Temperature Stability and Effectiveness of Plasma-Activated Liquids over an 18 Months Period
Abstract
1. Introduction
2. Materials and Methods
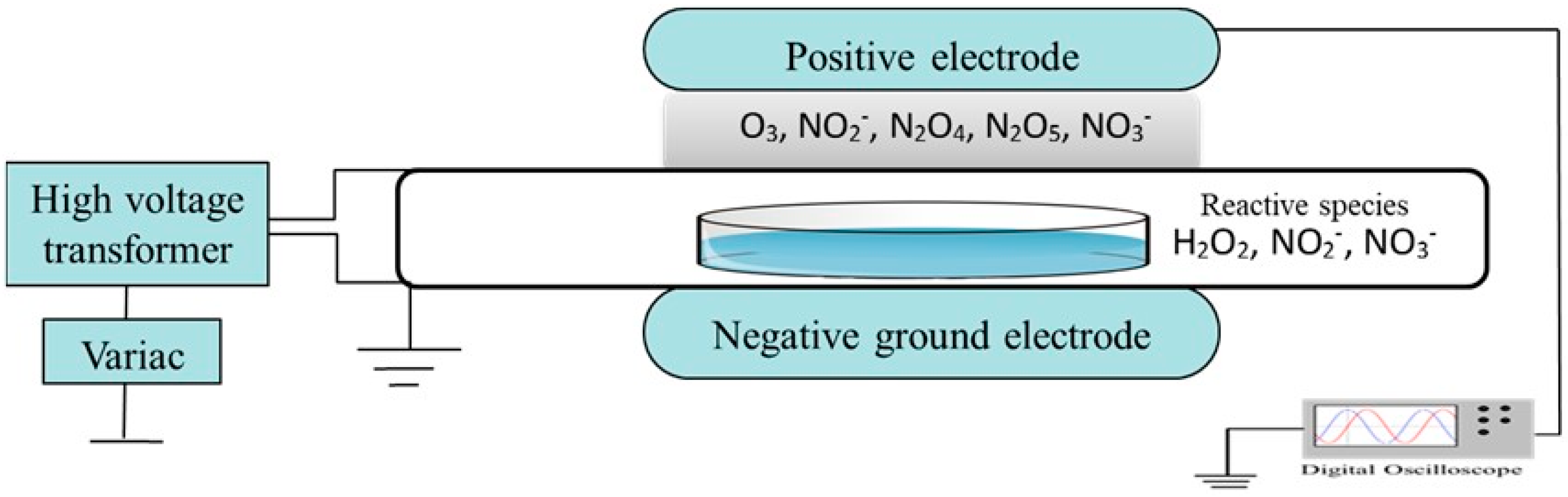
2.1. Plasma Source and Sample Treatment
2.2. Antimicrobial Efficacy
2.2.1. Bacterial Strains and Growth Conditions
2.2.2. Preparation of Cell Suspensions
2.2.3. Microbiological Analysis
2.3. Investigation of the Chemical Composition of PAL
2.3.1. Determination of Hydrogen Peroxide and Reactive Oxygen Species (ROS)
2.3.2. Determination of Nitrite and Nitrate
2.3.3. pH and Temperature Measurements
2.4. Data Analysis
3. Results
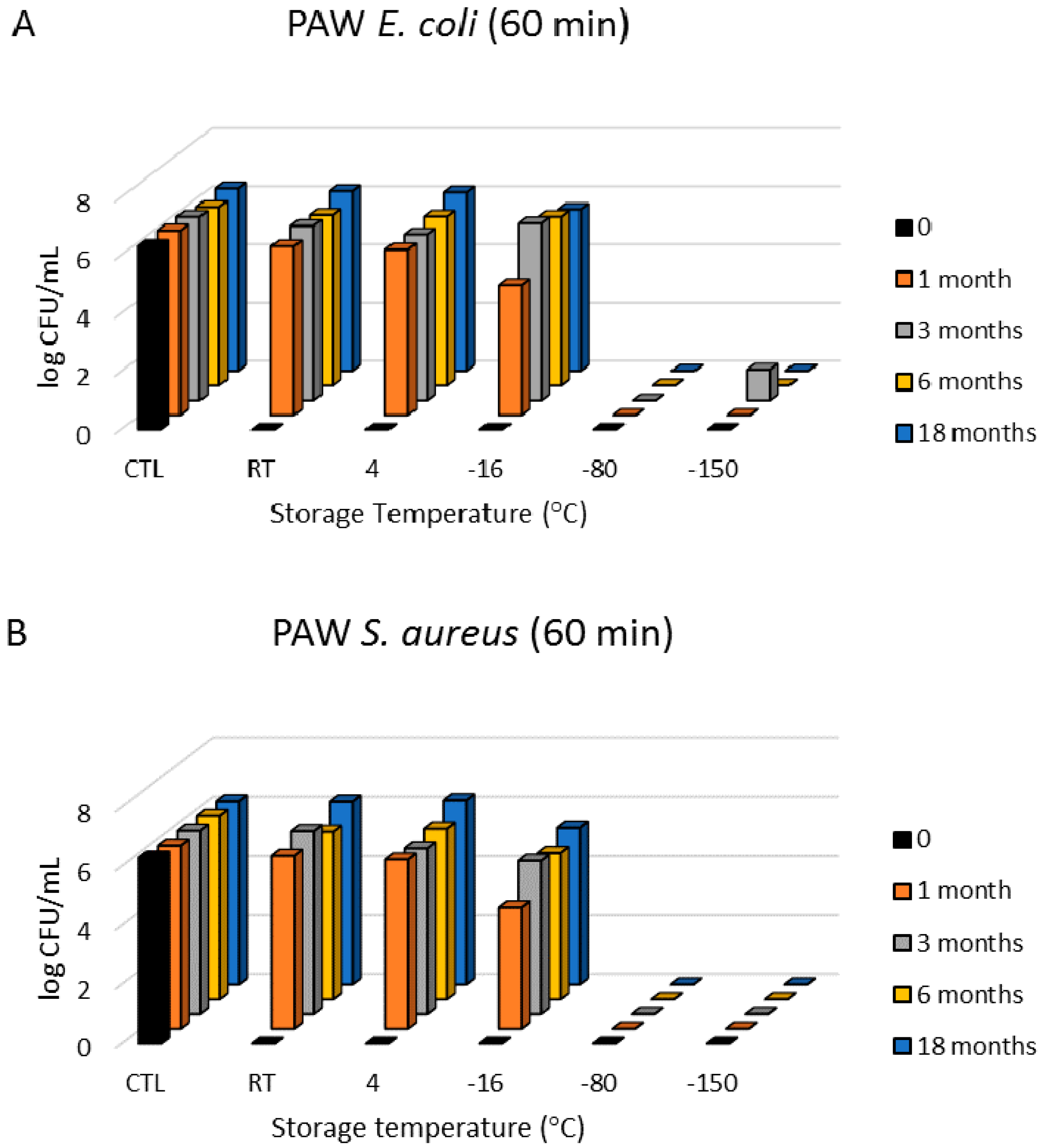
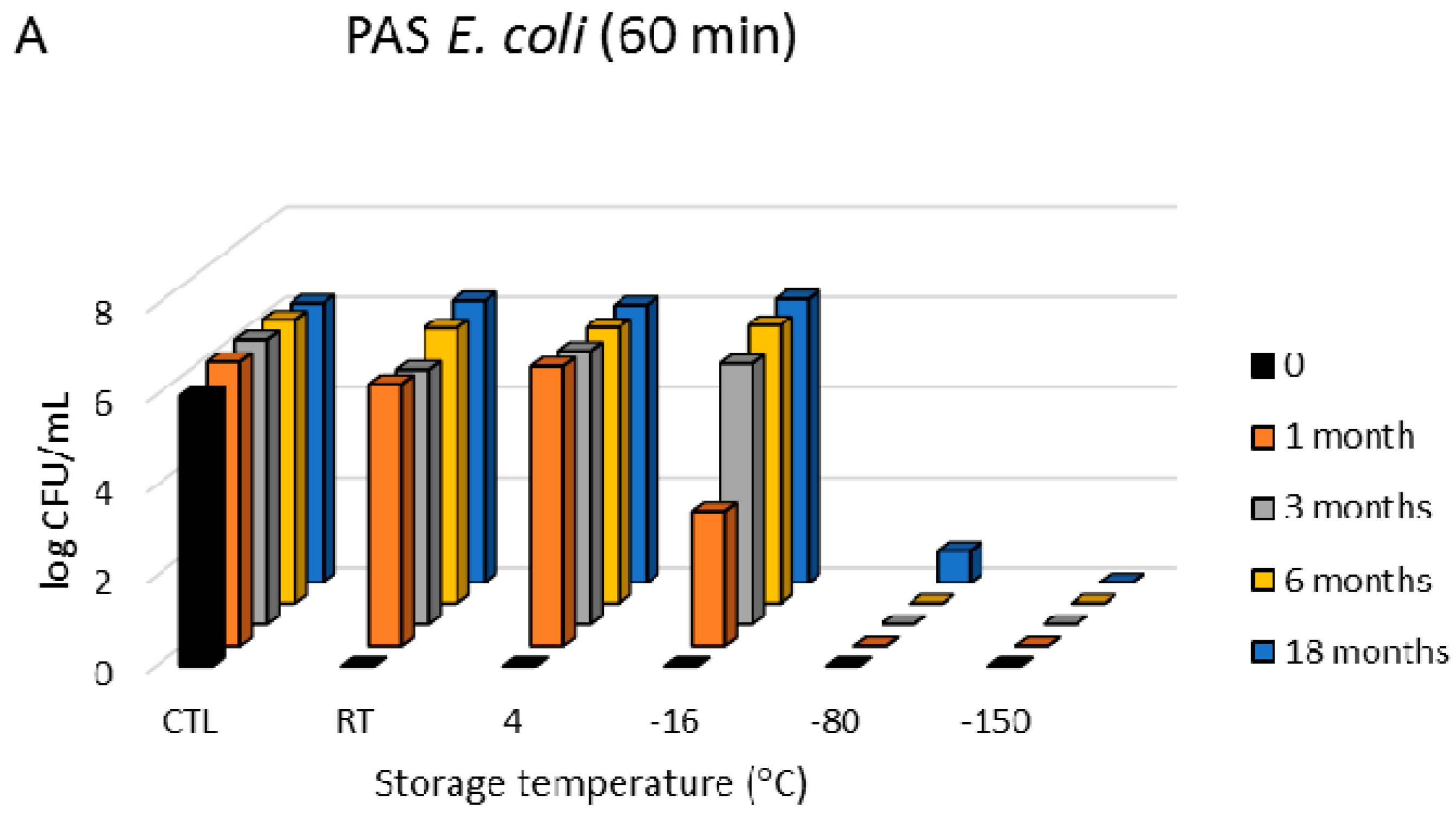
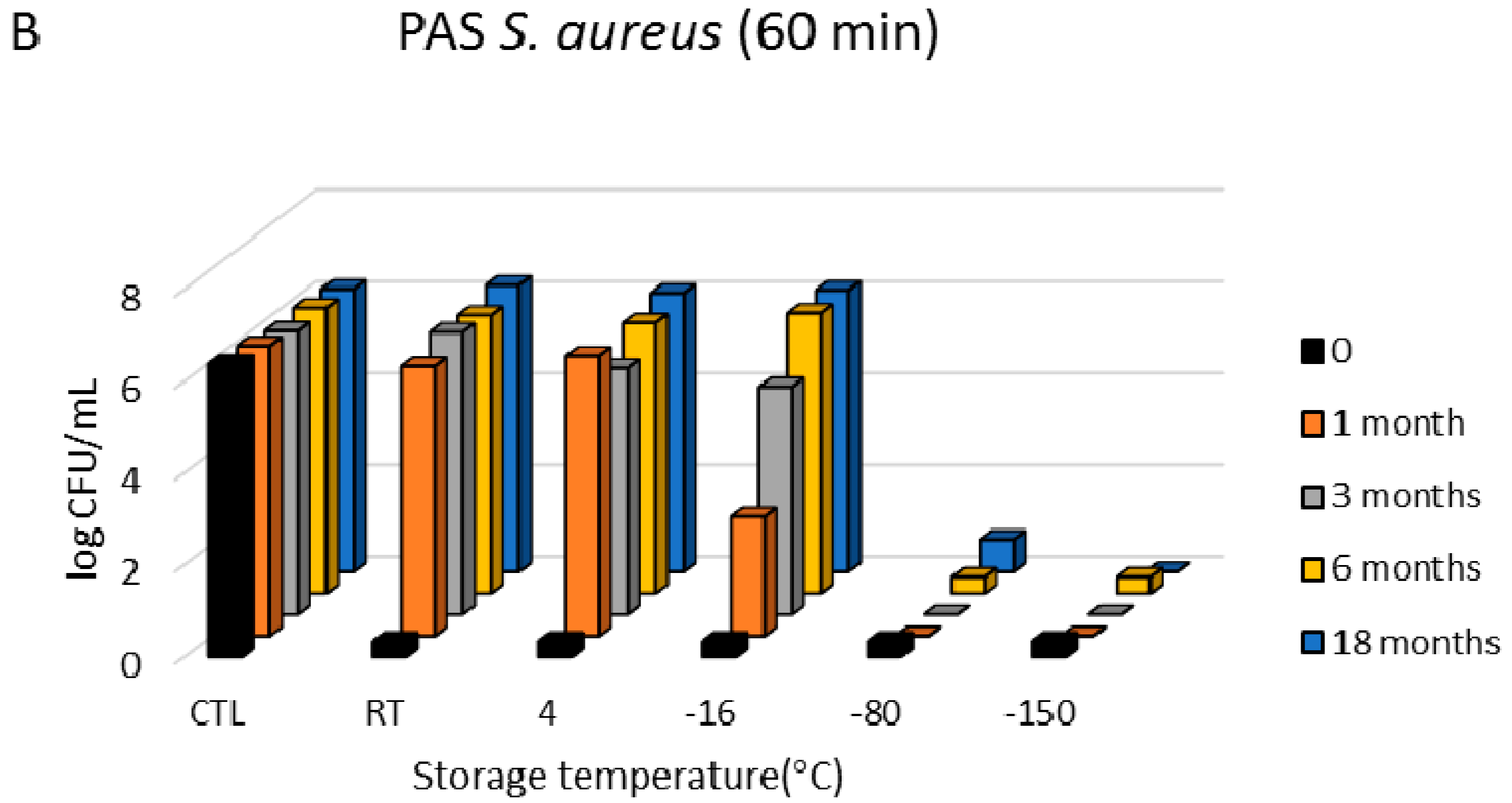
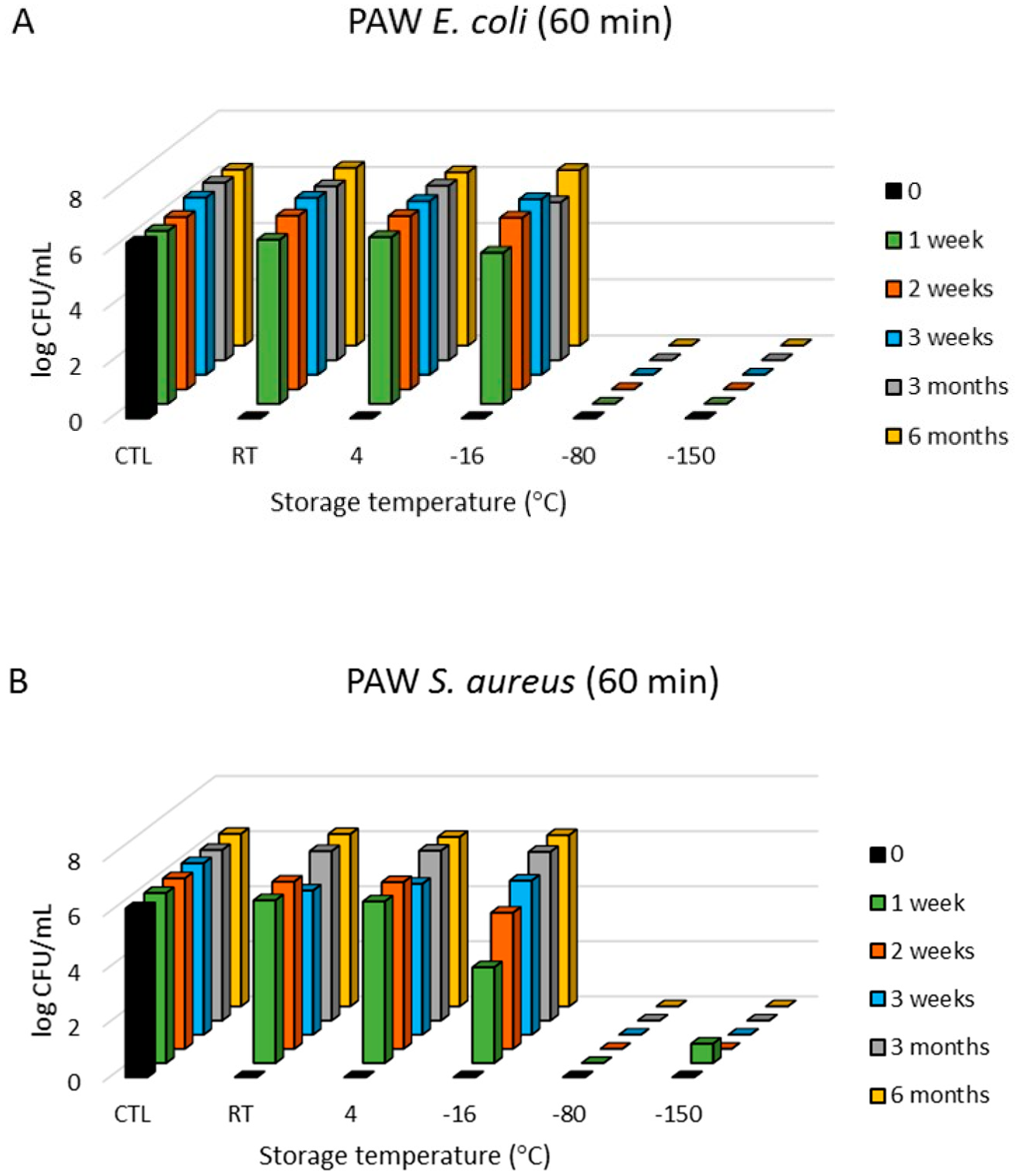
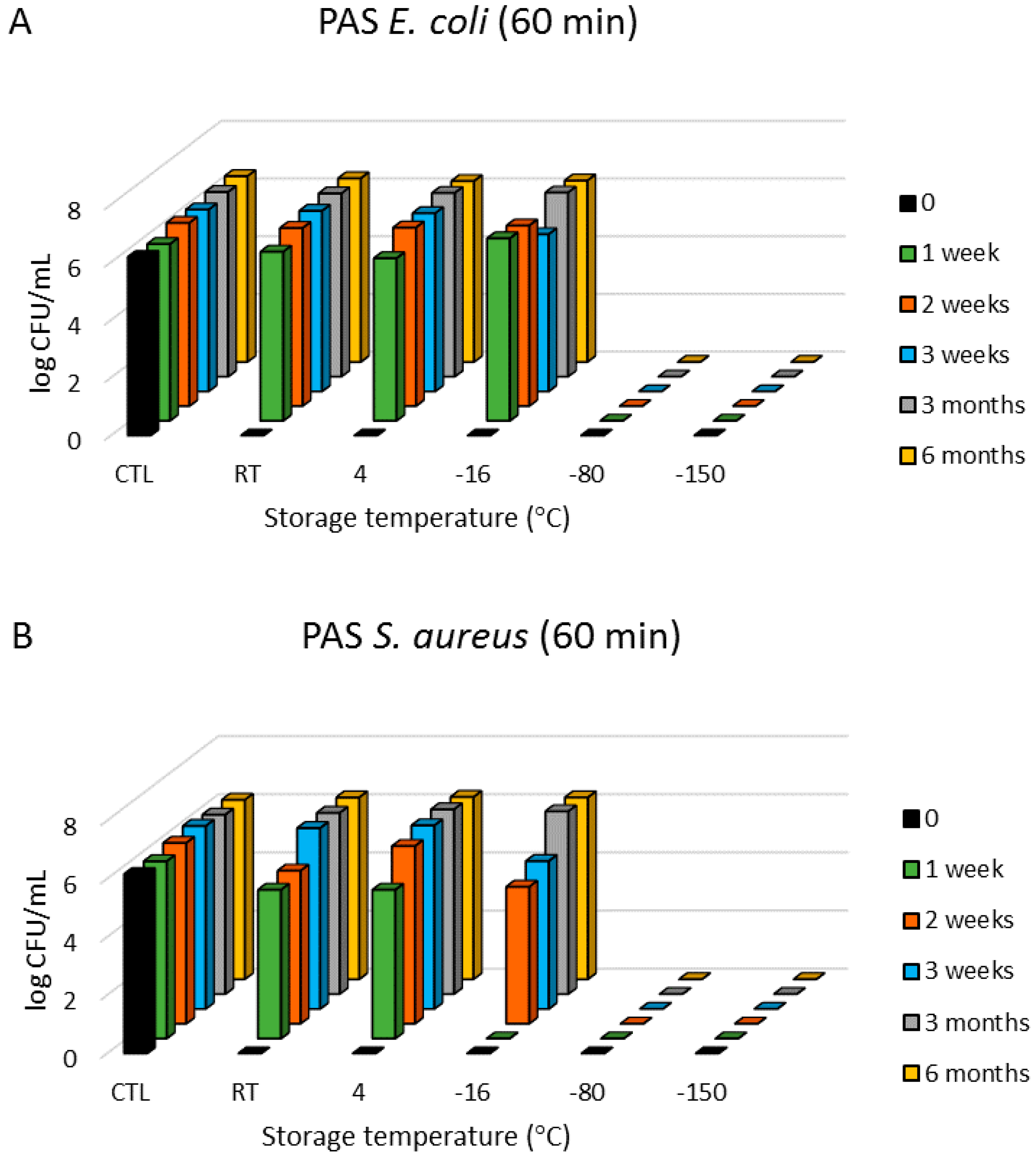
3.1. Antimicrobial Effects
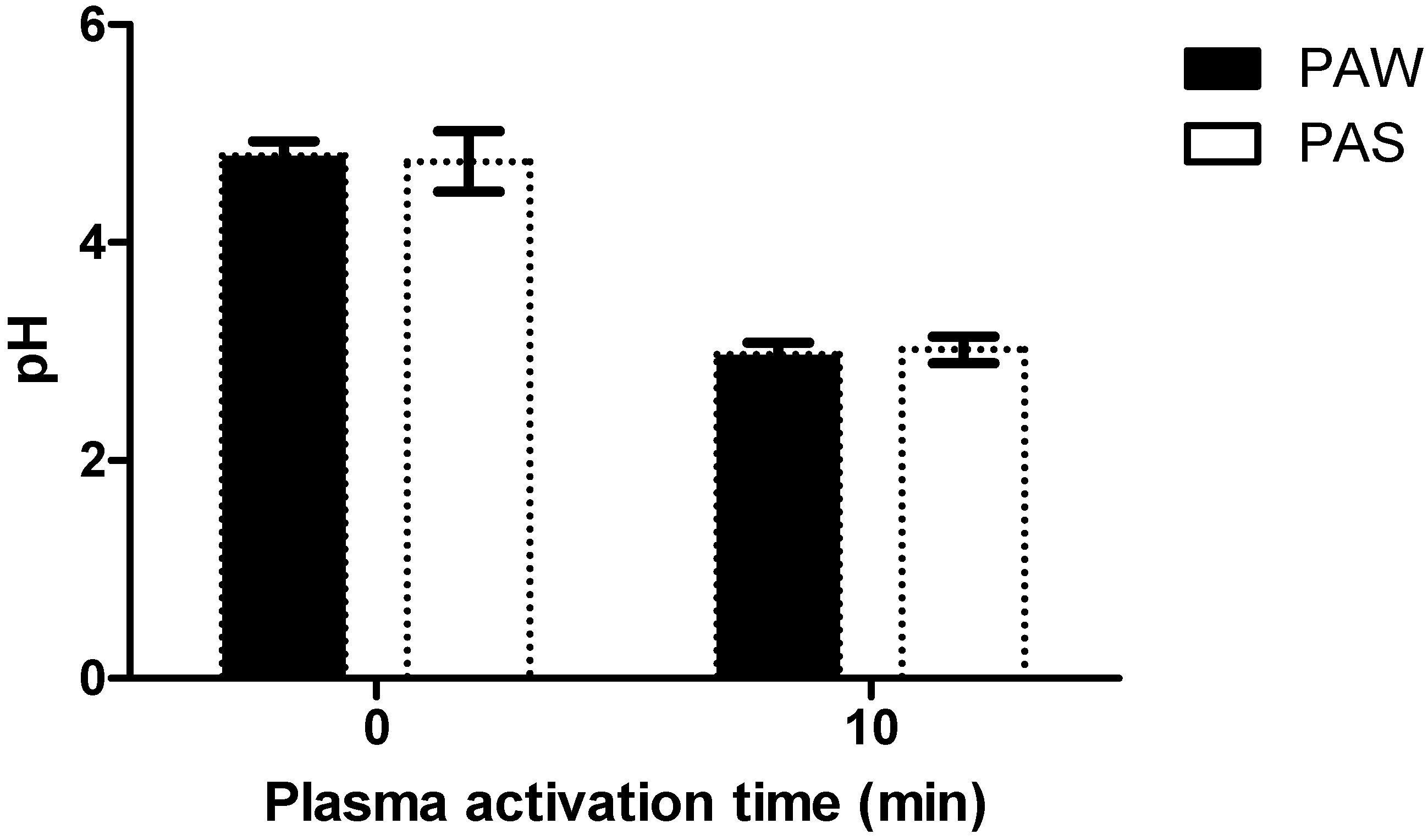
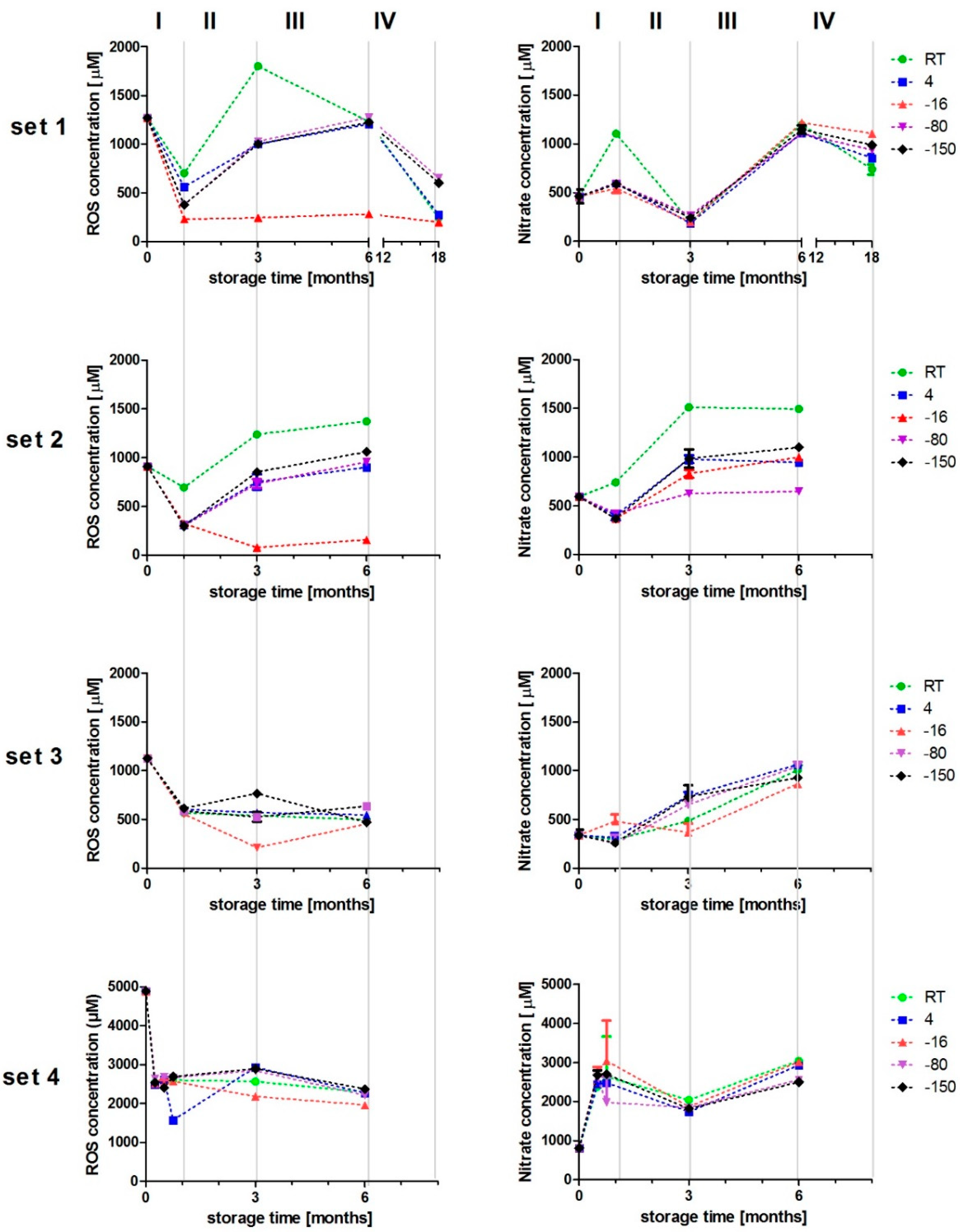
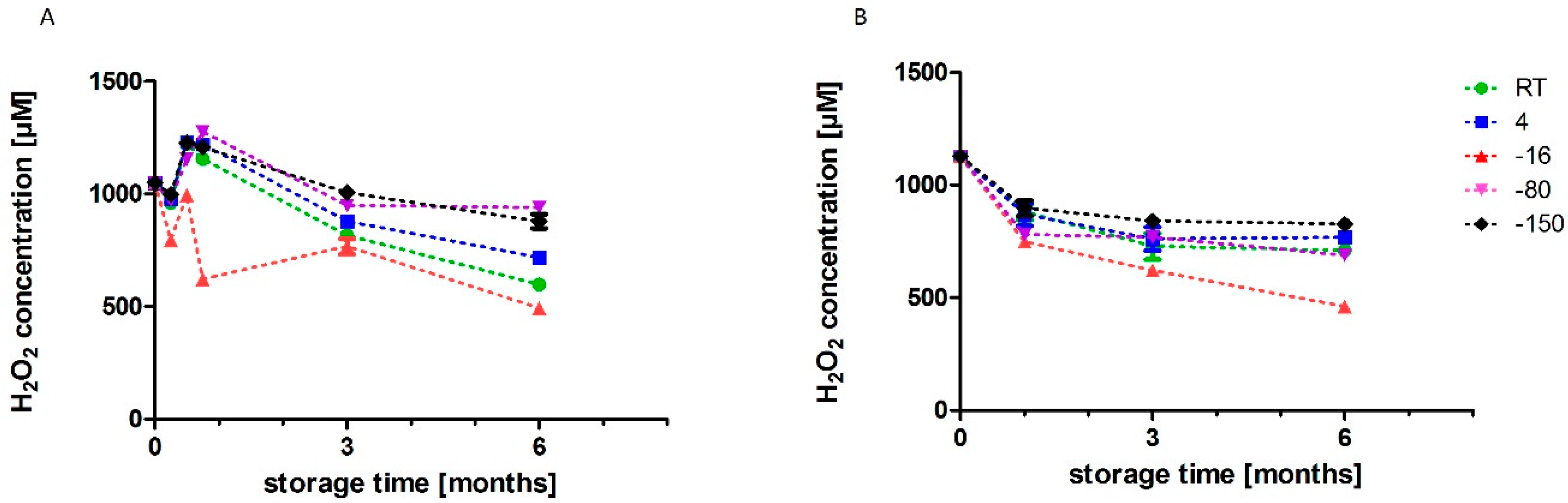
3.2. Chemical Characterisation of PAL
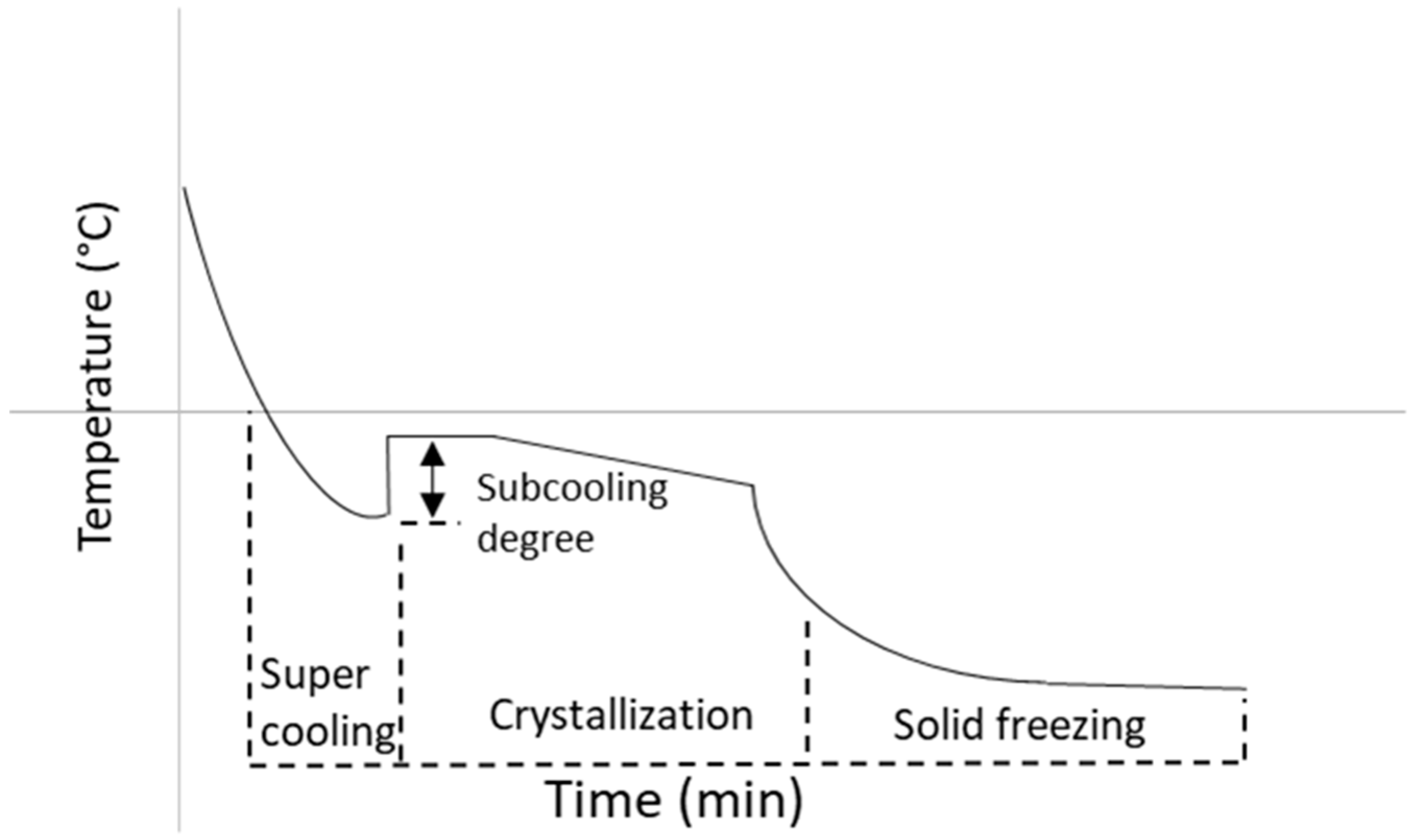
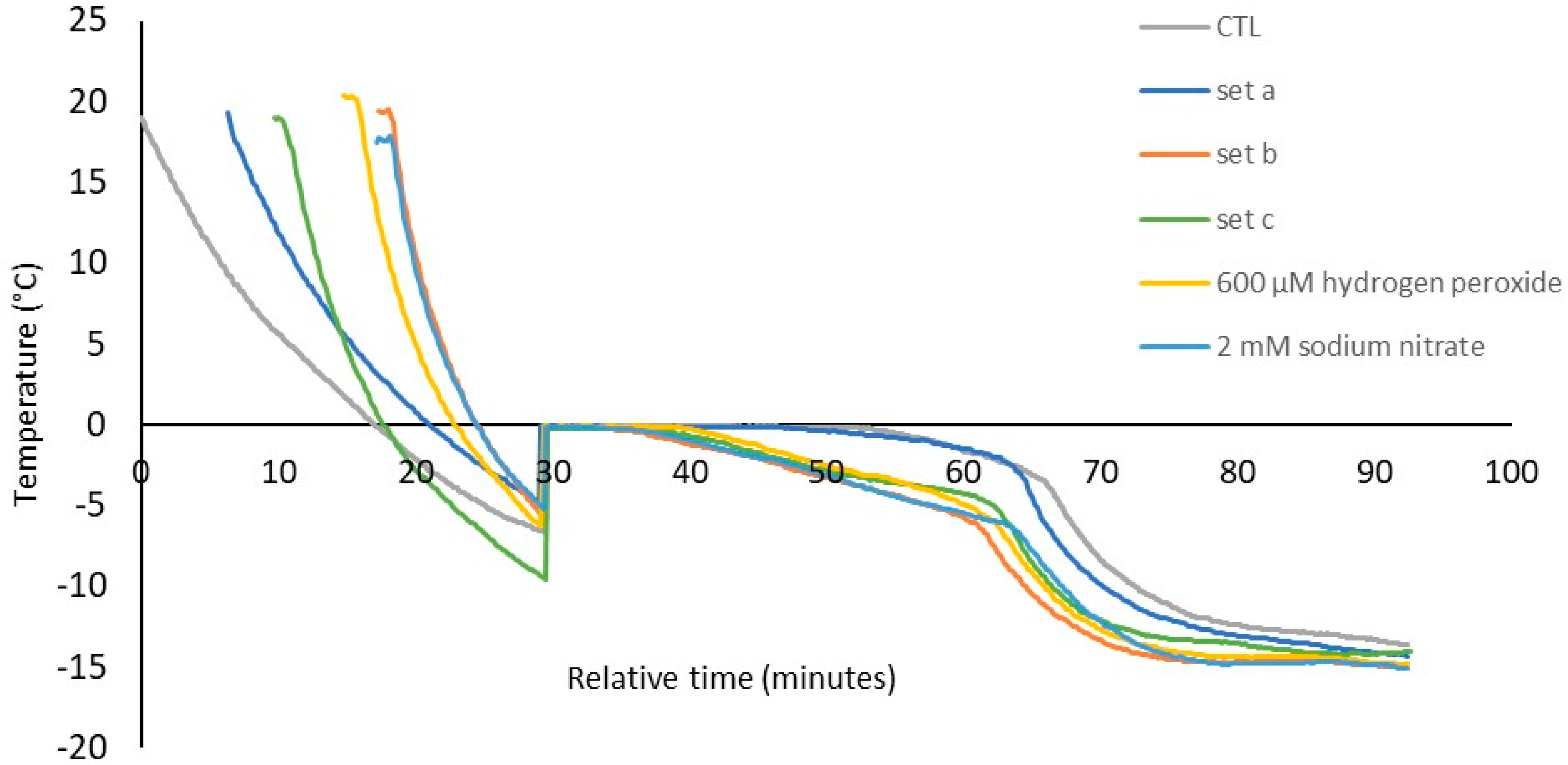
3.3. Freezing Behaviour
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kampf, G. Acquired resistance to chlorhexidine—Is it time to establish an “antiseptic stewardship” initiative? J. Hosp. Infect. 2016, 94, 213–227. [Google Scholar] [CrossRef] [PubMed]
- Verraes, C.; Van Boxstael, S.; Van Meervenne, E.; Van Coillie, E.; Butaye, P.; Catry, B.; de Schaetzen, M.-A.; Van Huffel, X.; Imberechts, H.; Dierick, K.; et al. Antimicrobial resistance in the food chain: A review. Int. J. Environ. Res. Public Health 2013, 10, 2643–2669. [Google Scholar] [CrossRef] [PubMed]
- Wand, M.E.; Bock, L.J.; Bonney, L.C.; Sutton, J.M. Mechanisms of Increased Resistance to Chlorhexidine and Cross-Resistance to Colistin following Exposure of Klebsiella pneumoniae Clinical Isolates to Chlorhexidine. Antimicrob. Agents Chemother. 2016, 61, e01162-16. [Google Scholar] [CrossRef] [PubMed]
- Carey, D.E.; McNamara, P.J. The impact of triclosan on the spread of antibiotic resistance in the environment. Front. Microbiol. 2015, 5, 780. [Google Scholar] [CrossRef] [PubMed]
- Buffet-Bataillon, S.; Tattevin, P.; Bonnaure-Mallet, M.; Jolivet-Gougeon, A. Emergence of resistance to antibacterial agents: The role of quaternary ammonium compounds—A critical review. Int. J. Antimicrob. Agents 2012, 39, 381–389. [Google Scholar] [CrossRef]
- Lu, P.; Boehm, D.; Cullen, P.J.; Bourke, P. Controlled cytotoxicity of plasma treated water formulated by open-air hybrid mode discharge. Appl. Phys. Lett. 2017, 110, 264102. [Google Scholar] [CrossRef]
- Zhou, R.; Zhou, R.; Yu, F.; Xi, D.; Wang, P.; Li, J.; Wang, X.; Zhang, X.; Bazaka, K.; Ostrikov, K. Removal of organophosphorus pesticide residues from Lycium barbarum by gas phase surface discharge plasma. Chem. Eng. J. 2018, 342, 401–409. [Google Scholar] [CrossRef]
- Sarangapani, C.; Ziuzina, D.; Behan, P.; Boehm, D.; Gilmore, B.F.; Cullen, P.J.; Bourke, P. Degradation kinetics of cold plasma-treated antibiotics and their antimicrobial activity. Sci. Rep. 2019, 9, 3955. [Google Scholar] [CrossRef]
- Schnabel, U.; Handorf, O.; Stachowiak, J.; Boehm, D.; Weit, C.; Weihe, T.; Schäfer, J.; Below, H.; Bourke, P.; Ehlbeck, J. Plasma-Functionalized Water: From Bench to Prototype for Fresh-Cut Lettuce. Food Eng. Rev. 2020, 1–21. [Google Scholar] [CrossRef]
- Lin, C.-M.; Chu, Y.-C.; Hsiao, C.-P.; Wu, J.S.; Hsieh, C.-W.; Hou, C.-Y. The Optimization of Plasma-Activated Water Treatments to Inactivate Salmonella Enteritidis (ATCC 13076) on Shell Eggs. Foods 2019, 8, 520. [Google Scholar] [CrossRef]
- Los, A.; Ziuzina, D.; Boehm, D.; Cullen, P.J.; Bourke, P. A Comparison of Inactivation Efficacy and Mechanisms of Gas Plasma and Plasma Activated Water against Aspergillus flavus Spores and Biofilms. Appl. Environ. Microbiol. 2020, 86, e02619-19. [Google Scholar] [CrossRef]
- Smet, C.; Govaert, M.; Kyrylenko, A.; Easdani, M.; Walsh, J.; Van Impe, J. Inactivation of Single Strains of Listeria monocytogenes and Salmonella Typhimurium Planktonic Cells Biofilms With Plasma Activated Liquids. Front. Microbiol. 2019, 10, 1539. [Google Scholar] [CrossRef] [PubMed]
- Filipić, A.; Gutierrez-Aguirre, I.; Primc, G.; Mozetič, M.; Dobnik, D. Cold Plasma, a New Hope in the Field of Virus Inactivation. Trends Biotechnol. 2020, 38, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Perinban, S.; Orsat, V.; Raghavan, V. Nonthermal Plasma–Liquid Interactions in Food Processing: A Review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1985–2008. [Google Scholar] [CrossRef]
- Thirumdas, R.; Kothakota, A.; Annapure, U.; Siliveru, K.; Blundell, R.; Gatt, R.; Valdramidis, V.P. Plasma activated water (PAW): Chemistry, physico-chemical properties, applications in food and agriculture. Trends Food Sci. Technol. 2018, 77, 21–31. [Google Scholar] [CrossRef]
- National Research, C. Drinking Water and Health. In Disinfectants and Disinfectant By-Products; The National Academies Press: Washington, DC, USA, 1987; Volume 7. [Google Scholar]
- Ng, S.W.; Tsoukou, E.; Chaple, S.; Boehm, D.; Bourke, P. Plasma Treatment of Liquids. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Ikawa, S.; Tani, A.; Nakashima, Y.; Kitano, K. Physicochemical properties of bactericidal plasma-treated water. J. Phys. D Appl. Phys. 2016, 49, 425401. [Google Scholar] [CrossRef]
- Lukes, P.; Dolezalova, E.; Sisrova, I.; Clupek, M. Aqueous-phase chemistry and bactericidal effects from an air discharge plasma in contact with water: Evidence for the formation of peroxynitrite through a pseudo-second-order post-discharge reaction of H2O2 and HNO2. Plasma Sources Sci. Technol. 2014, 23, 015019. [Google Scholar] [CrossRef]
- Boehm, D.; Heslin, C.; Cullen, P.J.; Bourke, P. Cytotoxic and mutagenic potential of solutions exposed to cold atmospheric plasma. Sci. Rep. 2016, 6, 21464. [Google Scholar] [CrossRef]
- Vlad, I.-E.; Anghel, S.D. Time stability of water activated by different on-liquid atmospheric pressure plasmas. J. Electrost. 2017, 87, 284–292. [Google Scholar] [CrossRef]
- Traylor, M.J.; Pavlovich, M.; Karim, S.; Hait, P.; Sakiyama, Y.; Clark, D.S.; Graves, D. Long-term antibacterial efficacy of air plasma-activated water. J. Phys. D Appl. Phys. 2011, 44, 472001. [Google Scholar] [CrossRef]
- Shen, J.; Tian, Y.; Li, Y.; Ma, R.; Zhang, Q.; Zhang, J.; Fang, J. Bactericidal Effects against S. aureus and Physicochemical Properties of Plasma Activated Water stored at different temperatures. Sci. Rep. 2016, 6, 28505. [Google Scholar] [CrossRef]
- Gelinas, P.; Goulet, J.; Tastayre, G.M.; Picard, G.A. Effect of Temperature and Contact Time on the Activity of Eight Disinfectants—A Classification. J. Food Prot. 1984, 47, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Tsoukou, E.; Bourke, P.; Boehm, D. Understanding the differences between antimicrobial and cytotoxic properties of plasma activated liquids. Plasma Med. 2018, 8, 299–320. [Google Scholar] [CrossRef]
- Moiseev, T.; Misra, N.N.; Patil, S.; Cullen, P.J.; Bourke, P.; Keener, K.; Mosnier, J.P. Post-discharge gas composition of a large-gap DBD in humid air by UV-Vis absorption spectroscopy. Plasma Sources Sci. Technol. 2014, 23, 065033. [Google Scholar] [CrossRef]
- Patil, S.; Moiseev, T.; Misra, N.N.; Cullen, P.J.; Mosnier, J.P.; Keener, K.; Bourke, P. Influence of high voltage atmospheric cold plasma process parameters and role of relative humidity on inactivation of Bacillus atrophaeus spores inside a sealed package. J. Hosp. Infect. 2014, 88, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Milosavljevic, V.; Cullen, P.J. Spectroscopic investigation of a Dielectric Barrier Discharge in Modified Atmosphere Packaging. Eur. Phys. J. Appl. Phys. 2017, 80, 20801. [Google Scholar] [CrossRef]
- Ziuzina, D.; Patil, S.; Cullen, P.J.; Keener, K.; Bourke, P. Atmospheric Cold Plasma Inactivation of Escherichia coli in Liquid Media inside a Sealed Package. J. Appl. Microbiol. 2012, 114, 778–787. [Google Scholar] [CrossRef]
- Pan, G.Y.; Chen, C.L.; Chang, H.M.; Gratzl, J.S. Studies on Ozone Bleaching. I. The Effect of PH, Temperature, Buffer Systems and Heavy Metal-Ions on Stability of Ozone in Aqueous Solution. J. Wood Chem. Technol. 1984, 4, 367–387. [Google Scholar] [CrossRef]
- Jablonowski, H.; Sousa, J.; Weltmann, K.-D.; Wende, K.; Reuter, S. Quantification of the ozone and singlet delta oxygen produced in gas and liquid phases by a non-thermal atmospheric plasma with relevance for medical treatment. Sci. Rep. 2018, 8, 12195. [Google Scholar] [CrossRef]
- Han, L.; Patil, S.; Boehm, D.; Milosavljevic, V.; Cullen, P.J.; Bourke, P. Mechanism of Inactivation by High Voltage Atmospheric Cold Plasma Differs between Escherichia coli and Staphylococcus aureus. Appl. Environ. Microbiol. 2015, 82, 450–458. [Google Scholar] [CrossRef]
- Khadre, M.A.; Yousef, A.E.; Kim, J.G. Microbiological Aspects of Ozone Applications in Food: A Review. J. Food Sci. 2001, 66, 1242–1252. [Google Scholar] [CrossRef]
- Gorbanev, Y.; O’Connell, D.; Chechik, V. Non-Thermal Plasma in Contact with Water: The Origin of Species. Chemistry 2016, 22, 3496–3505. [Google Scholar] [CrossRef] [PubMed]
- Joshi, I.; Salvi, D.; Schaffner, D.W.; Karwe, M.V. Characterization of Microbial Inactivation Using Plasma-Activated Water and Plasma-Activated Acidified Buffer. J. Food Prot. 2018, 81, 1472–1480. [Google Scholar] [CrossRef] [PubMed]
- Figueira, F.; Doria, A.; Khouri, S.; Maciel, H.; Pessoa, R.; Antonio, R.-R. Effect of Storage Temperature on pH and Conductivity of Reverse Osmosis Water Treated with Atmospheric Plasma. Plasma Med. 2018, 8, 237–244. [Google Scholar] [CrossRef]
- Zhou, R.; Zhou, R.; Prasad, K.; Fang, Z.; Speight, R.; Bazaka, K.; Ostrikov, K. Cold atmospheric plasma activated water as a prospective disinfectant the crucial role of peroxynitrite. Green Chem. 2018, 20, 5276–5284. [Google Scholar] [CrossRef]
- Schairer, D.O.; Chouake, J.S.; Nosanchuk, J.D.; Friedman, A.J. The potential of nitric oxide releasing therapies as antimicrobial agents. Virulence 2012, 3, 271–279. [Google Scholar] [CrossRef]
- Lukes, P.; Locke, B.; Brisset, J.-L. Aqueous-Phase Chemistry of Electrical Discharge Plasma in Water and in Gas-Liquid Environments. In Plasma Chemistry and Catalysis in Gases and Liquids; Parvulescu, P.I., Magureanu, M., Lukes, P., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2012; Volume 7, pp. 243–308. [Google Scholar]
- Kissner, R.; Koppenol, W. Product Distribution of Peroxynitrite Decay as a Function of pH, Temperature, and Concentration. J. Am. Chem. Soc. 2002, 124, 234–239. [Google Scholar] [CrossRef]
- Jia, X.; Chen, Q.; Yang, Y.; Tang, Y.; Wang, R.; Xu, Y.; Zhu, W.; Qian, X. FRET-Based Mito-Specific Fluorescent Probe for Ratiometric Detection and Imaging of Endogenous Peroxynitrite: Dyad of Cy3 and Cy5. J. Am. Chem. Soc. 2016, 138, 10778–10781. [Google Scholar] [CrossRef]
- Huie, R.E.; Padmaja, S. The reaction of no with superoxide. Free Radic. Res. Commun. 1993, 18, 195–199. [Google Scholar] [CrossRef]
- Zhang, Q.; Ma, R.; Tian, Y.; Su, B.; Wang, K.; Yu, S.; Zhang, J.; Fang, J. Sterilization Efficiency of a Novel Electrochemical Disinfectant against Staphylococcus aureus. Environ. Sci. Technol. 2016, 50, 3184–3192. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, A.; Héroux, P.; Sand, W.; Sun, Z.; Zhan, J.; Cihao, W.; Hao, S.; Li, Z.; Li, Z.; et al. Effect of Dielectric Barrier Discharge Cold Plasma on Pea Seed Growth. J. Agric. Food Chem. 2019, 67, 10813–10822. [Google Scholar] [CrossRef] [PubMed]
- Judée, F.; Fongia, C.; Ducommun, B.; Yousfi, M.; Lobjois, V.; Merbahi, N. Short and long time effects of low temperature Plasma Activated Media on 3D multicellular tumor spheroids. Sci. Rep. 2016, 6, 21421. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsoukou, E.; Bourke, P.; Boehm, D. Temperature Stability and Effectiveness of Plasma-Activated Liquids over an 18 Months Period. Water 2020, 12, 3021. https://doi.org/10.3390/w12113021
Tsoukou E, Bourke P, Boehm D. Temperature Stability and Effectiveness of Plasma-Activated Liquids over an 18 Months Period. Water. 2020; 12(11):3021. https://doi.org/10.3390/w12113021
Chicago/Turabian StyleTsoukou, Evanthia, Paula Bourke, and Daniela Boehm. 2020. "Temperature Stability and Effectiveness of Plasma-Activated Liquids over an 18 Months Period" Water 12, no. 11: 3021. https://doi.org/10.3390/w12113021
APA StyleTsoukou, E., Bourke, P., & Boehm, D. (2020). Temperature Stability and Effectiveness of Plasma-Activated Liquids over an 18 Months Period. Water, 12(11), 3021. https://doi.org/10.3390/w12113021





