Skip the Dip—Avoid the Risk? Integrated Microbiological Water Quality Assessment in the South-Eastern Baltic Sea Coastal Waters
Abstract
:1. Introduction
2. Materials and Methods
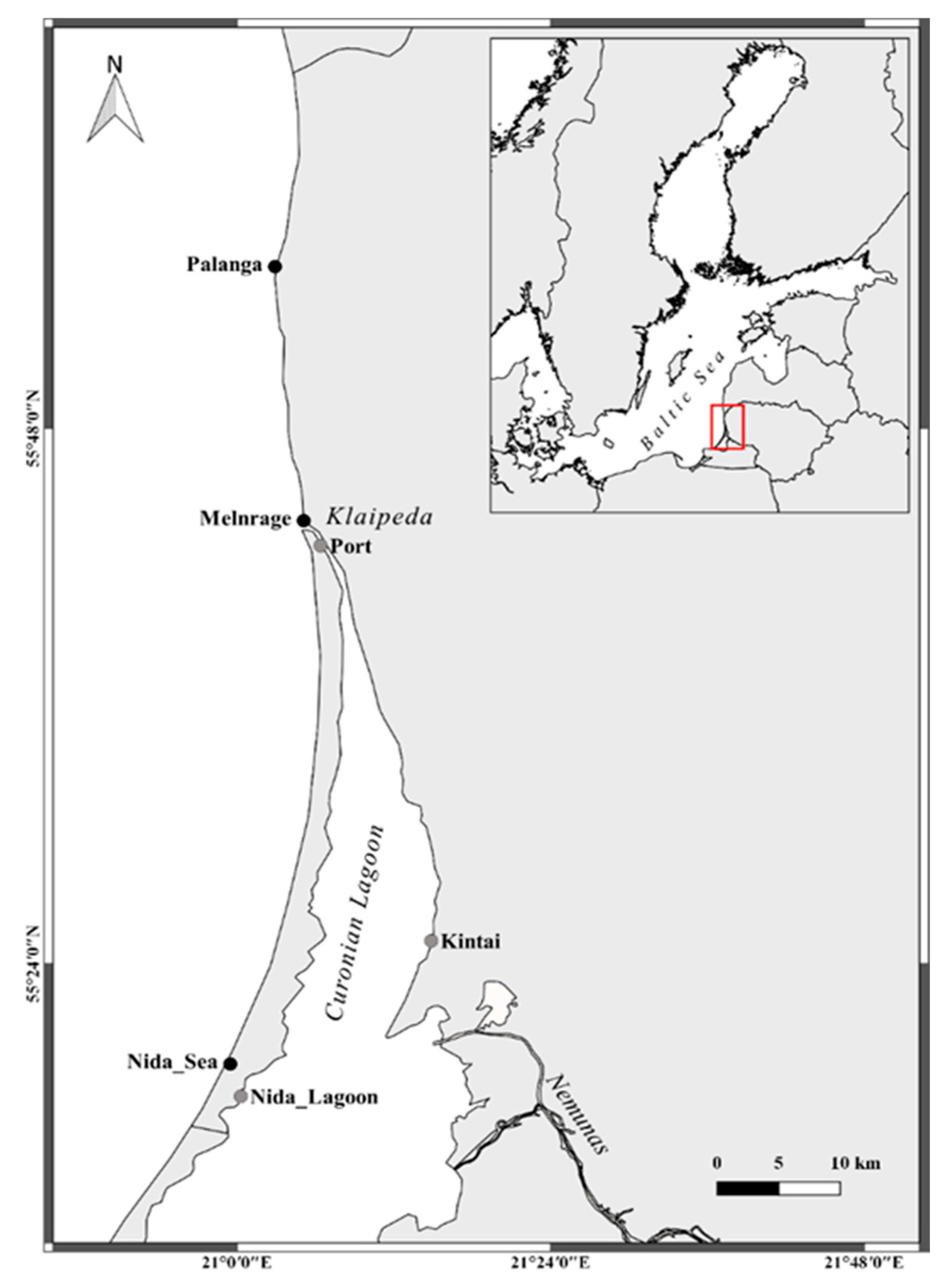
2.1. Study Area and Sample Collection
2.2. Identification and Enumeration of Microorganisms
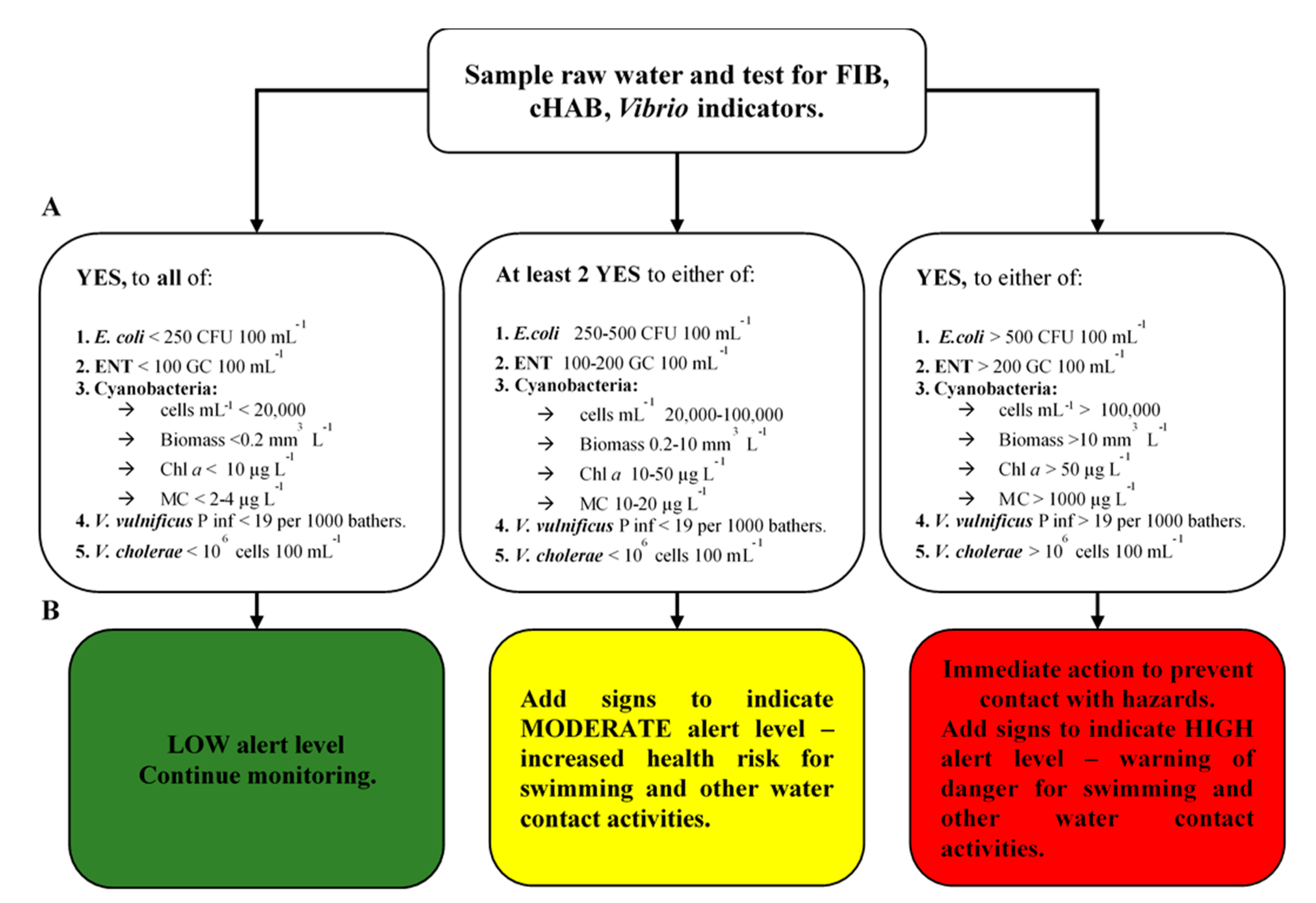
2.3. Integrated Microbial Risk Assessment of Swimming-Associated Illnesses
- (a)
- bathing water suitability for recreational use was defined by established limits of fecal indicator bacteria (FIB) such as E. coli and Enterococcus (BWD, 2006/7/EC) [7];
- (b)
- (c)
- for safe practice in managing bathing waters, WHO guideline values classifying severity and probability of cyanobacteria impact on health at three levels of risk (‘low’, ‘moderate’, and ‘high’; see Table 2) were applied. Total cells of cyanobacteria (mL−1), Chl a (µg L−1), cyanobacterial biomass (mm3 L−1), and the total concentration of MCs (µg L−1) were used as indicators of potential risk for adverse health outcome from exposure to cyanobacteria blooms [1,56,57]; and
- (d)
- due to a lack of existing thresholds for water-based potentially pathogenic Vibrio spp. bacteria, minimum infectious dose (104 cells mL−1 or 106 cells 100 mL−1), as defined by Hornick et al. [58], was used for V. cholerae risk assessment. In this study, we assume that GC corresponds to a cell number. For potentially pathogenic V. vulnificus, QMRA was applied.
2.4. Statistical Analysis
3. Results
3.1. Spatial and Temporal Variability of FIB
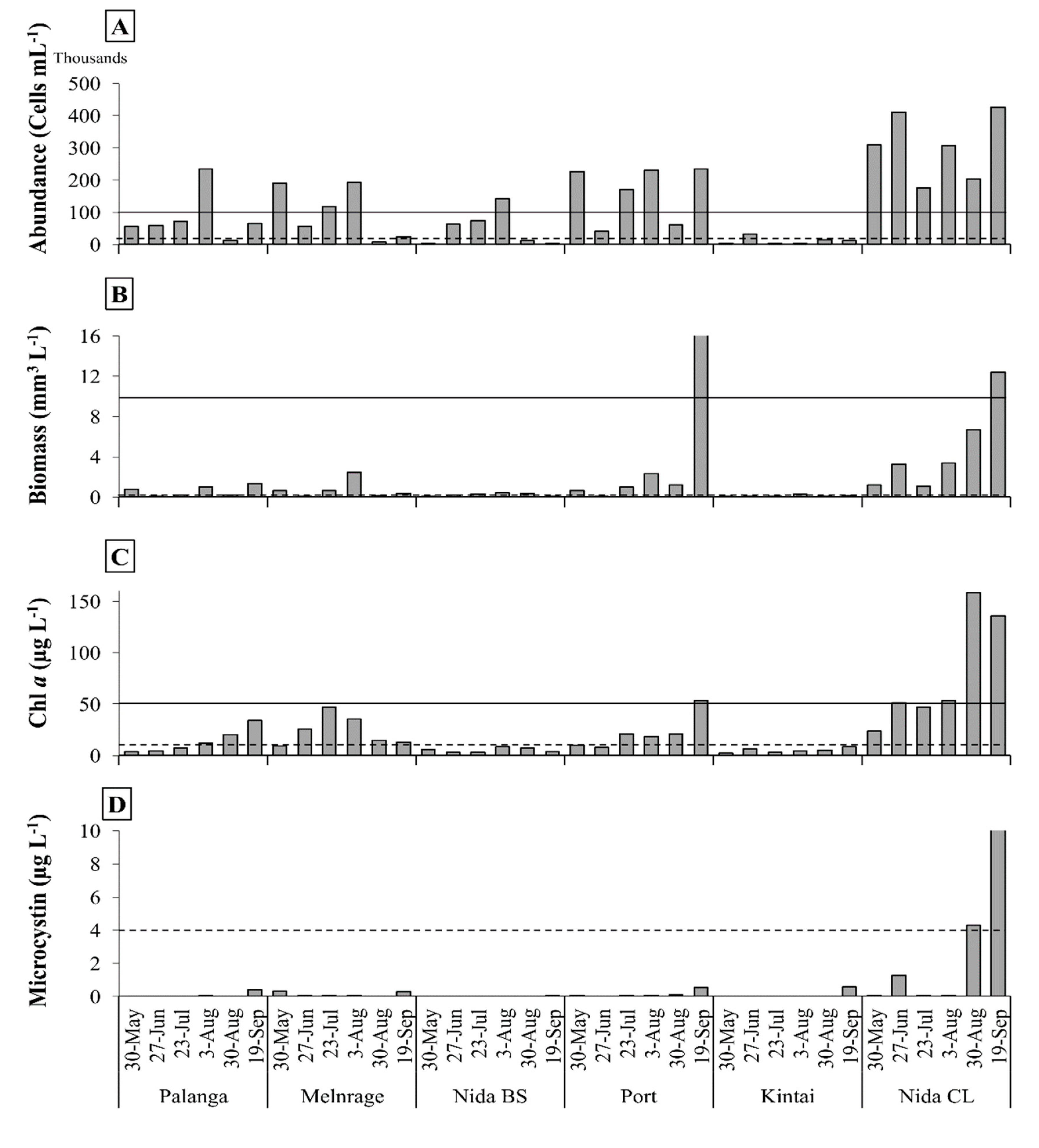
3.2. Spatial and Temporal Variability of cHAB
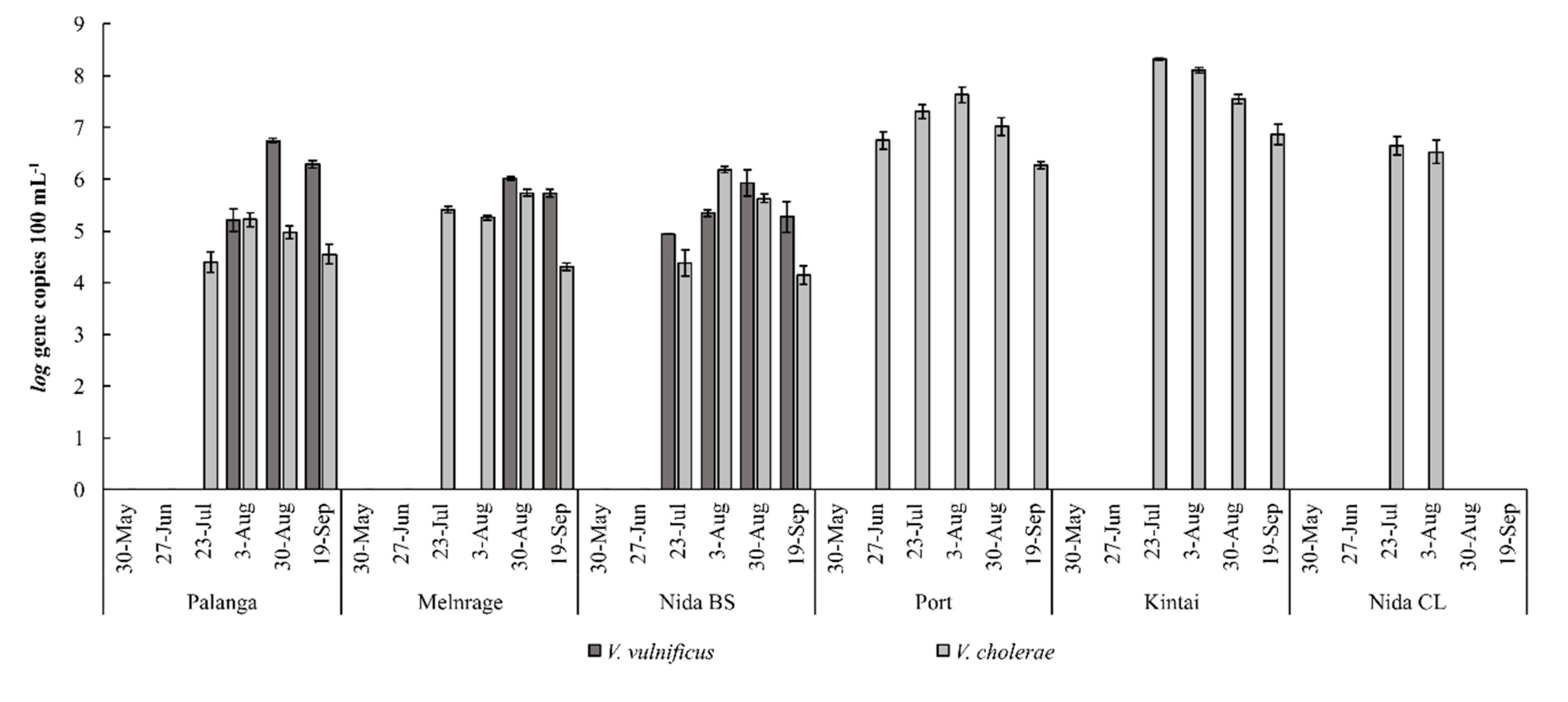
3.3. V. vulnificus and V. cholerae Abundance and the Risk of Infection
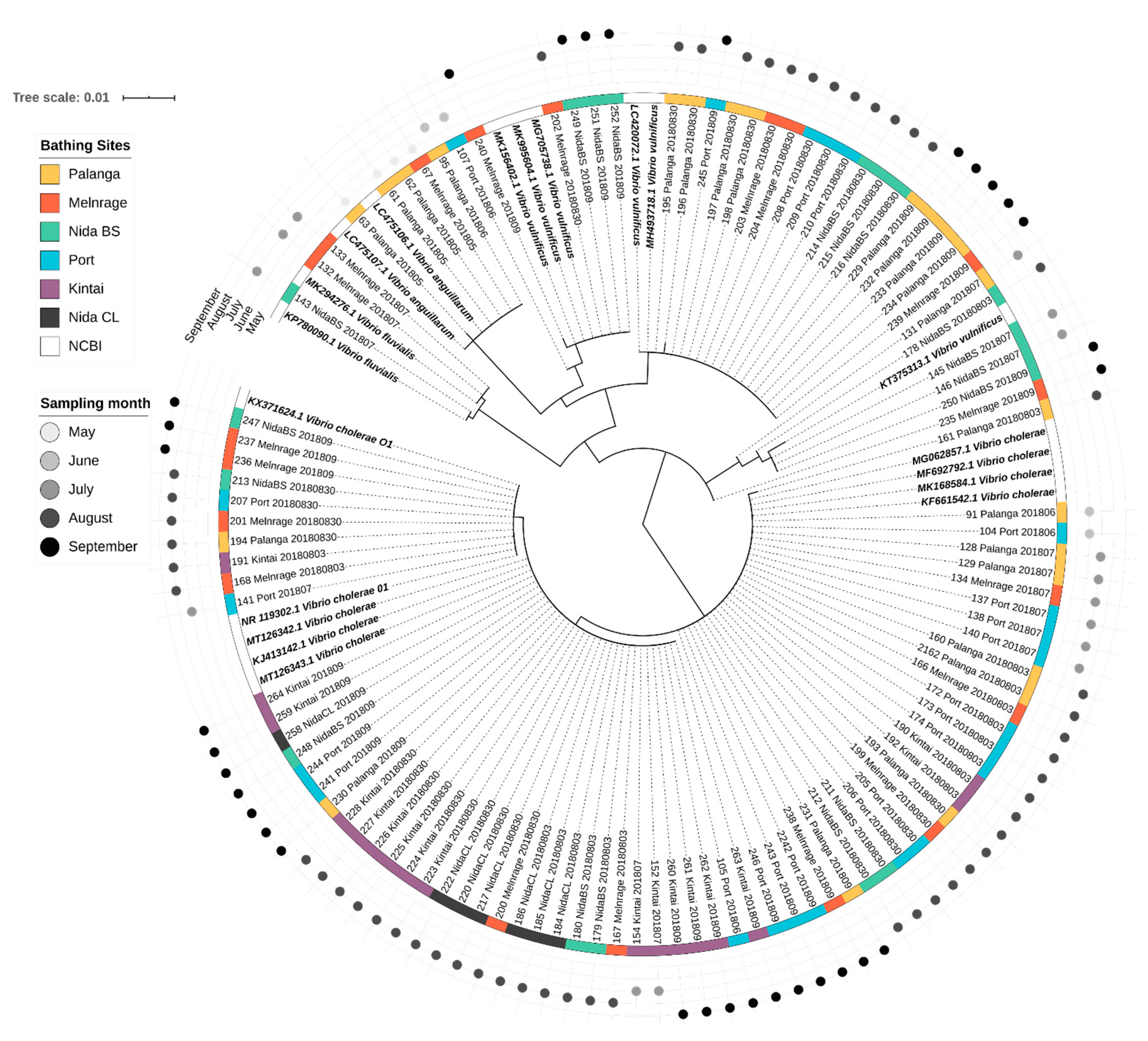
3.4. Diversity of Potentially Pathogenic Bacteria
3.5. Decision Scheme for Integrated Microbial Risk Assessment
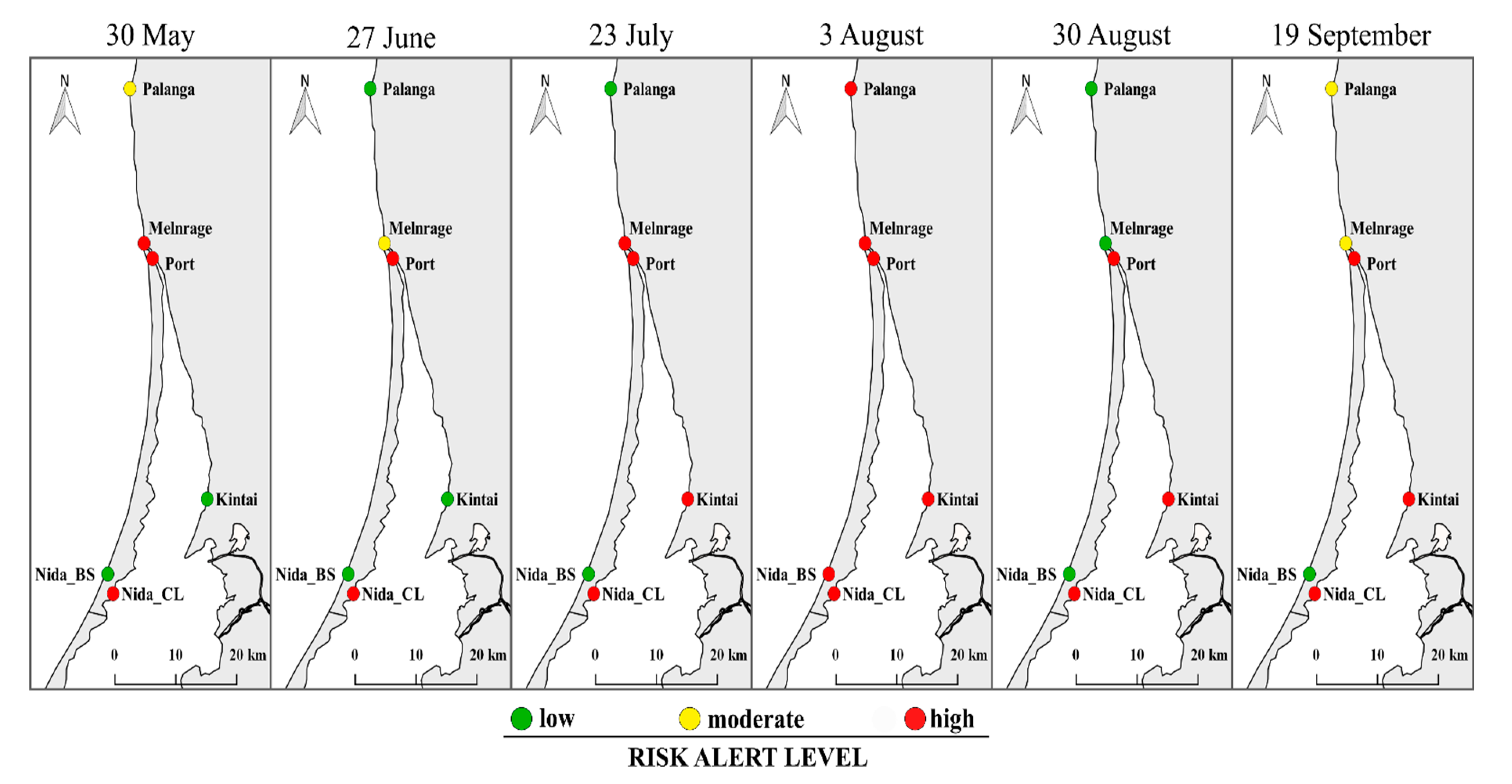
3.6. Integrated Microbial Risk Assessment of the Recreational Waters
4. Discussion
5. Conclusions
- This is the first study presenting an integrated microbial risk analysis in the Lithuanian coastal and Curonian Lagoon bathing sites.
- The E. coli concentration in all sampling sites over the study period was below the ‘moderate’ risk alert level, except in June in Nida CL. Exceedances of ‘moderate’ and ‘high’ Enterococcus risk thresholds were observed for the Curonian Lagoon sites.
- cHAB abundance, biomass, and Chl a exceeded ‘moderate’ and ‘high’ risk thresholds in more than 50% of all samples at all the sampling sites, except Kintai. The concentration of MC reached the ‘moderate’ alert level threshold only in Nida CL.
- In 2018, the potential infection risk from V. vulnificus was below the acceptable illness benchmark (19 out of 1000 bathers), although its abundance at some sampling sites was two times higher than in 2017 [32]. In the Curonian Lagoon sites, the minimum infectious dose of V. cholerae was exceeded over the study period, except at the beginning of the bathing season in Kintai.
- A new approach for bathing water profiling based on more target indicators for water quality, such as cHAB and Vibrio, revealed more bathing sites of ‘high’ and ‘moderate’ risk alert level. In contrast, the FIB indicators showed an excellent water quality.
- The integrated microbial risk assessment presented in this study covers a wider spectrum of risks caused by potentially harmful organisms and could be applied in similar fresh–brackish water systems.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Faecal Pollution and Water Quality, Guidelines for Safe Recreational Environments. Volume 1: Coastal and Fresh Waters. 2003. Available online: https://www.who.int/water_sanitation_health/bathing/srwg1.pdf (accessed on 2 September 2020).
- Testai, E.; Buratti, F.M.; Funari, E.; Manganelli, M.; Vichi, S.; Arnich, N.; Biré, R.; Fessard, V.; Sialehaamoa, A. Review and analysis of occurrence, exposure and toxicity of cyanobacteria toxins in food. EFSA Support. Publ. 2017, 13. [Google Scholar] [CrossRef]
- Sunger, N.; Hamilton, K.A.; Morgan, P.M.; Haas, C.N. Comparison of pathogen-derived ‘total risk’ with indicator-based correlations for recreational (swimming) exposure. Environ. Sci. Pollut. Res. 2019, 26, 30614–30624. [Google Scholar] [CrossRef] [PubMed]
- Gerba, C.P.; Pepper, I.L. Microbial Contaminants. In Environmental and Pollution Science; Elsevier: Amsterdam, The Netherlands, 2019; pp. 191–217. [Google Scholar]
- Shannon, K.E.; Lee, D.Y.; Trevors, J.T.; Beaudette, L.A. Application of real-time quantitative PCR for the detection of selected bacterial pathogens during municipal wastewater treatment. Sci. Total Environ. 2007, 382, 121–129. [Google Scholar] [CrossRef] [PubMed]
- European Union, Directive 76/160/ECC of the European Parliament and the Council. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:31976L0160&from=EN (accessed on 2 September 2020).
- European Union. Directive 2006/7/EC of the European Parliament and the Council. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32006L0007 (accessed on 2 September 2020).
- Wade, T.J.; Sams, E.; Brenner, K.P.; Haugland, R.; Chern, E.; Beach, M.; Wymer, L.; Rankin, C.C.; Love, D.; Li, Q.; et al. Rapidly measured indicators of recreational water quality and swimming-associated illness at marine beaches: A prospective cohort study. Environ. Health Glob. Access Sci. Source 2010, 9, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Union. Support to the Assessment of Member States’ Compliance with the Bathing Water Directive 2006/7/EC. Final EU Overview Report. Available online: https://ec.europa.eu/environment/water/water-bathing/pdf/BWD_EU_Report.pdf (accessed on 2 September 2020).
- Byappanahalli, M.N.; Nevers, M.B.; Korajkic, A.; Staley, Z.R.; Harwood, V.J. Enterococci in the Environment. Microbiol. Mol. Biol. Rev. 2012, 76, 685–706. [Google Scholar] [CrossRef] [Green Version]
- Bonadonna, L.; Briancesco, R.; Suffredini, E.; Coccia, A.; Della Libera, S.; Carducci, A.; Verani, M.; Federigi, I.; Iaconelli, M.; Bonanno Ferraro, G.; et al. Enteric viruses, somatic coliphages and Vibrio species in marine bathing and non-bathing waters in Italy. Mar. Pollut. Bull. 2019, 149, 110570. [Google Scholar] [CrossRef]
- Oliver, D.M.; van Niekerk, M.; Kay, D.; Heathwaite, A.L.; Porter, J.; Fleming, L.E.; Kinzelman, J.L.; Connolly, E.; Cummins, A.; McPhail, C.; et al. Opportunities and limitations of molecular methods for quantifying microbial compliance parameters in EU bathing waters. Environ. Int. 2014, 64, 124–128. [Google Scholar] [CrossRef]
- Whitman, R.L.; Harwood, V.J.; Edge, T.A.; Nevers, M.B.; Byappanahalli, M.; Vijayavel, K.; Brandao, J.; Sadowsky, M.J.; Alm, E.W.; Crowe, A.; et al. Microbes in Beach Sands: Integrating Environment, Ecology and Public Health. Building 2008, 45, 258–266. [Google Scholar] [CrossRef] [Green Version]
- Harwood, V.J.; Staley, C.; Badgley, B.D.; Borges, K.; Korajkic, A. Microbial source tracking markers for detection of fecal contamination in environmental waters: Relationships between pathogens and human health outcomes. FEMS Microbiol. Rev. 2014, 38, 1–40. [Google Scholar] [CrossRef] [Green Version]
- Tan, B.F.; Ng, C.; Nshimyimana, J.P.; Loh, L.L.; Gin, K.Y.H.; Thompson, J.R. Next-generation sequencing (NGS) for assessment of microbial water quality: Current progress, challenges, and future opportunities. Front. Microbiol. 2015, 6, 1027. [Google Scholar] [CrossRef]
- Haugland, R.A.; Varma, M.; Sivaganesan, M.; Kelty, C.; Peed, L.; Shanks, O.C. Evaluation of genetic markers from the 16S rRNA gene V2 region for use in quantitative detection of selected Bacteroidales species and human fecal waste by qPCR. Syst. Appl. Microbiol. 2010, 33, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Ashbolt, N.J.; Schoen, M.E.; Soller, J.A.; Roser, D.J. Predicting pathogen risks to aid beach management: The real value of quantitative microbial risk assessment (QMRA). Water Res. 2010, 44, 4692–4703. [Google Scholar] [CrossRef] [PubMed]
- WHO Recommendations on Scientific, Analytical and Epidemiological Developments Relevant to the Parameters for Bathing Water qualIty in the Bathing Water Directive. Available online: https://circabc.europa.eu/d/d/workspace/SpacesStore/9e89152c-7cfe-4391-9bcf-c173519e8181/WHO Recommendations on EC BWD (accessed on 2 September 2020).
- Šulčius, S.; Pilkaityte, R.; Mazur-Marzec, H.; KasperovičienE, J.; Ezhova, E.; Błaszczyk, A.; Paškauskas, R. Increased risk of exposure to microcystins in the scum of the filamentous cyanobacterium Aphanizomenon flos-aquae accumulated on the western shoreline of the Curonian Lagoon. Mar. Pollut. Bull. 2015, 99, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Overlingė, D.; Kataržytė, M.; Vaičiūtė, D.; Gyraite, G.; Gečaitė, I.; Jonikaitė, E.; Mazur-Marzec, H. Are there concerns regarding cHAB in coastal bathing waters affected by freshwater-brackish continuum? Mar. Pollut. Bull. 2020, 159, 111500. [Google Scholar] [CrossRef] [PubMed]
- Mazur-Marzec, H.; Sutryk, K.; Kobos, J.; Hebel, A.; Hohlfeld, N.; Błaszczyk, A.; Toruńska, A.; Kaczkowska, M.J.; Łysiak-Pastuszak, E.; Kraśniewski, W.; et al. Occurrence of cyanobacteria and cyanotoxin in the Southern Baltic Proper. Filamentous cyanobacteria versus single-celled picocyanobacteria. Hydrobiologia 2013, 701, 235–252. [Google Scholar] [CrossRef] [Green Version]
- Baker-Austin, C.; Lemm, E.; Hartnell, R.; Lowther, J.; Onley, R.; Amaro, C.; Oliver, J.D.; Lees, D. PilF polymorphism-based real-time PCR to distinguish Vibrio vulnificus strains of human health relevance. Food Microbiol. 2012, 30, 17–23. [Google Scholar] [CrossRef] [Green Version]
- Lukinmaa, S.; Mattila, K.; Lehtinen, V.; Hakkinen, M.; Koskela, M.; Siitonen, A. Territorial waters of the Baltic Sea as a source of infections caused by Vibrio cholerae non-O1, non-O139: Report of 3 hospitalized cases. Diagn. Microbiol. Infect. Dis. 2006, 54, 1–6. [Google Scholar] [CrossRef]
- Baker-Austin, C.; Trinanes, J.A.; Salmenlinna, S.; Löfdahl, M.; Siitonen, A.; Taylor, N.G.H.; Martinez-Urtaza, J. Heat wave-associated vibriosis, Sweden and Finland, 2014. Emerg. Infect. Dis. 2016, 22, 1216–1220. [Google Scholar] [CrossRef]
- Huehn, S.; Eichhorn, C.; Urmersbach, S.; Breidenbach, J.; Bechlars, S.; Bier, N.; Alter, T.; Bartelt, E.; Frank, C.; Oberheitmann, B.; et al. Pathogenic Vibrios in Environmental, Seafood and Clinical Sources in Germany. Available online: https://www.clinicalkey.com/#!/content/journal/1-s2.0-S1438422114000927 (accessed on 2 September 2020).
- Belkin, S.; Colwell, R.R. Oceans and Health: Pathogens in the Marine Environment; Springer Science + Business Media: New York, NY, USA, 2005. [Google Scholar]
- Jacobsen, B.P. (Ed.) State of the Tourism Industry in the Baltic Sea Region, 2018 ed. Available online: https://bstc.eu/fileadmin/bstc.eu/Downloads/Final_Report_Tourism_Industry_in_BSR_2018.pdf (accessed on 2 September 2020).
- European Union. The 2018 Annual Economic Report on EU Blue Economy. Available online: https://op.europa.eu/en/publication-detail/-/publication/79299d10-8a35-11e8-ac6a-01aa75ed71a1 (accessed on 2 September 2020).
- Baker-Austin, C.; Oliver, J.D.; Alam, M.; Ali, A.; Waldor, M.K.; Qadri, F.; Martinez-Urtaza, J. Vibrio spp. infections. Nat. Rev. Dis. Prim. 2018, 4. [Google Scholar] [CrossRef]
- Greenfield, D.I.; Gooch Moore, J.; Stewart, J.R.; Hilborn, E.D.; George, B.J.; Li, Q.; Dickerson, J.; Keppler, C.K.; Sandifer, P.A. Temporal and environmental factors driving Vibrio vulnificus and V. parahaemolyticus populations and their associations with harmful algal blooms in South Carolina detention ponds and receiving tidal creeks. GeoHealth 2017, 1, 306–317. [Google Scholar] [CrossRef]
- Olsen, H. Vibrio parahaemolyticus isolated from discharge from the ear in two patients exposed to sea water. Acta Pathol. Microbiol. Scand. Sect. B Microbiol. 1978, 86, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Gyraite, G.; Katarzyte, M.; Schernewski, G. First findings of potentially human pathogenic bacteria Vibrio in the south-eastern Baltic Sea coastal and transitional bathing waters. Mar. Pollut. Bull. 2019, 149, 110546. [Google Scholar] [CrossRef] [PubMed]
- FAO/WHO. Risk Assessment of Vibrio Vulnificus in Raw Oysters; FAO: Rome, Italy; WHO: Geneva, Switzerland, 2005; Volume 119, p. 73. [Google Scholar]
- Haas, C.N.; Rose, J.B.; Gerba, C.P. Quantitative Microbial Risk Assessment; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; Volume 9781118145296, ISBN 9781118910030. [Google Scholar]
- Federigi, I.; Bonadonna, L.; Bonanno Ferraro, G.; Briancesco, R.; Cioni, L.; Coccia, A.M.; Della Libera, S.; Ferretti, E.; Gramaccioni, L.; Iaconelli, M.; et al. Quantitative Microbial Risk Assessment as support for bathing waters profiling. Mar. Pollut. Bull. 2020, 157, 111318. [Google Scholar] [CrossRef] [PubMed]
- Kownacka, J.; Busch, S.; Göbel, J.; Gromisz, S.; Hällfors, H.; Höglander, H.; Huseby, S.; Jaanus, A.; Jakobsen, H.H.; Johansen, M.; et al. Cyanobacteria biomass, 1990–2018. HELCOM Baltic Sea Environment Fact Sheets. Available online: https://helcom.fi/baltic-sea-trends/environment-fact-sheets/eutrophication/cyanobacteria-biomass/ (accessed on 2 September 2020).
- Backer, L.C.; Manassaram-Baptiste, D.; LePrell, R.; Bolton, B. Cyanobacteria and algae blooms: Review of health and environmental data from the harmful algal bloom-related illness surveillance system (HABISS) 2007–2011. Toxins (Basel) 2015, 7, 1048–1064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eiler, A.; Johansson, M.; Bertilsson, S. Environmental influences on Vibrio populations in northern temperate and boreal coastal waters (Baltic and Skagerrak Seas). Appl. Environ. Microbiol. 2006, 72, 6004–6011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asplund, M.E.; Rehnstam-Holm, A.S.; Atnur, V.; Raghunath, P.; Saravanan, V.; Härnström, K.; Collin, B.; Karunasagar, I.; Godhe, A. Water column dynamics of Vibrio in relation to phytoplankton community composition and environmental conditions in a tropical coastal area. Environ. Microbiol. 2011, 13, 2738–2751. [Google Scholar] [CrossRef] [Green Version]
- Paldavičiene, A.; Mazur-Marzec, H.; Razinkovas, A. Toxic cyanobacteria blooms in the Lithuanian part of the Curonian Lagoon. Oceanologia 2009, 51, 203–216. [Google Scholar] [CrossRef] [Green Version]
- Kataržytė, M.; Mėžinė, J.; Vaičiūtė, D.; Liaugaudaitė, S.; Mukauskaitė, K.; Umgiesser, G.; Schernewski, G. Fecal contamination in shallow temperate estuarine lagoon: Source of the pollution and environmental factors. Mar. Pollut. Bull. 2018, 133, 762–772. [Google Scholar] [CrossRef]
- Kataržytė, M.; Vaičiūtė, D.; Nasvytis, P. Excellent bathing waters in coastal areas: Is microbial pollution the only important parameter? Ocean. Coast. Manag. 2019, 182, 104922. [Google Scholar] [CrossRef]
- Umgiesser, G.; Čerkasova, N.; Erturk, A.; Mėžinė, J.; Kataržytė, M. New beach in a shallow estuarine lagoon: A model-based E. coli pollution risk assessment. J. Coast. Conserv. 2018, 22, 573–586. [Google Scholar] [CrossRef]
- Schernewski, G.; Baltranaitė, E.; Kataržytė, M.; Balčiūnas, A.; Čerkasova, N.; Mėžinė, J. Establishing new bathing sites at the Curonian Lagoon coast: An ecological-social-economic assessment. J. Coast. Conserv. 2019, 23, 899–911. [Google Scholar] [CrossRef] [Green Version]
- Yáñez, M.A.; Valor, C.; Catalán, V. A simple and cost-effective method for the quantification of total coliforms and Escherichia coli in potable water. J. Microbiol. Methods 2006, 65, 608–611. [Google Scholar] [CrossRef] [PubMed]
- Fricker, C.R.; Bullock, S.; Murrin, K.; Niemela, S.I. Use of the ISO 9308-1 procedure for the detection of E. coli in water utilizing two incubation temperatures and two confirmation procedures and comparison with defined substrate technology. J. Water Health 2008, 6, 389–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haugland, R.A.; Siefring, S.; Lavender, J.; Varma, M. Influences of sample interference and interference controls on quantification of enterococci fecal indicator bacteria in surface water samples by the qPCR method. Water Res. 2012, 46, 5989–6001. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, H.; Muyzer, G. Denaturing gradient gel electrophoresis in marine microbial ecology. Methods Microbiol. 2001, 30, 425–468. [Google Scholar] [CrossRef]
- Fykse, E.M.; Skogan, G.; Davies, W.; Olsen, J.S.; Blatny, J.M. Detection of Vibrio cholerae by real-time nucleic acid sequence-based amplification. Appl. Environ. Microbiol. 2007, 73, 1457–1466. [Google Scholar] [CrossRef] [Green Version]
- Fykse, E.M.; Nilsen, T.; Nielsen, A.D.; Tryland, I.; Delacroix, S.; Blatny, J.M. Real-time PCR and NASBA for rapid and sensitive detection of Vibrio cholerae in ballast water. Mar. Pollut. Bull. 2012, 64, 200–206. [Google Scholar] [CrossRef]
- HELCOM. Guidelines for Monitoring Phytoplankton Species Composition, Abundance and Biomass. Available online: https://helcom.fi/media/publications/Guidelines-for-monitoring-phytoplankton-species-composition-abundance-and-biomass.pdf (accessed on 2 September 2020).
- Romero, J.; Navarrete, P. 16S rDNA-based analysis of dominant bacterial populations associated with early life stages of coho salmon (Oncorhynchus kisutch). Microb. Ecol. 2006, 51, 422–430. [Google Scholar] [CrossRef]
- Campbell, M.S.; Wright, A.C. Real-Time PCR Analysis of Vibrio vulnificus from Oysters. Appl. Environ. Microbial. 2003, 69, 7137–7144. [Google Scholar] [CrossRef] [Green Version]
- Wade, T.J.; Calderon, R.L.; Brenner, K.P.; Sams, E.; Beach, M.; Haugland, R.; Wymer, L.; Dufour, A.P. High Sensitivity of Children to Swimming-Associated Gastrointestinal Illness. Epidemiology 2008, 19, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Hassard, F.; Andrews, A.; Jones, D.L.; Parsons, L.; Jones, V.; Cox, B.A.; Daldorph, P.; Brett, H.; McDonald, J.E.; Malham, S.K. Physicochemical factors influence the abundance and culturability of human enteric pathogens and fecal indicator organisms in estuarine water and sediment. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Codd, G.A.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins: Risk management for health protection. Toxicol. Appl. Pharmacol. 2005, 203, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Churro, C.; Dias, E.; Valrio, E. Risk Assessment of Cyanobacteria and Cyanotoxins, the Particularities and Challenges of Planktothrix spp. Monitoring. In Novel Approaches and Their Applications in Risk Assessment; IntechOpen Limited: London, UK, 2012. [Google Scholar] [CrossRef] [Green Version]
- Hornick, R.; Music, S.; Wenzel, R.; Cash, R.; Libonati, J.P.; Snyder, M.; Woodward, T.E. The Broad Street Pump Revisited: Response of Volunteers to Ingested Cholera Vibrios. Symp. Cholera 1971, 47, 1181–1191. [Google Scholar]
- US EPA. Action Plan for Beaches and Recreational Waters. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi/30003NQI.PDF?Dockey=30003NQI.PDF (accessed on 2 September 2020).
- US EPA. Ambient Water Quality Criteria for Bacteria—1986. Available online: https://www.epa.gov/sites/production/files/2019-03/documents/ambient-wqc-bacteria-1986.pdf (accessed on 2 September 2020).
- Mouriño-Pérez, R.R.; Worden, A.Z.; Azam, F. Growth of Vibrio cholerae O1 in red tide waters off California. Appl. Environ. Microbiol. 2003, 69, 6923–6931. [Google Scholar] [CrossRef] [Green Version]
- Prato, S.; La Valle, P.; De Luca, E.; Lattanzi, L.; Migliore, G.; Morgana, J.G.; Munari, C.; Nicoletti, L.; Izzo, G.; Mistri, M. The “one-out, all-out” principle entails the risk of imposing unnecessary restoration costs: A study case in two Mediterranean coastal lakes. Mar. Pollut. Bull. 2014, 80, 30–40. [Google Scholar] [CrossRef]
- Addinsoft, XLSTAT Statistical and Data Analysis Solution. Available online: https://www.xlstat.com/en/ (accessed on 2 September 2020).
- Boehm, A.B.; Ashbolt, N.J.; Colford, J.M.; Dunbar, L.E.; Fleming, L.E.; Gold, M.A.; Hansel, J.A.; Hunter, P.R.; Ichida, A.M.; McGee, C.D.; et al. A sea change ahead for recreational water quality criteria. J. Water Health 2009, 7, 9–20. [Google Scholar] [CrossRef] [Green Version]
- Petkuviene, J.; Vaiciute, D.; Katarzyte, M.; Gecaite, I.; Rossato, G.; Vybernaite-Lubiene, I.; Bartoli, M. Feces from Piscivorous and Herbivorous Birds Stimulate Differentially Phytoplankton Growth. Water 2019, 11, 2567. [Google Scholar] [CrossRef] [Green Version]
- Buer, A.L.; Gyraite, G.; Wegener, P.; Lange, X.; Katarzyte, M.; Hauk, G.; Schernewski, G. Long term development of Bathing Water Quality at the German Baltic coast: Spatial patterns, problems and model simulations. Mar. Pollut. Bull. 2018, 135, 1055–1066. [Google Scholar] [CrossRef]
- Preece, E.P.; Hardy, F.J.; Moore, B.C.; Bryan, M. A review of microcystin detections in Estuarine and Marine waters: Environmental implications and human health risk. Harmful Algae 2017, 61, 31–45. [Google Scholar] [CrossRef] [Green Version]
- Giannuzzi, L.; Sedan, D.; Echenique, R.; Andrinolo, D. An acute case of intoxication with cyanobacteria and cyanotoxins in recreational water in Salto Grande Dam, Argentina. Mar. Drugs 2011, 9, 2164–2175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landsberg, J.H. The effects of harmful algal blooms on aquatic organisms. Rev. Fish. Sci. 2002, 10, 113–390. [Google Scholar] [CrossRef]
- Allen, S.A.; Bertilsson, S.; Goedkoop, W. Factors Determining Distribution and Abundance of Pathogenic Bacteria, with a Focus on Vibrio spp. Available online: https://www.ibg.uu.se/digitalAssets/90/a_90474-f_Allen-Anna-Sophie-arbete.pdf (accessed on 2 September 2020).
- Jutla, A.S.; Akanda, A.S.; Islam, S. Tracking cholera in coastal regions using satellite observations. J. Am. Water Resour. Assoc. 2010, 46, 651–662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jutla, A.; Akanda, A.S.; Huq, A.; Faruque, A.S.G.; Colwell, R.; Islam, S. A water Marker monitored by satellites to predict seasonal endemic cholera. Remote Sens. Lett. 2013, 4, 822–831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lobitz, B.; Beck, L.; Huq, A.; Wood, B.; Fuchs, G.; Faruque, A.S.G.; Colwell, R. Climate and infectious disease: Use of remote sensing for detection of Vibrio cholerae by indirect measurement. Proc. Natl. Acad. Sci. USA 2000, 97, 1438–1443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ULAC. Baltijos Jūroje Susidarė Palankios Sąlygos Daugintis Vibrio vulnificus Bakterijoms: Kyla Grėsmė Poilsiautojų Sveikatai. Available online: http://www.ulac.lt/naujienos/pranesimai-spaudai/baltijos-juroje-susidare-palankios-salygos-daugintis-vibrio-vulnificus-bakterijoms-kyla-gresme-poilsiautoju-sveikatai (accessed on 2 September 2020).
- Tamminen, M.; Karkman, A.; Corander, J.; Paulin, L.; Virta, M. Differences in bacterial community composition in Baltic Sea sediment in response to fish farming. Aquaculture 2011, 313, 15–23. [Google Scholar] [CrossRef]
- Ripabelli, G.; Sammarco, M.L.; Grasso, G.M.; Fanelli, I.; Caprioli, A.; Luzzi, I. Occurrence of Vibrio and other pathogenic bacteria in Mytilus galloprovincialis (mussels) harvested from Adriatic Sea, Italy. Int. J. Food Microbiol. 1999, 49, 43–48. [Google Scholar] [CrossRef]
- Romalde, J.L.; Diéguez, A.L.; Lasa, A.; Balboa, S. New Vibrio species associated to molluscan microbiota: A review. Front. Microbiol. 2014, 4, 1–11. [Google Scholar] [CrossRef]
- Halpern, M.; Izhaki, I. Fish as Hosts of Vibrio cholerae. Front. Microbiol. 2017, 8, 282. [Google Scholar] [CrossRef] [Green Version]
- Laviad -Shitrit, S.; Lev-Ari, T.; Katzir, G.; Sharaby, Y.; Izhaki, I.; Halpern, M. Great cormorants (Phalacrocorax carbo) as potential vectors for the dispersal of Vibrio cholerae. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Das, B.; Verma, J.; Kumar, P.; Ghosh, A.; Ramamurthy, T. Antibiotic resistance in Vibrio cholerae: Understanding the ecology of resistance genes and mechanisms. Vaccine 2020, 38, A83–A92. [Google Scholar] [CrossRef] [PubMed]
- Ibelings, B.W.; Backer, L.C.; Kardinaal, W.E.A.; Chorus, I. Current approaches to cyanotoxin risk assessment and risk management around the globe. Harmful Algae 2014, 40, 63–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brylinsky, M. Evaluation of Two Test Kits for Measurement of Microcystin Concentrations; Acadia University: Wolfville, NS, Canada, 2012; p. 19. Available online: https://19january2017snapshot.epa.gov/sites/production/files/2014-08/documents/microcystins-testkit-novascotia.pdf (accessed on 2 September 2020).
- FernÁndez-Delgado, M.; GarcÍa-Amado, M.A.; Contreras, M.; Incani, R.N.; Chirinos, H.; Rojas, H.; SuÁrez, P. Survival, Induction and resuscitation of vibrio cholerae from the viable but non-culturable state in the Southern Caribbean Sea. Rev. Inst. Med. Trop. Sao Paulo 2015, 57, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Mahmuda, S.; Morshed, M.G.; Bakht, H.B.; Khan, M.N.; Sack, R.B.; Sack, D.A. Role of cyanobacteria in the persistence of Vibrio cholerae O139 in saline microcosms. Can. J. Microbiol. 2004, 50, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Collin, B.; Rehnstam-Holm, A.S. Occurrence and potential pathogenesis of Vibrio cholerae, Vibrio parahaemolyticus and Vibrio vulnificus on the South Coast of Sweden. FEMS Microbiol. Ecol. 2011, 78, 306–313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wade, T.J.; Calderon, R.L.; Sams, E.; Beach, M.; Brenner, K.P.; Williams, A.H.; Dufour, A.P. Rapidly measured indicators of recreational water quality are predictive of swimming-associated gastrointestinal illness. Environ. Health Perspect. 2006, 114, 24–28. [Google Scholar] [CrossRef] [Green Version]
- Karunasagar, A.; Jalastagi, R.; Naik, A.; Rai, P. Detection of bacteria by 16S rRNA PCR and sequencing in culture-negative chronic rhinosinusitis. Laryngoscope 2018, 128, 2223–2225. [Google Scholar] [CrossRef]
- Liang, Z.; Keeley, A. Filtration recovery of extracellular DNA from environmental water samples. Environ. Sci. Technol. 2013, 47, 9324–9331. [Google Scholar] [CrossRef]
- Rudi, K.; Moen, B.; Drømtorp, S.M.; Holck, A.L. Use of ethidium monoazide and PCR in combination for quantification of viable and dead cells in complex samples. Appl. Environ. Microbiol. 2005, 71, 1018–1024. [Google Scholar] [CrossRef] [Green Version]
- Šyvokiene, J.; Mickeniene, L. Hydrocarbon-degrading bacteria associated with intestinal tract of fish from the baltic sea. J. Environ. Eng. Landsc. Manag. 2011, 19, 244–250. [Google Scholar] [CrossRef]
- Bock, T.; Christensen, N.; Eriksen, N.H.R.; Winter, S.; Rygaard, H.; Jorgensen, F. The first fatal case of Vibrio vulnificus infection in Denmark. APMIS 1994, 102, 874–876. [Google Scholar] [CrossRef] [PubMed]
- Reilly, G.D.; Reilly, C.A.; Smith, E.G.; Baker-Austin, C. Vibrio alginolyticus-associated wound infection acquired in British waters, Guernsey, July 2011. Eurosurveillance 2011, 16, 19994. [Google Scholar] [PubMed]
- Schets, F.M.; van den Berg, H.H.J.L.; Marchese, A.; Garbom, S.; de Roda Husman, A.M. Potentially human pathogenic vibrios in marine and fresh bathing waters related to environmental conditions and disease outcome. Int. J. Hyg. Environ. Health 2011, 214, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Shaw, K.S.; Sapkota, A.R.; Jacobs, J.M.; He, X.; Crump, B.C. Recreational swimmers’ exposure to Vibrio vulnificus and Vibrio parahaemolyticus in the Chesapeake Bay, Maryland, USA. Environ. Int. 2015, 74, 99–105. [Google Scholar] [CrossRef]
- Semenza, J.C.; Trinanes, J.; Lohr, W.; Sudre, B.; Löfdahl, M.; Martinez-Urtaza, J.; Nichols, G.L.; Rocklöv, J. Environmental suitability of vibrio infections in a warming climate: An early warning system. Environ. Health Perspect. 2017, 125, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Efstratiou, M.A. Managing Coastal Bathing Water Quality: The Contribution of Microbiology and Epidemiology. Mar. Pollut. Bull. 2001, 42, 424–431. [Google Scholar] [CrossRef]

| Target Organism | Primer Sequences | Base Pairs (Bp) | Tm (°C) | Reference |
|---|---|---|---|---|
| Conventional PCR | ||||
| Bacterial 16S rRNA | 341F: 5′-GCCTACGGGAGGCAGCAG-3′ | 630 | 59 | Romero and Navarrete [52] |
| 907R: 5′-CCGTCAATTCMTTTGAGTTT-3′ | ||||
| Real-time PCR | ||||
| V. vulnificus | vvha_F: 5′-GTTTATGGTGAGAACGGTGACA-3′ | - | 60 | Campbell et al. [53] |
| vvha_R: 5′-TTCTTTATCTAGGCCCCAAACTTG-3′ | ||||
| vvha_P: (FAM)-CCGTTAACCGAACCACCCGCAA-(TAMRA) | ||||
| V. cholerae | groEL_F: 5′-GGTTATCGCTGCGGTAGAAG-3′ | 117 | 58 | Fykse et al. [50] |
| groEL_R: 5′-ATGATGTTGCCCACGCTAGA-3′ | ||||
| groEL_P: (FAM)-CTGTCTGTACCTTGTGCCGATACTAAAGC-BBQ) | ||||
| Enterococcus | ENT_F: 5′-GAGAAATTCCAAACGAACTTG-’3 | 92 | 60 | Haugland et al. [47] |
| ENT_R: 5′-CAGTGCTCTACCTCCATCATT-’3 | ||||
| ENT_P: (FAM)-GGTTCTCTCCGAAATAGCTTTAGGGCTA-(TAMRA) | ||||
| Bacteroides | HF183_F: 5′-ATCATGAGTTCACATGTCCG-’3 | 167 | 60 | Haugland et al. [16] |
| HF183_R: 5′-CGTAGGAGTTTGGACCGTGT-’3 | ||||
| Probe: (FAM)-CTGAGAGGAAGGTCCCCCACATTGGA-(MGB) | ||||
| Indicator | Unit | Risk Level | Reference | ||
|---|---|---|---|---|---|
| FIB | Low | Moderate | High | ||
| Guidance level | 0–3% probability of gastrointestinal illness | 3–5% probability of gastrointestinal illness | 5–8.4% probability of gastrointestinal illness | ||
| E. coli | CFU 100 mL−1 | <250 | 250–500 | >500 | BWD [7] |
| Enterococcus | GC 100 mL−1 | <100 | 101–200 | >200 | |
| Bacteroides | GC 100 mL−1 | <860 | >860 | Ashbolt et al. [17] | |
| QMRA | Acceptable benchmark | Not acceptable | |||
| V. vulnificus | Probability (Infection) (%) | <19 infections per 1000 bathers | >19 | U.S. EPA [60] | |
| V. cholerae | Minimum infectious dose | <106 GC 100 mL−1 | >106 cells 100 mL−1 | Mouriño-Pérez et al. [61] | |
| cHABs | Low | Moderate | High | ||
| Guidance level | Relatively low probability of adverse health effects | Moderate probability of adverse health effects | High probability of adverse health effects | ||
| Cyanobacteria | Cells mL−1 | <20,000 | 20,000–100,000 | >100,000 | Churro et al. [57] WHO [1] |
| Chl a µg L−1 | <10 | 10–50 | >50 | ||
| Biomass mm3 L−1 | <0.2 | 0.2–10 | >10 | ||
| MC | µg L−1 | 2–4 | 10–20 | >1000 | Codd et al. [56] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gyraite, G.; Kataržytė, M.; Overlingė, D.; Vaičiūtė, D.; Jonikaitė, E.; Schernewski, G. Skip the Dip—Avoid the Risk? Integrated Microbiological Water Quality Assessment in the South-Eastern Baltic Sea Coastal Waters. Water 2020, 12, 3146. https://doi.org/10.3390/w12113146
Gyraite G, Kataržytė M, Overlingė D, Vaičiūtė D, Jonikaitė E, Schernewski G. Skip the Dip—Avoid the Risk? Integrated Microbiological Water Quality Assessment in the South-Eastern Baltic Sea Coastal Waters. Water. 2020; 12(11):3146. https://doi.org/10.3390/w12113146
Chicago/Turabian StyleGyraite, Greta, Marija Kataržytė, Donata Overlingė, Diana Vaičiūtė, Eglė Jonikaitė, and Gerald Schernewski. 2020. "Skip the Dip—Avoid the Risk? Integrated Microbiological Water Quality Assessment in the South-Eastern Baltic Sea Coastal Waters" Water 12, no. 11: 3146. https://doi.org/10.3390/w12113146






