Removal of COD and SO42− from Oil Refinery Wastewater Using a Photo-Catalytic System—Comparing TiO2 and Zeolite Efficiencies
Abstract
1. Introduction
2. Materials and Methods
2.1. Effluent Sample and Analytical Methods
2.2. Chemicals Used
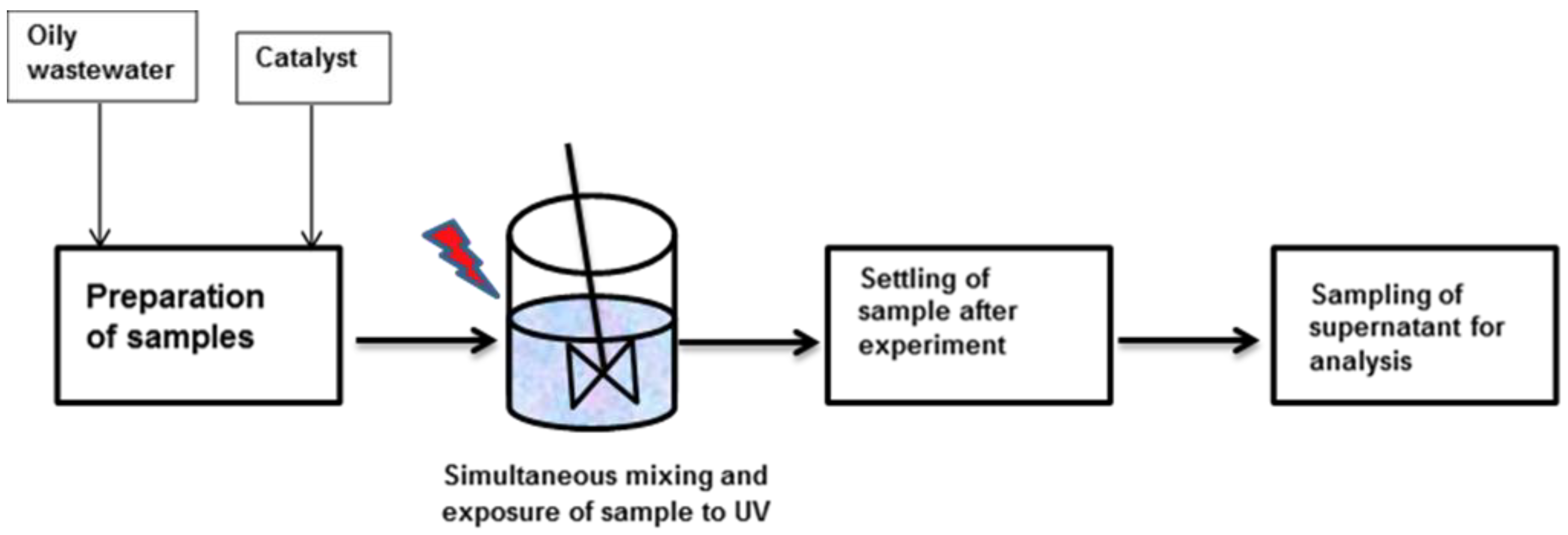
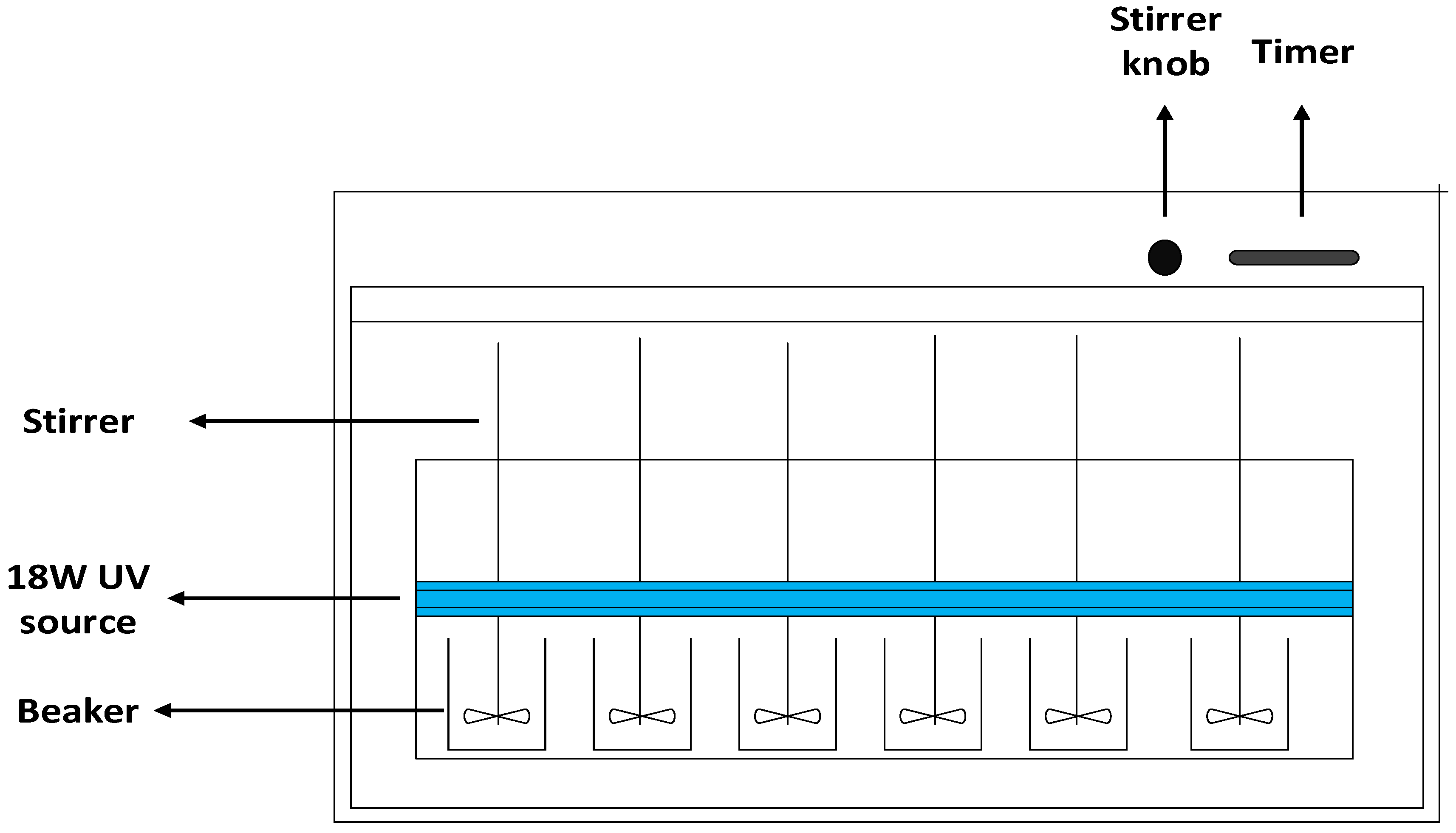
2.3. Experimental Setup
2.4. Response Surface Methodology (RSM)
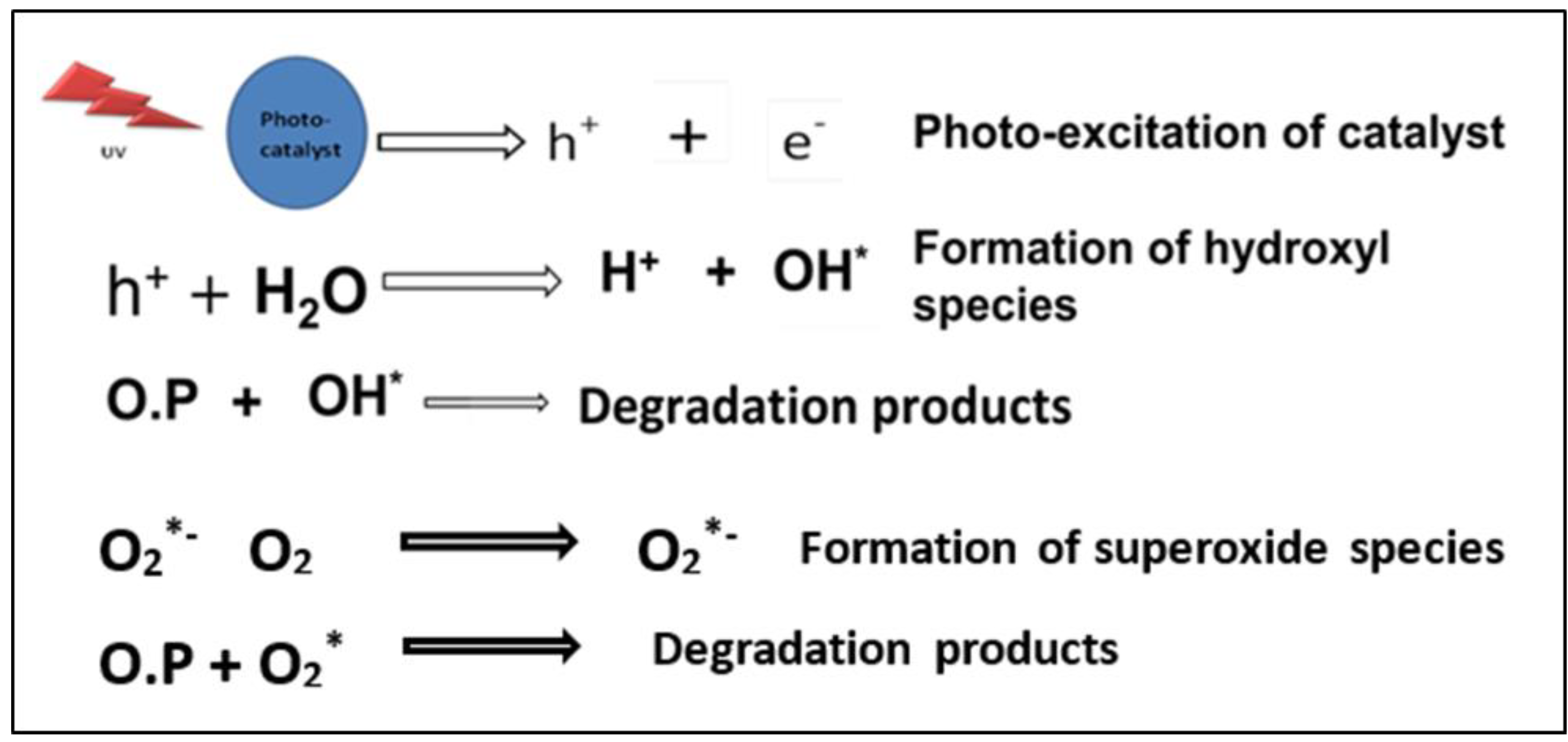
3. Results and Discussion
3.1. Model Fitting and Statistical Analysis
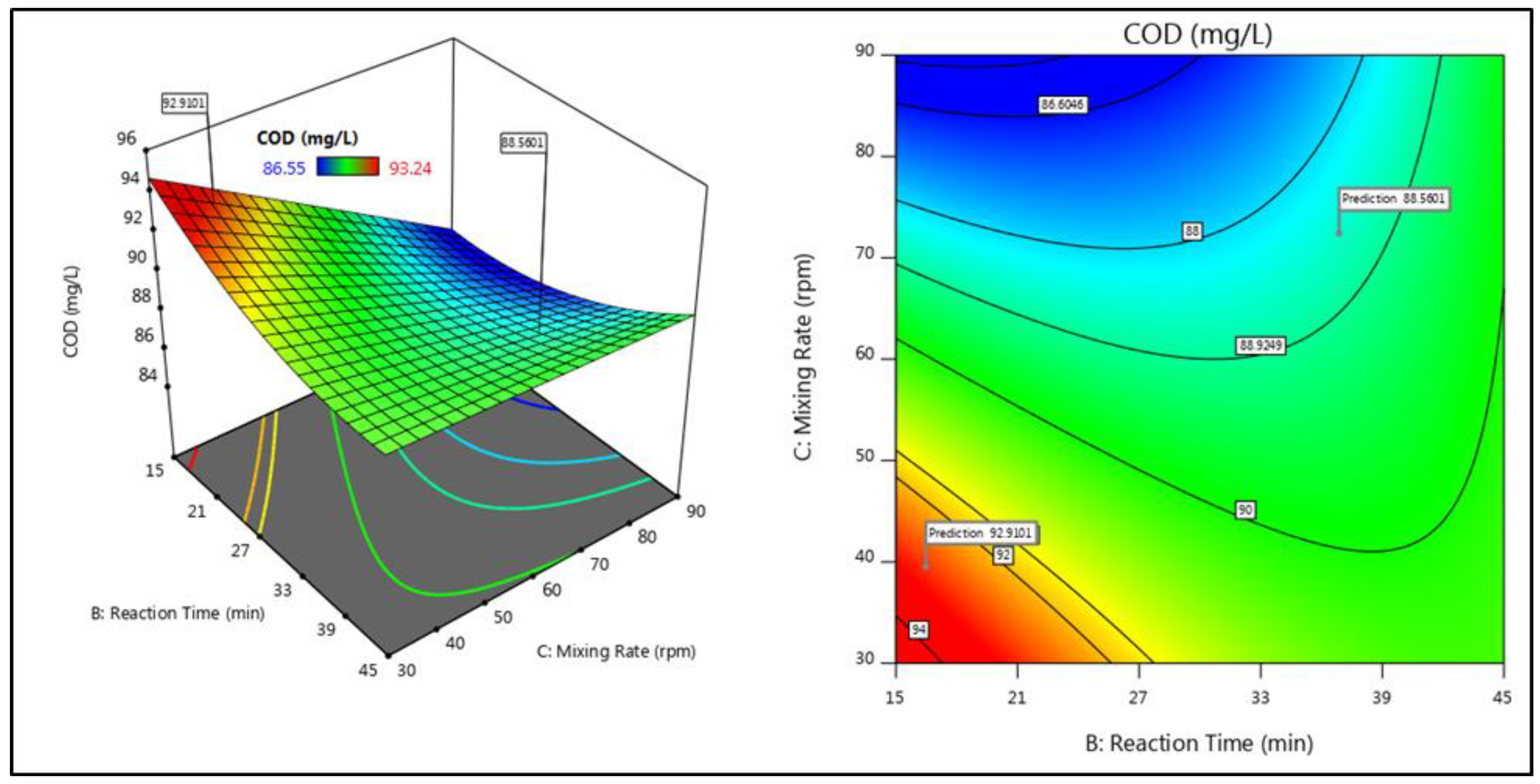
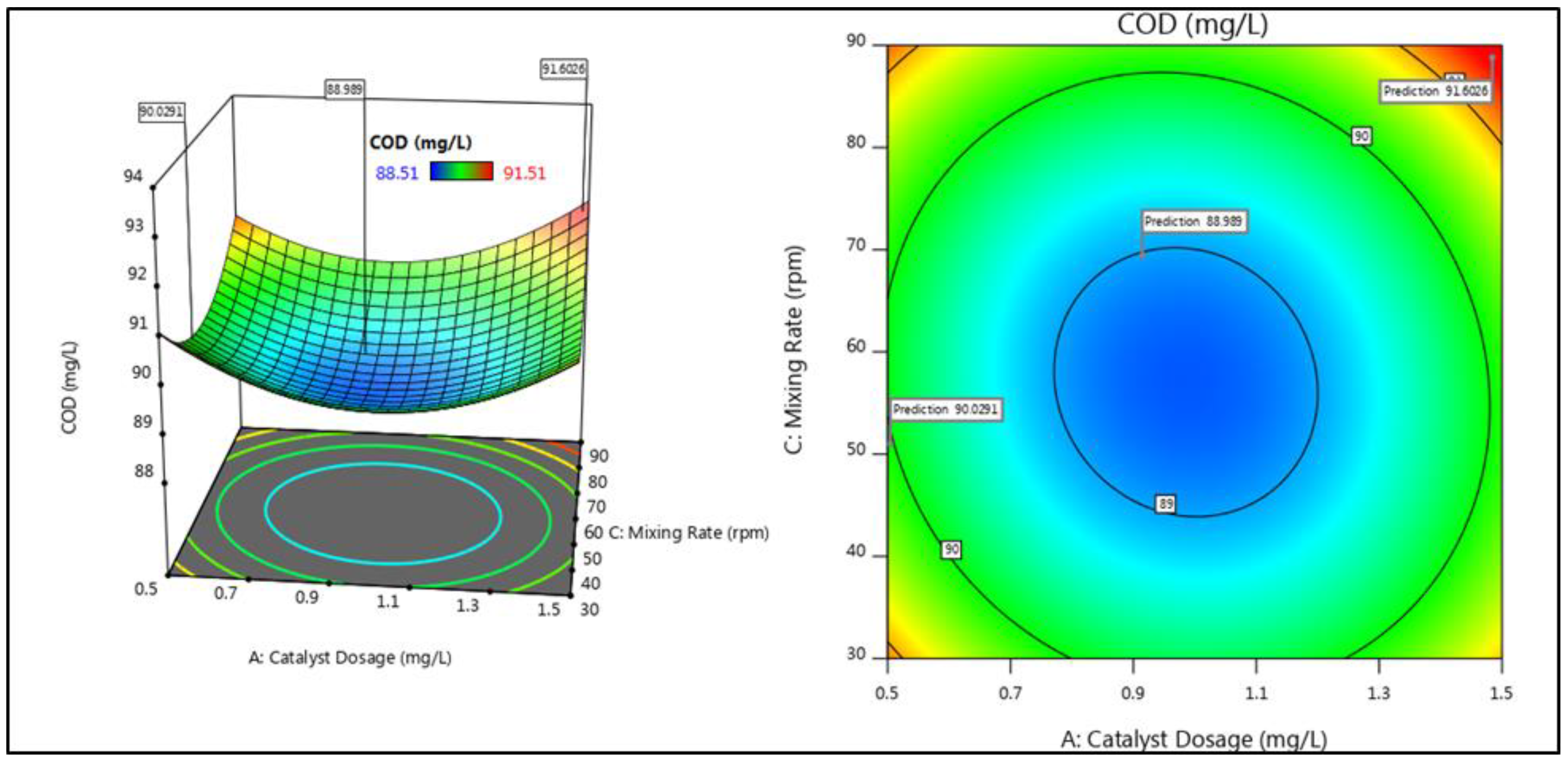
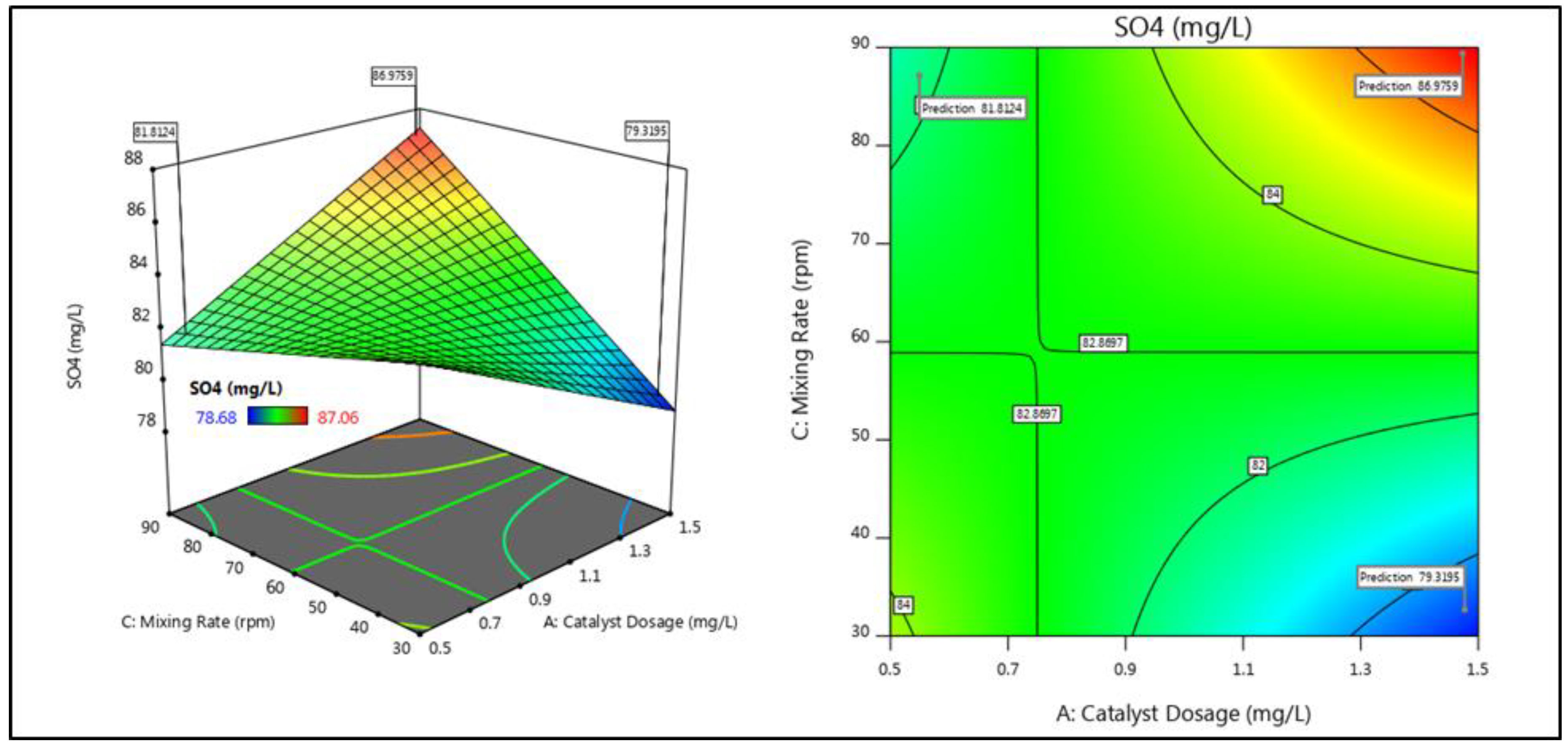
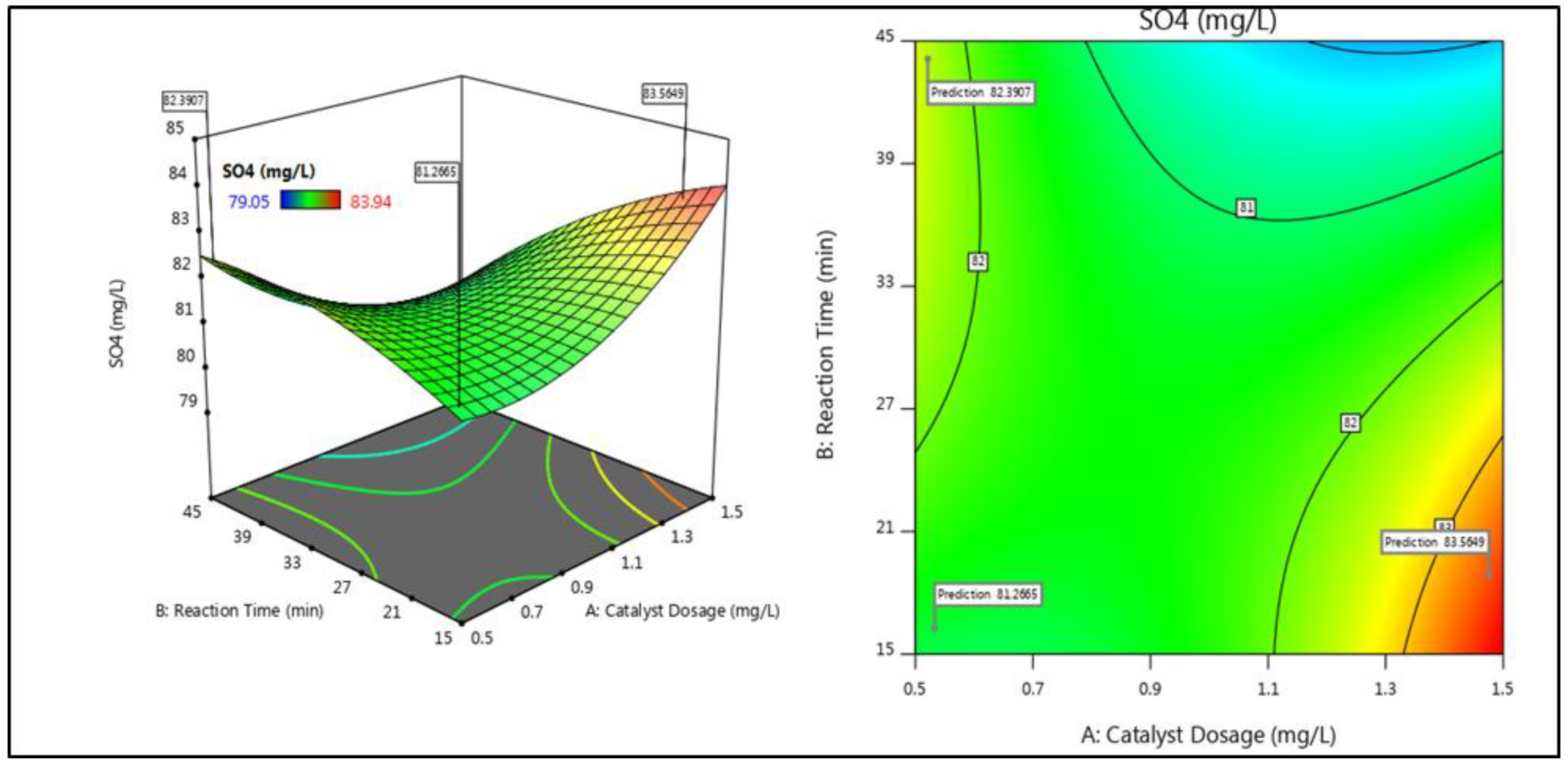
3.2. Interactive Effects of Parameters
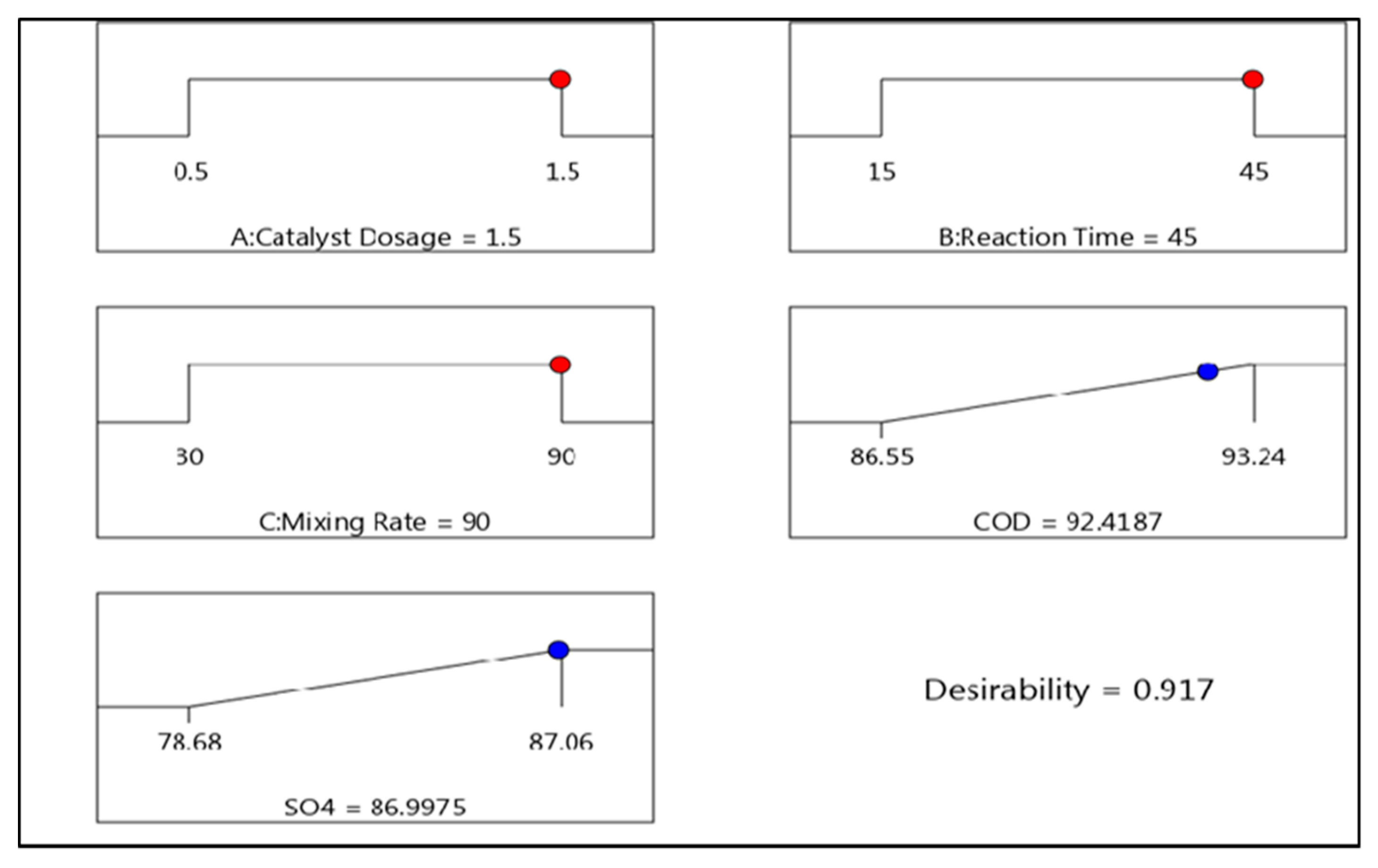
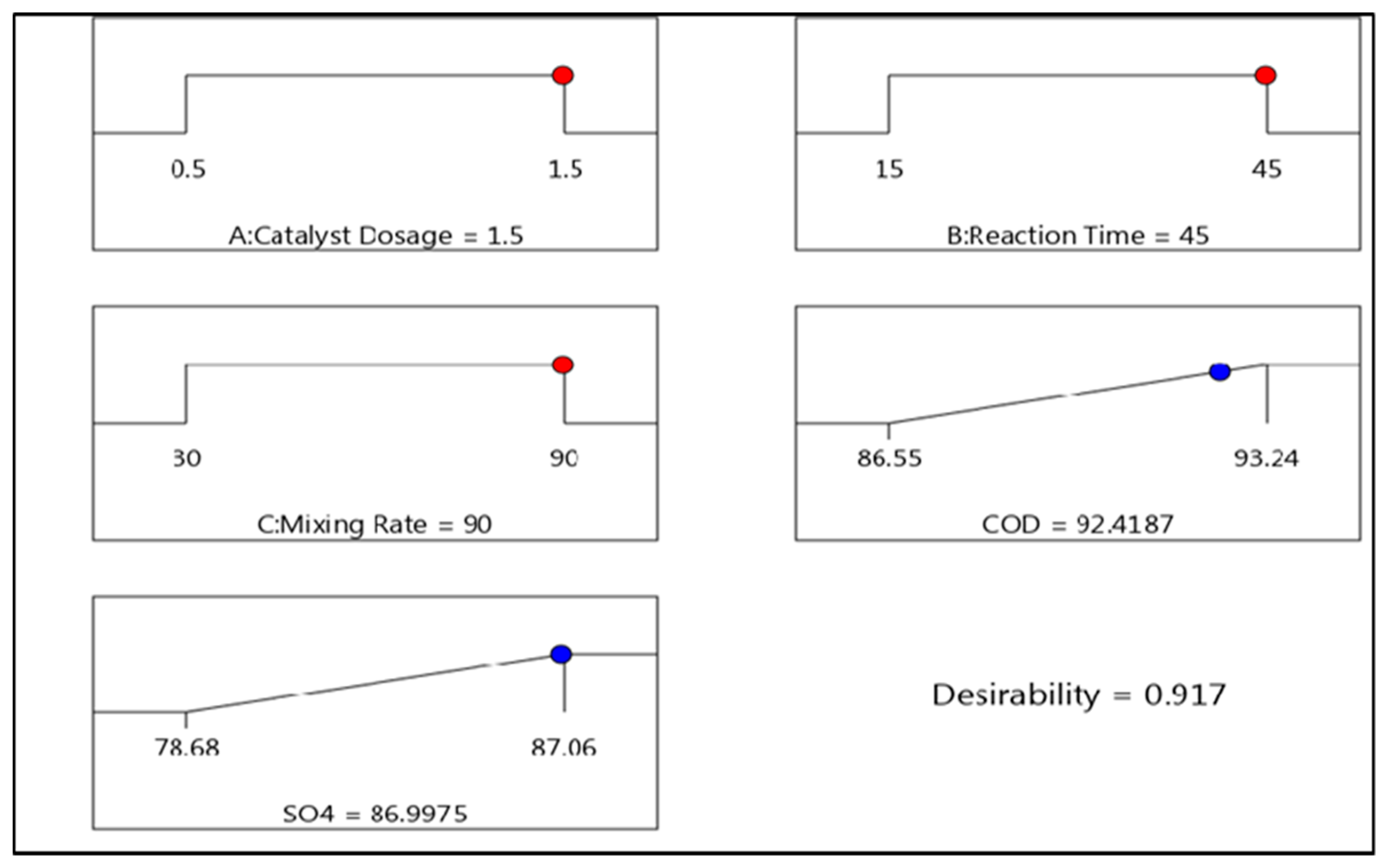
4. Optimum Conditions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cath, T.Y.; Drewes, J.E.; Lundin, C.D. A Novel Hybrid Forward Osmosis Process for Drinking Water Augmentation using Impaired Water and Saline Water Sources; Water Research Foundation: Las Cruces, NM, USA; Denver, CO, USA, 2009; Available online: http://inside.mines.edu/~tcath/research/projects/Cath_WRS_2009_AwwaRF4150.pdf (accessed on 12 January 2020).
- Pendashteh, A.R.; Fakhru’l-Razi, A.; Madaeni, S.S.; Abdullah, L.C.; Abidin, Z.Z.; Biak, D.R.A. Membrane foulants characterization in a membrane bioreactor (MBR) treating hypersaline oily wastewater. Chem. Eng. J. 2011, 168, 140–150. [Google Scholar] [CrossRef]
- Lefebvre, O.; Moletta, R. Treatment of organic pollution in industrial saline wastewater: A literature review. Water Res. 2006, 40, 3671–3682. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Zhang, X.; Liu, H.; Liu, S.; Yeung, K.L. Preparation and application of zeolite/ceramic microfiltration membranes for treatment of oil contaminated water. J. Membr. Sci. 2008, 325, 420–426. [Google Scholar] [CrossRef]
- Fakhru’l-Razi, A.; Pendashteh, A.; Abdullah, L.C.; Biak, D.R.A.; Madaeni, S.S.; Abidin, Z.Z. Review of technologies for oil and gas produced water treatment. J. Hazard. Mater. 2009, 170, 530–551. [Google Scholar] [CrossRef] [PubMed]
- Aljuboury, D.A.D.A.; Palaniandy, P.; Abdul Aziz, H.B.; Feroz, S. Treatment of petroleum wastewater by conventional and new technologies—A review. Glob. Nest J. 2017, 19, 439–452. [Google Scholar]
- Zhao, X.; Wang, Y.; Ye, Z.; Borthwick, A.G.; Ni, J. Oil field wastewater treatment in biological aerated filter by immobilized microorganisms. Process Biochem. 2006, 41, 1475–1483. [Google Scholar] [CrossRef]
- Hassan, A.A.; Naeem, H.T.; Hadi, R.T. Degradation of oily waste water in aqueous phase using solar (ZnO, TiO2 and Al2O3) catalysts. Biotechnology 2018, 15, 909–916. [Google Scholar]
- Aljuboury, D.D.; Palaniandy, P.; Feroz, S. Advanced Oxidation Processes (AOPs) to Treat the Petroleum Wastewater; IGI Global: Hershey, PA, USA, 2018. [Google Scholar]
- Samsudin, E.M.; Goh, S.N.; Wu, T.Y.; Ling, T.T.; Hamid, S.A.; Juan, J.C. Evaluation on the Photocatalytic Degradation Activity of Reactive Blue 4 using Pure Anatase Nano-TiO2. Sains Malays. 2015, 44, 1011–1019. [Google Scholar] [CrossRef]
- Kumar, K.; Chowdhury, A. Use of Novel Nanostructured Photocatalysts for the Environmental Sustainability of Wastewater Treatments. In Reference Module in Materials Science and Materials Engineering; Elsevier: Dublin, Ireland, 2018. [Google Scholar]
- Stasinakis, A. Use of selected advanced oxidation processes (AOPs) for wastewater treatment—A mini review. Glob. Nest J. 2008, 10, 376–385. [Google Scholar]
- El Hajjouji, H.; Barje, F.; Pinelli, E.; Bailly, J.R.; Richard, C.; Winterton, P.; Revel, J.C.; Hafidi, M. Photochemical UV/TiO2 treatment of olive mill wastewater (OMW). Bioresour. Technol. 2008, 99, 7264–7269. [Google Scholar] [CrossRef]
- Chatzisymeon, E.; Stypas, E.; Bousios, S.; Xekoukoulotakis, N.P.; Mantzavinos, D. Photocatalytic treatment of black table olive processing wastewater. J. Hazard. Mater. 2008, 154, 1090–1097. [Google Scholar] [CrossRef]
- Kalló, D. Applications of natural zeolites in water and wastewater treatment. Rev. Mineral. 2001, 45, 519–550. [Google Scholar] [CrossRef]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Nascimento, M.; Soares, P.S.M.; Souza, V.P.D. Adsorption of heavy metal cations using coal fly ash modified by hydrothermal method. Fuel 2009, 88, 1714–1719. [Google Scholar] [CrossRef]
- Petrus, R.; Warchoł, J.K. Heavy metal removal by clinoptilolite. An equilibrium study in multi-component systems. Water Res. 2005, 39, 819–830. [Google Scholar] [CrossRef]
- Wang, S.; Soudi, M.; Li, L.; Zhu, Z.H. Coal ash conversion into effective adsorbents for removal of heavy metals and dyes from wastewater. J. Hazard. Mater. 2006, 133, 243–251. [Google Scholar] [CrossRef]
- Woolard, C.D.; Strong, J.; Erasmus, C.R. Evaluation of the use of modified coal ash as a potential sorbent for organic waste streams. Appl. Geochem. 2002, 17, 1159–1164. [Google Scholar] [CrossRef]
- Guo, Y.; Zu, B.; Dou, X. Zeolite-based photocatalysts: A promising strategy for efficient photocatalysis. J. Thermodyn. Catal. 2013, 4, 1. [Google Scholar] [CrossRef]
- Dutta, P.K.; Turbeville, W. Intrazeolitic photoinduced redox reactions between tris (2,2′-bipyridine) ruthenium (2+) and methylviologen. J. Phys. Chem. 1992, 96, 9410–9416. [Google Scholar] [CrossRef]
- Dubey, N.; Rayalu, S.S.; Labhsetwar, N.K.; Naidu, R.R.; Chatti, R.V.; Devotta, S. Photocatalytic properties of zeolite-based materials for the photoreduction of methyl orange. Appl. Catal. A Gen. 2006, 303, 152–157. [Google Scholar] [CrossRef]
- Tetteh, E.K.; Rathilal, S. Effects of a polymeric organic coagulant for industrial mineral oil wastewater treatment using response surface methodology (RSM). Water SA 2018, 44, 155–161. [Google Scholar] [CrossRef]
- Sharifi, H.; Zabihzadeh, S.M.; Ghorbani, M. The application of response surface methodology on the synthesis of conductive polyaniline/cellulosic fiber nanocomposites. Carbohydr. Polym. 2018, 194, 384–394. [Google Scholar] [CrossRef]
- Houshmand, A.; Daud, W.M.A.W.; Shafeeyan, M.S. Tailoring the surface chemistry of activated carbon by nitric acid: Study using response surface method. Bull. Chem. Soc. Jpn. 2011, 84, 1251–1260. [Google Scholar] [CrossRef]
- Abdi, H. Coefficient of variation. Encycl. Res. Des. 2010, 1, 169–171. [Google Scholar]
- Farzadkia, M.; Bazrafshan, E.; Esrafili, A.; Yang, J.K.; Shirzad-Siboni, M. Photocatalytic degradation of Metronidazole with illuminated TiO2 nanoparticles. J. Environ. Health Sci. Eng. 2015, 13, 35. [Google Scholar] [CrossRef]
- Shahrezaei, F.; Mansouri, Y.; Zinatizadeh, A.A.L.; Akhbari, A. Process modeling and kinetic evaluation of petroleum refinery wastewater treatment in a photocatalytic reactor using TiO2 nanoparticles. Powder Technol. 2012, 221, 203–212. [Google Scholar] [CrossRef]
- Coelho, A.; Castro, A.V.; Dezotti, M.; Sant’Anna, G.L., Jr. Treatment of petroleum refinery sourwater by advanced oxidation processes. J. Hazard. Mater. 2006, 137, 178–184. [Google Scholar] [CrossRef]
- Saien, J.; Nejati, H. Enhanced photocatalytic degradation of pollutants in petroleum refinery wastewater under mild conditions. J. Hazard. Mater. 2007, 148, 491–495. [Google Scholar] [CrossRef]
- Topare, N.S.; Joy, M.; Joshi, R.R.; Jadhav, P.B.; Kshirsagar, L.K. Treatment of petroleum industry wastewater using TiO2/UV photocatalytic process. Indian Chem. Soc. 2015, 92, 219–222. [Google Scholar]
| Component | Value |
|---|---|
| pH | 8.09 |
| Conductivity (mg/L) | 613.38 |
| Sulphates (mg/L) | 895.00 |
| Sulphide (mg/L) | 1.26 |
| COD (mg/L) | 1226.8 |
| SOG (mg/L) | 33.23 |
| Properties | Value |
|---|---|
| White powder content | 94% purity |
| Phase mixture | Rutile 94%, anatase 6% |
| Surface treatment | Alumina, zirconia |
| Organic treatment | Present |
| Surface gravity | 4.1 g/cm3 |
| Crystal size | 10 nm |
| Loss at 105 °C | 0.60% |
| Bulk density | 1.1 g/cm3 |
| Oil absorption | 18 cm3 /100 g pigment |
| Durability | Highly durable |
| ISO 591 classification | R2 |
| Chemical Abstract Service (CAS) Number | 13463-67-7 |
| Factor A (Coded) | Factor B (Coded) | Factor C (Coded) | Response 1 (COD) % | Response 2 (SO42−) % | |||
|---|---|---|---|---|---|---|---|
| Run | Catalyst Dosage (g/L) | Reaction Time (min) | Mixing Rate (rpm) | Actual | Predicted | Actual | Predicted |
| 1 | −1 | 1 | 0 | 90.1 | 90.07 | 82.87 | 82.89 |
| 2 | −1 | −1 | 0 | 90.28 | 90.31 | 82.87 | 82.81 |
| 3 | −1 | 0 | −1 | 91.38 | 91.35 | 84.26 | 84.24 |
| 4 | 0 | 0 | 0 | 88.84 | 88.84 | 82.87 | 82.89 |
| 5 | 0 | −1 | 1 | 87.07 | 87.07 | 84.26 | 84.33 |
| 6 | 1 | 1 | 0 | 90.1 | 90.01 | 82.87 | 82.81 |
| 7 | 1 | 0 | 1 | 89.08 | 89.11 | 87.06 | 87.12 |
| 8 | 1 | −1 | 0 | 90.04 | 90.01 | 83.2 | 83.06 |
| 9 | 0 | −1 | −1 | 93.24 | 93.24 | 81.47 | 81.54 |
| 10 | 0 | 1 | 1 | 91 | 91.06 | 84.26 | 84.25 |
| 11 | 0 | 1 | −1 | 88.94 | 89.01 | 81.47 | 81.45 |
| 12 | 1 | 0 | −1 | 88.37 | 88.4 | 78.68 | 78.74 |
| 13 | −1 | 0 | 1 | 86.55 | 86.52 | 81.47 | 81.45 |
| 14 | 0 | 0 | 0 | 88.84 | 88.84 | 82.87 | 82.89 |
| 15 | 0 | 0 | 0 | 88.84 | 88.84 | 82.87 | 82.89 |
| Factor A (Coded) | Factor B (Coded) | Factor C (Coded) | Response 1 (COD) % | Response 2 (SO4) % | |||
|---|---|---|---|---|---|---|---|
| Run | Catalyst Dosage (g/L) | Reaction Time (min) | Mixing Rate (rpm) | Actual | Predicted | Actual | Predicted |
| 1 | 0 | −1 | 1 | 90.59 | 90.59 | 80.45 | 80.42 |
| 2 | −1 | 0 | −1 | 91.51 | 91.57 | 81.14 | 81.11 |
| 3 | 1 | 0 | −1 | 90.41 | 90.47 | 83.94 | 83.97 |
| 4 | −1 | 0 | 1 | 91.17 | 91.11 | 82.54 | 82.51 |
| 5 | 0 | 0 | 0 | 88.51 | 88.51 | 81.38 | 81.38 |
| 6 | 0 | 1 | 1 | 89.63 | 89.76 | 79.05 | 79.08 |
| 7 | 1 | 0 | 1 | 90.88 | 90.82 | 79.75 | 79.78 |
| 8 | −1 | 1 | 0 | 91.25 | 91.19 | 82.54 | 82.54 |
| 9 | 0 | 0 | 0 | 88.51 | 88.51 | 81.38 | 81.38 |
| 10 | −1 | −1 | 0 | 89.96 | 90.02 | 81.14 | 81.21 |
| 11 | 0 | −1 | −1 | 90.01 | 89.88 | 81.84 | 81.81 |
| 12 | 1 | −1 | 0 | 90.49 | 90.55 | 83.94 | 83.94 |
| 13 | 0 | 0 | 0 | 88.51 | 88.51 | 81.38 | 81.38 |
| 14 | 1 | 1 | 0 | 89.33 | 89.27 | 80.01 | 79.94 |
| 15 | 0 | 1 | −1 | 90.59 | 90.59 | 80.44 | 80.47 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 39.05 | 7 | 5.58 | 1614.82 | <0.0001 | significant |
| A—Catalyst dosage | 0.0648 | 1 | 0.0648 | 18.76 | 0.0034 | |
| B—Reaction time | 0.03 | 1 | 0.03 | 8.69 | 0.0215 | |
| C—Mixing rate | 8.47 | 1 | 8.47 | 2451.01 | <0.0001 | |
| AB | 0.0144 | 1 | 0.0144 | 4.17 | 0.0805 | |
| AC | 7.67 | 1 | 7.67 | 2221.24 | <0.0001 | |
| BC | 16.93 | 1 | 16.93 | 4902.02 | <0.0001 | |
| B2 | 5.87 | 1 | 5.87 | 1697.88 | <0.0001 | |
| Residual | 0.0242 | 7 | 0.0035 | |||
| Lack of fit | 0.0242 | 5 | 0.0048 | |||
| Pure error | 0 | 2 | 0 | |||
| Cor total | 39.07 | 14 | ||||
| R2 0.9994 | Adjusted R2 0.9988 | CV% 0.0657 | Predicted R2 0.9955 | Adeq. Pr 156.67 | Mean 89.51 | SD 0.0588 |
| Source | Sum of Squares | Degree of Dreedom | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 46.84 | 5 | 9.37 | 1764.91 | <0.0001 | significant |
| A—Catalyst dosage | 0.0144 | 1 | 0.0144 | 2.72 | 0.1334 | |
| B—Reaction time | 0.0136 | 1 | 0.0136 | 2.56 | 0.1438 | |
| C—Mixing rate | 15.6 | 1 | 15.6 | 2938.04 | <0.0001 | |
| AB | 0.0272 | 1 | 0.0272 | 5.13 | 0.0498 | |
| AC | 31.19 | 1 | 31.19 | 5876.09 | <0.0001 | |
| Residual | 0.0478 | 9 | 0.0053 | |||
| Lack of fit | 0.0478 | 7 | 0.0068 | |||
| Pure error | 0 | 2 | 0 | |||
| Cor total | 46.89 | 14 | ||||
| R2 0.999 | Adjusted R2 0.9984 | CV% 0.0879 | Predicted R2 0.9953 | Adeq. Pr 181.80 | Mean 82.89 | SD 0.0729 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 14.15 | 9 | 1.57 | 120.91 | <0.0001 | significant |
| A—Catalyst dosage | 0.9661 | 1 | 0.9661 | 74.28 | 0.0003 | |
| B—Reaction time | 0.0078 | 1 | 0.0078 | 0.6007 | 0.4733 | |
| C—Mixing rate | 0.0078 | 1 | 0.0078 | 0.6007 | 0.4733 | |
| AB | 1.5 | 1 | 1.5 | 115.39 | 0.0001 | |
| AC | 0.164 | 1 | 0.164 | 12.61 | 0.0164 | |
| BC | 0.5929 | 1 | 0.5929 | 45.59 | 0.0011 | |
| A2 | 5.93 | 1 | 5.93 | 456.12 | <0.0001 | |
| B2 | 0.8507 | 1 | 0.8507 | 65.41 | 0.0005 | |
| C2 | 5.45 | 1 | 5.45 | 419.12 | <0.0001 | |
| Residual | 0.065 | 5 | 0.013 | |||
| Lack of fit | 0.065 | 3 | 0.0217 | |||
| Pure error | 0 | 2 | 0 | |||
| Cor total | 14.22 | 14 | ||||
| R2 0.9954 | Adjusted R2 0.9872 | CV% 0.1266 | Predicted R2 0.9268 | Adeq. Pr 32.9036 | Mean 90.09 | SD 0.114 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 27.82 | 8 | 3.48 | 1187.01 | <0.0001 | significant |
| A—Catalyst dosage | 0.0098 | 1 | 0.0098 | 3.35 | 0.1171 | |
| B—Reaction time | 3.55 | 1 | 3.55 | 1212.33 | <0.0001 | |
| C—Mixing rate | 3.88 | 1 | 3.88 | 1323.96 | <0.0001 | |
| AB | 7.1 | 1 | 7.1 | 2424.66 | <0.0001 | |
| AC | 7.81 | 1 | 7.81 | 2666.98 | <0.0001 | |
| A2 | 3.42 | 1 | 3.42 | 1167.76 | <0.0001 | |
| B2 | 0.6987 | 1 | 0.6987 | 238.52 | <0.0001 | |
| C2 | 0.9231 | 1 | 0.9231 | 315.13 | <0.0001 | |
| Residual | 0.0176 | 6 | 0.0029 | |||
| Lack of fit | 0.0176 | 4 | 0.0044 | |||
| Pure error | 0 | 2 | 0 | |||
| Cor total | 27.83 | 14 | ||||
| R2 0.9994 | Adjusted R2 0.9985 | CV% 0.0665 | Predicted R2 0.9919 | Adeq. Pr 116.61 | Mean 81.39 | SD 0.054 |
| Photo-Catalytic Degradation of Petroleum Refinery Effluent | |||
|---|---|---|---|
| Contaminant Investigated | Conditions | Results (Removal %) | Reference |
| * TCOD | pH (4), catalyst concentration (100 mg/L), temperature (45 °C), and reaction time (120 min) | Over 83 | [29] |
| ** DOC | Catalyst concentration (0.2 g/L), reaction time (60 min), reactor volume (100 mL) | 21 | [30] |
| COD | Temperature (45 °C), catalyst concentration (100 mg/L), pH (3), reaction time (240 min) | 90 | [31] |
| COD | Catalyst concentration (1 g/L), pH (3), temperature (50 °C) | 60 | [32] |
| COD | Catalyst concentration (1.5 g/L), pH (6), reaction time (15 min), temperature (25 ± 2 °C), mixing rate (30 rpm) | 92 | * This study |
| SO42− | Catalyst concentration (1.5 g/L), pH (6), reaction time (45 min), temperature (25 ± 2 °C), mixing rate (90 rpm) | 87 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tetteh, E.K.; Obotey Ezugbe, E.; Rathilal, S.; Asante-Sackey, D. Removal of COD and SO42− from Oil Refinery Wastewater Using a Photo-Catalytic System—Comparing TiO2 and Zeolite Efficiencies. Water 2020, 12, 214. https://doi.org/10.3390/w12010214
Tetteh EK, Obotey Ezugbe E, Rathilal S, Asante-Sackey D. Removal of COD and SO42− from Oil Refinery Wastewater Using a Photo-Catalytic System—Comparing TiO2 and Zeolite Efficiencies. Water. 2020; 12(1):214. https://doi.org/10.3390/w12010214
Chicago/Turabian StyleTetteh, Emmanuel K., Elorm Obotey Ezugbe, Sudesh Rathilal, and Dennis Asante-Sackey. 2020. "Removal of COD and SO42− from Oil Refinery Wastewater Using a Photo-Catalytic System—Comparing TiO2 and Zeolite Efficiencies" Water 12, no. 1: 214. https://doi.org/10.3390/w12010214
APA StyleTetteh, E. K., Obotey Ezugbe, E., Rathilal, S., & Asante-Sackey, D. (2020). Removal of COD and SO42− from Oil Refinery Wastewater Using a Photo-Catalytic System—Comparing TiO2 and Zeolite Efficiencies. Water, 12(1), 214. https://doi.org/10.3390/w12010214








