Using Freshwater Bivalves (Corbicula Fluminea) to Alleviate Harmful Effects of Small-Sized Crucian Carp (Carassius Carassius) on Growth of Submerged Macrophytes during Lake Restoration by Biomanipulation
Abstract
1. Introduction
2. Materials and Methods
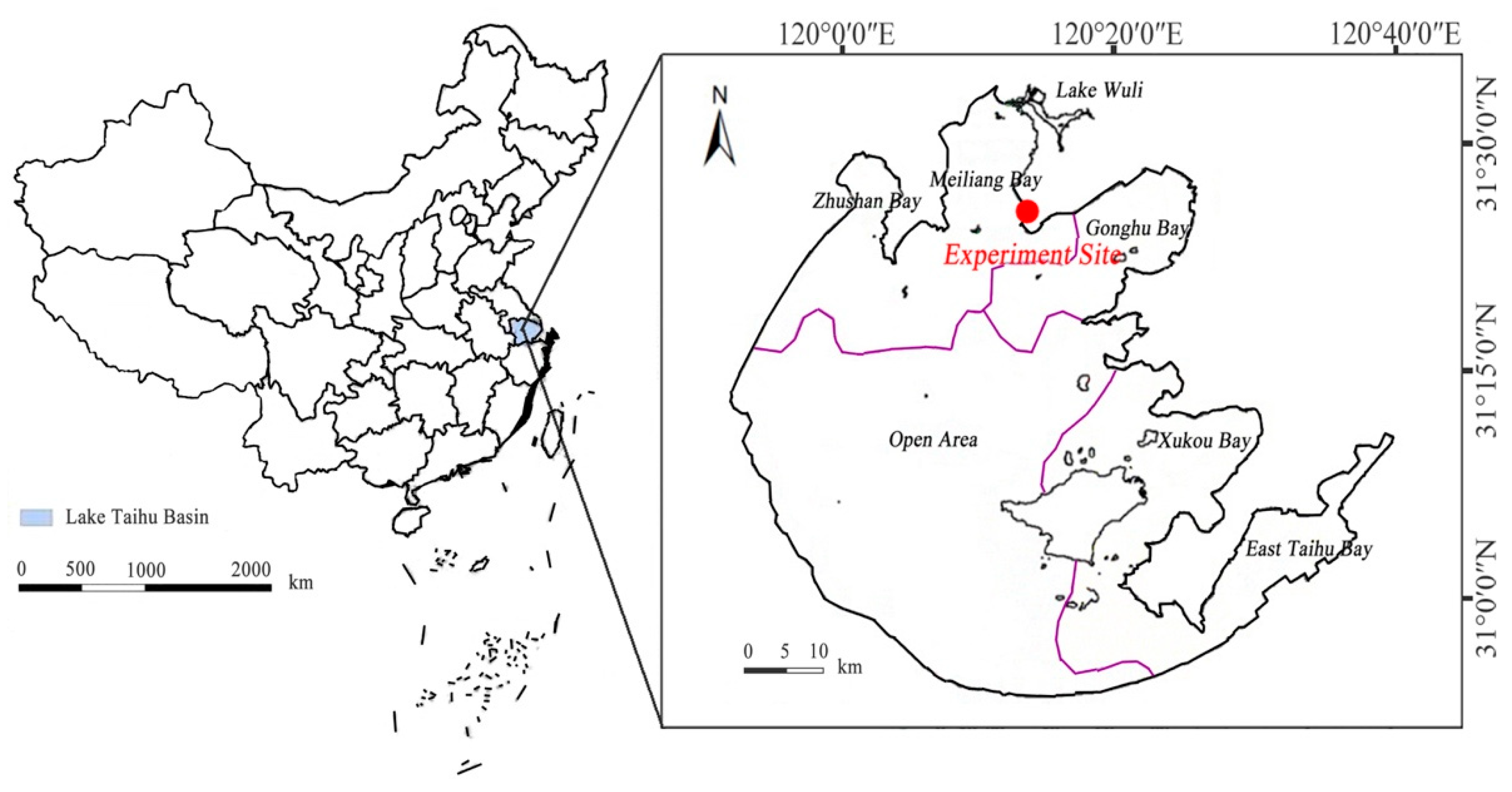
2.1. Experimental Materials
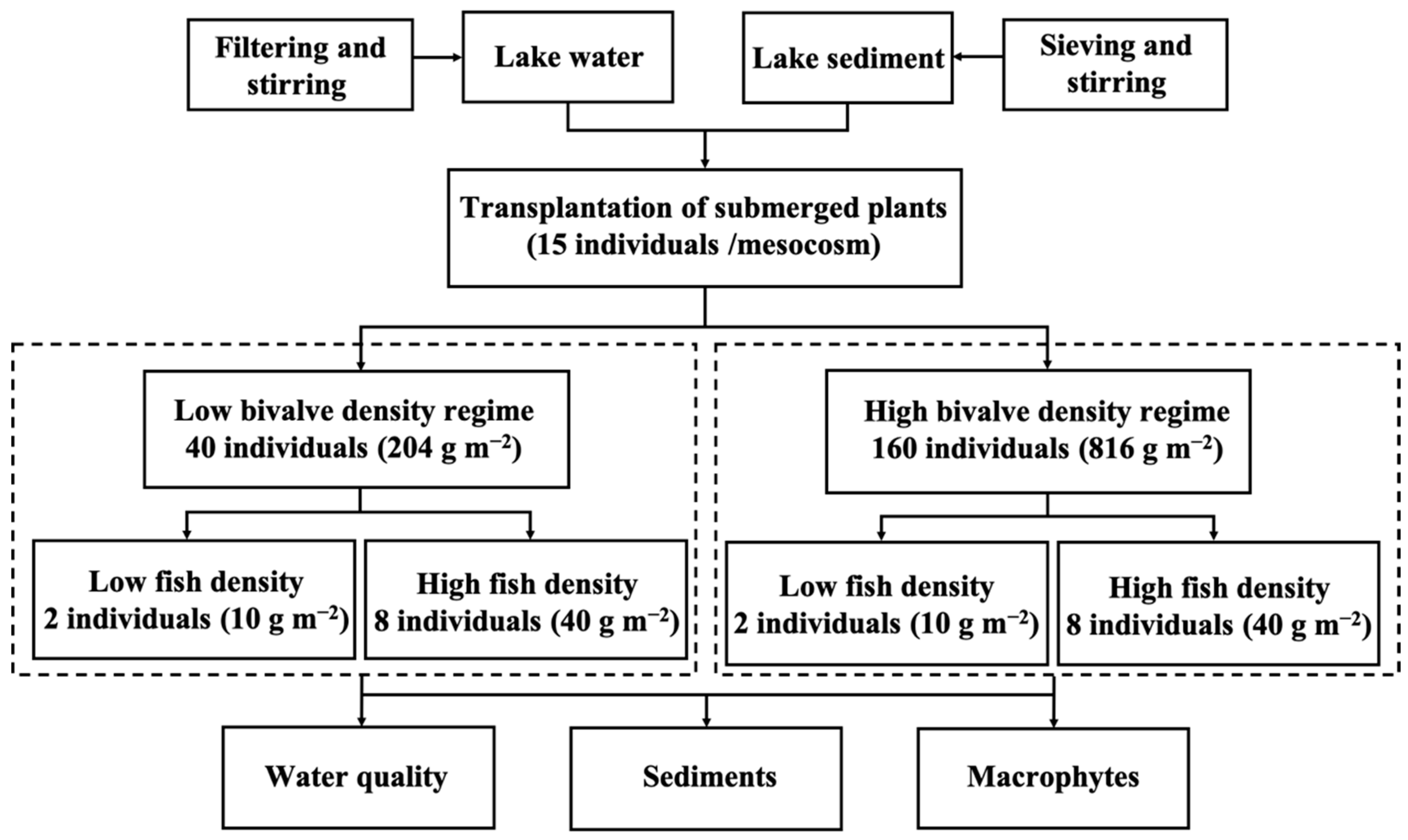
2.2. Experimental Design
2.3. Samplings
2.4. Data Analyses
3. Results
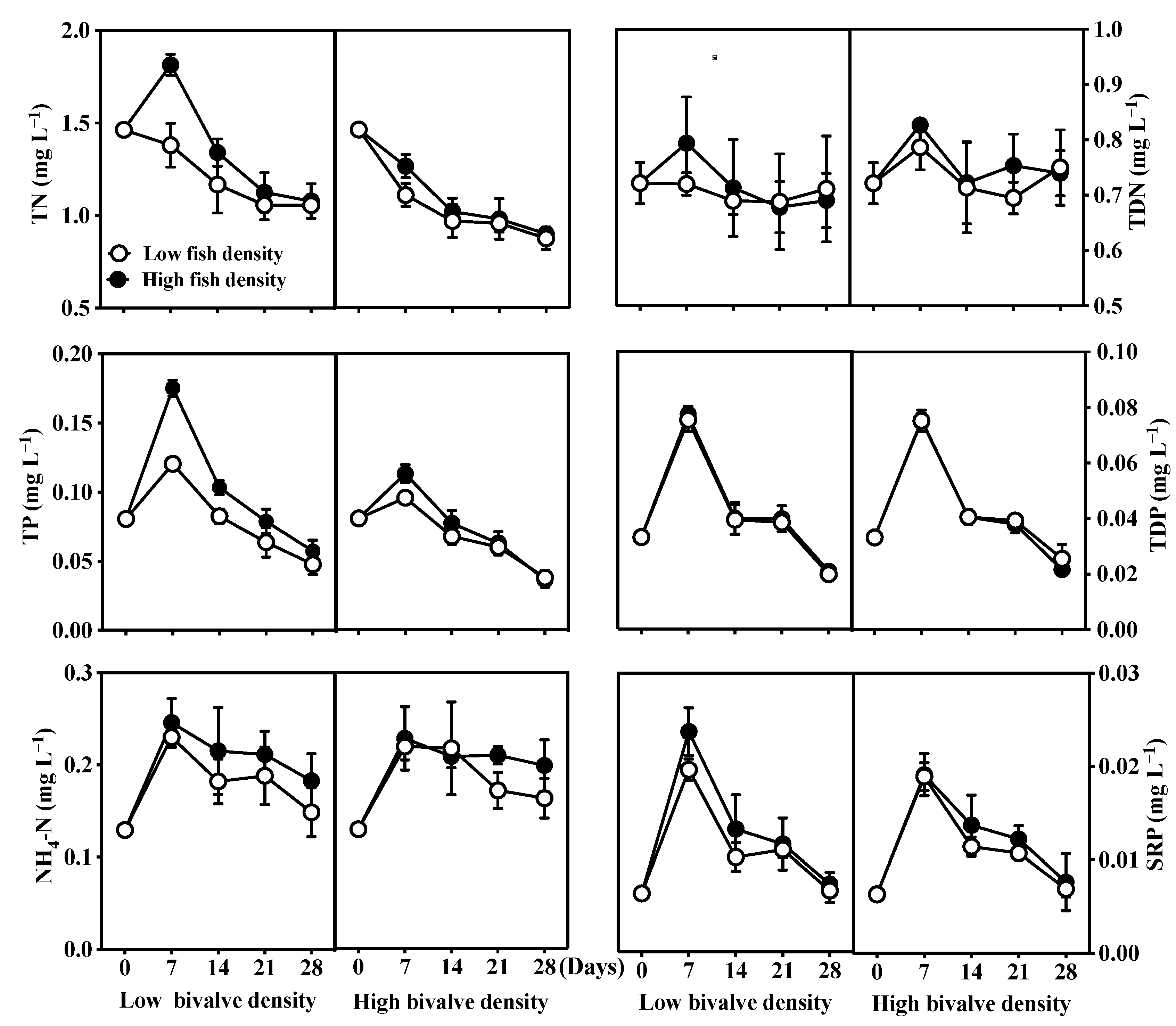
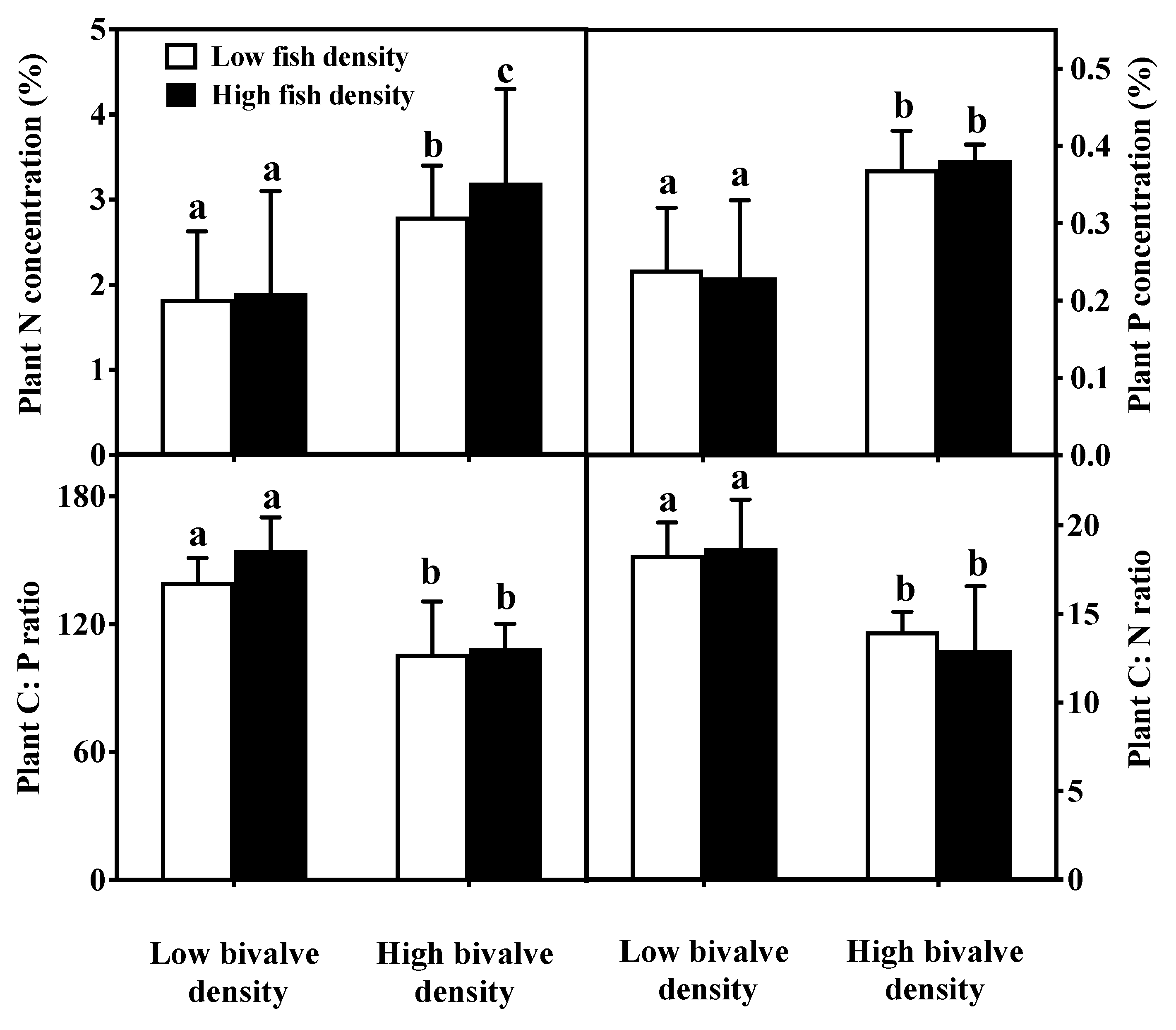
3.1. Nutrients
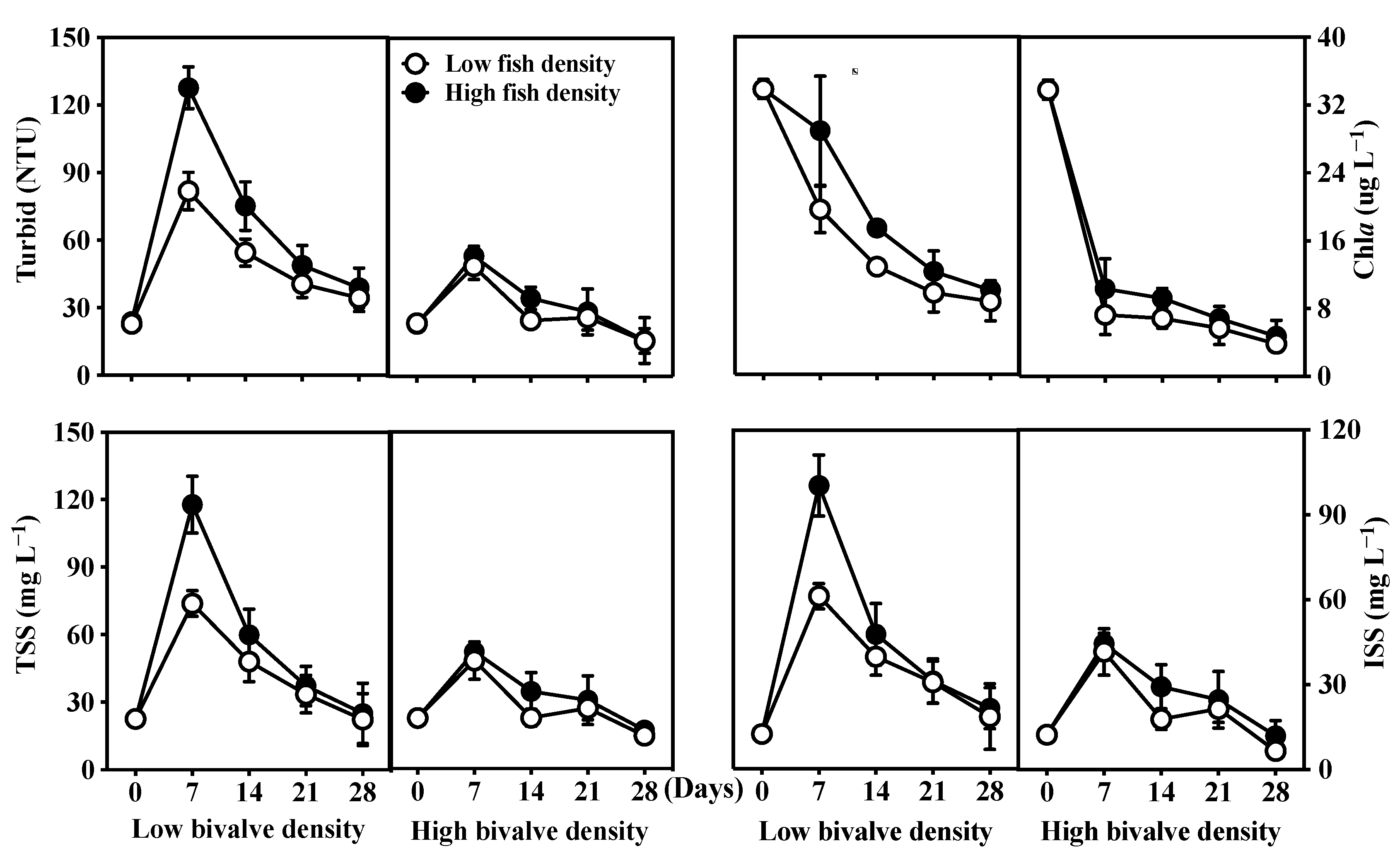
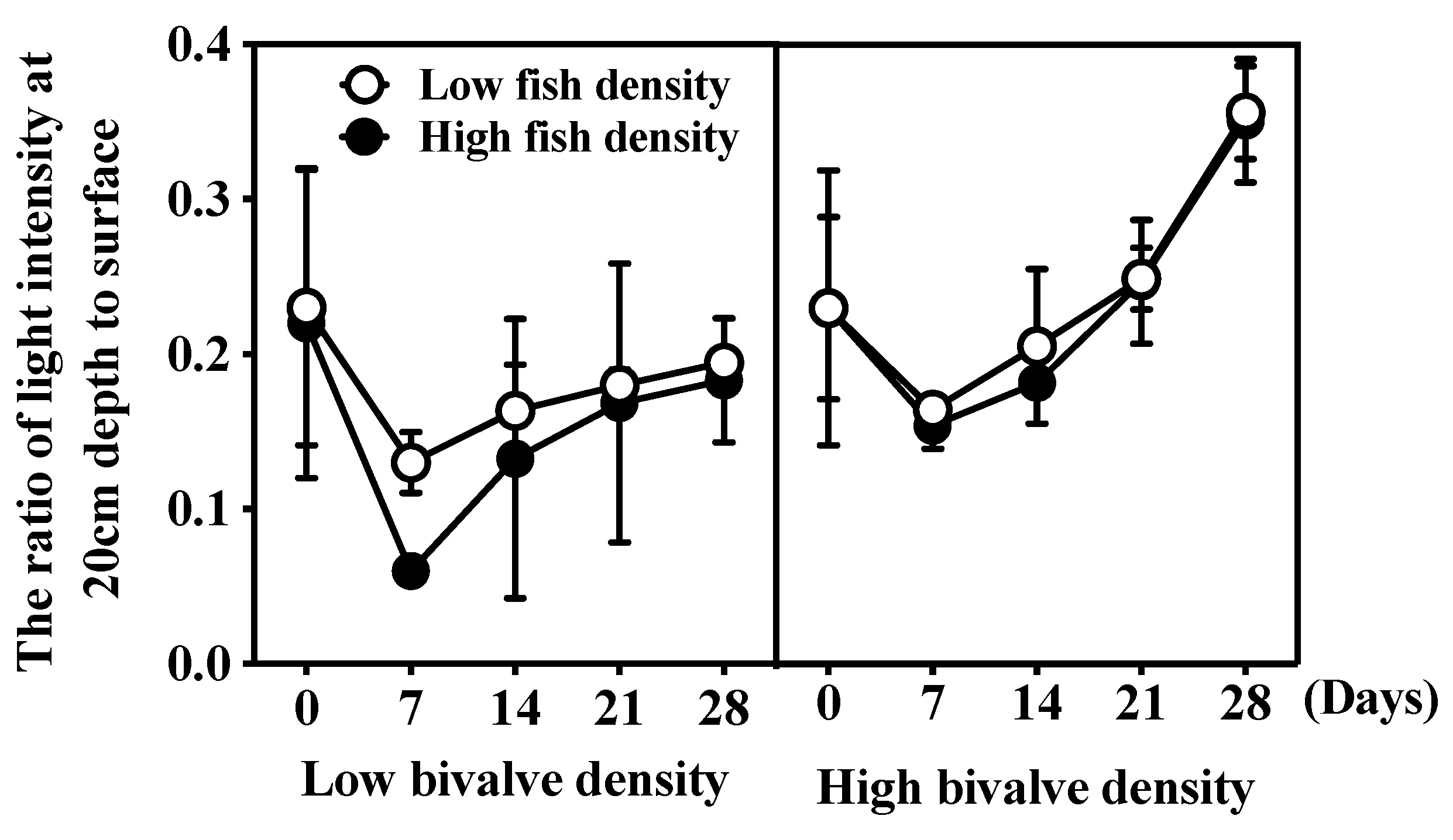
3.2. Water Clarity and Light Intensity
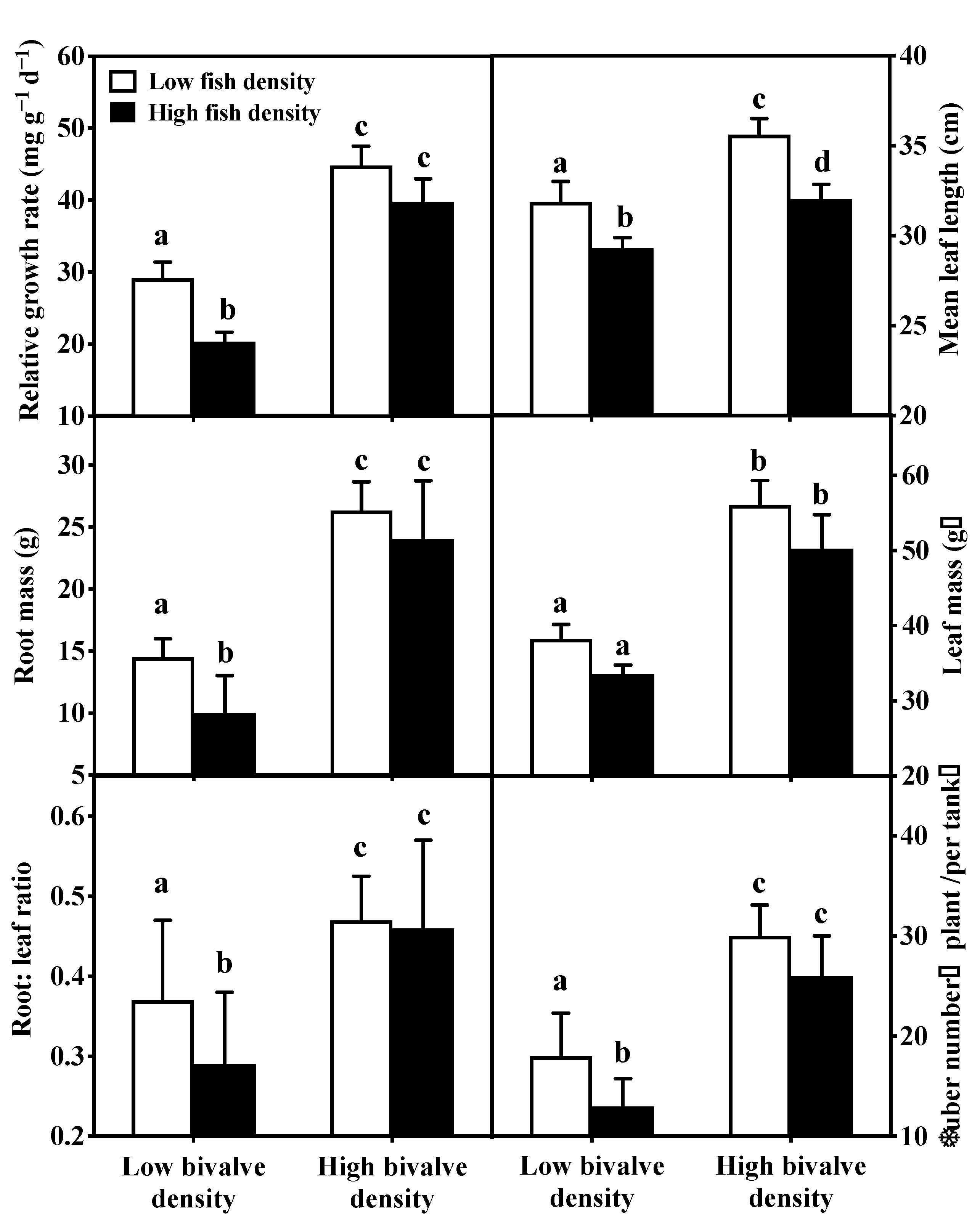
3.3. Macrophytes
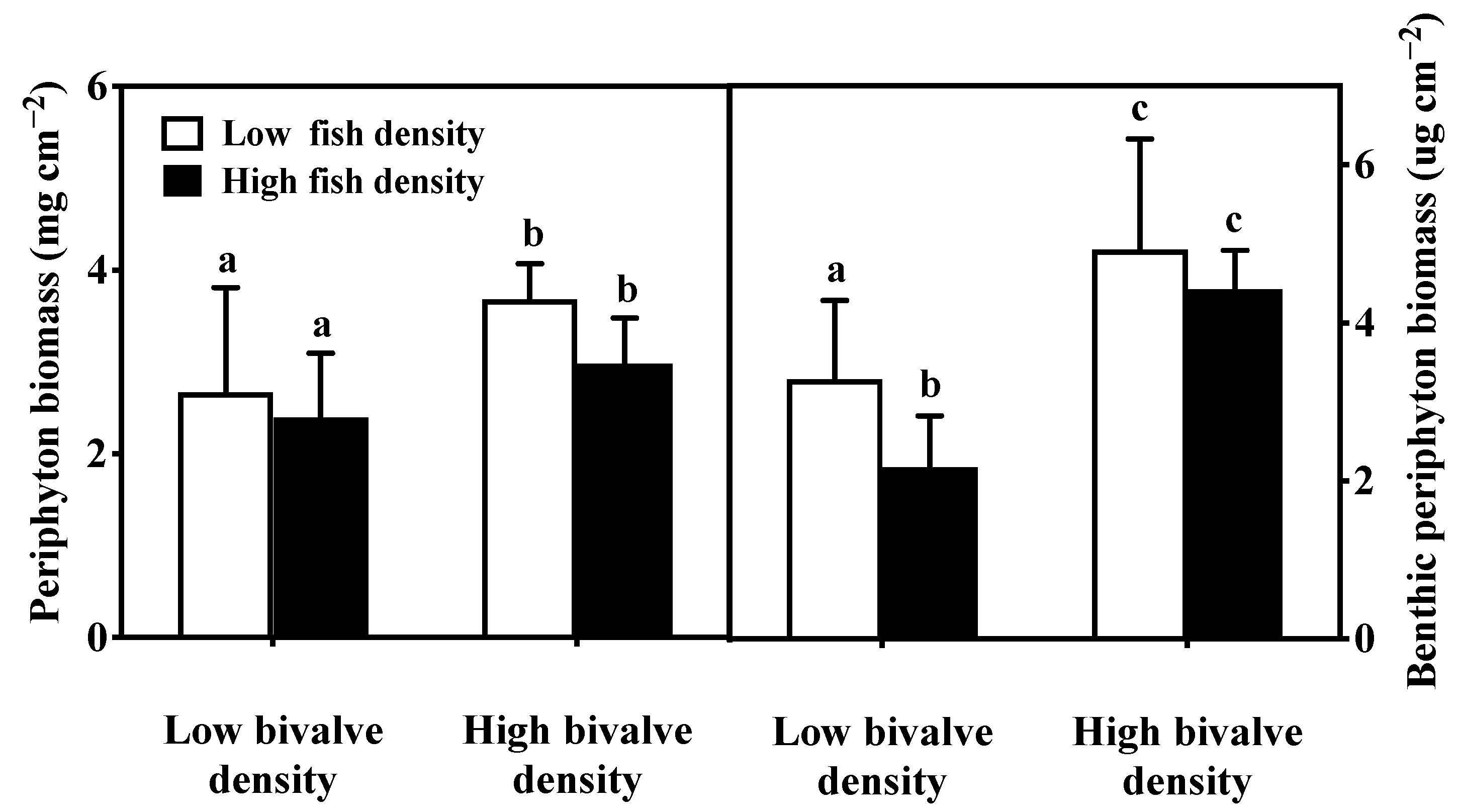
3.4. Periphyton and Benthic Algae
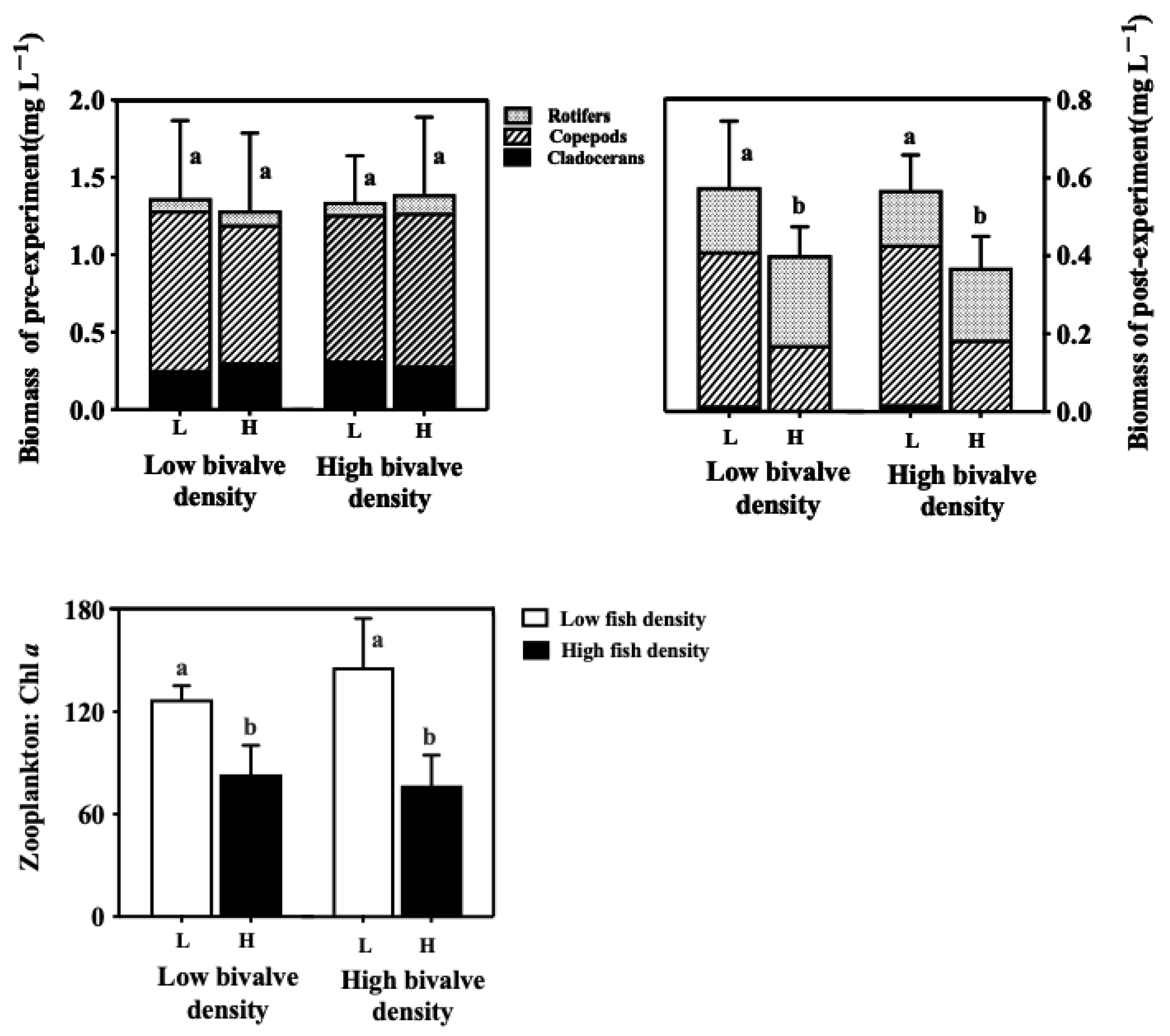
3.5. Zooplankton
3.6. Fish Guts
3.7. Sediments
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Waajen, G.W.A.M.; Van Bruggen, N.C.B.; Pires, L.M.D.; Lengkeek, W.; Lürling, M. Biomanipulation with quagga mussels (Dreissena rostriformis bugensis) to control harmful algal blooms in eutrophic urban ponds. Ecol. Eng. 2016, 90, 141–150. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Lauridsen, T.L.; Davidson, T.A.; Liu, Z.W.; Mazzeo, N.; Trochine, C.; Özkan, K.; Jensen, H.S.; Trolle, D.; et al. Biomanipulation as a restoration tool to combat eutrophication: Recent advances and future challenges. Adv. Ecol. Res. 2012, 47, 411–488. [Google Scholar]
- Chen, Z.; Zhao, D.; Li, M.; Tu, W.; Liu, X. A field study on the effects of combined biomanipulation on the water quality of a eutrophic lake. Environ. Pollut. 2020, 265, 115091. [Google Scholar] [CrossRef]
- Scheffer, M.; Hosper, S.H.; Meijer, M.L.; Moss, B.; Jeppesen, E. Alternative equilibria in shallow lakes. Trends. Ecol. Evol. 1993, 8, 275–279. [Google Scholar] [CrossRef]
- Vanderstukken, M.; Mazzeo, N.; Van Colen, W.; Declerck, S.A.J.; Muylaert, K. Biological control of phytoplankton by the subtropical submerged macrophytes Egeria densa and Potamogeton illinoensis: A mesocosm study. Freshw. Biol. 2011, 56, 1837–1849. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Søndergaard, M.; Christoffersen, K. (Eds.) The Structuring Role of Submerged Macrophytes in Lakes; Springer-Verlag New York, Inc.: New York, NY, USA, 1998. [Google Scholar]
- Hosper, S.H.; Meijer, M.L.; Gulati, R.D.; Van Donk, E. Biomanipulation in shallow lakes: Concepts, case studies and perspectives. Lakes Handb. 2005. [Google Scholar] [CrossRef]
- Ferreira, T.F.; Crossetti, L.O.; Motta Marques, D.M.L.; Cardoso, L.; Fragoso, C.R., Jr.; van Nes, E.H. The structuring role of submerged macrophytes in a large subtropical shallow lake: Clear effects on water chemistry and phytoplankton structure community along a vegetated-pelagic gradient. Limnologica 2018, 69, 142–154. [Google Scholar] [CrossRef]
- Søndergaard, M.; Liboriussen, L.; Pedersen, A.R.; Jeppesen, E. Lake restoration by fish removal: Short- and long-term effects in 36 Danish Lakes. Ecosystems 2008, 11, 1291–1305. [Google Scholar] [CrossRef]
- Meijer, M.L.; de Boois, I.; Scheffer, M.; Portielje, R.; Hosper, H. Biomanipulation in shallow lakes in The Netherlands: An evaluation of 18 case studies. In Shallow Lakes’ 98; Springer: Berlin/Heidelberg, Germany, 1999; pp. 13–30. [Google Scholar]
- Setubal, R.B.; Riccardi, N. Long-term effects of fish biomanipulation and macrophyte management on zooplankton functional diversity and production in a temperate shallow lake. Limnology 2020, 21, 305–317. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Mazzeo, N.; Meerhoff, M.; Christina, C.B.; Huszar, V.; Scasso, F. Lake restoration and biomanipulation in temperate lakes: Relevance for subtropical and tropical lakes. In Their Restoration and Management of Tropical Eutrophic Lakes; Vikram, R., Ed.; Science Publishers Inc.: Enfield, NH, USA, 2005; pp. 341–359. [Google Scholar]
- Liu, Z.; Hu, J.; Zhong, P.; Zhang, X.; Ning, J.; Larsen, S.E.; Chen, D.; Gao, Y.; He, H.; Jeppesen, E. Successful restoration of a tropical shallow eutrophic lake: Strong bottom-up but weak top-down effects recorded. Water Res. 2018, 146, 88–97. [Google Scholar] [CrossRef]
- Jeppesen, E.; Meerhoff, M.; Holmgren, K.; González-Bergonzoni, I.; Mello, T.D.; Declerck, S.A.J.; Meester, L.D.; Søndergaard, M.; Lauridsen, T.L.; Bjerring, R.; et al. Impacts of climate warming on lake fish community structure and potential effects on ecosystem function. Hydrobiologia 2010, 646, 73–90. [Google Scholar] [CrossRef]
- Meerhoff, M.; Clemente, J.M.; Mello, F.T.D.; Iglesias, C.; Pedersen, A.R.; Jeppesen, E. Can warm climate-related structure of littoral predator assemblies weaken the clear water state in shallow lakes? Glob. Chang. Biol. 2007, 13, 1888–1897. [Google Scholar] [CrossRef]
- Gulati, R.D.; Pires, L.M.D.; Van Donk, E. Lake restoration studies: Failures, bottlenecks and prospects of new ecotechnological measures. Limnologica 2008, 38, 233–247. [Google Scholar] [CrossRef]
- Nagdali, S.S.; Gupta, P.K. Impact of mass mortality of a mosquito fish, Gambusia affinis on the ecology of a fresh water eutrophic lake (Lake Naini Tal, India). Hydrobiologia 2002, 468, 45–51. [Google Scholar] [CrossRef]
- Liu, Z.W.; Zhong, P.; Zhang, X.; Ning, J.; Larsen, S.E.; Jeppesen, E. Successful restoration of a tropical shallow eutrophic lake: Strong bottom-up but weak top-down effects recorded. In Proceedings of the Australia-China Wetland Network Research Partnership Symposium, Nanjing, China, 23–28 March 2014; pp. 78–86. [Google Scholar]
- Chen, F.; Shu, T.; Jeppesen, E.; Liu, Z.; Chen, Y. Restoration of a subtropical eutrophic shallow lake in China: Effects on nutrient concentrations and biological communities. Hydrobiologia 2013, 718, 59–71. [Google Scholar] [CrossRef]
- Zhang, M.; Yu, J.; He, H.; Li, K.; Chen, F.; Guan, B.; Hu, Y.; Su, Y.; Du, Y.; Liu, Z. Effects of ecological restoration on water quality of Wuli Bay, Lake Taihu. Ecol. Sci. 2012, 31, 240–244. [Google Scholar]
- Søndergaard, M.; Jeppesen, E.; Lauridsen, T.L.; Skov, C.; Van Nes, E.H.; Roijackers, R.; Lammens, E.; Portielje, R. Lake restoration: Successes, failures and long-term effects. J. Appl. Ecol. 2007, 44, 1095–1105. [Google Scholar] [CrossRef]
- Gulati, R.D.; Van Donk, E. Lakes in the Netherlands, their origin, eutrophication and restoration: State-of-the-art review. In Ecological Restoration of Aquatic and Semi-Aquatic Ecosystems in the Netherlands (NW Europe); Springer: Berlin/Heidelberg, Germany, 2002; Volume 478, pp. 73–106. [Google Scholar]
- Ozersky, T.; Evans, D.O.; Barton, D.R. Invasive mussels alter the littoral food web of a large lake: Stable isotopes reveal drastic shifts in sources and flow of energy. PLoS ONE 2012, 7, e51249. [Google Scholar] [CrossRef]
- De Stasio, B.T.; Schrimpf, M.B.; Beranek, A.E.; Daniels, W.C. Increased Chlorophyll a, phytoplankton abundance, and cyanobacteria occurrence following invasion of Green Bay, Lake Michigan by dreissenid mussels. Aquat. Invasions 2008, 3, 21–27. [Google Scholar] [CrossRef]
- Vaughn, C.C.; Hoellein, T.J. Bivalve impacts in freshwater and marine ecosystems. Annu. Rev. Ecol. Evol. Syst. 2018, 49, 183–208. [Google Scholar] [CrossRef]
- Mclaughlan, C.; Aldridge, D.C. Cultivation of zebra mussels (Dreissena polymorpha) within their invaded range to improve water quality in reservoirs. Water Res. 2013, 47, 4357–4369. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.P.; Lillebø, A.I.; Crespo, D.; Leston, S.; Dolbeth, M. Effect of the alien invasive bivalve Corbicula fluminea on the nutrient dynamics under climate change scenarios. Estuar. Coast. Shelf Sci. 2018, 204, 273–282. [Google Scholar] [CrossRef]
- Mayer, C.M.; Keats, R.A.; Rudstam, L.G.; Mills, E.L. Scale-dependent effects of zebra mussels on benthic invertebrates in a large eutrophic lake. J. N. Am. Benthol. Soc. 2002, 21, 616–633. [Google Scholar] [CrossRef]
- Zhu, B.; Fitzgerald, D.G.; Mayer, C.M.; Rudstam, L.G.; Mills, E.L. Alteration of ecosystem function by zebra mussels in Oneida Lake: Impacts on submerged macrophytes. Ecosystems 2006, 9, 1017–1028. [Google Scholar] [CrossRef]
- Ye, C.; Li, C.H.; Yu, H.C.; Song, X.F.; Zou, G.Y.; Liu, J. Study on ecological restoration in near-shore zone of a eutrophic lake, Wuli Bay, Taihu Lake. Ecol. Eng. 2011, 37, 1434–1437. [Google Scholar] [CrossRef]
- Li, W. Ecological restoration of shallow, eutrophic lakes-experimental studies on the recovery of aquatic vegetation in Wuli Lake. J. Lake Sci. 1996, 8, 1–10. [Google Scholar]
- Yu, J.; Liu, Z.; He, H.; Zhen, W.; Guan, B.; Chen, F.; Li, K.; Zhong, P.; Teixeira-de Mello, F.; Jeppesen, E. Submerged macrophytes facilitate dominance of omnivorous fish in a subtropical shallow lake: Implications for lake restoration. Hydrobiologia 2016, 775, 97–107. [Google Scholar] [CrossRef]
- Jin, X.; Xu, Q.; Yan, C. Restoration scheme for macrophytes in a hypertrophic water body, Wuli Lake, China. Lakes Reserv. Res. Manag. 2006, 11, 21–27. [Google Scholar] [CrossRef]
- Cai, Y.; Gong, Z.; Qin, B. Benthic macroinvertebrate community structure in Lake Taihu, China: Effects of trophic status, wind-induced disturbance and habitat complexity. J. Great Lakes Res. 2012, 38, 39–48. [Google Scholar] [CrossRef]
- Beasley, C.R.; Tagliaro, C.H.; Figueiredo, W.B. The occurrence of the Asian Clam Corbicula fluminea in the lowe amazon basin. Acta Amaz. 2003, 33, 317–324. [Google Scholar] [CrossRef][Green Version]
- Williams, E.H.; Bunkley-Williams, L.; Lilyestrom, C.G.; Ortiz-Corps, E.A.R. A review of recent introductions of aquatic invertebrates in Puerto Rico and implications for the management of nonindigenous species. Caribb. J. Sci. 2001, 37, 246–251. [Google Scholar]
- Marroni, S.; Iglesias, C.; Mazzeo, N.; Clemente, J.; Mello, F.T.D.; Pacheco, J.P. Alternative food sources of native and non-native bivalves in a subtropical eutrophic lake. Hydrobiologia 2014, 735, 263–276. [Google Scholar] [CrossRef]
- McMahon, R.; Bogan, A. Mollusca: Bivalvia. In Ecology and Classification of North American Freshwater Invertebrates, 2nd ed.; Thorp, J.H., Covich, A.P., Eds.; Academic Press, Inc.: San Diego, CA, USA, 2001; pp. 331–430. [Google Scholar]
- Boltovskoy, D.; Izaguirre, I.; Correa, N. Feeding selectivity of Corbicula fluminea (Bivalvia) on natural phytoplankton. Hydrobiologia 1995, 312, 171–182. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Z.; Jeppesen, E. The response of Vallisneria spinulosa (Hydrocharitaceae) to different loadings of ammonia and nitrate at moderate phosphorus concentration: A mesocosm approach. Freshw. Biol. 2008, 53, 2321–2330. [Google Scholar]
- Yu, J.; Liu, Z.; Li, K.; Chen, F.; Guan, B.; Hu, Y.; Zhong, P.; Tang, Y.; Zhao, X.; He, H.; et al. Restoration of shallow lakes in subtropical and tropical China: Response of nutrients and water clarity to biomanipulation by fish removal and submerged plant transplantation. Water 2016, 8, 438. [Google Scholar] [CrossRef]
- Gao, J.; Liu, Z.W.; Jeppesen, E. Fish community assemblages changed but biomass remained similar after lake restoration by biomanipulation in a Chinese tropical eutrophic lake. Hydrobiologia 2014, 724, 127–140. [Google Scholar] [CrossRef]
- Liu, E.; Liu, Z.; Bao, C. The changes of catches of Carassius auratus and relationship with environment in Lake Taihu, China. J. Lake Sci. 2007, 19, 345–350. [Google Scholar]
- Cai, Y.; Gong, Z.; Qin, B. Community structure and diversity of macrozoobenthos in Lake Taihu, a large shallow eutrophic lake in China. Biodivers. Sci. 2010, 18, 50–59. [Google Scholar]
- Jin, X.; Tu, Q. The Standard Methods for Observation and Analysis in Lake Eutrophication; Chinese Environmental Science Press: Beijing, China, 1990. [Google Scholar]
- Wang, J.J. Fauna Sinica, Rotifer; Science Press: Beijing, China, 1961. [Google Scholar]
- Chiang, S.C.; Du, N.S. Fauna Sinica, Crustacea; Science Press: Beijing, China, 1979. [Google Scholar]
- Shen, J.R.; Du, N.S. Fauna Sinica, Crustacea, Freshwater Copepoda; Science Press: Beijing, China, 1979. [Google Scholar]
- Jeppesen, E.; Noges, P.; Davidson, T.A.; Haberman, J.; Noges, T.; Blank, K.; Lauridsen, T.L.; Sondergaard, M.; Sayer, C.D.; Laugaste, R. Zooplankton as indicators in lakes: A scientific-based plea for including zooplankton in the ecological quality assessment of lakes according to the European Water Framework Directive (WFD). Hydrobiologia 2011, 676, 279–297. [Google Scholar] [CrossRef]
- Huang, X.F. Servey Observation and Analysis of Lake Ecology; Standards Press of China: Beijing, China, 2000. [Google Scholar]
- Weber, M.J.; Brown, M.L. Biomass-dependent effects of age-0 common carp on aquatic ecosystems. Hydrobiologia 2015, 742, 71–80. [Google Scholar] [CrossRef]
- Mehner, T.; Thiel, R. A review of predation impact by 0+ fish on zooplankton in fresh and brackish waters of the temperate northern hemisphere. Environ. Biol. Fish 1999, 56, 169–181. [Google Scholar] [CrossRef]
- Meerhoff, M.; Fosalba, C.; Bruzzone, C.; Mazzeo, N.; Noordoven, W.; Jeppesen, E. An experimental study of habitat choice by Daphnia: Plants signal danger more than refuge in subtropical lakes. Freshw. Biol. 2006, 51, 1320–1330. [Google Scholar] [CrossRef]
- Williams, A.E.; Moss, B. Effects of different fish species and biomass on plankton interactions in a shallow lake. Hydrobiologia 2003, 491, 331–346. [Google Scholar] [CrossRef]
- Havens, K.E.; Beaver, J.R. Composition, size, and biomass of zooplankton in large productive Florida lakes. Hydrobiologia 2011, 668, 49–60. [Google Scholar] [CrossRef]
- Meerhoff, M.; Mello, T.D.; Kruk, C.; Alonso, C.; González-Bergonzoni, I.; Pacheco, J.P.; Lacerot, G.; Arim, M.; Beklioğlu, M.; Brucet, S.; et al. Environmental warming in shallow lakes: A review of potential changes in community structure as evidenced from space-for-time substitution approaches. Adv. Ecol. Res. Acad. Press 2012, 46, 259–349. [Google Scholar]
- Zambrano, L.; Hinojosa, D. Direct and indirect effects of carp (Cyprinus carpio L.) on macrophyte and benthic communities in experimental shallow ponds in central Mexico. Hydrobiologia 1999, 408, 131–138. [Google Scholar]
- Dorenbosch, M.; Bakker, E.S. Effects of contrasting omnivorous fish on submerged macrophyte biomass in temperate lakes: A mesocosm experiment. Freshw. Biol. 2012, 57, 1360–1372. [Google Scholar] [CrossRef]
- Korner, S.; Dugdale, T. Is roach herbivory preventing re-colonization of submerged macrophytes in a shallow lake? Hydrobiologia 2003, 506, 497–501. [Google Scholar] [CrossRef]
- van der Wal, J.E.M.; Dorenbosch, M.; Immers, A.K.; Forteza, C.V.; Geurts, J.J.M.; Peeters, E.T.H.M.; Koese, B.; Bakker, E.S. Invasive crayfish threaten the development of submerged macrophytes in lake restoration. PLoS ONE 2013, 8, e78579. [Google Scholar] [CrossRef]
- Yu, J.; Xia, M.; Zhen, W.; He, H.; Shen, R.; Guan, B.; Liu, Z. A small omnivore fish (Acheilognathus macropterus) reduces both growth and biomass of submerged macrophytes: Implications for shallow lake restoration. Knowl. Manag. Aquat. Ecosyst. 2020, 421, 34. [Google Scholar] [CrossRef]
- Zhu, B.; Fitzgerald, D.G.; Hoskins, S.B.; Rudstam, L.G.; Mayer, C.M.; Mills, E.L. Quantification of historical changes of submerged aquatic vegetation cover in two bays of lake Ontario with three complementary methods. J. Great Lakes Res. 2007, 33, 122–135. [Google Scholar] [CrossRef]
- Hwang, S.J.; Kim, H.S.; Park, J.H.; Kim, B.H. Shift in nutrient and plankton community in eutrophic lake following introduction of a freshwater bivalve. J. Environ. Biol. 2011, 32, 227–234. [Google Scholar] [PubMed]
- Cronin, G.; Lodge, D.M. Effects of light and nutrient availability on the growth, allocation, carbon/nitrogen balance, phenolic chemistry, and resistance to herbivory of two freshwater macrophytes. Oecologia 2003, 137, 32–41. [Google Scholar] [CrossRef]
- Chappell, J.; Whitmire, S.; Sotomayor-Ramírez, D.; Martínez-Rodríguez, G. Effects of Corbicula fluminea on the nutrient concentration and phytoplankton biomass of tropical reservoirs. Int. Aquat. Res. 2019, 11, 401–415. [Google Scholar] [CrossRef]
- Mcmahon, R.F. Ecology of an invasive pest bivalve. In Corbicula. Biology of Mollusca, Ecology; Russell-Hunter, W.D., Ed.; Academic Press: New York, NY, USA, 1983; Volume 6. [Google Scholar]
- Vanderploeg, H.A.; Liebig, J.R.; Carmichael, W.W.; Agy, M.A.; Johengen, T.H.; Fahnenstiel, G.L.; Nalepa, T.F. Zebra mussel (Dreissena polymorpha) selective filtration promoted toxic blooms in Saginaw Bay (Lake Huron) and Lake Erie. Can. J. Fish. Aquat. Sci. 2001, 58, 1208–1221. [Google Scholar] [CrossRef]
- Basen, T.; Martincreuzburg, D.; Rothhaupt, K. Role of essential lipids in determining food quality for the invasive freshwater clam Corbicula fluminea. J. N. Am. Benthol. Soc. 2011, 30, 653–664. [Google Scholar] [CrossRef]
- Sousa, R.; Gutiérrez, J.L.; Aldridge, D.C. Non-indigenous invasive bivalves as ecosystem engineers. Biol. Invasions 2009, 11, 2367–2385. [Google Scholar] [CrossRef]
- Vaughn, C.C.; Hakenkamp, C.C. The functional role of burrowing bivalves in freshwater ecosystems. Freshw. Biol. 2010, 46, 1431–1446. [Google Scholar] [CrossRef]
- Peterson, B.J.; Heck, K.L.J. Positive interactions between suspension-feeding bivalves and seagrassa facultative mutualism. Mar. Ecol. Prog. Ser. 1999, 213, 143–155. [Google Scholar] [CrossRef]
- Fourqurean, J.W.; Zieman, J.C. Nutrient content of the seagrass Thalassia testudinum reveals regional patterns of relative availability of nitrogen and phosphorus in the Florida Keys USA. Biogeochemistry 2002, 61, 229–245. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Z.; Jeppesen, E.; Taylor, W.D. Effects of deposit-feeding tubificid worms and filter-feeding bivalves on benthic-pelagic coupling: Implications for the restoration of eutrophic shallow lakes. Water Res. 2014, 50, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Nalepa, T.F.; Gardner, W.S.; Malczyk, J.M. Phosphorus cycling by mussels (Unionidae: Bivalvia) in Lake St. Clair. Hydrobiologia 1991, 219, 239–250. [Google Scholar] [CrossRef]
- Shen, R.; Gu, X.; Chen, H.; Mao, Z.; Jeppesen, E. Combining bivalve (Corbicula fluminea) and filter-feeding fish (Aristichthys nobilis) enhances the bioremediation effect of algae: An outdoor mesocosm study. Sci. Total Environ. 2020, 727, 138692. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.L.; Zhang, G.F.; Liu, Q.G.; Li, J.L. Optimization of culturing the freshwater pearl mussels, Hyriopsis cumingii with filter feeding Chinese carps (bighead carp and silver carp) by orthogonal array design. Aquaculture 2009, 292, 60–66. [Google Scholar] [CrossRef]
- Søndergaard, M.; Lauridsen, T.L.; Johansson, L.S.; Jeppesen, E. Repeated fish removal to restorelakes: Case study of Lake Væng, Denmark-two biomanipulations during 30 years of monitoring. Water 2017, 9, 43. [Google Scholar] [CrossRef]
- Higgins, S.N.; Zanden, M.J.V. What a difference a species makes: A meta–analysis of dreissenid mussel impacts on freshwater ecosystems. Ecol. Monogr. 2010, 80, 179–196. [Google Scholar] [CrossRef]
- Gu, J.; Jin, H.; He, H.; Ning, X.Y.; Yu, J.L.; Tan, B.C.; Jeppesen, E.; Li, K.Y. Effects of small-sized crucian carp (Carassius carassius) on the growth of submerged macrophytes: Implications for shallow lake restoration. Ecol. Eng. 2016, 95, 567–573. [Google Scholar] [CrossRef]
- Zhu, X.L.; Gu, J.; Jin, H.; Ning, X.Y.; He, H.; Tan, B.C.; Yang, G.J.; Li, K.Y. Effects of Corbicula fluminea in Lake Taihu on improvement of eutrophic water. J. Lake Sci. 2009, 21, 314–328. [Google Scholar]
- Wu, Z.; Qiu, X.; Zhang, X.; Liu, Z.; Tang, Y. Effects of Anodonta woodiana on water quality improvement in restoration of eutrophic shallow lakes. J. Lake Sci. 2018, 30, 1610–1615. [Google Scholar]
- Kang, Y.H.; Song, X.L.; Liu, Z.W. Sediment resuspension dampens the effect of nutrient inputs on the phytoplankton community: A mesocosm experiment study. Hydrobiologia 2013, 710, 117–127. [Google Scholar] [CrossRef]
| Effect | DF | F Value | Pr > F | DF | F Value | Pr > F | ||
|---|---|---|---|---|---|---|---|---|
| TN | Fish (F) | 1 | 37.80 | ** | Chl a | 1 | 14.59 | ** |
| Bivalve (B) | 1 | 154.55 | ** | 1 | 51.27 | ** | ||
| F × B | 1 | 8.54 | * | 1 | 11.74 | * | ||
| TP | Fish (F) | 1 | 48.80 | ** | Turbid | 1 | 28.78 | ** |
| Bivalve (B) | 1 | 94.43 | ** | 1 | 151.89 | ** | ||
| F × B | 1 | 14.66 | ** | 1 | 11.81 | ** | ||
| TDN | Fish (F) | 1 | 1.12 | n.s. | TSS | 1 | 25.20 | ** |
| Bivalve (B) | 1 | 3.37 | n.s. | 1 | 98.46 | ** | ||
| F × B | 1 | 1.28 | n.s. | 1 | 5.12 | * | ||
| TDP | Fish (F) | 1 | 0.01 | n.s. | ISS | 1 | 16.07 | ** |
| Bivalve (B) | 1 | 1.26 | n.s. | 1 | 61.65 | ** | ||
| F × B | 1 | 3.76 | n.s. | 1 | 2.53 | n.s. | ||
| NH4-N | Fish (F) | 1 | 6.07 | * | Lux | 1 | 19.11 | ** |
| Bivalve (B) | 1 | 0.05 | n.s. | 1 | 239.7 | ** | ||
| F × B | 1 | 0.24 | n.s. | 1 | 17.90 | ** | ||
| SRP | Fish (F) | 1 | 10.60 | ** | ||||
| Bivalve (B) | 1 | 0.54 | n.s. | |||||
| F × B | 1 | 0.81 | n.s. |
| Fish | Bivalve | Fish × Bivalve | |||||||
|---|---|---|---|---|---|---|---|---|---|
| DF | F | Pr > F | DF | F | Pr > F | DF | F | Pr > F | |
| Plant N concentration | 1 | 10.65 | * | 1 | 197.66 | ** | 1 | 1.36 | n.s. |
| Plant P concentration | 1 | 0.78 | n.s. | 1 | 370.36 | ** | 1 | 0.35 | n.s. |
| Plant C:N ratio | 1 | 1.23 | n.s. | 1 | 174.11 | ** | 1 | 0.70 | n.s. |
| Plant C:P ratio | 1 | 2.29 | n.s. | 1 | 18.04 | * | 1 | 0.38 | n.s. |
| Relative growth rate | 1 | 30.94 | ** | 1 | 147.13 | ** | 1 | 20.22 | ** |
| Mean leaf length | 1 | 48.62 | ** | 1 | 53.48 | ** | 1 | 1.29 | n.s. |
| Leaf mass | 1 | 20.82 | ** | 1 | 95.36 | ** | 1 | 2.24 | n.s. |
| Root mass | 1 | 9.12 | * | 1 | 47.34 | ** | 1 | 8.18 | * |
| Root:shoot ratio | 1 | 10.59 | * | 1 | 20.5 | ** | 1 | 7.62 | * |
| Tuber numbers | 1 | 80.66 | ** | 1 | 726.00 | ** | 1 | 10.67 | ** |
| Periphyton biomass | 1 | 6.25 | n.s. | 1 | 29.90 | ** | 1 | 6.10 | n.s. |
| Chl a of benthic algae | 1 | 32.70 | ** | 1 | 58.60 | ** | 1 | 12.97 | * |
| Zooplankton biomass | 1 | 44.60 | * | 1 | 0.41 | n.s. | 1 | 0.07 | n.s. |
| Zooplankton:Chl a | 1 | 26.18 | ** | 1 | 0.37 | n.s. | 1 | 0.01 | n.s. |
| Sediment TN content | 1 | 0.002 | n.s. | 1 | 6.21 | * | 1 | 0.04 | n.s. |
| Sediment TP content | 1 | 0.01 | n.s. | 1 | 8.24 | * | 1 | 0.39 | n.s. |
| Sediment OM content | 1 | 0.27 | n.s. | 1 | 0.22 | n.s. | 1 | 2.90 | n.s. |
| Low Bivalve Density | High Bivalve Density | |||
|---|---|---|---|---|
| Prey Items | Low Fish Density | High Fish Density | Low Fish Density | High Fish Density |
| Macrophytes | 50 | 56.3 | 50 | 62.5 |
| Zooplankton | 100 | 81.3 | 100 | 75 |
| Filamentous algae | 25 | 31.3 | 50 | 56.3 |
| Low Density Bivalve | High Density Bivalve | |||
|---|---|---|---|---|
| Prey Items | Low Density Fish | High Density Fish | Low Density Fish | High Density Fish |
| TN (mg kg−1) | 1508 ± 119 | 1525 ± 102 | 1688 ± 253 | 1744 ± 99 |
| TP (mg kg−1) | 235 ± 17 | 237 ± 60 | 266 ± 33 | 260 ± 37 |
| OM% | 1.51 ± 0.10 | 1.72 ± 0.05 | 1.66 ± 0.06 | 1.77 ± 0.13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gu, J.; Li, K.; Jeppesen, E.; Han, Y.; Jin, H.; He, H.; Ning, X. Using Freshwater Bivalves (Corbicula Fluminea) to Alleviate Harmful Effects of Small-Sized Crucian Carp (Carassius Carassius) on Growth of Submerged Macrophytes during Lake Restoration by Biomanipulation. Water 2020, 12, 3161. https://doi.org/10.3390/w12113161
Gu J, Li K, Jeppesen E, Han Y, Jin H, He H, Ning X. Using Freshwater Bivalves (Corbicula Fluminea) to Alleviate Harmful Effects of Small-Sized Crucian Carp (Carassius Carassius) on Growth of Submerged Macrophytes during Lake Restoration by Biomanipulation. Water. 2020; 12(11):3161. https://doi.org/10.3390/w12113161
Chicago/Turabian StyleGu, Jiao, Kuanyi Li, Erik Jeppesen, Yanqing Han, Hui Jin, Hu He, and Xiaoyu Ning. 2020. "Using Freshwater Bivalves (Corbicula Fluminea) to Alleviate Harmful Effects of Small-Sized Crucian Carp (Carassius Carassius) on Growth of Submerged Macrophytes during Lake Restoration by Biomanipulation" Water 12, no. 11: 3161. https://doi.org/10.3390/w12113161
APA StyleGu, J., Li, K., Jeppesen, E., Han, Y., Jin, H., He, H., & Ning, X. (2020). Using Freshwater Bivalves (Corbicula Fluminea) to Alleviate Harmful Effects of Small-Sized Crucian Carp (Carassius Carassius) on Growth of Submerged Macrophytes during Lake Restoration by Biomanipulation. Water, 12(11), 3161. https://doi.org/10.3390/w12113161






