How Elevated CO2 Shifts Root Water Uptake Pattern of Crop? Lessons from Climate Chamber Experiments and Isotopic Tracing Technique
Abstract
1. Introduction
2. Materials and Methods
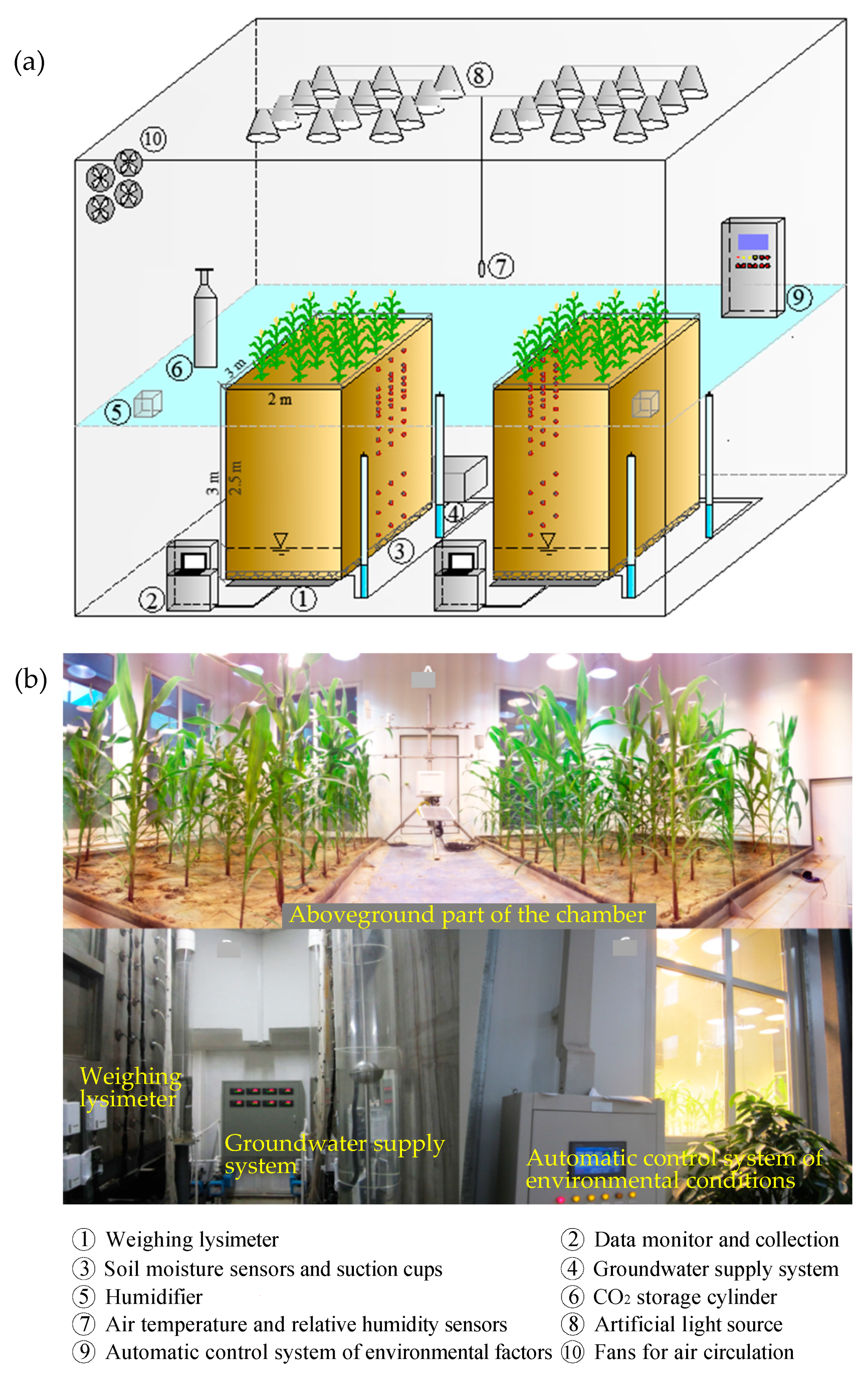
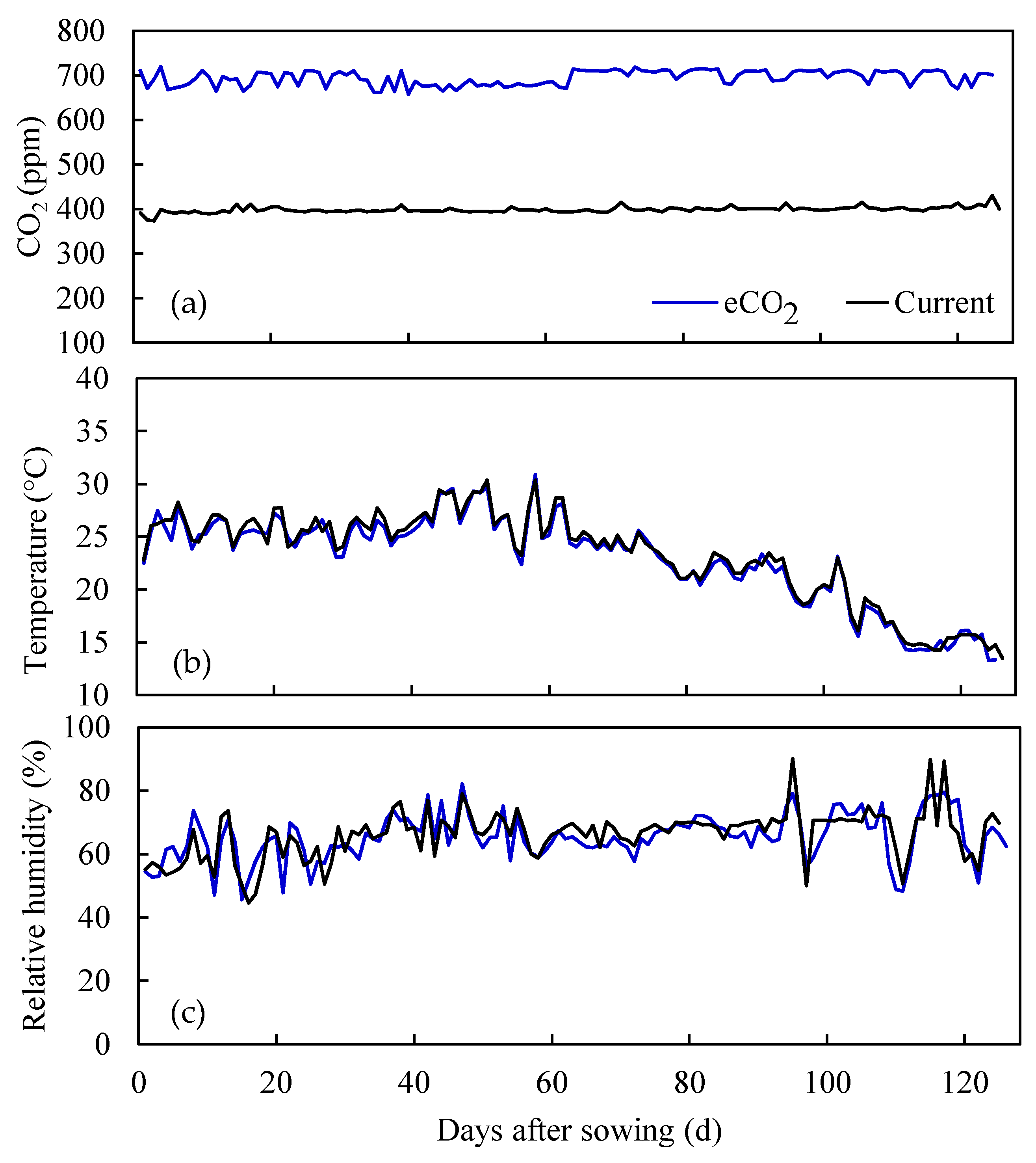
2.1. Description of the Experiment
2.2. Water Sampling and Isotope Analysis
2.3. Measurements of Plant Ecophysiological Indices
2.4. Quantification of Water Source Contributions to Maize
2.5. Statistical Analysis
3. Results
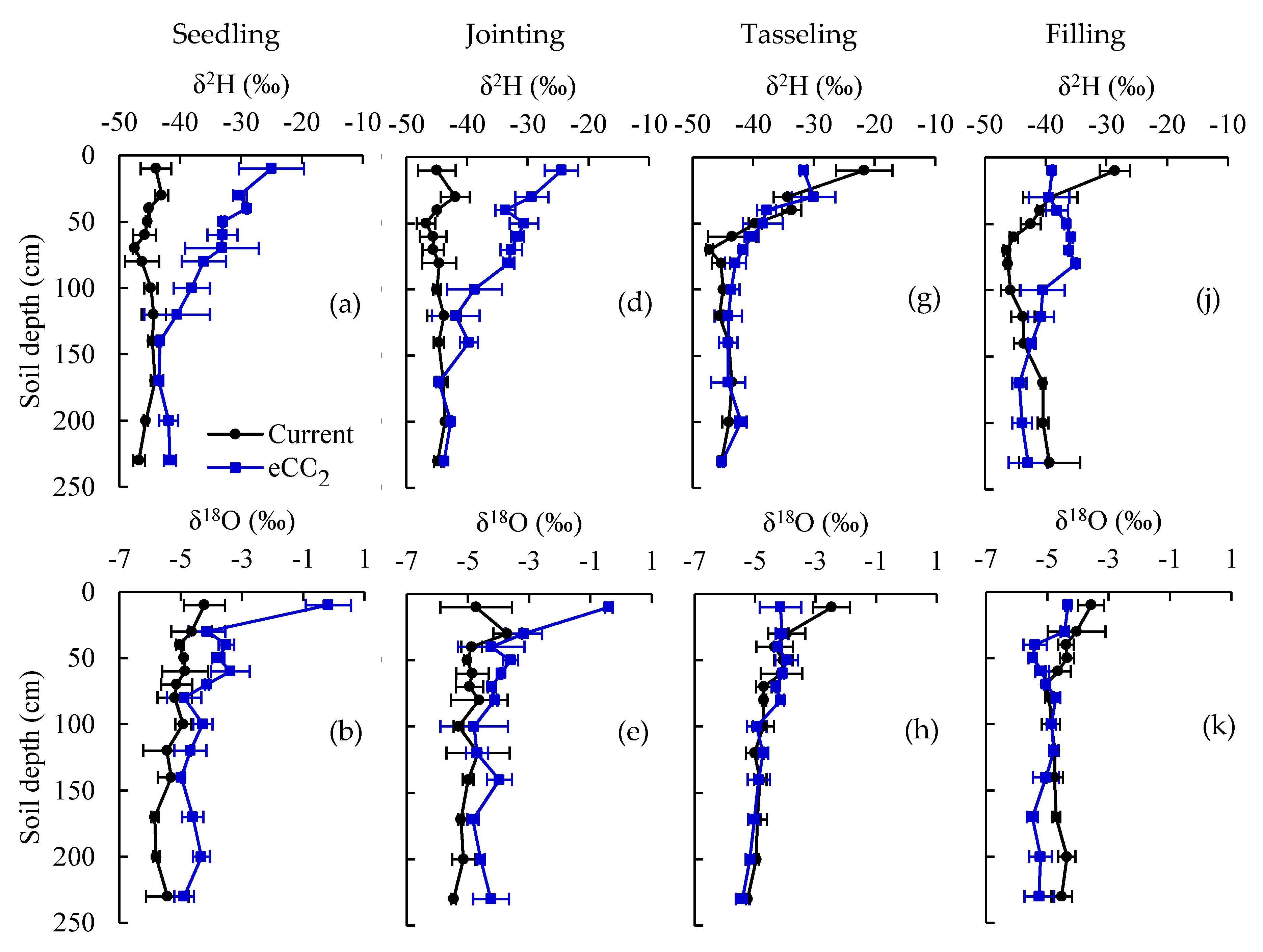
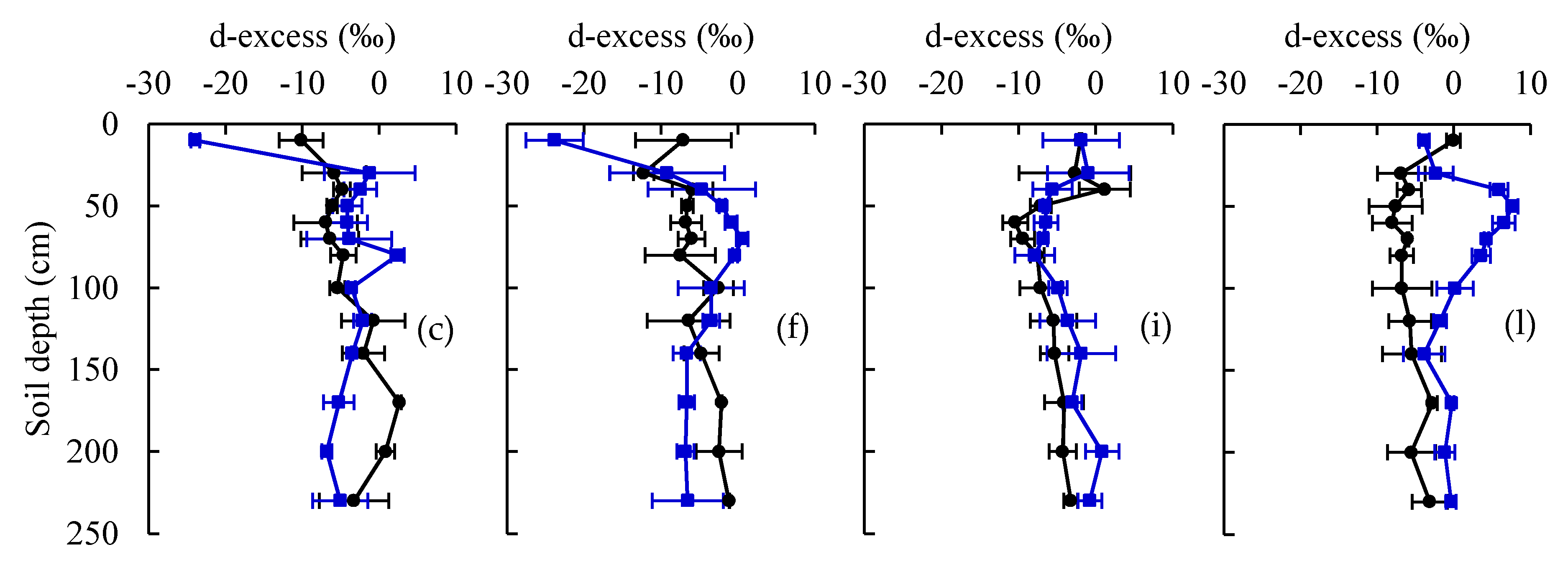
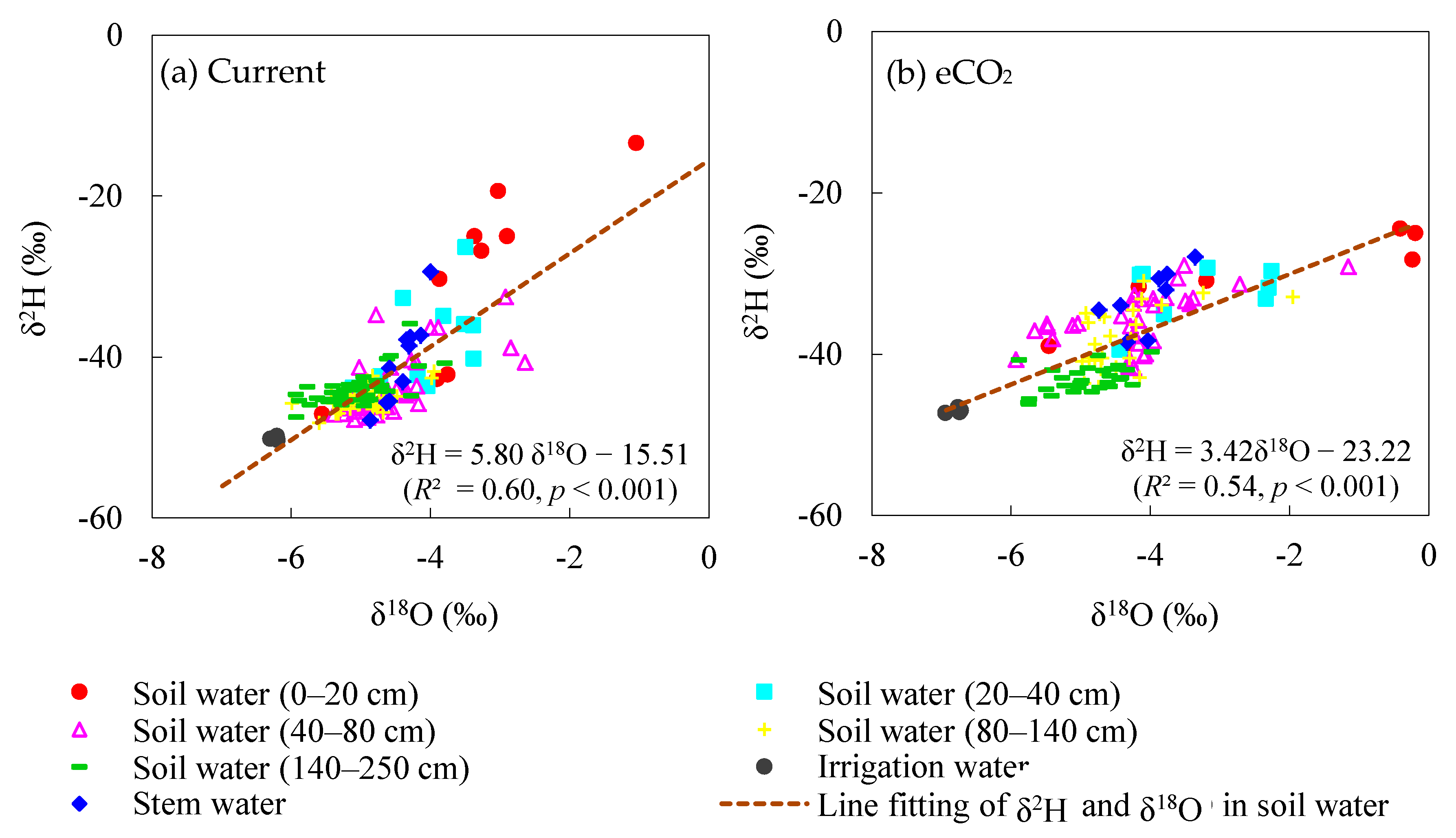
3.1. Water Stable Isotopes under Current CO2 and ECO2 Treatments
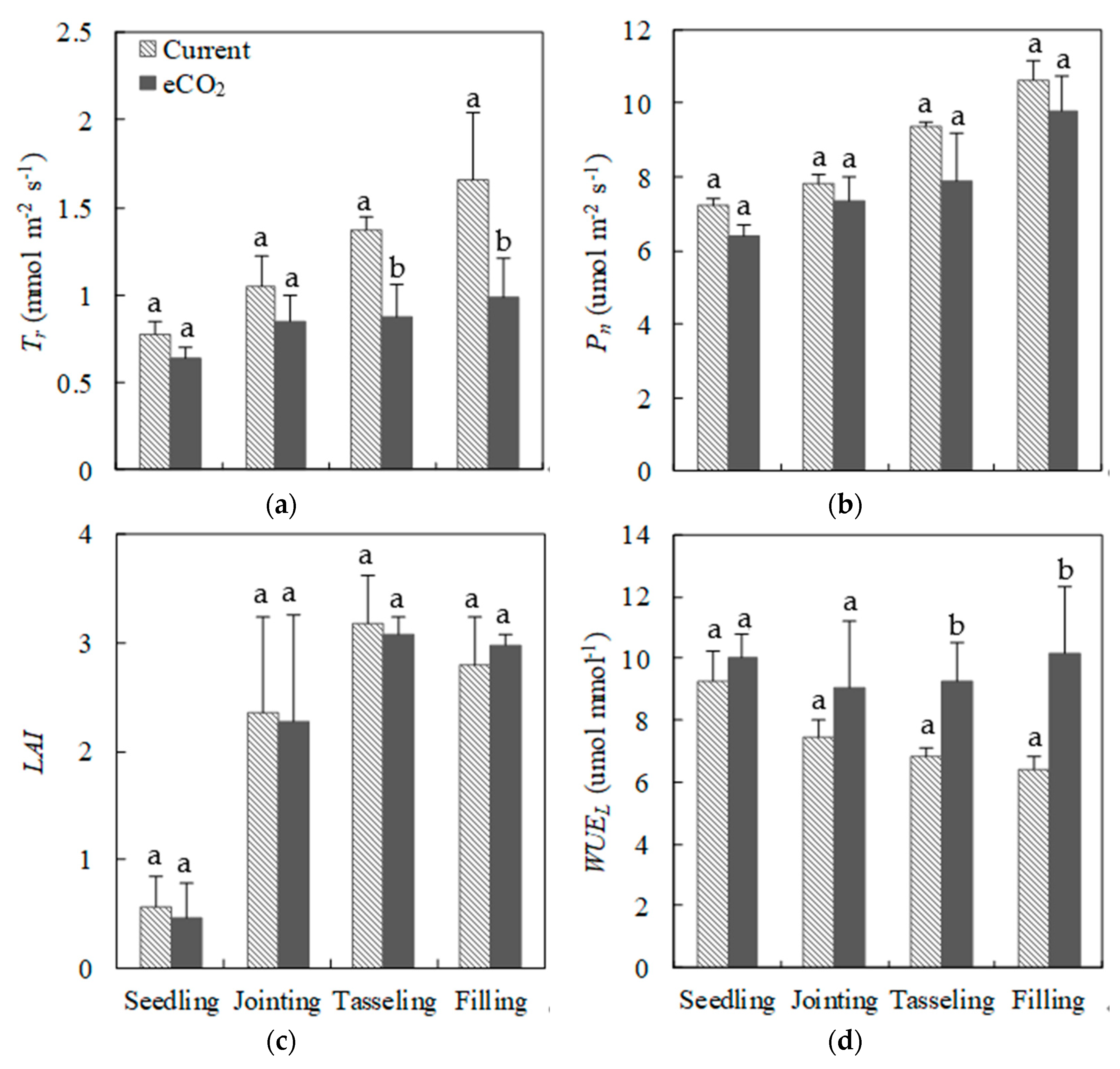
3.2. Plant Ecophysiological Indices under Current CO2 and eCO2 Treatments
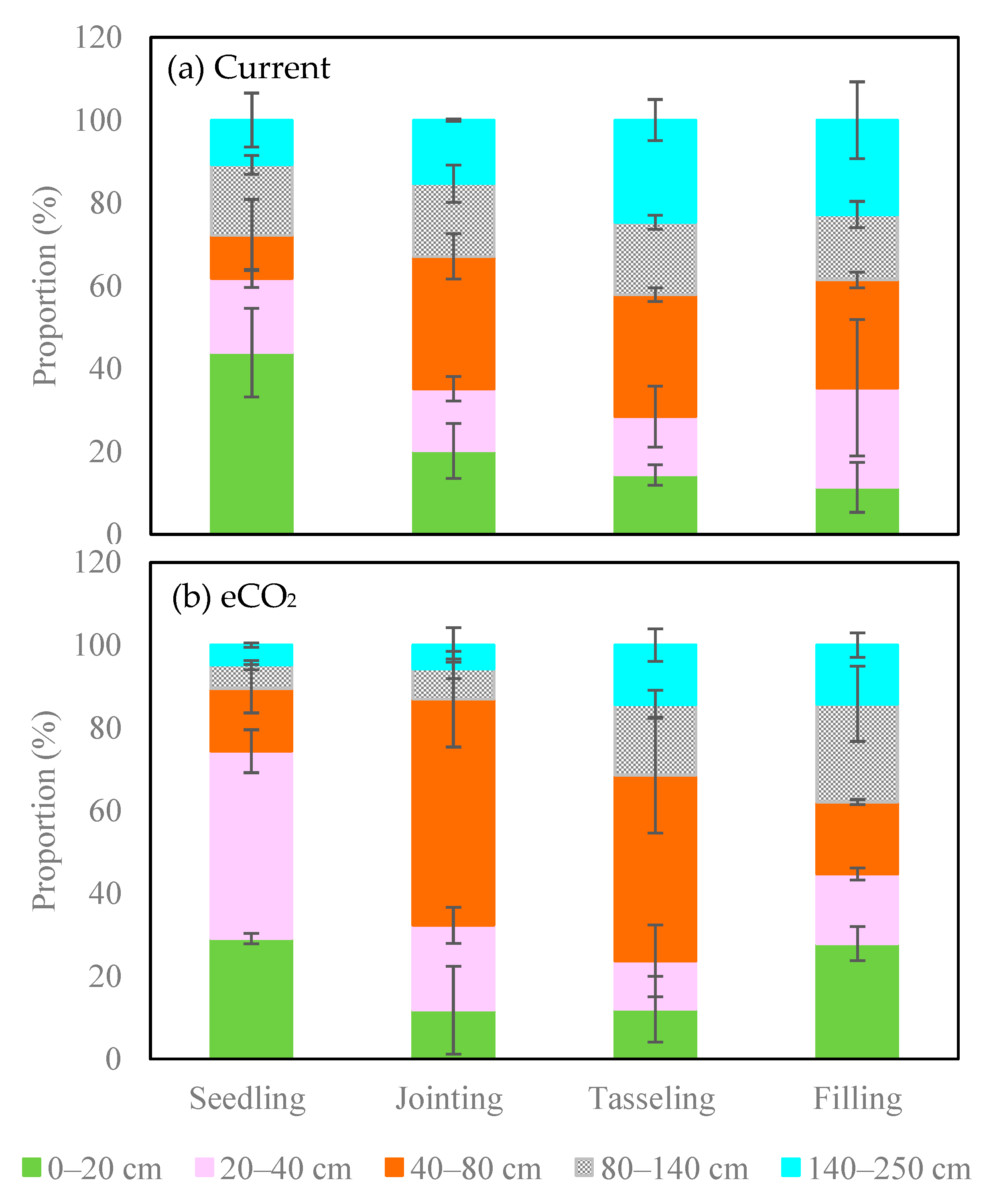
3.3. Root Water Uptake Patterns under Current CO2 and eCO2 Treatments
3.4. Correlations between Root Water Uptake Patterns and Crop Growth under Current and eCO2 Treatments
4. Discussion
4.1. Impacts of eCO2 on Crop Water Uptake
4.2. Implications of this Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grossiord, C.; Sevanto, S.; Dawson, T.E.; Adams, H.D.; Collins, A.D.; Dickman, L.T.; Newman, B.D.; Stockton, E.A.; McDowell, N.G. Warming combined with more extreme precipitation regimes modifies the water sources used by trees. New Phytol. 2017, 213, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Uddin, S.; Löw, M.; Parvin, S.; Fitzgerald, G.; Bahrami, H.; Tausz-Posch, S.; Armstrong, R.; O’Leary, G.; Tausz, M. Water use and growth responses of dryland wheat grown under elevated [CO2] are associated with root length in deeper, but not upper soil layer. Field Crops Res. 2018, 224, 170–181. [Google Scholar] [CrossRef]
- Dubey, S.K.; Sharma, D. Assessment of climate change impact on yield of major crops in the Banas River Basin, India. Sci. Total Environ. 2018, 635, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Asbjornsen, H.; Mora, G.; Helmers, M.J. Variation in water uptake dynamics among contrasting agricultural and native plant communities in the Midwestern US. Agric. Ecosyst. Environ. 2007, 121, 343–356. [Google Scholar] [CrossRef]
- Wang, J.; Fu, B.J.; Lu, N.; Zhang, L. Seasonal variation in water uptake patterns of three plant species based on stable isotopes in the semi-arid Loess Plateau. Sci. Total Environ. 2017, 609, 27–37. [Google Scholar] [CrossRef]
- IPCC, Core Writing Team. Climate Change: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; p. 151. [Google Scholar]
- Tilman, D.; Kenneth, G.C.; Pamela, A.M.; Rosamond, N.; Stephen, P. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef]
- Manderscheid, R.; Erbs, M.; Weigel, H. Interactive effects of free-air CO2 enrichment and drought stress on maize growth. Eur. J. Agron. 2014, 52, 11–21. [Google Scholar] [CrossRef]
- Beyer, M.; Hamutoko, J.T.; Wanke, H.; Gaj, M.; Koeniger, P. Examination of deep root water uptake using anomalies of soil water stable isotopes, depth-controlled isotopic labeling and mixing models. J. Hydrol. 2018, 566, 122–136. [Google Scholar] [CrossRef]
- Amin, A.; Zuecco, G.; Geris, J.; Schwendenmann, L.; McDonnell, J.J.; Borga, M.; Penna, D. Depth distribution of soil water sourced by plants at the global scale: A new direct inference approach. Ecohydrology 2020, 13, e2177. [Google Scholar] [CrossRef]
- Leakey, A.D.B.; Uribelarrea, M.; Ainsworth, E.A.; Naidu, S.L.; Rogers, A.; Ort, D.R.; Long, S.P. Photosynthesis, productivity, and yield of maize are not affected by open-air elevation of CO2 concentration in the absence of drought. Plant Physiol. 2006, 140, 779–790. [Google Scholar] [CrossRef]
- Oliver, R.J.; Finch, J.W.; Taylor, G. Second generation bioenergy crops and climate change: A review of the effects of elevated atmospheric CO2 and drought on water use and the implications for yield. Glob. Chang. Biol. Bioenergy 2009, 1, 97–114. [Google Scholar] [CrossRef]
- Drewry, D.T.; Kumar, P.; Long, S.; Bernacchi, C.; Liang, X.-Z.; Sivapalan, M. Ecohydrological responses of dense canopies to environmental variability: 2. Role of acclimation under elevated CO2. J. Geophys. Res. 2010, 115, G04023. [Google Scholar] [CrossRef]
- Markelz, R.J.C.; Strellner, R.S.; Leakey, A.D.B. Impairment of C4 photosynthesis by drought is exacerbated by limiting nitrogen and ameliorated by elevated [CO2] in maize. J. Exp. Bot. 2011, 62, 3235–3246. [Google Scholar] [CrossRef] [PubMed]
- Le, P.V.V.; Kumar, P.; Drewry, D.T. Implications for the hydrologic cycle under climate change due to the expansion of bioenergy crops in the Midwestern United States. Proc. Natl. Acad. Sci. USA 2011, 108, 15085–15090. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.H., Jr.; Kakani, V.G.; Vu, J.C.V.; Boote, K.J. Elevated CO2 increases water use efficiency by sustaining photosynthesis of water-limited maize and sorghum. J. Plant Physiol. 2011, 168, 1909–1918. [Google Scholar] [CrossRef]
- Kim, S.-H.; Sicher, R.C.; Bae, H.; Gitz, D.C.; Baker, J.T.; Timlin, D.J.; Reddy, V.R. Canopy photosynthesis, evapotranspiration, leaf nitrogen, and transcription profiles of maize in response to CO2 enrichment. Glob. Chang. Biol. 2006, 12, 588–600. [Google Scholar] [CrossRef]
- Kang, S.Z.; Zhang, F.C.; Hu, X.T.; Zhang, J.H. Benefits of CO2 enrichment on crop plants are modified by soil water status. Plant Soil 2002, 238, 69–77. [Google Scholar] [CrossRef]
- Leakey, A.D.B.; Bernacchi, C.J.; Dohleman, W.F.G.; Ort, D.R.; Long, S.P. Will photosynthesis of maize (Zea mays) in the US Corn Belt increase in future [CO2] rich atmospheres? An analysis of diurnal courses of CO2 uptake under free-air concentration enrichment (FACE). Glob. Chang. Biol. 2004, 10, 951–962. [Google Scholar] [CrossRef]
- Driscoll, S.P.; Prins, A.; Olmos, E.; Kunert, K.J.; Foyer, C.H. Specification of adaxial and abaxial stomata, epidermal structure and photosynthesis to CO2 enrichment in maize leaves. J. Exp. Bot. 2006, 57, 381–390. [Google Scholar] [CrossRef]
- van der Kooi, C.J.; Reich, M.; Löw, M.; De Kok, L.J.; Tausza, M. Growth and yield stimulation under elevated CO2 and drought: A meta-analysis on crops. Environ. Exp. Bot. 2016, 122, 150–157. [Google Scholar] [CrossRef]
- Li, X.J.; Kang, S.Z.; Zhang, X.T.; Li, F.S.; Lu, H.N. Deficit irrigation provokes more pronounced responses of maize photosynthesis and water productivity to elevated CO2. Agric. Water Manag. 2018, 195, 71–83. [Google Scholar] [CrossRef]
- Wang, P.; Song, X.F.; Han, D.M.; Zhang, Y.H.; Liu, X. A study of root water uptake of crops indicated by hydrogen and oxygen stable isotopes: A case in Shanxi Province, China. Agric. Water Manag. 2010, 97, 475–482. [Google Scholar] [CrossRef]
- Ma, Y.; Song, X.F. Using stable isotopes to determine seasonal variations in water uptake of summer maize under different fertilization treatments. Sci. Total Environ. 2016, 550, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.J.; Du, T.S.; Li, F.S.; Li, S.E.; Ding, R.S.; Tong, L. Quantification of maize water uptake from different layers and root zones under alternate furrow irrigation using stable oxygen isotope. Agric. Water Manag. 2016, 168, 35–44. [Google Scholar] [CrossRef]
- Zhao, X.; Li, F.D.; Ai, Z.P.; Li, J.; Gu, C.K. Stable isotope evidences for identifying crop water uptake in a typical winter wheat–summer maize rotation field in the North China Plain. Sci. Total Environ. 2018, 618, 121–131. [Google Scholar] [CrossRef]
- Sprenger, M.; Leistert, H.; Gimbel, K.; Weiler, M. Illuminating hydrological processes at the soil-vegetation-atmosphere interface with water stable isotopes. Rev. Geophys. 2016, 54. [Google Scholar] [CrossRef]
- Wang, J.; Lu, N.; Fu, B.J. Inter-comparison of stable isotope mixing models for determining plant water source partitioning. Sci. Total Environ. 2019, 666, 685–693. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, Z.S. Stable isotopic analysis on water utilization of two xerophytic shrubs in a revegetated desert area: Tengger Desert, China. Water 2015, 7, 1030–1045. [Google Scholar] [CrossRef]
- Wu, J.B.; Li, H.P.; Niu, J.M.; Liu, T.J.; Zheng, H.X.; Xu, X.T.; Miao, S. Water uptake patterns of alfalfa under winter irrigation in cold and arid grassland. Water 2020, 12, 1093. [Google Scholar] [CrossRef]
- Penna, D.; Geris, J.; Hopp, L.; Scandellari, F. Water sources for root water uptake: Using stable isotopes of hydrogen and oxygen as a research tool in agricultural and agroforestry systems. Agric. Ecosyst. Environ. 2020, 291, 106790. [Google Scholar] [CrossRef]
- Dansgaard, W. Stable isotopes in precipitation. Tellus 1964, 16, 436–468. [Google Scholar] [CrossRef]
- Wang, L.; Good, S.P.; Caylor, K.K. Global synthesis of vegetation control on evapotranspiration partitioning. Geophys. Res. Lett. 2014, 41, 6753–6757. [Google Scholar] [CrossRef]
- Ritchie, S.W.; Hanway, J.J. How a Corn Plant Develops; Special Report No. 48; Iowa State University Press: Ames, IA, USA, 1982. [Google Scholar]
- Stock, B.C.; Semmens, B.X. MixSIAR GUI User Manual, Version 1.0. 2013. Available online: http://conserver.iugo-cafe.org/user/brice.semmens/MixSIAR (accessed on 7 February 2020).
- Rothfuss, Y.; Javaux, M. Reviews and syntheses: Isotopic approaches to quantify root water uptake: A review and comparison of methods. Biogeosciences 2017, 14, 2199–2224. [Google Scholar] [CrossRef]
- Mahindawansha, A.; Orlowski, N.; Kraft, P.; Rothfuss, Y.; Racela, H.; Breuer, L. Quantification of plant water uptake by water stable isotopes in rice paddy systems. Plant Soil 2018, 429, 281–302. [Google Scholar] [CrossRef]
- Newton, P.C.D.; Clark, H.; Bell, C.C.; Glasgow, E.M. Interaction of soil moisture and elevated CO2 on the above-ground growth rate root length density and gas exchange of curves from temperate pasture. J. Exp. Bot. 1996, 47, 771–779. [Google Scholar] [CrossRef]
- de Graaff, M.A.; van Groenigen, K.J.; Six, J.; Hungate, B.; van Kessel, C. Interactions between plant growth and soil nutrient cycling under elevated CO2: A meta-analysis. Glob. Chang. Biol. 2006, 12, 2077–2091. [Google Scholar] [CrossRef]
- Norby, R.J.; Wullschleger, S.D.; Gunderson, C.A.; Johnson, D.W.; Ceulemans, R. Tree responses to rising CO2 in field experiments: Implications for the future forest. Plant Cell Environ. 1999, 22, 683–714. [Google Scholar] [CrossRef]
- Matamala, R.; Gonzàlez-Meler, M.A.; Jastrow, J.D.; Norby, R.J.; Schlesinger, W.H. Impacts of fine root turnover on forest NPP and soil C sequestration potential. Science 2003, 302, 1385–1387. [Google Scholar] [CrossRef]
- Wilson, K.B.; Carlson, T.N.; Bunce, J.A. Feedback significantly influences the simulated effect of CO2 on seasonal evapotranspiration from two agricultural species. Glob. Chang. Biol. 1999, 5, 903–917. [Google Scholar] [CrossRef]
- Liu, C.M.; Zhang, X.Y.; Zhang, Y.Q. Determination of daily evaporation and evapotranspiration of winter wheat and maize by large-scale weighing lysimeter and micro-lysimeter. Agric. For. Meteorol. 2002, 111, 109–120. [Google Scholar] [CrossRef]
- Hammer, G.L.; Dong, Z.S.; McLean, G.; Doherty, A.; Messina, C.; Schusler, J.; Zinselmeier, C.; Paszkiewicz, S.; Cooper, M. Can changes in canopy and/or root system architecture explain historical maize yield trends in the US Corn Belt? Crop Sci. 2009, 49, 299–312. [Google Scholar] [CrossRef]
- Wall, G.W.; Brooks, T.J.; Adam, R.; Cousins, A.B.; Kimball, B.A.; Pinter, P.J.; LaMorte, R.L.; Triggs, J.; Ottman, M.J.; Leavitt, S.W.; et al. Elevated atmospheric CO2 improved sorghum plant water status by ameliorating the adverse effects of drought. New Phytol. 2001, 152, 231–248. [Google Scholar] [CrossRef]
| Stage | Days after Sowing | Amount of Irrigation (mm) | Fertilizer | ||
|---|---|---|---|---|---|
| Current | eCO2 | Type | Amount (kg ha−1) | ||
| Seedling | 0–32 | 0–33 | 20 | Urea | 300 |
| Jointing | 33–64 | 34–66 | 60 | None | None |
| Tasseling | 65–79 | 67–82 | 60 | Urea | 300 |
| Filling | 80–103 | 83–105 | None | None | None |
| Maturity | 104–125 | 106–126 | 60 | None | None |
| Proportions in Each Layer | Tr | Pn | LAI | WUEL | ||||
|---|---|---|---|---|---|---|---|---|
| Current | eCO2 | Current | eCO2 | Current | eCO2 | Current | eCO2 | |
| 0–20 cm | −0.84 ** | 0.00 | −0.92 ** | 0.00 | −0.91 ** | −0.14 | 0.64 * | 0.00 |
| 20–40 cm | 0.00 | −0.54 * | 0.01 | −0.58 * | −0.01 | −0.88 ** | 0.00 | −0.05 |
| 40–80 cm | 0.53 * | 0.00 | 0.61 * | 0.00 | 0.83 * | 0.09 | −0.38 | 0.01 |
| 80–140 cm | −0.06 | 0.67 * | −0.01 | 0.67 * | 0.00 | 0.58 * | 0.13 | 0.01 |
| 140–250 cm | 0.69 * | 0.82 ** | 0.60 * | 0.72 ** | 0.60 * | 0.60 * | −0.73 ** | −0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Y.; Wu, Y.; Song, X. How Elevated CO2 Shifts Root Water Uptake Pattern of Crop? Lessons from Climate Chamber Experiments and Isotopic Tracing Technique. Water 2020, 12, 3194. https://doi.org/10.3390/w12113194
Ma Y, Wu Y, Song X. How Elevated CO2 Shifts Root Water Uptake Pattern of Crop? Lessons from Climate Chamber Experiments and Isotopic Tracing Technique. Water. 2020; 12(11):3194. https://doi.org/10.3390/w12113194
Chicago/Turabian StyleMa, Ying, Yali Wu, and Xianfang Song. 2020. "How Elevated CO2 Shifts Root Water Uptake Pattern of Crop? Lessons from Climate Chamber Experiments and Isotopic Tracing Technique" Water 12, no. 11: 3194. https://doi.org/10.3390/w12113194
APA StyleMa, Y., Wu, Y., & Song, X. (2020). How Elevated CO2 Shifts Root Water Uptake Pattern of Crop? Lessons from Climate Chamber Experiments and Isotopic Tracing Technique. Water, 12(11), 3194. https://doi.org/10.3390/w12113194






