Conservation and Management of Isolated Pools in Temporary Rivers
Abstract
1. Introduction
2. Hydrological Characterization of Isolated Pools
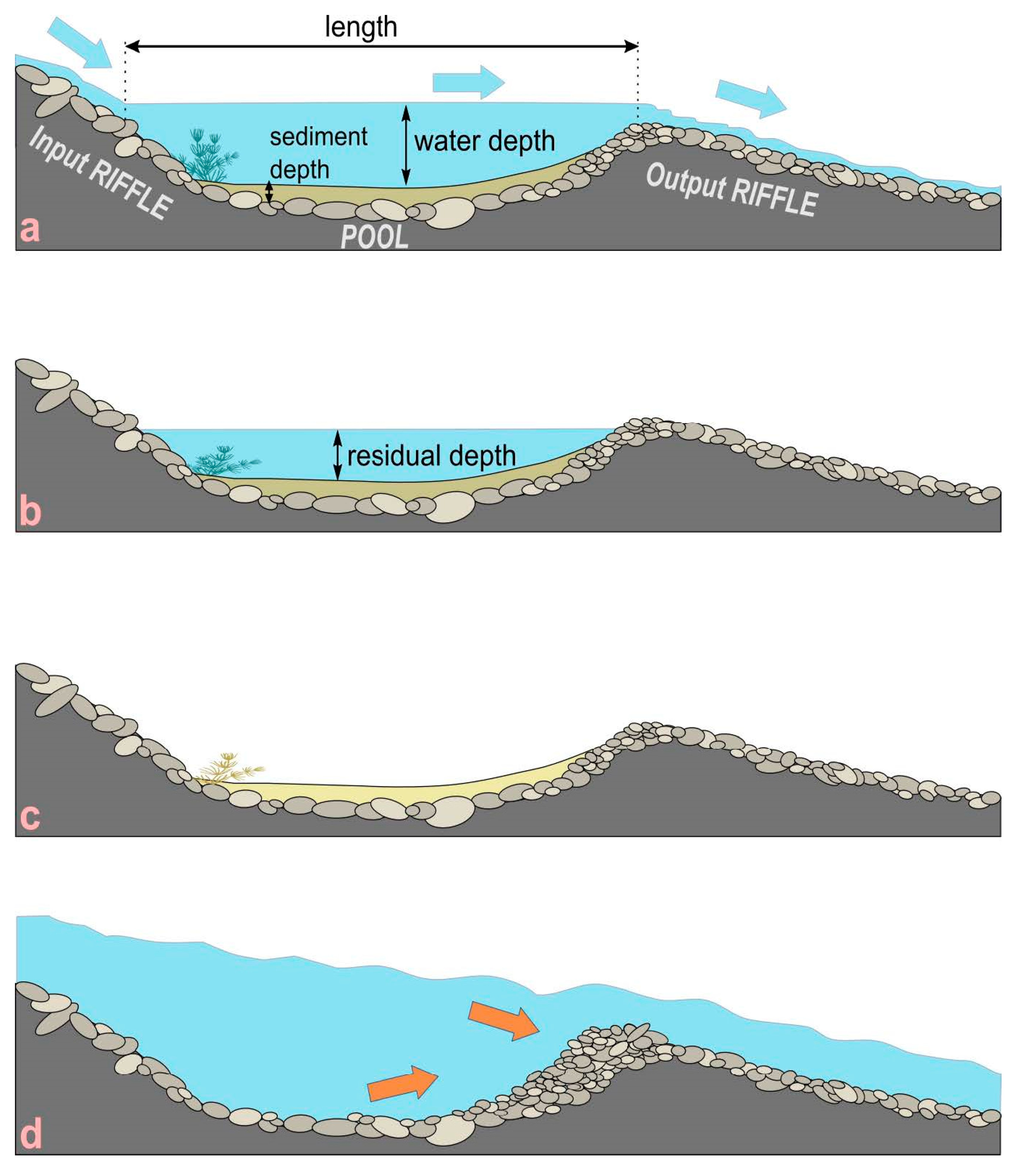
3. Geomorphological Characterization of Isolated Pools
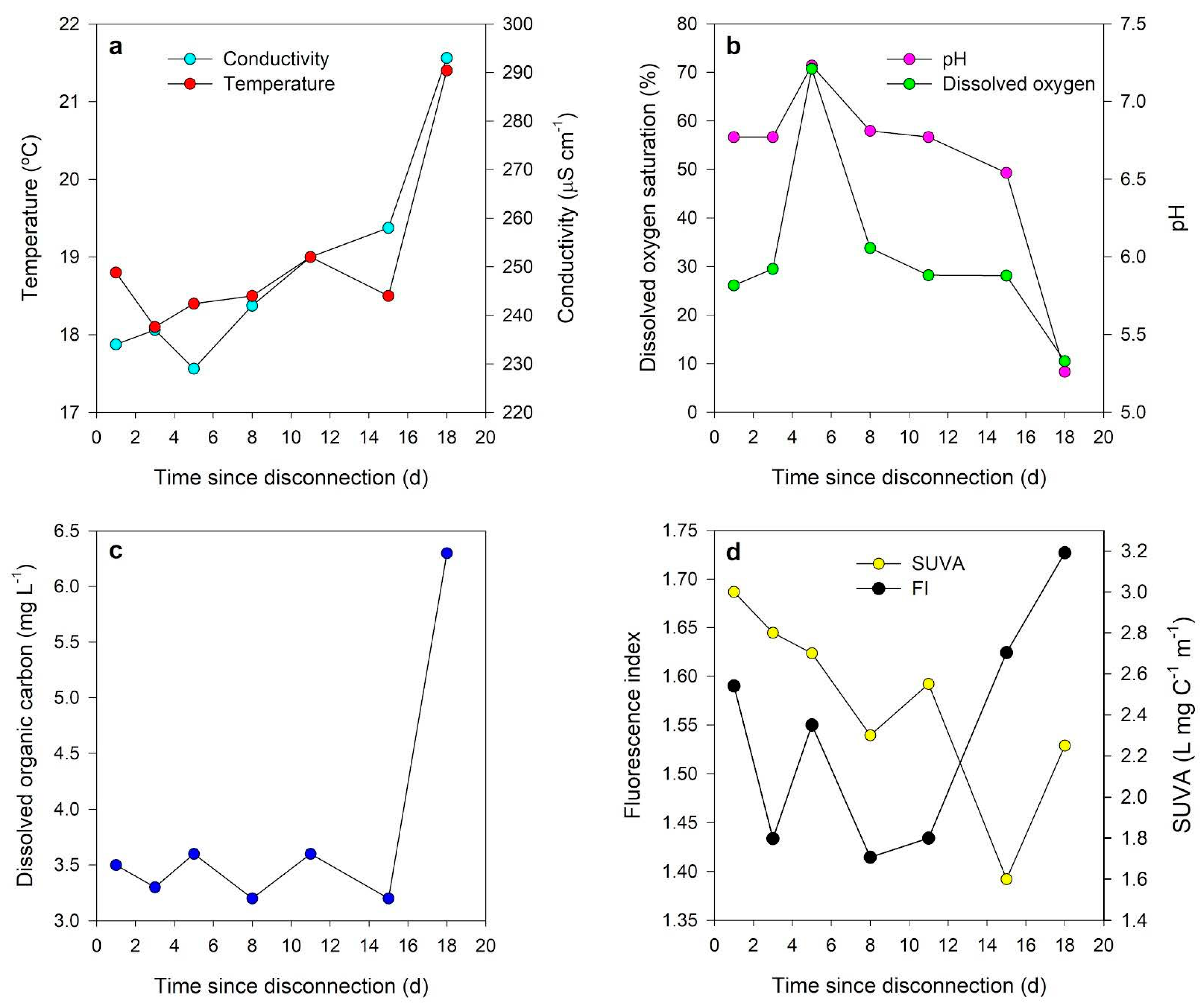
4. Physicochemical and Biogeochemical Characterization of Isolated Pools
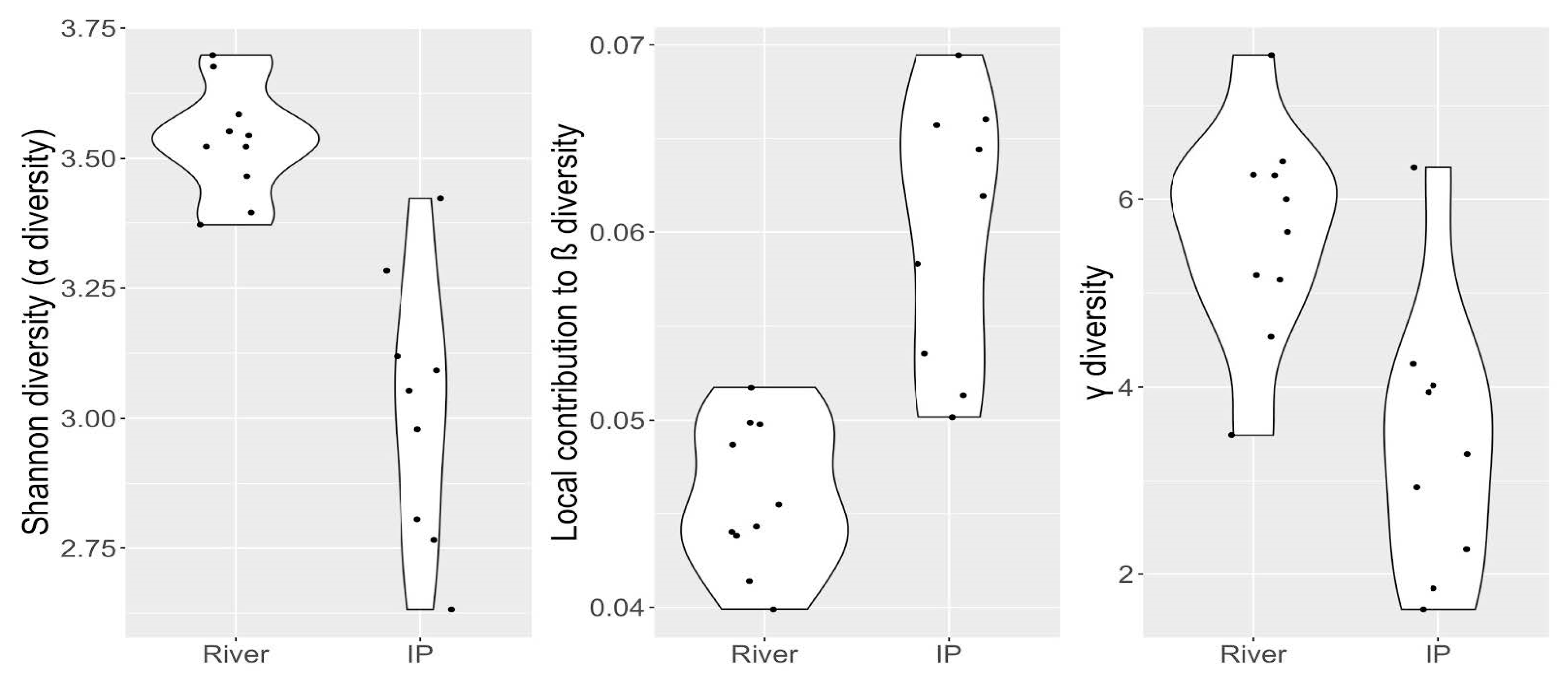
5. Biodiversity Characterization of Isolated Pools
6. Conservation of Isolated Pools: Current Management Needs and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Datry, T.; Larned, S.T.; Tockner, K. Intermittent rivers: A challenge for freshwater ecology. BioScience 2014, 64, 229–235. [Google Scholar] [CrossRef]
- Döll, P.; Schmied, H.M. How is the impact of climate change on river flow. Environ. Res. Lett. 2012, 7, 014037. [Google Scholar] [CrossRef]
- Pumo, D.; Caracciolo, D.; Viola, F.; Noto, L.V. Climate change effects on the hydrological regime of small non-perennial river basins. Sci. Total Environ. 2016, 542, 76–92. [Google Scholar] [CrossRef] [PubMed]
- Datry, T.; Boulton, A.J.; Bonada, N.; Fritz, K.; Leigh, C.; Sauquet, E.; Tockner, K.; Hugueny, B.; Dahm, C.N. Flow intermittence and ecosystem services in rivers of the Anthropocene. J. Appl. Ecol. 2018, 55, 353–364. [Google Scholar] [CrossRef]
- Magand, C.; Alves, M.H.; Calleja, E.; Datry, T.; Dörflinger, G.; England, J.; Gallart, F.; Gómez, R.; Jorda-Capdevila, D.; Marti, E.; et al. Intermittent Rivers and Ephemeral Streams: What Water Managers Need to Know; Technical Report—Cost ACTION CA; European Cooperation in Science and Technology: Brussels, Belgium, 2020. [Google Scholar] [CrossRef]
- Leigh, C.; Boulton, A.J.; Courtwright, J.L.; Fritz, K.; May, C.L.; Walker, R.H.; Datry, T. Ecological research and management of intermittent rivers: An historical review and future directions. Freshw. Biol. 2016, 61, 1181–1199. [Google Scholar] [CrossRef]
- Datry, T.; Bonada, N.; Boulton, A.J. General introduction. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 1–20. [Google Scholar] [CrossRef]
- Datry, T.; Bonada, N.; Boulton, A.J. Conclusions: Recent advances and future prospects in the ecology and management of intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 563–584. [Google Scholar] [CrossRef]
- Skoulikidis, N.T.; Sabater, S.; Datry, T.; Morais, M.M.; Buffagni, A.; Dörflinger, G.; Zogaris, S.; Sánchez-Montoya, M.M.; Bonada, N.; Kelogianni, E.; et al. Non-perennial Mediterranean rivers in Europe: Status, pressures, and challengers for research and management. Sci. Total Environ. 2017, 577, 1–18. [Google Scholar] [CrossRef]
- Gallart, F.; Prat, N.; García-Roger, E.M.; Latron, J.; Rieradevall, M.; Llorens, P.; Barberá, G.G.; Brito, D.; De Girolamo, A.M.; Lo Porto, A.; et al. A novel approach to analysing the regimes of temporary streams in relation to their controls on the composition and structure of aquatic biota. Hydrol. Earth Syst. Sci. 2012, 16, 3165–3182. [Google Scholar] [CrossRef]
- Bonada, N.; Rieradevall, M.; Prat, N. Macroinvertebrate community structure and biological traits related to flow permanence in a Mediterranean river network. Hydrobiologia 2007, 589, 91–106. [Google Scholar] [CrossRef]
- Datry, T.; Pella, H.; Leigh, C.; Bonada, N.; Hugueny, B. A landscape approach to advance intermittent river ecology. Freshw. Biol. 2016, 61, 1200–1213. [Google Scholar] [CrossRef]
- Gallart, F.; Cid, N.; Latron, J.; Llorens, P.; Bonada, N.; Jeuffroy, J.; Jiménez-Argudo, S.M.; Vega, R.M.; Solà, C.; Soria, M.; et al. TREHS: An open-access software tool for investigating and evaluating temporary river regimes as a first step for their ecological status assessment. Sci. Total Environ. 2017, 607, 519–540. [Google Scholar] [CrossRef]
- Baldwin, D.S.; Mitchell, A.M. The effects of drying and re-flooding on the sediment and soil nutrient dynamics of lowland river–floodplain systems: A synthesis. Regul. Rivers Res. Manag. 2000, 16, 457–467. [Google Scholar] [CrossRef]
- Bourke, S.A.; Shanafield, M.; Hedley, P.; Dogramaci, S. A hydrological framework for persistent river pools in semi-arid environments. Hydrol. Earth Syst. Sci. Discuss. 2020. [Google Scholar] [CrossRef]
- Roshier, D.A.; Whetton, P.H.; Allan, R.J.; Robertson, A.I. Distribution and persistence of temporary wetland habitats in arid Australia in relation to climate. Austral Ecol. 2001, 26, 371–384. [Google Scholar] [CrossRef]
- Jocque, M.; Vanschoenwinkel, B.; Brendonck, L. Freshwater rock pools: A review of habitat characteristics, faunal diversity and conservation value. Freshw. Biol. 2010, 55, 1587–1602. [Google Scholar] [CrossRef]
- Holzapfel, G.; Weihs, P.; Rauch, H.P. Use of the Shade-a-lator 6.2 model to assess the shading potential of riparian purple willow (Salix purpurea) coppices on small to medium sized rivers. Ecol. Eng. 2013, 61, 697–705. [Google Scholar] [CrossRef]
- Trimmel, H.; Weihs, P.; Leidinger, D.; Formayer, H.; Kalny, G.; Melcher, A. Can riparian vegetation shade mitigate the expected rise in stream temperatures due to climate change during heat waves in a human-impacted pre-alpine river? Hydrol. Earth Sysy. Sci. 2018, 22, 437–461. [Google Scholar] [CrossRef]
- Leibowitz, S.G.; Brooks, R.T. Hydrology and landscape connectivity of vernal pools. In Science and Conservation of Vernal Pools in Northeastern North America; Calhouh, A.J.K., deMaynadier, P.G., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 31–53. [Google Scholar]
- Lupon, A.; Bernal, S.; Poblador, S.; Martí, E.; Sabater, F. The influence of riparian evapotranspiration on stream hydrology and nitrogen retention in a subhumid Mediterranean catchment. Hydrol. Earth Syst. Sci. 2016, 20, 3831–3842. [Google Scholar] [CrossRef]
- Bonada, N.; Rieradevall, M.; Prat, N.; Resh, V.H. Benthic macroinvertebrate assemblages and macrohabitat connectivity in Mediterranean-climate streams of northern California. J. N. Am. Benthol. Soc. 2006, 25, 32–43. [Google Scholar] [CrossRef]
- Boulton, A.J.; Rolls, R.J.; Jaeger, K.L.; Datry, T. Hydrological connectivity in intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc: Cambridge, MA, USA, 2017; pp. 79–108. [Google Scholar] [CrossRef]
- Jaeger, K.L.; Sutfin, N.A.; Tooth, S.; Michaelides, K.; Singer, M. Geomorphology and sediment regimes of intermittent rivers and ephemeral streams. 2017. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 21–49. [Google Scholar] [CrossRef]
- Chin, A. The morphologic structure of step-pools in mountain streams. Geomorphology 1999, 27, 191–204. [Google Scholar] [CrossRef]
- Chiu, M.-C.; Leigh, C.; Mazor, R.; Cid, N.; Resh, V. Anthropogenic threats to intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 433–454. [Google Scholar] [CrossRef]
- Bourgeau-Chavez, L.L.; Lee, Y.M.; Battaglia, M.; Endres, S.L.; Laubach, Z.M.; Scarbrough, K. Identification of woodland vernal pools with seasonal change PALSAR data for habitat conservation. Remote Sens. 2016, 8, 490. [Google Scholar] [CrossRef]
- Schnack, J.A. Why wetlands? Oecol. Bras. 2006, 10, 127–134. [Google Scholar] [CrossRef]
- Leibowitz, S.G.; Wigington, P.J., Jr.; Schofield, K.A.; Alexander, L.C.; Vanderhoof, M.K.; Golden, H.E. Connectivity of streams and wetlands to downstream waters: An integrated systems framework. J. Am. Water Resour. Assoc. 2018, 54, 298–322. [Google Scholar] [CrossRef] [PubMed]
- Brooks, R.T. A review of basin morphology and pool hydrology of isolated ponded wetlands: Implications for seasonal forest pools of the northeastern United States. Wetl. Ecol. Manag. 2005, 13, 335–348. [Google Scholar] [CrossRef]
- Fellman, J.B.; Dogramaci, S.; Skrzypek, G.; Dodson, W.; Grierson, P.F. Hydrologic control of dissolved organic matter biogeochemistry in pools of a subtropical dryland river. Water Resour. Res. 2011, 47, W06501. [Google Scholar] [CrossRef]
- Brooks, R.T.; Hayashi, M. Depth-area-volume and hydroperiod relationships of ephemeral (vernal) forest pools in southern New England. Wetlands 2002, 22, 247–255. [Google Scholar] [CrossRef]
- Hamilton, S.K.; Bunn, S.E.; Thoms, M.C.; Marshall, J.C. Persistence of aquatic refugia between flow pulses in a dryland river system (Cooper Creek, Australia). Limnol. Oceanogr. 2005, 50, 743–754. [Google Scholar] [CrossRef]
- Schofield, K.A.; Alexander, L.C.; Ridley, C.E.; Vanderhoof, M.K.; Fritz, K.M.; Autrey, B.C.; DeMeester, J.; Kepner, W.G.; Lane, C.R.; Leibowitz, S.; et al. Biota connect aquatic habitats throughout freshwater ecosystem mosaics. JAWRA 2018, 54, 372–399. [Google Scholar] [CrossRef]
- Zeug, S.C.; Winemiller, K.O.; Tarim, S. Response of Brazos River oxbow fish assemblages to patterns of hydrologic connectivity and environmental variability. Trans. Am. Fish. Soc. 2005, 134, 1389–1399. [Google Scholar] [CrossRef]
- Leopold, L.B.; Miller, J.P. Ephemeral Streams. Hydraulic Factors and Their Relation to the Drainage Net; U.S. Geological Survey Professional Paper 282-A; US Government Printing Office: Washington, DC, USA, 1956. Available online: https://pubs.usgs.gov/pp/0282a/report.pdf (accessed on 14 October 2020).
- Dogramaci, S.; Firmani, G.; Hedley, P.; Skrzypek, G.; Grierson, P.F. Evaluating recharge to an ephemeral dryland stream using a hydraulic model and water, chloride and isotope mass balance. J. Hydrol. 2015, 521, 520–532. [Google Scholar] [CrossRef]
- Rau, G.C.; Halloran, L.J.; Cuthbert, M.O.; Andersen, M.S.; Acworth, R.I.; Tellam, J.H. Characterising the dynamics of surface water-groundwater interactions in intermittent and ephemeral streams using streambed thermal signatures. Adv. Water Resour. 2017, 107, 354–369. [Google Scholar] [CrossRef]
- Smakhtin, V.U. Low flow hydrology: A review. J. Hydrol. 2001, 240, 147–186. [Google Scholar] [CrossRef]
- Gallart, F.; Llorens, P.; Latron, J.; Cid, N.; Rieradevall, M.; Prat, N. Validating alternative methodologies to estimate the regime of temporary rivers when flow data are unavailable. Sci. Total Environ. 2016, 565, 1001–1010. [Google Scholar] [CrossRef]
- WMO (World Meteorological Organization). Manual on Stream Gauging. Volume I-Fieldwork; WMO-No. 1044; WMO: Geneva, Switzerland, 2010; 254p, Available online: http://www.wmo.int/pages/prog/hwrp/publications/stream_gauging/1044_Vol_I_en.pdf (accessed on 14 October 2020).
- Zimmer, M.A.; Kaiser, K.E.; Blaszczak, J.R.; Zipper, S.C.; Hammond, J.C.; Fritz, K.M.; Costigan, K.H.; Hosen, J.; Godsey, S.E.; Allen, G.H.; et al. Zero or not? Causes and consequences of zero-flow stream gage readings. WIRES Water 2020, e1436. [Google Scholar] [CrossRef] [PubMed]
- Puckridge, J.T.; Walker, K.F.; Costelloe, J.F. Hydrological persistence and the ecology of dryland rivers. Regul. Rivers Res. Manag. 2000, 16, 385–402. [Google Scholar] [CrossRef]
- Callow, J.N.; Boggs, G. Studying reach-scale spatial hydrology in ungauged catchments. J. Hydrol. 2013, 496, 31–46. [Google Scholar] [CrossRef]
- Fritz, K.M.; Johnson, B.R.; Walters, D.M. Field Operations Manual for Assessing the Hydrologic Permanence and Ecological Condition of Headwater Streams; EPA/600/ R-06/126; US Environmental Protection Agency, Office of Research and Development: Washington, DC, USA, 2006.
- Sefton, C.E.; Parry, S.; England, J.; Angell, G. Visualising and quantifying the variability of hydrological state in intermittent rivers. Fundam. Appl. Limnol. Archiv Hydrobiol. 2019, 193, 21–38. [Google Scholar] [CrossRef]
- Blasch, K.W.; Ferré, T.P.; Christensen, A.H.; Hoffmann, J.P. New field method to determine streamflow timing using electrical resistance sensors. Vadose Zone J. 2002, 1, 289–299. [Google Scholar] [CrossRef]
- Gungle, B. Timing and Duration of Flow in Ephemeral Streams of the Sierra Vista Subwatershed of the Upper San Pedro Basin, Cochise County, Southeastern Arizona; U.S. Geological Survey Scientific Investigations, Report 2005–5190; US Geological Survey: Reston, VA, USA, 2006; 47p. [CrossRef]
- Chapin, T.P.; Todd, A.S.; Zeigler, M.P. Robust, low-cost data loggers for stream temperature, flow intermittency, and relative conductivity monitoring. Water Resour. Res. 2014, 50, 6542–6548. [Google Scholar] [CrossRef]
- Bhamjee, R.; Lindsay, J.B.; Cockburn, J. Monitoring ephemeral headwater streams: A paired-sensor approach. Hydrol. Process. 2016, 30, 888–898. [Google Scholar] [CrossRef]
- Assendelft, R.S.; van Meerveld, H.J. A Low-Cost, multi-Sensor system to monitor temporary stream dynamics in mountainous headwater catchments. Sensors 2019, 19, 4645. [Google Scholar] [CrossRef]
- Straka, M.; Polášek, M.; Syrovátka, V.; Stubbington, R.; Zahrádková, S.; Němejcová, D.; Šikulová, L.; Řezníčková, P.; Opatřilová, L.; Datry, T.; et al. Recognition of stream drying based on benthic macroinvertebrates: A new tool in Central Europe. Ecol. Indic. 2019, 106, 105486. [Google Scholar] [CrossRef]
- Skrzypek, G.; Mydłowski, A.; Dogramaci, S.; Hedley, P.; Gibson, J.J.; Grierson, P.F. Estimation of evaporative loss based on the stable isotope composition of water using Hydrocalculator. J. Hydrol. 2015, 523, 781–789. [Google Scholar] [CrossRef]
- Keller, E.A.; Melhorn, W.M. Rhythmic spacing and origin of pools and riffles. Geol. Soc. Am. Bull. 1978, 89, 723–730. [Google Scholar] [CrossRef]
- Carling, P.A.; Orr, H.G. Morphology of riffle-pool sequences in the river Severn, England. Earth Surf. Process. 2000, 25, 369–384. [Google Scholar] [CrossRef]
- Keller, E.A. Pools, riffles, and meanders: Discussion. Geol. Soc. Am. Bull. 1971, 82, 279–280. [Google Scholar] [CrossRef]
- Richards, K.S. Channel width and the riffle-pool sequence. Geol. Soc. Am. Bull. 1976, 87, 883–890. [Google Scholar] [CrossRef]
- Knighton, D. Fluvial Forms and Processes; Arnold: London, UK, 1984; 218p. [Google Scholar]
- Thompson, D.M. Pool-Riffle sequences. Ref. Modul. Earth Syst. Environ. Sci. 2020. [Google Scholar] [CrossRef]
- Ferguson, R.I. Understanding braiding processes in gravel-bed rivers: Progress and unsolved problems. Geol. Soc. Sp. Publ. 1993, 75, 73–87. [Google Scholar] [CrossRef]
- Lisle, T.E.; Hilton, S. The volume of fine sediment in pools: An index of sediment supply in gravel-bed streams. J. Am. Water Resour. Assoc. 1992, 28, 371–383. [Google Scholar] [CrossRef]
- Lisle, T.E. Using ‘Residual Depths’ to Monitor Pool Depths Independently of Discharge; U.S. Forest Service Research Note PSW-394; U.S. Department of Agriculture, Forest Service, Pacific Southwest Forest and Range Experiment Station: Berkeley, CA, USA, 1987.
- Beesley, L. Environmental Stability: Its Role in Structuring Fish Communities and Life History Strategies in the Fortescue River, Western Australia. Ph.D. Thesis, The University of Western Australia, Perth, Australia, 2006. [Google Scholar]
- Marimon, M. Effects of Flow Intermittence on Fish Fauna in Mediterranean-Climate Rivers. Master’s Thesis, University of Barcelona, Barcelona, Spain, 2018. Available online: http://www.ub.edu/fem/docs/treballs/TFM_Marc_Marimon.pdf (accessed on 14 October 2020).
- Gómez, R.; Arce, M.I.; Baldwin, D.S.; Dahm, C.N. Water physicochemistry in intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 109–134. [Google Scholar] [CrossRef]
- von Schiller, D.; Bernal, S.; Dahm, C.N.; Martí, E. Nutrient and Organic Matter Dynamics in Intermittent Rivers and Ephemeral Streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 135–160. [Google Scholar] [CrossRef]
- Bestland, E.; George, A.; Green, G.; Olifent, V.; Mackay, D.; Whalen, M. Groundwater dependent pools in seasonal and permanent streams in the Clare Valley of South Australia. J. Hydrol. Reg. Stud. 2017, 9, 216–235. [Google Scholar] [CrossRef]
- Siebers, A.R.; Pettit, N.E.; Skrzypek, G.; Fellman, J.B.; Dogramaci, S.; Grierson, P.F. Alluvial ground water influences dissolved organic matter biogeochemistry of pools within intermittent dryland streams. Freshw. Biol. 2016, 61, 1228–1241. [Google Scholar] [CrossRef]
- Larned, S.T.; Datry, T.; Arscott, D.B.; Tockner, K. Emerging concepts in temporary-river ecology. Freshw. Biol. 2010, 55, 717–738. [Google Scholar] [CrossRef]
- Fisher, S.G.; Grimm, N.B.; Martí, E.; Holmes, R.M.; Jones, J.B., Jr. Material spiraling in stream corridors: A telescoping ecosystem model. Ecosystems 1998, 1, 19–34. [Google Scholar] [CrossRef]
- Bernal, S.; von Schiller, D.; Sabater, F.; Martí, E. Hydrological extremes modulate nutrient dynamics in mediterranean climate streams across different spatial scales. Hydrobiologia 2013, 719, 31–42. [Google Scholar] [CrossRef]
- Harjung, A.; Sabater, F.; Butturini, A. Hydrological connectivity drives dissolved organic matter processing in an intermittent stream. Limnologica 2018, 68, 71–81. [Google Scholar] [CrossRef]
- Acuña, V.; Giorgi, A.; Muñoz, I.; Sabater, F.; Sabater, S. Meteorological and riparian influences on organic matter dynamics in a forested Mediterranean stream. J. N. Am. Benthol. Soc. 2007, 26, 54–69. [Google Scholar] [CrossRef]
- Turner, L.; Erskine, W.D. Variability in the development, persistence and breakdown of thermal, oxygen and salt stratification on regulated rivers of southeastern Australia. River Res. Appl. 2005, 21, 151–168. [Google Scholar] [CrossRef]
- Overton, I.C.; Colloff, M.J.; Doody, T.M.; Henderson, B.; Cuddy, S.M. Ecological Outcomes of Flow Regimes in the Murray-Darling Basin; Report prepared for the National Water Commission by CSIRO Water for a Healthy Country Flagship; CSIRO: Canberra, Australia, 2009; 422p. [Google Scholar] [CrossRef]
- Gómez, R.; García, V.; Vidal-Abarca, R.; Suárez, L. Effect of intermittency on N spatial variability in an arid Mediterranean stream. J. N. Am. Benthol. Soc. 2009, 28, 572–583. [Google Scholar] [CrossRef]
- Seaman, M.T.; Avenant, M.F.; Watson, M.; King, J.; Armour, J.; Barker, C.H.; Dollar, E.; Du Preez, P.J.; Hughes, D.; Rossouw, L.; et al. Developing a Method for Determining the Environmental Water Requirements for Non-Perennial Systems; Report Number: WRC TT459/10; Centre for Environmental Management University of the Free State: Bloemfontein, South Africa, 2009. [Google Scholar]
- von Schiller, D.; Acuña, V.; Graeber, D.; Martí, E.; Ribot, M.; Sabater, S.; Timoner, X.; Tockner, K. Contraction, fragmentation and expansion dynamics determine nutrient availability in a Mediterranean forest stream. Aquat. Sci. 2011, 73, 485–497. [Google Scholar] [CrossRef]
- Day, J.A.; Malan, H.L.; Malijani, E.; Abegunde, A.P. Water quality in non-perennial rivers. Water SA 2019, 45, 487–500. [Google Scholar] [CrossRef]
- Zale, A.V.; Leslie, D.M., Jr.; Fisher, W.L.; Merrifield, S.G. The Physicochemistry, Flora, and Fauna of Intermittent Prairie Streams: A Review of the Literature; US Department of the Interior, Fish and Wildlife Service: Washington, DC, USA, 1989; Volume 89.
- Williams, D.D. The Biology of Temporary Waters; Oxford University Press: Oxford, UK, 2006. [Google Scholar] [CrossRef]
- Lillebø, A.I.; Morais, M.; Guilherme, P.; Fonseca, R.; Serafim, A.; Neves, R. Nutrient dynamics in Mediterranean temporary streams: A case study in Pardiela catchment (Degebe River, Portugal). Limnologica 2007, 37, 337–348. [Google Scholar] [CrossRef]
- Burrows, R.M.; Rutlidge, H.; Bond, N.R.; Eberhard, S.M.; Auhl, A.; Andersen, M.S.; Valdez, D.G.; Kennard, M.J. High rates of organic carbon processing in the hyporheic zone of intermittent streams. Sci. Rep. 2017, 7, 13198. [Google Scholar] [CrossRef]
- Fazi, S.; Vázquez, E.; Casamayor, E.O.; Amalfitano, S.; Butturini, A. Stream hydrological fragmentation drives bacterioplankton community composition. PLoS ONE 2013, 8, e64109. [Google Scholar] [CrossRef]
- Sabater, S.; Timoner, X.; Borrego, C.; Acuña, V. Stream biofilm responses to flow intermittency: From cells to ecosystems. Front. Environ. Sci. 2016, 4, 14. [Google Scholar] [CrossRef]
- Freixa, A.; Ejarque, E.; Crognale, S.; Amalfitano, S.; Fazi, S.; Butturini, A.; Romaní, A.M. Sediment microbial communities rely on different dissolved organic matter sources along a Mediterranean river continuum. Limnol. Oceanogr. 2016, 61, 1389–1405. [Google Scholar] [CrossRef]
- Baker, M.A.; Dahm, C.N.; Valett, H.M. Anoxia, anaerobic metabolism, and biogeochemistry of the stream-water–ground-water interface. In Streams and Ground Waters; Jones, J.B., Mulholland, P.J., Eds.; Academic Press: San Diego, CA, USA, 2000; pp. 259–283. [Google Scholar] [CrossRef]
- Baldwin, D.S.; Rees, G.N.; Wilson, J.S.; Colloff, M.J.; Whitworth, K.L.; Pitman, T.L.; Wallace, T.A. Provisioning of bioavailable carbon between the wet and dry phases in a semi-arid floodplain. Oecologia 2013, 172, 539–550. [Google Scholar] [CrossRef]
- Boulton, A.J.; Lake, P.S. Benthic organic matter and detritivorous macroinvertebrates in two intermittent streams in south-eastern Australia. Hydrobiologia 1992, 241, 107–118. [Google Scholar] [CrossRef]
- Acuña, V.; Muñoz, I.; Giorgi, A.; Omella, M.; Sabater, F.; Sabater, S. Drought and postdrought recovery cycles in an intermittent Mediterranean stream: Structural and functional aspects. J. N. Am. Benthol. Soc. 2005, 24, 919–933. [Google Scholar] [CrossRef]
- Romaní, A.M.; Amalfitano, S.; Artigas, J.; Fazi, S.; Sabater, S.; Timoner, X.; Ylla, I.; Zoppini, A. Microbial biofilm structure and organic matter use in mediterranean streams. Hydrobiologia 2013, 719, 43–58. [Google Scholar] [CrossRef]
- Pattee, E.; Maamri, A.; Chergui, H. Leaf litter processing and its agents in a temporary Moroccan river. Int. Ver. Theor. Angew. Limnol. Verh. 2001, 27, 3054–3057. [Google Scholar] [CrossRef]
- Schlief, J.; Mutz, M. Leaf decay processes during and after a supra-seasonal hydrological drought in a temperate lowland stream. Int. Rev. Hydrobiol. 2011, 96, 633–655. [Google Scholar] [CrossRef]
- Abril, M.; Muñoz, I.; Menéndez, M. Heterogeneity in leaf litter decomposition in a temporary Mediterranean stream during flow fragmentation. Sci. Total Environ. 2016, 553, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Arias-Real, R.; Muñoz, I.; Gutierrez-Cánovas, C.; Granados, V.; Lopez-Laseras, P.; Menéndez, M. Subsurface zones in intermittent streams are hotspots of microbial decomposition during the non-flow period. Sci. Total Environ. 2020, 703, 135485. [Google Scholar] [CrossRef] [PubMed]
- Canhoto, C.; Laranjeira, C. Leachates of Eucalyptus globulus in intermittent streams affect water parameters and invertebrates. Int. Rev. Hydrobiol. 2007, 92, 173–182. [Google Scholar] [CrossRef]
- Casas-Ruiz, J.P.; Tittel, J.; von Schiller, D.; Catalán, N.; Obrador, B.; Gómez-Gener, L.; Zwirnmann, E.; Sabater, S.; Marcé, R. Drought-induced discontinuities in the source and degradation of dissolved organic matter in a Mediterranean river. Biogeochemistry 2016, 127, 125–139. [Google Scholar] [CrossRef]
- Dahm, V.; Hering, D.; Nemitz, D.; Graf, W.; Schmidt-Kloiber, A.; Leitner, P.; Melcher, A.; Feld, C.K. Effects of physico-chemistry, land use and hydromorphology on three riverine organism groups: A comparative analysis with monitoring data from Germany and Austria. Hydrobiologia 2013, 704, 389–415. [Google Scholar] [CrossRef]
- Vazquez, E.; Amalfitano, S.; Fazi, S.; Butturini, A. Dissolved organic matter composition in a fragmented Mediterranean fluvial system under severe drought conditions. Biogeochemistry 2011, 102, 59–72. [Google Scholar] [CrossRef]
- von Schiller, D.; Graeber, D.; Ribot, M.; Timoner, X.; Acuña, V.; Martí, E.; Sabater, S.; Tockner, K. Hydrological transitions drive dissolved organic matter quantity and composition in a temporary Mediterranean stream. Biogeochemistry 2015, 123, 429–446. [Google Scholar] [CrossRef]
- Granados, V.; Gutiérrez-Cánovas, C.; Arias-Real, R.; Obrador, B.; Harjung, A.; Butturini, A. The interruption of longitudinal hydrological connectivity causes delayed responses in dissolved organic matter. Sci. Total Environ. 2020, 713, 136619. [Google Scholar] [CrossRef]
- Catalán, N.; Casas-Ruiz, J.P.; von Schiller, D.; Proia, L.; Obrador, B.; Zwirnmann, E.; Marcé, R. Biodegradation kinetics of dissolved organic matter chromatographic fractions in an intermittent river. J. Geophys. Res. Biogeosci. 2017, 122. [Google Scholar] [CrossRef]
- Gómez-Gener, L.; Obrador, B.; von Schiller, D.; Marcé, R.; Casas-Ruiz, J.P.; Proia, L.; Acuña, V.; Catalán, N.; Muñoz, I.; Koschorreck, M. Hot spots for carbon emissions from Mediterranean fluvial networks during summer drought. Biogeochemistry 2015, 125, 409–426. [Google Scholar] [CrossRef]
- Romaní, A.M.; Chauvet, E.; Febria, C.; Mora-Gómez, J.; Risse-Buhl, U.; Timoner, X.; Weitere, M.; Zeglin, L. The biota of intermittent rivers and ephemeral streams: Prokaryotes, fungi, and protozoans. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 161–188. [Google Scholar] [CrossRef]
- Sabater, S.; Timoner, X.; Bornette, G.; De Wilde, M.; Stromberg, J.C.; Stella, J.C. The biota of intermittent rivers and ephemeral streams: Algae and vascular plants. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 189–216. [Google Scholar] [CrossRef]
- Stubbington, R.; Bogan, M.T.; Bonada, N.; Boulton, A.J.; Datry, T.; Leigh, C.; Vander Vorste, R. The biota of intermittent rivers and ephemeral streams: Aquatic invertebrates. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 217–243. [Google Scholar] [CrossRef]
- Kerezsy, A.; Gido, K.; Magalhaes, M.F.; Skelton, P.H. The biota of intermittent rivers and ephemeral streams: Fishes. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 273–298. [Google Scholar] [CrossRef]
- Mora-Gómez, J.; Elosegi, A.; Duarte, S.; Cássio, F.; Pascoal, C.; Romaní, A.M. Differences in the sensitivity of fungi and bacteria to season and invertebrates affect leaf litter decomposition in a Mediterranean stream. FEMS Microbiol. Ecol. 2016, 92. [Google Scholar] [CrossRef] [PubMed]
- Drummond, L.R.; McIntosh, A.R.; Larned, S.T. Invertebrate community dynamics and insect emergence in response to pool drying in a temporary river. Freshw. Biol. 2015, 60, 1596–1612. [Google Scholar] [CrossRef]
- Aparicio, E.; Sostoa, A. Pattern of movements of adult Barbus haasi in a small Mediterranean stream. J. Fish Biol. 1999, 55, 1086–1095. [Google Scholar] [CrossRef]
- Hill, M.J.; Milner, V.S. Ponding in intermittent streams: A refuge for lotic taxa and a habitat for newly colonising taxa? Sci. Total Environ. 2018, 628-629, 1308–1316. [Google Scholar] [CrossRef]
- Van Leeuwen, C.H.; van der Velde, G. Prerequisites for flying snails: External transport potential of aquatic snails by waterbirds. Freshw. Sci. 2012, 31, 963–972. [Google Scholar] [CrossRef]
- Arthington, A.H.; Balcombe, S.R. Extreme flow variability and the ‘boom and bust’ ecology of fish in arid-zone floodplain rivers: A case history with implications for environmental flows, conservation and management. Ecohydrology 2011, 4, 708–720. [Google Scholar] [CrossRef]
- Ribera, I.; Foster, G.N.; Vogler, A.P. Does habitat use explain large scale species richness patterns of aquatic beetles in Europe? Ecography 2003, 26, 145–152. [Google Scholar] [CrossRef]
- Bogan, M.T.; Chester, E.T.; Datry, T.; Murphy, A.L.; Robson, B.J.; Ruhí, A.; Stubbington, R.; Whitney, J.E. Resistance, resilience, and community recovery in intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 349–376. [Google Scholar] [CrossRef]
- Bazzanti, M.; Seminara, M.; Baldoni, S. Chironomids (Diptera: Chironomidae) from three temporary ponds of different wet phase duration in central Italy. J. Freshw. Ecol. 1997, 12, 89–99. [Google Scholar] [CrossRef]
- Woelfle-Erskine, C.; Larsen, L.G.; Carlson, S.M. Abiotic habitat thresholds for salmonid over-summer survival in intermittent streams. Ecosphere 2017, 8, e01645. [Google Scholar] [CrossRef]
- Anusa, A.; Ndagurwa, H.G.T.; Magadza, C.H.D. The influence of pool size on species diversity and water chemistry in temporary rock pools on Domboshawa Mountain, northern Zimbawe. Afr. J. Aquat. Sci. 2012, 37, 89–99. [Google Scholar] [CrossRef]
- Angermeier, P.L.; Schlosser, I.J. Species-area relationship for stream fishes. Ecology 1989, 70, 1450–1462. [Google Scholar] [CrossRef]
- Taylor, C.M. Fish species richness and incidence patterns in isolated and connected stream pools: Effects of pool volume and spatial position. Oecologia 1997, 110, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Arthington, A.H.; Olden, J.D.; Balcombe, S.R.; Thoms, M.C. Multi-scale environmental factors explain fish losses and refuge quality in drying waterholes of Cooper Creek, an Australian arid-zone river. Mar. Freshw. Res. 2010, 61, 842–856. [Google Scholar] [CrossRef]
- García-Roger, E.M.; Sánchez-Montoya, M.M.; Gómez, R.; Suárez, M.L.; Vidal-Abarca, M.R.; Latron, J.; Rieradevall, M.; Prat, N. Do seasonal changes in habitat features influence aquatic macroinvertebrate assemblages in perennial versus temporary Mediterranean streams? Aquat. Sci. 2011, 73, 567–579. [Google Scholar] [CrossRef]
- Morey, S.R. Pool duration influences age and body mass at metamorphosis in the western spadefoot toad: Implications for vernal pool conservation. In Ecology, Conservation, and Management of Vernal Pool Ecosystem, Proceedings from a 1996 Conference, Sacramento, CA, USA, 1996; Witham, C.W., Bauder, E.T., Belk, D., Ferren, W.R., Jr., Ornduff, R., Eds.; California Native Plant Society: Sacramento, CA, USA, 1998; pp. 86–91. [Google Scholar]
- Bonada, N.; Carlson, S.M.; Datry, T.; Finn, D.S.; Leigh, C.; Lytle, D.A.; Monaghan, M.T.; Tedesco, P.A. Genetic, evolutionary, and biogeographical processes in intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 405–431. [Google Scholar] [CrossRef]
- McIntosh, A.R.; Leigh, C.; Boersma, K.S.; McHugh, P.A.; Febria, C.; García-Berthou, E. Food webs and trophic interactions in intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 323–347. [Google Scholar] [CrossRef]
- DuBose, T.P.; Ashford, K.; Vaughn, C.C. Freshwater mussels increase survival of largemouth bass (Micropterus salmoides) in drying pools. Ecol. Freshw. Fish 2019, 29, 220–229. [Google Scholar] [CrossRef]
- Williams, L.R.; Taylor, C.M.; Warren, M.L., Jr. Influence of fish predation on assemblage structure of macroinvertebrates in an intermittent stream. Trans. Am. Fish. Soc. 2003, 132, 120–130. [Google Scholar] [CrossRef]
- Driver, L.J.; Hoeinghaus, D.J. Fish metacommunity responses to experimental drought are determined by habitat heterogeneity and connectivity. Freshw. Biol. 2016, 61, 533–548. [Google Scholar] [CrossRef]
- Vanschoenwinkel, B.; Hulsmans, A.; De Roeck, E.; De Vries, C.; Seaman, M.; Brendonck, L. Community structure in temporary freshwater pools: Disentangling the effects of habitat size and hydroregime. Freshw. Biol. 2009, 54, 1487–1500. [Google Scholar] [CrossRef]
- Tonkin, J.D.; Stoll, S.; Sundermann, A.; Haase, P. Dispersal distance and the pool of taxa, but not barriers, determine the colonisation of restored river reaches by benthic invertebrates. Freshw. Biol. 2014, 59, 1843–1855. [Google Scholar] [CrossRef]
- Heino, J.; Melo, A.S.; Siqueira, T.; Soininen, J.; Valanko, S.; Bini, L.M. Metacommunity organisation, spatial extent and dispersal in aquatic systems: Patterns, processes and prospects. Freshw. Biol. 2015, 60, 845–869. [Google Scholar] [CrossRef]
- Chase, J.M. Drought mediates the importance of stochastic community assembly. Proc. Natl. Acad. Sci. USA 2007, 104, 17430–17434. [Google Scholar] [CrossRef]
- Cañedo-Argüelles, M.; Bogan, M.T.; Lytle, D.A.; Prat, N. Are Chironomidae (Diptera) good indicators of water scarcity? Dryland streams as a case study. Ecol. Indic. 2016, 71, 155–162. [Google Scholar] [CrossRef]
- Cañedo-Argüelles, M.; Boersma, K.S.; Bogan, M.T.; Olden, J.D.; Phillipsen, I.; Schriever, T.A.; Lytle, D.A. Dispersal strength determines meta-community structure in a dendritic riverine network. J. Biogeogr. 2015, 42, 778–790. [Google Scholar] [CrossRef]
- Phillipsen, I.C.; Lytle, D.A. Aquatic insects in a sea of desert: Population genetic structure is shaped by limited dispersal in a naturally fragmented landscape. Ecography 2013, 36, 731–743. [Google Scholar] [CrossRef]
- Tornés, E.; Ruhí, A. Flow intermittency decreases nestedness and specialisation of diatom communities in Mediterranean rivers. Freshw. Biol. 2013, 58, 2555–2566. [Google Scholar] [CrossRef]
- Ruhí, A.; Datry, T.; Sabo, J.L. Interpreting beta-diversity components over time to conserve metacommunities in highly dynamic ecosystems. Conserv. Biol. 2017, 31, 1459–1468. [Google Scholar] [CrossRef]
- Rogosch, J.S.; Olden, J.D. Dynamic contributions of intermittent and perennial streams to fish beta diversity in dryland rivers. J. Biogeogr. 2019, 46, 2311–2322. [Google Scholar] [CrossRef]
- Ebert, T.A.; Balko, M.L. Temporary pools as islands in space and in time: The biota of vernal pools in San Diego, Southern California, USA. Arch. Hydrobiol. 1987, 110, 101–123. [Google Scholar]
- Boix, D.; Sala, J.; Moreno-Amich, R. The faunal composition of Espolla pond (NE Iberian peninsula): The neglected biodiversity of temporary waters. Wetlands 2001, 21, 577–592. [Google Scholar] [CrossRef]
- Lumbreras, A.; Marques, J.; Belo, A.F.; Cristo, M.; Fernandes, M.; Galioto, D.; Machado, M.; Mira, A.; Sá-Sousa, L.G.; Pinto-Cruz, C. Assessing the conservation status of Mediterranean temporary ponds using biodiversity: A new tool for practitioners. Hydrobiologia 2016, 782, 187–199. [Google Scholar] [CrossRef]
- Rodríguez-Lozano, P.; Woelfle-Erskine, C.; Bogan, M.T.; Carlson, S.M. Are non-perennial rivers considered as valuable and worthy of conservation as perennial rivers? Sustainability 2020, 12, 5782. [Google Scholar] [CrossRef]
- Hermoso, V.; Ward, D.P.; Kennard, M.J. Prioritizing refugia for freshwater biodiversity conservation in highly seasonal ecosystems. Divers. Distrib. 2013, 19, 1031–1042. [Google Scholar] [CrossRef]
- Nicolet, P.; Biggs, J.; Fox, G.; Hodson, M.J.; Reynolds, C.; Whitfield, M.; Williams, P. The wetland plant and macroinvertebrate assemblages of temporary ponds in England and Wales. Biol. Conserv. 2004, 120, 261–278. [Google Scholar] [CrossRef]
- Lukács, B.A.; Sramkó, G.; Molnár, A. Plant diversity and conservation value of continental temporary pools. Biol. Conserv. 2013, 158, 393–400. [Google Scholar] [CrossRef]
- IPBES. Global Assessment Report on Biodiversity and Ecosystem Services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services; Brondizio, E.S., Settele, J., Díaz, S., Ngo, H.T., Eds.; IPBES Secretariat: Bonn, Germany, 2019. [Google Scholar]
- Rhazi, L.; Grillas, P.; Saber, E.R.; Rhazi, M.; Brendonck, L.; Waterkeyn, A. Vegetation of Mediterranean temporary pools: A fading jewel? Hydrobiologia 2012, 689, 23–36. [Google Scholar] [CrossRef]
- Van den Broeck, M.; Waterkeyn, A.; Rhazi, L.; Grillas, P.; Brendonck, L. Assessing the ecological integrity of endorheic wetlands, with focus on Mediterranean temporary ponds. Ecol. Indic. 2015, 54, 1–11. [Google Scholar] [CrossRef]
- Biggs, J.; Von Fumetti, S.; Kelly-Quinn, M. The importance of small waterbodies for biodiversity and ecosystem services: Implications for policy makers. Hydrobiologia 2017, 793, 3–39. [Google Scholar] [CrossRef]
- Hill, M.J.; Hassall, C.; Oertli, B.; Fahrig, L.; Robson, B.J.; Biggs, J.; Samways, M.J.; Usio, N.; Takamura, N.; Krishnaswamy, J.; et al. New policy directions for global pond conservation. Conserv. Lett. 2018, 11, e12447. [Google Scholar] [CrossRef]
- Céréghino, R.; Biggs, J.; Oertli, B.; Declerck, S. The ecology of European ponds: Defining the characteristics of a neglected freshwater habitat. Hydrobiologia 2008, 597, 1–6. [Google Scholar] [CrossRef]
- Hermoso, V.; Cattarino, L.; Kennard, M.J.; Watts, M.; Linke, S. Catchment zoning for freshwater conservation: Refining plans to enhance action on the ground. J. Appl. Ecol. 2015, 52, 940–949. [Google Scholar] [CrossRef]
- Soria, M.; Gutiérrez-Cánovas, C.; Bonada, N.; Acosta, R.; Rodríguez-Lozano, P.; Fortuño, P.; Burgazzi, G.; Vinyoles, D.; Gallart, F.; Latron, J.; et al. Natural disturbances can produce misleading bioassessment results: Identifying metrics to detect anthropogenic impacts in intermittent rivers. J. Appl. Ecol. 2020, 57, 283–295. [Google Scholar] [CrossRef]
- Cid, N.; Bonada, N.; Carlson, S.; Grantham, T.E.; Gasith, A.; Resh, V.H. High variability is a defining component of Mediterranean-climate rivers and their biota. Water 2017, 9, 52. [Google Scholar] [CrossRef]
- Fritz, K.; Cid, N.; Autrey, B. Governance, legislation, and protection of intermittent rivers and ephemeral streams. In Intermittent Rivers and Ephemeral Streams: Ecology and Management; Datry, T., Bonada, N., Boulton, A.J., Eds.; Elsevier, Inc.: Cambridge, MA, USA, 2017; pp. 477–507. [Google Scholar] [CrossRef]
- Stubbington, R.; Chadd, R.; Cid, N.; Csabai, Z.; Milisa, M.; Morais, M.; Munné, A.; Paril, P.; Pesic, V.; Tziortzis, I.; et al. Biomonitoring of intermittent rivers and ephemeral streams in Europe: Current practice and priorities to enhance ecological status assessments. Sci. Total Environ. 2018, 618, 1096–1113. [Google Scholar] [CrossRef] [PubMed]
- Cid, N.; Bonada, N.; Heino, J.; Cañedo-Argüelles, M.; Crabot, J.; Sarremejane, R.; Soininen, J.; Stubbington, R.; Datry, T. A metacommunity approach to improve biological assessments in highly dynamic freshwater ecosystems. BioScience 2020, 70, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Pires, D.F.; Beja, P.; Magalhães, M.F. Out of pools: Movement patterns of mediterranean stream fish in relation to dry season refugia. River Res. Appl. 2014, 30, 1269–1280. [Google Scholar] [CrossRef]
- Mesnage, V.; Bonneville, S.; Laignel, B.; Lefebvre, D.; Dupont, J.P.; Mikes, D. Filling of a wetland (Seine estuary, France): Natural eutrophication or anthropogenic process? A sedimentological and geochemical study of wetland organic sediments. Hydrobiologia 2002, 475/476, 423–435. [Google Scholar] [CrossRef]
- Sánchez-Carrillo, S.; Angeler, D.G.; Álvarez-Cobelas, M.; Sánchez-Andrés, R. Freshwater wetland eutrophication. In Eutrophication: Causes, Consequences and Control; Ansari, A., Singh Gill, S., Lanza, G., Rast, W., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 195–210. [Google Scholar] [CrossRef]
- Costelloe, J.F.; Western, A.W.; Irvine, E.C. Development of hypersaline groundwater in alluvial aquifers of ephemeral rivers. In MODSIM 2007: International Congress on Modelling and Simulation; Oxley, L., Kulasiri, D., Eds.; Modelling and Simulation Society of Australia and New Zealand: Christchurch, New Zealand, 2007; pp. 1416–1422. [Google Scholar]
- Luthy, R.G.; Sedlak, D.L.; Plumlee, M.H.; Austin, D.; Resh, V.H. Wastewater-effluent-dominated streams as ecosystem-management tools in a drier climate. Front. Ecol. Environ. 2015, 13, 477–485. [Google Scholar] [CrossRef]
- Poff, N.L.; Richter, B.D.; Arthington, A.H.; Bunn, S.E.; Naiman, R.J.; Kendy, E.; Acreman, M.; Apse, C.; Bledsoe, B.P.; Freeman, M.C.; et al. The ecological limits of hydrologic alteration (ELOHA): A new framework for developing regional environmental flow standards. Freshw. Biol. 2010, 55, 147–170. [Google Scholar] [CrossRef]
- Suárez-Alonso, M.L.; Vidal-Abarca, M.R. Aplicación del índice de calidad del bosque de ribera, QBR, a los cauces fluviales de la cuenca del río Segura. Tecnol. Agua 2000, 201, 33–45. [Google Scholar]
- Sala, J.; Gascón, S.; Boix, D.; Gesti, J.; Quintana, X. Proposal of a rapid methodology to assess the conservation status of Mediterranean wetlands and its application in Catalunya (NE Iberian peninsula). Arch. Sci. 2004, 57, 141–152. [Google Scholar]
- Quintana, X.D.; Cañedo-Argüelles, M.; Nebra, A.; Gascón, S.; Rieradevall, M.; Caiola, N.; Sala, J.; Ibàñez, C.; Sánchez-Millaruelo, N.; Boix, D. New tools to analyse the ecological status of Mediterranean wetlands and shallow lakes. In Experiences from Surface Water Quality Monitoring; Munné, A., Ginebreda, A., Prat, N., Eds.; Springer: Cham, Switzerland, 2015; pp. 171–199. [Google Scholar] [CrossRef]
- Angeler, D.G.; García, G. Using emergence from soil propagule banks as indicators of ecological integrity in wetlands: Advantages and limitations. J. N. Am. Benthol. Soc. 2005, 24, 740–752. [Google Scholar] [CrossRef]
- Hajibabaei, M.; Shokralla, S.; Zhou, X.; Singer, G.; Baird, D.J. Environmental barcoding: A next-generation sequencing approach for biomonitoring applications using river benthos. PLoS ONE 2011, 6, e17497. [Google Scholar] [CrossRef] [PubMed]
- Elbrecht, V.; Vamos, E.E.; Meissner, K.; Aroviita, J.; Leese, F. Assessing strengths and weaknesses of DNA metabarcoding-based macroinvertebrate identification for routine stream monitoring. Methods Ecol. Evol. 2017, 8, 1265–1275. [Google Scholar] [CrossRef]
- Harper, L.R.; Lawson Handley, L.; Carpenter, A.I.; Ghazali, M.; Di Muri, C.; Macgregor, C.J.; Logan, T.W.; Law, A.; Breithaupt, T.; Read, D.S.; et al. Environmental DNA (eDNA) metabarcoding of pond water as a tool to survey conservation and management priority mammals. Biol. Conserv. 2019, 238, 108225. [Google Scholar] [CrossRef]
- Jerde, C.L.; Wilson, E.A.; Dressler, T.L. Measuring global fish species richness with eDNA metabarcoding. Mol. Ecol. Resour. 2019, 19, 19–22. [Google Scholar] [CrossRef]
- Furlan, E.M.; Davis, J.; Duncan, R.P. Identifying error and accurately interpreting environmental DNA metabarcoding results: A case study to detect vertebrates at arid zone waterholes. Mol. Ecol. Resour. 2020, 20, 1259–1276. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonada, N.; Cañedo-Argüelles, M.; Gallart, F.; von Schiller, D.; Fortuño, P.; Latron, J.; Llorens, P.; Múrria, C.; Soria, M.; Vinyoles, D.; et al. Conservation and Management of Isolated Pools in Temporary Rivers. Water 2020, 12, 2870. https://doi.org/10.3390/w12102870
Bonada N, Cañedo-Argüelles M, Gallart F, von Schiller D, Fortuño P, Latron J, Llorens P, Múrria C, Soria M, Vinyoles D, et al. Conservation and Management of Isolated Pools in Temporary Rivers. Water. 2020; 12(10):2870. https://doi.org/10.3390/w12102870
Chicago/Turabian StyleBonada, Núria, Miguel Cañedo-Argüelles, Francesc Gallart, Daniel von Schiller, Pau Fortuño, Jérôme Latron, Pilar Llorens, Cesc Múrria, Maria Soria, Dolors Vinyoles, and et al. 2020. "Conservation and Management of Isolated Pools in Temporary Rivers" Water 12, no. 10: 2870. https://doi.org/10.3390/w12102870
APA StyleBonada, N., Cañedo-Argüelles, M., Gallart, F., von Schiller, D., Fortuño, P., Latron, J., Llorens, P., Múrria, C., Soria, M., Vinyoles, D., & Cid, N. (2020). Conservation and Management of Isolated Pools in Temporary Rivers. Water, 12(10), 2870. https://doi.org/10.3390/w12102870






