Hydrochemical Characteristics and Irrigation Suitability Evaluation of Groundwater with Different Degrees of Seawater Intrusion
Abstract
:1. Introduction
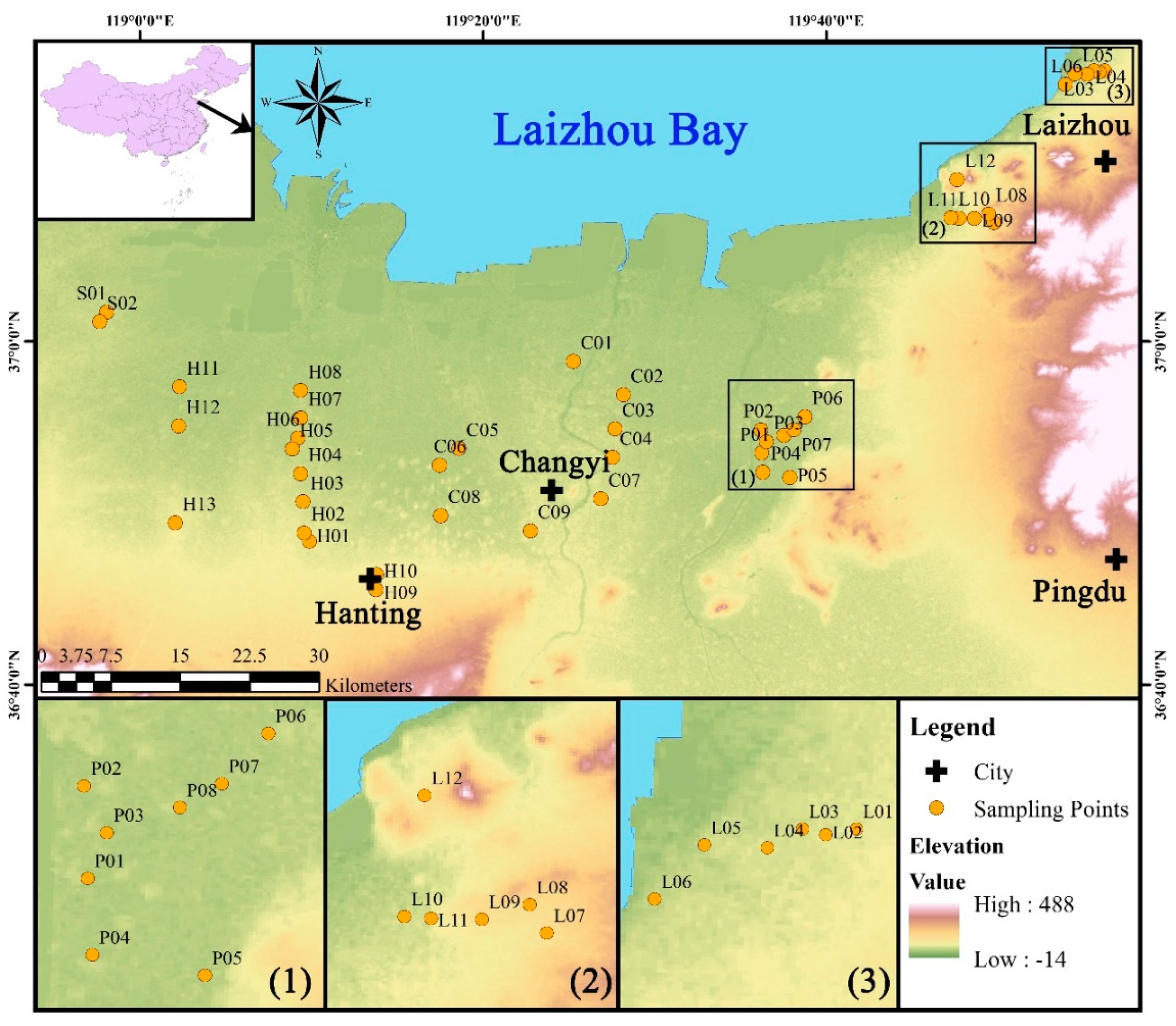
2. Study Area
2.1. Physiographic Condition
2.2. Hydrogeology
3. Materials and Methods
3.1. Sampling and Testing
3.2. Analytical Methods
4. Results
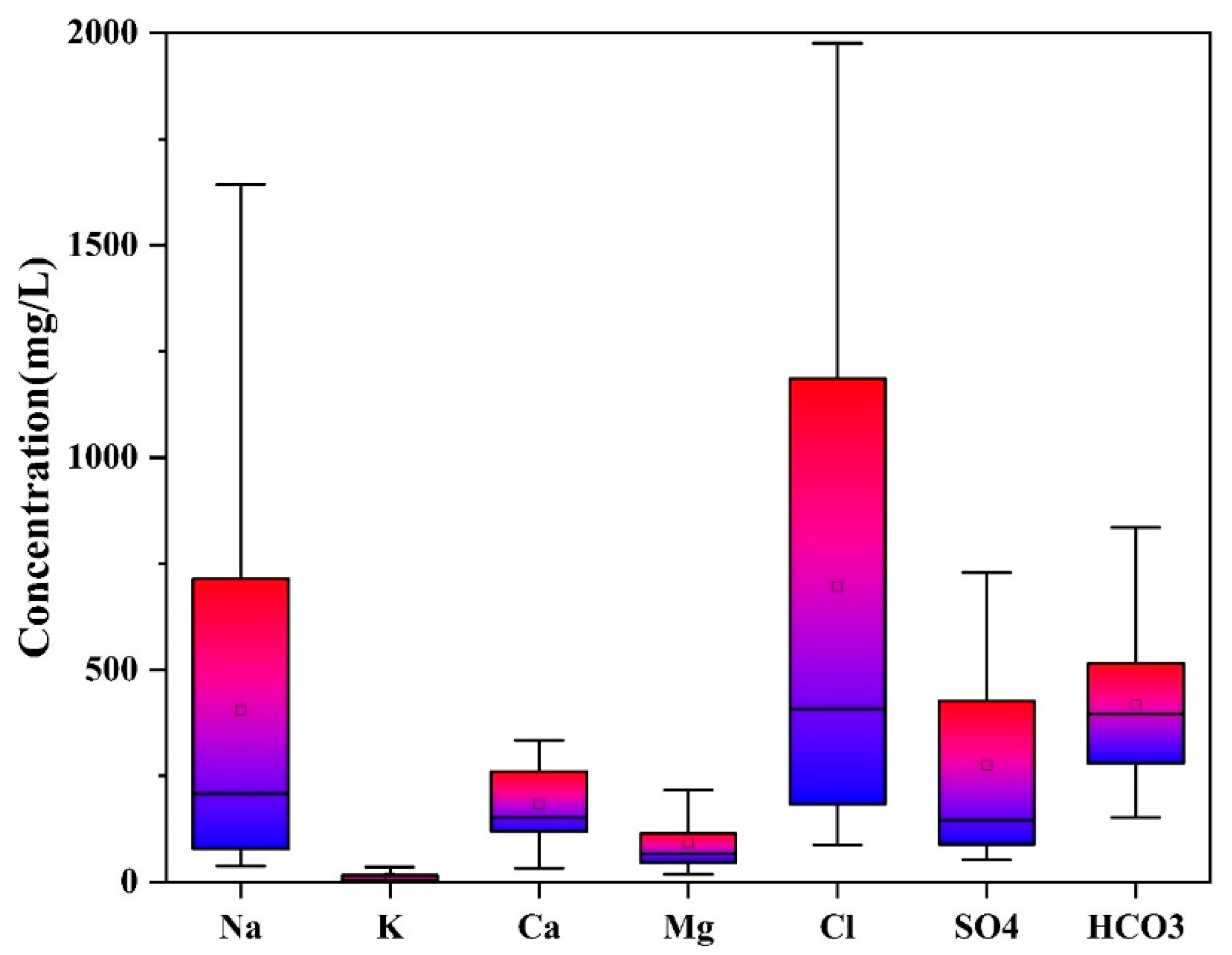
4.1. Statistical Analysis
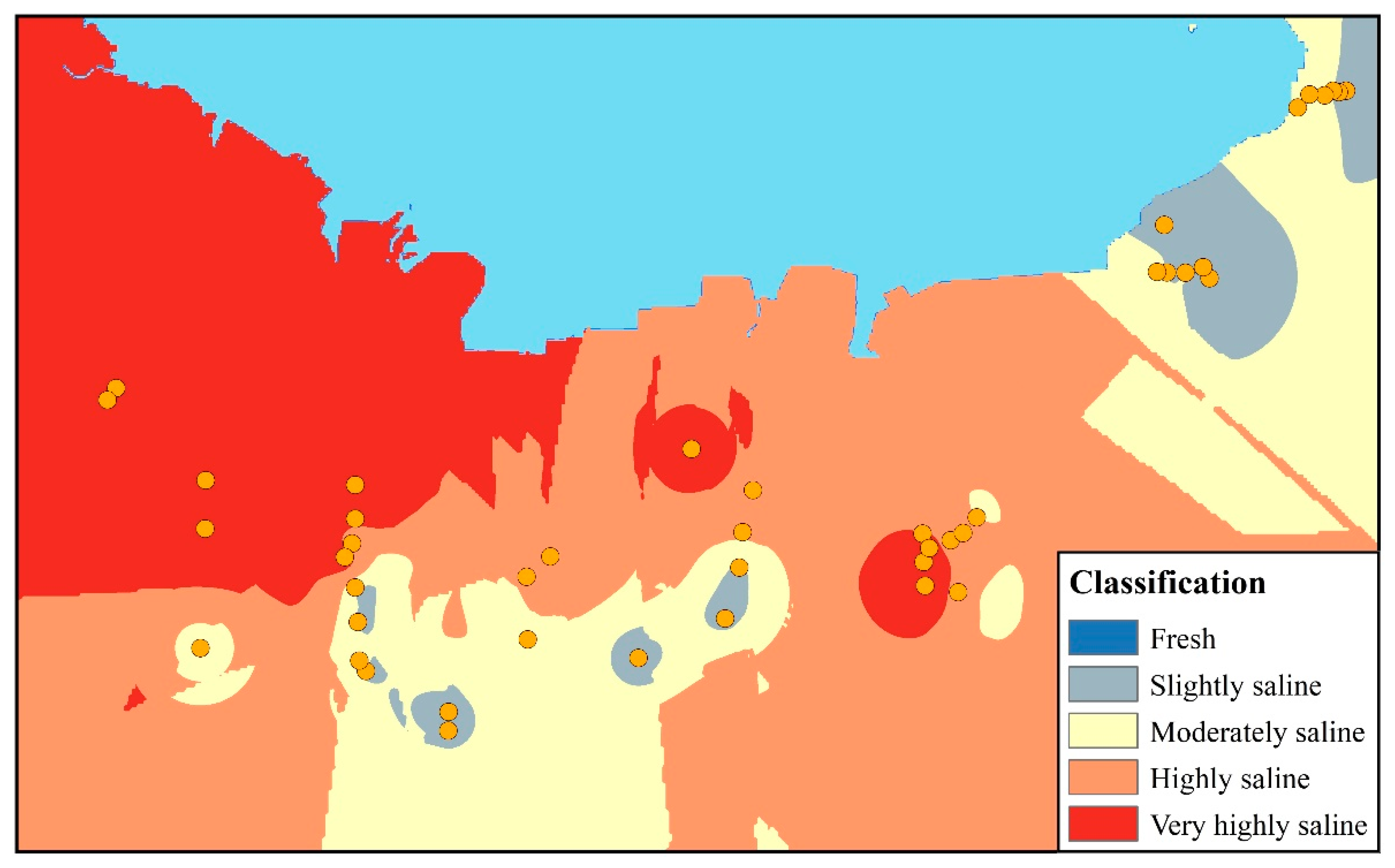
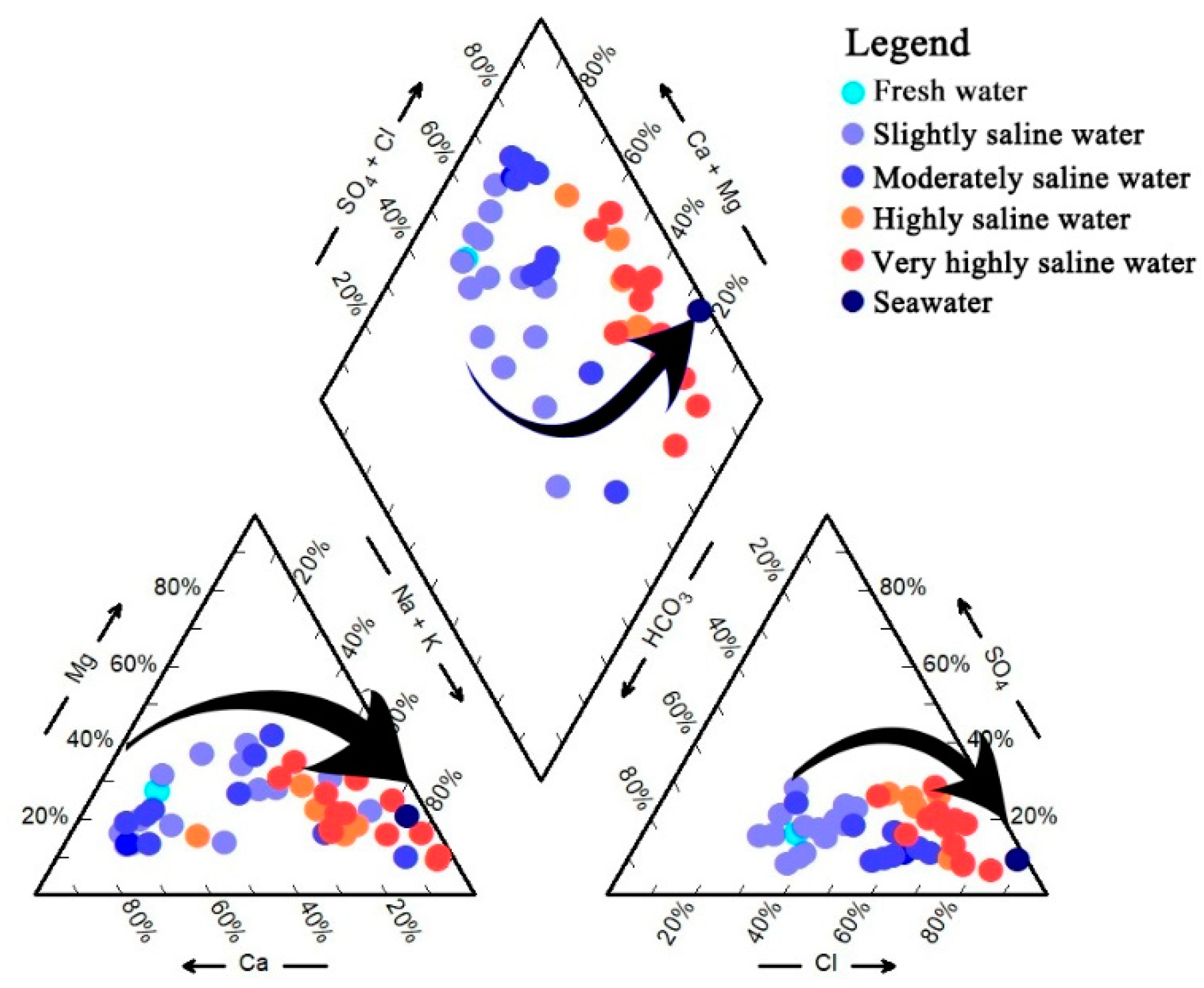
4.2. Status of Seawater Intrusion
5. Discussion
5.1. Hydrochemical Type Changes with the Deepening of Seawater Intrusion
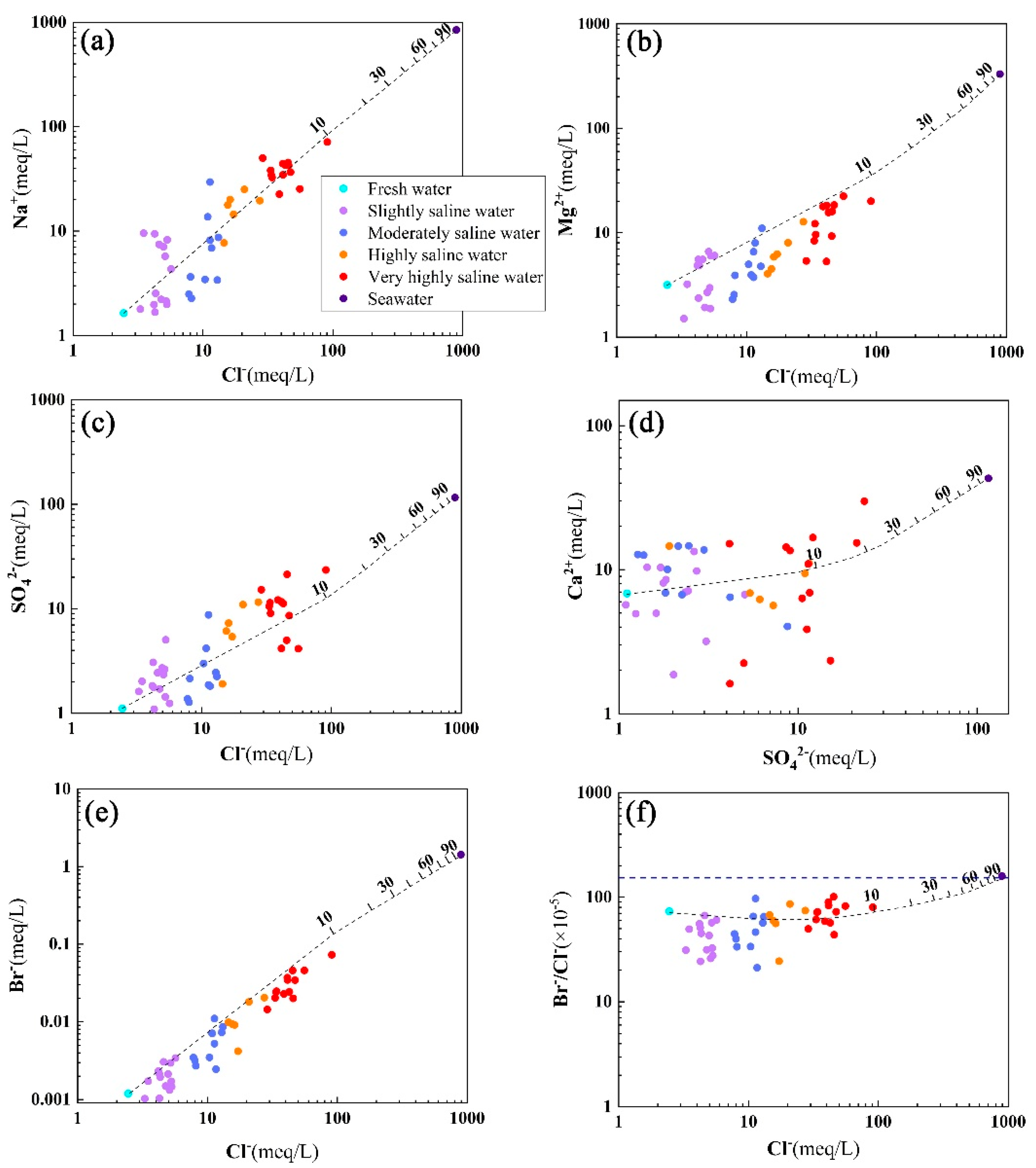
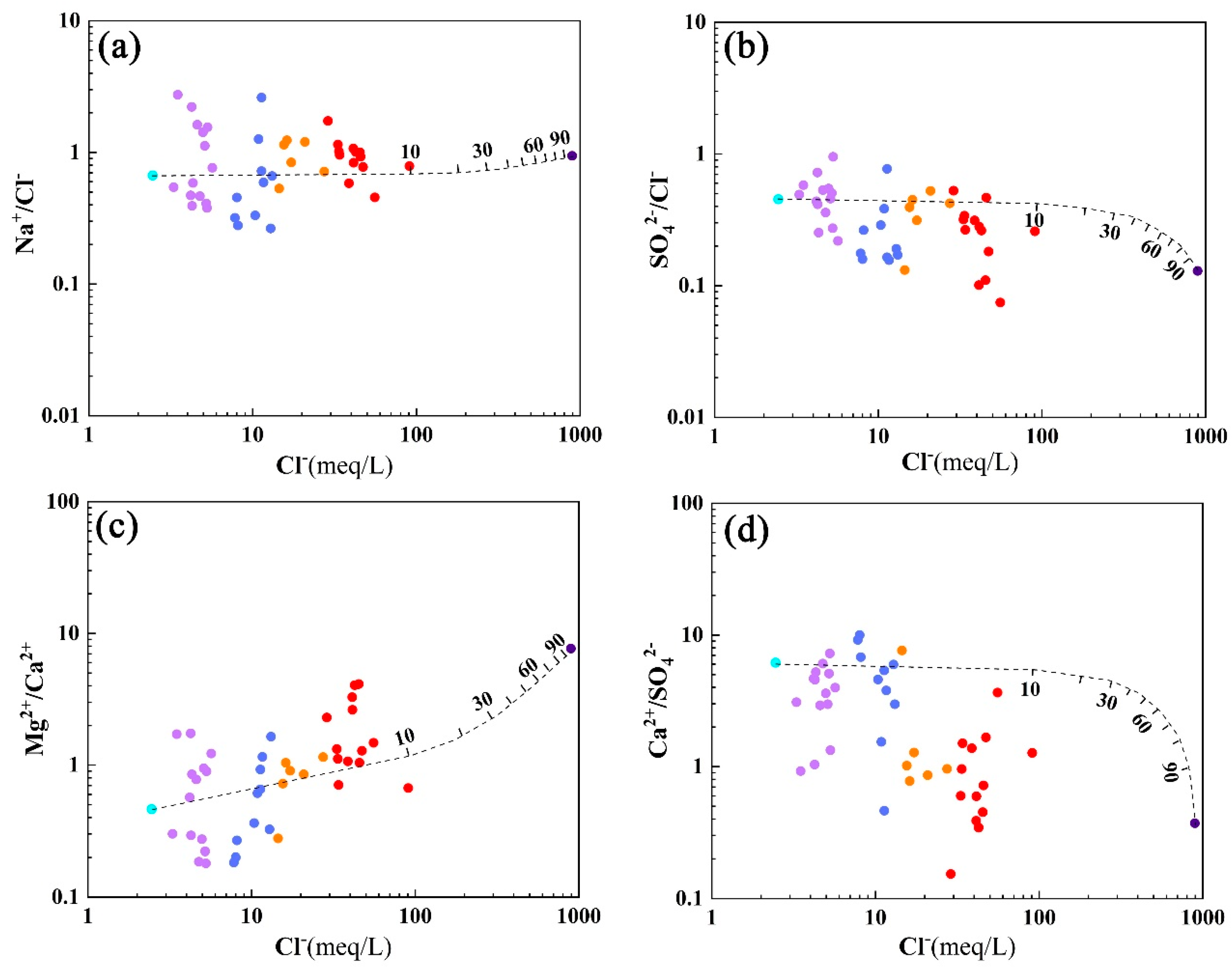
5.2. Variation Trends of Groundwater Ions
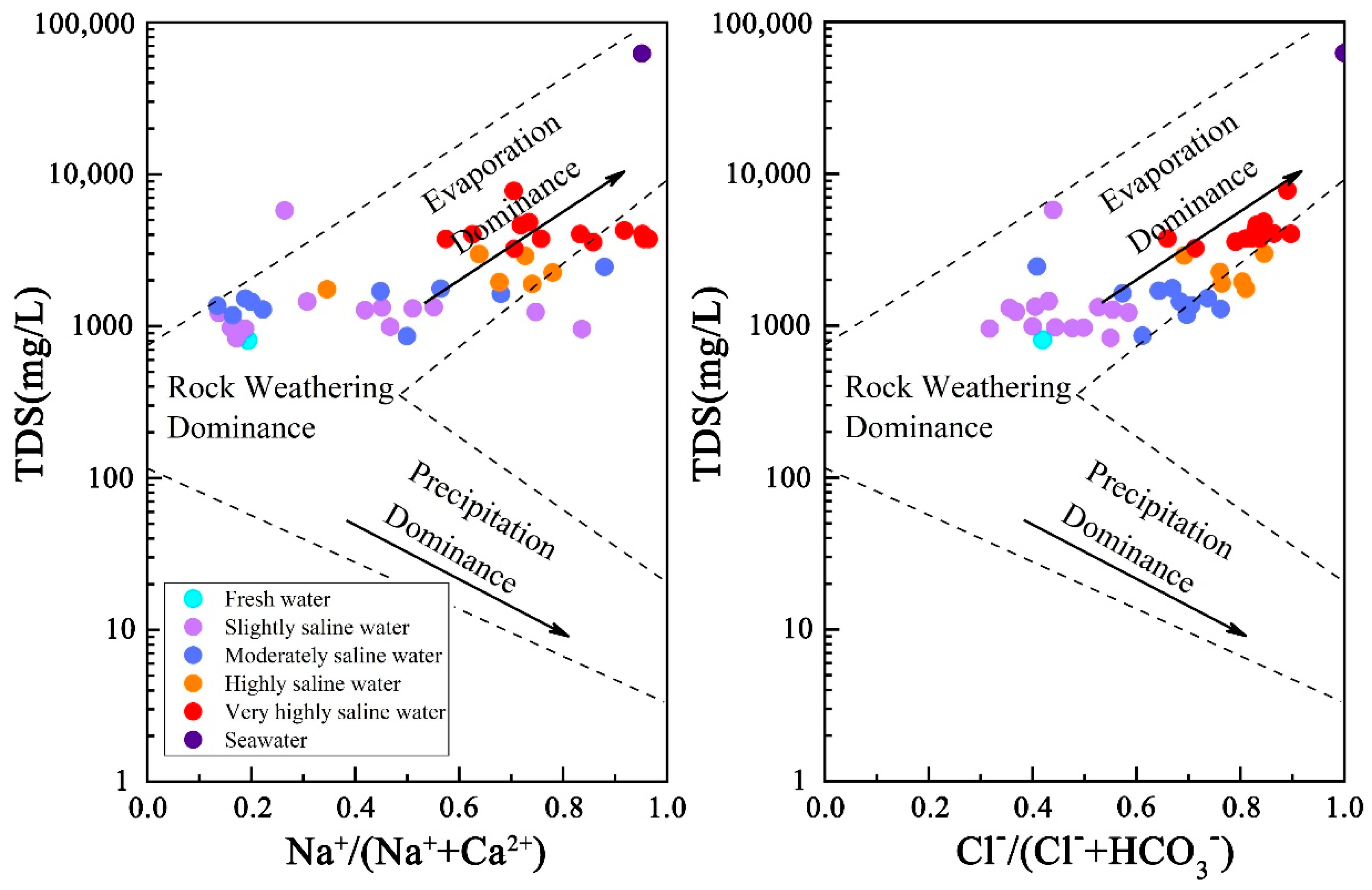
5.3. Material Sources of Groundwater
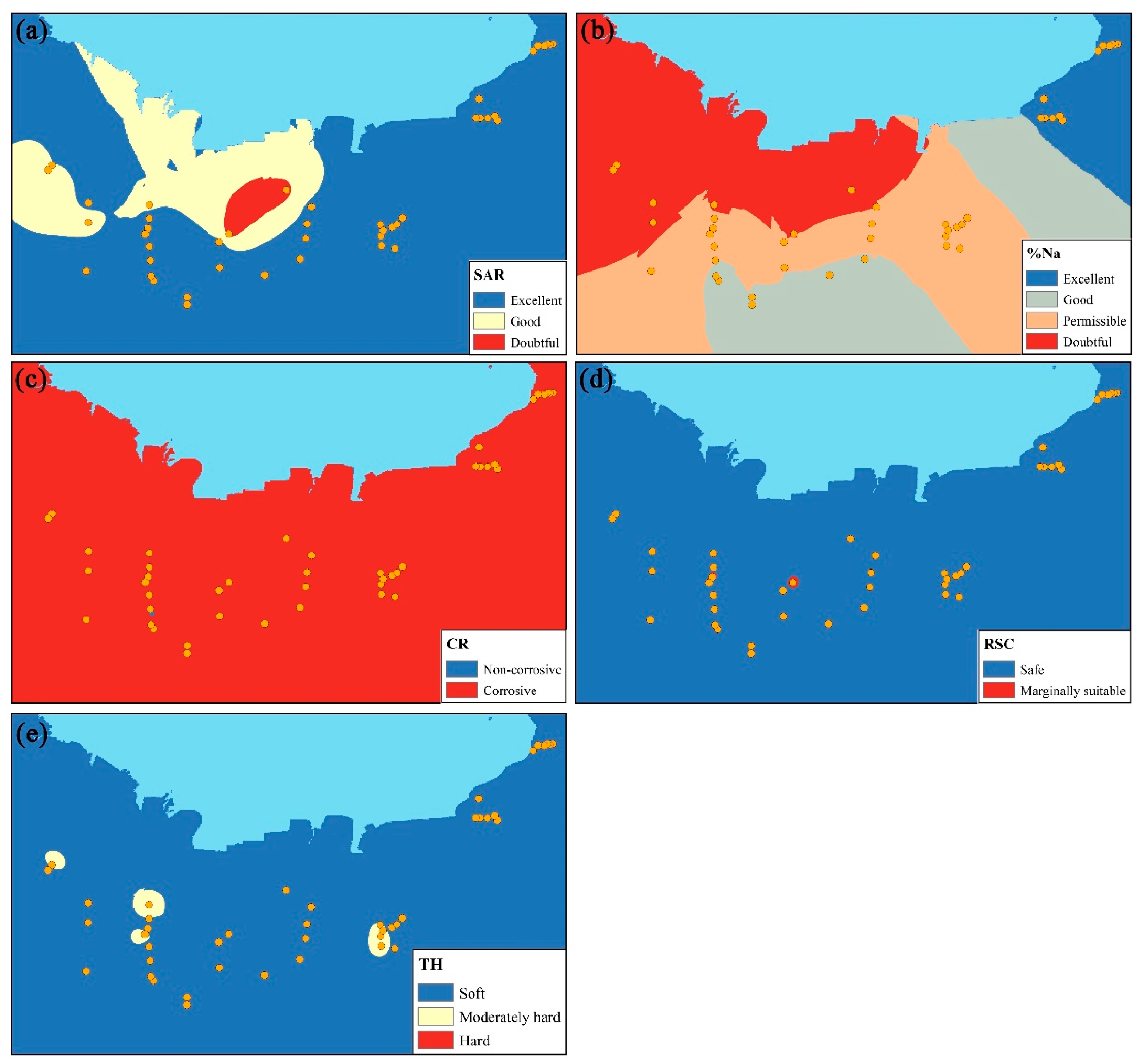
5.4. Groundwater Evaluation for Irrigation Purpose and Its Distribution
6. Conclusions
- (1)
- With the deepening of seawater intrusion, the contents of Na+, Ca2+, Mg2+, K+, Cl−, and SO42− in groundwater increase accordingly, and the hydrochemical type gradually changes from Ca-HCO3·Cl to Na·Mg-Cl·SO4 and then to Na-Cl type. Seawater intrusion is not an ideal mixing process between seawater and fresh groundwater. In the process of seawater intrusion, the chemical composition of groundwater is also changing due to the constant water–rock interaction of groundwater and aquifers. However, the influence of water–rock interaction on the composition of groundwater ions is lower than that of groundwater–seawater mixing.
- (2)
- The chemical composition of groundwater in areas invaded by seawater is obviously affected by seawater. With the deepening of seawater intrusion, the composition of substances in groundwater affected gradually changes from “Rock weathering dominated” to “Evaporation dominated”, showing that with the deepening of seawater intrusion, the influence of seawater on the hydrochemical composition of groundwater gradually exceeds that of mineral dissolution.
- (3)
- Affected by seawater intrusion, groundwater may not be suitable for irrigation, mainly due to high salinity and high hardness. When the Cl− concentration is greater than 7.1 meq/L, groundwater will corrode pipelines and instruments. When the Cl− concentration is greater than 14.1 meq/L, the groundwater hardness is too high, which may make the groundwater unsuitable for cultivation. When the Cl− concentration is greater than 28.2 meq/L, excessively high salinity of groundwater will have adverse effects on planting. The areas affected by seawater intrusion should actively take measures to curb the intensification of seawater intrusion to maintain the sustainable development of local society and economy.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Sample | Location | T (°C) | EC | TDS | pH | Na+ | K+ | Ca2+ | Mg2+ | Cl− | SO42− | HCO3− | Br− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S01 | Shouguang | 22.50 | 5.88 | 4030.00 | 7.02 | 794.49 | 3.09 | 138.39 | 221.11 | 1467.56 | 557.85 | 396.07 | 2.75 |
| S02 | Shouguang | 18.60 | 5.39 | 4030.00 | 6.85 | 1041.12 | 7.62 | 44.96 | 112.65 | 1606.18 | 239.26 | 530.13 | 3.65 |
| H01 | Hanting | 17.80 | 1.92 | 1449.50 | 6.69 | 58.20 | 0.93 | 113.94 | 58.87 | 153.33 | 52.41 | 347.33 | 0.16 |
| H02 | Hanting | 15.40 | 1.08 | 858.00 | 6.48 | 158.49 | 1.19 | 137.98 | 96.82 | 413.57 | 87.33 | 450.92 | 0.20 |
| H03 | Hanting | 15.60 | 1.24 | 988.00 | 6.42 | 99.74 | 1.65 | 98.90 | 73.48 | 201.35 | 59.61 | 517.94 | 0.27 |
| H04 | Hanting | 15.80 | 1.65 | 1306.50 | 6.35 | 171.28 | 3.33 | 142.59 | 67.32 | 162.90 | 117.04 | 505.76 | 0.24 |
| H05 | Hanting | 14.50 | 5.65 | 4608.50 | 6.30 | 842.97 | 9.34 | 286.55 | 223.94 | 1679.22 | 412.59 | 584.97 | 2.73 |
| H06 | Hanting | 19.40 | 3.37 | 2450.50 | 6.56 | 678.87 | 2.37 | 80.73 | 45.35 | 401.76 | 418.95 | 999.33 | 0.88 |
| H07 | Hanting | 17.90 | 4.30 | 3237.00 | 6.37 | 749.91 | 6.24 | 271.29 | 116.46 | 1208.71 | 433.53 | 834.80 | 1.97 |
| H08 | Hanting | 18.50 | 10.45 | 7767.50 | 6.50 | 1643.10 | 32.07 | 596.93 | 242.73 | 3222.22 | 1127.08 | 682.47 | 5.81 |
| H09 | Hanting | 13.00 | 1.47 | 1235.00 | 6.43 | 216.41 | 0.68 | 63.61 | 67.30 | 150.84 | 147.24 | 444.82 | 0.17 |
| H10 | Hanting | 12.70 | 1.56 | 1326.00 | 6.51 | 131.95 | 0.91 | 139.41 | 80.03 | 181.41 | 112.35 | 280.30 | 0.11 |
| H11 | Hanting | 18.30 | 5.04 | 3757.00 | 6.47 | 786.86 | 5.91 | 218.87 | 148.21 | 1194.13 | 548.17 | 444.82 | 1.93 |
| H12 | Hanting | 15.10 | 5.31 | 4270.50 | 6.50 | 988.89 | 7.76 | 77.08 | 189.39 | 1515.83 | 537.91 | 536.22 | 1.94 |
| H13 | Hanting | 15.10 | 1.20 | 955.50 | 6.76 | 219.77 | 0.71 | 37.38 | 38.98 | 123.77 | 97.02 | 457.01 | 0.14 |
| C01 | Changyi | 16.80 | 4.86 | 3744.00 | 6.80 | 1016.12 | 38.10 | 32.33 | 64.37 | 1463.37 | 200.07 | 487.48 | 2.95 |
| C02 | Changyi | 16.50 | 3.74 | 2899.00 | 6.58 | 576.77 | 18.31 | 188.29 | 97.23 | 740.64 | 525.38 | 566.69 | 1.43 |
| C03 | Changyi | 14.80 | 2.78 | 2249.00 | 6.64 | 460.62 | 20.59 | 112.90 | 71.35 | 574.68 | 349.11 | 310.77 | 0.72 |
| C04 | Changyi | 16.20 | 1.71 | 1332.50 | 6.55 | 189.73 | 18.59 | 134.23 | 73.13 | 188.35 | 242.16 | 475.29 | 0.12 |
| C05 | Changyi | 14.20 | 4.57 | 3750.50 | 6.21 | 1151.08 | 28.31 | 46.70 | 65.16 | 1023.94 | 728.92 | 907.93 | 1.15 |
| C06 | Changyi | 16.20 | 2.35 | 1898.00 | 6.11 | 408.65 | 10.20 | 124.37 | 54.70 | 551.41 | 293.71 | 292.49 | 0.74 |
| C07 | Changyi | 15.40 | 0.73 | 5785.00 | 6.61 | 41.17 | 1.41 | 99.73 | 18.23 | 116.80 | 77.54 | 255.93 | 0.08 |
| C08 | Changyi | 14.80 | 2.18 | 1761.50 | 6.36 | 199.83 | 0.87 | 134.07 | 133.86 | 466.56 | 108.06 | 396.07 | 0.69 |
| C09 | Changyi | 15.80 | 1.02 | 806.00 | 6.59 | 37.73 | 1.01 | 136.56 | 38.34 | 87.10 | 53.28 | 207.18 | 0.14 |
| P01 | Pingdu | 15.00 | 4.63 | 3744.00 | 6.37 | 518.81 | 29.88 | 334.28 | 216.58 | 1375.38 | 581.81 | 554.50 | 1.82 |
| P02 | Pingdu | 14.30 | 3.63 | 2977.00 | 6.54 | 450.29 | 40.66 | 221.71 | 154.77 | 971.94 | 554.35 | 304.67 | 1.63 |
| P03 | Pingdu | 15.00 | 5.99 | 4823.00 | 6.43 | 975.70 | 35.69 | 306.57 | 194.11 | 1621.67 | 1022.47 | 511.85 | 1.59 |
| P04 | Pingdu | 14.20 | 4.87 | 4017.00 | 6.48 | 582.38 | 46.96 | 302.82 | 271.60 | 1975.69 | 199.00 | 389.98 | 3.66 |
| P05 | Pingdu | 15.30 | 2.43 | 1943.50 | 6.49 | 332.72 | 12.74 | 137.65 | 75.68 | 611.01 | 258.77 | 255.93 | 0.34 |
| P06 | Pingdu | 15.10 | 2.11 | 1696.50 | 6.39 | 187.88 | 3.74 | 200.63 | 79.82 | 402.21 | 89.50 | 383.89 | 0.42 |
| P07 | Pingdu | 13.60 | 4.28 | 3568.50 | 6.49 | 877.15 | 7.01 | 126.45 | 101.63 | 1178.07 | 506.45 | 536.22 | 1.62 |
| P08 | Pingdu | 17.40 | 2.15 | 1631.50 | 6.66 | 315.39 | 21.53 | 128.84 | 47.87 | 385.74 | 200.49 | 493.57 | 0.57 |
| L01 | Laizhou | 14.30 | 1.19 | 968.50 | 6.22 | 50.93 | 1.20 | 207.20 | 23.35 | 169.08 | 82.07 | 292.49 | 0.12 |
| L02 | Laizhou | 14.10 | 1.18 | 976.00 | 6.22 | 45.81 | 1.20 | 207.99 | 22.79 | 186.68 | 68.93 | 402.17 | 0.14 |
| L03 | Laizhou | 14.20 | 1.44 | 1176.50 | 6.26 | 57.19 | 1.17 | 252.34 | 28.04 | 277.03 | 65.99 | 207.18 | 0.28 |
| L04 | Laizhou | 14.90 | 1.60 | 1287.00 | 6.29 | 83.71 | 0.72 | 254.53 | 30.98 | 284.32 | 61.11 | 152.34 | 0.25 |
| L05 | Laizhou | 21.90 | 2.54 | 1748.50 | 6.52 | 177.42 | 1.34 | 291.72 | 49.21 | 515.41 | 91.75 | 207.18 | 0.78 |
| L06 | Laizhou | 16.40 | 1.94 | 1521.00 | 6.36 | 78.25 | 1.16 | 292.91 | 57.85 | 457.18 | 117.92 | 280.30 | 0.58 |
| L07 | Laizhou | 14.70 | 1.02 | 832.00 | 6.56 | 38.53 | 0.87 | 161.88 | 28.74 | 151.61 | 84.92 | 213.27 | 0.08 |
| L08 | Laizhou | 13.90 | 1.47 | 1222.00 | 6.48 | 49.06 | 1.15 | 267.38 | 36.02 | 184.86 | 126.06 | 225.46 | 0.24 |
| L09 | Laizhou | 14.30 | 1.65 | 1358.50 | 6.59 | 52.13 | 4.75 | 291.39 | 47.39 | 288.69 | 103.06 | 207.18 | 0.22 |
| L10 | Laizhou | 13.20 | 1.73 | 1443.00 | 6.51 | 79.21 | 1.05 | 274.37 | 60.51 | 367.82 | 143.36 | 292.49 | 0.28 |
| L11 | Laizhou | 14.10 | 1.54 | 1274.00 | 6.55 | 162.48 | 1.76 | 195.84 | 32.54 | 176.57 | 130.45 | 243.74 | 0.17 |
| L12 | Laizhou | 12.40 | 1.12 | 962.00 | 6.52 | 45.41 | 2.07 | 170.58 | 58.90 | 148.64 | 87.86 | 280.30 | 0.19 |
References
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Todd, D.K.; Mays, L.W. Groundwater Hydrology; John Wiley & Sons: Hoboken, NJ, USA, 2004. [Google Scholar]
- Wang, S.; Gao, Z.; Wang, Z.; Wu, X.; An, Y.; Ren, X.; He, M.; Wang, W.; Liu, J. Hydrodynamic characteristics of groundwater aquifer system under recharge and discharge conditions. Arab. J. Geosci. 2020, 13, 1–12. [Google Scholar] [CrossRef]
- Merkel, B.J.; Planer-Friedrich, B.; Nordstrom, D.K. Groundwater Geochemistry. In A Practical Guide to Modeling of Natural and Contaminated Aquatic Systems; Springer: Berlin, Germany, 2005; p. 2. [Google Scholar]
- Matthess, G.; Frimmel, F.; Hirsch, P.; Schulz, H.; Usdowski, H. Progress in Hydrogeochemistry: Organics, Carbonate Systems, Silicate Systems, Microbiology, Models; Springer: Berlin, Germany, 1992. [Google Scholar]
- Gao, Z.; Wang, Z.; Wang, S.; Wu, X.; An, Y.; Wang, W.; Liu, J. Factors that influence the chemical composition and evolution of shallow groundwater in an arid region: A case study from the middle reaches of the Heihe River, China. Environ. Earth Sci. 2019, 78, 390. [Google Scholar] [CrossRef]
- Fitts, C.R. Groundwater Science; Elsevier: Amsterdam, The Netherlands, 2002. [Google Scholar]
- Famiglietti, J.S. The global groundwater crisis. Nat. Clim. Chang. 2014, 4, 945–948. [Google Scholar] [CrossRef]
- Liu, J.; Gao, Z.; Wang, Z.; Xu, X.; Su, Q.; Wang, S.; Qu, W.; Xing, T. Hydrogeochemical processes and suitability assessment of groundwater in the Jiaodong Peninsula, China. Environ. Monit. Assess. 2020, 192, 384. [Google Scholar] [CrossRef] [PubMed]
- Bear, J.; Cheng, A.H.-D.; Sorek, S.; Ouazar, D.; Herrera, I. Seawater Intrusion in Coastal Aquifers: Concepts, Methods and Practices; Springer Science & Business Media: Berlin, Germany, 1999. [Google Scholar]
- Mahmoodzadeh, D.; Karamouz, M. Seawater intrusion in heterogeneous coastal aquifers under flooding events. J. Hydrol. 2019, 568, 1118–1130. [Google Scholar] [CrossRef]
- Badaruddin, S.; Werner, A.D.; Morgan, L.K. Characteristics of active seawater intrusion. J. Hydrol. 2017, 551, 632–647. [Google Scholar] [CrossRef] [Green Version]
- Xiao, H.; Wang, D.; Medeiros, S.C.; Bilskie, M.V.; Hagen, S.C.; Hall, C.R. Exploration of the effects of storm surge on the extent of saltwater intrusion into the surficial aquifer in coastal east-central Florida (USA). Sci. Total Environ. 2019, 648, 1002–1017. [Google Scholar] [CrossRef]
- Parizi, E.; Hosseini, S.M.; Ataie-Ashtiani, B.; Simmons, C.T. Vulnerability mapping of coastal aquifers to seawater intrusion: Review, development and application. J. Hydrol. 2019, 570, 555–573. [Google Scholar] [CrossRef]
- Arslan, H.; Demir, Y. Impacts of seawater intrusion on soil salinity and alkalinity in Bafra Plain, Turkey. Environ. Monit. Assess. 2013, 185, 1027–1040. [Google Scholar] [CrossRef]
- Hussain, M.S.; Abd-Elhamid, H.F.; Javadi, A.A.; Sherif, M.M. Management of seawater intrusion in coastal aquifers: A review. Water 2019, 11, 2467. [Google Scholar] [CrossRef] [Green Version]
- Shi, W.; Lu, C.; Ye, Y.; Wu, J.; Li, L.; Luo, J. Assessment of the impact of sea-level rise on steady-state seawater intrusion in a layered coastal aquifer. J. Hydrol. 2018, 563, 851–862. [Google Scholar] [CrossRef]
- Herzberg, A. Die Wasserversorgung einiger Nordseebäder: Journal für Gasbeleuchtung und Wasserversorgung. J. Hydrol. 2018, 26, 1789–1799. [Google Scholar]
- Luyun Jr, R.; Momii, K.; Nakagawa, K. Effects of recharge wells and flow barriers on seawater intrusion. Groundwater 2011, 49, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elhamid, H.; Javadi, A.; Abdelaty, I.; Sherif, M. Simulation of seawater intrusion in the Nile Delta aquifer under the conditions of climate change. Hydrol. Res. 2016, 47, 1198–1210. [Google Scholar] [CrossRef]
- Shi, W.; Lu, C.; Werner, A.D. Assessment of the impact of sea-level rise on seawater intrusion in sloping confined coastal aquifers. J. Hydrol. 2020, 586, 124872. [Google Scholar] [CrossRef]
- Liu, S.; Tang, Z.; Gao, M.; Hou, G. Evolutionary process of saline-water intrusion in Holocene and Late Pleistocene groundwater in southern Laizhou Bay. Sci. Total Environ. 2017, 607, 586–599. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wei, J.; Wang, H.; Shi, L.; Gao, Z.; Liu, S.; Ning, F.; Jia, C.; Ji, Y.; Dong, F. Discussion on the fluorosis in seawater-intrusion areas along coastal zones in Laizhou Bay and other parts of China. Int. J. Environ. Res. 2019, 13, 435–442. [Google Scholar] [CrossRef]
- Qi, H.; Ma, C.; He, Z.; Hu, X.; Gao, L. Lithium and its isotopes as tracers of groundwater salinization: A study in the southern coastal plain of Laizhou Bay, China. Sci. Total Environ. 2019, 650, 878–890. [Google Scholar] [CrossRef]
- Al Maliki, A.A.; Abbass, Z.D.; Hussain, H.M.; Al-Ansari, N. Assessment of the groundwater suitability for irrigation near Al Kufa City and preparing the final water quality maps using spatial distribution tools. Environ. Earth Sci. 2020, 79, 1–12. [Google Scholar] [CrossRef]
- Jeon, C.; Raza, M.; Lee, J.-Y.; Kim, H.; Kim, C.-S.; Kim, B.; Kim, J.-W.; Kim, R.-H.; Lee, S.-W. Countrywide Groundwater Quality Trend and Suitability for Use in Key Sectors of Korea. Water 2020, 12, 1193. [Google Scholar] [CrossRef]
- Piper, A. A Graphic Procedure in the Geochemical Interpretation of Water-Analyses. Neurochem. Int. 1984, 6, 27–39. [Google Scholar] [CrossRef]
- Gibbs, R.J. Mechanisms controlling world water chemistry. Science 1970, 170, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Mahanta, N.; Mishra, I.; Hatui, A.; Mahanta, P.; Sahoo, H.; Goswami, S. Geochemical appraisal of groundwater qualities and its uses in and around Maneswar Block of Sambalpur District, Odisha, India. Environ. Earth Sci. 2020, 79, 5. [Google Scholar] [CrossRef]
- Rao, N.S. Controlling factors of fluoride in groundwater in a part of South India. Arab. J. Geosci. 2017, 10, 524. [Google Scholar]
- Richard, L. Diagnosis and Improvement of Saline and Alkali Soils. USDA Hand Book. No. 60; US Goverment. Press: Washington, DC, USA, 1954; p. 160. [Google Scholar]
- Wilcox, L. Classification and Use of Irrigation Waters; US Department of Agriculture: Washington, DC, USA, 1955. [Google Scholar]
- Raman, V. Impact of corrosion in the conveyance and distribution of water. J. Indian Water Works Assoc. 1983, 15, 115–121. [Google Scholar]
- Tomaszkiewicz, M.; Abou Najm, M.; El-Fadel, M. Development of a groundwater quality index for seawater intrusion in coastal aquifers. Environ. Model. Softw. 2014, 57, 13–26. [Google Scholar] [CrossRef]
- Khan, A.F.; Srinivasamoorthy, K.; Prakash, R.; Rabina, C. Hydrochemical and statistical techniques to decode groundwater geochemical interactions and saline water intrusion along the coastal regions of Tamil Nadu and Puducherry, India. Environ. Geochem. Health 2020, 1–17. [Google Scholar] [CrossRef]
- Sae-Ju, J.; Chotpantarat, S.; Thitimakorn, T. Assessment of seawater intrusion using multivariate statistical, hydrochemical and geophysical techniques in coastal aquifer, Cha-am district, Thailand. Hydrol. Earth Syst. Sci. Discuss. 2018, 1–50. [Google Scholar] [CrossRef]
- Papatheodorou, G.; Lambrakis, N.; Panagopoulos, G. Application of multivariate statistical procedures to the hydrochemical study of a coastal aquifer: An example from Crete, Greece. Hydrol. Process. Int. J. 2007, 21, 1482–1495. [Google Scholar] [CrossRef]
- Sridharan, M.; Nathan, D.S. Hydrochemical facies and ionic exchange in coastal aquifers of Puducherry region, India: Implications for seawater intrusion. Earth Syst. Environ. 2017, 1, 5. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Song, S.-H. Groundwater chemistry and ionic ratios in a western coastal aquifer of Buan, Korea: Implication for seawater intrusion. Geosci. J. 2007, 11, 259–270. [Google Scholar] [CrossRef]
- Park, S.-C.; Yun, S.-T.; Chae, G.-T.; Yoo, I.-S.; Shin, K.-S.; Heo, C.-H.; Lee, S.-K. Regional hydrochemical study on salinization of coastal aquifers, western coastal area of South Korea. J. Hydrol. 2005, 313, 182–194. [Google Scholar] [CrossRef]
- Han, D.; Kohfahl, C.; Song, X.; Xiao, G.; Yang, J. Geochemical and isotopic evidence for palaeo-seawater intrusion into the south coast aquifer of Laizhou Bay, China. Appl. Geochem. 2011, 26, 863–883. [Google Scholar] [CrossRef]
- Kharroubi, A.; Tlahigue, F.; Agoubi, B.; Azri, C.; Bouri, S. Hydrochemical and statistical studies of the groundwater salinization in Mediterranean arid zones: Case of the Jerba coastal aquifer in southeast Tunisia. Environ. Earth Sci. 2012, 67, 2089–2100. [Google Scholar] [CrossRef]
- Davis, S.N.; Whittemore, D.O.; Fabryka-Martin, J. Uses of chloride/bromide ratios in studies of potable water. Groundwater 1998, 36, 338–350. [Google Scholar] [CrossRef]
- Han, D.M.; Song, X.F.; Currell, M.J.; Yang, J.L.; Xiao, G.Q. Chemical and isotopic constraints on evolution of groundwater salinization in the coastal plain aquifer of Laizhou Bay, China. J. Hydrol. 2014, 508, 12–27. [Google Scholar] [CrossRef]
- Chafouq, D.; El Mandour, A.; Elgettafi, M.; Himi, M.; Chouikri, I.; Casas, A. Hydrochemical and isotopic characterization of groundwater in the Ghis-Nekor plain (northern Morocco). J. Afr. Earth Sci. 2018, 139, 1–13. [Google Scholar] [CrossRef]
- Terzić, J.; Marković, T.; Pekaš, Ž. Influence of sea-water intrusion and agricultural production on the Blato Aquifer, Island of Korčula, Croatia. Environ. Geol. 2008, 54, 719–729. [Google Scholar] [CrossRef]
- Sappa, G.; Ergul, S.; Ferranti, F.; Sweya, L.N.; Luciani, G. Effects of seasonal change and seawater intrusion on water quality for drinking and irrigation purposes, in coastal aquifers of Dar es Salaam, Tanzania. J. Afr. Earth Sci. 2015, 105, 64–84. [Google Scholar] [CrossRef]
- Milnes, E.; Renard, P. The problem of salt recycling and seawater intrusion in coastal irrigated plains: An example from the Kiti aquifer (Southern Cyprus). J. Hydrol. 2004, 288, 327–343. [Google Scholar] [CrossRef] [Green Version]
- Abdoulhalik, A.; Ahmed, A.; Hamill, G. A new physical barrier system for seawater intrusion control. J. Hydrol. 2017, 549, 416–427. [Google Scholar] [CrossRef] [Green Version]
- Nishikawa, T.; Siade, A.J.; Reichard, E.G.; Ponti, D.J.; Canales, A.; Johnson, T. Stratigraphic controls on seawater intrusion and implications for groundwater management, Dominguez Gap area of Los Angeles, California, USA. Hydrogeol. J. 2009, 17, 1699. [Google Scholar] [CrossRef]

| Indicator | Equation | Range | Classification | Reference |
|---|---|---|---|---|
| SAR | <10 | Excellent | [31] | |
| 10–18 | Good | |||
| 18–26 | Doubtful | |||
| >26 | Unsuitable | |||
| %Na | <20 | Excellent | [32] | |
| 20–40 | Good | |||
| 40–60 | Permissible | |||
| 60–80 | Doubtful | |||
| >80 | Unsuitable | |||
| CR | ≤1 | Non-corrosive | [33] | |
| >1 | Corrosive | |||
| RSC | <1.25 | Safe | [31] | |
| 1.25–2.5 | Marginally suitable | |||
| 2.5–5 | Unsuitable | |||
| >5 | Harmful | |||
| TH | <75 | Soft | [2] | |
| 75–150 | Moderately hard | |||
| 150–300 | Hard | |||
| >300 | Very hard |
| Parameters | Maximum | Minimum | Average | CV |
|---|---|---|---|---|
| Ca2+ | 596.93 | 32.33 | 183.75 | 0.57 |
| Mg2+ | 271.60 | 18.23 | 91.30 | 0.73 |
| Na+ | 1643.10 | 37.73 | 405.10 | 0.98 |
| K+ | 46.96 | 0.68 | 9.95 | 1.30 |
| Cl− | 3222.22 | 87.10 | 696.03 | 0.97 |
| SO42− | 1127.08 | 52.41 | 275.07 | 0.94 |
| HCO3− | 999.33 | 152.34 | 416.99 | 0.45 |
| Br− | 5.81 | 0.08 | 1.04 | 1.19 |
| TDS | 7767.50 | 806.00 | 2401.48 | 0.65 |
| EC | 10.45 | 0.73 | 2.91 | 0.68 |
| pH | 7.02 | 6.11 | 6.49 | 0.03 |
| Classification | Cl− (meq/L) | TDS (mg/L) | EC (μs/cm) | Sample ID | (%) |
|---|---|---|---|---|---|
| Freshwater | <2.8 | 0–500 | <700 | C09 | 2.27 |
| Slightly saline water | 2.8–7.1 | 500–1500 | 700–2000 | C04,C07,H01,H03,H04,H09,H10, H13,L01,L02,L07,L08,L11,L12 | 31.82 |
| Moderately saline water | 7.1–14.1 | 1500–7000 | 2000–10,000 | C08,H02,H06,L03,L04,L06,L09, L10,P06,P08 | 22.73 |
| Highly saline water | 14.1–28.2 | 7000–15,000 | 10,000–25,000 | C02,C03,C06,L05,P02,P05 | 13.64 |
| Very highly saline water | 28.2–282.2 | 15,000–35,000 | 25,000–45,000 | C01,C05,H05,H07,H08,H11,H12, P01,P03,P04,P07,S01,S02 | 29.54 |
| Seawater | >282.2 | >35,000 | >45,000 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; Wang, S.; Liu, W.; Su, Q.; Tong, H.; Xu, X.; Gao, Z.; Liu, J. Hydrochemical Characteristics and Irrigation Suitability Evaluation of Groundwater with Different Degrees of Seawater Intrusion. Water 2020, 12, 3460. https://doi.org/10.3390/w12123460
Wang Z, Wang S, Liu W, Su Q, Tong H, Xu X, Gao Z, Liu J. Hydrochemical Characteristics and Irrigation Suitability Evaluation of Groundwater with Different Degrees of Seawater Intrusion. Water. 2020; 12(12):3460. https://doi.org/10.3390/w12123460
Chicago/Turabian StyleWang, Zhenyan, Shu Wang, Wenyue Liu, Qiao Su, Hui Tong, Xingyong Xu, Zongjun Gao, and Jiutan Liu. 2020. "Hydrochemical Characteristics and Irrigation Suitability Evaluation of Groundwater with Different Degrees of Seawater Intrusion" Water 12, no. 12: 3460. https://doi.org/10.3390/w12123460







