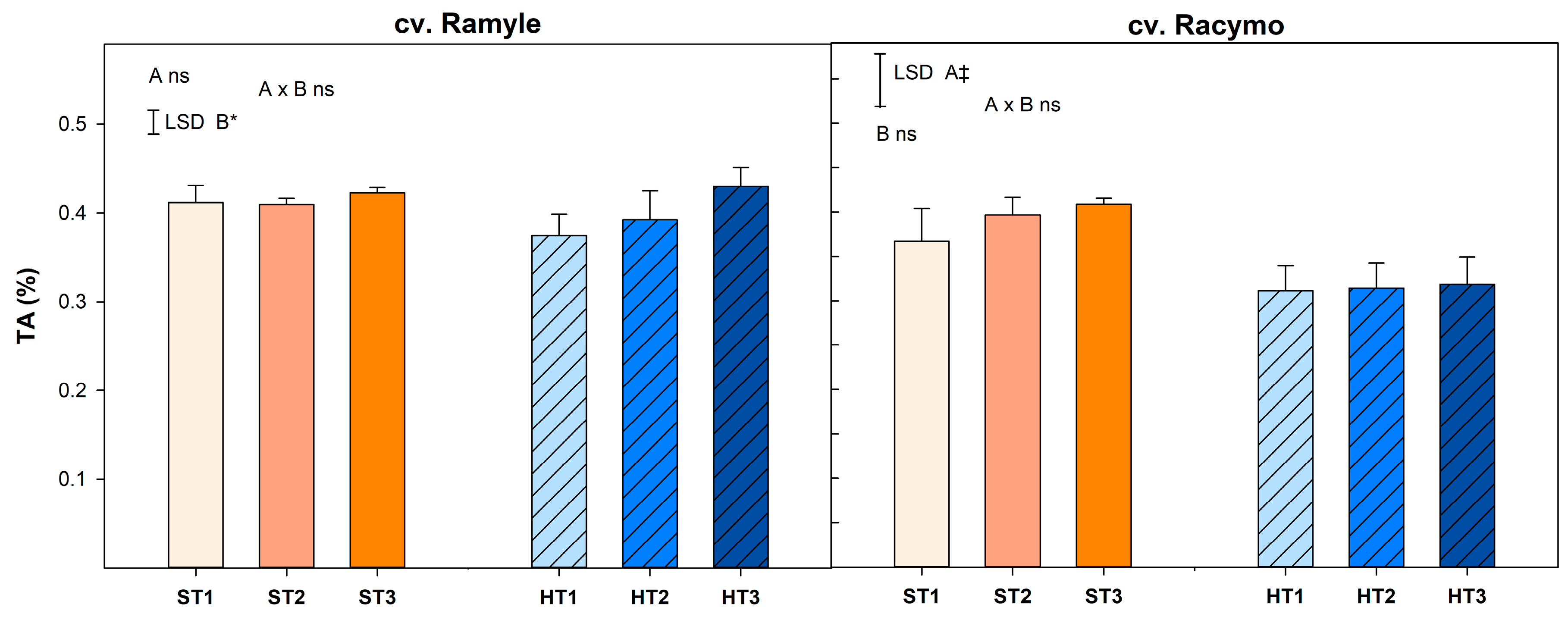
3.2. Titratable Acidity
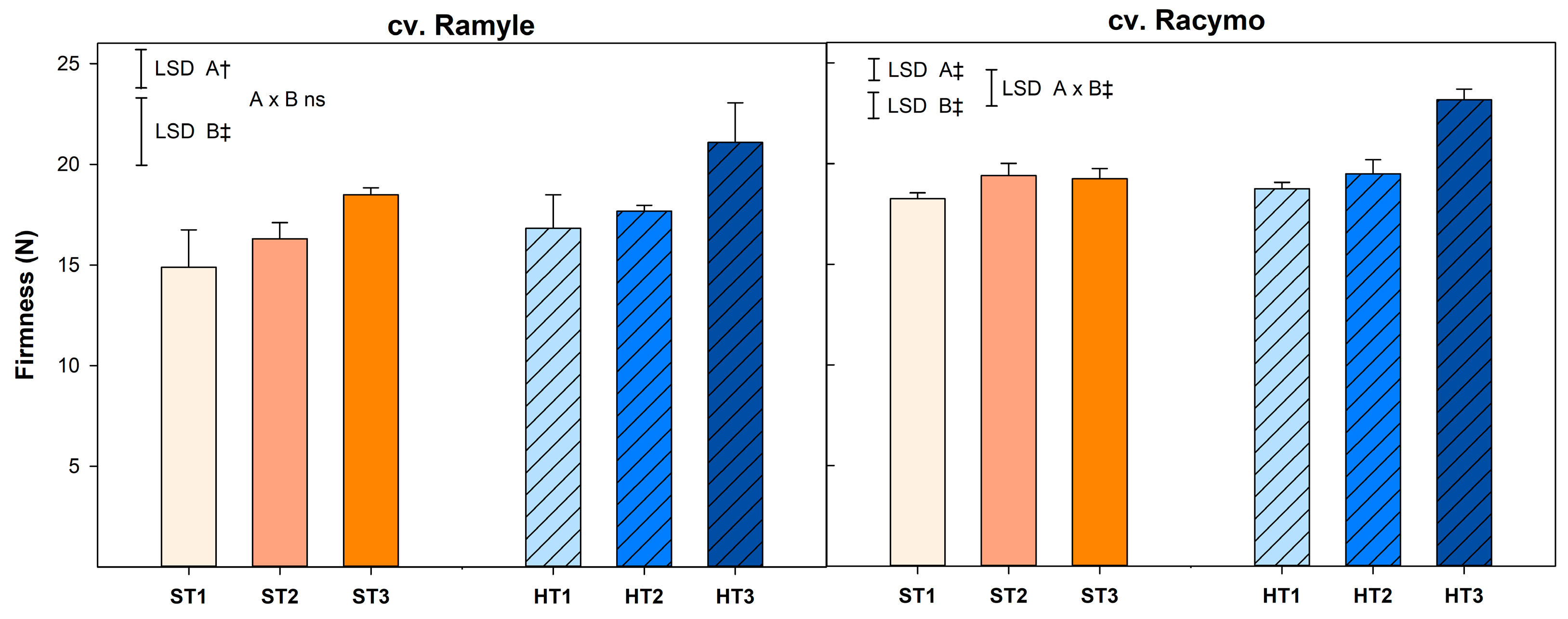
3.3. Firmness
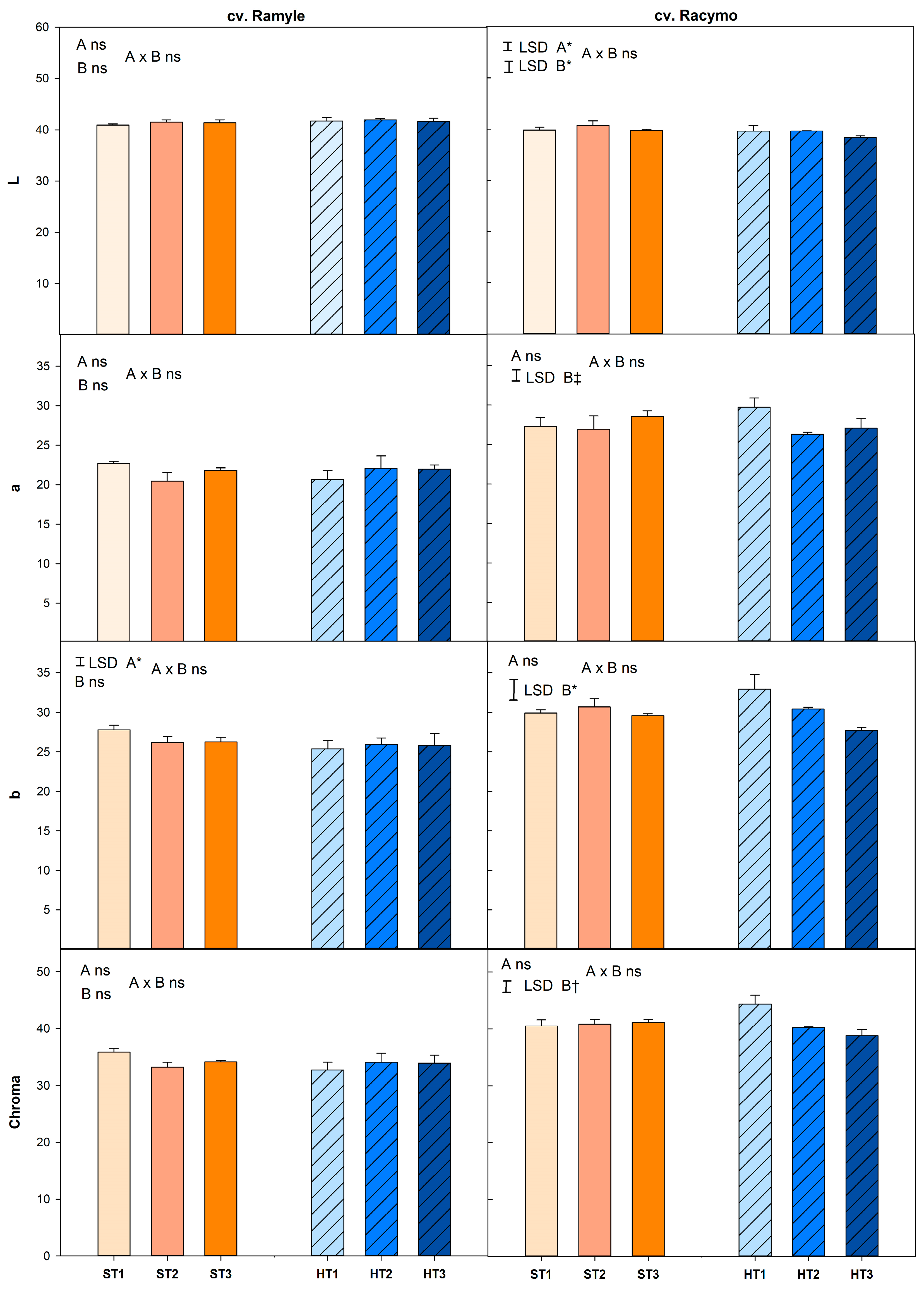
3.4. Color and Lycopene Contents
3.5. Total Vitamin C
3.6. Total Phenolic Content
3.7. Total Antioxidant Capacity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Martí, R.; Roselló, S.; Cebolla-Cornejo, J. Tomato as a source of carotenoids and polyphenols targeted to cancer prevention. Cancers (Basel) 2016, 8, 58. [Google Scholar] [CrossRef]
- FAOSTAT. Available online: http://www.fao.org/faostat/en/#data (accessed on 17 October 2018).
- Burton-Freeman, B.; Reimers, K. Tomato consumption and health: Emerging benefits. Am. J. Lifestyle Med. 2011, 5, 182–191. [Google Scholar] [CrossRef]
- Pagliarini, E.; Monteleone, E.; Ratti, S. Sensory profile of eight tomato cultivars (Lycopersicon Esculentum) and its relationship to consumer preference. Ital. J. Food Sci. 2001, 13, 285–296. [Google Scholar]
- Luthria, D.L.; Mukhopadhyay, S.; Krizek, D.T. Content of total phenolics and phenolic acids in tomato (Lycopersicon esculentum Mill.) fruits as influenced by cultivar and solar UV radiation. J. Food Compos. Anal. 2006, 19, 771–777. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Boluda-Aguilar, M.; Taboada-Rodríguez, A.; Soto-Jover, S.; Marín-Iniesta, F.; López-Gómez, A. Processing, packaging, and storage of tomato products: Influence on the lycopene content. Food Eng. Rev. 2016, 8, 52–75. [Google Scholar] [CrossRef]
- Dorais, M.; Papadopoulos, A.P.; Gosselin, A. Greenhouse tomato fruit quality. In Horticultural Reviews; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 239–319. [Google Scholar]
- Montesano, F.; Parente, A.; Santamaria, P. Closed cycle subirrigation with low concentration nutrient solution can be used for soilless tomato production in saline conditions. Sci. Hortic. (Amst.) 2010, 124, 338–344. [Google Scholar] [CrossRef]
- CHS. Hydrological Plan of the Demarcation of Segura 2015/21. Annex 6 Operating System and Balances; Hydrographic Confederation of Segura: Murcia, Spain, 2015. [Google Scholar]
- Bates, B.; Kundzewicz, Z.W.; Wu, S.; Palutikof, J.P. Climate Change and Water. Technical Paper of the Intergovernmental Panel on Climate Change; IPCC Secretariat: Geneva, Switzerland, 2008. [Google Scholar]
- Aznar-Sánchez, J.; Belmonte-Ureña, L.; Valera, D. Perceptions and acceptance of desalinated seawater for irrigation: A case study in the Níjar district (Southeast Spain). Water 2017, 9, 408. [Google Scholar] [CrossRef]
- Martínez-Alvarez, V.; González-Ortega, M.J.; Martin-Gorriz, B.; Soto-García, M.; Maestre-Valero, J.F. The use of desalinated seawater for crop irrigation in the Segura River Basin (south-eastern Spain). Desalination 2017, 422, 153–164. [Google Scholar] [CrossRef]
- Martínez-Alvarez, V.; Maestre-Valero, J.F.; González-Ortega, M.J.; Gallego-Elvira, B.; Martin-Gorriz, B. Characterization of the agricultural supply of desalinated seawater in Southeastern Spain. Water 2019, 11, 1233. [Google Scholar] [CrossRef]
- Silber, A.; Israeli, Y.; Elingold, I.; Levi, M.; Levkovitch, I.; Russo, D.; Assouline, S. Irrigation with desalinated water: A step toward increasing water saving and crop yields. Water Resour. Res. 2015, 51, 450–464. [Google Scholar] [CrossRef]
- Martínez-Álvarez, V.; González-Ortega, M.J.; Martín-Gorriz, B.; Soto-García, M.; Maestre-Valero, J.F. Seawater desalination for crop irrigation-Current status and perspectives. In Emerging Technologies for Sustainable Desalination Handbook; Elsevier: Amsterdam, The Netherlands, 2018; pp. 461–492. ISBN 9780128167120. [Google Scholar]
- Fanasca, S.; Colla, G.; Maiani, G.; Venneria, E.; Rouphael, Y.; Azzini, E.; Saccardo, F. Changes in antioxidant content of tomato fruits in response to cultivar and nutrient solution composition. J. Agric. Food Chem. 2006, 54, 4319–4325. [Google Scholar] [CrossRef]
- Martínez-Alvarez, V.; Martin-Gorriz, B.; Soto-García, M. Seawater desalination for crop irrigation—A review of current experiences and revealed key issues. Desalination 2016, 381, 58–70. [Google Scholar] [CrossRef]
- Maestre-Valero, J.F.; Martin-Gorriz, B.; Soto-García, M.; Martinez-Mate, M.A.; Martinez-Alvarez, V. Producing lettuce in soil-based or in soilless outdoor systems. Which is more economically profitable? Agric. Water Manag. 2018, 206, 48–55. [Google Scholar] [CrossRef]
- Benoit, F.; Ceustermans, N. Some qualitative aspects of tomatoes grown on NFT. Soil. Cult. 1987, 3, 3–7. [Google Scholar]
- Adams, P.; Winsor, G.W. Further studies on the composition and quality of tomato fruit. Rep. Glas. Crop Res. Inst. 1976, 133–138. [Google Scholar]
- Sonneveld, C.; Straver, N. Nutrient Solutions for Vegetables and Flowers Grown in Water or Substrates, 10th ed.; Glasshouse Crops Research Station: Naaldwijk, The Netherlands, 1994. [Google Scholar]
- Urrestarazu, M. Treaty of Cultivation Without Soil; Mundi Pren: Madrid, Spain, 2004. [Google Scholar]
- USDA. Tomatoes. Shipping Point and Market Inspection Instructions; United States Department of Agriculture: Washington, DC, USA, 2005.
- Buendía-Moreno, L.; Sánchez-Martínez, M.J.; Antolinos, V.; Ros-Chumillas, M.; Navarro-Segura, L.; Soto-Jover, S.; Martínez-Hernández, G.B.; López-Gómez, A. Active cardboard box with a coating including essential oils entrapped within cyclodextrins and/or halloysite nanotubes. A case study for fresh tomato storage. Food Control 2020, 107, 106763. [Google Scholar] [CrossRef]
- López-Camelo, A.F.; Gómez, P.A. Comparison of color indexes for tomato ripening. Hortic. Bras. 2004, 22, 534–537. [Google Scholar] [CrossRef]
- Pathare, P.B.; Opara, U.L.; Al-Said, F.A.-J. Colour measurement and analysis in fresh and processed foods: A review. Food Bioprocess Technol. 2013, 6, 36–60. [Google Scholar] [CrossRef]
- Barrett, D.M.; Anthon, G. Lycopene content of california-grown tomato varieties. Acta Hortic. 2001, 542, 165–174. [Google Scholar] [CrossRef]
- Zapata, S.; Dufour, J.P. Ascorbic, dehydroascorbic and isoascorbic acid simultaneous determinations by reverse phase ion interaction HPLC. J. Food Sci. 1992, 57, 506–511. [Google Scholar] [CrossRef]
- Vallejo, F.; Tomás-Barberán, F.A.; García-Viguera, C. Potential bioactive compounds in health promotion from broccoli cultivars grown in Spain. J. Sci. Food Agric. 2002, 82, 1293–1297. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Artés-Hernández, F.; Colares-Souza, F.; Gómez, P.A.; García-Gómez, P.; Artés, F. Innovative cooking techniques for improving the overall quality of a kailan-hybrid broccoli. Food Bioprocess Technol. 2013, 6, 2135–2149. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Gómez, P.A.; Pradas, I.; Artés, F.; Artés-Hernández, F. Moderate UV-C pretreatment as a quality enhancement tool in fresh-cut Bimi® broccoli. Postharvest Biol. Technol. 2011, 62, 327–337. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Kowalczyk, K.; Mirgos, M.; Baczek, K.; Niedzińska, M.; Gajewski, M. Effect of different growing media in hydroponic culture on the yield and biological quality of lettuce (Lactuca sativa var. capitata). Acta Hortic. 2016, 1142, 105–110. [Google Scholar] [CrossRef]
- Reshma, T.; Sarath, P.S. Standardization of growing media for the hydroponic cultivation of tomato. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 626–631. [Google Scholar] [CrossRef]
- Petersen, K.K.; Willumsen, J.; Kaack, K. Composition and taste of tomatoes as affected by increased salinity and different salinity sources. J. Hortic. Sci. Biotechnol. 1998, 73, 205–215. [Google Scholar] [CrossRef]
- Davies, J.N.; Hobson, G.E. The constituents of tomato fruit—The influence of environment, nutrition, and genotype. Crit. Rev. Food Sci. Nutr. 1981, 15, 205–280. [Google Scholar] [CrossRef]
- Dorais, M.; Ehret, D.L.; Papadopoulos, A.P. Tomato (Solanum lycopersicum) health components: From the seed to the consumer. Phytochem. Rev. 2008, 7, 231–250. [Google Scholar] [CrossRef]
- Ho, L.C. The physiological basis for improving tomato fruit quality. Acta Hortic. 1999, 487, 33–40. [Google Scholar] [CrossRef]
- Harker, F.R.; Marsh, K.B.; Young, H.; Murray, S.H.; Gunson, F.A.; Walker, S.B. Sensory interpretation of instrumental measurements 2: Sweet and acid taste of apple fruit. Postharvest Biol. Technol. 2002, 24, 241–250. [Google Scholar] [CrossRef]
- Stevens, M.A.; Kader, A.A.; Albright, M. Potential for increasing tomato flavor via increased sugar and acid content. J. Am. Soc. Hortic. Sci. 1979, 104, 40–42. [Google Scholar]
- Sams, C.E. Preharvest factors affecting postharvest texture. Postharvest Biol. Technol. 1999, 15, 249–254. [Google Scholar] [CrossRef]
- Demarty, M.; Morvan, C.; Thellier, M. Calcium and the cell wall. Plant. Cell Environ. 1984, 7, 441–448. [Google Scholar] [CrossRef]
- Hao, X.; Papadopoulos, A.P. Effects of calcium and magnesium on growth, fruit yield and quality in a fall greenhouse tomato crop grown on rockwool. Can. J. Plant Sci. 2003, 83, 903–912. [Google Scholar]
- Bertin, N.; Génard, M. Tomato quality as influenced by preharvest factors. Sci. Hortic. (Amst.) 2018, 233, 264–276. [Google Scholar] [CrossRef]
- Perveen, R.; Suleria, H.A.R.; Anjum, F.M.; Butt, M.S.; Pasha, I.; Ahmad, S. Tomato (Solanum lycopersicum) carotenoids and lycopenes chemistry; metabolism, absorption, nutrition, and allied health claims—A comprehensive review. Crit. Rev. Food Sci. Nutr. 2015, 55, 919–929. [Google Scholar] [CrossRef]
- Llorente, B.; D’Andrea, L.; Ruiz-Sola, M.A.; Botterweg, E.; Pulido, P.; Andilla, J.; Loza-Alvarez, P.; Rodriguez-Concepcion, M. Tomato fruit carotenoid biosynthesis is adjusted to actual ripening progression by a light-dependent mechanism. Plant J. 2016, 85, 107–119. [Google Scholar] [CrossRef]
- Al-Ajmi, A.; Al-Karaki, G.; Othman, Y. Effect of different substrates on yield and fruit quality of tomato grown in soilless culture. Acta Hortic. 2009, 807, 491–494. [Google Scholar] [CrossRef]
- Lee, S.K.; Kader, A.A. Preharvest and postharvest factors influencing vitamin C content of horticultural crops. Postharvest Biol. Technol. 2000, 20, 207–220. [Google Scholar] [CrossRef]
- Rouphael, Y.; Cardarelli, M.; Bassal, A.; Leonardi, C.; Giuffrida, F.; Colla, G. Vegetable quality as affected by genetic agronomic and environmental factors. J. Food Agric. Environ. 2012, 10, 680–688. [Google Scholar]
- Suvo, T.; Biswas, H.; Jewel, M.; Islam, M.; Khan, M. Impact of substrate on soilless tomato cultivation. Int. J. Agric. Res. Innov. Technol. 2017, 6, 82–86. [Google Scholar] [CrossRef][Green Version]
- EFSA. Scientific opinion on dietary reference values for vitamin C. EFSA J. 2013, 11, 3418. [Google Scholar]
- Slimestad, R.; Verheul, M. Review of flavonoids and other phenolics from fruits of different tomato (Lycopersicon esculentum Mill.) cultivars. J. Sci. Food Agric. 2009, 89, 1255–1270. [Google Scholar] [CrossRef]
- Dannehl, D.; Suhl, J.; Ulrichs, C.; Schmidt, U. Evaluation of substitutes for rock wool as growing substrate for hydroponic tomato production. J. Appl. Bot. Food Q. 2015, 88, 68–77. [Google Scholar]
- Klunklin, W.; Savage, G. Effect on quality characteristics of tomatoes grown under well-watered and drought stress conditions. Foods 2017, 6, 56. [Google Scholar] [CrossRef]
- Falagán, N.; Artes, F.; Gomez, P.A.; Artàes-Hernàandez, F.; Pàerez-Pastor, A.; de la Rosa, J.M.; Aguayo, E. Individual phenolics and enzymatic changes in response to regulated deficit irrigation of extra-early nectarines. J. Am. Soc. Hortic. Sci. 2016, 141, 222–232. [Google Scholar] [CrossRef]
- Nasrabadi, M.; Ramezanian, A.; Eshghi, S.; Kamgar-Haghighi, A.A.; Vazifeshenas, M.R.; Valero, D. Biochemical changes and winter hardiness in pomegranate (Punica granatum L.) trees grown under deficit irrigation. Sci. Hortic. (Amst.) 2019, 251, 39–47. [Google Scholar] [CrossRef]
- Spencer, J.P.E.; Kuhnle, G.G.C.; Hajirezaei, M.; Mock, H.P.; Sonnewald, U.; Rice-Evans, C. The genotypic variation of the antioxidant potential of different tomato varieties. Free Radic. Res. 2005, 39, 1005–1016. [Google Scholar] [CrossRef]
| Irrigation Treatment | EC | Cl− - | NO3− (10.49) | SO42− - | HPO42− (1.50) | Na+ - | Mg2+ (0.82) | K+ (6.91) | Ca2+ (3.43) | NH4+ (0.50) |
|---|---|---|---|---|---|---|---|---|---|---|
| T1 | 2.2 | 3.66 | 10.50 | 1.25 | 1.50 | 3.48 | 1.00 | 7.00 | 3.75 | 0.50 |
| T2 | 2.5 | 5.31 | 10.50 | 1.46 | 1.50 | 5.13 | 1.58 | 7.00 | 3.75 | 0.50 |
| T3 | 3.5 | 7.77 | 10.50 | 3.56 | 1.50 | 7.59 | 3.68 | 7.00 | 7.34 | 0.50 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antolinos, V.; Sánchez-Martínez, M.J.; Maestre-Valero, J.F.; López-Gómez, A.; Martínez-Hernández, G.B. Effects of Irrigation with Desalinated Seawater and Hydroponic System on Tomato Quality. Water 2020, 12, 518. https://doi.org/10.3390/w12020518
Antolinos V, Sánchez-Martínez MJ, Maestre-Valero JF, López-Gómez A, Martínez-Hernández GB. Effects of Irrigation with Desalinated Seawater and Hydroponic System on Tomato Quality. Water. 2020; 12(2):518. https://doi.org/10.3390/w12020518
Chicago/Turabian StyleAntolinos, Vera, María J. Sánchez-Martínez, José F. Maestre-Valero, Antonio López-Gómez, and Ginés B. Martínez-Hernández. 2020. "Effects of Irrigation with Desalinated Seawater and Hydroponic System on Tomato Quality" Water 12, no. 2: 518. https://doi.org/10.3390/w12020518
APA StyleAntolinos, V., Sánchez-Martínez, M. J., Maestre-Valero, J. F., López-Gómez, A., & Martínez-Hernández, G. B. (2020). Effects of Irrigation with Desalinated Seawater and Hydroponic System on Tomato Quality. Water, 12(2), 518. https://doi.org/10.3390/w12020518








