Groundwater Quality Assessment of a Multi-Layered Aquifer in a Desert Environment: A Case Study in Wadi ad-Dawasir, Saudi Arabia
Abstract
1. Introduction
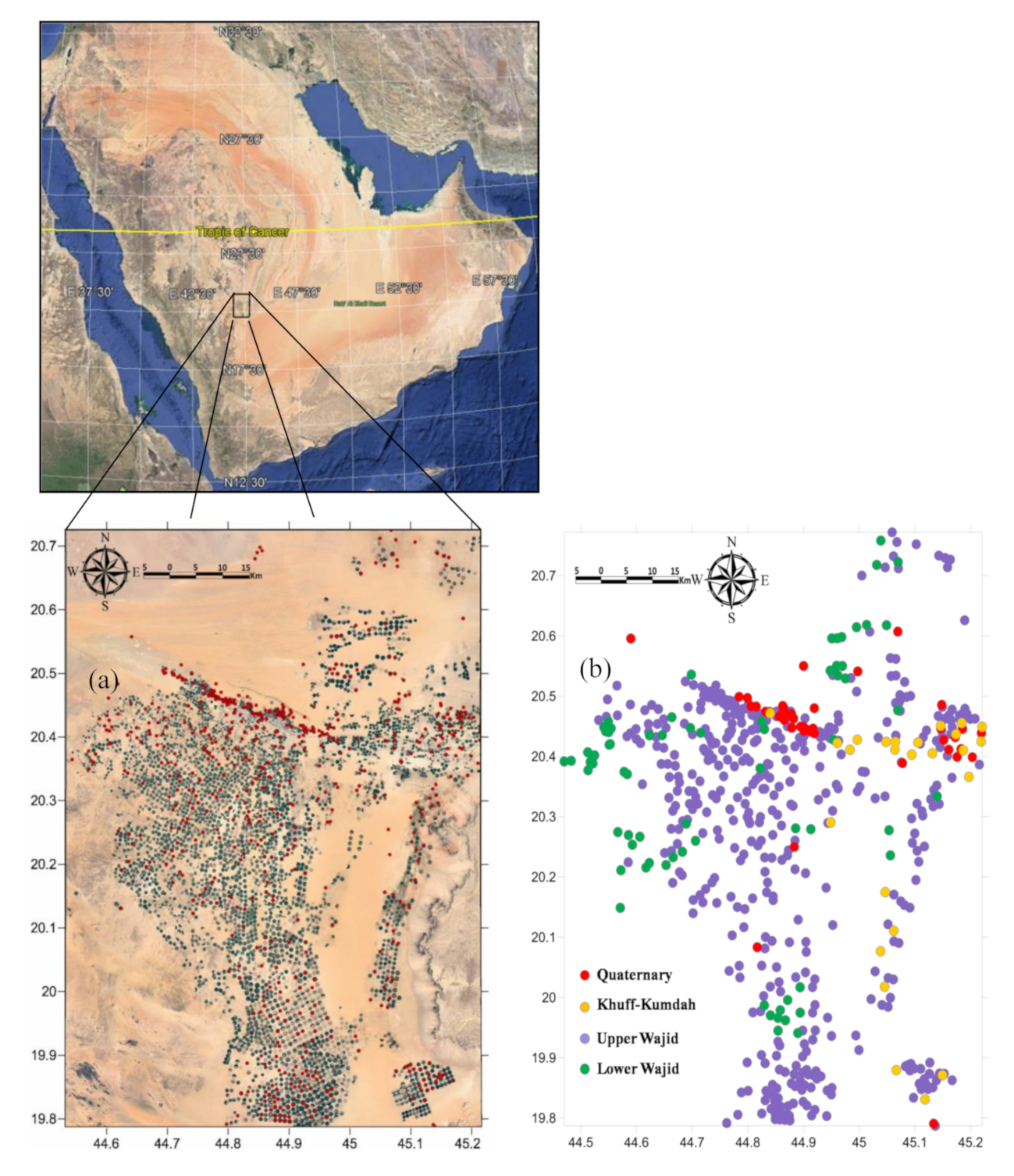
2. Study Area
2.1. Physiography and Area Characteristics
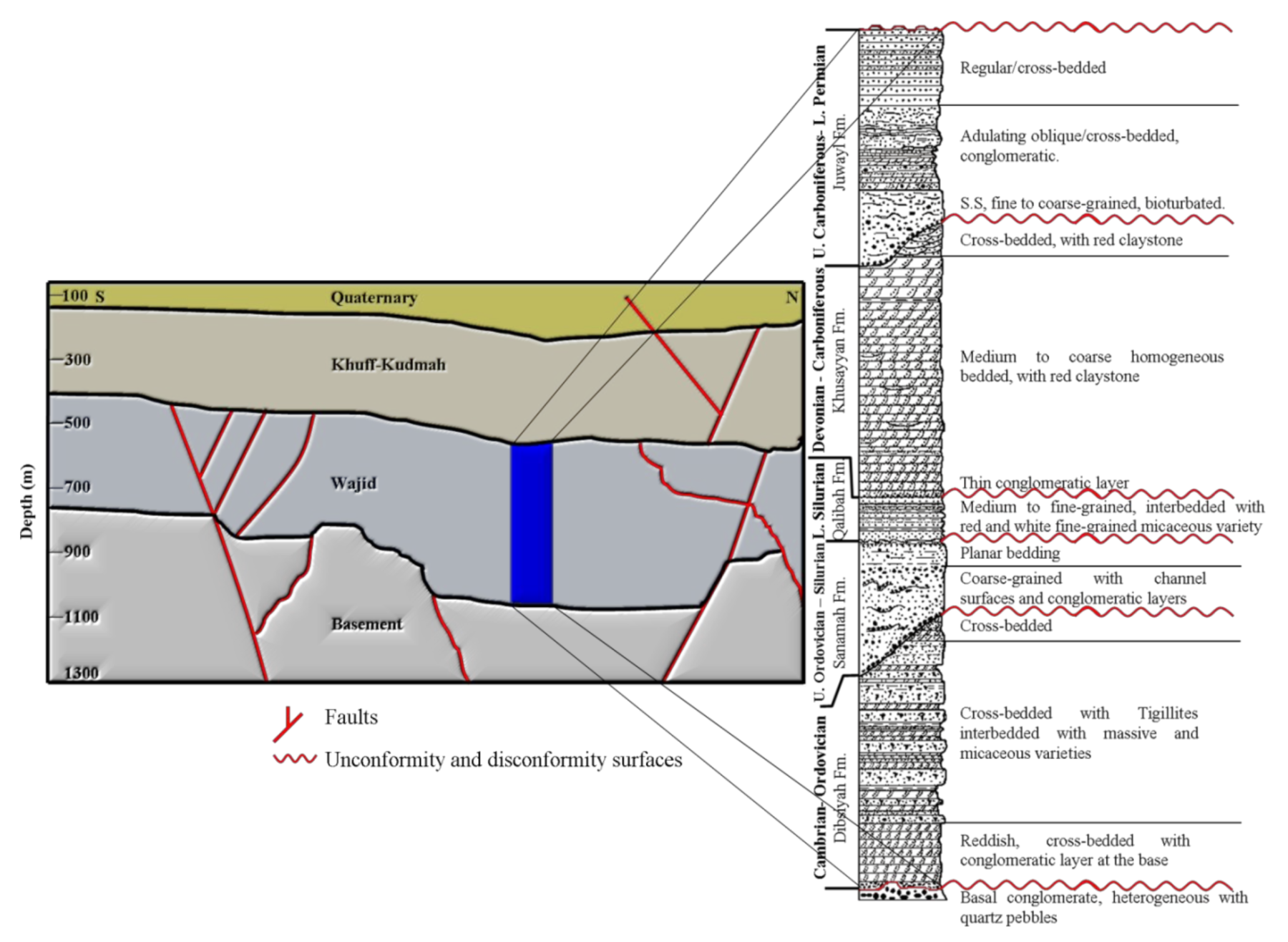
2.2. Geologic Setup
2.3. Hydrogeology
3. Materials and Methods
3.1. Hydrochemistry
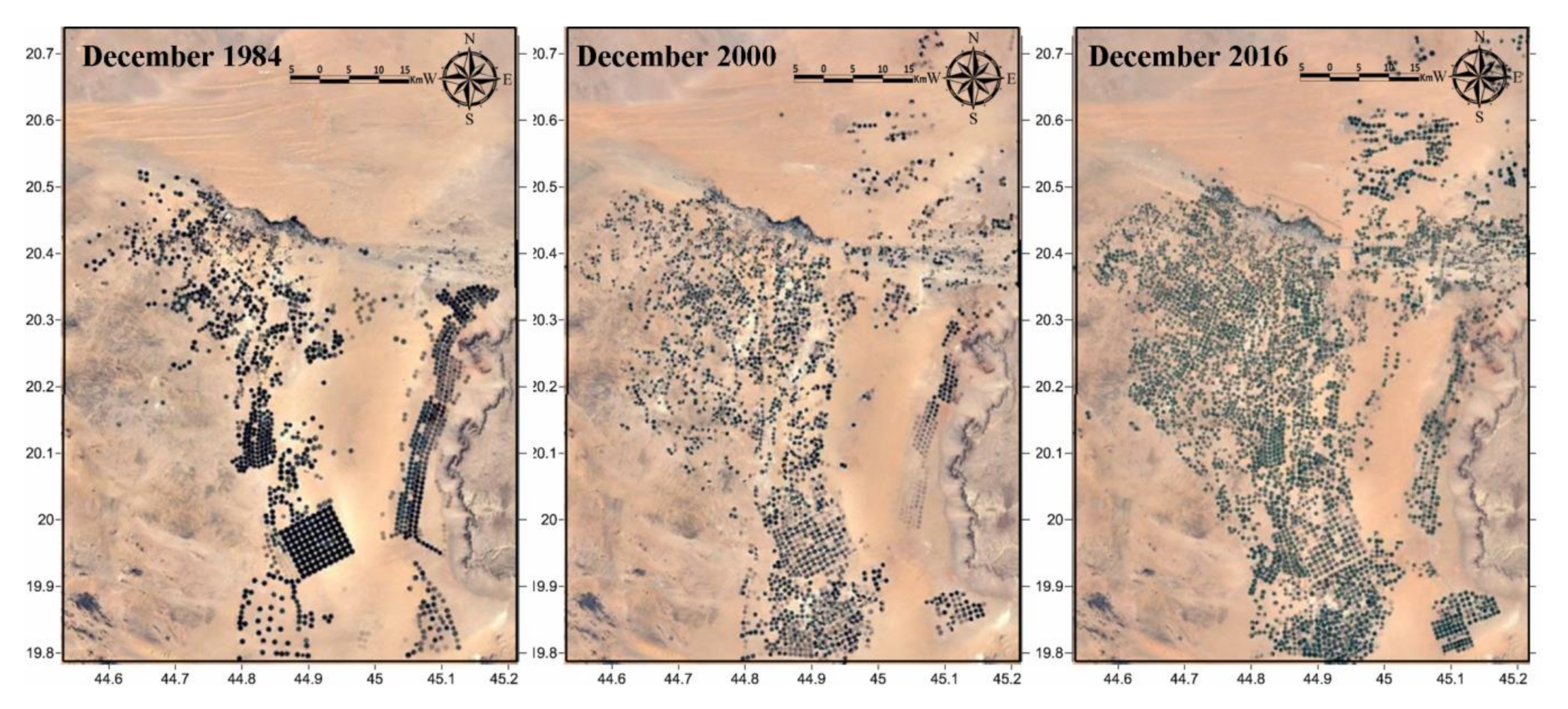
3.2. Land-Use Change and Hydrodynamics
4. Results
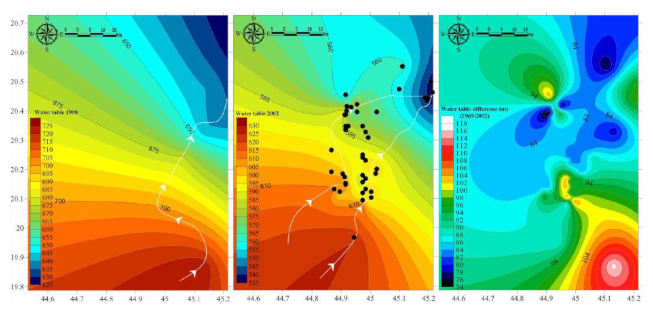
4.1. Agricultural Expansion and Hydrodynamics
4.2. Hydrochemical Characteristics and Health Risks
4.3. Emergence and Spatial Distribution of Pollutants
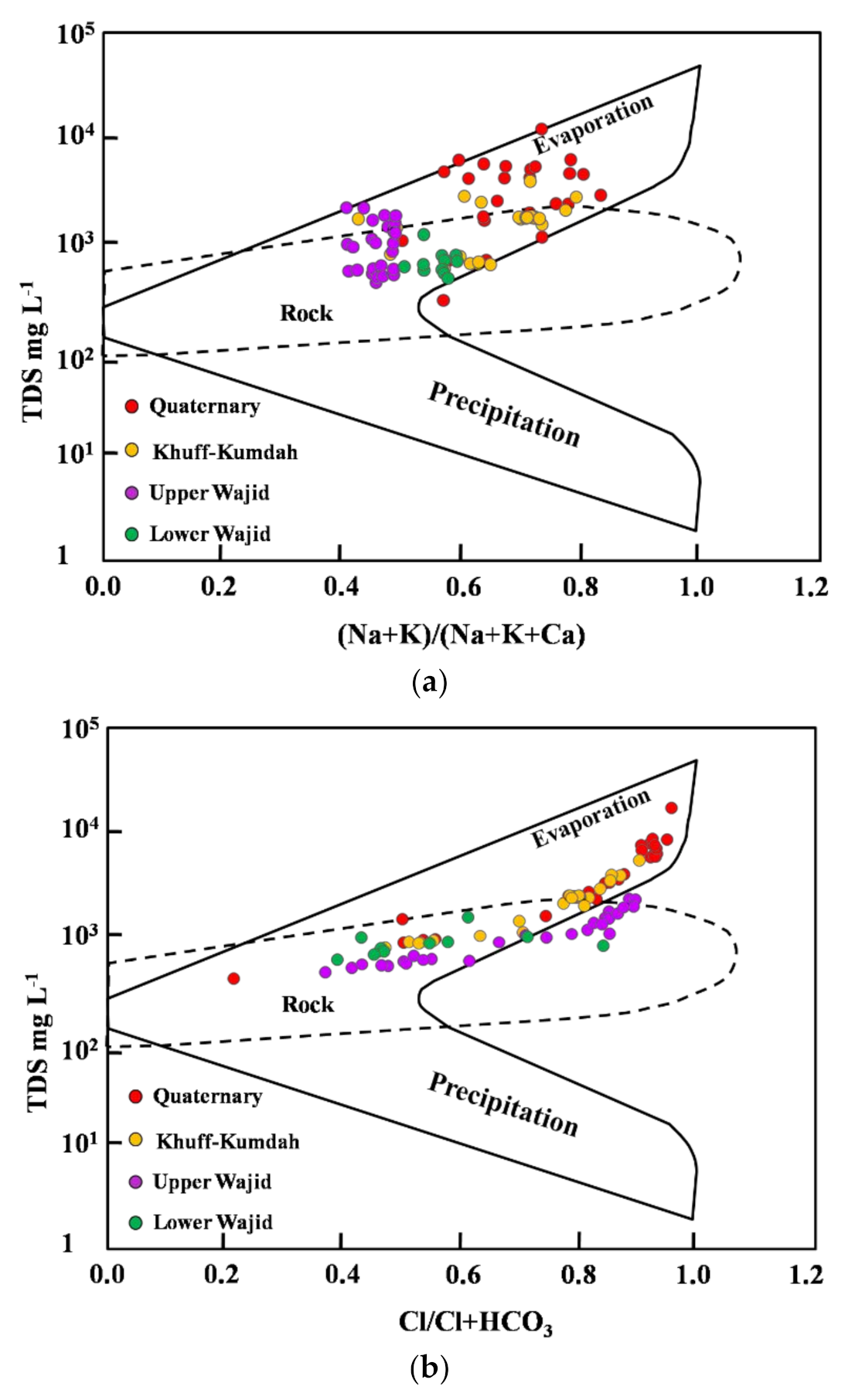
4.4. Mechanisms Controlling Hydrochemistry
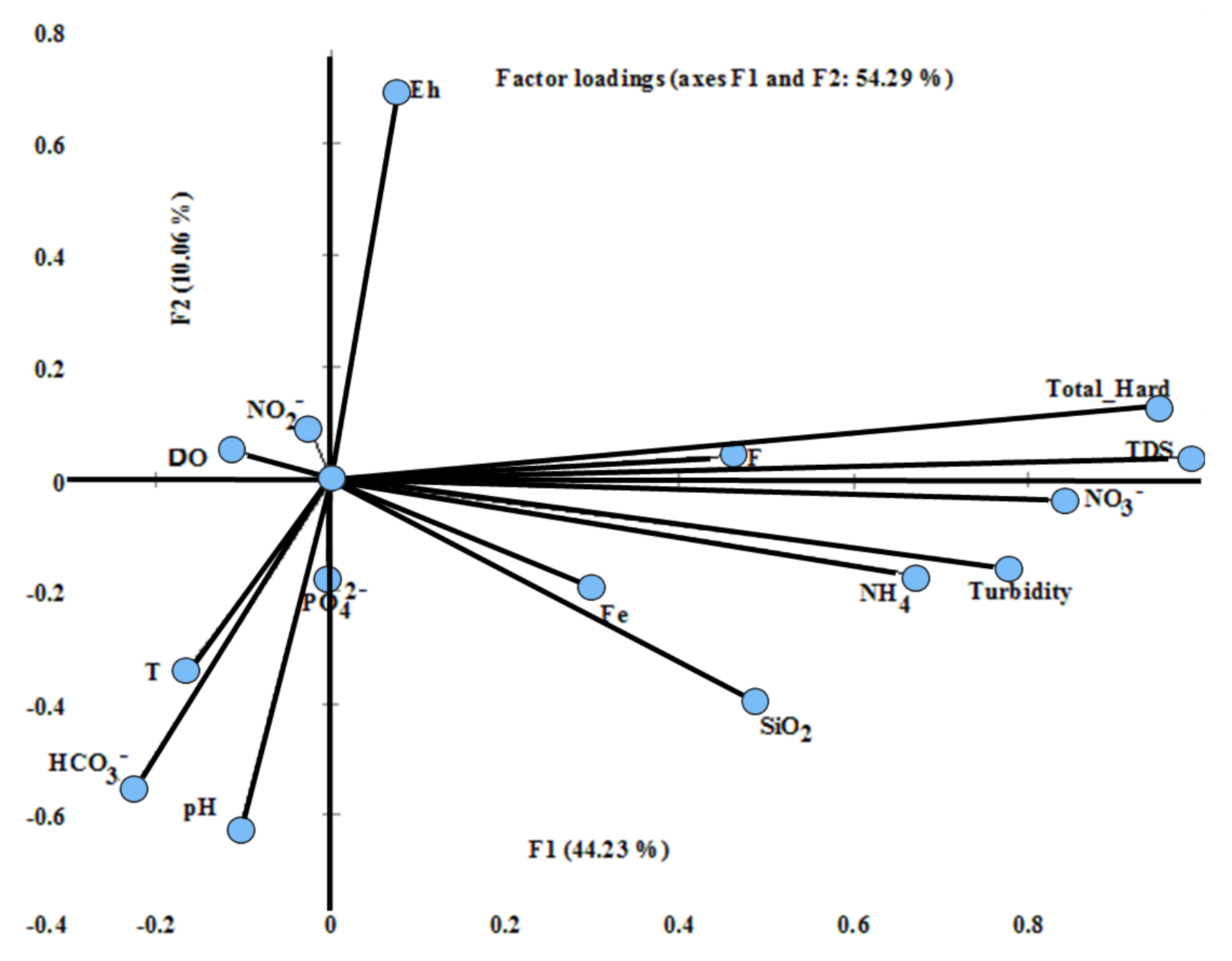
4.5. Factor Analysis (FA)
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kajenthira, A.; Siddiqi, A.; Anadon, L.D. A new case for promoting wastewater reuse in Saudi Arabia: Bringing energy into the water equation. J. Environ. Manag. 2012, 102, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Drewes, J.E.; Garduño, C.P.R.; Amy, G.L. Water reuse in the Kingdom of Saudi Arabia—Status, prospects and research needs. Water Supply 2012, 12, 926–936. [Google Scholar] [CrossRef]
- Bernáldez, F.; Benayas, J.M.R. Geochemical relationships between groundwater and wetland soils and their effects on vegetation in central Spain. Geoderma 1992, 55, 273–288. [Google Scholar] [CrossRef]
- Salama, R.B.; Otto, C.J.; Fitzpatrick, R.W. Contributions of groundwater conditions to soil and water salinization. Hydrogeol. J. 1999, 7, 46–64. [Google Scholar] [CrossRef]
- Wang, T.; Zlotnik, V.A.; Wedin, D.A.; Wally, K.D. Spatial trends in saturated hydraulic conductivity of vegetated dunes in the Nebraska Sand Hills: Effects of depth and topography. J. Hydrol. 2008, 349, 88–97. [Google Scholar] [CrossRef]
- Han, Z.; Tang, C.; Wu, P.; Zhang, R.; Zhang, C.; Sun, J. Hydrogeochemical characteristics and associated mechanism based on groundwater dating in a karstic basin, Guizhou Province, China. Environ. Earth Sci. 2015, 73, 67–76. [Google Scholar] [CrossRef]
- Singh, V.; Chandel, C.P.S. Analysis of wastewater of Jaipur cityfor agricultural use. Res. J. Chem. Environ. 2006, 10, 30–33. [Google Scholar]
- Nisi, B.; Buccianti, A.; Vaselli, O.; Perini, G.; Tassi, F.; Minissale, A. Hydrogeochemistry and strontium isotopes in the ArnoRiver Basin (Tuscany, Italy): Constraints on natural controlsbystatistical modeling. J. Hydrol. 2008, 360, 166–183. [Google Scholar] [CrossRef]
- Jiang, Y.; Yan, J. Effects of land use on hydrochemistry andcontamination of Karst groundwater from Nandongundergroundriver system, China. Water Air Soil Pollut. 2010, 210, 123–141. [Google Scholar] [CrossRef]
- Guendouz, A.; Moulla, A.S.; Edmunds, W.M.; Zouari, K.; Shand, P.; Mamou, A. Hydrogeochemical and isotopic evolution of water in the Complexe Terminal aquifer in the Algerian Sahara. Hydrogeol. J. 2003, 11, 483–495. [Google Scholar] [CrossRef]
- Bozdağ, A.; Göçmez, G. Evaluation of groundwater quality in the Cihanbeyli basin, Konya, Central Anatolia, Turkey. Environ. Earth Sci. 2013, 69, 921–937. [Google Scholar] [CrossRef]
- Missimer, T.M.; Drewes, J.E.; Amy, G.; Maliva, R.G.; Keller, S. Restoration of wadi aquifers by artificial recharge with treated wastewater. Groundwater 2012, 50, 514–526. [Google Scholar] [CrossRef] [PubMed]
- AL-Ailawi, J.; Abdulrazzik, M. Water in the Arabian Peninsula: Problems and perspectives. In Water in the Arab World, Perspectives and Prognoses; Harvard University, Division of Applied Sciences: Cambridge, MA, USA, 1994. [Google Scholar]
- Edgell, H.S. Aquifers of Saudi Arabia and their geological framework. Arab. J. Sci. Eng. 1997, 22, 3–31. [Google Scholar]
- Al-Faifi, H.J.A. Evaluation of Groundwater Resources in Wajid Qquifer in WadiDawasir Area Southern Saudi Arabia, Using Computer Simulation Data and Methods. Master’s Thesis, Department of Geology at the College of Science-King Saud University, Riyadh, Saudi Arabia, 2005; p. 164. [Google Scholar]
- Al-Kahtani, S.H.; Ismaiel, S.M. Groundwater irrigation management in the Kingdom of Saudi Arabia: A case study of Al-Wajid aquifer. J. US-Cn. Public Admin. 2010, 54–65. [Google Scholar]
- Elhag, M. Evaluation of Different Soil Salinity Mapping Using Remote Sensing Techniques in Arid Ecosystems, Saudi Arabia. J. Sens. 2016, 14, 1495–1515. [Google Scholar] [CrossRef]
- Dabbagh, M.E.; Rogers, J.J. Depositional environments and tectonic significance of the Wajid Sandstone of southern Saudi Arabia. J. Afr. Earth Sci. 1983, 1, 47–57. [Google Scholar] [CrossRef]
- Moshrif, M.; El-Hiti, A. Lithofacies and petrography of Wajid sandstone (Cambrian-Ordovician) Saudi Arabia. J. Afr. Earth Sci. 1989, 9, 401–412. [Google Scholar] [CrossRef]
- Vaslet, D. Upper Ordovician glacial deposits in Saudi Arabia. Episodes 1990, 13, 147–161. [Google Scholar] [CrossRef]
- Stump, T.; Van Der Eem, J. The stratigraphy, depositional environments and periods of deformation of the Wajid outcrop belt, southwestern Saudi Arabia. J. Afr. Earth Sci. 1994, 21, 421–441. [Google Scholar] [CrossRef]
- Sharland, P.; Archer, R.; Casey, D.M.; Davies, R.B.; Hall, S.; Heward, A.P.; Horbury, A.D.; Simmons, M.D. Arabian Plate Sequence Stratigraphy; GeoArabia SP2, Gulf PetroLink: Manama, Bahrain, 2001. [Google Scholar]
- Windley, B.F.; Whitehouse, M.J.; Stoeser, D.B.; ALkhirbash, S.; BA-Bttat, M.A.O.; Wanas, H.A.; Abdel-Maguid, N.M. Petrography and geochemistry of the Cambro-Ordovician Wajid Sandstone, SW Saudi Arabia: Implication for provenance and tectonic setting. J. Asian Earth Sci. 2006, 27, 416–429. [Google Scholar]
- Al-Ajmi, H.F.; Keller, M.; Hinderer, M.; Filomena, C.M. Lithofacies, depositional environments and stratigraphic architecture of the Wajid Group outcrops in southern Saudi Arabia. GeoArabia 2015, 20, 49–94. [Google Scholar]
- Al Asmari, A.; Mogren, S.; Lashin, A.; Hussein, M.; Al Fouzan, F. An integrated geophysical study for inferring the subsurface setting of water-bearing aquifers: Case study at Wadi al-Dawasir area, Saudi Arabia. Environ. Earth Sci. 2016, 75, 1028. [Google Scholar] [CrossRef]
- Benaafi, M.; Hariri, M.; Bertotti, G.; Al-Shaibani, A.; Abdullatif, O.; Makkawi, M. Natural fracture system of the Cambro-Permian Wajid Group, Wadi Al-Dawasir, SW Saudi Arabia. J. Pet. Sci. Eng. 2019. [Google Scholar] [CrossRef]
- MOP (Ministry of Planning). In Fourth Development Plan: 1985–1990MOP; MOP: Riyadh, Saudi Arabia.
- Gibbs, R.J. Mechanisms Controlling World Water Chemistry. Science 1970, 170, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Kynčlová, P.; Hron, K.; Filzmoser, P. Correlation Between Compositional Parts Based on Symmetric Balances. Math. Geol. 2017, 49, 777–796. [Google Scholar] [CrossRef]
- Reimann, C.; Filzmoser, P.; Hron, K.; Kynčlová, P.; Garrett, R.G. A new method for correlation analysis of compositional (environmental) data—A worked example. Sci. Total Environ. 2017, 607, 965–971. [Google Scholar] [CrossRef]
- Rock, N.M.S. Numerical Geology; Lecture Notes in Earth Sciences 18; Springer Verlag: New York, NY, USA; Berlin/Heidelberg, Germany, 1988. [Google Scholar]
- Ravikumar, P.; Somashekar, R.K.; Angami, M. Hydrochemistry and evaluation of groundwater suitability for irrigation and drinking purposes in the Markandeya River basin, Belgaum District, Karnataka State, India. Environ. Monit. Assess. 2011, 173, 459–487. [Google Scholar] [CrossRef]
- Selvam, S.; Manimaran, G.; Sivasubramanian, P.; Balasubramanian, N.; Seshunarayana, T. GIS-based Evaluation of Water Quality Index of groundwater resources around Tuticorin coastal city, south India. Environ. Earth Sci. 2014, 71, 2847–2867. [Google Scholar] [CrossRef]
- Ramesh, K.; Elango, L. Groundwater quality assessment in Tondiar basin. Indian J. Environ. Prot. 2006, 26, 497–504. [Google Scholar]
- World Health Organization—WHO. Guidelines for Drinking Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011; pp. 1–518. [Google Scholar]
- Kumar, S.K.; Chandrasekar, N.; Seralathan, P.; Godson, P.S.; Magesh, N.S. Hydrogeochemical study of shallow carbonate aquifers, Rameswaram Island, India. Environ. Monit. Assess. 2012, 184, 4127–4138. [Google Scholar] [CrossRef]
- Fewtrell, L. Drinking-Water Nitrate, Methemoglobinemia, and Global Burden of Disease: A Discussion. Environ. Health Perspect. 2004, 112, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Pirsaheb, M.; Fazlzadehdavil, M.; Hazrati, S.; Sharafi, K.; Khodadadi, T.; Safari, Y. A survey on nitrogen and phosphor compound variation processes in wastewater stabilization ponds. Polish J. Environ. Stud. 2014, 23, 831–834. [Google Scholar]
- Carney, M. European Drinking Water Standards. J. Am. Water Work. Assoc. 1991, 83, 48–55. [Google Scholar] [CrossRef]
- Krapac, I.G.; Dey, W.S.; Roy, W.R.; Smyth, C.A.; Storment, E.; Sargent, S.L.; Steele, J. Impacts of swine manure pits on groundwater quality. Environ. Pollut. 2002, 120, 475–492. [Google Scholar] [CrossRef]
- Daesslé, L.W.; Mendoza-Espinosa, L.; Camacho-Ibar, V.; Rozier, W.; Morton, O.; Van Dorst, L.; Lugo-Ibarra, K.C.; Quintanilla-Montoya, A.L.; Rodríguez-Pinal, A. The hydrogeochemistry of a heavily used aquifer in the Mexican wine-producing Guadalupe Valley, Baja California. Environ. Geol. 2006, 51, 151–159. [Google Scholar] [CrossRef]
- Smedley, P.; Nicolli, H.; Macdonald, D.; Barros, A.; Tullio, J. Hydrogeochemistry of arsenic and other inorganic constituents in groundwaters from La Pampa, Argentina. Appl. Geochem. 2002, 17, 259–284. [Google Scholar] [CrossRef]
- Edmunds, W.M.; Smedley, P.L. Fluoride in natural waters. In Essentials of Medical Geology: Revised Edition; Alloway, B., Centeno, J., Finkelman, R., Fuge, R., Lindh, U., Smedley, P., Selinus, O., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 311–350. [Google Scholar]
- Viswanathan, G.; Jaswanth, A.; Gopalakrishnan, S.; Ilango, S.S.; Aditya, G. Determining the optimal fluoride concentration in drinking water for fluoride endemic regions in South India. Sci. Total. Environ. 2009, 407, 5298–5307. [Google Scholar] [CrossRef]
- Dissanayake, C.B. The fluoride problem in the ground water of Sri Lanka—Environmental management and health. Int. J. Environ. Stud. 1991, 38, 137–155. [Google Scholar] [CrossRef]
- Kezao, C.; Bowler, J. Late pleistocene evolution of salt lakes in the Qaidam basin, Qinghai province, China. Palaeogeogr. Palaeoclim. Palaeoecol. 1986, 54, 87–104. [Google Scholar] [CrossRef]
- British Geological Survey-BGS. Fluoride in Groundwater. 2019. Available online: http://www.bgs.ac.uk/research/groundwater/health/fluoride.html (accessed on 19 May 2019).
- Redwan, M.; Moneim, A.A.A. Factors controlling groundwater hydrogeochemistry in the area west of Tahta, Sohag, Upper Egypt. J. Afr. Earth Sci. 2015, 118, 328–338. [Google Scholar] [CrossRef]
- Verma, S.; Mukherjee, A.; Mahanta, C.; Choudhury, R.; Mitra, K. Influence of geology on groundwater–sediment interactions in arsenic enriched tectono-morphic aquifers of the Himalayan Brahmaputra river basin. J. Hydrol. 2016, 540, 176–195. [Google Scholar] [CrossRef]
- Awaleh, M.O.; Baudron, P.; Soubaneh, Y.D.; Boschetti, T.; Hoch, F.B.; Egueh, N.M.; Mohamed, J.; Dabar, O.A.; Masse-Dufresne, J.; Gassani, J. Recharge, groundwater flow pattern and contamination processes in an arid volcanic area: Insights from isotopic and geochemical tracers (Bara aquifer system, Republic of Djibouti). J. Geochem. Explor. 2017, 175, 82–98. [Google Scholar] [CrossRef]
- Guo, X.; Feng, Q.; Liu, W.; Li, Z.; Wen, X.H.; Si, J.; Xi, H.; Guo, R.; Jia, B. Stable isotopic and geochemical identification of groundwater evolution and recharge sources in the arid Shule River Basin of Northwestern China. Hydrol. Process. 2015, 29, 4703–4718. [Google Scholar] [CrossRef]
- Dogramaci, S.; Skrzypek, G.; Dodson, W.; Grierson, P.F. Stable isotope and hydrochemical evolution of groundwater in the semi-arid Hamersley Basin of sub-tropical northwest Australia. J. Hydrol. 2012, 475, 281–293. [Google Scholar] [CrossRef]
- Balugani, E.; Lubczynski, M.; Reyes-Acosta, L.; Van Der Tol, C.; Francés, A.; Metselaar, K. Groundwater and unsaturated zone evaporation and transpiration in a semi-arid open woodland. J. Hydrol. 2017, 547, 54–66. [Google Scholar] [CrossRef]
- Kamai, T.; Assouline, S. Evaporation From Deep Aquifers in Arid Regions: Analytical Model for Combined Liquid and Vapor Water Fluxes. Water Resour. Res. 2018, 54, 4805–4822. [Google Scholar] [CrossRef]
- Menz, C. Oxygen Delivering Processes in Groundwater and Their Relevance for Iron-Related Clogging Processes—A Case Study on the Quaternary Aquifers of Berlin. Ph.D. Thesis, Freie Universität Berlin, Berlin, Germany, 2016; p. 185. [Google Scholar]
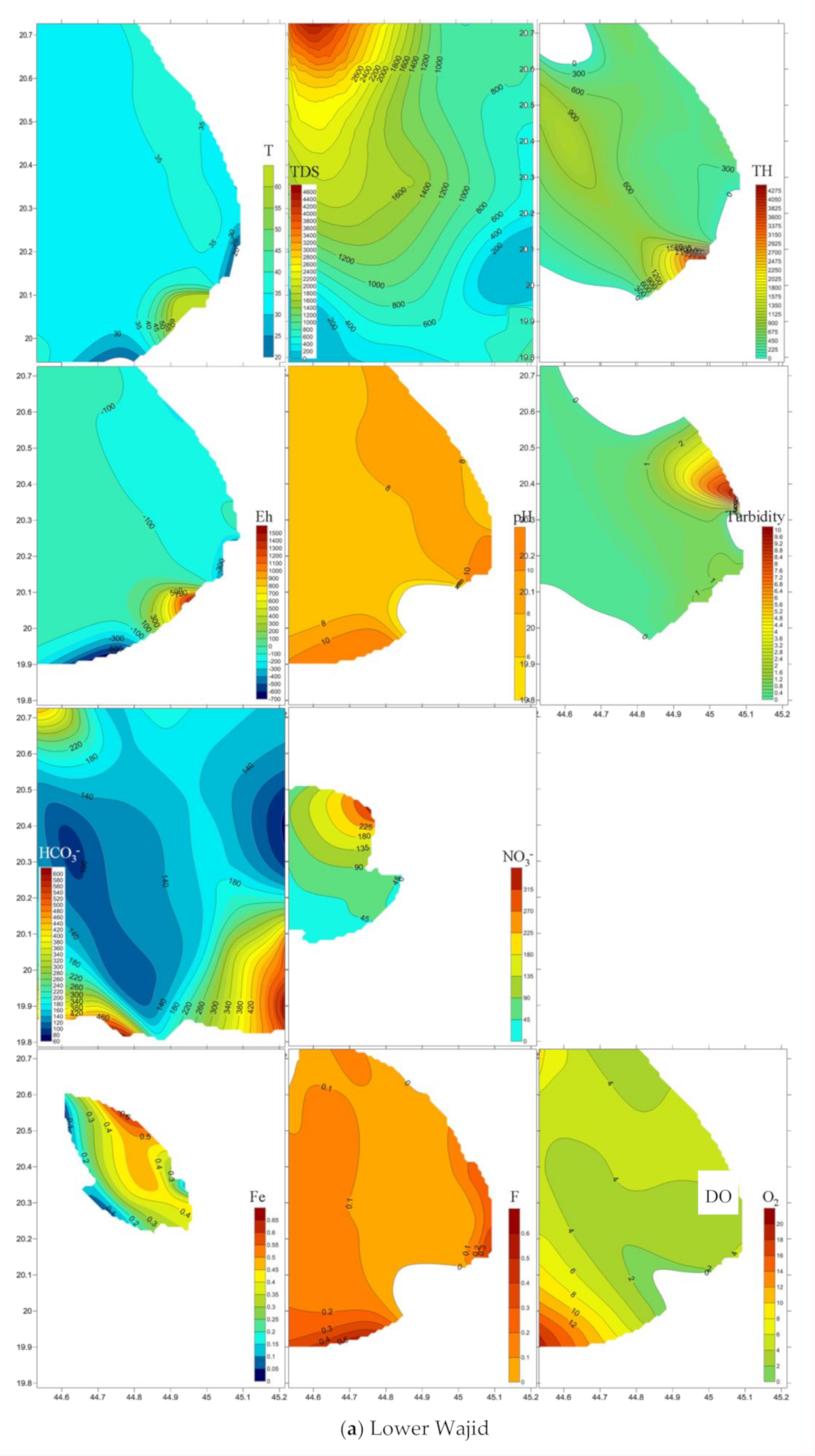
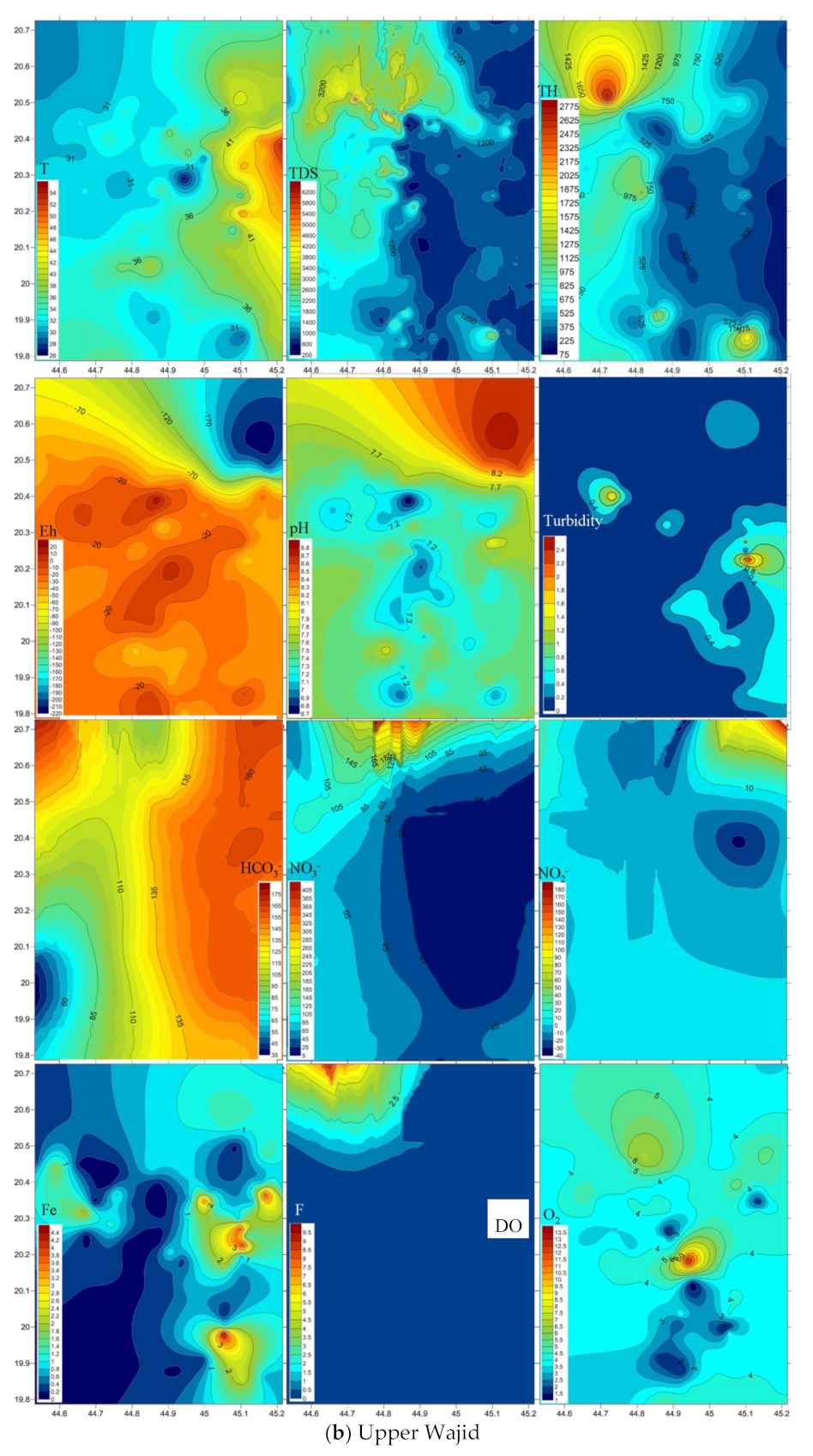
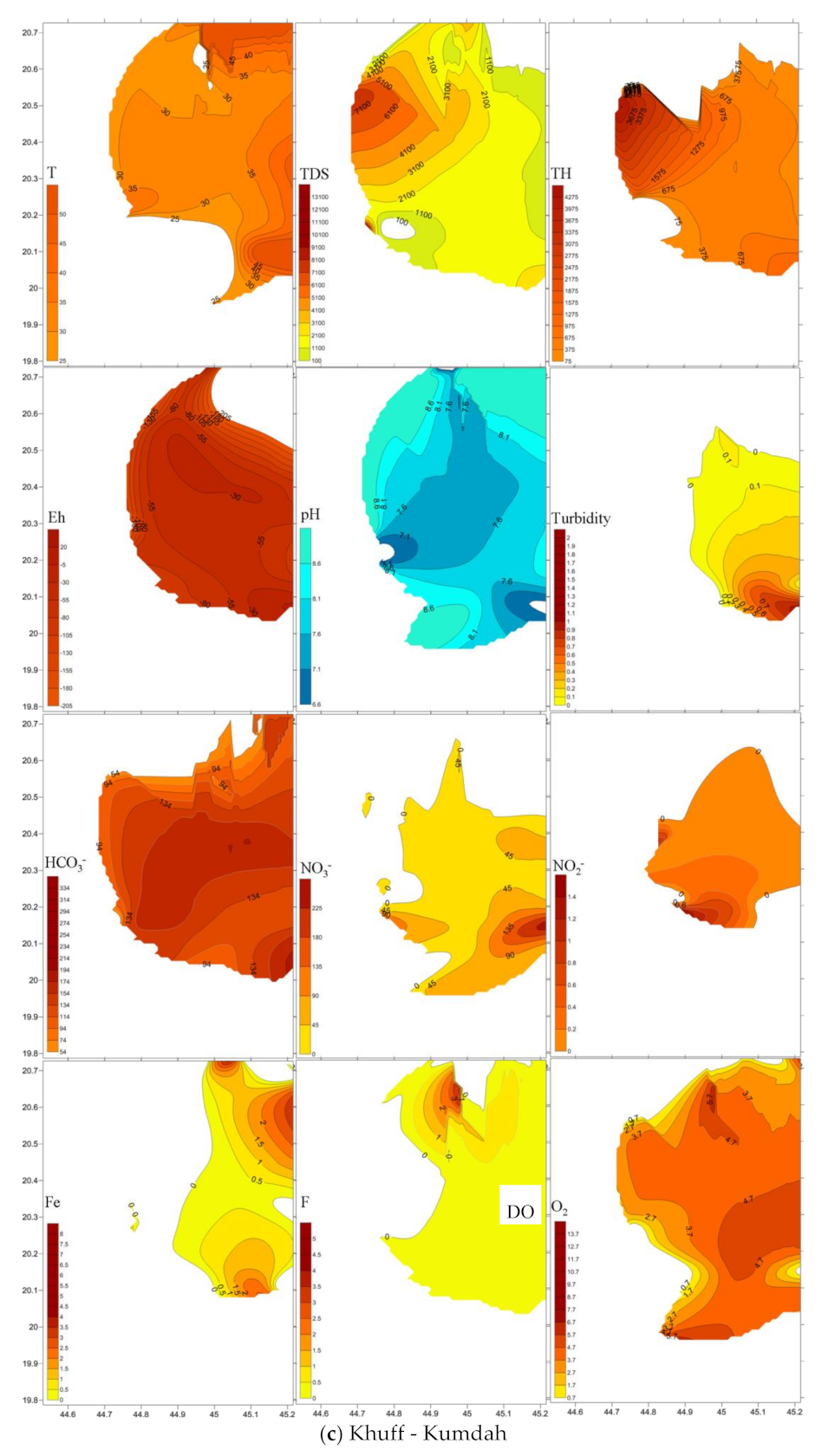
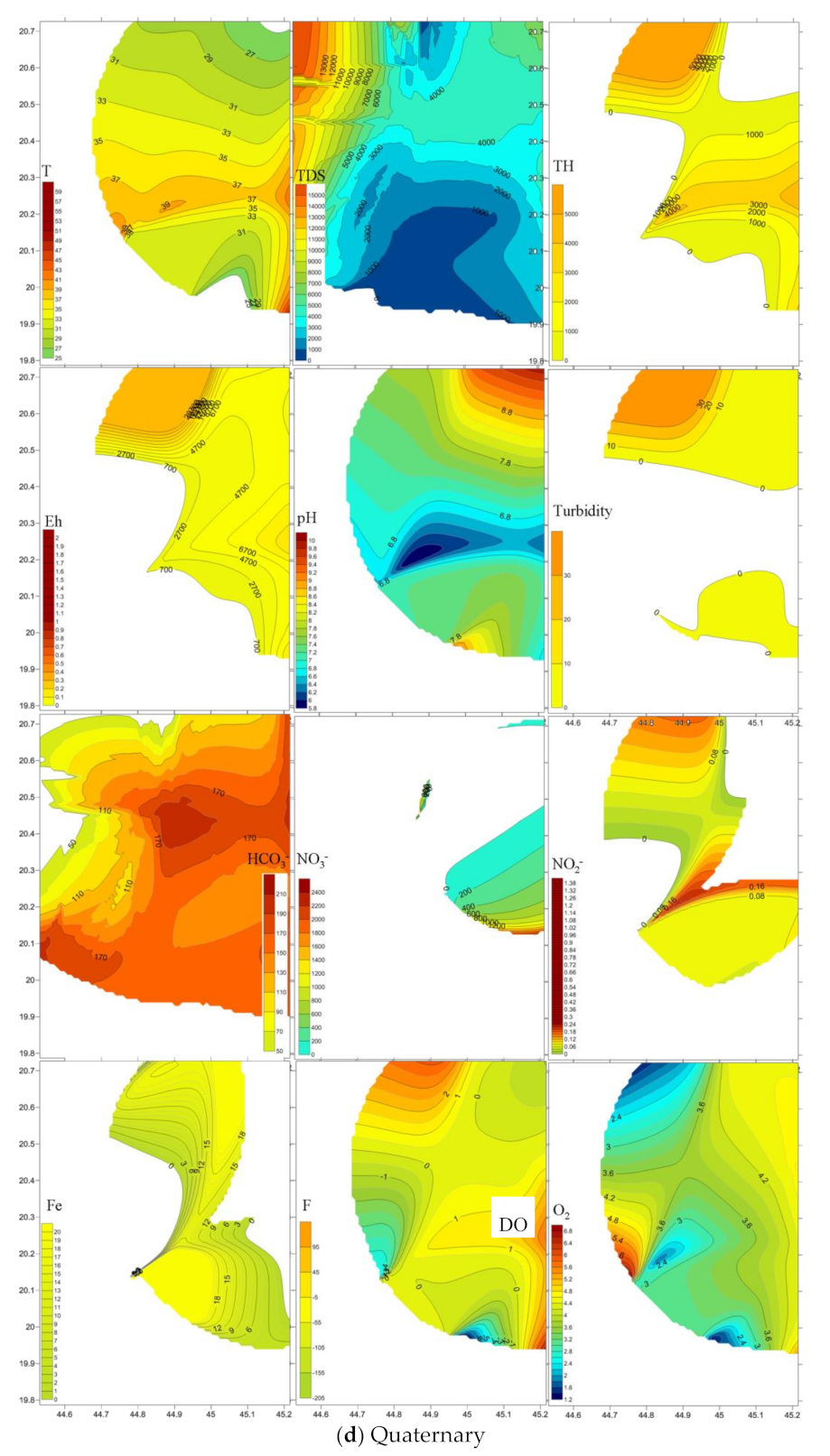
| T | pH | DO | Eh | Turbidity | Total Hardness | TDS | Fe2+ | NH4 | HCO3− | NO3− | F | NO2− | PO42− | SiO2 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Quaternary | Min | 30.9 | 7.15 | 2.01 | −145 | 0.02 | 314 | 355 | 0.17 | 0.17 | 62 | 10.8 | 0.11 | 0.04 | 0.23 | 13.8 |
| Max | 39.1 | 7.96 | 5.82 | −22 | 29.05 | 4737 | 16,500 | 8.53 | 3.54 | 335 | 2781 | 2.98 | 0.36 | 0.29 | 59.4 | |
| Mean | 32.8 | 7.5 | 4.3 | −52.9 | 2.7 | 1519 | 4659.0 | 2.8 | 1.8 | 177.0 | 483 | 0.8 | 0.1 | 0.3 | 28.0 | |
| SD | 2.1 | 0.2 | 1.0 | 42.3 | 8.3 | 1343.6 | 3734.2 | 3.3 | 1.7 | 62.1 | 952.4 | 1.0 | 0.1 | 0.0 | 12.2 | |
| count | 13 | 13 | 13 | 10 | 12 | 12 | 45 | 8 | 3 | 45 | 10 | 12 | 6 | 2 | 12 | |
| Khuff-Kumdah | Min | 30.1 | 6.7 | 1.9 | −180 | 0.0 | 92 | 472 | 0.0 | 2.4 | 105 | 2.6 | 0.1 | 0.0 | 0.1 | 15.7 |
| Max | 46 | 8.4 | 7.3 | −17 | 0.5 | 1479 | 5405 | 1.8 | 2.4 | 255 | 145.1 | 0.7 | 1.4 | 0.4 | 31.4 | |
| Mean | 32.5 | 7.6 | 4.4 | −50 | 0.1 | 629.3 | 1906.6 | 0.5 | 2.4 | 152.5 | 33.2 | 0.3 | 0.2 | 0.2 | 22.6 | |
| SD | 6.3 | 0.3 | 1.1 | 39.6 | 0.1 | 302.1 | 1061 | 0.6 | 25 | 32.3 | 0.2 | 0.3 | 0.1 | 4.5 | ||
| count | 36 | 36 | 36 | 24 | 35 | 35 | 36 | 17 | 1 | 36 | 33 | 35 | 17 | 5 | 35 | |
| Upper Wajid | Min | 25 | 6.97 | 1.73 | −68 | 0.02 | 188 | 414 | 0.06 | 0 | 73 | 13.7 | 0.07 | 0.04 | 0.35 | 14.2 |
| Max | 54.8 | 8.06 | 8.22 | 1 | 0.3 | 1387 | 3045 | 1.1 | 0 | 222 | 270 | 0.48 | 0.91 | 0.35 | 17.3 | |
| Mean | 34.4 | 7.4 | 4 | −25.5 | 0.1 | 728.1 | 1541.5 | 0.4 | 118.5 | 71.3 | 0.2 | 0.3 | 0.4 | 15.8 | ||
| SD | 4.8 | 0.3 | 1.6 | 21.5 | 0.1 | 353.1 | 670.8 | 0.4 | 41.8 | 70.8 | 0.1 | 0.5 | 0.8 | |||
| count | 119 | 11 | 11 | 8 | 11 | 11 | 46 | 6 | 0 | 24 | 11 | 9 | 3 | 1 | 11 | |
| Lower Wajid | Min | 30 | 6.9 | 0.4 | −205 | 0.0 | 245 | 563 | 0.0 | 0.5 | 56 | 11.2 | 0.0 | 0.1 | 0.5 | 14.1 |
| Max | 40.7 | 8.8 | 6.2 | 5.0 | 2.0 | 1097 | 4167 | 0.9 | 0.5 | 259 | 118 | 0.4 | 0.1 | 0.5 | 18.7 | |
| Mean | 33.6 | 7.7 | 3.8 | −75.5 | 0.2 | 558.9 | 1518.2 | 0.4 | 0.5 | 140.7 | 52.4 | 0.1 | 0.1 | 0.5 | 16.7 | |
| SD | 3 | 0.7 | 1.3 | 84.6 | 0.5 | 243.4 | 799.7 | 0.3 | 45.7 | 29.8 | 0.1 | 0.0 | 1.1 | |||
| count | 17 | 17 | 17 | 14 | 17 | 17 | 73 | 10 | 1 | 23 | 13 | 16 | 2 | 1 | 17 | |
| W. Dawasir | Min | 3.1 | 6.63 | 0.41 | −205 | 0.02 | 92 | 83 | 0.03 | 0.05 | 54 | 2.6 | 0.04 | 0.01 | 0.01 | 10.1 |
| Max | 54.8 | 8.79 | 13.8 | 22 | 29.05 | 4737 | 16,500 | 8.53 | 5.66 | 335 | 2781 | 5.27 | 1.38 | 16.1 | 59.4 | |
| Mean | 33.9 | 7.5 | 4.4 | −46.7 | 0.3 | 607.2 | 1838.3 | 0.9 | 1.2 | 148.5 | 68.5 | 0.3 | 0.1 | 0.8 | 18.7 | |
| SD | 4.9 | 0.4 | 1.6 | 51.9 | 2.2 | 530.0 | 1541.8 | 1.4 | 1.6 | 42.4 | 259.5 | 0.5 | 0.2 | 3.1 | 5.3 | |
| count | 185 | 186 | 175 | 125 | 179 | 179 | 686 | 104 | 16 | 238 | 153 | 173 | 78 | 26 | 178 |
| T | pH | DO | Eh | Turbidity | Total Hardness | TDS | Fe2+ | NH4 | HCO3− | NO3− | F | NO2− | PO42− | SiO2 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T | 1 | ||||||||||||||
| pH | 0.09 | 1 | |||||||||||||
| DO | −0.04 | 0.06 | 1 | ||||||||||||
| Eh | −0.17 | −0.90 | −0.09 | 1 | |||||||||||
| Turbidity | −0.01 | −0.04 | −0.12 | −0.10 | 1 | ||||||||||
| Total Hardness | −0.21 | −0.14 | −0.09 | 0.14 | 0.57 | 1 | |||||||||
| TDS | −0.21 | −0.11 | −0.03 | 0.10 | 0.51 | 0.92 | 1 | ||||||||
| Fe2+ | 0.07 | 0.07 | −0.11 | 0.00 | 0.46 | 0.17 | 0.24 | 1 | |||||||
| NH4 | 0.0 | 0.05 | 0.02 | 0.49 | 0.84 | 0.90 | 0.80 | 0.98 | 1 | ||||||
| HCO3− | 0.32 | 0.11 | −0.02 | −0.26 | −0.14 | −0.30 | 0.11 | −0.05 | −0.61 | 1 | |||||
| NO3− | −0.08 | −0.10 | −0.01 | 0.19 | 0.50 | 0.66 | 0.85 | 0.51 | 0.28 | −0.33 | 1 | ||||
| F | −0.08 | −0.04 | 0.10 | −0.02 | 0.16 | 0.60 | 0.60 | 0.02 | 0.66 | −0.04 | 0.39 | 1 | |||
| NO2− | −0.01 | −0.02 | 0.00 | 0.07 | −0.02 | 0.06 | 0.08 | −0.08 | −0.14 | −0.24 | 0.10 | −0.02 | 1 | ||
| PO42− | −0.02 | −0.27 | −0.27 | 0.48 | −0.09 | −0.02 | −0.04 | −0.25 | −0.23 | −0.32 | 0.05 | −0.24 | 0.14 | 1 | |
| SiO2 | 0.06 | 0.08 | −0.05 | −0.13 | 0.29 | 0.36 | 0.40 | 0.43 | 0.90 | 0.18 | 0.12 | 0.19 | −0.06 | −0.13 | 1 |
| pH | Turbidity NTU | Total Hardness | TDS | Fe2+ | NH4 | NO3− | F | |
|---|---|---|---|---|---|---|---|---|
| WHO 2011 | 8.5 | 1 | 200 | 1000 | 0.3 | 1.5 | 50 | 1.5 |
| Quaternary | 16.6 | 100 | 85.7 | 62.5 | 2.3 | 33.3 | 18.2 | |
| Khuff-Kumdah | 91.4 | 70.2 | 50 | 14.7 | ||||
| Upper Wajid | 3.4 | 1.8 | 100 | 63.4 | 68.1 | 0.5 | 33.7 | |
| Lower Wajid | 33.3 | 6.6 | 100 | 59.7 | 44.4 | 33.3 | ||
| W. Dawasir | 5.5 | 2.8 | 98.8 | 65.7 | 63 | 0.7 | 32 | 1.2 |
| F1 | F2 | F3 | F4 | F5 | F6 | Communality | |
|---|---|---|---|---|---|---|---|
| T | −0.17 | −0.34 | 0.01 | −0.14 | 0.13 | 0.14 | 0.20 |
| pH | −0.10 | −0.63 | −0.37 | 0.40 | −0.33 | −0.12 | 0.83 |
| DO | −0.11 | 0.05 | −0.06 | 0.13 | 0.05 | 0.06 | 0.04 |
| Eh | 0.08 | 0.70 | 0.35 | −0.37 | 0.30 | −0.02 | 0.85 |
| Turbidity | 0.78 | −0.16 | −0.32 | −0.49 | −0.09 | 0.02 | 0.99 |
| Total Hardness | 0.95 | 0.13 | 0.10 | 0.18 | −0.08 | 0.07 | 0.98 |
| TDS | 0.98 | 0.04 | 0.08 | 0.16 | 0.02 | 0.00 | 1.00 |
| Fe2+ | 0.31 | −0.20 | −0.21 | −0.26 | 0.19 | −0.18 | 0.31 |
| NH4 | 0.67 | −0.17 | −0.27 | −0.43 | −0.08 | 0.08 | 0.75 |
| HCO3− | −0.22 | −0.55 | 0.06 | 0.00 | 0.42 | 0.37 | 0.67 |
| NO3− | 0.84 | −0.04 | −0.27 | −0.37 | −0.19 | 0.05 | 0.96 |
| F | 0.47 | 0.04 | 0.10 | 0.33 | 0.05 | 0.29 | 0.42 |
| NO2− | −0.02 | 0.08 | 0.02 | 0.01 | −0.07 | −0.10 | 0.02 |
| PO42− | −0.01 | −0.18 | 0.65 | −0.27 | −0.42 | −0.08 | 0.70 |
| SiO2 | 0.49 | −0.40 | 0.11 | 0.13 | 0.54 | −0.38 | 0.87 |
| Eigenvalue | 9.73 | 2.21 | 1.54 | 1.37 | 1.02 | 0.49 | |
| Variability (%) | 44.23 | 10.06 | 7 | 6.24 | 4.63 | 2.24 | |
| Cumulative% | 44.23 | 54.29 | 61.29 | 67.52 | 72.15 | 74.39 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masoud, A.A.; Aldosari, A.A. Groundwater Quality Assessment of a Multi-Layered Aquifer in a Desert Environment: A Case Study in Wadi ad-Dawasir, Saudi Arabia. Water 2020, 12, 3020. https://doi.org/10.3390/w12113020
Masoud AA, Aldosari AA. Groundwater Quality Assessment of a Multi-Layered Aquifer in a Desert Environment: A Case Study in Wadi ad-Dawasir, Saudi Arabia. Water. 2020; 12(11):3020. https://doi.org/10.3390/w12113020
Chicago/Turabian StyleMasoud, Alaa A., and Ali A. Aldosari. 2020. "Groundwater Quality Assessment of a Multi-Layered Aquifer in a Desert Environment: A Case Study in Wadi ad-Dawasir, Saudi Arabia" Water 12, no. 11: 3020. https://doi.org/10.3390/w12113020
APA StyleMasoud, A. A., & Aldosari, A. A. (2020). Groundwater Quality Assessment of a Multi-Layered Aquifer in a Desert Environment: A Case Study in Wadi ad-Dawasir, Saudi Arabia. Water, 12(11), 3020. https://doi.org/10.3390/w12113020





