Characterization of 1,4-Dioxane Biodegradation by a Microbial Community
Abstract
1. Introduction
2. Materials and Methods
2.1. Enrichment
2.2. Mineralization by Mixed Culture
2.3. Biodegradation at Various Initial Concentrations and Degradation Kinetics of 1,4-dioxane
2.4. Effect of Extra Carbon Sources on 1,4-Dioxane Biodegradation
2.5. Analytical Procedures
3. Results and Discussions
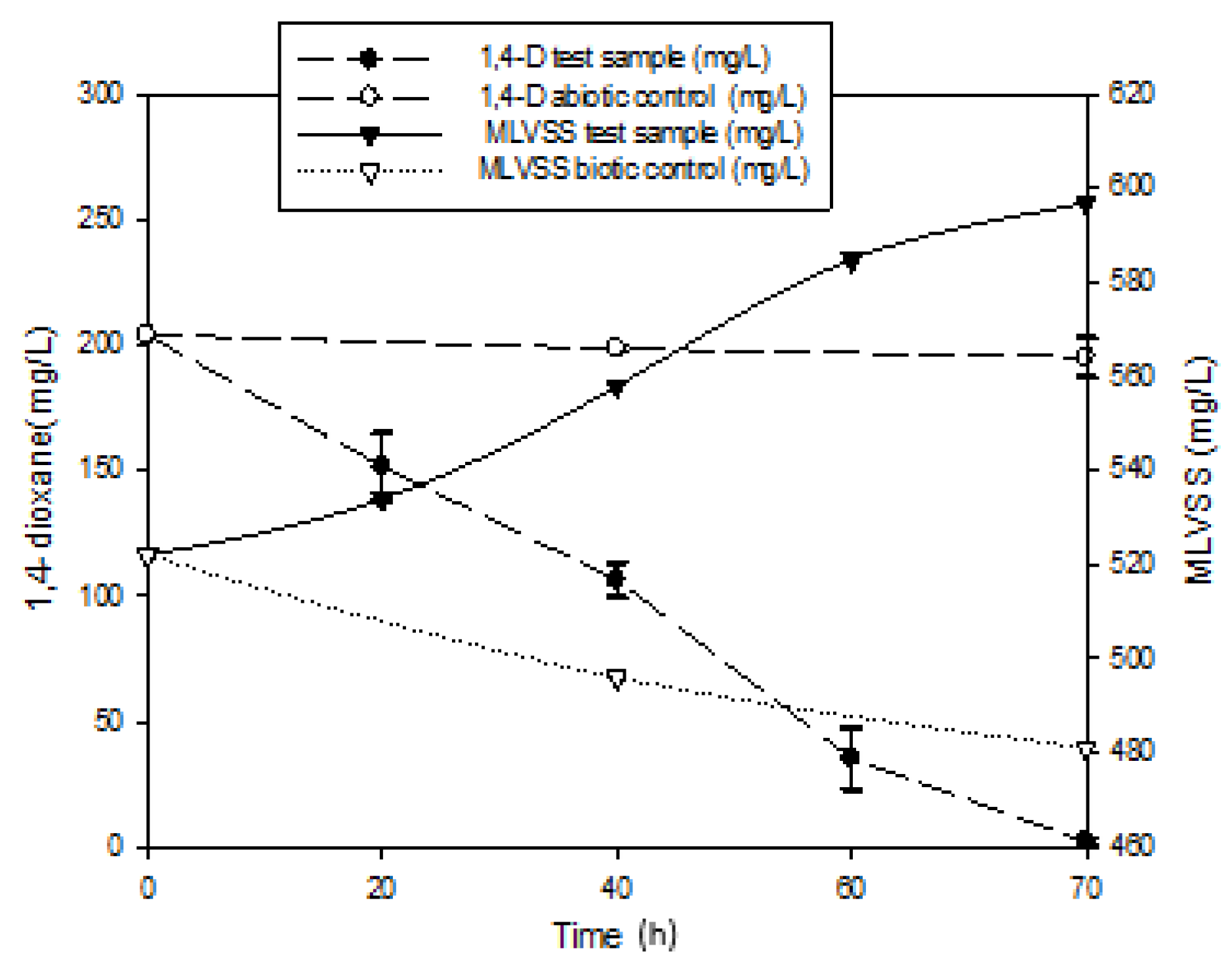
3.1. Mineralization of 1,4-Dioxane
- Cell synthesis from 1,4-dioxane:3C4H8O2 + 5O2 + 2NH3 = 2C5H7NO2 + 2CO2 + 8H2O
- Energy production reaction: (CO2 recovered from 1,4-dioxane)C4H8O2 + 5O2 = 4CO2 + 4H2O
- Biomass decayC5H7NO2 + 5O2 = 5CO2 + NH3 + 2H2O
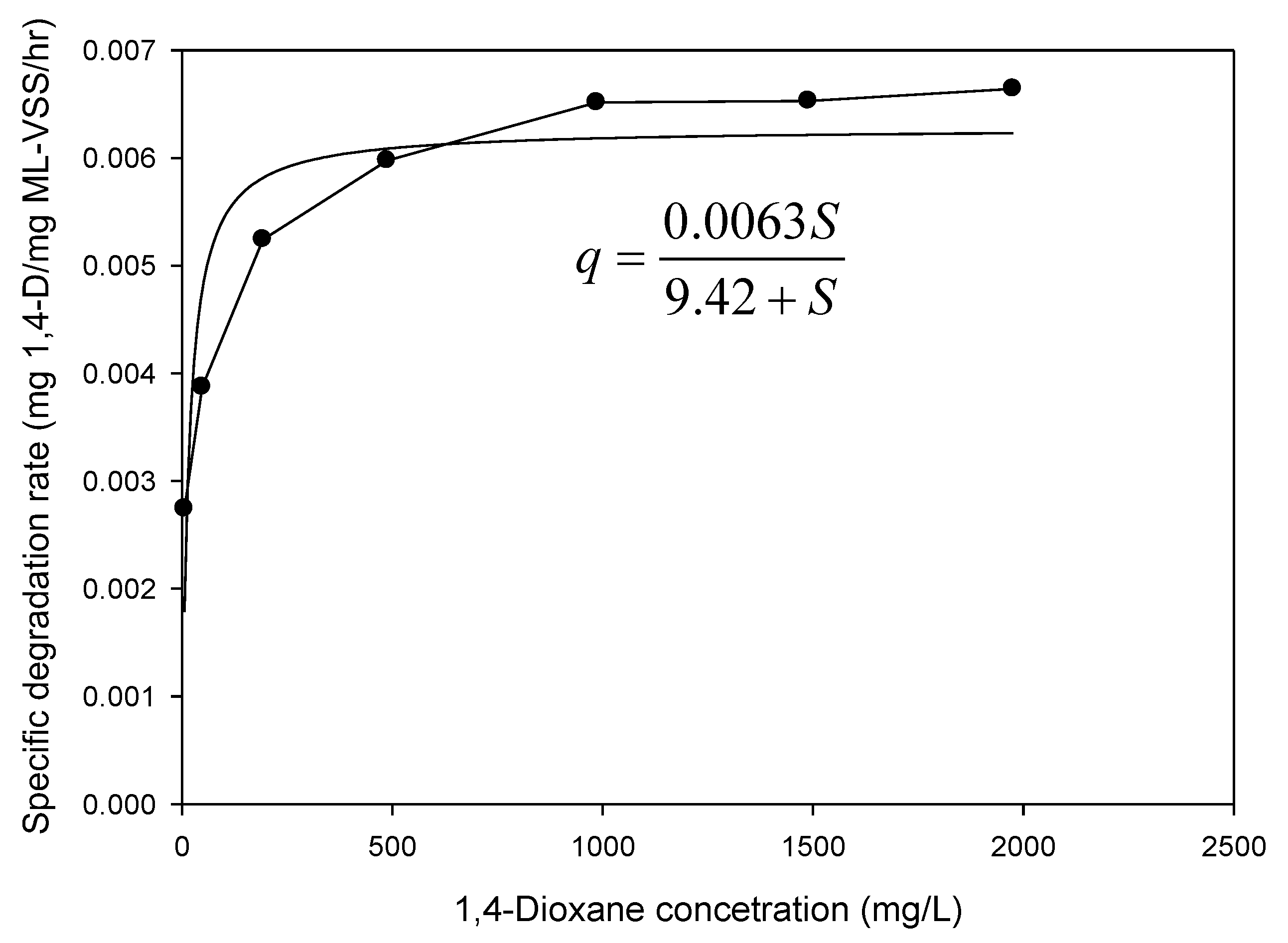
3.2. Kinetics Parameters Estimation
3.2.1. Endogenous or Decay Rate
3.2.2. Observed Cell Yield
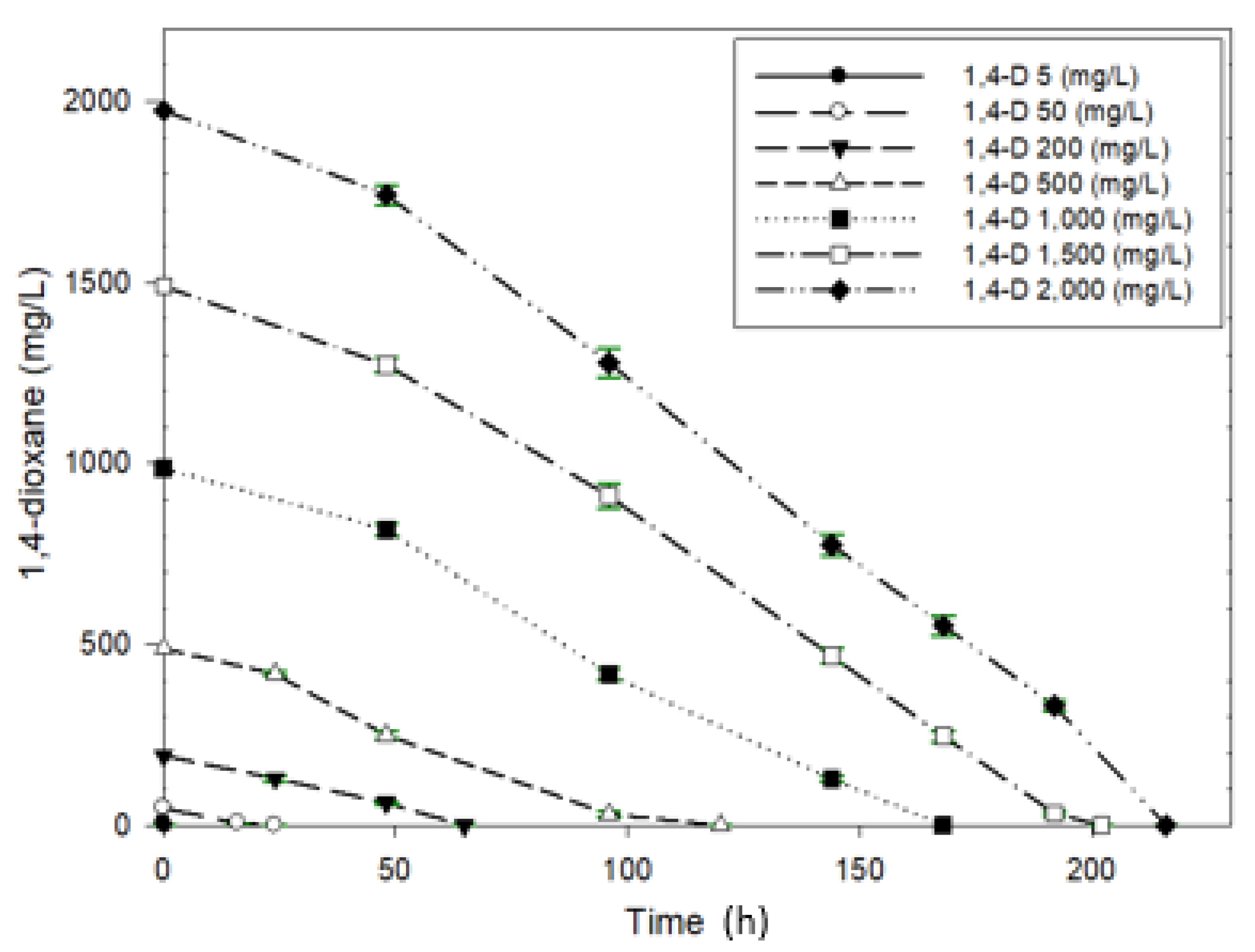
3.2.3. Biodegradation of 1,4-Dioxane with Different Concentrations and Kinetics
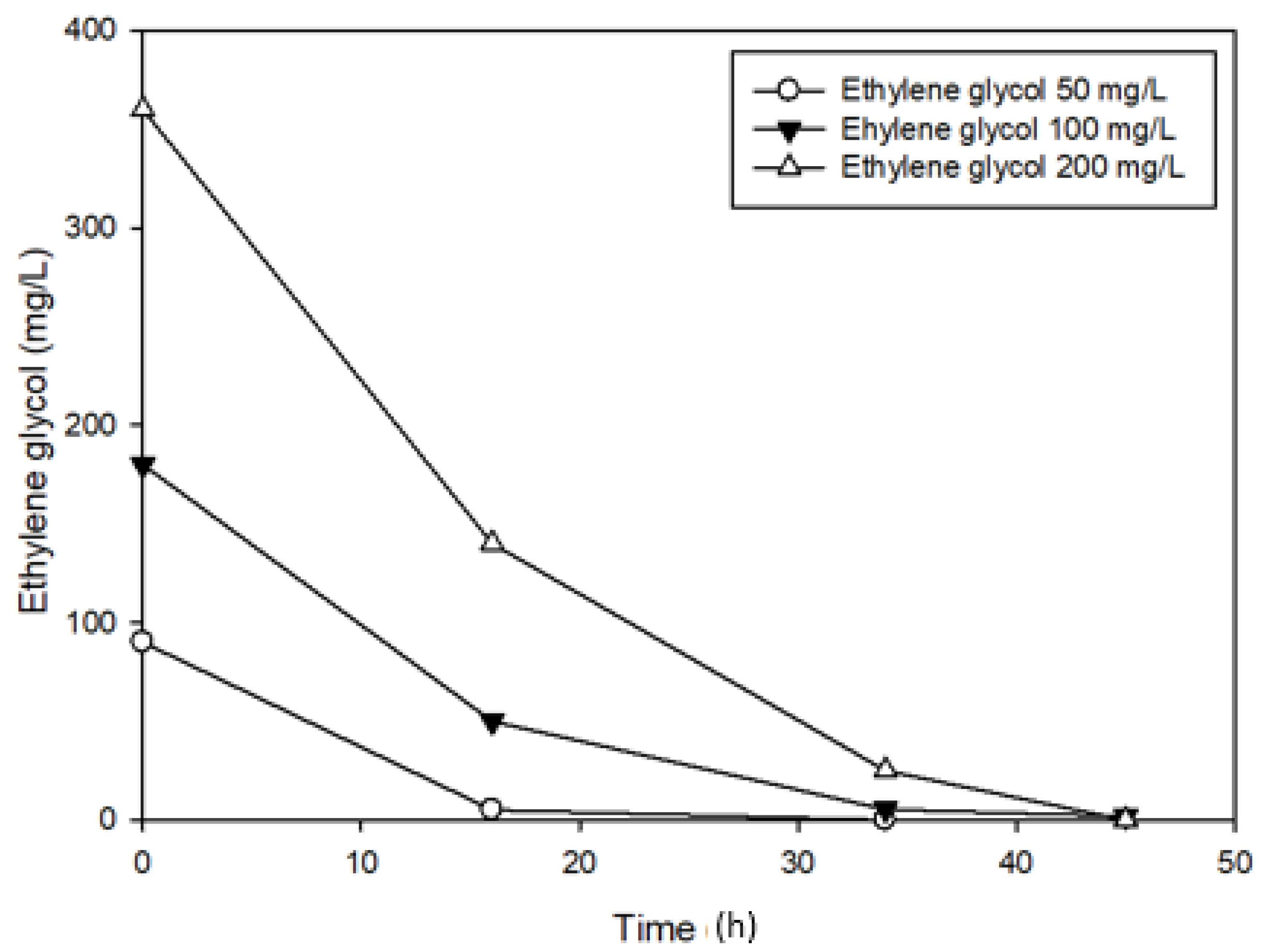
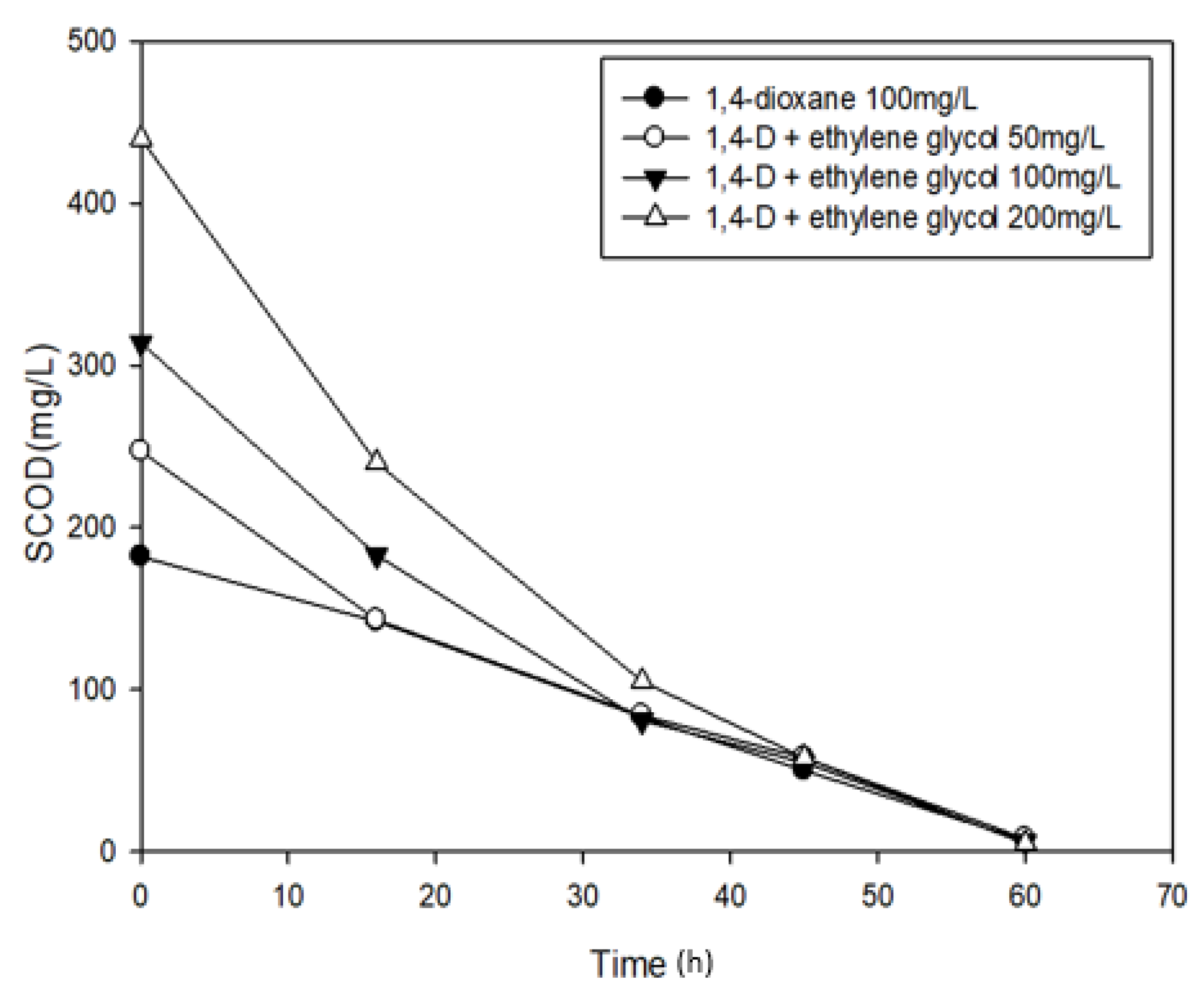
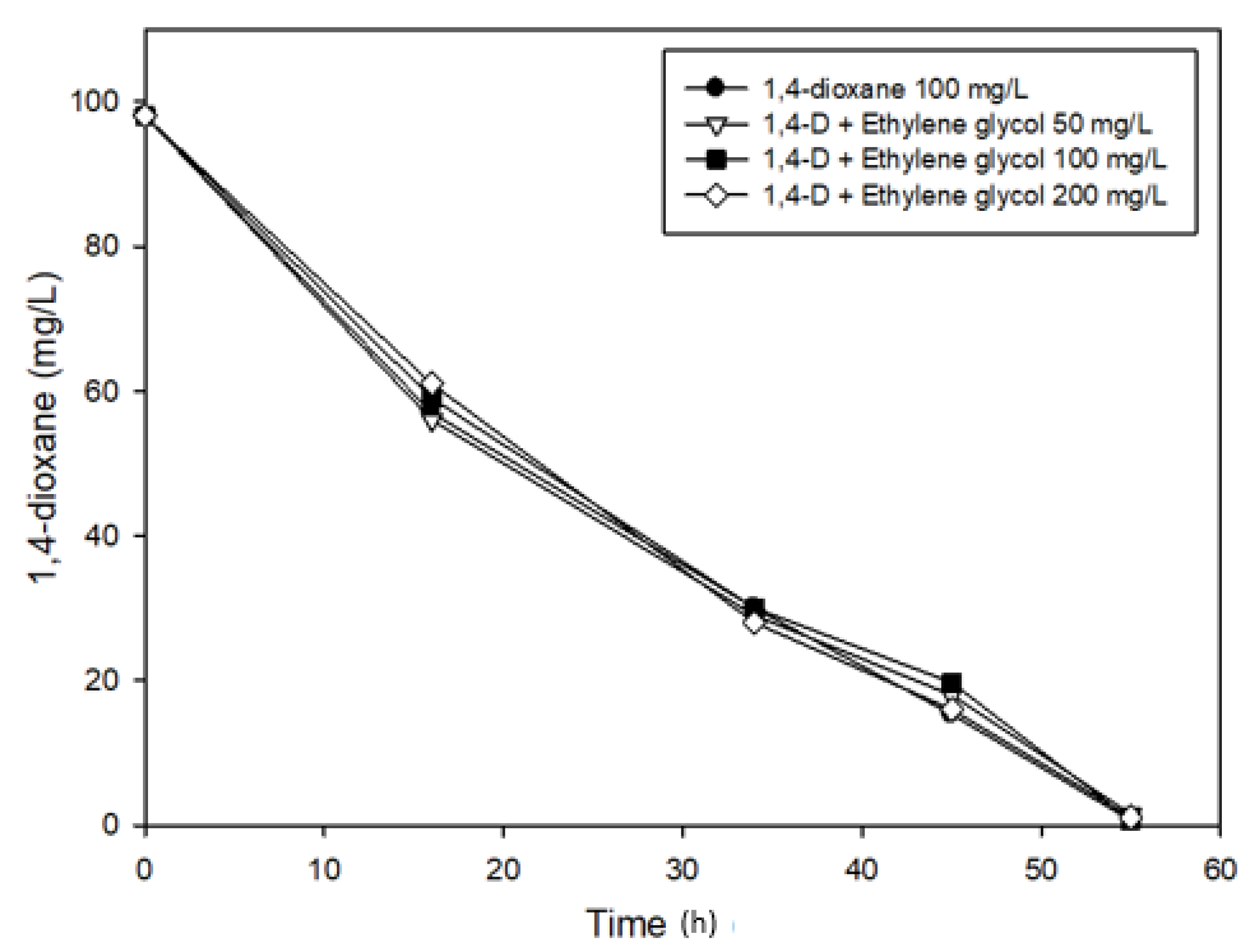
3.3. Effects of Ethylene glycol on 1,4-Dioxane Biodegradation
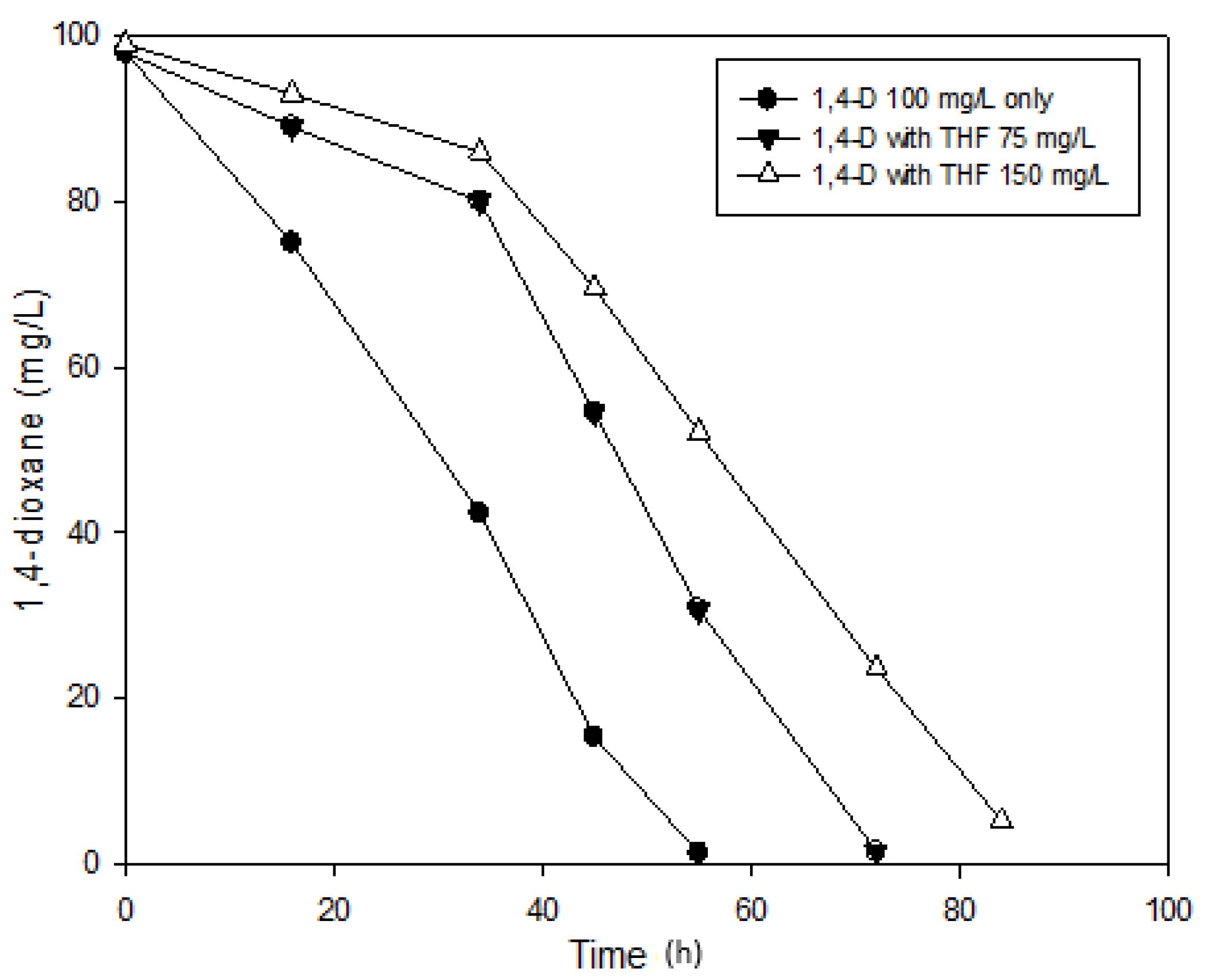
3.4. Effects of THF on 1,4-Dioxane Biodegradation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bernhardt, D.; Diekmann, H. Degradation of dioxane, tetrahydrofuran and other cyclic ethers by an environmental Rhodococcus strain. Appl. Microbiol. Biotechnol. 1991, 36, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Popoola, A.V. Mechanism of the reaction involving the formation of dioxane byproduct during the production of poly (ethylene terephthalate). J. Appl. Polym. Sci. 1991, 43, 1875–1877. [Google Scholar] [CrossRef]
- DiGuiseppi, W.; Walecka-Hutchison, C.; Hatton, J. 1,4-Dioxane Treatment Technologies. Remediat. J. 2016, 27, 71–92. [Google Scholar] [CrossRef]
- Sock, S.M. A Comprehensive Evaluation of Biodegradation as a Treatment Alternative for the Removal of 1,4-Dioxane. Master’s Thesis, Clemson University, Clemson, SC, USA, 1993. [Google Scholar]
- Abe, A. Distribution of 1,4-dioxane in relation to possible sources in the water environment. Sci. Total Environ. 1999, 227, 41–47. [Google Scholar] [CrossRef]
- Isaka, K.; Udagawa, M.; Sei, K.; Ike, M. Pilot test of biological removal of 1,4-dioxane from a chemical factory wastewater by gel carrier entrapping Afipia sp. strain D1. J. Hazard. Mater. 2016, 304, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Johnson, N.W.; Gedalanga, P.B.; Adamson, D.; Newell, C.; Mahendra, S. Response and recovery of microbial communities subjected to oxidative and biological treatments of 1,4-dioxane and co-contaminants. Water Res. 2019, 149, 74–85. [Google Scholar] [CrossRef] [PubMed]
- U.E.P. Agency. Treatment Technologies for 1,4-Dioxane Fundamentals and Field Application; Environmental Protection Agency Publication, EPA Cincinnati: Cincinnati, OH, USA, 2006. [Google Scholar]
- Alexander, M. Nonbiodegradable and other racalcitrant molecules. Biotechnol. Bioeng. 1973, 15, 611–647. [Google Scholar] [CrossRef]
- Inoue, D.; Tsunoda, T.; Sawada, K.; Yamamoto, N.; Sei, K.; Ike, M. Stimulatory and inhibitory effects of metals on 1,4-dioxane degradation by four different 1,4-dioxane-degrading bacteria. Chemosphere 2019, 238, 124606. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, Q.; Wu, L.; Cao, J.; Feng, Q.; Fang, F.; Chen, Y. Inhibition of 1,4-dioxane on the denitrification process by altering the viability and metabolic activity of Paracoccus denitrificans. Environ. Sci. Pollut. Res. 2018, 25, 27274–27282. [Google Scholar] [CrossRef]
- Zhao, L.; Lu, X.; Polasko, A.; Johnson, N.W.; Miao, Y.; Yang, Z.; Mahendra, S.; Gu, B. Co-contaminant effects on 1,4-dioxane biodegradation in packed soil column flow-through systems. Environ. Pollut. 2018, 243, 573–581. [Google Scholar] [CrossRef]
- Isaka, K.; Udagawa, M.; Kimura, Y.; Sei, K.; Ike, M. Biological 1,4-Dioxane Wastewater Treatment by Immobilized Pseudonocardia sp. D17 on Lower 1,4-Dioxane Concentration. J. Water Environ. Technol. 2016, 14, 289–301. [Google Scholar] [CrossRef]
- Skinner, K.; Cuiffetti, L.; Hyman, M.R. Metabolism and Cometabolism of Cyclic Ethers by a Filamentous Fungus, a Graphium sp. Appl. Environ. Microbiol. 2009, 75, 5514–5522. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Ko, K.; A Ramsay, J. Biodegradation of 1,4-dioxane by a Flavobacterium. Biodegradation 2011, 22, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Parales, R.E.; Adamus, J.E.; White, N.; May, H.D. Degradation of 1,4-dioxane by an actinomycete in pure culture. Appl. Environ. Microbiol. 1994, 60, 4527–4530. [Google Scholar] [CrossRef]
- Kämpfer, P.; Kroppenstedt, R.M. Pseudonocardia benzenivorans sp. nov. Int. J. Syst. Evol. Microbiol. 2004, 54, 749–751. [Google Scholar] [CrossRef]
- Mahendra, S.; Petzold, C.J.; Baidoo, E.E.; Keasling, J.D.; Alvarez-Cohen, L. Identification of the Intermediates of in Vivo Oxidation of 1,4-Dioxane by Monooxygenase-Containing Bacteria. Environ. Sci. Technol. 2007, 41, 7330–7336. [Google Scholar] [CrossRef]
- Mahendra, S.; Cohen, L.A. Pseudonocardia dioxanivorans sp. nov., a novel actinomycete that grows on 1,4-dioxane. Int. J. Syst. Evol. Microbiol. 2005, 55, 593–598. [Google Scholar] [CrossRef]
- Kim, Y.-M.; Jeon, J.-R.; Murugesan, K.; Kim, E.-J.; Chang, Y.-S. Biodegradation of 1,4-dioxane and transformation of related cyclic compounds by a newly isolated Mycobacterium sp. PH-06. Biodegradation 2008, 20, 511–519. [Google Scholar] [CrossRef]
- Inoue, D.; Tsunoda, T.; Yamamoto, N.; Ike, M.; Sei, K. 1,4-Dioxane degradation characteristics of Rhodococcus aetherivorans JCM 14343. Biodegradation 2018, 29, 301–310. [Google Scholar] [CrossRef]
- Kohlweyer, U.; Thiemer, B.; Schräder, T.; Andreesen, J.R. Tetrahydrofuran degradation by a newly isolated culture of Pseudonocardia sp. strain K1. FEMS Microbiol. Lett. 2000, 186, 301–306. [Google Scholar] [CrossRef]
- Inoue, D.; Tsunoda, T.; Sawada, K.; Yamamoto, N.; Saito, Y.; Sei, K.; Ike, M. 1,4-Dioxane degradation potential of members of the genera Pseudonocardia and Rhodococcus. Biodegradation 2016, 27, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Zenker, M.J.; Borden, R.; Barlaz, M.A. Modeling Cometabolism of Cyclic Ethers. Environ. Eng. Sci. 2002, 19, 215–228. [Google Scholar] [CrossRef]
- Nakamiya, K.; Hashimoto, S.; Ito, H.; Edmonds, J.S.; Morita, M. Degradation of 1,4-dioxane and cyclic ethers by an isolated fungus. Appl. Environ. Microbiol. 2005, 71, 1254. [Google Scholar] [CrossRef] [PubMed]
- Polasko, A.L.; Zulli, A.; Gedalanga, P.B.; Pornwongthong, P.; Mahendra, S. A Mixed Microbial Community for the Biodegradation of Chlorinated Ethenes and 1,4-Dioxane. Environ. Sci. Technol. Lett. 2018, 6, 49–54. [Google Scholar] [CrossRef]
- Han, J.S.; So, M.H.; Kim, C.-G. Optimization of biological wastewater treatment conditions for 1,4-dioxane decomposition in polyester manufacturing processes. Water Sci. Technol. 2009, 59, 995–1002. [Google Scholar] [CrossRef]
- Sei, K.; Kakinoki, T.; Inoue, D.; Soda, S.; Fujita, M.; Ike, M. Evaluation of the biodegradation potential of 1,4-dioxane in river, soil and activated sludge samples. Biodegradation 2010, 21, 585–591. [Google Scholar] [CrossRef]
- Barajas-Rodriguez, F.J.; Murdoch, L.C.; Falta, R.W.; Freedman, D.L. Simulation of in situ biodegradation of 1,4-dioxane under metabolic and cometabolic conditions. J. Contam. Hydrol. 2019, 223, 103464. [Google Scholar] [CrossRef]
- Li, F.; Deng, D.; Li, M. Distinct Catalytic Behaviors between Two 1,4-Dioxane-Degrading Monooxygenases: Kinetics, Inhibition, and Substrate Range Environ. Sci. Technol. 2020, 54, 1898–1908. [Google Scholar] [CrossRef]
- Rittmann, B.E.; McCarthy, P.L. Environmental Biotechnology: Principles and Applications. In McGraw-Hill Series in Water Resources and Environmental Engineering; Tata McGraw Hill Education Private Limited: New York, NY, USA, 2001. [Google Scholar]
- Mahendra, S.; Alvarez-Cohen, L. Kinetics of 1,4-dioxane biodegradation by monooxygenase-expressing bacteria. Environ. Sci. Technol. 2006, 40, 5435–5442. [Google Scholar] [CrossRef]
- Zenker, M.J.; Borden, R.; Barlaz, M.A. Biodegradation of 1,4-Dioxane Using Trickling Filter. J. Environ. Eng. 2004, 130, 926–931. [Google Scholar] [CrossRef]



| Content | Values (mg) | Percentage (%) |
|---|---|---|
| Cells associated as 1,4-D | 36.4 ± 2.4 | 36.9 ± 6.6 |
| CO2 recovered as 1,4-dioxane | 57.5 ± 5.4 | 58.3 ± 9.4 |
| Volatile | 3.9 ± 0.1 | 4.0 ± 2.5 |
| 1,4-dioxane residual in liquid | 0.8 ± 0.1 | 0.8 |
| 1,4-dioxane | 98.6 | 100 |
| 1,4-Dioxane Concentration (mg/L) | Degradation Rate (mg/L/h) |
|---|---|
| 5 | 1.793 |
| 50 | 2.158 |
| 200 | 3.056 |
| 500 | 4.375 |
| 1000 | 6.167 |
| 1500 | 7.83 |
| 2000 | 9.125 |
| Kinetic Parameters | Value |
|---|---|
| Ks (mg 1,4-D/L) | 9.42 |
| qmax (mg 1,4-D/mg VSS/h) | 0.0063 |
| kd (mg VSS/mg VSS/day) | 0.023 |
| YT (mg VSS/mg1,4-D) | 0.43 |
| μmax (day)−1 | 0.061 |
| Kinetic Coefficients | 1,4-Dioxane as Sole Source | THF as Sole Source | 1,4-D&THF 75 mg/L | 1,4-D&THF 150 mg/L |
|---|---|---|---|---|
| qmax (mg COD/mgVSS/h) | 0.0088 | 0.0167 | 0.0171 | 0.0149 |
| Keff (mg/L) | 9.08 | 6.4 | 131.72 | 345.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, K.H.; Wie, Y.M.; Lee, Y.-S. Characterization of 1,4-Dioxane Biodegradation by a Microbial Community. Water 2020, 12, 3372. https://doi.org/10.3390/w12123372
Lee KH, Wie YM, Lee Y-S. Characterization of 1,4-Dioxane Biodegradation by a Microbial Community. Water. 2020; 12(12):3372. https://doi.org/10.3390/w12123372
Chicago/Turabian StyleLee, Kang Hoon, Young Min Wie, and Yong-Soo Lee. 2020. "Characterization of 1,4-Dioxane Biodegradation by a Microbial Community" Water 12, no. 12: 3372. https://doi.org/10.3390/w12123372
APA StyleLee, K. H., Wie, Y. M., & Lee, Y.-S. (2020). Characterization of 1,4-Dioxane Biodegradation by a Microbial Community. Water, 12(12), 3372. https://doi.org/10.3390/w12123372





