Quality and Health Risk Assessment Associated with Water Consumption—A Case Study on Karstic Springs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Description of the Study Area
2.2. Field Sampling and Analytical Procedures
2.3. Data Treatment and Statistical Analysis
2.3.1. Water Quality Index (WQI)
2.3.2. Heavy Metal Pollution Index (HPI) and Heavy Metal Evaluation Index (HEI)
2.3.3. Human Health Risk Assessment
3. Results and Discussion
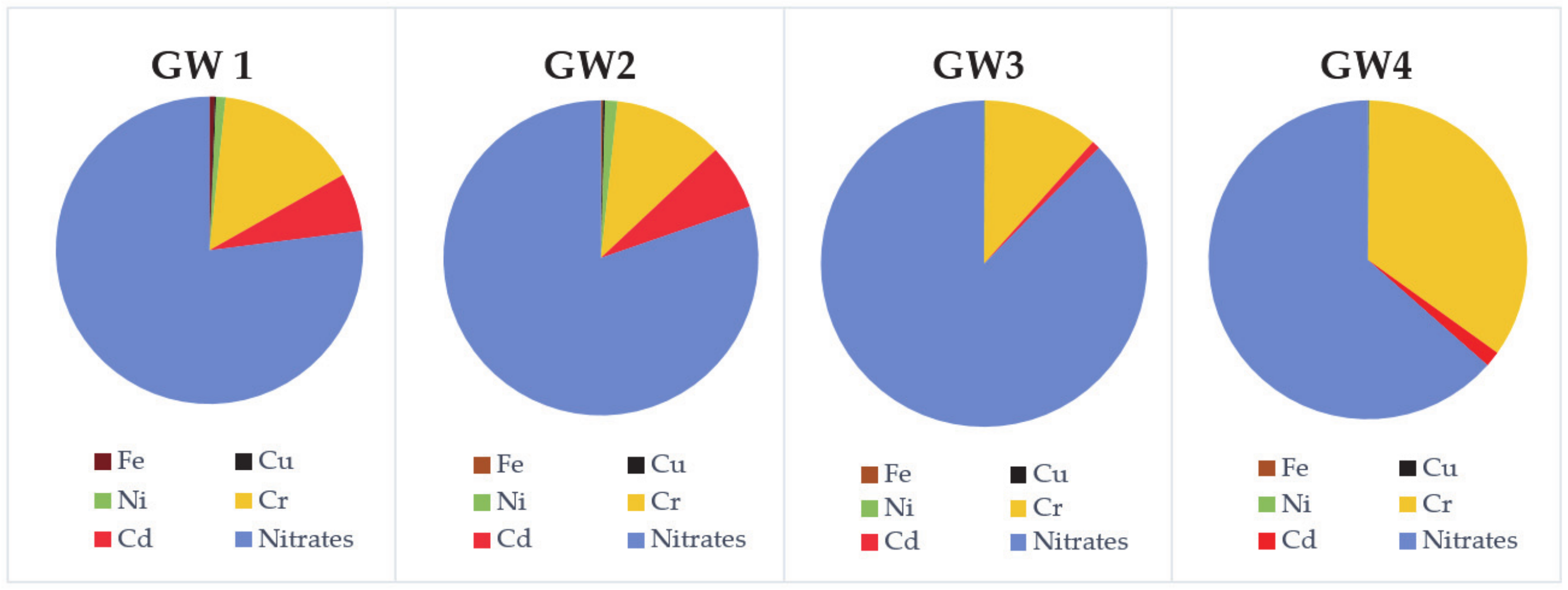
3.1. Physico-Chemical Parameters of the Spring’s Water
3.2. Water Quality Indices
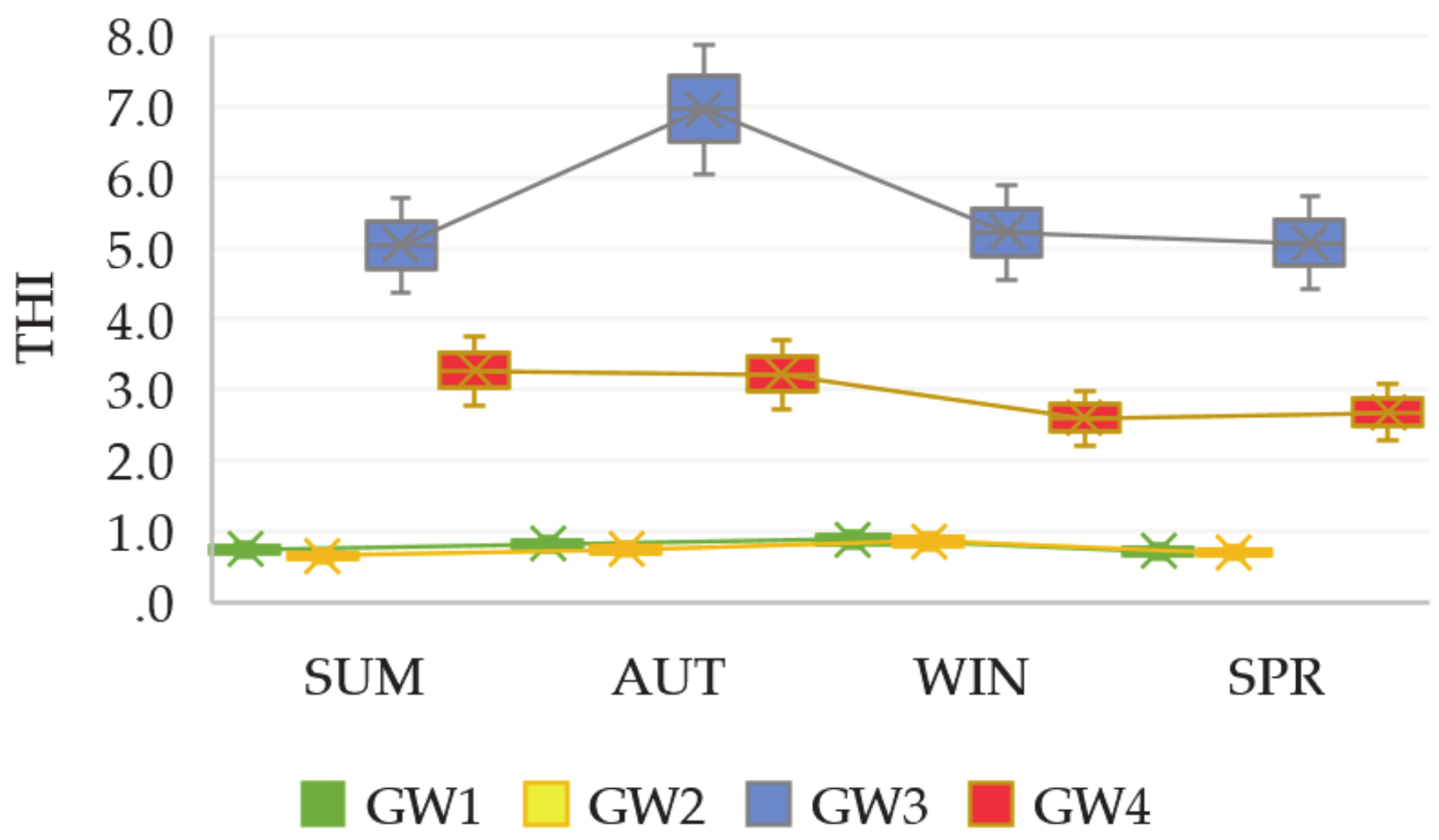
3.3. Exposures and Health Risk Assessments
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Adams, B.; Foster, S.S.D. Land-surface zoning for groundwater protection. Water Environ. J. 1992, 6, 312–319. [Google Scholar] [CrossRef]
- Doerfliger, N.; Jeannin, P.; Zwahlen, F. Water vulnerability assessment in karst environments: A new method of defining protection areas using a multi-attribute approach and GIS tools (EPIK method). Environ. Geol. 1999, 39, 165–176. [Google Scholar] [CrossRef] [Green Version]
- Kazakis, N.; Voudouris, K. Groundwater vulnerability and pollution risk assessment of porous aquifers to nitrate: Modifying the DRASTIC method using quantitative parameters. J. Hydrol. 2015, 525, 13–25. [Google Scholar] [CrossRef]
- Wu, J.; Sun, Z. Evaluation of shallow groundwater contamination and associated human health risk in an alluvial plain impacted by agricultural and industrial activities, Mid-west China. Expo. Health 2016, 8, 311–329. [Google Scholar] [CrossRef]
- Gamvroula, D.; Alexakis, D.; Stamatis, G. Diagnosis of groundwater quality and assessment of contamination sources in the Megara basin (Attica, Greece). Arab. J. Geosci. 2013, 6, 2367–2381. [Google Scholar] [CrossRef]
- Alexakis, D.; Tsakiris, G. Drought impacts on karstic spring annual water potential. Application on Almyros (Heraklion Crete) brackish spring. Desalin. Water Treat. 2010, 16, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Feng, J.; Sun, H.; He, M.; Gao, Z.; Liu, J.; Wu, X.; An, Y. Quality Assessments of Shallow Groundwaters for Drinking and Irrigation Purposes: Insights from a Case Study (Jinta Basin, Heihe Drainage Area, Northwest China). Water 2020, 12, 2704. [Google Scholar] [CrossRef]
- Vespasiano, G.; Cianflone, G.; Cannata, C.B.; Apollaro, C.; Dominici, R.; De Rosa, R. Analysis of groundwater pollution in the Sant’Eufemia Plain (Calabria—South Italy). Ital. J. Eng. Geol. Environ. 2016, 2. [Google Scholar] [CrossRef]
- Vespasiano, G.; Cianflone, G.; Romanazzi, A.; Apollaro, C.; Dominici, R.; Polemio, M.; De Rosa, R. A multidisciplinary approach for sustainable management of a complex coastal plain: The case of Sibari Plain (Southern Italy). Mar. Pet. Geol. 2019, 109, 740–759. [Google Scholar] [CrossRef]
- Vespasiano, G.; Notaro, P.; Cianflone, G. Water-mortar interaction in a tunnel located in southern Calabria (Southern Italy). Environ. Eng. Geosci. 2018, 24, 305–315. [Google Scholar] [CrossRef]
- Wagh, V.M.; Panaskar, D.B.; Mukate, S.V.; Gaikwad, S.K.; Muley, A.A.; Varade, A.M. Health risk assessment of heavy metal contamination in groundwater of Kadava River Basin, Nashik, India. Model Earth Syst. Environ. 2018, 4, 959–980. [Google Scholar] [CrossRef]
- Cadar, O.; Miclean, M.; Cadar, S.; Tanaselia, C.; Senila, L.; Senila, M. Assessment of heavy metals in cow’s milk Rodnei mountains area, Romania. Environ. Eng. Manag. J. 2015, 14, 2523–2528. [Google Scholar] [CrossRef]
- Ayotte, J.D.; Szabo, Z.; Focazio, M.J.; Eberts, S.M. Effects of human-induced alteration of groundwater flow on concentrations of naturally-occurring trace elements at water-supply wells. Appl. Geochem. 2011, 26, 747–762. [Google Scholar] [CrossRef] [Green Version]
- Nyambura, C.; Hasim, N.O.; Chege, M.W.; Tokonami, S.; Omonya, F.W. Cancer and non-cancer health risks from carcinogenic heavy metal exposures in underground water from Kilimambogo, Kenya. Groundw. Sustain. Dev. 2020, 10, 100315. [Google Scholar] [CrossRef]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, Mechanism and Health Effects of Some Heavy Metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, S.; Shah, M.T.; Khan, S. Health risk assessment of heavy metals and their source apportionment in drinking water of Kohistan region, northern Pakistan. Microchem. J. 2011, 98, 334–343. [Google Scholar] [CrossRef]
- Cao, X.; Lu, L.; Wang, C.; Zhang, M.; Yuan, J.; Zhang, A.; Song, S.; Baninla, Y.; Khan, K.; Wang, Y. Hydrogeochemistry and quality of surface water and groundwater in the drinking water source area of an urbanizing region. Ecotoxi. Environ. Safe 2019, 186, 109628. [Google Scholar] [CrossRef]
- Hossain, M.; Patra, P.K. Contamination zoning and health risk assessment of trace elements in groundwater through geostatistical modelling. Ecotoxicol. Environ. Saf. 2020, 189, 110038. [Google Scholar] [CrossRef]
- Chowdhury, H.; Mazumder, M.A.J.; Al-Attas, O.; Husain, T. Heavy metals in drinking water: Occurrences, implications, and future needs in developing countries. Sci. Total Environ. 2016, 569, 476–488. [Google Scholar] [CrossRef]
- Deepanjau, M.; Navindu, C. Nitrate pollution of groundwater and associated human health disorders. Indian J. Environ. Health 2000, 42, 28–39. [Google Scholar]
- Saha, N.; Rahman, M.S.; Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W. Industrial metal pollution in water and probabilistic assessment of human health risk. J. Environ. Manag. 2017, 10, 100315. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, A.; Hassani, H.; Hayati, M.; Jabbari, N.; Barzegar, R. Risk assessment and ranking of heavy metals concentration in Iran’s Rayen groundwater basin using linear assignment method. Stoch. Environ. Res. Risk Assess. 2017, 32, 1317–1336. [Google Scholar] [CrossRef]
- Dippong, T.; Mihali, C.; Hoaghia, M.A.; Cical, E.; Cosma, A. Chemical modeling of groundwater quality in the aquifer of Seini town—Someș Plain, Northwestern Romania. Ecotoxicol. Environ. Saf. 2019, 168, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Islam, A.R.M.T.; Siddiqua, M.T.; Zahid, A.; Tasnim, S.S.; Rahman, M.M. Drinking appraisal of coastal groundwater in Bangladesh: An approach of multi-hazards towards water security and health safety. Chemosphere 2020, 255, 126933. [Google Scholar] [CrossRef]
- National Institute of Statistics 2018. Water Distribution and Wastewater Disposal. 2018. Available online: https://insse.ro/cms/sites/default/files/field/publicatii/distributia_apei_si_evacuarea_apelor_uzate_in_anul_2018_0.pdf (accessed on 30 September 2020).
- Water Framework Directive, Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 Establishing a Framework for Community Action in the Field of Water Policy, Official Journal of the European Communities. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32000L0060 (accessed on 30 September 2020).
- Groundwater Directive, Directive 2006/118/CE on the European Parliament and of the Council of 12 December 2006, on the Protection of Groundwater against Pollution and Deterioration, Official Journal of the European Communities. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:372:0019:0031:EN:PDF (accessed on 30 September 2020).
- Council Directive 98/83/EC of 3 November 1998 on the Quality of Water Intended for Human Consumption. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:31998L0083&from=EN (accessed on 30 September 2020).
- Onac, B.P.; Goran, C. Karst and Caves of Romania: A Brief Overview. In Caves and Karst Systems of Romania; Ponta, G.M.L., Onac, B.P., Eds.; Springer: Cham, Switzerland, 2019; pp. 21–35. [Google Scholar]
- Orășeanu, I. Hydrogeological regional classification of the Romanian karst. Theor. Appl. Karst. 1993, 6, 7–86. [Google Scholar]
- Țenu, A.; Davidescu, F.; Eichinger, L.; Voerkelius, S. Quality evaluation of groundwaters in Southern Dobrogea (Romania). Theor. Appl. Karst. 1997, 10, 63–77. [Google Scholar]
- Ghenea, C.; Bandrabur, T.; Ghenea, A. Atlas of Romania: The Underground and Mineral Waters Map; Sheet V-2; Romanian Academy, Institute of Geography: Bucharest, Romania, 1981. [Google Scholar]
- Moldovan, O.T.; Baricz, A.; Szekeres, E.; Kenesz, M.; Hoaghia, M.A.; Levei, E.A.; Mirea, I.C.; Năstase-Bucur, R.; Brad, T.; Chiciudean, I.; et al. Testing Different Membrane Filters for 16S rRNA Gene-Based Metabarcoding in Karstic Springs. Water 2020, 12, 3400. [Google Scholar] [CrossRef]
- Bandoc, G.; Prăvălie, R. Climatic water balance dynamics over the last five decades in Romania’s most arid region, Dobrogea. J. Geogr. Sci. 2015, 25, 1307–1327. [Google Scholar] [CrossRef]
- Klein Tank, A.M.G.; Wijngaard, J.B.; Können, G.P.; Böhm, R.; Demarée, G.; Gocheva, A.; Mileta, M.; Pashiardis, S.; Hejkrlik, L.; Kern-Hansen, C.; et al. Daily dataset of 20th-century surface air temperature and precipitation series for the European Climate Assessment. Int. J. Climatol. 2002, 22, 1441–1453. [Google Scholar] [CrossRef]
- Beck, H.; Zimmermann, N.; McVicar, T. Present and future Köppen-Geiger climate classification maps at 1-km resolution. Sci. Data 2018, 5, 180214. [Google Scholar] [CrossRef] [Green Version]
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 2007, 11, 1633–1644. [Google Scholar] [CrossRef] [Green Version]
- © European Union, Copernicus Land Monitoring Service 2020, European Environment Agency (EEA). Available online: https://land.copernicus.eu/ (accessed on 3 December 2020).
- Sener, S.; Sener, E.; Davraz, A. Evaluation of water quality using water quality index (WQI) method and GIS in Aksu River (SW-Turkey). Sci. Total Environ. 2017, 584, 131–144. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; Incorporating first addendum; World Health Organization: Geneva, Switzerland, 2017; Available online: https://www.who.int/publications/i/item/9789241549950 (accessed on 30 September 2020).
- Singh, D.D.; Thind, P.S.; Sharma, M.; Sahoo, S.; John, S. Environmentally sensitive elements in groundwater of an industrial town in India: Spatial distribution and human health risk. Water 2019, 11, 2350. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M.; Nagdev, R.; Tripathi, R.; Singh, V.B.; Ranjan, P.; Soheb, M.; Ramanathan, A.L. Geospatial and multivariate analysis of trace metals in tubewell water using for drinking purpose in the upper Gangetic basin, India: Heavy metal pollution index. Groundw. Sustain. Dev. 2019, 8, 122–133. [Google Scholar] [CrossRef]
- Rezaei, A.; Hassani, H.; Hassani, S.; Jabbari, N.; Far, S.B.; Rezaei, M.S. Evaluation of groundwater quality and heavy metal pollution indices in Bazman basin, southeastern Iran. Groundw. Sustain. Dev. 2019, 9, 100245. [Google Scholar] [CrossRef]
- Bodrud-Doza, M.; Islam, S.M.D.; Rume, T.; Quraishi, S.B.; Rahman, M.S.; Bhuiyan, M.A.H. Groundwater quality and human health risk assessment for safe and sustainable water supply of Dhaka City dwellers in Bangladesh. Groundw. Sustain. Dev. 2020, 10, 100374. [Google Scholar] [CrossRef]
- Giri, S.; Singh, A.K. Human health risk assessment via drinking water pathway due to metal contamination in the groundwater of Subarnarekha River Basin, India. Environ. Monit. Assess. 2015, 187, 63. [Google Scholar] [CrossRef]
- Mukherjee, I.; Singh, U.M.; Singh, R.P.; Kumari, D.; Jha, P.K.; Mehta, P. Characterization of heavy metal pollution in an anthropogenically and geologically influenced semi-arid region of east India and assessment of ecological and human health risks. Sci. Total Environ. 2020, 705, 135801. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. USEPA Baseline Human Health Risk Assessment Vasquez Boulevard and I-70 Superfund Site Denver CO. 2001. Available online: https://hero.epa.gov/hero/index.cfm/reference/details/reference_id/786143 (accessed on 30 September 2020).
- USEPA. Risk Assessment Guidance for Superfund Volume I: Human Health Evaluation Manual (Part E). 2004. Available online: https://www.epa.gov/sites/production/files/2015-09/documents/part_e_final_revision_10-03-07.pdf (accessed on 30 September 2020).
- Mohammadi, A.A.; Zarei, A.; Majidi, S.; Ghaderpoury, A.; Hashempur, Y.; Saghi, M.H.; Alinejad, A.; Yousefi, M.; Hosseingholizadeh, N.; Ghaderpoori, M. Carcinogenic and non-carcinogenic health risk assessment of heavy metals in drinking water of Khorramabad, Iran. MethodsX 2019, 6, 1642–1651. [Google Scholar] [CrossRef]
- Wu, B.; Zhang, Y.; Zhang, X.; Cheng, S. Health Risk from Exposure of Organic Pollutants Through Drinking Water Consumption in Nanjing, China. Bull. Environ. Contam. Toxicol. 2010, 84, 46–50. [Google Scholar] [CrossRef]
- Ahmed, A.N.; Othman, F.B.; Afan, H.A.; Ibrahim, R.K.; Fai, C.M.; Hossain, M.S.; Ehteram, M.; Elshafie, A. Machine learning methods for better water quality prediction. J. Hydrol. 2019, 578, 124084. [Google Scholar] [CrossRef]
- Mahler, B.J.; Bourgeais, R. Dissolved oxygen fluctuations in karst spring flow and implications for endemic species: Barton Springs, Edwards aquifer, Texas, USA. J. Hydrol. 2013, 505, 291–298. [Google Scholar] [CrossRef]
- Rajwa-Kuligiewicz, A.; Bialik, R.; Rowiński, P. Dissolved oxygen and water temperature dynamics in lowland rivers over various timescales. J. Hydrol. Hydromech. 2015, 63, 353–363. [Google Scholar] [CrossRef] [Green Version]
- Hassan, S.H.A.; Gurung, A.; Kang, W.; Shin, B.; Rahimnejad, M.; Jeon, B.; Kin, J.R.; Oh, S. Real-time monitoring of water quality of stream water using sulfur-oxidizing bacteria as bio-indicator. Chemosphere 2019, 223, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Jha, M.K.; Shekhar, A.; Jenifer, M.A. Assessing groundwater quality for drinking water supply using hybrid fuzzy-GIS-based water quality index. Water Res. 2020, 179, 115867. [Google Scholar] [CrossRef]
- Miyittah, M.K.; Tulashie, S.K.; Tsyawo, F.W.; Sarfo, J.K.; Darko, A.A. Assessment of surface water quality status of the Aby Lagoon System in the Western Region of Ghana. Heliyon 2020, 6, e04466. [Google Scholar] [CrossRef]
- Egbi, C.D.; Anornu, G.K.; Ganyaglo, S.Y.; Appiah-Adjei, E.K.; Li, S.L.; Dampare, S.B. Nitrate contamination of groundwater in the Lower Volta River Basin of Ghana: Sources and related human health risks. Ecotoxic. Environ. Safe 2020, 191, 110227. [Google Scholar] [CrossRef]
- Rout, C.; Sharma, A. Assessment of drinking water quality: A case study of Ambala cantonment area, Haryana, India. Int. J. Environ. Sci. 2011, 2, 2. [Google Scholar]
- Vetrimurugan, E.; Elango, L.; Rajmohan, N. Sources of contaminants and groundwater quality in the coastal part of a river delta. Int. J. Environ. Sci. Technol. 2013, 10, 473–486. [Google Scholar] [CrossRef] [Green Version]
- Moisa, C.; Cadar, O.; Barabas, R.; Vicas, L.G.; Hoaghia, M.A.; Levei, E.A.; Jurca, C.; Berce, C. Influence of magnesium compounds on sodium, potassium and calcium levels in different mice organs. Farmacia 2019, 67, 2. [Google Scholar] [CrossRef]
- Blaine, J.; Chonchol, M.; Levi, M. Renal control of calcium, phosphate, and magnesium homeostasis. Clin. J. Am. Soc. Nephrol. 2015, 10, 1257–1272. [Google Scholar] [CrossRef] [PubMed]
- Kozisek, F. Regulations for calcium, magnesium or hardness in drinking water in the European Union member states. Regul. Toxicol. Pharmacol. 2020, 112, 104589. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, P. Potential health impacts of hard water. Int. J. Prev. Med. 2013, 4, 866–875. [Google Scholar] [PubMed]
- Manoj, K.; Ghosh, S.; Padhy, P.K. Characterization and classification of hydrochemistry using graphical and hydrostatical techniques. Res. J. Chem. Sci. 2013, 3, 32–42. [Google Scholar]
- Hoaghia, M.A.; Cadar, O.; Hognogi, G.G.; Levei, E.; Moisa, C.; Roman, C. Quality and human health risk assessment if metals and nitrogen compounds in drinking water from an urban area near a former non-ferrous ore smelter. Anal. Lett. 2019, 52, 1268–1281. [Google Scholar] [CrossRef]
- Verma, P.; Singh, P.K.; Sinha, R.R.; Tiwari, A.K. Assessment of groundwater quality status by using water quality index (WQI) and geographic information system (GIS) approaches: A case study of the Bokaro district, India. Appl. Water Sci. 2020, 10, 27. [Google Scholar] [CrossRef] [Green Version]
- Adamou, H.; Ibrahim, B.; Salack, S.; Adamou, R.; Sanfo, S.; Liersch, S. Physico-chemical and bacteriological quality of groundwater in a rural area of Western Niger: A case study of Bonkoukou. J. Water Health 2020, 1, 18. [Google Scholar] [CrossRef]
- Baba, M.E.; Kayastha, P.; Huysmans, M.; Smedt, F.D. Evaluation of the groundwater quality using the water quality index and geostatistical analysis in the Dier al-Balah Governorate, Gaza Strip, Palestine. Water 2020, 12, 262. [Google Scholar] [CrossRef] [Green Version]
- Koc, C. Water quality index for measuring drinking water quality of Bodrum Peninsula—Turkey. Ohu J. Eng. Sci. 2018, 7, 694–702. [Google Scholar]
- Alexakis, D.E. Meta-evaluation of water quality indices. Application into groundwater resources. Water 2020, 12, 1890. [Google Scholar] [CrossRef]
- Zakhem, B.A.; Hafez, R. Heavy metal pollution index for groundwater quality assessment in Damascus Oasis, Syria. Environ. Earth Sci. 2015, 73, 6591–6600. [Google Scholar] [CrossRef]
- Boateng, T.K.; Opoku, F.; Akoto, O. Heavy metal contamination assessment of groundwater quality: A case study of Oti landfill site, Kumasi. Appl. Water Sci. 2019, 9, 33. [Google Scholar] [CrossRef] [Green Version]
- Chen, G.; Wang, X.; Wang, R.; Liu, G. Health risk assessment of potentially harmful elements in subsidence water bodies using a Monte Carlo approach: An example from the Huainan coal mining area, China. Ecotoxicol. Environ. Saf. 2019, 171, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Narsimha, A.; Veskatayogi, S.; Geeta, S. Hydrogeochemical data on groundwater quality with special emphasis on fluoride enrichment in Munneru river basin (MRB), Telangana State, South India. Data Brief 2018, 17, 339–346. [Google Scholar] [CrossRef]
- Tiwari, A.K.; De Maio, M. Assessment of risk to human health due to intake of chromium in the groundwater of the Aosta Valley region, Italy. Hum. Ecol. Risk Assess. 2017, 23, 1153–1163. [Google Scholar] [CrossRef]
- Marsidi, N.; Abu Hasan, H.; Abdullah, S.R.S. A review of biological aerated filters for iron and manganese ions removal in water treatment. J. Water Process. Eng. 2018, 23, 1–12. [Google Scholar] [CrossRef]
- Neag, E.; Moldovan, A.; Băbălău-Fuss, V.; Török, A.; Cadar, O.; Roman, C. Kinetic, Equilibrium and Phytotoxicity Studies for Dyes Removal by Low Cost Natural Activated Plant-Based Carbon. Acta Chim. Slov. 2019, 66, 850–858. [Google Scholar] [CrossRef]
- Faisal, A.A.H.; Sulaymon, A.H.; Khaliefa, Q.M. A review of permeable reactive barrier as passive sustainable technology for groundwater remediation. Int. J. Environ. Sci. Technol. 2018, 15, 1123–1138. [Google Scholar] [CrossRef]
- Kalhor, K.; Ghasemizadeh, R.; Rajic, L.; Alshawabkeh, K. Assessment of groundwater quality and remediation in karst aquifers: A review. Groundw. Sustain. Dev. 2019, 8, 104–121. [Google Scholar] [CrossRef]
- Moldovan, O.T.; Skoglund, R.O.; Banciu, H.L.; Dinu Cucos, A.; Levei, E.A.; Persoiu, A.; Lauritzan, S.E. Monitoring and risk assessment for groundwater sources in rural communities of Romania (GROUNDWATERISK). Res. Ideas Outcomes 2019, 5, e48898. [Google Scholar] [CrossRef] [Green Version]
- Sharma, A.S.K.; Sobti, R.C. Nitrate Removal from Ground Water: A Review. J. Chem. 2012, 9, 154616. [Google Scholar]
- Mohsenipour, M.; Shahid, S.; Ebrahimi, K. Removal Techniques of Nitrate from Water. Asian J. Chem. 2014, 26, 7881–7886. [Google Scholar] [CrossRef]
- Duan, S.; Tong, T.; Zheng, S.; Zhang, X.; Li, S. Achieving low-cost, highly selective nitrate removal with standard anion exchange resin by tuning recycled brine composition. Water Res. 2020, 173, 115571. [Google Scholar] [CrossRef] [PubMed]
- Labarca, F.; Borquaez, R. Comparative study of nanofiltration and ion exchange for nitrate reduction in the presence of chloride and iron in groundwater. Sci. Total Environ. 2020, 723, 137809. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.S.; Dahms, H.U.; Won, E.J.; Lee, J.S.; Shin, K.H. Microalgae—A promising tool for heavy metal remediation. Ecotoxic. Environ. Safe 2015, 113, 329–352. [Google Scholar] [CrossRef] [PubMed]
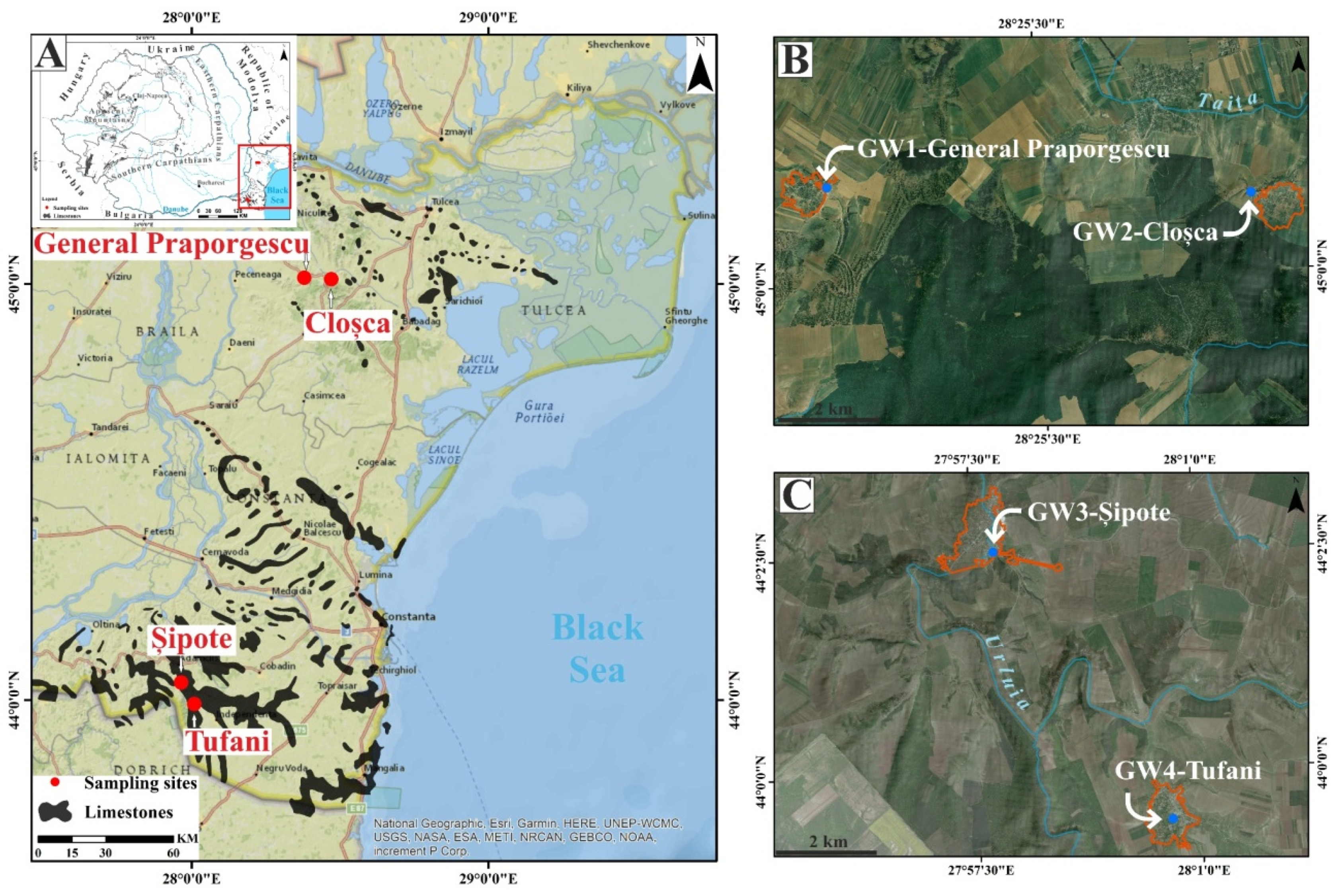
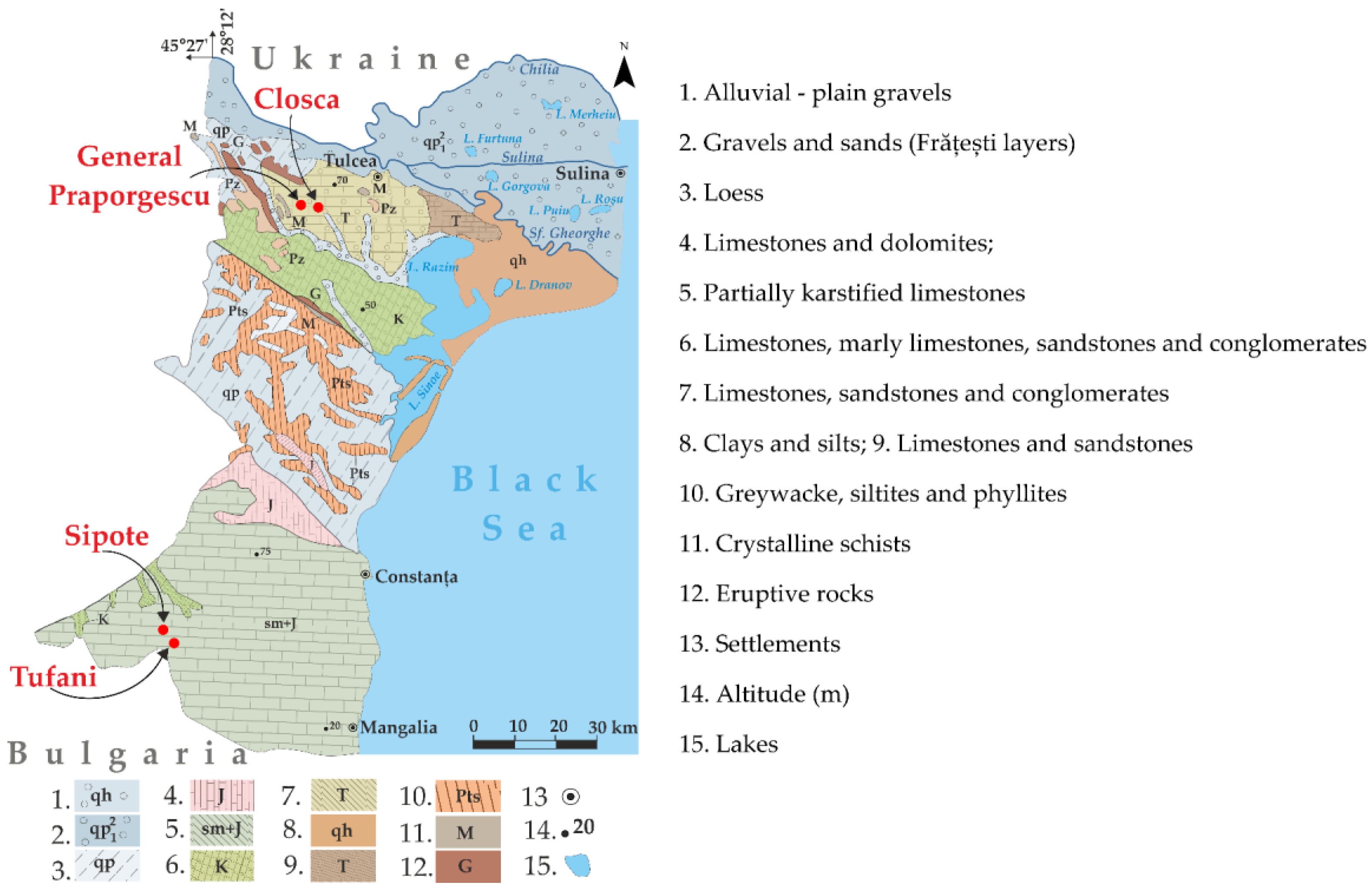
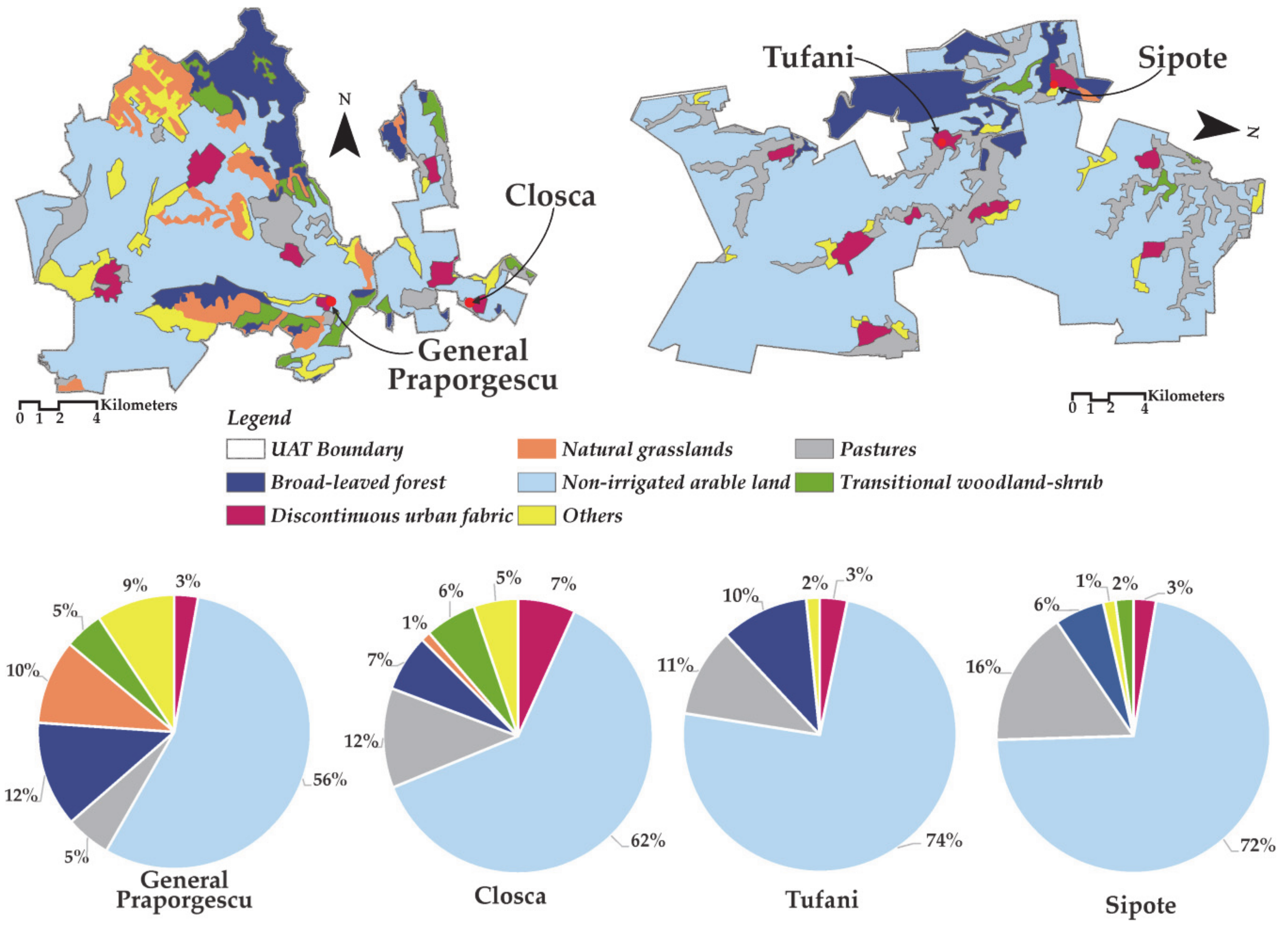
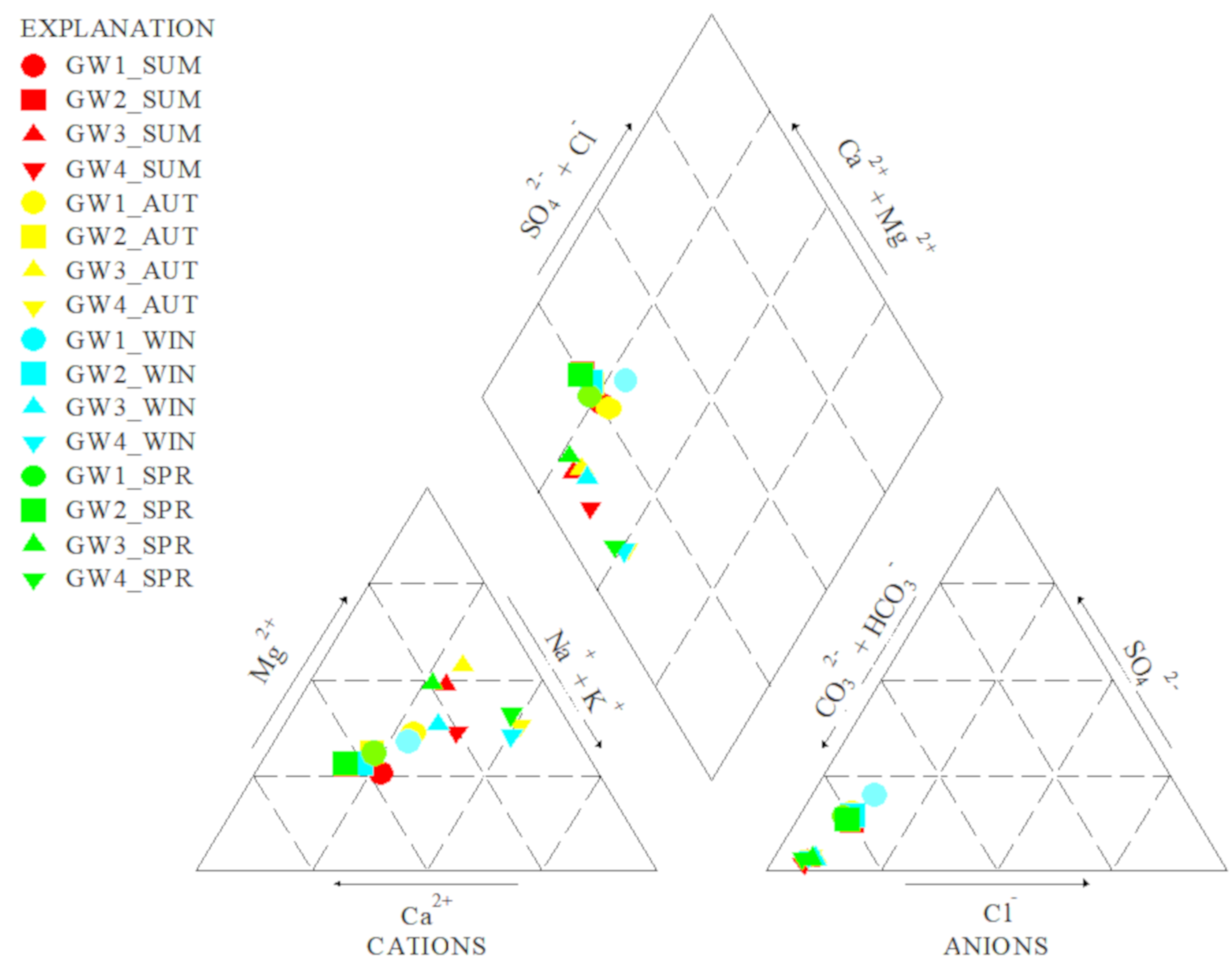
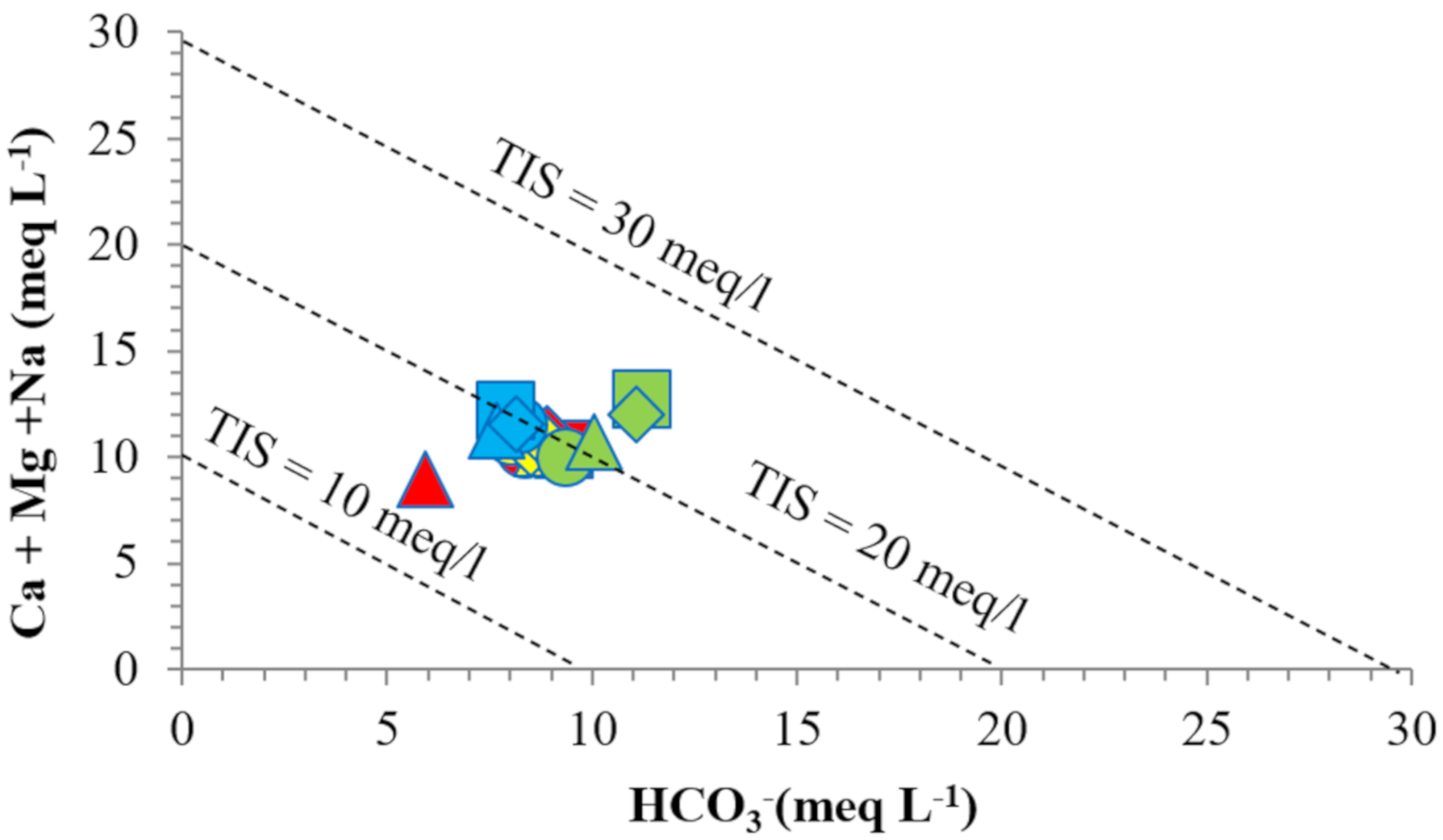
| Spring | GW1 | GW2 | GW3 | GW4 |
|---|---|---|---|---|
| Area | Northern Dobrogea | Southern Dobrogea | ||
| Altitude (m a.s.l.) | 139 | 88 | 57 | 78 |
| Geology | Sandstone, conglomerate limestones | Sandstone, limestone | Limestone, marl limestone, clays, dolomites | Lumashelic limestone, Nubeculoria limestone, clays, diatomites |
| Soil | Carbonate chernozems | Chernozems, alluvial soils | Rendzinas, rocks | Alluvial soils |
| Surface use | Arable land/forest/soft loam | Arable land/forest/soft loam | Arable land | Arable land/soft loam |
| No. of inhabitants | 181 | 101 | 566 | 362 |
| Water flow (L/min) | 42–120 | 120–200 | 2.3–21 | 6.0–12.5 |
| Temperature (°C) | 12.9–15.8 | 14.2–15.5 | 4.7–20.4 | 10.5–15.5 |
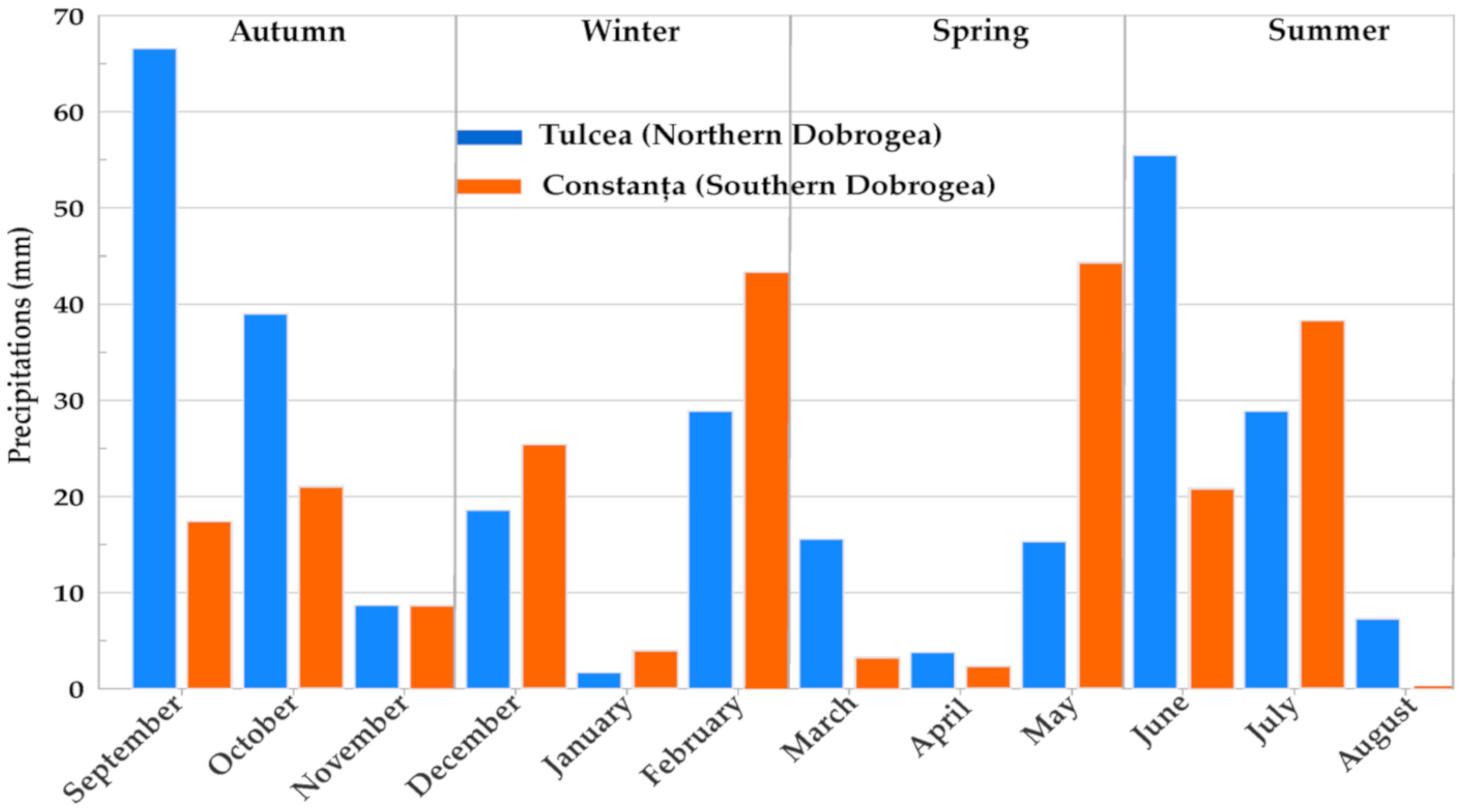
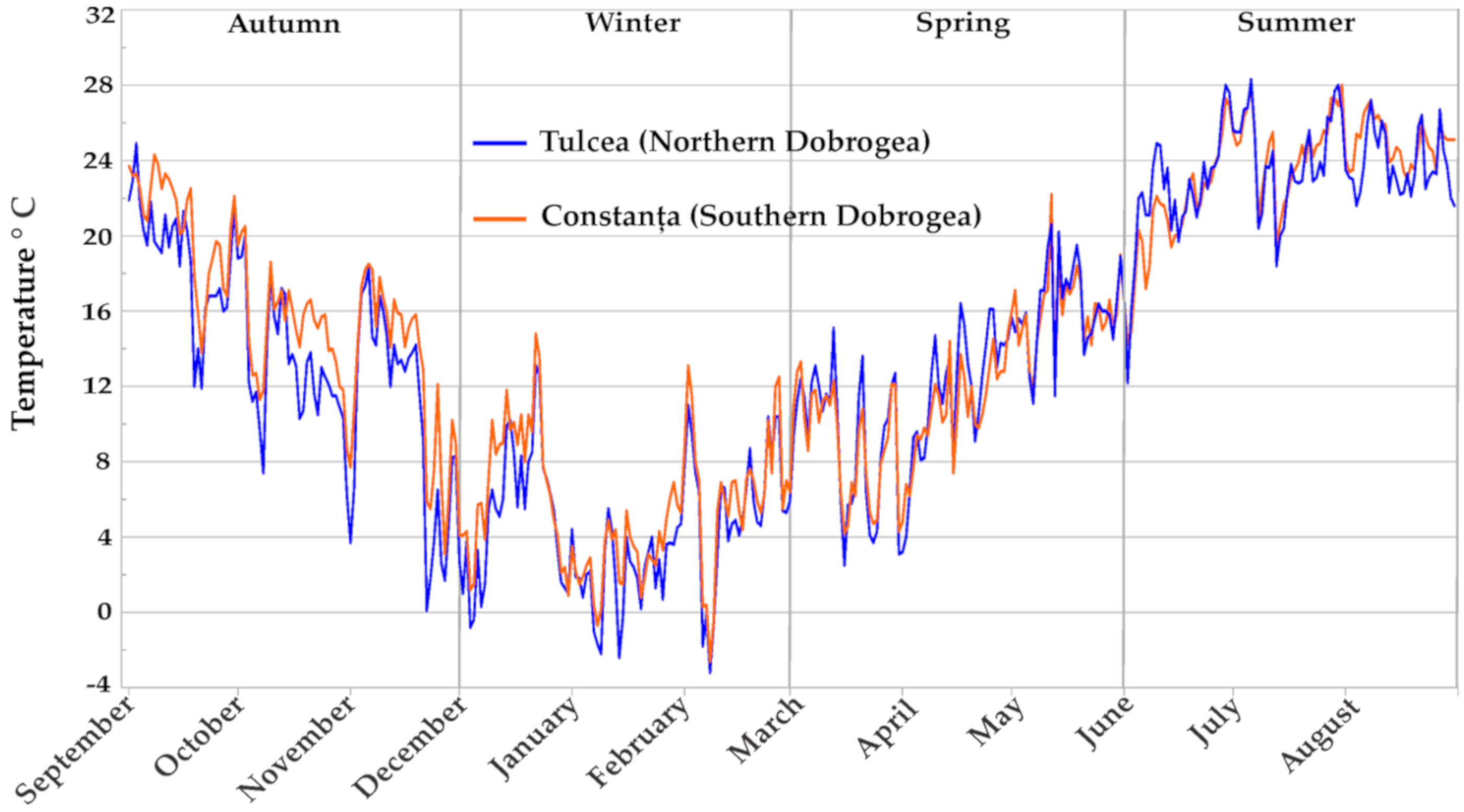
| Parameters | Meteorological Station | Autumn | Winter | Spring | Summer |
|---|---|---|---|---|---|
| Temperatures (°C) | Northern Dobrogea | 14.2 | 4.31 | 12.3 | 23.3 |
| Southern Dobrogea | 16.3 | 5.52 | 11.9 | 23.5 | |
| Precipitations (mm) | Northern Dobrogea | 114 | 49.2 | 34.7 | 91.7 |
| Southern Dobrogea | 47.2 | 72.7 | 50.1 | 59.2 |
| Heavy Metal | Si * (µg L−1) | Ii * (µg L−1) | Wi |
|---|---|---|---|
| Cr | 50 | - | 0.02 |
| Cd | 3 | - | 0.333 |
| Cu | 1500 | 50 | 0.001 |
| Fe | 300 | - | 0.003 |
| Ni | 70 | 20 | 0.01 |
| Pb | 10 | - | 0.100 |
| Zn | 15,000 | 5000 | 0.00007 |
| Springs | Season | pH | EC | DO | HCO3−− | DOC | TU | TH | Cl− | SO42− | NO3− | F− | Na | K | Ca | Mg | Fe | Cu | Zn | Ni | Cr | Co | Cd | Pb |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| µS cm−1 | mg L−1 | mg L−1 | mg L−1 | NTU | mg L−1 | mgL−1 | mg L−1 | mg L−1 | mgL−1 | mg L−1 | mg L−1 | mg L−1 | mg L−1 | mg L−1 | µg L−1 | µg L−1 | µg L−1 | µg L−1 | µg L−1 | µg L−1 | µg L−1 | |||
| GW1 | SUM | 7.7 | 1036 | 9.72 | 510 | 4.8 | 0.07 | 245 | 59.5 | 90.5 | 24.7 | 0.54 | 63.9 | 2.23 | 98.2 | 32.2 | 0.03 | 1.63 | 2.86 | 5.57 | 5.35 | <0.21 | <0.32 | <0.20 |
| AUT | 7.5 | 1124 | 8.75 | 567 | 4.0 | 0.01 | 179 | 55.1 | 109 | 29.4 | 0.59 | 68.3 | 1.66 | 71.7 | 45.4 | <0.01 | <0.71 | 8.26 | 4.28 | 4.52 | <0.21 | <0.32 | <0.20 | |
| WIN | 7.6 | 1052 | 7.15 | 363 | 3.0 | 0.07 | 165 | 52.5 | 103 | 27.5 | 0.55 | 58.6 | 1.69 | 66.0 | 36.6 | 0.09 | 2.15 | 3.35 | 5.08 | 6.09 | 1.67 | 0.79 | <0.20 | |
| SPR | 7.8 | 1217 | 7.79 | 543 | 3.4 | 0.04 | 252 | 52.0 | 106 | 26.5 | 0.47 | 58.5 | 1.36 | 101 | 41.1 | <0.01 | <0.71 | <1.00 | 2.31 | 3.08 | 0.263 | <0.32 | 0.21 | |
| Mean | 7.7 | 1107 | 8.35 | 496 | 3.8 | 0.05 | 210 | 54.8 | 102 | 27.0 | 0.54 | 62.3 | 1.73 | 84.1 | 38.8 | 0.02 | 1.74 | 4.82 | 4.31 | 4.76 | 0.94 | 0.41 | <0.20 | |
| GW2 | SUM | 7.5 | 967 | 7.20 | 490 | 3.0 | 0.01 | 295 | 53.5 | 77.8 | 23.1 | 0.25 | 46.6 | 2.25 | 118 | 36.9 | 0.02 | 1.68 | 3.17 | 6.42 | 3.37 | <0.21 | <0.32 | <0.20 |
| AUT | 7.2 | 1134 | 6.33 | 537 | 1.2 | 0.01 | 242 | 51.7 | 88.8 | 28.6 | 0.28 | 51.9 | 2.00 | 96.7 | 38.9 | <0.01 | <0.71 | <1.00 | 5.31 | 2.27 | <0.21 | <0.32 | <0.20 | |
| WIN | 7.2 | 1006 | 5.44 | 519 | 1.9 | 0.06 | 260 | 50.3 | 86.5 | 27.2 | 0.29 | 50.1 | 1.93 | 104 | 35.5 | 0.04 | 3.34 | 2.91 | 6.48 | 5.61 | 1.62 | 0.83 | <0.20 | |
| SPR | 7.4 | 1099 | 4.59 | 531 | 2.6 | 0.02 | 295 | 50.0 | 83.0 | 26.7 | 0.22 | 45.2 | 1.54 | 118 | 37.5 | <0.01 | <0.71 | <1.00 | 2.62 | 2.08 | 0.57 | <0.32 | 0.24 | |
| Mean | 7.3 | 1051 | 5.89 | 519 | 2.2 | 0.02 | 273 | 51.4 | 84.0 | 26.4 | 0.26 | 48.5 | 1.93 | 109 | 37.2 | 0.02 | 2.15 | 3.04 | 5.21 | 3.33 | 1.08 | 0.41 | <0.20 | |
| GW3 | SUM | 8.5 | 1204 | 7.80 | 500 | 4.5 | 1.70 | 126 | 65.1 | 30.0 | 192 | 0.54 | 76.3 | 5.29 | 50.4 | 68.3 | 0.14 | 1.54 | 1.64 | 3.30 | 26.7 | <0.21 | <0.32 | <0.20 |
| AUT | 8.4 | 1362 | 8.57 | 480 | 3.7 | 1.40 | 95 | 66.4 | 33.0 | 268 | 0.73 | 84.5 | 4.62 | 38.1 | 79.9 | <0.01 | <0.71 | <1.00 | 2.00 | 35.4 | <0.21 | <0.32 | <0.20 | |
| WIN | 8.4 | 1224 | 10.1 | 470 | 2.9 | 0.40 | 161 | 65.0 | 31.0 | 207 | 0.66 | 82.8 | 4.87 | 64.6 | 52.0 | <0.01 | 1.87 | 1.26 | 3.79 | 18.8 | 1.51 | 0.70 | <0.20 | |
| SPR | 8.3 | 1335 | 8.29 | 497 | 3.9 | 0.32 | 142 | 56.0 | 27.0 | 200 | 0.66 | 68.5 | 4.91 | 56.7 | 68.4 | <0.01 | <0.71 | <1.00 | 1.37 | 21.0 | 0.52 | <0.32 | 0.23 | |
| Mean | 8.4 | 1281 | 8.69 | 487 | 3.8 | 0.95 | 131 | 63.1 | 30.0 | 215 | 0.65 | 78.0 | 4.92 | 52.4 | 67.1 | 0.04 | 1.74 | 1.45 | 2.62 | 25.5 | 0.77 | 0.35 | <0.20 | |
| GW4 | SUM | 8.1 | 1173 | 10.2 | 570 | 4.4 | 0.22 | 130 | 43.3 | 15.7 | 93.5 | 1.13 | 82.1 | 14.1 | 51.9 | 45.5 | 0.02 | 2.41 | 1.47 | 2.67 | 44.7 | <0.21 | <0.32 | <0.20 |
| AUT | 7.9 | 1311 | 8.70 | 683 | 3.3 | 0.30 | 69 | 47.7 | 35.4 | 85.4 | 1.34 | 147 | 3.15 | 27.6 | 58.7 | <0.01 | <0.71 | <1.00 | <0.48 | 50.1 | <0.21 | <0.32 | <0.20 | |
| WIN | 8.0 | 1154 | 8.33 | 615 | 2.8 | 0.28 | 75 | 43.6 | 32.0 | 75.0 | 1.05 | 124 | 3.35 | 30.2 | 46.3 | <0.01 | 1.96 | <1.00 | 2.14 | 32.8 | 1.41 | 0.69 | <0.20 | |
| SPR | 8.1 | 1298 | 8.72 | 677 | 3.4 | 1.17 | 68 | 43.5 | 32.5 | 78.0 | 1.00 | 131 | 2.70 | 27.1 | 59.6 | <0.01 | <0.71 | <1.00 | 0.89 | 35.1 | 0.23 | <0.32 | 0.29 | |
| Mean | 8.0 | 1234 | 8.99 | 636 | 3.5 | 0.44 | 85 | 44.5 | 28.9 | 82.2 | 1.13 | 121 | 5.80 | 34.2 | 52.5 | <0.01 | 1.62 | 0.62 | 1.90 | 43.1 | 0.82 | 0.42 | <0.20 | |
| DWSi | 6.5–9.5 | 2500 | NA | 500 | NA | NA | NA | NA | 250 | 50 | 1.5 | 200 | NA | NA | NA | 0.2 | 2000 | NA | 20 | 50 | NA | 5.0 | 10 | |
| Si | 6.5–8.5 | 2500 | NA | NA | NA | 5.0 | NA | NA | 250 | 50 | 1.5 | 50 | NA | NA | NA | 2.0 | 2000 | NA | 70 | 50 | NA | 3.0 | 10 | |
| Indices | Season | GW1 | GW2 | GW3 | GW4 |
|---|---|---|---|---|---|
| WQI | SUM | 42.2 | 25.7 | 51.6 | 78.8 |
| AUT | 45.0 | 27.3 | 65.8 | 89.8 | |
| WIN | 42.8 | 27.7 | 58.5 | 73.3 | |
| SPR | 38.6 | 24.1 | 58.4 | 70.7 | |
| HPI | SUM | 94.8 | 89.2 | 142 | 179 |
| AUT | 95.5 | 89.4 | 162 | 196 | |
| WIN | 123 | 119 | 151 | 179 | |
| SPR | 97.0 | 94.4 | 135 | 164 | |
| HEI | SUM | 0.19 | 0.17 | 0.59 | 0.94 |
| AUT | 0.16 | 0.13 | 0.74 | 1.01 | |
| WIN | 0.46 | 0.47 | 0.70 | 0.92 | |
| SPR | 0.12 | 0.10 | 0.47 | 0.73 |
| GW1 | GW2 | GW3 | GW4 | |||||
| Oral | Dermal | Oral | Dermal | Oral | Dermal | Oral | Dermal | |
| Adults | ||||||||
| Fe | 1.10 | 3.00 × 10−3 | 3.94 × 10−1 | 1.07 × 10−3 | 6.30 × 10−1 | 1.72 × 10−3 | 3.94 × 10−1 | 1.07 × 10−3 |
| Cu | 4.09 × 10−2 | 1.86 × 10−4 | 5.07 × 10−2 | 2.30 × 10−4 | 3.80 × 10−2 | 1.73 × 10−4 | 4.56 × 10−2 | 2.07 × 10−4 |
| Pb | 6.38 × 10−3 | 2.90 × 10−6 | 6.61 × 10−3 | 3.00 × 10−6 | 6.53 × 10−3 | 2.97 × 10−6 | 7.01 × 10−3 | 3.18 × 10−6 |
| Zn | 1.22 × 10−1 | 5.53 × 10−5 | 6.63 × 10−2 | 2.89 × 10−5 | 3.86 × 10−2 | 1.75 × 10−5 | 3.52 × 10−2 | 1.60 × 10−5 |
| Ni | 1.36 × 10−1 | 6.16 × 10−4 | 1.64 × 10−1 | 7.45 × 10−4 | 8.23 × 10−2 | 3.74 × 10−4 | 4.87 × 10−2 | 2.21 × 10−4 |
| Cr | 1.50 × 10−1 | 6.81 × 10−4 | 1.05 × 10−1 | 4.77 × 10−4 | 8.02 × 10−1 | 3.64 × 10−3 | 1.28 | 5.82 × 10−3 |
| Cd | 1.38 × 10−2 | 6.26 × 10−5 | 1.41 × 10−2 | 6.40 × 10−5 | 1.31 × 10−2 | 5.94 × 10−5 | 1.30 × 10−2 | 5.90 × 10−5 |
| NO3− | 8.51 × 102 | 3.86 | 8.31 × 102 | 3.78 | 6.83 × 103 | 3.10 × 101 | 2.46 × 103 | 1.12 × 101 |
| Children | ||||||||
| Fe | 1.40 | 5.32 × 10−3 | 5.00 × 10−1 | 1.90 × 10−3 | 8.00 × 10−1 | 3.04 × 103 | 5.00 × 10−1 | 1.90 × 10−3 |
| Cu | 5.20 × 10−2 | 3.29 × 10−4 | 6.44 × 10−2 | 4.08 × 10−4 | 4.83 × 10−2 | 3.06 × 10−4 | 5.79 × 10−2 | 2.07 × 10−4 |
| Pb | 8.10 × 10−3 | 5.13 × 10−6 | 3.00 × 10−6 | 5.32 × 10−6 | 9.00 × 10−3 | 8.30 × 10−3 | 8.90 × 10−3 | 5.63 × 10−6 |
| Zn | 1.55 × 10−1 | 9.79 × 10−5 | 8.08 × 10−2 | 5.11 × 10−5 | 4.90 × 10−2 | 3.10 × 10−5 | 4.47 × 10−2 | 2.83 × 10−5 |
| Ni | 1.72 × 10−1 | 1.09 × 10−3 | 2.08 × 10−1 | 1.32 × 10−3 | 1.05 × 10−1 | 6.62 × 10−4 | 6.18 × 10−2 | 3.91 × 10−2 |
| Cr | 1.90 × 10−1 | 1.20 × 10−3 | 1.33 × 10−1 | 8.44 × 10−4 | 1.02 | 6.45 × 10−3 | 1.63 | 1.03 × 10−2 |
| Cd | 1.72 × 10−2 | 1.11 × 10−4 | 1.79 × 10−2 | 1.13 × 10−4 | 1.66 × 10−2 | 1.05 × 10−4 | 1.65 × 10−2 | 1.04 × 10−4 |
| NO3− | 1.08 × 103 | 6.84 | 1.06 × 103 | 6.68 | 8.67 × 103 | 5.49 × 101 | 3.12 × 103 | 1.97 × 101 |
| GW1 | GW2 | GW3 | GW4 | ||||||
| Oral | Dermal | Oral | Dermal | Oral | Dermal | Oral | Dermal | ||
| Adults | |||||||||
| HQ | Fe | 3.67 × 10−3 | 6.67 × 10−5 | 1.31 × 10−3 | 2.38 × 10−5 | 2.10 × 10−3 | 3.81 × 10−5 | 1.31 × 10−3 | 2.38 × 10−5 |
| Cu | 1.02 × 10−3 | 1.55 × 10−5 | 1.27 × 10−3 | 1.92 × 10−5 | 9.51 × 10−4 | 1.44 × 10−5 | 1.14 × 10−3 | 1.73 × 10−5 | |
| Pb | 2.13 × 10−5 | 4.83 × 10−8 | 2.20 × 10−5 | 5.01 × 10−8 | 2.18 × 10−5 | 4.95 × 10−8 | 2.34 × 10−5 | 5.30 × 10−8 | |
| Zn | 4.06 × 10−4 | 9.22 × 10−7 | 2.12 × 10−4 | 4.81 × 10−7 | 1.29 × 10−4 | 2.92 × 10−7 | 1.17 × 10−4 | 2.66 × 10−7 | |
| Ni | 6.79 × 10−3 | 1.14 × 10−4 | 8.20 × 10−3 | 1.38 × 10−4 | 4.12 × 10−3 | 6.93 × 10−5 | 2.43 × 10−3 | 4.09 × 10−5 | |
| Cr | 5.00 × 10−2 | 4.54 × 10−2 | 3.50 × 10−2 | 3.21 × 10−2 | 2.67 × 10−1 | 2.43 × 10−1 | 4.27 × 10−1 | 3.88 × 10−1 | |
| Cd | 2.76 × 10−2 | 1.25 × 10−2 | 2.82 × 10−2 | 1.28 × 10−2 | 2.61 × 10−2 | 1.19 × 10−2 | 2.60 × 10−2 | 1.18 × 10−2 | |
| NO3− | 5.32 × 10−1 | 3.51 × 10−3 | 5.20 × 10−1 | 3.43 × 10−3 | 4.27 | 2.82 × 10−2 | 1.63 | 1.08 × 10−2 | |
| HI | 6.21 × 10−1 | 6.16 × 10−2 | 5.94 × 10−1 | 4.82 × 10−2 | 4.57 | 2.83 × 10−1 | 2.09 | 6.16 × 10−2 | |
| THI | 0.683 | 0.642 | 4.85 | 2.50 | |||||
| Children | |||||||||
| HQ | Fe | 4.67 × 10−3 | 1.18 × 10−4 | 1.67 × 10−3 | 4.22 × 10−5 | 2.67 × 10−3 | 6.75 × 10−5 | 1.67 × 10−3 | 4.22 × 10−5 |
| Cu | 1.30 × 10−3 | 2.74 × 10−5 | 1.61 × 10−3 | 3.40 × 10−5 | 1.21 × 10−3 | 2.55 × 10−5 | 1.45 × 10−3 | 3.05 × 10−5 | |
| Pb | 2.70 × 10−5 | 8.54 × 10−8 | 2.80 × 10−5 | 8.86 × 10−8 | 2.77 × 10−5 | 8.75 × 10−8 | 2.97 × 10−5 | 9.39 × 10−8 | |
| Zn | 5.16 × 10−4 | 1.63 × 10−6 | 2.69 × 10−4 | 8.52 × 10−7 | 1.63 × 10−4 | 5.17 × 10−7 | 1.49 × 10−4 | 4.71 × 10−7 | |
| Ni | 8.62 × 10−3 | 2.02 × 10−4 | 1.04 × 10−2 | 2.44 × 10−4 | 5.23 × 10−3 | 1.23 × 10−4 | 3.09 × 10−3 | 7.24 × 10−5 | |
| Cr | 6.35 × 10−2 | 8.03 × 10−2 | 4.44 × 10−2 | 5.62 × 10−2 | 3.40 × 10−1 | 4.30 × 10−1 | 5.42 × 10−1 | 6.86 × 10−1 | |
| Cd | 3.50 × 10−2 | 2.22 × 10−2 | 3.58 × 10−2 | 2.27 × 10−2 | 3.32 × 10−2 | 2.10 × 10−2 | 3.30 × 10−2 | 2.09 × 10−2 | |
| NO3− | 6.76 × 10−1 | 6.22 × 10−3 | 6.60 × 10−1 | 6.08 × 10−3 | 5.42 | 4.99 × 10−2 | 2.07 | 1.91 × 10−2 | |
| HI | 7.89 × 10−1 | 1.09 × 10−1 | 7.54 × 10−1 | 8.53 × 10−2 | 5.8 | 5.01 × 10−1 | 2.66 | 7.27 × 10−1 | |
| THI | 0.898 | 0.840 | 6.30 | 3.38 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moldovan, A.; Hoaghia, M.-A.; Kovacs, E.; Mirea, I.C.; Kenesz, M.; Arghir, R.A.; Petculescu, A.; Levei, E.A.; Moldovan, O.T. Quality and Health Risk Assessment Associated with Water Consumption—A Case Study on Karstic Springs. Water 2020, 12, 3510. https://doi.org/10.3390/w12123510
Moldovan A, Hoaghia M-A, Kovacs E, Mirea IC, Kenesz M, Arghir RA, Petculescu A, Levei EA, Moldovan OT. Quality and Health Risk Assessment Associated with Water Consumption—A Case Study on Karstic Springs. Water. 2020; 12(12):3510. https://doi.org/10.3390/w12123510
Chicago/Turabian StyleMoldovan, Ana, Maria-Alexandra Hoaghia, Eniko Kovacs, Ionuț Cornel Mirea, Marius Kenesz, Răzvan Adrian Arghir, Alexandru Petculescu, Erika Andrea Levei, and Oana Teodora Moldovan. 2020. "Quality and Health Risk Assessment Associated with Water Consumption—A Case Study on Karstic Springs" Water 12, no. 12: 3510. https://doi.org/10.3390/w12123510
APA StyleMoldovan, A., Hoaghia, M.-A., Kovacs, E., Mirea, I. C., Kenesz, M., Arghir, R. A., Petculescu, A., Levei, E. A., & Moldovan, O. T. (2020). Quality and Health Risk Assessment Associated with Water Consumption—A Case Study on Karstic Springs. Water, 12(12), 3510. https://doi.org/10.3390/w12123510








