Biochar as an Eco-Friendly and Economical Adsorbent for the Removal of Colorants (Dyes) from Aqueous Environment: A Review
Abstract
:1. Introduction
2. Colorants in Water and Toxicity
2.1. Natural and Synthetic Dyes
2.2. Classification of Dyes
2.2.1. Chromophores and Color Index (C.I.)
2.2.2. Disperse Dyes
2.2.3. Direct Dyes
2.2.4. Reactive Dyes
2.3. Toxicity of Dyes
3. Treatment Technologies for Dyes Removal from Water
3.1. Coagulation
3.2. Advanced Oxidation Processes (AOPs)
3.2.1. Ozonation
3.2.2. Fenton’s Reagent and Fenton-Like Processes
3.3. Membrane Processes
3.3.1. Nanofiltration
3.3.2. Forward Osmosis (FO)
3.4. Biological Process
3.5. Adsorption Process
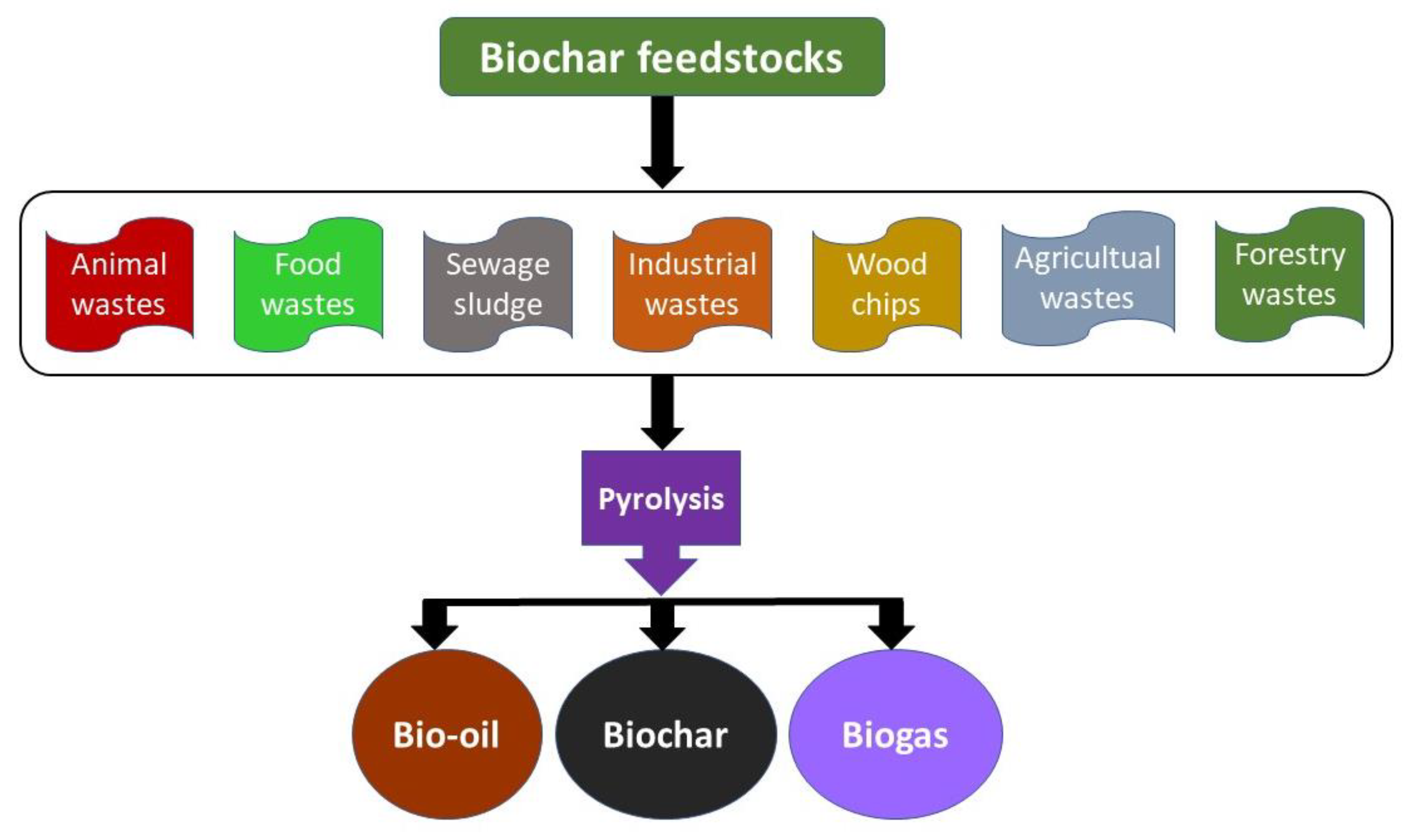
4. Biochar
4.1. Biochar Synthesis by Various Methods
4.1.1. Hydrothermal Carbonization
4.1.2. Hydrothermal Liquefaction
4.1.3. Pyrolysis
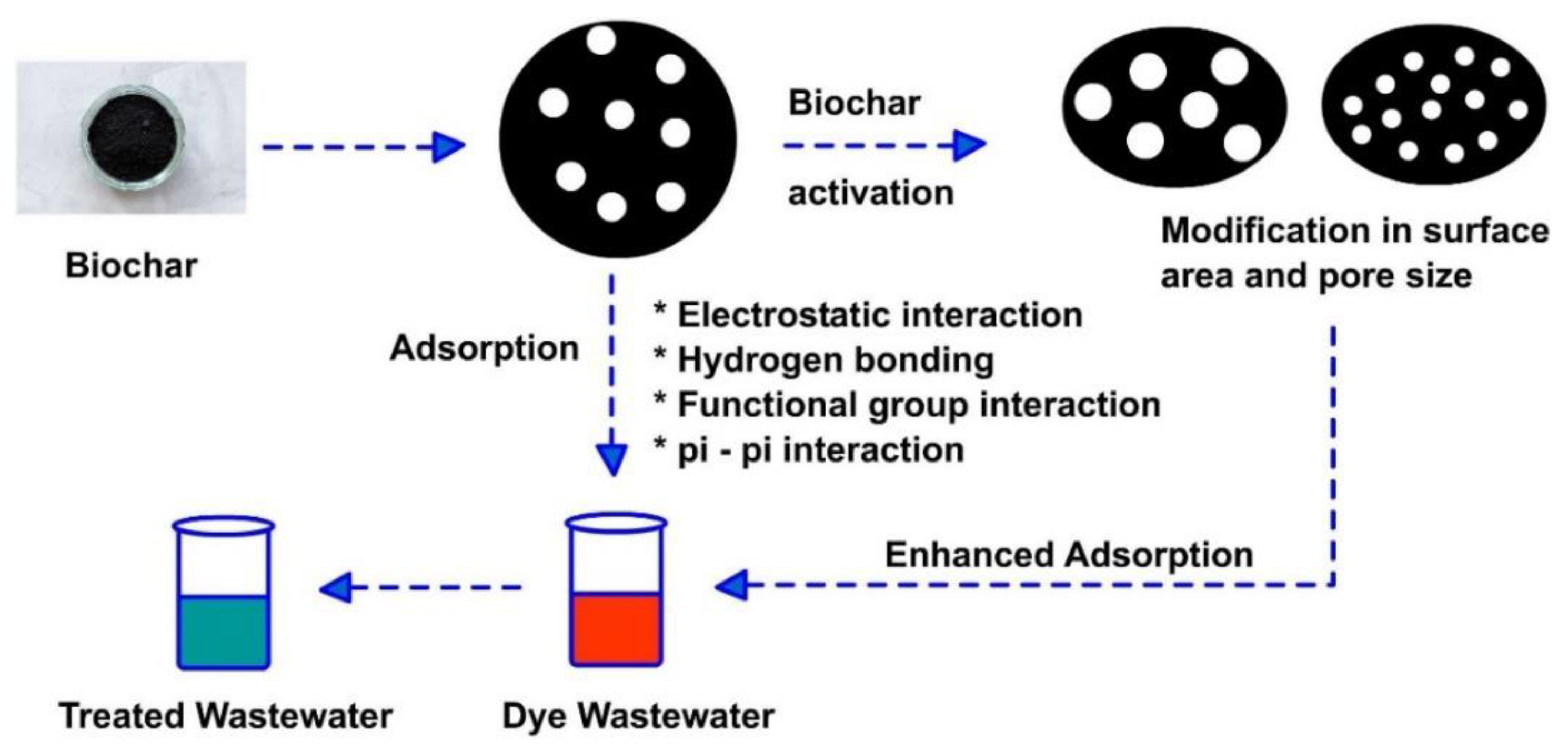
5. Biochar as an Adsorbent
5.1. Effect of Operational Parameters on the Adsorption of Dyes by Biochar
5.2. Adsorption Kinetic Studies
5.3. Adsorption Isotherm Studies
5.4. Mechanisms of Dyes Adsorption onto Biochar
5.5. Potential Drawbacks of Using Biochar as an Adsorbent
6. Conclusions and Future Perspective
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lin, Y.T.; Kao, F.Y.; Chen, S.H.; Wey, M.Y.; Tseng, H.H. A facile approach from waste to resource: Reclaimed rubber-derived membrane for dye removal. J. Taiwan Inst. Chem. Eng. 2020, 112, 286–295. [Google Scholar] [CrossRef]
- Kazemi Shariat Panahi, H.; Dehhaghi, M.; Ok, Y.S.; Nizami, A.S.; Khoshnevisan, B.; Mussatto, S.I.; Aghbashlo, M.; Tabatabaei, M.; Lam, S.S. A comprehensive review of engineered biochar: Production, characteristics, and environmental applications. J. Clean. Prod. 2020, 270, 122462. [Google Scholar] [CrossRef]
- Chi, N.T.L.; Anto, S.; Ahamed, T.S.; Kumar, S.S.; Shanmugam, S.; Samuel, M.S.; Mathimani, T.; Brindhadevi, K.; Pugazhendhi, A. A review on biochar production techniques and biochar based catalyst for biofuel production from algae. Fuel 2020, 119411. [Google Scholar] [CrossRef]
- Dai, Y.; Zhang, N.; Xing, C.; Cui, Q.; Sun, Q. The adsorption, regeneration and engineering applications of biochar for removal organic pollutants: A review. Chemosphere 2019, 223, 12–27. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.A.; Lustosa Filho, J.F.; Melo, L.C.A.; de Assis, I.R.; de Oliveira, T.S. Influence of pyrolysis temperature and feedstock on the properties of biochars produced from agricultural and industrial wastes. J. Anal. Appl. Pyrolysis 2020, 149, 104839. [Google Scholar] [CrossRef]
- Xu, Y.; Luo, G.; He, S.; Deng, F.; Pang, Q.; Xu, Y.; Yao, H. Efficient removal of elemental mercury by magnetic chlorinated biochars derived from co-pyrolysis of Fe(NO3)3-laden wood and polyvinyl chloride waste. Fuel 2019, 239, 982–990. [Google Scholar] [CrossRef]
- Yang, H.; Ye, S.; Zeng, Z.; Zeng, G.; Tan, X.; Xiao, R.; Wang, J.; Song, B.; Du, L.; Qin, M.; et al. Utilization of biochar for resource recovery from water: A review. Chem. Eng. J. 2020, 397. [Google Scholar] [CrossRef]
- Amen, R.; Yaseen, M.; Mukhtar, A.; Ullah, S.; Al-sehemi, A.G.; Ra, S.; Babar, M.; Lai, C.; Ibrahim, M.; Asif, S.; et al. Lead and cadmium removal from wastewater using eco-friendly biochar adsorbent derived from rice husk, wheat straw, and corncob. Clean. Eng. Technol. 2020. [Google Scholar] [CrossRef]
- Santos, G.E.D.S.D.; Lins, P.V.D.S.; Oliveira, L.M.T.D.M.; Da Silva, E.O.; Anastopoulos, I.; Erto, A.; Giannakoudakis, D.A.; De Almeida, A.R.F.; Duarte, J.L.D.S.; Meili, L. Layered double hydroxides/biochar composites as adsorbents for water remediation applications: Recent trends and perspectives. J. Clean. Prod. 2020, 124755. [Google Scholar] [CrossRef]
- Leichtweis, J.; Silvestri, S.; Carissimi, E. New composite of pecan nutshells biochar-ZnO for sequential removal of acid red 97 by adsorption and photocatalysis. Biomass Bioenergy 2020, 140, 105648. [Google Scholar] [CrossRef]
- Fan, S.; Tang, J.; Wang, Y.; Li, H.; Zhang, H.; Tang, J.; Wang, Z.; Li, X. Biochar prepared from co-pyrolysis of municipal sewage sludge and tea waste for the adsorption of methylene blue from aqueous solutions: Kinetics, isotherm, thermodynamic and mechanism. J. Mol. Liq. 2016, 220, 432–441. [Google Scholar] [CrossRef]
- Xiang, W.; Zhang, X.; Chen, J.; Zou, W.; He, F.; Hu, X.; Tsang, D.C.W.; Ok, Y.S.; Gao, B. Biochar technology in wastewater treatment: A critical review. Chemosphere 2020, 252, 126539. [Google Scholar] [CrossRef] [PubMed]
- Benkhaya, S.; M’ rabet, S.; El Harfi, A. A review on classifications, recent synthesis and applications of textile dyes. Inorg. Chem. Commun. 2020, 115, 107891. [Google Scholar] [CrossRef]
- Patel, B.H. Handbook of Textile and Industrial Dyeing; Principles, Processes and Types of Dyes, Volume 1 in Woodhead Publishing Series in Textiles; Clark, M., Ed.; Woodhead Publishing: Sawston, UK, 2011; pp. 395–424. [Google Scholar] [CrossRef]
- Zerin, I.; Farzana, N.; Sayem, A.S.M.; Anang, D.M.; Haider, J. Potentials of Natural Dyes for Textile Applications; Encyclopedia of Renewable and Sustainable Materials, Volume 2; Hashmi, S., Choudhury, I.A., Eds.; Elsevier: Oxford, UK, 2020; pp. 873–883. ISBN 978-0-12-813196-1. [Google Scholar] [CrossRef]
- Nambela, L.; Haule, L.V.; Mgani, Q. A review on source, chemistry, green synthesis and application of textile colorants. J. Clean. Prod. 2020, 246, 119036. [Google Scholar] [CrossRef]
- Saxena, S.; Raja, A.S.M. Natural Dyes: Sources, Chemistry, Application and Sustainability Issues BT—Roadmap to Sustainable Textiles and Clothing: Eco-friendly Raw Materials, Technologies, and Processing Methods; Muthu, S.S., Ed.; Springer Singapore: Singapore, 2014; pp. 37–80. ISBN 978-981-287-065-0. [Google Scholar]
- Degano, I.; Mattonai, M.; Sabatini, F.; Colombini, M.P. A mass spectrometric study on tannin degradation within dyed woolen yarns. Molecules 2019, 24, 2318. [Google Scholar] [CrossRef]
- Christie, R. Colour Chemistry; Royal Society of Chemistry: Cambridge, UK, 2014; ISBN 1849733287. [Google Scholar]
- Raman, C.D.; Kanmani, S. Textile dye degradation using nano zero valent iron: A review. J. Environ. Manag. 2016, 177, 341–355. [Google Scholar] [CrossRef]
- Gürses, A.; Açıkyıldız, M.; Güneş, K.; Gürses, M.S. Classification of Dye and Pigments BT—Dyes and Pigments; Gürses, A., Açıkyıldız, M., Güneş, K., Gürses, M.S., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 31–45. ISBN 978-3-319-33892-7. [Google Scholar]
- Clark, M. Fundamental Principles of Dyeing. In Handbook of Textile and Industrial Dyeing; Woodhead Publishing: Cambridge, UK, 2011; Volume 1, pp. 1–27. [Google Scholar]
- Burkinshaw, S.M. Physico-Chemical Aspects of Textile Coloration; SDC-Society of Dyers and Colourists; Wiley: Hoboken, NJ, USA, 2015; ISBN 9781118725634. [Google Scholar]
- Roy Choudhury, A.K. 2—Dyeing of synthetic fibres. In Woodhead Publishing Series in Textiles; Clark, M., Ed.; Woodhead Publishing: Cambridge, UK, 2011; Volume 2, pp. 40–128. ISBN 978-1-84569-696-2. [Google Scholar]
- Jamil, A.; Bokhari, T.H.; Javed, T.; Mustafa, R.; Sajid, M.; Noreen, S.; Zuber, M.; Nazir, A.; Iqbal, M.; Jilani, M.I. Photocatalytic degradation of disperse dye Violet-26 using TiO2 and ZnO nanomaterials and process variable optimization. J. Mater. Res. Technol. 2020, 9, 1119–1128. [Google Scholar] [CrossRef]
- Broadbent, A. Basic principles of textile coloration. Color Res. Appl. 2003, 28, 230–231. [Google Scholar] [CrossRef]
- Chattopadhyay, D.P. 4—Chemistry of dyeing. In Woodhead Publishing Series in Textiles; Clark, M., Ed.; Woodhead Publishing: Cambridge, UK, 2011; Volume 1, pp. 150–183. ISBN 978-1-84569-695-5. [Google Scholar]
- Jalandoni-Buan, A.C.; Decena-Soliven, A.L.A.; Cao, E.P.; Barraquio, V.L.; Barraquio, W.L. Characterization and identification of Congo red decolorizing bacteria from monocultures and consortia. Philipp. J. Sci. 2010, 139, 71–78. [Google Scholar]
- Pereira, L.; Alves, M. Dyes—Environmental Impact and Remediation BT—Environmental Protection Strategies for Sustainable Development; Malik, A., Grohmann, E., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 111–162. ISBN 978-94-007-1591-2. [Google Scholar]
- Pal, P. Chapter 6—Industry-Specific Water Treatment: Case Studies; Pal, P., Ed.; Butterworth-Heinemann: Oxford, UK, 2017; pp. 243–511. ISBN 978-0-12-810391-3. [Google Scholar]
- Khatri, A.; Peerzada, M.H.; Mohsin, M.; White, M. A review on developments in dyeing cotton fabrics with reactive dyes for reducing effluent pollution. J. Clean. Prod. 2015, 87, 50–57. [Google Scholar] [CrossRef]
- Chavan, R.B. 16—Environmentally friendly dyes. In Woodhead Publishing Series in Textiles; Clark, M., Ed.; Woodhead Publishing: Cambridge, UK, 2011; Volume 1, pp. 515–561. ISBN 978-1-84569-695-5. [Google Scholar]
- Desore, A.; Narula, S.A. An overview on corporate response towards sustainability issues in textile industry. Environ. Dev. Sustain. 2018, 20, 1439–1459. [Google Scholar] [CrossRef]
- Liang, Z.; Wang, J.; Zhang, Y.; Han, C.; Ma, S.; Chen, J.; Li, G.; An, T. Removal of volatile organic compounds (VOCs) emitted from a textile dyeing wastewater treatment plant and the attenuation of respiratory health risks using a pilot-scale biofilter. J. Clean. Prod. 2020, 253, 120019. [Google Scholar] [CrossRef]
- Khatri, J.; Nidheesh, P.V.; Anantha Singh, T.S.; Suresh Kumar, M. Advanced oxidation processes based on zero-valent aluminium for treating textile wastewater. Chem. Eng. J. 2018, 348, 67–73. [Google Scholar] [CrossRef]
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnol. Res. Innov. 2019, 3, 275–290. [Google Scholar] [CrossRef]
- Vikrant, K.; Giri, B.S.; Raza, N.; Roy, K.; Kim, K.-H.; Rai, B.N.; Singh, R.S. Recent advancements in bioremediation of dye: Current status and challenges. Bioresour. Technol. 2018, 253, 355–367. [Google Scholar] [CrossRef]
- Xiang, X.; Chen, X.; Dai, R.; Luo, Y.; Ma, P.; Ni, S.; Ma, C. Anaerobic digestion of recalcitrant textile dyeing sludge with alternative pretreatment strategies. Bioresour. Technol. 2016, 222, 252–260. [Google Scholar] [CrossRef]
- Issa Hamoud, H.; Finqueneisel, G.; Azambre, B. Removal of binary dyes mixtures with opposite and similar charges by adsorption, coagulation/flocculation and catalytic oxidation in the presence of CeO2/H2O2 Fenton-like system. J. Environ. Manag. 2017, 195, 195–207. [Google Scholar] [CrossRef]
- Furlan, F.R.; de Melo da Silva, L.G.; Morgado, A.F.; de Souza, A.A.U.; Guelli Ulson de Souza, S.M.A. Removal of reactive dyes from aqueous solutions using combined coagulation/flocculation and adsorption on activated carbon. Resour. Conserv. Recycl. 2010, 54, 283–290. [Google Scholar] [CrossRef]
- Babu, D.S.; Srivastava, V.; Nidheesh, P.V.; Kumar, M.S. Detoxification of water and wastewater by advanced oxidation processes. Sci. Total Environ. 2019, 696, 133961. [Google Scholar] [CrossRef]
- Nidheesh, P.V.; Zhou, M.; Oturan, M.A. An overview on the removal of synthetic dyes from water by electrochemical advanced oxidation processes. Chemosphere 2018, 197, 210–227. [Google Scholar] [CrossRef] [PubMed]
- Colindres, P.; Yee-Madeira, H.; Reguera, E. Removal of Reactive Black 5 from aqueous solution by ozone for water reuse in textile dyeing processes. Desalination 2010, 258, 154–158. [Google Scholar] [CrossRef]
- Muniyasamy, A.; Sivaporul, G.; Gopinath, A.; Lakshmanan, R.; Altaee, A.; Achary, A.; Velayudhaperumal Chellam, P. Process development for the degradation of textile azo dyes (mono-, di-, poly-) by advanced oxidation process—Ozonation: Experimental & partial derivative modelling approach. J. Environ. Manag. 2020, 265. [Google Scholar] [CrossRef]
- Mascolo, G.; Lopez, A.; Bozzi, A.; Tiravanti, G. By-products formation during the ozonation of the reactive dye uniblu-A. Ozone Sci. Eng. 2002, 24, 439–446. [Google Scholar] [CrossRef]
- Xu, X.; Geng, A.; Yang, C.; Carabineiro, S.A.C.; Lv, K.; Zhu, J. One-pot synthesis of La—Fe—O @ CN composites as photo-Fenton catalysts for highly e ffi cient removal of organic dyes in wastewater. Ceram. Int. 2020, 46, 10740–10747. [Google Scholar] [CrossRef]
- Jin, J.; Du, X.; Yu, J.; Qin, S.; He, M.; Zhang, K.; Chen, G. High performance nanofiltration membrane based on SMA-PEI cross-linked coating for dye/salt separation. J. Memb. Sci. 2020, 611, 118307. [Google Scholar] [CrossRef]
- Qi, Y.; Zhu, L.; Shen, X.; Sotto, A.; Gao, C.; Shen, J. Polythyleneimine-modified original positive charged nano filtration membrane: Removal of heavy metal ions and dyes. Sep. Purif. Technol. 2019, 222, 117–124. [Google Scholar] [CrossRef]
- Meng, L.; Wu, M.; Chen, H.; Xi, Y.; Huang, M.; Luo, X. Rejection of antimony in dyeing and printing wastewater by forward osmosis. Sci. Total Environ. 2020, 745. [Google Scholar] [CrossRef]
- Lin, C.; Tung, K.; Lin, Y.; Dong, C.; Wu, C. l P re of. Process Saf. Environ. Prot. 2020. [Google Scholar] [CrossRef]
- Marzbali, M.H.; Mir, A.A.; Pazoki, M.; Pourjamshidian, R.; Tabeshnia, M. Removal of direct yellow 12 from aqueous solution by adsorption onto spirulina algae as a high-efficiency adsorbent. J. Environ. Chem. Eng. 2017, 5, 1946–1956. [Google Scholar] [CrossRef]
- Ihsanullah, I.; Jamal, A.; Ilyas, M.; Zubair, M.; Khan, G.; Atieh, M.A. Bioremediation of dyes: Current status and prospects. J. Water Process Eng. 2020, 38, 101680. [Google Scholar] [CrossRef]
- Shao, H.; Li, Y.; Zheng, L.; Chen, T.; Liu, J. Removal of methylene blue by chemically modified defatted brown algae Laminaria japonica. J. Taiwan Inst. Chem. Eng. 2017, 80, 525–532. [Google Scholar] [CrossRef]
- Cui, D.; Guo, Y.Q.; Lee, H.S.; Cheng, H.Y.; Liang, B.; Kong, F.Y.; Wang, Y.Z.; Huang, L.P.; Xu, M.Y.; Wang, A.J. Efficient azo dye removal in bioelectrochemical system and post-aerobic bioreactor: Optimization and characterization. Chem. Eng. J. 2014, 243, 355–363. [Google Scholar] [CrossRef]
- Sirajudheen, P.; Karthikeyan, P.; Vigneshwaran, S.; Meenakshi, S. Synthesis and characterization of La(III) supported carboxymethylcellulose-clay composite for toxic dyes removal: Evaluation of adsorption kinetics, isotherms and thermodynamics. Int. J. Biol. Macromol. 2020, 161, 1117–1126. [Google Scholar] [CrossRef] [PubMed]
- Saxena, M.; Sharma, N.; Saxena, R. Highly efficient and rapid removal of a toxic dye: Adsorption kinetics, isotherm, and mechanism studies on functionalized multiwalled carbon nanotubes. Surf. Interfaces 2020, 21, 100639. [Google Scholar] [CrossRef]
- Morais da Silva, P.M.; Camparotto, N.G.; Grego Lira, K.T.; Franco Picone, C.S.; Prediger, P. Adsorptive removal of basic dye onto sustainable chitosan beads: Equilibrium, kinetics, stability, continuous-mode adsorption and mechanism. Sustain. Chem. Pharm. 2020, 18. [Google Scholar] [CrossRef]
- Deniz, F. Adsorption Properties of Low-Cost Biomaterial Derived from Prunus amygdalus L. for Dye Removal from Water. Sci. World J. 2013, 2013, 961671. [Google Scholar] [CrossRef]
- Fan, H.; Ma, Y.; Wan, J.; Wang, Y.; Li, Z.; Chen, Y. Adsorption properties and mechanisms of novel biomaterials from banyan aerial roots via simple modification for ciprofloxacin removal. Sci. Total Environ. 2020, 708, 134630. [Google Scholar] [CrossRef]
- Niinipuu, M.; Latham, K.G.; Boily, J.F.; Bergknut, M.; Jansson, S. The impact of hydrothermal carbonization on the surface functionalities of wet waste materials for water treatment applications. Environ. Sci. Pollut. Res. 2020, 27, 24369–24379. [Google Scholar] [CrossRef]
- Sharma, H.B.; Sarmah, A.K.; Dubey, B. Hydrothermal carbonization of renewable waste biomass for solid biofuel production: A discussion on process mechanism, the influence of process parameters, environmental performance and fuel properties of hydrochar. Renew. Sustain. Energy Rev. 2020, 123, 109761. [Google Scholar] [CrossRef]
- Stirling, R.J.; Snape, C.E.; Meredith, W. The impact of hydrothermal carbonisation on the char reactivity of biomass. Fuel Process. Technol. 2018, 177, 152–158. [Google Scholar] [CrossRef]
- Yusuf, I.; Flagiello, F.; Ward, N.I.; Arellano-García, H.; Avignone-Rossa, C.; Felipe-Sotelo, M. Valorisation of banana peels by hydrothermal carbonisation: Potential use of the hydrochar and liquid by-product for water purification and energy conversion. Bioresour. Technol. Rep. 2020, 100582. [Google Scholar] [CrossRef]
- Bisinoti, C.; Moreira, A.B.; Laranja, J.; Renata, C.J.; Ferreira, O.P.; Melo, C.A. Semivolatile organic compounds in the products from hydrothermal carbonisation of sugar cane bagasse and vinasse by gas chromatography-mass spectrometry. Bioresour. Technol. Rep. 2020, 12. [Google Scholar] [CrossRef]
- Fregolente, L.G.; Miguel, T.B.A.R.; de Castro Miguel, E.; de Almeida Melo, C.; Moreira, A.B.; Ferreira, O.P.; Bisinoti, M.C. Toxicity evaluation of process water from hydrothermal carbonization of sugarcane industry by-products. Environ. Sci. Pollut. Res. 2019, 26, 27579–27589. [Google Scholar] [CrossRef] [PubMed]
- Ischia, G.; Fiori, L. Hydrothermal Carbonization of Organic Waste and Biomass: A Review on Process, Reactor, and Plant Modeling. Waste Biomass Valorization 2020. [Google Scholar] [CrossRef]
- Marzbali, M.H.; Paz-ferreiro, J.; Kundu, S.; Ramezani, M.; Halder, P.; Patel, S.; White, T.; Madapusi, S.; Shah, K. Investigations into distribution and characterisation of products formed during hydrothermal carbonisation of paunch waste. Biochem. Pharmacol. 2020, 104672. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, Z.; Shen, B.; Liu, L. Insights into biochar and hydrochar production and applications: A review. Energy 2019, 171, 581–598. [Google Scholar] [CrossRef]
- Wang, L.; Chang, Y.; Liu, Q. Fate and distribution of nutrients and heavy metals during hydrothermal carbonization of sewage sludge with implication to land application. J. Clean. Prod. 2019, 225, 972–983. [Google Scholar] [CrossRef]
- Maniscalco, M.P.; Volpe, M.; Messineo, A. Hydrothermal carbonization as a valuable tool for energy and environmental applications: A review. Energies 2020, 13, 4098. [Google Scholar] [CrossRef]
- Ponnusamy, V.K.; Nagappan, S.; Bhosale, R.R.; Lay, C.H.; Duc Nguyen, D.; Pugazhendhi, A.; Chang, S.W.; Kumar, G. Review on sustainable production of biochar through hydrothermal liquefaction: Physico-chemical properties and applications. Bioresour. Technol. 2020, 310, 123414. [Google Scholar] [CrossRef]
- Mathimani, T.; Mallick, N. A review on the hydrothermal processing of microalgal biomass to bio-oil - Knowledge gaps and recent advances. J. Clean. Prod. 2019, 217, 69–84. [Google Scholar] [CrossRef]
- Brindhadevi, K.; Anto, S.; Rene, E.R.; Sekar, M. Effect of reaction temperature on the conversion of algal biomass to bio-oil and biochar through pyrolysis and hydrothermal liquefaction. Fuel 2021, 285, 119106. [Google Scholar] [CrossRef]
- Liu, H.; Ma, M.; Xie, X. New materials from solid residues for investigation the mechanism of biomass hydrothermal liquefaction. Ind. Crop. Prod. 2017, 108, 63–71. [Google Scholar] [CrossRef]
- Sun, Y.; Yang, G.; Zhang, L.; Sun, Z. Preparation of high performance H2S removal biochar by direct fluidized bed carbonization using potato peel waste. Process Saf. Environ. Prot. 2017, 107, 281–288. [Google Scholar] [CrossRef]
- Li, F.; Gui, X.; Ji, W.; Zhou, C. Effect of calcium dihydrogen phosphate addition on carbon retention and stability of biochars derived from cellulose, hemicellulose, and lignin. Chemosphere 2020, 251, 126335. [Google Scholar] [CrossRef]
- Li, J.; Li, Y.; Wu, Y.; Zheng, M. A comparison of biochars from lignin, cellulose and wood as the sorbent to an aromatic pollutant. J. Hazard. Mater. 2014, 280, 450–457. [Google Scholar] [CrossRef]
- Kim, D.; Lee, K.; Park, K.Y. Upgrading the characteristics of biochar from cellulose, lignin, and xylan for solid biofuel production from biomass by hydrothermal carbonization. J. Ind. Eng. Chem. 2016, 42, 95–100. [Google Scholar] [CrossRef]
- Qin, L.; Wu, Y.; Hou, Z.; Jiang, E. Influence of biomass components, temperature and pressure on the pyrolysis behavior and biochar properties of pine nut shells. Bioresour. Technol. 2020, 313, 123682. [Google Scholar] [CrossRef]
- Muppaneni, T.; Reddy, H.K.; Selvaratnam, T.; Dandamudi, K.P.R.; Dungan, B.; Nirmalakhandan, N.; Schaub, T.; Omar Holguin, F.; Voorhies, W.; Lammers, P.; et al. Hydrothermal liquefaction of Cyanidioschyzon merolae and the influence of catalysts on products. Bioresour. Technol. 2017, 223, 91–97. [Google Scholar] [CrossRef]
- Cui, Z.; Cheng, F.; Jarvis, J.M.; Brewer, C.E.; Jena, U. Roles of Co-solvents in hydrothermal liquefaction of low-lipid, high-protein algae. Bioresour. Technol. 2020, 310, 123454. [Google Scholar] [CrossRef]
- Shen, Y.; Yu, S.; Yuan, R.; Wang, P. Biomass pyrolysis with alkaline-earth-metal additive for co-production of bio-oil and biochar-based soil amendment. Sci. Total Environ. 2020, 743, 140760. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Hong, M.; Li, H.; Ye, Z.; Gong, H.; Zhang, J.; Huang, Q.; Tan, Z. Contributions and mechanisms of components in modified biochar to adsorb cadmium in aqueous solution. Sci. Total Environ. 2020, 733, 139320. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhang, S.; Wang, J.; Ding, X. Phosphorus retention using iron (II/III) modified biochar in saline-alkaline soils: Adsorption, column and field tests. Environ. Pollut. 2020, 261, 114223. [Google Scholar] [CrossRef] [PubMed]
- Nakarmi, A.; Bourdo, S.E.; Ruhl, L.; Kanel, S.; Nadagouda, M.; Kumar Alla, P.; Pavel, I.; Viswanathan, T. Benign zinc oxide betaine-modified biochar nanocomposites for phosphate removal from aqueous solutions. J. Environ. Manag. 2020, 272, 111048. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xing, B.; Ding, Y.; Han, X.; Wang, S. A critical review of the production and advanced utilization of biochar via selective pyrolysis of lignocellulosic biomass. Bioresour. Technol. 2020, 312, 123614. [Google Scholar] [CrossRef] [PubMed]
- Elkhalifa, S.; Al-Ansari, T.; Mackey, H.R.; McKay, G. Food waste to biochars through pyrolysis: A review. Resour. Conserv. Recycl. 2019, 144, 310–320. [Google Scholar] [CrossRef]
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of process parameters on production of biochar from biomass waste through pyrolysis: A review. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Wang, X.; Li, C.; Li, Z.; Yu, G.; Wang, Y. Effect of pyrolysis temperature on characteristics, chemical speciation and risk evaluation of heavy metals in biochar derived from textile dyeing sludge. Ecotoxicol. Environ. Saf. 2019, 168, 45–52. [Google Scholar] [CrossRef]
- Park, J.H.; Wang, J.J.; Meng, Y.; Wei, Z.; DeLaune, R.D.; Seo, D.C. Adsorption/desorption behavior of cationic and anionic dyes by biochars prepared at normal and high pyrolysis temperatures. Colloids Surf. A Physicochem. Eng. Asp. 2019, 572, 274–282. [Google Scholar] [CrossRef]
- Chen, X.L.; Li, F.; Chen, H.Y.; Wang, H.J.; Li, G.G. Fe2O3/TiO2 functionalized biochar as a heterogeneous catalyst for dyes degradation in water under Fenton processes. J. Environ. Chem. Eng. 2020, 8, 103905. [Google Scholar] [CrossRef]
- Yek, P.N.Y.; Peng, W.; Wong, C.C.; Liew, R.K.; Ho, Y.L.; Wan Mahari, W.A.; Azwar, E.; Yuan, T.Q.; Tabatabaei, M.; Aghbashlo, M.; et al. Engineered biochar via microwave CO2 and steam pyrolysis to treat carcinogenic Congo red dye. J. Hazard. Mater. 2020, 395. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.H.; Kang, J.K.; Park, S.J.; Lee, C.G. Application of magnetic biochar derived from food waste in heterogeneous sono-Fenton-like process for removal of organic dyes from aqueous solution. J. Water Process Eng. 2020, 37, 101455. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Abdelfattah, A.M.; Tharwat, R.M.; Nabil, G.M. Adsorption of negatively charged food tartrazine and sunset yellow dyes onto positively charged triethylenetetramine biochar: Optimization, kinetics and thermodynamic study. J. Mol. Liq. 2020, 318, 114297. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, T.; Wang, M.; Jin, R.; Song, Y.; Zhou, Y.; Qi, Z.; Chen, W. Inhibitory role of citric acid in the adsorption of tetracycline onto biochars: Effects of solution pH and Cu2+. Colloids Surf. A Physicochem. Eng. Asp. 2020, 595. [Google Scholar] [CrossRef]
- Yao, X.; Ji, L.; Guo, J.; Ge, S.; Lu, W.; Chen, Y.; Cai, L.; Wang, Y.; Song, W. An abundant porous biochar material derived from wakame (Undaria pinnatifida) with high adsorption performance for three organic dyes. Bioresour. Technol. 2020, 318, 124082. [Google Scholar] [CrossRef] [PubMed]
- Rubeena, K.K.; Prasad, P.H.; Laiju, A.R.; Nidheesh, P. V Iron impregnated biochars as heterogeneous Fenton catalyst for the degradation of acid red 1 dye. J. Environ. Manag. 2018, 226, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Zazycki, M.A.; Borba, P.A.; Silva, R.N.F.; Peres, E.C.; Perondi, D.; Collazzo, G.C.; Dotto, G.L. Chitin derived biochar as an alternative adsorbent to treat colored effluents containing methyl violet dye. Adv. Powder Technol. 2019, 30, 1494–1503. [Google Scholar] [CrossRef]
- Nautiyal, P.; Subramanian, K.A.; Dastidar, M.G. Adsorptive removal of dye using biochar derived from residual algae after in-situ transesteri fi cation: Alternate use of waste of biodiesel industry. J. Environ. Manag. 2016, 182, 187–197. [Google Scholar] [CrossRef]
- Damertey, D.; Jung, H.; Soo, S.; Sung, D.; Han, S. Decolorization of cationic and anionic dye-laden wastewater by steam- activated biochar produced at an industrial-scale from spent mushroom substrate. Bioresour. Technol. 2019, 277, 77–86. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, W.; Chen, Y.; Li, Y. Nickel aluminum layered double oxides modified magnetic biochar from waste corncob for efficient removal of acridine orange. Bioresour. Technol. 2020, 315, 123834. [Google Scholar] [CrossRef]
- Yu, K.L.; Lee, X.J.; Ong, H.C.; Chen, W.-H.; Chang, J.-S.; Lin, C.-S.; Show, P.L.; Ling, T.C. Adsorptive removal of cationic methylene blue and anionic Congo red dyes using wet-torrefied microalgal biochar: Equilibrium, kinetic and mechanism modeling. Environ. Pollut. 2020, 115986. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Shah, N.S.; Sayed, M.; Niazi, N.K.; Imran, M.; Khan, J.A.; Khan, Z.U.H.; Hussien, A.G.S.; Polychronopoulou, K.; Howari, F. Nano-zerovalent manganese/biochar composite for the adsorptive and oxidative removal of Congo-red dye from aqueous solutions. J. Hazard. Mater. 2021, 403, 123854. [Google Scholar] [CrossRef]
- Ganguly, P.; Sarkhel, R.; Das, P. Synthesis of pyrolyzed biochar and its application for dye removal: Batch, kinetic and isotherm with linear and non-linear mathematical analysis. Surf. Interfaces 2020, 20, 100616. [Google Scholar] [CrossRef]
- Kumar, S.; Abdel-fattah, T.M. Kinetics, isotherm, and thermodynamic studies of the adsorption of reactive red 195 A dye from water by modified Switchgrass Biochar adsorbent. J. Ind. Eng. Chem. 2016. [Google Scholar] [CrossRef]
- Sahu, S.; Pahi, S.; Tripathy, S.; Singh, S.K.; Behera, A.; Sahu, U.K.; Patel, R. Adsorption of methylene blue on chemically modified lychee seed biochar: Dynamic, equilibrium, and thermodynamic study. J. Mol. Liq. 2020, 315, 113743. [Google Scholar] [CrossRef]
- Zhu, K.; Wang, X.; Chen, D.; Ren, W.; Lin, H.; Zhang, H. Wood-based biochar as an excellent activator of peroxydisulfate for Acid Orange 7 decolorization. Chemosphere 2019, 231, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Guo, X. Adsorption kinetic models: Physical meanings, applications, and solving methods. J. Hazard. Mater. 2020, 390, 122156. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Mohapatra, S.S.; Kumari, U.; Meikap, B.C.; Sen, T.K. Batch and continuous closed circuit semi-fluidized bed operation: Removal of MB dye using sugarcane bagasse biochar and alginate composite adsorbents. J. Environ. Chem. Eng. 2020, 8, 103637. [Google Scholar] [CrossRef]
- Côrtes, L.N.; Druzian, S.P.; Streit, A.F.M.; Godinho, M.; Perondi, D.; Collazzo, G.C.; Oliveira, M.L.S.; Cadaval, T.R.S.; Dotto, G.L. Biochars from animal wastes as alternative materials to treat colored effluents containing basic red 9. J. Environ. Chem. Eng. 2019, 7. [Google Scholar] [CrossRef]
- Xu, X.; Xu, Z.; Huang, J.; Gao, B.; Zhao, L.; Qiu, H.; Cao, X. Sorption of reactive red by biochars ball milled in different atmospheres: Co-effect of surface morphology and functional groups. Chem. Eng. J. 2020, 127468. [Google Scholar] [CrossRef]
- Wang, H.; Wang, S.; Gao, Y. Cetyl trimethyl ammonium bromide modified magnetic biochar from pine nut shells for efficient removal of acid chrome blue K. Bioresour. Technol. 2020, 312, 123564. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yan, W.; He, C.; Wen, H.; Cai, Z.; Wang, Z.; Chen, Z.; Liu, W. Microwave-assisted preparation of nitrogen-doped biochars by ammonium acetate activation for adsorption of acid red 18. Appl. Surf. Sci. 2018, 433, 222–231. [Google Scholar] [CrossRef]
- Chen, L.; Jiang, X.; Xie, R.; Zhang, Y.; Jin, Y.; Jiang, W. A novel porous biochar-supported Fe-Mn composite as a persulfate activator for the removal of acid red 88. Sep. Purif. Technol. 2020, 250, 117232. [Google Scholar] [CrossRef]
- Hoslett, J.; Ghazal, H.; Mohamad, N.; Jouhara, H. Removal of methylene blue from aqueous solutions by biochar prepared from the pyrolysis of mixed municipal discarded material. Sci. Total Environ. 2020, 714, 136832. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.N. Comments on “High-efficiency removal of dyes from wastewater by fully recycling litchi peel biochar”. Chemosphere 2020, 257, 126444. [Google Scholar] [CrossRef] [PubMed]
- Ang, T.N.; Young, B.R.; Taylor, M.; Burrell, R.; Aroua, M.K.; Chen, W.-H.; Baroutian, S. Enrichment of surface oxygen functionalities on activated carbon for adsorptive removal of sevoflurane. Chemosphere 2020, 260, 127496. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, M.; Kumar, R.; Neogi, S. Activated biochar derived from Opuntia ficus-indica for the efficient adsorption of malachite green dye, Cu+2 and Ni+2 from water. J. Hazard. Mater. 2020, 392, 122441. [Google Scholar] [CrossRef]
- Shan, R.; Lu, L.; Gu, J.; Zhang, Y.; Yuan, H.; Chen, Y.; Luo, B. Photocatalytic degradation of methyl orange by Ag/TiO2/biochar composite catalysts in aqueous solutions. Mater. Sci. Semicond. Process. 2020, 114, 105088. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Da’ana, D.A. Guidelines for the use and interpretation of adsorption isotherm models: A review. J. Hazard. Mater. 2020, 393, 122383. [Google Scholar] [CrossRef]
- Choi, J.; Won, W.; Capareda, S.C. The economical production of functionalized Ashe juniper derived-biochar with high hazardous dye removal efficiency. Ind. Crop. Prod. 2019, 137, 672–680. [Google Scholar] [CrossRef]
- Ji, B.; Wang, J.; Song, H.; Chen, W. Removal of methylene blue from aqueous solutions using biochar derived from a fallen leaf by slow pyrolysis: Behavior and mechanism. J. Environ. Chem. Eng. 2019, 7, 103036. [Google Scholar] [CrossRef]
- Jabar, J.M.; Odusote, Y.A. Removal of cibacron blue 3G-A (CB) dye from aqueous solution using chemo-physically activated biochar from oil palm empty fruit bunch fiber. Arab. J. Chem. 2020, 13, 5417–5429. [Google Scholar] [CrossRef]
- Zubair, M.; Manzar, M.S.; Mu’azu, N.D.; Anil, I.; Blaisi, N.I.; Al-Harthi, M.A. Functionalized MgAl-layered hydroxide intercalated date-palm biochar for Enhanced Uptake of Cationic dye: Kinetics, isotherm and thermodynamic studies. Appl. Clay Sci. 2020, 190, 105587. [Google Scholar] [CrossRef]
- Khan, N.; Chowdhary, P.; Ahmad, A.; Giri, B.S.; Chaturvedi, P. Hydrothermal liquefaction of rice husk and cow dung in Mixed-Bed-Rotating Pyrolyzer and application of biochar for dye removal. Bioresour. Technol. 2020, 309, 123294. [Google Scholar] [CrossRef]
- Oladipo, A.A.; Ifebajo, A.O. Highly efficient magnetic chicken bone biochar for removal of tetracycline and fluorescent dye from wastewater: Two-stage adsorber analysis. J. Environ. Manag. 2018, 209, 9–16. [Google Scholar] [CrossRef]
- Zhang, P.; O’Connor, D.; Wang, Y.; Jiang, L.; Xia, T.; Wang, L.; Tsang, D.C.W.; Ok, Y.S.; Hou, D. A green biochar/iron oxide composite for methylene blue removal. J. Hazard. Mater. 2020, 384, 121286. [Google Scholar] [CrossRef]
- Hou, Y.; Huang, G.; Li, J.; Yang, Q.; Huang, S.; Cai, J. Hydrothermal conversion of bamboo shoot shell to biochar: Preliminary studies of adsorption equilibrium and kinetics for rhodamine B removal. J. Anal. Appl. Pyrolysis 2019, 143, 104694. [Google Scholar] [CrossRef]
- Muhammad, N.Z.; Qamar, D.; Faisal, N.; Muhammad, N.Z.; Munawar, I.; Muhammad, F.N. Effective adsorptive removal of azo dyes over spherical ZnO nanoparticles. J. Mater. Res. Technol. 2019, 8, 713–725. [Google Scholar]
- Pal, P.; Pal, A. Dye removal using waste beads: Efficient utilization of surface-modified chitosan beads generated after lead adsorption process. J. Water Process Eng. 2019, 31. [Google Scholar] [CrossRef]
- Dai, L.; Zhu, W.; He, L.; Tan, F.; Zhu, N.; Zhou, Q.; He, M.; Hu, G. Calcium-rich biochar from crab shell: An unexpected super adsorbent for dye removal. Bioresour. Technol. 2018, 267, 510–516. [Google Scholar] [CrossRef]
- Boudechiche, N.; Fares, M.; Ouyahia, S.; Yazid, H.; Trari, M.; Sadaoui, Z. Comparative study on removal of two basic dyes in aqueous medium by adsorption using activated carbon from Ziziphus lotus stones. Microchem. J. 2019, 146, 1010–1018. [Google Scholar] [CrossRef]
- Chahinez, H.-O.; Abdelkader, O.; Leila, Y.; Tran, H.N. One-stage preparation of palm petiole-derived biochar: Characterization and application for adsorption of crystal violet dye in water. Environ. Technol. Innov. 2020, 19, 100872. [Google Scholar] [CrossRef]
- Gokulan, R.; Avinash, A.; Prabhu, G.G.; Jegan, J. Remediation of remazol dyes by biochar derived from Caulerpa scalpelliformis—An eco-friendly approach. J. Environ. Chem. Eng. 2019, 7, 103297. [Google Scholar] [CrossRef]
- Kah, M.; Sigmund, G.; Xiao, F.; Hofmann, T. Sorption of ionizable and ionic organic compounds to biochar, activated carbon and other carbonaceous materials. Water Res. 2017, 124, 673–692. [Google Scholar] [CrossRef] [PubMed]
- Spagnoli, A.A.; Giannakoudakis, D.A.; Bashkova, S. Adsorption of methylene blue on cashew nut shell based carbons activated with zinc chloride: The role of surface and structural parameters. J. Mol. Liq. 2017, 229, 465–471. [Google Scholar] [CrossRef]
- Del Bubba, M.; Anichini, B.; Bakari, Z.; Bruzzoniti, M.C.; Camisa, R.; Caprini, C.; Checchini, L.; Fibbi, D.; El Ghadraoui, A.; Liguori, F.; et al. Physicochemical properties and sorption capacities of sawdust-based biochars and commercial activated carbons towards ethoxylated alkylphenols and their phenolic metabolites in effluent wastewater from a textile district. Sci. Total Environ. 2020, 708, 135217. [Google Scholar] [CrossRef]
- Santra, B.; Ramrakhiani, L.; Kar, S.; Ghosh, S.; Majumdar, S. Ceramic membrane-based ultrafiltration combined with adsorption by waste derived biochar for textile effluent treatment and management of spent biochar. J. Environ. Health Sci. Eng. 2020. [Google Scholar] [CrossRef]
- De Gisi, S.; Notarnicola, M. Industrial Wastewater Treatment; Abraham, M., Ed.; Elsevier: Oxford, UK, 2017; pp. 23–42. ISBN 978-0-12-804792-7. [Google Scholar]
- Odinga, E.S.; Waigi, M.G.; Gudda, F.O.; Wang, J.; Yang, B.; Hu, X.; Li, S.; Gao, Y. Occurrence, formation, environmental fate and risks of environmentally persistent free radicals in biochars. Environ. Int. 2020, 134, 105172. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Z.; Xu, W.; Liao, Q.; Zhang, H.; Hao, S.; Chen, S. Pyrolysis of various phytoremediation residues for biochars: Chemical forms and environmental risk of Cd in biochar. Bioresour. Technol. 2020, 299, 122581. [Google Scholar] [CrossRef]
- Zhang, Q.; Ye, X.; Li, H.; Chen, D.; Xiao, W.; Zhao, S.; Xiong, R.; Li, J. Cumulative effects of pyrolysis temperature and process on properties, chemical speciation, and environmental risks of heavy metals in magnetic biochar derived from coagulation-flocculation sludge of swine wastewater. J. Environ. Chem. Eng. 2020, 8, 104472. [Google Scholar] [CrossRef]
| Treatment Technology for Dyes Removal | Advantages | Disadvantages |
|---|---|---|
| Coagulation |
|
|
| Advanced Oxidation Processes (AOP) |
|
|
| Membrane Processes |
|
|
| Biological Processes |
|
|
| Adsorption Processes |
|
|
| Adsorbent | Adsorbate | Applicable Kinetic Models | Reference |
|---|---|---|---|
| Steam activated spent mushroom substrate (SMS) | Congo red and crystal violet | Pseudo first order Pseudo second order | [100] |
| Nickel aluminum layered double oxides modified magnetic biochar | Acridine orange | Pseudo first order and Pseudo second order | [101] |
| Chemically modified lychee seed biochar | Methylene blue | Pseudo second order | [117] |
| Triethylenetetramine biochar | Sunset yellow dye | Pseudo second order | [94] |
| Orange peel waste microwave activated biochar | Congo red dye | Pseudo second order | [92] |
| Fe2O3/TiO2 functionalized biochar | Methylene blue, Methyl orange, Rhodamine B | Pseudo first order | [91] |
| Switchgrass biochar | Methylene Blue, Orange G, Congo red | Pseudo first and second order model, Intra-particle diffusion model | [90] |
| Biochar derived from Opuntia ficus-indica (OFI) cactus | Malachite green | Pseudo second order model, Elovich model | [118] |
| N-doped biochar | Acid red 18 | Intra-particle diffusion model | [113] |
| Ag-TiO2 biochar | Methyl orange | Pseudo first order kinetic model | [119] |
| Biochar derived from mixed municipal discarded material | Methylene blue | Pseudo first and second order model Intra-particle diffusion model | [115] |
| Mesoporous nano-zerovalent manganese (nZVMn) and Phoenix dactylifera leaves biochar (PBC) composite. | Congo red | Pseudo first and second order model | [103] |
| Cetyl trimethyl ammonium bromide modified magnetic biochar derived from pine nutshells | Acid chrome blue K | Pseudo first order model Pseudo second order model | [112] |
| Pristine and ball milled biochar | Reactive red 120 | Pseudo-second order kinetic model Intra-particle diffusion model | [111] |
| Wood based biochar | Acid orange 7 | Langmuir-Hinshelwood (L-H) kinetic mode | [107] |
| Pyrolyzed rice husk biochar | Malachite green | Intraparticle Diffusion Model Elovich model Pseudo second order model Pseudo-first order model | [104] |
| Animal waste biochar | Basic red 9 | Pseudo first order model Pseudo second order model Pseudo Nth order model | [110] |
| Adsorbent | Adsorbate | Applicable Isotherm Models | Reference |
|---|---|---|---|
| Mg-Al-layered hydroxide intercalated date-palm biochar composites | Methylene blue | Langmuir, Freundlich | [124] |
| Rice husk biochar | Congo red | Langmuir, Freundlich | [125] |
| Cow dung biochar | Congo red | Langmuir, Freundlich | [125] |
| Magnetic chicken bone biochar | Rhodamine B | Freundlich | [126] |
| Chitin derived biochar | Methyl violet | Freundlich, Langmuir, Liu | [98] |
| Switchgrass biochar | Methylene blue | Langmuir, Freundlich | [90] |
| Switchgrass biochar | Orange G | Langmuir, Freundlich | [90] |
| Switchgrass biochar | Congo red | Langmuir, Freundlich | [90] |
| Banana peel extract and FeSO4 | Methylene blue | Langmuir, Freundlich, Dubinin-Radushkevich | [127] |
| Chemically modified lychee seed biochar | Methylene blue | Langmuir, Freundlich, Temkin, Dubinin-Radushkevich | [117] |
| Bamboo shoot shell biochar | Rhodamine B | Langmuir, Freundlich | [128] |
| Cetyl trimethyl ammonium bromide modified magnetic biochar from pine nut shells | Acid chrome blue K | Langmuir, Freundlich | [112] |
| Bovine bone biochar | Basic Red 9 | Langmuir, Freundlich | [110] |
| Fish scale biochar | Basic Red 9 | Langmuir, Freundlich | [110] |
| Adsorbent | Adsorbate | Adsorbent Dose | Contact Time (min) | Adsorption Capacity (mg/g) | Temperature (°C) | pH | References |
|---|---|---|---|---|---|---|---|
| Chitosan Based Material | Basic Blue 7 | 0.3 g/L | 60 | 1174 mg/g | 25 | 6 | [57] |
| Wet-torrefied microalgal biochar | Methylene blue | 1 g/L | 7200 | 113.00 mg/g | 25 | 6 | [129] |
| Wet-torrefied microalgal biochar | Congo red | 2 g/L | 240 | 164.35 mg/g | 25 | 6–8 | [129] |
| Surfactant Modified Chitosan Beads | Crystal violet | 0.36 g/L | 120 | 97.09 mg/g | 50 | 6 | [130] |
| Surfactant Modified Chitosan Beads | Tartrazine | 0.72 g/L | 120 | 30.03 mg/g | 50 | 3 | [130] |
| Calcium rich biochar from Crab Shell | Malachite green | 0.5 g/L | 150 | 12,501.98 mg/g | 25 | 7 | [131] |
| Calcium rich biochar from Crab Shell | Congo red | 0.5 g/L | 2 | 20,317 mg/g | 25 | 4 | [131] |
| Ziziphus Lotus stones | Basic Yellow 28 | 0.5 g/L | 180 | 424 mg/g | 20 | 8 | [132] |
| Ziziphus Lotus stones | Basic Red 46 | 0.5 g/L | 180 | 307 mg/g | 20 | 8 | [132] |
| Date palm petiole derived biochar | Crystal violet | NA | 15 | 209 mg/g | 30 | 7 | [133] |
| Activated wakame biochar material | Methylene blue | 0.06 g/50 mL | NA | 841.64 mg/g | 20 | 2–12 | [96] |
| Activated wakame biochar material | Rhodamine B | 0.06 g/50 mL | NA | 533.77 mg/g | 20 | 2–12 | [96] |
| Activated wakame biochar material | Malachite green | 0.06 g/50 mL | NA | 4066.96 mg/g | 20 | 2–7 | [96] |
| Activated biochar derived from Opuntia ficus-indica | Malachite green | 60 mg/100 mL | 120 | 1341.38 mg/g | 30 | 6 | [118] |
| Biochar derived from Caulerpa scalpelliformis | Remazol brilliant blue R | 2 g/L | 300 | 129.00 mg/g | 30 | 2 | [134] |
| Biochar derived from Caulerpa scalpelliformis | Remazol brilliant orange 3R | 2 g/L | 300 | 130.11 mg/g | 30 | 2 | [134] |
| Biochar derived from Caulerpa scalpelliformis | Remazol brilliant violet 5R | 2 g/L | 300 | 139.76 mg/g | 30 | 2 | [134] |
| Biochar derived from Caulerpa scalpelliformis | Remazol black B | 2 g/L | 300 | 159.08 mg/g | 30 | 2 | [134] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Srivatsav, P.; Bhargav, B.S.; Shanmugasundaram, V.; Arun, J.; Gopinath, K.P.; Bhatnagar, A. Biochar as an Eco-Friendly and Economical Adsorbent for the Removal of Colorants (Dyes) from Aqueous Environment: A Review. Water 2020, 12, 3561. https://doi.org/10.3390/w12123561
Srivatsav P, Bhargav BS, Shanmugasundaram V, Arun J, Gopinath KP, Bhatnagar A. Biochar as an Eco-Friendly and Economical Adsorbent for the Removal of Colorants (Dyes) from Aqueous Environment: A Review. Water. 2020; 12(12):3561. https://doi.org/10.3390/w12123561
Chicago/Turabian StyleSrivatsav, Prithvi, Bhaskar Sriharsha Bhargav, Vignesh Shanmugasundaram, Jayaseelan Arun, Kannappan Panchamoorthy Gopinath, and Amit Bhatnagar. 2020. "Biochar as an Eco-Friendly and Economical Adsorbent for the Removal of Colorants (Dyes) from Aqueous Environment: A Review" Water 12, no. 12: 3561. https://doi.org/10.3390/w12123561
APA StyleSrivatsav, P., Bhargav, B. S., Shanmugasundaram, V., Arun, J., Gopinath, K. P., & Bhatnagar, A. (2020). Biochar as an Eco-Friendly and Economical Adsorbent for the Removal of Colorants (Dyes) from Aqueous Environment: A Review. Water, 12(12), 3561. https://doi.org/10.3390/w12123561






