Integrated Aquaculture Recirculation System (IARS) Supported by Solar Energy as a Circular Economy Alternative for Resilient Communities in Arid/Semi-Arid Zones in Southern South America: A Case Study in the Camarones Town
Abstract
:1. Introduction
2. Implementation
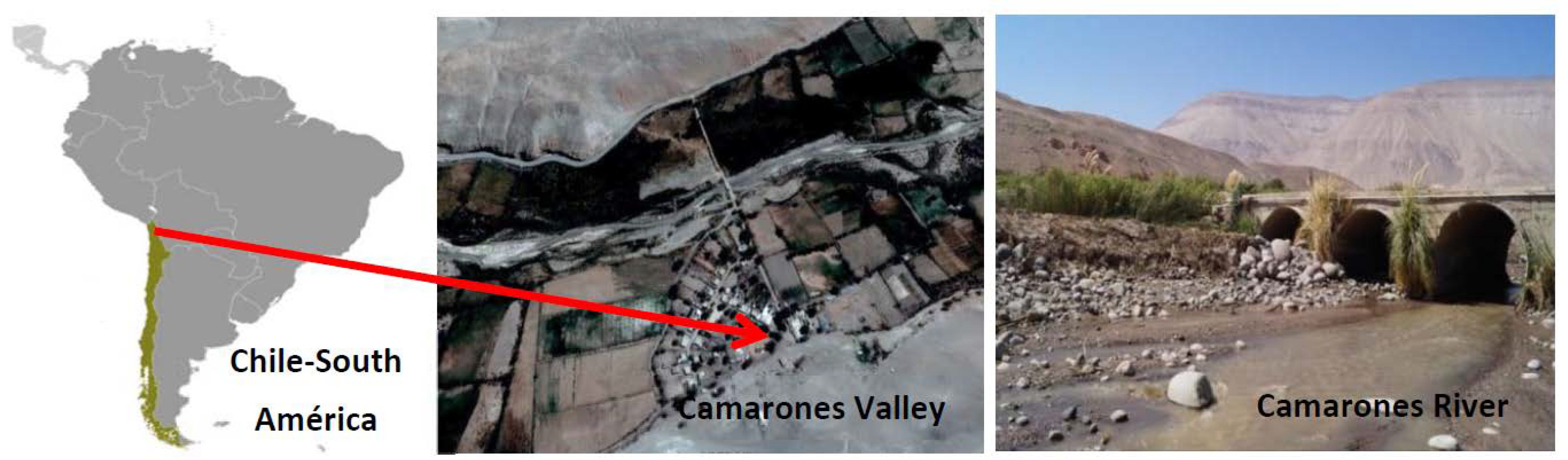
2.1. The Camarones Area
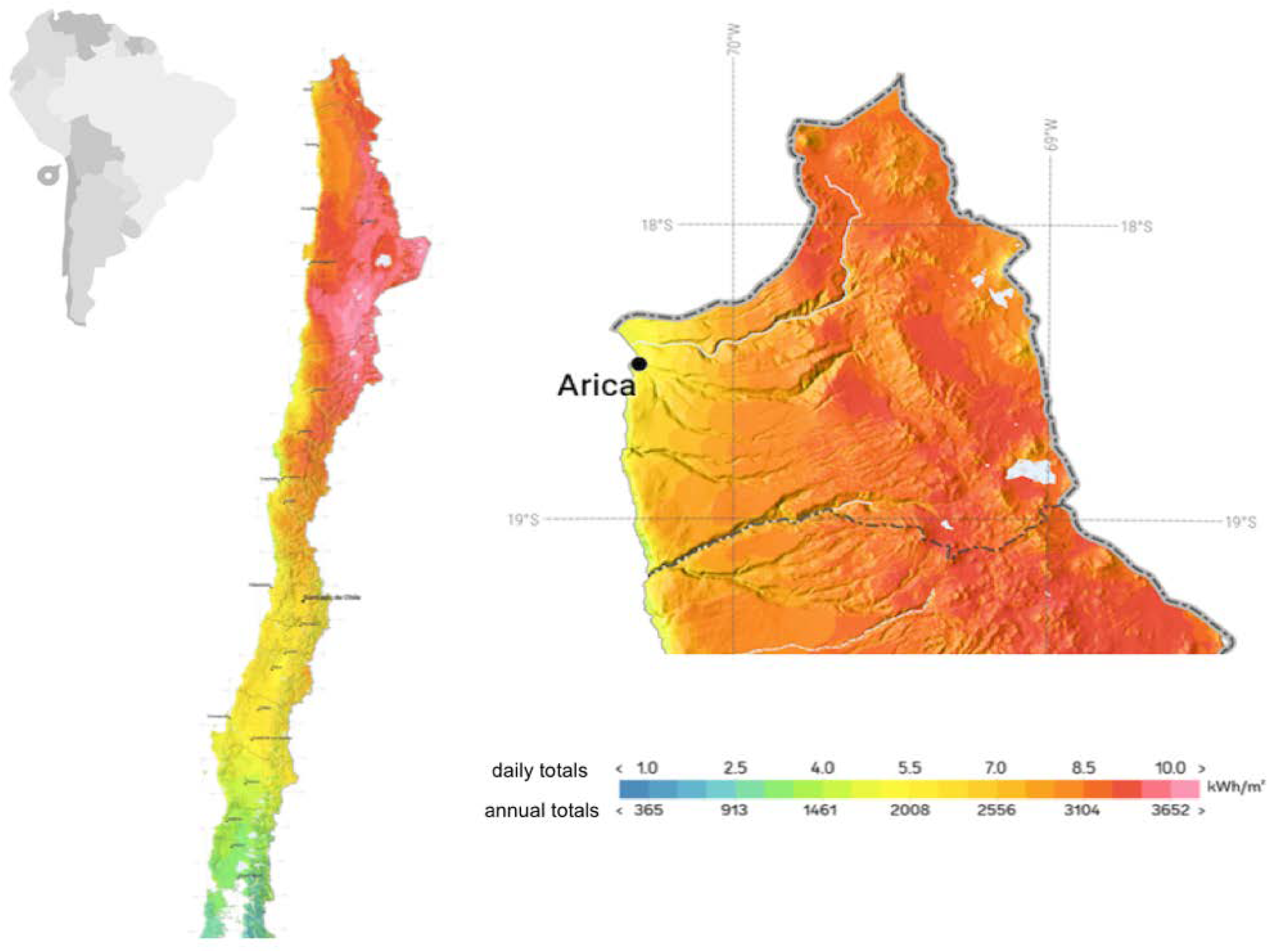
2.2. Solar Radiation
2.3. Water Quality in Hydrobiological Species Crops
2.4. Farmer Species
2.4.1. Rainbow Trout
2.4.2. Optimal Parameters for Trout Farming
2.4.3. River Shrimp
2.5. Presence of Arsenic in Aquaculture Species
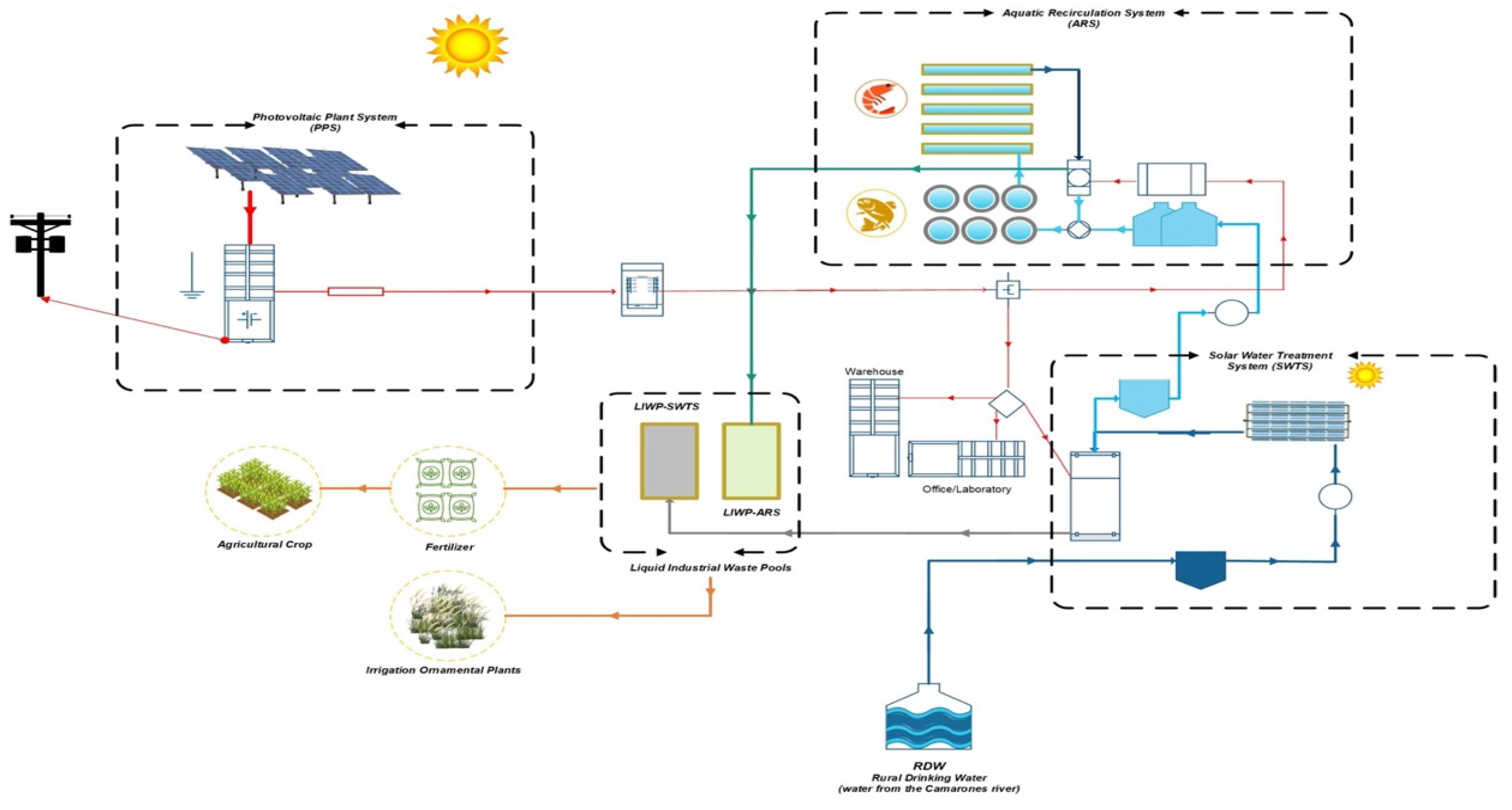
3. The Integrated Aquaculture Recirculation System
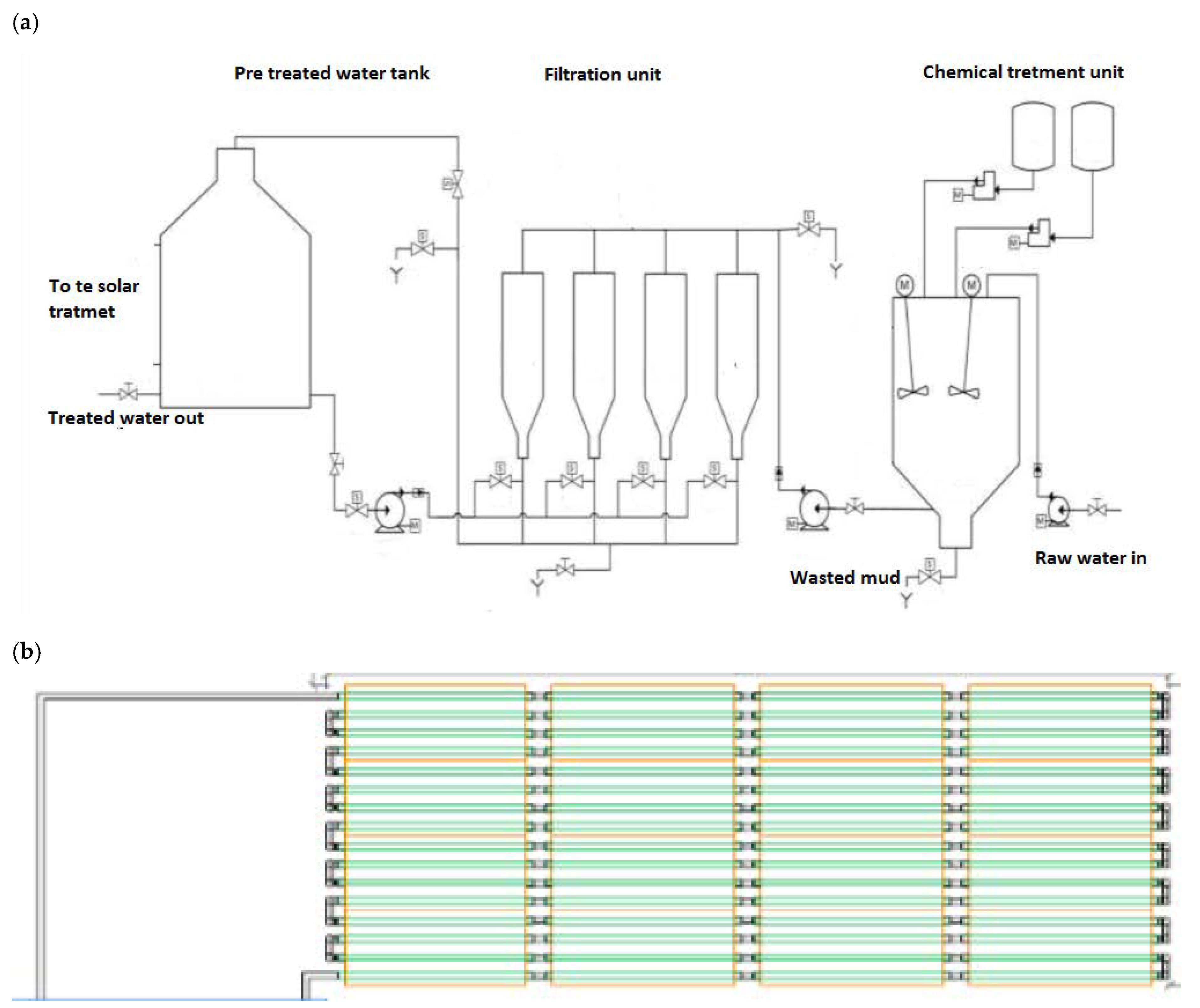
3.1. Component 1: Solar Water Treatment Plant
Optimal Performance
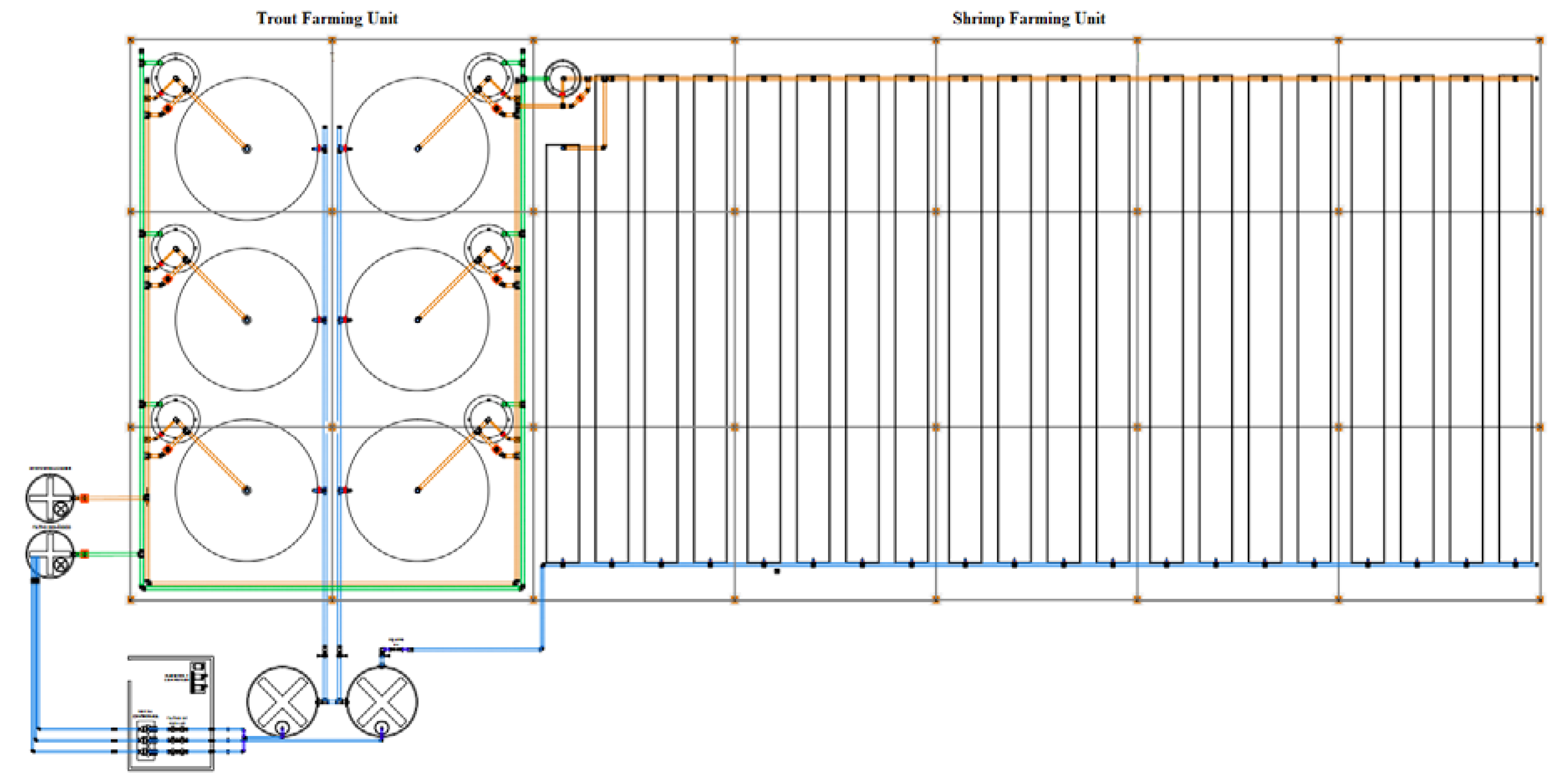
3.2. Component 2: Aquaculture Recirculation System
3.3. Component 3: Photovoltaic Plant
- (a)
- In-grid plant, to generate, consume, and sell surplus electricity to the local electricity company, this project was covered by law 20.571 regarding the distributed generation or net billing of the Chilean Ministry of Energy. This law allows self-generation of energy based on non-conventional renewable energies and efficient cogeneration. In addition, this plant was registered by the Superintendence of Electricity and Fuels (SEF), and has the following components (Table 5):
- (b)
- Off-grid plant, it is a backup system that allows connecting the critical loads of integrated aquaculture recirculation system plant, which cannot be interrupted due to the fact that there are biological species that must receive aeration for the growth and development of the species (Table 6).
4. Circular Economy
5. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Organización de Naciones Unidas, ONU. Agua. 2020. Available online: https://www.un.org/es/sections/issues-depth/water/index.html (accessed on 3 June 2020).
- PMA. Programa Mundial de Alimentos de la ONU. La COVID-19 Duplicaría el Número de Personas que Hacen Frente a Crisis Alimentarias si no se actúa con Rapidez. 2020. Available online: https://es.wfp.org/noticias/covid-19-duplicara-numero-personas-hambre-si-no-se-actua (accessed on 3 June 2020).
- FAO. El Estado Mundial de la Pesca y la Acuicultura 2018. Cumplir los Objetivos de Desarrollo Sostenible. 2018. Available online: http://www.fao.org/3/I9540ES/i9540es.pdf (accessed on 11 June 2020).
- UNESCO. Informe de las Naciones Unidas Sobre los Recursos Hídricos en el Mundo 2015. Agua Para un Mundo Sostenible. Programa Mundial de Evaluación de los Recursos Hídricos. 2015. Available online: http://www.unesco.org/new/fileadmin/MULTIMEDIA/HQ/SC/images/WWDR2015Facts_Figures_SPA_web.pdf (accessed on 18 April 2020).
- Multsch, S.; Grabowski, D.; Lüdering, J.; Alquwaizany, A.S.; Lehnert, K.; Frede, H.-G.; Winker, P.; Breuer, L. A practical planning software program for desalination in agriculture. Desalination 2017, 404, 121–131. [Google Scholar] [CrossRef]
- GEO6, Resumen para Responsables de Formular Políticas. Sexto informe Perspectivas del Medio Ambiente Mundial. 2019, p. 12. Available online: https://wedocs.unep.org/bitstream/handle/20.500.11822/27652/GEO6SPM_SP.pdf?sequence=6&isAllowed=y (accessed on 9 March 2020).
- Organización de la Naciones Unidas, ONU. Panorama de la Situación de la Pesca en el Mundo. In Conferencia de Revisión Continuada del Acuerdo Relativo a la Conservación y Ordenación de Poblaciones de Peces Transzonales y las Poblaciones de Peces altamente Migratorios; Publisher by Département de l’Information de Nations Unies. 2010, pp. 24–28. Available online: http://www.un.org/depts/los/convention_agreements/reviewconf/FishStocks_SP_A.pdf (accessed on 23 March 2020).
- FAO. La Resiliencia de los Medios de Vida–Programa Marco de Reducción del Riesgo de Desastres para la Seguridad Alimentaria y Nutricional. 2013. Available online: http://www.fao.org/3/a-i3270s.pdf (accessed on 27 March 2020).
- Cosgrove, C.E.; Cosgrove, W.J. The Dynamics of Global Water Futures Driving Forces 2011–2050; Report on the findings of Phase One of the UNESCO-WWAP Water Scenarios Project to 2050; Issues: Infrastructure, Health Natural Resources, World Water Assessment Programme (UNESCO-WWAP); UNESCO: Paris, France, 2012; ISBN 978-92-3-001035-5. [Google Scholar]
- Liu, H.; Lia, H.; Wei, H.; Zhu, X.; Hana, D.; Jin, J.; Yanga, Y.; Xie, S. Biofloc formation improves water quality and fish yield in a freshwater pond aquaculture system. Aquaculture 2019, 506, 256–269. [Google Scholar] [CrossRef]
- Biblioteca del Congreso Nacional de Chile, BCN. Clima y Vegetación Región Arica y Parinacota. Biblioteca del Congreso Nacional de Chile. 2018. Available online: https://www.bcn.cl/siit/nuestropais/region15/clima.htm (accessed on 10 May 2020).
- SERC. Solar Energy Research Center. 2020. Available online: http://serc.cl/ (accessed on 10 May 2020).
- García, S.I. Hidroarsenicismo Crónico Regional Endémico HACRE: Módulo de Capacitación, Programa Nacional de Prevención y Control de las Intoxicaciones, 1st Edición; Buenos Aires, Argentina. 2011, p. 68. Available online: https://bancos.salud.gob.ar/sites/default/files/2020-10/03-%202011-HACRE-modulo-capacitacion.pdf (accessed on 10 May 2020).
- Dirección General de Aguas, DGA. Estudio de Sintesis de Catastros de Ususarios de Agua e Infraestriuctura de Aprovechamiento. Dirección General de Aguas. Ministerio de Obras Públicas. 1991, p. 49. Available online: https://snia.mop.gob.cl/sad/INF313.pdf (accessed on 10 May 2020).
- Cornejo, L.; Lienqueo, H.; Arenas, M.J.; Acarapi, J.; Contreras, D.; Yáñez, J.; Mansilla, H. In field arsenic removal from natural water by zero-valent iron assisted by solar radiation. Environ. Pollut. 2008, 156, 827–831. [Google Scholar] [CrossRef]
- Cornejo, L.; Lienqueo, H.; Vilca, P. Hydro-chemical characteristics, water quality assessment and water relationship (HCA) of the Amuyo Lagoons, Andean Altiplano, Chile. Desalin. Water Treat. 2019, 153, 36–45. [Google Scholar] [CrossRef]
- Cornejo, L.; Acarapi, J.; Mella, U. Cuenca se Camarones: Identificación y Caracterización de Fuentes que Condicionan la Calidad de las Aguas Superficiales: Rol del Tranque Caritaya; UTA: Arica, Chile, 2010; pp. 1–222. [Google Scholar]
- Cornejo, L.; Acarapi, J.; Lienqueo, H.; Arena, M. Métodos Solares. Tecnología Hierro cero para la Remoción de arsénico en aguas Naturales con altos Niveles de Salinidad. Experiencias en Implementación y Transferencia Tecnológica en Comunidades Rurales del Norte de Chile, IBEROARSEN. Tecnologías Económicas para el Abatimiento de Arsénico en Aguas; Litter, M., Sancha, A.M., Ingallinella, A.M., Eds.; CYTED: Bueno Aires, Argentina, 2010; ISBN 978-84-96023-74-1. [Google Scholar]
- Instituto Nacional de Normalización, INN. Drinking water-Part 1: Requirements; NCh409/1:2005; Instituto Nacional de Normalización: Santiago, Chile, 2005. [Google Scholar]
- Instituto Nacional de Normalización. Water Quality Requirements for Different Uses; INN. NCh1333:1987; Instituto Nacional de Normalización: Santiago, Chile, 1987. [Google Scholar]
- Höll, W.; Litter, M.Y. Capitulo 1. Ocurrencia y Química del Arsénico en Aguas. Sumario de Tecnologías de Remoción de Arsénico de Aguas, IBEROARSEN. Tecnologís Económicas para el Abatimiento de ars√©nico en aguas; Litter, M., Sancha, A.M., Ingallinella, A.M., Eds.; CYTED: Buenos Aires, Argentina, 2010; ISBN 978-84-96023-74-1. [Google Scholar]
- WWAP. Programa Mundial de las Naciones Unidas de Evaluación de los Recursos Hídricos/ONU-Agua. Informe Mundial de las Naciones Unidas sobre el Desarrollo de los Recursos Hídricos 2018: Soluciones Basadas en la Naturaleza para la Gestión del Agua. París, UNESCO. 2018. Available online: https://unesdoc.unesco.org/ark:/48223/pf0000261494 (accessed on 10 August 2020).
- Instituto Nacional de Estadística, INE. Comuna de Camarones. Región de Arica y Parinacota. Resultados Censo 2017. Instituto Nacional Estadistica. 2017. Available online: http://resultados.censo2017.cl/Region?R=R15 (accessed on 10 May 2020).
- Palenzuela, P.; Miralles-Cuevas, S.; Cabrera-Reina, A.; Cornejo-Ponce, L. Techno-economic assessment of a multi-effect distillation plant installed for the production of irrigation water in Arica (Chile). Sci. Total Environ. 2018, 643, 423–434. [Google Scholar] [CrossRef]
- Cornejo, L.; Martín-Pomares, L.; Alarcón, D.; Blanco, J.; Polo, J. A through analysis of solar irradiation measurements in the region of Arica Parinacota, Chile. Renew. Energy 2017, 112, 197–208. [Google Scholar] [CrossRef]
- Solargis. Solar Resource Maps of Chile. Irradiación Directa Normal de Chile. © 2019 The World Bank, Source: Global Solar Atlas 2.0, Solar Resource Data: Solargis. 2019. Available online: https://solargis.com/es (accessed on 10 August 2020).
- Rosenthal, H. Water Quality: Problems and Solutions. “Aquaculture. A Biotechnology in Progress”; Pauw, N., Jaspers, E., Ackefors, H., Wilkins, N., Eds.; European Aquaculture Society: Bredene, Belgica, 1989. [Google Scholar]
- Tomasso, J.R.; Brune, D.E. Aquacultural Water Quality: The Emergence of an Applied Discipline. “Aquaculture and Water Quality”; Brune, D.E., Tomasso, J.R., Eds.; The World Aquaculture Society: Baton, Rouge, 1991. [Google Scholar]
- Boyd, C.E. Water Quality in Warmwater Fish Culture; Elsiever Scientific Pub. Co.: Amsterdam, The Netherlands, 1982; ISBN 0444420541. [Google Scholar]
- Blanco, M. La Trucha. Cria en Cautividad; Ediciones Mundi Prensa: Madrid, Spain, 1995; p. 503. [Google Scholar]
- Carballo, M.; Muñoz, M. Effect of sublethal concentrations of four chemicals on Susceptibility of juvenile rainbow trout (Oncorhynchus niykiss) tosaprolegniosis. Appl. Environm. Microbiol. 1991, 57, 1813–1816. [Google Scholar] [CrossRef] [Green Version]
- Becke, C.; Schumann, M.; Steinhagen, D.; Rojas-Tiradoc, P.; Geiste, J.; Brinkera, A. Effects of unionized ammonia and suspended solids on rainbow trout (Oncorhynchus mykiss) in recirculating aquaculture systems. Aquaculture 2019, 499, 348–357. [Google Scholar] [CrossRef] [Green Version]
- Avenue, T.C.; Kong, H. The environmental impact of marine fish culture: Towards a sustainable future. Mar. Pollut. Bull. 1995, 31, 159–166. [Google Scholar]
- Timmons, M.B.; Ebeling, J.M.; Piedrahita, R.H. Acuicultura en Sistemas de Recirculación; Edición en español de la Fundación Chile: Santiago, Chile, 2009; p. 748. [Google Scholar]
- Chen, S.; Stechey, D.; Malone, R. Suspended solids control in recirculating aquaculture systems. In Aquaculture Water Reuse Systems: Engineering Design and Management; Timmons, M.B., Losordo, T.M., Eds.; Elsevier: Amsterdam, The Netherlands, 1994; pp. 61–100. [Google Scholar]
- Lovell, T. Nutrition and Feeding of Fish; Chapman Hall: New York, NY, USA, 1988. [Google Scholar]
- Islam, M.S. Nitrogen and phosphorus budget in coastal and marine cage aquaculture and impact of effluent loading on ecosystem: Review and analysis towards model development. Mar. Pollut. Bull. 2005, 50, 48–61. [Google Scholar] [CrossRef]
- Sandu, S.; Hallerman, E. Charter 13, Biodegradation of nitrogen in a commercial recirculating aquaculture facility. In Biodegradation-Engineering and Technology; Chamyn, R., RosenKranz, F., Eds.; InTech: Rijeka, Croatia, 2013; pp. 341–364. ISBN 978-953-1153-5. [Google Scholar]
- Ebeling, J.M.; Timmons, M.B.; Bisogni, J.J. Engineering analysis of the stoichiometry of photoautotrophic, autotrophic, and heterotrophic removal of ammonia–nitrogen in aquaculture systems. Aquaculture 2006, 257, 346–358. [Google Scholar] [CrossRef] [Green Version]
- Randall, D.J.; Tsui, T.K.N. Ammonia toxicity in fish. Mar. Pollut. Bull. 2002, 45, 17–23. [Google Scholar] [CrossRef]
- Ministerio del Medio Ambiente, MMA. Oncorhynchus mykiss (Walbaum 1792). Inventario Nacional de especies de Chile. 2020. Available online: http://especies.mma.gob.cl/CNMWeb/Web/WebCiudadana/ficha_indepen.aspx?EspecieId=108 (accessed on 7 August 2020).
- Izquierdo, P.; Torres, G.; Gonzalez, E.; Barboza, Y.; Márquez, E. Caracteristicas físico-químicas de la carne de trucha (Oncorhynchus mykiss). Rev. Cient. FCV LUZ 1999, 9, 127–132. [Google Scholar]
- FAO. Oncorhynchus mykiss. In Cultured Aquatic Species Fact Sheets; Text by Cowx, I.G. Edited and Compiled by Valerio Crespi and Michael New; 2009; Available online: http://www.fao.org/tempref/FI/DOCUMENT/aquaculture/CulturedSpecies/file/es/es_rainbowtrout.htm (accessed on 2 June 2020).
- Cameron, J.N.; Davis, J.C. Gas exchange in rainbow trout (Salmo gairdneri) with varying blood oxygen capacity. J. Fish. Board Can. 1970, 27, 1069–1085. [Google Scholar] [CrossRef]
- Proyecto Fondo de Innovación para la Competitividad Regional FIC-R, Codigo BIP 30158872-0, 2013. Ficha de la Especie. Cultivo de Trucha Arcoiris (Oncorhynchus mykiss) en Sistema de Recirculación, como Alternativa Sustentable y de Desarrollo Productivo para Comunidades Precordilleranas de la Región de Arica y Parinacota, Arica, Chile. 2013. Available online: https://www.truchascurmi.cl/proyecto/ficha-paiche.html (accessed on 11 June 2020).
- Ministerio de Medio Ambiente, MMA. Cryphiops caementarius (Molina, 1782). Inventario Nacional de Especies de Chile. 2020. Available online: http://especies.mma.gob.cl/CNMWeb/Web/WebCiudadana/ficha_indepen.aspx?EspecieId=639&Version=1 (accessed on 11 June 2020).
- Centro de Ecología Aplicada, CEA. Ficha de Camaron de río del Norte de Chile. Centro de Estudios Agrarios y Ambientales, Ministerio de Medio Ambiente de Chile. 2013. Available online: http://www.mma.gob.cl/clasificacionespecies/fichas10proceso/fichas_10_pac/Cryphiops_caementarius_1RCE_01_PAC.pdf (accessed on 13 March 2020).
- Decreto Supremo N° 145/1986. Establece Tamaño Mínimo de Extracción Camarón de río. Normative of Extractive Fishing Regulations, SUBPESCA; Ministerio de Economia, Fomento y Turismo: Santiago, Chile, 1986. [Google Scholar]
- Meruane, J.A.; Morales, M.C.; Galleguillos, C.A.; Rivera, M.A.; Hosokawa, H. Experiencias y resultados de investigaciones sobre el camaron de rio del norte cryphiops caementarius (Molina 1782) (Decapoda: Palaemonidae): Historia natural y cultivo. Gayana 2006, 70, 280–292. [Google Scholar] [CrossRef]
- Cornejo, L.; Lienqueo, H.; Arriaza, B. Levels of total arsenic in edible fish and shellfish obtained from two coastal sectors of the Atacama Desert in the north of Chile: Use of non-migratory marine species as bioindicators of sea environmental pollution. J. Environ. Sci. Health A 2011, 46, 1274–1282. [Google Scholar] [CrossRef]
- Albert, L.A. Introducción a la Toxicología Ambiental; Gobierno del Estado de México: Edomex, Mexico, 1977; p. 471. ISBN1 9275322333. ISBN2 978927532. [Google Scholar]
- Lenihan, J.; Fletcher, W.W. The Chemical Environment. Environment and Man; Blackie Glasgow and Son: London, UK, 1977; Volume 6. [Google Scholar]
- Garcia, B. Contaminación del agua por metales pesados As, B, Cu, Pb, Cd y CN- en las cuencas de los ríos Tambo, Quilca, Camaná y Ocoño de la región de Arequipa. Ph.D. Thesis, Universidad Nacional de San Agustín de Arequipa, Arequipa, Peru, 2019; p. 195. [Google Scholar]
- Ministerio de Sanidad y Política Social Secretaría General Técnica, MSPS. Guía de Desalación: Aspectos técnicos y Sanitarios en la Producción de agua de Consumo Humano; Centro de Publicaciones Paseo del Prado: Madrid, Spain, 2009; p. 60. [Google Scholar]
- Wang, K.; Li, K.; Liu, L.; Tanase, C.; Mols, R.; Van der Meer, M. Effects of light intensity and photoperiod on the growth and stress response of juvenile Nile tilapia (Oreochromis niloticus) in a recirculating aquaculture system. Aquacult. Fish. 2020, in press. Available online: https://reader.elsevier.com/reader/sd/pii/S2468550X20300204?token=F75946E4B3F7EE3D231E218D11E55E315E4B4B31106519F5585DA76C85B9CCFF42C43FE16BA8981F80BCD928E7E242A1 (accessed on 30 July 2020).
- Pepe-Victoriano, R.; Aravena-Ambrosetti, H. Primer Centro de Cultivos de Trucha Arcoíris bajo un Sistema de Recirculación, en el sector de Copaquilla, Región de Arica y Parinacota. Revista Versión Diferente Salmón-Acuícola 2018, 29, 58–62. [Google Scholar]
- Industria Acuícola, Acuicultura y Negocios de México. Sistemas de recirculación en acuicultura: Una visión y retos diversos para Latinoamérica. Aqua Negocios 2012, 8, 4–8. Available online: https://issuu.com/industriaacuicola/docs/edicion8_2 (accessed on 8 April 2020).
- Zhang, S.Y.; Lia, G.; Wu, H.B.; Liu, X.G.; Yao, Y.H.; Tao, L.; Liu, H. An integrated recirculating aquaculture system (RAS) for land-based fish farming: The effects on water quality and fish production. Aquacult. Eng. 2011, 45, 93–102. [Google Scholar] [CrossRef]
- Solar Trust. 2020. Available online: www.Solartrust.cl (accessed on 15 August 2020).
- Sgroi, M.; Vagliasindi, F.; Roccaro, P. Feasibility, sustainability and circular economy concepts in water reuse. Environ. Sci. Health 2018, 2, 20–25. [Google Scholar] [CrossRef]
- Makropoulos, C.; Rozos, E.; Tsoukalas, I.; Plevri, A.; Karakatsanis, G.; Karagiannidis, L.; Makri, E.; Lioumis, C.; Noutsopoulos, D.; Mamais, D.; et al. Sewer-mining: A water reuse option supporting circular economy, public service provision and entrepreneurship. J. Environ. Manag. 2018, 216, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Laso, J.; García-Herrero, I.; Margallo, M.; Vázquez-Rowe, I.; Fullana, P.; Bala, L.; Gazulla, C.; Irabien, A.; Aldaco, R. Finding an economic and environmental balance in value chains based on circular economy thinking: An eco-efficiency methodology applied to the fish canning industry. Resour. Conserv. Recycl. 2018, 133, 428–437. [Google Scholar] [CrossRef]
- Atzori, G.; de Vos, A.C.; van Rijsselberghe, M.; Vignolini, P.; Rozema, J.; Mancuso, S.; van Bodeg, P.M. Effects of increased seawater salinity irrigation on growth and quality of the edible halophyte Mesembryanthemum crystallinum L. under field conditions. Agric. Water Manag. 2017, 187, 37–46. [Google Scholar] [CrossRef]
- Buschmann Alejandro, H. Impacto Ambiental de la Acuicultura el Estado de la Investigacion en Chile y el Mundo. 2001. Available online: https://www.cetmar.org/DOCUMENTACION/dyp/ImpactoChileacuicultura.pdf (accessed on 29 June 2020).
- Sánchez, A.S.; Nogueira, I.B.R.; Kalid, R.A. Uses of the reject brine from inland desalination for fish farming, Spirulina cultivation, and irrigation of forage shrub and crops. Desalination 2015, 364, 96–107. [Google Scholar] [CrossRef]
- Cornejo-Ponce, L.; Acarapi-Cartes, J.; Arenas-Herrera, M. Development and validation of a method for simultaneous arsenic, antimony, selenium and mercury determination in plants by energy dispersive X ray fluorescence spectrometry. Interciencia 2018, 43, 425–433. [Google Scholar]
- Bianchi, E.; Dalzochio, T.; Ressel, L.A.; Prado, G.Z.; Marques da Silva, C.E.; Gehlen, G.; do Nascimento, C.A.; Rosado, F.; Ziulkoski, A.L.; Basso da Silva, L. Water quality monitoring of the Sinos River Basin, Southern Brazil, using physicochemical and microbiological analysis and biomarkers in laboratory-exposed fish. Ecohydrol. Hydrobiol. 2019, 19, 328–338. [Google Scholar] [CrossRef]

| Physical–Chemical Parameters | Unit | Camarones River Water Values | NCh409/1 of 2005 Potable Water | NCh1333 of 1978 Modificada 1987 Irrigation Water |
|---|---|---|---|---|
| pH | - | 8.3 | 6.5–8.5 | 6.0–9.0 |
| Electrical conductivity | μs cm−1 | 2200 | - | - |
| Temperature | °C | 22.30 | - | 30.00 |
| Chloride | mg L−1 | 564.30 | 400.00 | 200.00 |
| Sulfate | mg L−1 | 178.50 | 500.00 | 250.00 |
| Sodium | mg L−1 | 557.70 | - | - |
| Potassium | mg L−1 | 34.20 | - | - |
| Manganese | mg L−1 | 0.10 | 0.10 | 0.20 |
| Magnesium | mg L−1 | 15.60 | 125.00 | - |
| Calcium | mg L−1 | 101.80 | - | - |
| Arsenic | mg L−1 | 1.20 | 0.01 | 0.10 |
| Total dissolved solids | mg L−1 | 1561 | 1500 | - |
| Parameter | Optimal Range | Parameter | Optimal Range |
|---|---|---|---|
| Temperature | 9–17 °C | Nitrites | Not greater than 0.05 mg L−1 |
| Dissolved oxygen | 5.50–9.00 mg L−1 | Ammonium Nitrogen | Not greater than 0.01 mg L−1 |
| pH | 6.5–8.5 | Phosphates | Greater than 500.00 mg L−1 |
| CO2 | Less than 7.00 mg L−1 | Sulfates | Greater than 45.00 mg L−1 |
| Alkalinity | 20.00–200.00 mg L−1 CaCO3 | Iron | Less than 0.10 mg L−1 |
| Hardness | 60.00–300.00 mg L−1 CaCO3 | Copper | Less than 0.05 mg L−1 |
| NH3 | Not greater than 0.02 mg L−1 | Lead | 0.03 mg L−1 |
| H2S | Maximum accepted 0.002 mg L−1 | Mercury | 0.05 mg L−1 |
| Nitrates | Not greater than 100.00 mg L−1 |
| Ammonium mg L−1 | Nitrate mg L−1 | pH | Oxygen mg L−1 | Temperature °C | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Input | Exit | Input | Exit | Input | Exit | Input | Exit | Input | Exit | |
| Month 1 | 0.08 | 0.21 | 0.31 | 0.38 | 6.8 | 6.9 | 5.8 | 5.5 | 14.8 | 15.3 |
| Month 2 | 0.07 | 0.14 | 0.60 | 0.80 | 6.9 | 6.9 | 5.7 | 5.7 | 15.3 | 15.8 |
| Month 3 | 0.16 | 0.19 | 0.36 | 0.36 | 6.9 | 7.0 | 5.6 | 5.5 | 16.7 | 17.9 |
| Month 4 | 0.08 | 0.19 | 0.38 | 0.09 | 6.9 | 7.0 | 5.8 | 5.6 | 17.5 | 18.3 |
| Month 5 | 0.11 | 0.31 | 0.44 | 0.09 | 7.0 | 6.9 | 6.3 | 6.2 | 17.1 | 17.8 |
| Month 6 | 0.08 | 0.21 | 0.31 | 0.38 | 6.8 | 6.9 | 6.4 | 6.0 | 16.1 | 16.6 |
| Month 7 | 0.07 | 0.14 | 0.60 | 0.80 | 6.9 | 7.0 | 6.3 | 6.0 | 14.7 | 14.9 |
| Month 8 | 0.16 | 0.19 | 0.36 | 0.36 | 7.0 | 7.1 | 6.2 | 6.0 | 12.5 | 12.8 |
| Parameter | Optimal Range |
|---|---|
| Temperature | 20–25 °C |
| Dissolved oxygen | 7.00–10.00 mg L−1 |
| pH | 7.5–8.0 |
| Ammonium | <0.50 mg L−1 |
| Nitrates | 4.00 mg L−1 |
| Nitrites | 0.25 mg L−1 |
| Salinity | 20 Practical Salinity Unit (PSU) during farming and reduction to 15 PSU in zoea 15 and to 12–10 PSU at the end of each cycle |
| Item | Characteristics |
|---|---|
| 80 polycrystalline silicon photovoltaic modules, brand Astronergy | Maximum power de 295 W |
| 1 Three-phase inverter SMA Sunny Tripower | 25,000 Wp |
| Power | 25 kVA |
| Item | Characteristics |
|---|---|
| 1 Studer hybrid inverter, single phase | 800 W |
| Battery bank | 24 Sonnenschein batteries (2 V cells) 1960 Ah |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cornejo-Ponce, L.; Vilca-Salinas, P.; Lienqueo-Aburto, H.; Arenas, M.J.; Pepe-Victoriano, R.; Carpio, E.; Rodríguez, J. Integrated Aquaculture Recirculation System (IARS) Supported by Solar Energy as a Circular Economy Alternative for Resilient Communities in Arid/Semi-Arid Zones in Southern South America: A Case Study in the Camarones Town. Water 2020, 12, 3469. https://doi.org/10.3390/w12123469
Cornejo-Ponce L, Vilca-Salinas P, Lienqueo-Aburto H, Arenas MJ, Pepe-Victoriano R, Carpio E, Rodríguez J. Integrated Aquaculture Recirculation System (IARS) Supported by Solar Energy as a Circular Economy Alternative for Resilient Communities in Arid/Semi-Arid Zones in Southern South America: A Case Study in the Camarones Town. Water. 2020; 12(12):3469. https://doi.org/10.3390/w12123469
Chicago/Turabian StyleCornejo-Ponce, Lorena, Patricia Vilca-Salinas, Hugo Lienqueo-Aburto, María J. Arenas, Renzo Pepe-Victoriano, Edward Carpio, and Juan Rodríguez. 2020. "Integrated Aquaculture Recirculation System (IARS) Supported by Solar Energy as a Circular Economy Alternative for Resilient Communities in Arid/Semi-Arid Zones in Southern South America: A Case Study in the Camarones Town" Water 12, no. 12: 3469. https://doi.org/10.3390/w12123469
APA StyleCornejo-Ponce, L., Vilca-Salinas, P., Lienqueo-Aburto, H., Arenas, M. J., Pepe-Victoriano, R., Carpio, E., & Rodríguez, J. (2020). Integrated Aquaculture Recirculation System (IARS) Supported by Solar Energy as a Circular Economy Alternative for Resilient Communities in Arid/Semi-Arid Zones in Southern South America: A Case Study in the Camarones Town. Water, 12(12), 3469. https://doi.org/10.3390/w12123469






