Electrochemical Oxidation of Effluents from Food Processing Industries: A Short Review and a Case-Study
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chatzisymeon, E.; Stypas, E.; Bousios, S.; Xekoukoulotakis, N.P.; Mantzavinos, D. Photocatalytic treatment of black table olive processing wastewater. J. Hazard. Mater. 2008, 154, 1090–1097. [Google Scholar] [CrossRef] [PubMed]
- Abdallh, M.N.; Abdelhalim, W.S.; Abdel-Halim, H.S. Industrial wastewater treatment of food industry using best techniques. Int. J. Eng. Sci. Invent. 2016, 5, 15–28. [Google Scholar]
- Iskandar, M.J.; Baharum, A.; Anuar, F.A.; Othaman, R. Palm oil industry in South East Asia and the effluent treatment technology—A review. Environ. Technol. Innov. 2018, 9, 169–185. [Google Scholar] [CrossRef]
- Commission Implementing Decision (EU) 2019/2031. Official Journal of the European Union. 2019, pp. L313/60–L313/93. Available online: http://data.europa.eu/eli/dec_impl/2019/2031/oj (accessed on 7 September 2020).
- Feitshans, T.A. Environmental Regulations and the Food Industry. In Guide to US Food Laws and Regulations, 2nd ed.; Curtis, P.A., Ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2013; pp. 121–141. [Google Scholar] [CrossRef]
- Hou, L.; Ji, D.; Zang, L. Inhibition of anaerobic biological treatment: A Review. Iop Conf. Ser. Earth Environ. Sci. 2018, 112, 012006. Available online: https://iopscience.iop.org/article/10.1088/1755-1315/112/1/012006/pdf (accessed on 10 September 2020). [CrossRef]
- Welz, P.J.; Holtman, G.; Haldenwang, R.; le Roes-Hill, M. Characterisation of winery wastewater from continuous flow settling basins and waste stabilisation ponds over the course of 1 year: Implications for biological wastewater treatment and land application. Water Sci. Technol. 2016, 74, 2036–2050. [Google Scholar] [CrossRef] [Green Version]
- Brillas, E.; Cabot, P.L.; Casado, J. Electrochemical methods for degradation of organic pollutants in aqueous media. In Chemical Degradation Methods for Wastes and Pollutants. Environmental and Industrial Applications; Tarr, M., Ed.; Marcel Dekker: New York, NY, USA, 2003; pp. 235–304. [Google Scholar]
- Comninellis, C.; Kapalka, A.; Malato, S.; Parsons, S.A.; Poulios, I.; Mantzavinos, D. Advanced oxidation processes for water treatment: Advances and trends for R&D. J. Chem. Technol. Biotechnol. 2008, 83, 769–776. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; Ferro, S. Electrochemical oxidation of organic pollutants for the wastewater treatment: Direct and indirect processes. Chem. Soc. Rev. 2006, 35, 1324–1340. [Google Scholar] [CrossRef]
- Ghazouani, M.; Akrout, H.; Bousselmi, L. Nitrate and carbon matter removals from real effluents using Si/BDD electrode. Environ. Sci. Pollut. Res. 2017, 24, 9895–9906. [Google Scholar] [CrossRef]
- Panizza, M.; Brillas, E.; Comninellis, C. Application of boron-doped diamond electrodes for wastewater treatment. J. Environ. Eng. Manag. 2008, 18, 139–153. [Google Scholar]
- Zambrano, J.; Min, B. Electrochemical treatment of leachate containing highly concentrated phenol and ammonia using a Pt/Ti anode at different current densities. Environ. Technol. Innov. 2020, 18, 100632. [Google Scholar] [CrossRef]
- Sultana, S.; Choudhury, M.R.; Bakr, A.R.; Anwar, N.; Rahaman, M.S. Effectiveness of electro-oxidation and electro-Fenton processes in removal of organic matter from high-strength brewery wastewater. J. Appl. Electrochem. 2018, 48, 519–528. [Google Scholar] [CrossRef]
- Cañizares, P.; Martínez, L.; Paz, R.; Sáez, C.; Lobato, J.; Rodrigo, M.A. Treatment of Fenton-refractory olive oil mill wastes by electrochemical oxidation with boron-doped diamond anodes. J. Chem. Technol. Biotechnol. 2006, 81, 1331–1337. [Google Scholar] [CrossRef]
- Deligiorgis, A.; Xekoukoulotakis, N.P.; Diamadopoulos, E.; Mantzavinos, D. Electrochemical oxidation of table olive processing wastewater over boron-doped diamond electrodes: Treatment optimization by factorial design. Water Res. 2008, 42, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Chatzisymeon, E.; Xekoukoulotakis, N.P.; Diamadopoulos, E.; Katsaounis, A.; Mantzavinos, D. Boron-doped diamond anodic treatment of olive mill wastewaters: Statistical analysis, kinetic modeling and biodegradability. Water Res. 2009, 43, 3999–4009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbosa, J.; Fernandes, A.; Ciríaco, L.; Lopes, A.; Pacheco, M.J. Electrochemical treatment of olive processing wastewater using a boron-doped diamond anode. Clean Soil Air Water 2016, 44, 1242–1249. [Google Scholar] [CrossRef]
- Israilides, C.J.; Vlyssides, A.G.; Mourafeti, V.N.; Karvouni, G. Olive oil wastewater treatment with the use of an electrolysis system. Bioresour. Technol. 1997, 61, 163–170. [Google Scholar] [CrossRef]
- Belaid, C.; Kallel, M.; Khadraoui, M.; Lalleve, G.; Elleuch, B.; Fauvarque, J.F. Electrochemical treatment of olive mill wastewaters: Removal of phenolic compounds and decolourization. J. Appl. Electrochem. 2006, 36, 1175–1182. [Google Scholar] [CrossRef]
- Kotta, E.; Kalogerakis, N.; Mantzavinos, D. The effect of solids on the electrochemical treatment of olive mill effluents. J. Chem. Technol. Biotechnol. 2007, 82, 504–511. [Google Scholar] [CrossRef]
- Belaid, C.; Khadraoui, M.; Mseddi, S.; Kallel, M.; Elleuch, B.; Fauvarque, J.F. Electrochemical treatment of olive mill wastewater: Treatment extent and effluent phenolic compounds monitoring using some uncommon analytical tools. J. Environ. Sci. 2013, 25, 220–230. [Google Scholar] [CrossRef]
- Kul, S.; Boncukcuoglu, R.; Yilmaz, A.E.; Fil, B.A. Treatment of olive mill wastewater with electro-oxidation method. J. Electrochem. Soc. 2015, 162, G41–G47. Available online: https://iopscience.iop.org/article/10.1149/2.0451508jes (accessed on 10 September 2020). [CrossRef]
- Gotsi, M.; Kalogerakis, N.; Psillakisa, E.; Samaras, P.; Mantzavinos, D. Electrochemical oxidation of olive oil mill wastewaters. Water Res. 2005, 39, 4177–4187. [Google Scholar] [CrossRef] [PubMed]
- Giannis, A.; Kalaitzakis, M.; Diamadopoulos, E. Electrochemical treatment of olive mill wastewater. J. Chem. Technol. Biotechnol. 2007, 82, 663–671. [Google Scholar] [CrossRef]
- Un, U.T.; Altay, U.; Koparal, A.S.; Ogutveren, U.B. Complete treatment of olive mill wastewaters by electrooxidation. Chem. Eng. J. 2008, 139, 445–452. [Google Scholar] [CrossRef]
- Papastefanakis, N.; Mantzavinos, D.; Katsaounis, A. DSA electrochemical treatment of olive mill wastewater on Ti/RuO2 anode. J. Appl. Electrochem. 2010, 40, 729–737. [Google Scholar] [CrossRef]
- Panizza, M.; Cerisola, G. Olive mill wastewater treatment by anodic oxidation with parallel plate electrodes. Water Res. 2006, 40, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- Chatzisymeon, E.; Dimou, A.; Mantzavinos, D.; Katsaounis, A. Electrochemical oxidation of model compounds and olive mill wastewater over DSA electrodes: 1. The case of Ti/IrO2 anode. J. Hazard. Mater. 2009, 167, 268–274. [Google Scholar] [CrossRef]
- Gonçalves, M.R.; Marques, I.P.; Correia, J.P. Electrochemical mineralization of anaerobically digested olive mill wastewater. Water Res. 2012, 46, 4217–4225. [Google Scholar] [CrossRef]
- Sharma, S.; Simsek, H. Treatment of canola-oil refinery effluent using electrochemical methods: A comparison between combined electrocoagulation + electrooxidation and electrochemical peroxidation methods. Chemosphere 2019, 221, 630–639. [Google Scholar] [CrossRef]
- Markou, V.; Kontogianni, M.-C.; Frontistis, Z.; Tekerlekopoulou, A.G.; Katsaounis, A.; Vayenas, D. Electrochemical treatment of biologically pre-treated dairy wastewater using dimensionally stable anodes. J. Environ. Manag. 2017, 202, 217–224. [Google Scholar] [CrossRef]
- Ghazouani, M.; Akrout, H.; Jellali, S.; Bousselmi, L. Comparative study of electrochemical hybrid systems for the treatment of real wastewaters from agri-food activities. Sci. Total Environ. 2019, 647, 1651–1664. [Google Scholar] [CrossRef]
- Candia-Onfray, C.; Espinoza, N.; Sabino da Silva, E.B.; Toledo-Neira, C.; Espinoza, L.C.; Santander, R.; García, V.; Salazar, R. Treatment of winery wastewater by anodic oxidation using BDD electrode. Chemosphere 2018, 206, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Manisankar, P.; Viswanathan, S.; Rani, C. Electrochemical treatment of distillery effluent using catalytic anodes. Green Chem. 2003, 5, 270–274. [Google Scholar] [CrossRef]
- Piya-Areetham, P.; Shenchunthichai, K.; Hunsom, M. Application of electrooxidation process for treating concentrated wastewater from distillery industry with a voluminous electrode. Water Res. 2006, 40, 2857–2864. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Pugliese, C.E.; Moreno-Wiedman, P.; Machuca-Martínez, F.; Marriaga-Cabrales, N. Distillery wastewater treated by electrochemical oxidation with boron-doped diamond electrodes. J. Adv. Oxid. Technol. 2011, 14, 213–219. [Google Scholar] [CrossRef]
- Alvarez-Pugliese, C.E.; Martinez-Hernández, L.; Imbachi-Ordoñez, S.; Marriaga-Cabrales, N.; Machuca-Martinez, F. Pilot scale anodic oxidation of pretreated vinasse using boron doped diamond electrodes. CTF Cienc. Tecnol. Y Futuro 2016, 6, 67–78. [Google Scholar] [CrossRef] [Green Version]
- Vijayaraghavan, K.; Ahmad, D.; Lesa, R. Electrolytic treatment of beer brewery wastewater. Ind. Eng. Chem. Res. 2006, 45, 6854–6859. [Google Scholar] [CrossRef]
- Indhumathi, P.; Chellammal, S.; Basha, C.A.; Raghavan, M. Electrochemical treatment of starch effluent using RuO2/Ti and PbO2/Ti electrodes. Bull. Electrochem. 2001, 17, 535–538. Available online: https://krc.cecri.res.in/ro_2001/30-2001.pdf (accessed on 14 September 2020).
- Sharma, S.; Simsek, H. Sugar beet industry process wastewater treatment using electrochemical methods and optimization of parameters using response surface methodology. Chemosphere 2020, 238, 124669. [Google Scholar] [CrossRef]
- Chiang, L.-C.; Chang, J.-E.; Wen, T.-C. Electrochemical oxidation process for the treatment of coke-plant wastewater. J. Environ. Sci. Health Part. A-Toxic/Hazard. Subst. Environ. Eng. 1995, 30, 753–771. [Google Scholar] [CrossRef]
- Oliveira, E.M.S.; Silva, F.R.; Morais, C.C.O.; Oliveira, T.M.B.F.; Martínez-Huitle, C.A.; Motheo, A.J.; Albuquerque, C.C.; Castro, S.S.L. Performance of (in)active anodic materials for the electrooxidation of phenolic wastewaters from cashew-nut processing industry. Chemosphere 2018, 201, 740–748. [Google Scholar] [CrossRef]
- Da Costa, P.R.F.; Costa, E.C.T.A.; Castro, S.S.L.; Fajardo, A.S.; Martínez-Huitle, C.A. A sequential process to treat a cashew-nut effluent: Electrocoagulation plus electrochemical oxidation. J. Electroanal. Chem. 2019, 834, 79–85. [Google Scholar] [CrossRef]
- Bensalah, N.; Abdel-Wahab, A. Electrochemical treatment of synthetic and actual dyeing wastewaters using BDD anodes. Air Soil Water Res. 2010, 3, 45–56. [Google Scholar] [CrossRef]
- Eaton, A.; Clesceri, L.; Rice, E.; Greenberg, A.; Franson, M.A. Standard Methods for Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Fernandes, A.; Pacheco, M.J.; Ciríaco, L.; Lopes, A. Anodic oxidation of a biologically treated leachate on a boron-doped diamond anode. J. Hazard. Mater. 2012, 199–200, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Huitle, C.A.; Panizza, M. Electrochemical oxidation of organic pollutants for wastewater treatment. Curr. Opin. Electrochem. 2018, 11, 62–71. [Google Scholar] [CrossRef]
- Fernandes, A.; Santos, D.; Pacheco, M.J.; Ciríaco, L.; Simões, R.; Gomes, A.C.; Lopes, A. Electrochemical treatment of cork boiling wastewater with a boron-doped diamond anode. Environ. Technol. 2015, 36, 26–35. [Google Scholar] [CrossRef]
- Fernandes, A.; Pereira, C.; Kozioł, V.; Pacheco, M.J.; Ciríaco, L.; Lopes, A. Emerging contaminants removal from effluents with complex matrices by electrooxidation. Sci. Total Environ. 2020, 740, 140153. [Google Scholar] [CrossRef]
- Brillas, E.; Martínez-Huitle, C.A. Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review. Appl. Catal. B Environ. 2014, 166–167, 603–643. [Google Scholar] [CrossRef]
- Mazellier, P.; Leroy, É.; De Laat, J.; Legube, B. Degradation of carbendazim by UV/H2O2 investigated by kinetic modelling. Environ. Chem. Lett. 2003, 1, 68–72. [Google Scholar] [CrossRef]
- Lacasa, E.; Llanos, J.; Cañizares, P.; Rodrigo, M.A. Electrochemical denitrificacion with chlorides using DSA and BDD anodes. Chem. Eng. J. 2012, 184, 66–71. [Google Scholar] [CrossRef]
- Caliari, P.C.; Pacheco, M.J.; Ciríaco, L.; Lopes, A. Tannery wastewater: Organic load and sulfide removal dynamics by electrochemical oxidation at different anode materials. Environ. Technol. Innov. 2019, 14, 100345. [Google Scholar] [CrossRef]



| Type of Effluent | Anode Material | Supporting Electrolyte | COD0 /mg L−1 | pH0 | Applied Current Intensity, Current Density or Voltage | Volume/L | Recirculation Flow Rate /L h−1 | Electrolysis Time /h | COD Removal/% | Energy Consumption | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Olive mill | Boron-doped diamond (BDD) | -- | 700 | 7.13 | 300 A m−2 | 0.6 | 150 | (1) | ≈100 (2) | 300 kWh m−3 (2) | [15] |
| wastewater | NaCl | 230 kWh m−3 (2) | |||||||||
| Na2SO4 | 100 kWh m−3 | ||||||||||
| -- | 9350 | (1) | ≈4286 A m−2 (2) | 10 | 1200 | 14 | 73 | 174 Wh gCOD−1 | [16] | ||
| Na2SO4 | 40,000 | 4.4 | ≈2857 A m−2 (2) | 10 | 600 | 15 | 19 | 96 Wh gCOD−1 | [17] | ||
| Na2SO4 | 15,178 | 5.76 | 200 A m−2 | 0.2 | 54 | 7 | 20.2 | 24 Wh gCOD−1 | [18] | ||
| 15,315 | 300 A m−2 | 21.1 | 30 Wh gCOD−1 | ||||||||
| Ti/Pt | NaCl | 250,000 (2) | 5.2 | 2600 A m−2 (2) | 15 | 40 | 10 | 93 | 12.3 Wh gCOD−1 | [19] | |
| -- | 42,000 | 5.2 | 350 A m−2 (2) | 0.3 | -- | 5 | 40 | (1) | [20] | ||
| NaCl | 234,000 | 5.1 | 3125 A m−2 (2) | 10 | 3600 | 72 | ≈50 | 246 Wh gPhenols−1 | [21] | ||
| -- | 65,000 | 5.27 | 350 A m−2 | 0.5 | 0.12 | 10 | 55 | (1) | [22] | ||
| -- | ≈50,000 | 4.6 | 76.9 A m−2 | 0.8 | -- | 24 | 86 | (1) | [23] | ||
| ≈10,000 | 5 | 26 | |||||||||
| 2 | 41 | ||||||||||
| 8 | 32 | ||||||||||
| Na2SO4 | 4.6 | 54 | |||||||||
| KCl | 100 | ||||||||||
| NaCl | 100 | ||||||||||
| Ti/Ta/Pt/Ir | NaCl | 1475 | (1) | 5 V | 10 | 2232 | 2 | 5 | 82.1 Wh gCOD−1 | [24] | |
| 7 V | 25 | 56.1 Wh gCOD−1 | |||||||||
| 9 V | 35 | 76.9 Wh gCOD−1 | |||||||||
| 3060 | 25 | 27.1 Wh gCOD−1 | |||||||||
| 5180 | 15 | 28.3 Wh gCOD−1 | |||||||||
| NaCl | 18,100 | 5.5 | 14 V | 45 | 15 | 8 | 58.9 | (1) | [25] | ||
| 16 V | 70.8 | ||||||||||
| 18 V | 63.4 | ||||||||||
| Na2SO4 | 38,100 | (1) | 20 | 7 | |||||||
| Na2SO4 + FeCl3 | 20,200 | 8.5 | 16 V | 7 | 39.1 | ||||||
| 20 V | 45.2 | ||||||||||
| 24 V | 60.2 | ||||||||||
| Ti/RuO2 | NaCl | 41,000 | 4.57 | 1350 A m−2 | 0.4 | 28.44 | 7 | 99.6 | 21.2 Wh gCOD−1 (2) | [26] | |
| HClO4 | 1220 | (1) | 500 A m−2 | 0.11 | -- | 5 | 52 | 32.5 Wh gCOD−1 (2) | [27] | ||
| HClO4 + NaCl | 150 A m−2 | 54 | 3.5 Wh gCOD−1 (2) | ||||||||
| HClO4 + FeCl3 | 39 | 7 Wh gCOD−1 (2) | |||||||||
| HClO4 + Na2SO4 | 31 (2) | 7.5 Wh gCOD−1 (2) | |||||||||
| Ti/TiRuO2 | -- | 26,750 | 5.0 | ≈617 A m−2 (2) | 0.4 | 180 | 33 (2) | ≈89 (2) | (1) | [28] | |
| NaCl | 21 (2) | ≈96 (2) | 800 kWh m−3 | ||||||||
| Ti/IrO2 | HClO4 | 1300 | (1) | 500 A m−2 | 0.11 | -- | 20 (2) | 60 | (1) | [29] | |
| HClO4 + NaCl | 7.5 | ≈20 (2) | 72 Wh gCOD−1 | ||||||||
| IrO2 based Dimensionally Stable Anode (DSA) | KNO3 | 1200 | (1) | 153 A m−2 | 0.03 | -- | ≈48 (2) | 16 | (1) | [30] | |
| RuO2 based DSA | 1100 | 69 A m−2 | 99 | ||||||||
| Canola-oil | BDD | NaCl | 1750 (2) | (1) | 9.1 A m−2 | 1 | -- | 7 | 23 | (1) | [31] |
| refinery | 91 A m−2 | 96 | |||||||||
| wastewater | 136.6 A m−2 | 96 | |||||||||
| Dairy | Pt/Ti | NaCl | 4000 | (1) | 2000 A m−2 | 0.15 | -- | 4 | ≈100 (2) | (1) | [32] |
| wastewater | Pt-IrO2/Ti | ≈78 (2) | |||||||||
| IrO2/Ti | ≈85 (2) | ||||||||||
| 15,000 | 1000 A m−2 | 6 | 47 | ||||||||
| 3750 | 89 | ||||||||||
| Na2SO4 | 57.7 | ||||||||||
| BDD | -- | 3350 | 2.17 | 357 A m−2 | 1 | 194.4 | 6 | 88 | 137 Wh gCOD−1 (2) | [33] | |
| Slaughter- house | BDD | -- | 2280 | 6.68 | 357 A m−2 | 1 | 194.4 | 3 | 85 | 100 Wh gCOD−1 (2) | [11] |
| wastewater | BDD | -- | 2366 | 6.3 | 357 A m−2 | 1 | 194.4 | 6 | 97 | 163 Wh gCOD−1 (2) | [33] |
| Winery | BDD | -- | 3490 | 5.6 | 600 A m−2 | 0.1 | -- | 7 | 70 | (1) | [34] |
| wastewater | Na2SO4 | 6 | ≈100 (2) | 96 kWh m−3 | |||||||
| NaCl | 6 | ≈100 (2) | (1) | ||||||||
| Distillery | PbO2-Ti | -- | ≈13,500 | ≈7.1 | 150 A m−2 | 0.2 | -- | 8 | 56.3 | 2.23 Wh gCOD−1 | [35] |
| wastewater | NaCl | 90.8 | 1.31 Wh gCOD−1 | ||||||||
| RuO2-Ti | -- | 62.0 | 1.91 Wh gCOD−1 | ||||||||
| NaCl | 92.1 | 1.19 Wh gCOD−1 | |||||||||
| Graphite | 80.6 | 1.13 Wh gCOD−1 | |||||||||
| Ti sponge | -- | ≈9200 | 1 | 9 A | 15 | 900 | 6 | 51.50 | 5.23 Wh gCOD−1 | [36] | |
| H2O2 | 67.71 | 3.71 Wh gCOD−1 | |||||||||
| NaCl | 89.62 | 2.82 Wh gCOD−1 | |||||||||
| BDD | -- | 3241 | 12.5 | 300 A m−2 | 0.1 | 78 | 4 | 71.9 | 22.8 Wh gCOD−1 | [37] | |
| 216 | 81.5 | 20.1 Wh gCOD−1 | |||||||||
| 400 A m−2 | 78 | 75.7 | 36.8 Wh gCOD−1 | ||||||||
| 216 | 88.3 | 30.0 Wh gCOD−1 | |||||||||
| BDD | Na2SO4 | 12,647 | 10.4 | 600 A m−2 | 25 | 4500 | 7 (2) | 81.3 | 173.19 kWh m−3 | [38] | |
| NaCl | 65.7 | 184.45 kWh m−3 | |||||||||
| Brewery | Graphite | -- | 2470 | 4.5 | 120 A m−2 | 24 | 180 | 0.83 | 18 | (1) | [39] |
| wastewater | NaCl | 35 | |||||||||
| 372 A m−2 | 93 (2) | ||||||||||
| 745 A m−2 | 97 (2) | ||||||||||
| BDD | -- | 1877 | 3.0 | 80 A m−2 | 0.45 | -- | 6 | 65 | ≈22 kWh m−3 (2) | [14] | |
| 160 A m−2 | 91 | ≈55 kWh m−3 (2) | |||||||||
| 240 A m−2 | 98 | ≈125 kWh m−3 (2) | |||||||||
| Starch | RuO2/Ti | NaCl | (1) | (1) | 750 A m−2 | 0.2 | -- | 4.2 | 75.8 | 23.4 Wh gCOD−1 | [40] |
| wastewater | PbO2/Ti | 50.5 | 44.8 Wh gCOD−1 | ||||||||
| Sugar beet wastewater | BDD | -- | 15,673 | 5 | 491 A m−2 | 1 | -- | 4.9 | 75 | 28.43 kWh m−3 | [41] |
| Coke-plant | DSA | -- | 2143 | 7.2 | (1) | 0.6 | -- | 4 | 57.80 | (1) | [42] |
| wastewater | Sn-Pd-Ru oxide coated titanium | 59.24 | |||||||||
| PbO2/Ti | 66.51 | ||||||||||
| NaCl | 250 A m−2 | 12 | 64.8 | ||||||||
| 750 A m−2 | 4 | 67.4 | |||||||||
| 1500 A m−2 | 2 | 75.2 | |||||||||
| Cashew-nut wastewater | Ti/RuO2-TiO2 | -- | 1540 | (1) | 1000 A m−2 | 0.03 | -- | 6 | 72 | (1) | [43] |
| BDD | 100 | (1) | |||||||||
| BDD | -- | 212 | 9.5 | 355 A m−2 | 0.35 | -- | 2 | 60 (2) | (1) | [44] |
| Parameter | Mean Value (±SD 1) | ||
|---|---|---|---|
| Olive Mill Wastewater | Slaughterhouse Wastewater | Winery Wastewater | |
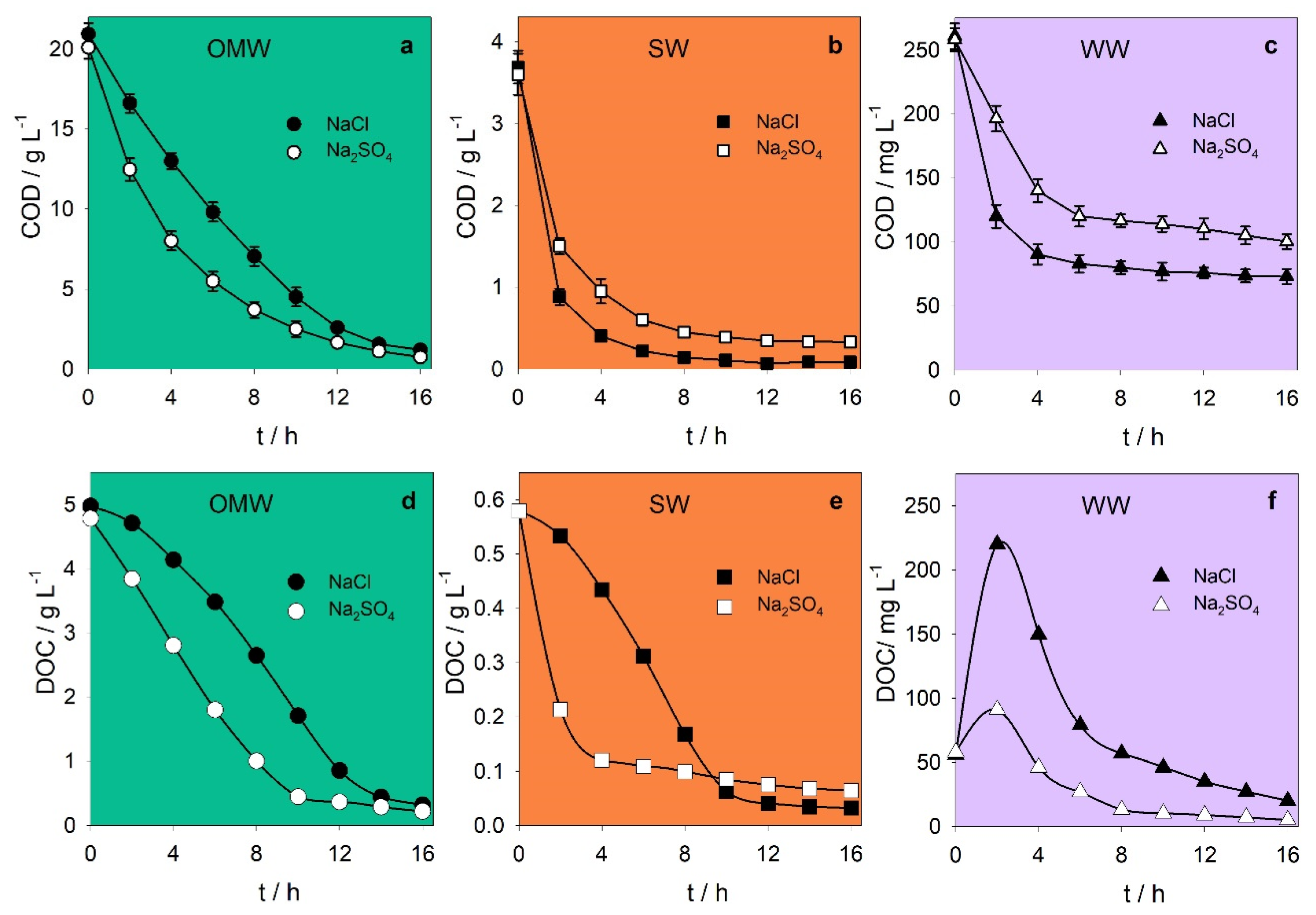
| Chemical oxygen demand/g L−1 | 20.5 ± 0.6 | 3.64 ± 0.06 | 0.259 ± 0.002 |
| Total dissolved carbon/g L−1 | 4.9 ± 0.1 | 0.677 ± 0.003 | 0.279 ± 0.007 |
| Dissolved organic carbon/g L−1 | 4.9 ± 0.1 | 0.579 ± 0.001 | 0.057 ± 0.001 |
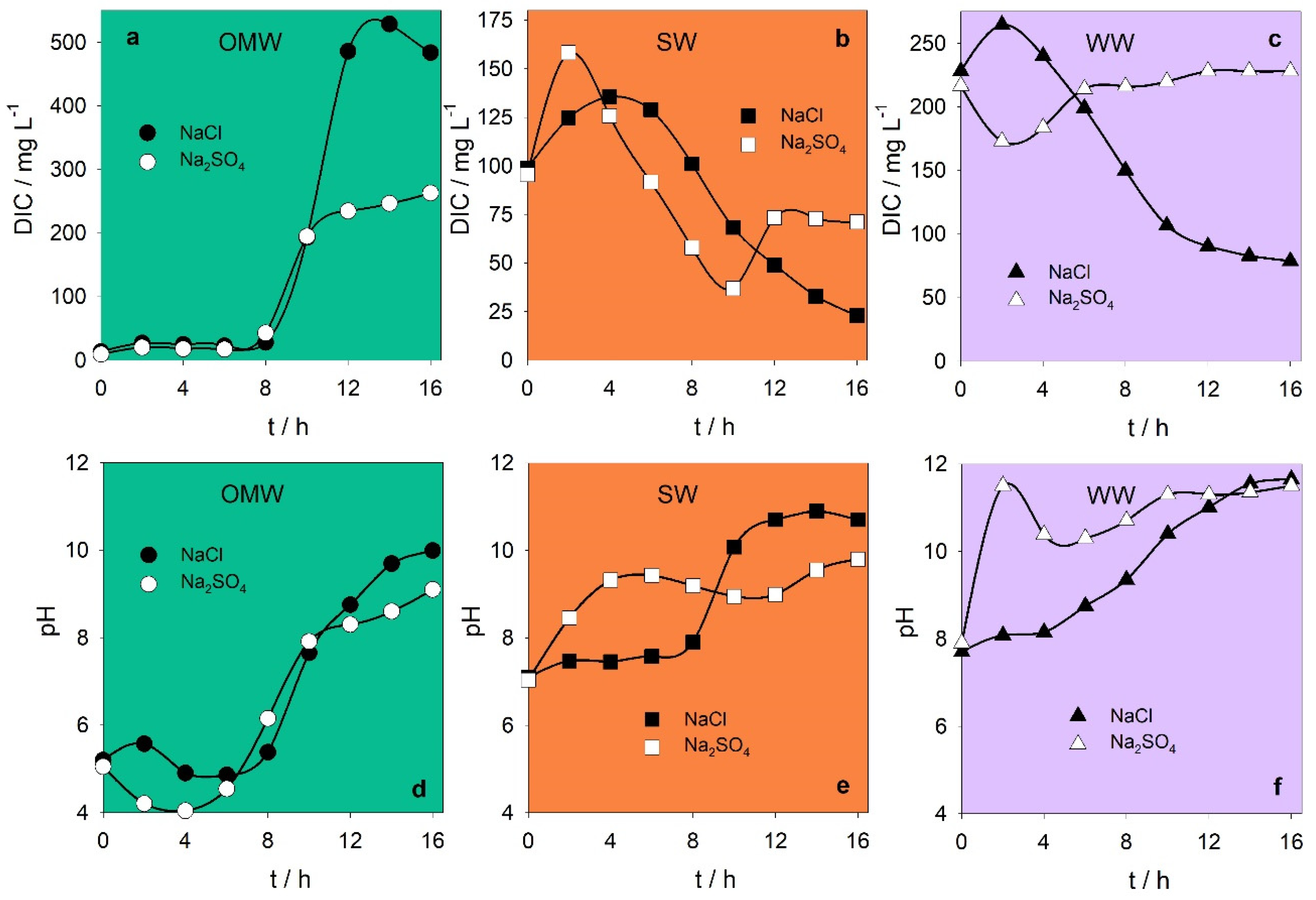
| Dissolved inorganic carbon/mg L−1 | 11 ± 3 | 97 ± 2 | 222 ± 8 |
| Total dissolved nitrogen/mg L−1 | 50 ± 1 | 129 ± 2 | 5 ± 1 |
| pH | 5.1 ± 0.1 | 7.06 ± 0.04 | 7.8 ± 0.1 |
| Conductivity/mS cm−1 | 1.0 ± 0.1 | 0.9 ± 0.1 | 0.6 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Cruz, A.; Fernandes, A.; Ciríaco, L.; Pacheco, M.J.; Carvalho, F.; Afonso, A.; Madeira, L.; Luz, S.; Lopes, A. Electrochemical Oxidation of Effluents from Food Processing Industries: A Short Review and a Case-Study. Water 2020, 12, 3546. https://doi.org/10.3390/w12123546
Martínez-Cruz A, Fernandes A, Ciríaco L, Pacheco MJ, Carvalho F, Afonso A, Madeira L, Luz S, Lopes A. Electrochemical Oxidation of Effluents from Food Processing Industries: A Short Review and a Case-Study. Water. 2020; 12(12):3546. https://doi.org/10.3390/w12123546
Chicago/Turabian StyleMartínez-Cruz, Alfredo, Annabel Fernandes, Lurdes Ciríaco, Maria José Pacheco, Fátima Carvalho, Alexandra Afonso, Luís Madeira, Silvana Luz, and Ana Lopes. 2020. "Electrochemical Oxidation of Effluents from Food Processing Industries: A Short Review and a Case-Study" Water 12, no. 12: 3546. https://doi.org/10.3390/w12123546








