Abstract
This work is part of a research project that aims to develop a method for combined radiocarbon and uranium-isotope dating of groundwater. The specific objective of this study was to investigate the mobility of uranium isotopes in the various mineral phases of rocks of an aquifer of sandy-clay deposits of the Vendian in the northwest of Russia, for which a partial extraction procedure was used. Representative aliquots of each core sample were treated with various reagents: distilled water, low mineralized fresh natural groundwater, minopolycarboxylic acid chelating agent (0.05 M EDTA), 0.5M HCl, 15 M HNO3, as well as total digestion, and the U isotope data for each procedure are reported in this study. The following mineral phases of core samples were characterized: sorbed material, carbonate minerals, amorphous iron oxides, partially soluble aluminosilicates and crystalline iron oxides, totally digestible aluminosilicates and a clay/quartz resistate. Red-colored siltstones and sandstones were depleted in uranium in relatively readily soluble mineral phases. The concentration of sorbed uranium was established in the range 15.8 ± 2.1–30.5 ± 3.9 μg/kg and carbonate minerals contained even less uranium. For iron hydroxides and the most readily soluble aluminosilicates, uranium concentrations were in the range 168 ± 24–212 ± 28 μg/kg. The most insoluble fraction contained uranium in the range 1.65 ± 0.21–4.32 ± 0.45 mg/kg. In green-colored siltstones, the concentration of sorbed uranium was much higher in the range 106 ± 14–364 ± 43 μg/kg. Carbonate minerals and amorphous iron oxides contained uranium in the range 1.91 ± 0.21–2.34 ± 0.26 mg/kg. In aluminosilicates and a clay/quartz resistate, uranium concentrations were in the range 5.6 ± 0.5–16.8 ± 1.4 mg/kg. Elevated values of 234U:238U activity ratio prevailed in the sorbed material and iron hydroxides. In aluminosilicates and clay/quartz resistate, the values decreased. This indicates the replacement of primary sedimentogenic uranium by secondary hydrogenic uranium sorbed on the surface of minerals and coprecipitation with iron hydroxides. The results obtained made it possible to carry out preliminary quantitative estimates of the retardation factor and recoil loss factor of uranium in the groundwater of siltstones and sandstones of the studied Vendian aquifer.
1. Introduction
Uranium isotopes are a powerful tool for refining conceptual models of groundwater [1,2,3,4], groundwater dating [5,6,7], and descriptions of chemical weathering processes [8,9,10] for periods of up to hundreds of thousands of years [11]. The main initial parameters are (i) measured concentration of 238U in the combined solution and solid phase, and (ii) measured 234U:238U activity ratio in the combined pore fluid and solid phase and they can be obtained by direct measurements in water and rock samples collected in the field.
However, the interpretation of the results is rather complicated due to the compositional heterogeneity of the rocks. For example, sandy clay sediments of aquifers can contain both poorly soluble grains of quartz and feldspar, as well as soluble carbonate and gypsum cements. Carbonates, iron oxides, and clay minerals are characterized by increased U sorption on their surfaces [12,13,14]. Further, daughter nuclides pass most actively into water from surface coatings [15], increasing the uranium isotopes activity ratio in water. In addition, the similarity of the activity ratios of 234U:238U in groundwater and in the most easily leached fractions of water-bearing rocks was noted in the experiments of Lowson et al. [16], Payne et al. [17], and Dabous et al. [18], with the fractions being defined as sorbed elements, carbonate minerals and amorphous iron minerals [19] which could indicate the opposite process which is the transition of nonequilibrium nuclides from water to rock with an increase in the U isotopes activity ratio in the rock [20]. Under such conditions, it is difficult to estimate the rate of chemical weathering and to date natural processes by using the currently developed methods [21,22].
Therefore, for a more complete understanding of the behavior of uranium isotopes in the water-rock system, a transition from the presentation of solely total U concentration and U isotopes activity data to an additional analysis using individual weak leach extractants or sequential extractions is necessary. In recent decades, leach data, rather than total decomposition data are widely used in a variety of fields of geoecology (see for example [23,24,25,26]). Therefore, in the present work, an attempt has been made to use the partial extraction procedure for reconstructing the processes of redistribution of uranium isotopes in certain mineral phases of an aquifer of sandy-clay deposits of the Vendian.
In previous papers [27,28], the possibility of sharing uranium and carbon isotopes for dating groundwater has been discussed and the transport of uranium in solution is reasonably well described with a standard advection-dispersion-exchange formulation: [5,6,7]. Among the many factors, the main ones are t—Groundwater residence time in the aquifer (time), v—Groundwater flow velocity (advection), Rd—Dissolution rate (weathering-precipitation), R—Retardation factor (sorption-desorption), p—Recoil loss factor (recoil + production), and λ4—Decay constants for 234U (decay). Suitable equations were derived by Andrews and Kay [29], Fröhlich and Gellermann [30], Ivanovich et al. [9], and Porcelli [5] to provide a 234U–238U dating method for groundwater under oxidizing conditions. However, the values of R, Rd (a−1) and p needed to be determined. In turn, the recoil loss factor value p depends on the specific surface area (SSA). The published SSA values in sandy aquifers range from 0.9–1.8 m2 kg−1 [31] to 330–390 m2 kg−1 [6]. Accordingly, the ranges of R and Rd can be calculated using these formulas. Therefore, we consider it more appropriate to show R and p in the form of their ratios: R:p. The main calculated equations are as follows [27]:
where is the concentration of uranium in water; is the concentration of uranium in the rock; -234U:238U is the activity ratio in the water sample; is the solid mass to fluid volume ratio.
In this case, two unknown parameters remain in Equation (1), which cannot be directly measured in water and rock samples: and . Therefore, in order to use Equation (1), it is necessary to make several determinations of the groundwater age by using other methods such as carbon isotopes. Then, we need to determine whether there is an increase in uranium concentrations in groundwater with a decrease in the concentration of radiocarbon. If this is the case, then the mean values for the studied aquifers can be determined. After this, uranium-isotopic dating of other groundwater samples, which is less labor-intensive and more accessible than radiocarbon dating, is carried out. The average value of the retardation factor/recoil loss factor ratio () in samples from the siltstone and sandstone aquifer of the upper Vendian strata and overlying horizons is assumed to be (24 ± 4) [27].
In the present study, by determining the amount of uranium sorbed on the surface of mineral particles, we planned to proceed to a separate assessment of the values of the retardation factor and recoil loss factor of uranium in the groundwater of siltstones and sandstones of the studied Vendian aquifer.
The Vendian aquifer was selected for this study due to the relatively homogeneous sandy composition of the host sedimentary sediments. In porous reservoirs, the indicators of the behavior of uranium during water-rock interaction are most pronounced. Groundwater is characterized by elevated uranium concentrations and uranium isotope ratios, which is favorable for groundwater dating. Rocks are also characterized by elevated uranium concentrations. The largest roll-type uranium deposits are confined to sandstones. Therefore, traditionally, the dating of groundwater by uranium isotopes was carried out in sandstone aquifers. We also carried out comprehensive isotope-geochemical studies in this direction with the aim of fundamental comprehensive justification of the method of dating groundwater by a complex of isotopes including isotopes of uranium, carbon, hydrogen, and oxygen.
2. Materials
The borehole GGS2-11 is located at the diamond deposit area with coordinates N65°20′48″ E41°06′10″ in the northwest of Russia and was drilled using a diamond drill bit (92 mm inner diameter, 112 mm outer diameter) and mud rotary methods to 101.2 m below the ground surface in July 2018. This core was selected because the sampled section traverses the aquifer of the Padun formation of the Vendian (Vpd) [27,28] which is of interest in the present study. Five samples were taken from the intervals of 66.7–66.8, 75.2–75.3, 83.9–84, 94–94.1, 99.5–99.6 m (Figure 1).

Figure 1.
Photos of samples of studied rocks. 1—Green siltstones, 66.7–66.8 m. 2—Red sandstones, 75.2–75.3 m. 3—Variegated siltstones, 83.9–84 m. 4—Red siltstones, 94–94.1 m. 5–6—Green siltstones, 99.5–99.6 m (photo S. Druzhinin).
The samples were quickly packed in airtight polythene bags. The sample mass collected in each case was about 1500 g. Sub-samples of the material were oven dried at 40 °C for 7 days and homogenized by grinding with an agate mortar and pestle to pass through a 125 μm sieve in a glove box with nitrogen. The prepared material was stored in glass bottles for sequential extractions and isotopic analyses.
The chemical composition and the mineralogical composition of the Vendian deposits in the area of the diamond deposit were studied in detail during different periods of geological exploration [32,33,34,35]. The Padun Formation which is 160 m thick mainly consists of sandstones (60–80%) and siltstones (20–30%) separated by mudstone interlayers. The rocks have a reddish-brown color with pale green lenses and patches and the sandstones are dominated by fine- and medium-grained varieties. The content of pelitic particles does not exceed 20%, clastic material is represented by quartz and feldspars, chalcedony, quartzite fragments, biotite, and clayey aggregates are insignificant. The cement has mainly a clayey (hydromicaceous)-ferruginous composition and carbonate and gypsum cements are also encountered. In the upper part of the sequence (thickness~50 m), the sandstones are poorly cemented and often represented by sands. The siltstones are dominated by the coarse-grained fraction and the clastic grains consist of quartz (up to 98%), feldspars (up to 10%), and micas (~1%) while the cement has clayey-ferruginous, carbonate-clayey, and less common gypsum compositions. Clay minerals are observed as hydromicas, kaolinite, and chlorite. Bitumen and organic carbon are nearly absent. The ratio of Fe2O3 and FeO forms of iron in red siltstones is ~ 17: 1 and in green siltstones, the Fe2O3 content is 2.7 times lower (Table 1).

Table 1.
Average chemical composition of the red siltstones and sandstones of the Padun Formation of the Vendian in the research area (from 18 determinations) [34] and green siltstones of the sample GGS2-11 (depth 99.5 m), %.
The description of the samples of the studied rocks was performed on five thin sections of core samples (see Appendix A).
3. Methods
Representative aliquots of each core sample were treated with various reagents, with U isotopes being reported in this study for each procedure. The various procedures are described hereinafter.
3.1. Partial Extractions
3.1.1. Distilled Water
A volume of 500 mL distilled water was mixed with 50 g of core sample in centrifuge tubes and shaken for 1 h on an end-over-end shaker at room temperature. The short duration of the laboratory experiments suggests that dissolution depends upon the ease with which the fine particulates may be freed from the rock surface [36,37]. First, this procedure refers to sorbed particles.
3.1.2. Low Mineralized Fresh Natural Groundwater
A volume of 500 mL of low mineralized water of Ca-Mg-Na-HCO3 composition from the borehole had total dissolved solids (TDS) of 285 mg/L (see [28]) and was mixed with 50 g of core sample in centrifuge tubes and shaken for 1 h on an end-over-end shaker at room temperature. The boundary conditions when using low-mineralized fresh natural groundwater were as follows: temperature 20 °C, pH 7.7 and redox potential of 230 mV. First, sorbed material was released.
The following four stages were carried out according to the method proposed by Sutherland et al. [26].
3.1.3. 0.05M EDTA
Aminopolycarboxylic acid chelating agent (ethylenediaminetetraacetic acid, EDTA) at the concentration and pH level used in this study was the weakest extractant after fresh groundwater [26] and EDTA has been widely used in environmental geochemistry. Complexants such as EDTA are frequently used to release the readily available (labile) fraction of materials [26], which in the present study is carbonate cement. The procedure outlined by Singh et al. [38] was followed in the present study and 500 mL of 0.05M EDTA (pH 7) was mixed with 50 g of core sample in centrifuge tubes and shaken for 1 h on an end-over-end shaker at room temperature.
3.1.4. 0.5M HCl
According to a review by Sutherland et al. [26], hydrochloric acid (HCl) dissolves complexed, adsorbed, precipitated, amorphous or poorly crystallized Fe compounds without significant attack on the crystal lattice. In the present study, 500 mL of 0.5M HCl was mixed with 50 g of core sample and shaken at room temperature for 1 h on an end-over-end shaker.
3.1.5. 15M. HNO3
Nitric acid (HNO3) is an oxidizing agent that is not as powerful in its attack on aluminosilicates as hydrofluoric acid (HF) and is therefore described as a partial digestion. Also, crystalline iron oxide is digested by hot HNO3. In the present study, 500 mL of 15 M HNO3 was mixed with 25 g of core sample and heated for 1 h while stirring. The resulting solution was evaporated to wet salts, topped up with 500 mL of distilled water and acidified with HCl to pH 1–2.
3.2. Measurements of Uranium Isotopes after Five Partial Extractions
Determinations of uranium isotopes in the resulting solution were made in accordance with Malyshev et al. [39] which is also described in Fröhlich [40]. Spectrometric detection of alpha particles was performed using an alpha spectrometer (PROGRESS-ALPHA, DOZE, Russia) with an uncertainty of 10–15%. Total error of analysis is defined by δ = δst + δsys (statistical + systematic), measurement of uncertainties for U are reported individually (Table 2), and efficiency of 232U extraction was 40–50%.

Table 2.
Distribution of uranium and measured 234U:238U activity ratios in extracted phases.
3.3. Total Digestion
Measurements of uranium isotopes were made in accordance with Malyshev et al. [41]. For these analyses, 10 g of core sample was placed in a porcelain crucible and calcined at 500 °C until complete combustion of organic substances. The residue was transferred into a Teflon cup wetted with distilled water. A weighed amount of spiked solution (with known activity of internal standard 232U) was added, followed by 10 cm3 of 15M HNO3 and this was then heated until the reddish brown NO2 gas vapor disappeared. Following cooling, 40 cm3 of 29M HF and 10 cm3 of 12M HClO4 were added. The covered Teflon cup was heated until HClO4 vapor appeared. HF treatment was repeated twice; the cup was cooled each time before adding acid and heating until HClO4 fumes were visible. After dissolution and cooling, the edge of the cup and the cover were rinsed with distilled water and the content was transferred to an open Teflon™ dish, where the solution was evaporated again until dense white fumes appeared. This procedure was repeated twice. Finally, the residue was evaporated to give moist salts, which were dissolved in boiling 7 M HNO3 (50 cm3). The undissolved residue was filtered off and washed three times with 5–10 cm3 portions of hot 7M HNO3 and added to the solution. The U solution was transferred to a separatory funnel and a 30% solution of freshly purified tributylphosphate in toluene was added to give an aqueous to organic phase ratio of 4:1. Radionuclides were extracted for 5 min. After phase separation, the lower layer was poured back into the beaker. The organic extract was washed twice with an equal volume of 7M HNO3, then with an equal volume of 0.25M HNO3 solution in 0.04M HF. The back-extraction of uranium isotopes was performed by washing the organic phase with an equal volume of distilled water three times for 1 min each. The pooled aqueous extracts were evaporated to dryness, treated with 5 cm3 of concentrated HNO3 to remove trace organic substances, and evaporated to dryness. The dry residue containing uranium was dissolved in 10 mL of 2% sodium carbonate with heating, then filtered. Dissolved uranium was electrodeposited on polished stainless steel plates for alpha spectrometry.
4. Results
Table 2 and Figure 2 and Figure 3 show the results of five partial extractions and total digestion of the five core samples.
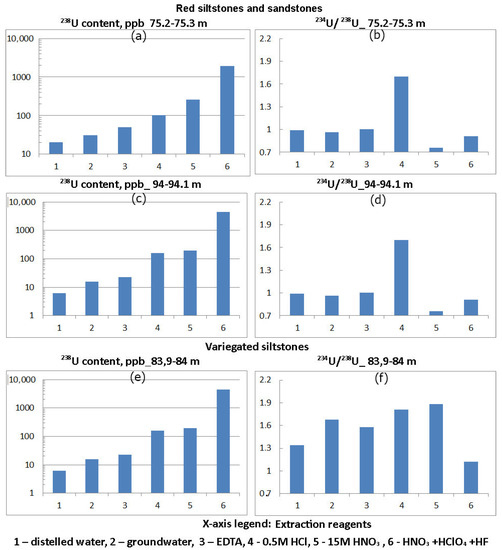
Figure 2.
Distribution of 238U content and 234U:238U activity ratio in phases 1–6, leached from crushed red and variegated siltstone of Vendian (Ediacaran) Padun Formations.
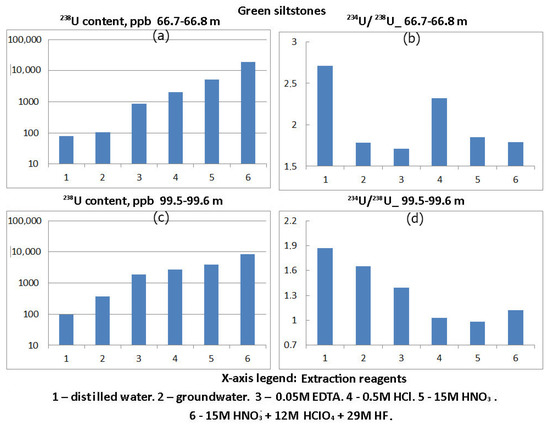
Figure 3.
Distribution of 238U content and 234U:238U activity ratio in phases 1–6, leached from crushed green siltstone of Vendian (Ediacaran) Padun Formations.
4.1. Substance Dissolution
As can be seen from Table 2, water dissolved on average about 1% of the samples of siltstones and sandstones while green siltstones dissolved approximately two-fold more intensively. The solvents, EDTA and 0.5M HCl, are further dissolved in approximately 1% of each. The ratio of red and green siltstones and sandstones dissolved in hydrochloric acid remains~1:2. The impact of EDTA on both types of rocks is approximately the same. Nitric acid additionally dissolves an average of 3.36% of the rock. Green siltstone dissolves with nitric acid 2.5-fold more intensively than with hydrochloric acid. The bulk of the rock, from 96.58 to 98.18% red siltstones and sandstones and from 84.6 to 93.15% green siltstones, is only dissolved with a mixture of 15M HNO3 + 12M HClO4 + 29M HF acids.
4.2. Uranium Concentration
In red siltstones and sandstones, the total uranium content was 1910 ± 243–4510 ± 467 μg/kg. The material dissolved in groundwater contained 15.8 ± 2.1–30.5 ± 3.9 μg/kg. Three subsequent solvents bring the uranium content in the material dissolved by them to 191 ± 27–262 ± 34 μg/kg, which is only 4.2–13.7% of the total uranium content in red siltstones and sandstones. The main supply of uranium is contained in these rocks in the highest degree of barely soluble fraction, extractable by complete digestion.
In green siltstones, the total uranium content was 8328 ± 707–18,800 ± 1504 μg/kg, the material dissolved in groundwater contained 106 ± 14–364 ± 43 μg/kg, while three subsequent solvents brought the uranium content in the material dissolved by them to 3957 ± 415–5086 ± 512 μg/kg, which is already 27.1–47.5% of the total uranium content in green siltstones. The most insoluble fraction, respectively, contained 52.5–72.9% of uranium.
4.3. 234U:238U Activity Ratio
In red siltstones and sandstones, the maximum values of 234U:238U activity ratio (1.7 ± 0.25–2.3 ± 0.32) were noted in the material dissolved in groundwater and 0.5 M HCl. Minimum values (0.76 ± 0.11–0.91 ± 0.13) are characteristic of the most difficultly soluble 15 M HNO3 and 15 M HNO3 + 12 M HClO4 + 29 M HF fractions of the studied rocks, and in other cases, the values are close to unity.
In green siltstones, one increased value of 234U:238U activity ratio (2.32 ± 0.33) was noted in a material dissolved in 0.5 M HCl. Three values close to unity were obtained by dissolving 0.5 M HCl, 15 M HNO3 and 15 M HNO3 + 12 M HClO4 + 29 M HF. However, in the remaining eight experiments using groundwater, 0.05 M EDTA, 15 M HNO3, and 15 M HNO3 + 12 M HClO4 + 29 M HF as a solvent, the values of 234U:238U activity ratio were high: 1.39 ± 0.2–2.71 ± 0.36.
5. Discussion
5.1. Substance Dissolution
In general, the percentage values of the substance dissolved under the influence of various solvents are approximately consistent with the average chemical composition of siltstones and sandstones of the Padun Formation of the Vendian in the study area. EDTA, the weakest extractant after fresh groundwater, dissolved up to 3.4% of the substance (Table 2). This corresponds to a loss on ignition (LOI) of 2.74–3.36% (Table 1), i.e., the carbonate material of the cement of the studied rocks and other labile fractions. Up to 3% of the substance was additionally dissolved by 0.5 M HCl, which is consistent with the content of Fe2O3 (2–5.3%) in Table 1, that is, it corresponds to amorphous iron oxides. Up to 9% of a substance that can only be represented by the most readily soluble aluminosilicates was additionally dissolved by15 M HNO3. A mixture of 15 M HNO3 + 12M HClO4 + 29 M HF acids dissolved the remainder of the aluminosilicates and a clay/quartz resistate.
5.2. Uranium Concentration
Table 3 shows the uranium concentrations converted to mineral phases in μg/kg and %. In red siltstones and sandstones, the concentrations of sorbed uranium were established in the amount of 15.8 ± 2.1–30.5 ± 3.9 μg/kg. This amounts to 0.35–1.6% of its total amount in rocks of this type. Carbonate minerals contain even less uranium: 7 ± 1–19.8 ± 2.5 μg/kg. In iron hydroxides and the most readily soluble aluminosilicates, uranium concentrations are in the range 52.7 ± 7.2–133 ± 19 and 35 ± 5–159 ± 21 μg/kg, respectively. This amounts to a total of 3.7–11.1% of its total concentration. The most insoluble fraction contains 1.6 ± 0.2–4.3 ± 0.4 mg/kg of uranium. In green siltstones, the concentration of adsorbed uranium is much higher: 106 ± 14–364 ± 43 μg/kg (0.56–4.37%). The carbonate minerals and amorphous iron oxides contain a total of 1.91 ± 0.21–2.34 ± 0.26 mg/kg of uranium (10.15–28.2%). In aluminosilicates and a clay/quartz resistate, the concentration of uranium amounts to 5.6 ± 0.5–16.8 ± 1.4 mg/kg.

Table 3.
Distribution of uranium in the mineral phases.
5.3. 234U:238U Activity Ratio
Increased values of 234U:238U activity ratio were noted in the material of red siltstones and sandstones dissolved in water and 0.5M HCl. That is, uranium isotopes with elevated values of 234U:238U activity ratio (greater than 1) were deposited from groundwater on sorbent material and with iron hydroxides. Reduced values of 234U:238U activity ratio (less than 1) are characteristic of the most difficultly soluble fractions within which the dissolution of uranium by groundwater was practically absent and only depletion of 234U atoms occurred due to recoil loss factor. In green siltstones, there is also a tendency towards a decrease in 234U:238U activity ratio in the direction from easily soluble fractions to sparingly soluble fractions (Figure 3, interval 99.5–99.6 m). However, for a sample taken from the interval 66.7–66.8 m, high values of 234U:238U activity ratio are observed in all mineral phases. Additional studies are needed to explain this fact.
5.4. Evolution of Uranium Isotopic Compositions
The results obtained partially confirm previously expressed ideas about the evolution of U isotopic compositions of the Vendian rock near the study area [20]. It has been established that the processes of chemical weathering of Vendian deposits led to the formation of a strong oxidation zone, developed above 250 m.b.s.l. The inverse correlation between the concentrations of U and Fe (see Table 1 and Table 2) in the red siltstones and sandstones (increased Fe concentrations and reduced U contents) is a result of removal of U in oxidizing conditions, and accumulation of Fe. In red siltstones and sandstones, the concentration of Fe2O3 is 5.3%, and the content of U is 1.9-4.5 mg/kg, in green siltstones the concentration of Fe2O3 is 2%, and the content of U is 5.8–18.8 mg/kg. Almost all U in the red siltstones and sandstones has been replaced by a newly formed hydrogenic U (precipitated from groundwater), with an initial 234U:238U activity ratio ≈ activity ratio of modern fresh groundwater = 3. The ending of the period of co-precipitation of hydrogenic uranium with iron hydroxide was estimated as 0.9 Ma, which should roughly correspond to the period of a sharp cold snap in the region and after that, dissolution and desorption of hydrogenic U occurred during periods with no glaciations and marine transgressions.
Our results are consistent with the experimental results of Lowson et al. [16], Payne et al. [17], and Dabous et al. [18], which note the similarity of the 234U:238U activity ratios in groundwater and in the most easily leached fractions of water-bearing rocks.
At the same time, the lower average value of 234U:238U activity ratio 0.92 [20] was established in the samples of green siltstones that were collected in the paleovalley, which was screened by the layer of sea clays. It is explainable by the fact that these deposits have reached a steady state of the 234U:238U activity ratio that depends only on their size (the average grain size ≈ 30 µm), because they were under reducing conditions for over 1 Ma. A significantly higher content of uranium in them compared to red siltstones and sandstones shows a considerable variability in the permeability values of the aquifer, whereby they were away from the paths of groundwater filtration and have retained uranium. The green siltstones that were studied in the present work occur in the oxidative conditions of the aquifer which contains young fresh water [28]. They have high concentrations of uranium in readily soluble fractions and high 234U:238U activity ratios. This can be explained by their deep processing by groundwater in a relatively recent period (tens of thousands to the first hundreds of thousands of years). During this processing, they were in a loose state, as a result of which U was evenly precipitated from water on the surface of disintegrated rock particles in the entire volume of the current green siltstones. Then there was cementation of rocks with Fe hydroxides, carbonates and clay material. After this, the rocks were again compacted under glacial loading. As the second option, it can be assumed that the green siltstones replace lenticular accumulations of organic matter with a high content of U adsorbed from groundwater. The place where 5 samples were taken for this study was the sea shelf of the Mikulinian (Eemian) Sea 115–130 ka ago and marine sediments up to 50–70 m thick contained a large amount (up to 10%) of organic residues of iodine-containing algae. Organic matter could have entered sediments of the upper part of the Vendian Formation during the diagenesis of marine precipitation [42].
5.5. Retardation Factor
Retardation factor is determined by the formula [6,43]:
where ; -solute concentration of U in the stationary solid, µg/kg [44]; -measured concentration of 238U in solution at the point of sampling, µg/kg; -mineral density, g/cm3; -pore fluid density, ~1 g/cm3; -porosity.
For the study area, the following values of the parameters included in Equation (2) were previously obtained: = 5 µg/kg (from 23 definitions of uranium concentrations in fresh water in a Vendian aquifer [28]; n = 0.23 (from 52 determinations) and ρm = 2.75 g/cm3 (from 52 determinations); = 9.2065 [27].
can be taken as the average value of U content in adsorbed elements of the red siltstones = 23 µg/kg (Table 3), because green siltstones occupy an insignificant volume in the deposits of the Padun formation of the Vendian compared to red siltstones. However, this value characterizes the entire volume of rock that was crushed before the experiments. Under natural conditions, the movement of water in siltstones and sandstones occurs along pores and cracks, characterized by the value of open porosity. From this open volume, uranium adsorption occurs, which determines the value of the retardation factor. Therefore, as a first approximation, we can take:
Calculations using Equation (3) give the value of the retardation factor 10.73.
In Malov [27], the average value of the ratio R:p = 24 was determined for a Vendian aquifer, where p is the recoil loss factor. That is, at R = 10.73, the value of p will be 0.45.
The obtained values (10.73 and 0.45) apparently characterize the upper limits of R and p, repectively.
6. Conclusions
The partial extraction procedure was used to reconstruct the redistribution processes of uranium isotopes in certain mineral phases of the aquifer of sandy-clay deposits of the Vendian.
Red siltstones and sandstones are depleted in uranium in relatively readily soluble mineral phases. The concentration of sorbed uranium was established as 15.8 ± 2.1–30.5 ± 3.9 μg/kg, which accounts for 0.35–1.6% of its total amount in rocks of this type. Carbonate minerals contain even less uranium: 7 ± 1–19.8 ± 2.5 μg/kg, i.e., 0.16–1% and in iron hydroxides and the most readily soluble aluminosilicates, uranium concentrations are in the range 168 ± 24–212 ± 28 μg/kg but the most insoluble fraction contains 1.65 ± 0.21–4.32 ± 0.45 mg/kg of uranium. In green siltstones, the concentration of adsorbed uranium is much higher: 106 ± 14–364 ± 43 μg/kg (0.56–4.37%), carbonate minerals and amorphous iron oxides contain 1.91 ± 0.21–2.34 ± 0.26 mg/kg of uranium and in aluminosilicates and a clay/quartz resistate, uranium concentrations are 5.6 ± 0.5–16.8 ± 1.4 mg/kg. Such a distribution of uranium in various types of rocks is consistent with the earlier assumption about the removal of uranium from red siltstones in the last 0.9 Ma and its conservation in green siltstones.
Elevated values of 234U:238U activity ratio prevail in the adsorbed material and iron hydroxides. In aluminosilicates and clay/quartz resistate, the values decrease. This indicates the replacement of primary sedimentogenic uranium by secondary hydrogenic uranium adsorbed on the surface of minerals and coprecipitated with iron hydroxides.
The results obtained made it possible to carry out preliminary quantitative estimates of the retardation factor and recoil loss factor of uranium in the groundwater of siltstones and sandstones of the Vendian aquifer. In the future, it is planned to investigate the effect of changes in the boundary conditions (t, pH, Eh, TDS) of the low mineralized fresh natural groundwater on partial uranium extraction.
At the same time, some uncertainty in the interpretation of the high U contents and high 234U:238U activity ratios in green siltstones obtained in this work should be noted. This requires additional research on a more representative number of samples. Also, the relationship between the extraction solutions and the mineral phases has not yet been precisely established. The mineral phases that were dissolved during the partial extraction procedure should be examined using X-ray powder diffraction (XRD) analysis and scanning electron microscopy (SEM).
Author Contributions
Conceptualization, formal analysis, writing—original draft preparation, A.I.M.; methodology and investigation, S.B.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
This work was supported by the Russian Ministry of Education and Science (project no. № AAAA-A19-119011890018-3), the UB RAS (project no. AAAA-A18-118012390242-5), the Russian Foundation for Basic Research (projects no. 20-05-00045_A, 18-05-60151_Arctic; no. 18-05-01041_A). The authors are grateful to T.N. Markova for the description of the samples of the studied rocks was performed on five thin sections of core samples.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Appendix A. Description of the Thin Sections of Core Samples
Thin sections of core samples (n = 5) were prepared in epoxy resin. Thin sections were examined under transmitted, polarized, and reflected light using optical microscopy to determine the minerals and the textural features of the rocks.
1. Green siltstones 66.7–66.8 m
The sandy siltstone has a light greenish-gray color. The texture of the rock is heterogeneous, due to the chaotic arrangement of clastic grains and the presence of spots up to 0.4 mm in size with an iron-carbonate aggregate. Clastic material is 70–80% and is represented by grains of quartz, feldspar and quartzite rocks; the composition is dominated by quartz grains. Plagioclases prevail among feldspars where plagioclases and potassium feldspars are diagnosed. The grain size varies from 0.02 to 0.15 mm, grains up to 0.2 mm in size are rarely found and the surface of the grains is corrosive. Single grains of accessory minerals are noted - tourmaline, ilmenite, epidote, zircon. On separate grains of quartz and feldspar, a regenerative rim is fixed. Some fragments of quartz and feldspar are sericitized. In the rock, single plates of colorless and colored mica up to 0.15–0.25 mm in size are diagnosed; this mica is diagenetic. Film-pore clay-ferruginous cement predominates, and cementation due to regeneration is also fixed. Marks, lenses (not exceeding 0.4–0.5 mm in size) with limonite-goethite and ferruginous-carbonate cement are noted. Carbonate is represented by calcite and it is formed later than limonite-goethite aggregate. Individual detrital grains are partially replaced by a ferruginous carbonate aggregate. Limonite-goethite and carbonate aggregates in cement are epigenetic. Most likely, compaction of sedimentary material, hydromica of cement in siltstone, and a high content of deformed plates are associated with dynamic changes. That is, the structural adjustment of cement is due to the influence of tectonic processes and hydrothermal solutions in the process of formation of kimberlite fields.
2. Red sandstones 75.2–75.3 m
Oligomictic fine-grained silty sandstone has brown color with light streaks. Rounded spots 1.0–2.0 mm in size with carbonate cement are fixed in the rock; their location is subparallel. The slanting texture due to particle size distribution is not clearly expressed. In some areas, a random distribution of clastic material is noted. Clastic material is represented by grains of quartz, feldspar and quartzite rocks; the composition is dominated by quartz grains. The size of clastic grains varies from 0.03–0.04 mm to 0.2–0.25 mm. Grain rounding is mostly good and medium. The grain surface is corrosive; regenerative rim is fixed on separate fragments of quartz and feldspar. Separate fragments of quartz and feldspar are sericitized. There are single plates of green mica up to 0.12–0.15 mm in size, which fill individual pores and sometimes partially replace detrital grains. Most likely this mica is epigenetic. Film-pore clay-ferruginous cement predominates, and cementation due to regeneration is also fixed. Spots with limonite-goethite and ferruginous-carbonate cement are noted. Carbonate is represented by calcite and is formed later than limonite-goethite aggregate. Individual detrital grains are partially replaced by a ferruginous carbonate aggregate. Limonite-goethite and carbonate aggregates in cement are epigenetic.
3. Variegated siltstones 83.9–84 m
Siltstone has indistinctly expressed oblique and banded textures. Striping does not coincide with layering; alternation of brown and gray, greenish-gray rocks does not form layers. Cross-bedding is due to the presence of argillite microlenses. The composition of this rock does not differ from the sandstones and siltstones that are described in the above.
4. Red siltstones 94–94.1 m
Oligomictic siltstone of brown color with indistinctly expressed cross-layered texture. The oblique texture is due to a poorly defined granulometric sorting of clastic material and the presence of argillite microlenses. Breccia is due to the presence of areas with a random distribution of clastic material, which is associated with sedimentation processes. The presence of sites with limonite-goethite cement is due to epigenetic processes. Clastic material, as in previous thin sections, is represented by grains of quartz, feldspar and quartzite rocks, quartz grains dominate in the composition. The size of clastic grains varies from 0.03-0.04 mm to 0.15-0.2 mm. Grain rounding is mostly average and good. The grain surface is corrosive; a regenerative rim is fixed on separate fragments of quartz and feldspar. Some fragments of quartz and feldspar are sericitized. Rare plates of colorless mica, which are diagenetic, are fixed in this strain. By the nature of the filling, the cement is not continuous, the number of hollow pores is insignificant. Film-pore clay-ferruginous cement predominates, and cementation due to regeneration is also fixed. Spots with limonite-goethite and ferruginous-carbonate cement are noted. Carbonate is represented by calcite and it is later than limonite-goethite aggregate. Limonite-goethite and carbonate aggregates in cement are epigenetic.
5. Green siltstones 99.5–99.6 m
Oligomictic siltstone sandy with indistinctly expressed oblique and banded textures. The color of the strain is light greenish gray. In terms of composition and structural-textural features, siltstone does not differ from the rocks that are described in the above.
References
- Chkir, N.; Guendouz, A.; Zouari, K.; Hadj Ammar, F.; Moulla, A.S. Uranium isotopes in groundwater from the continental intercalaire aquifer in Algerian Tunisian Sahara (northern Africa). J. Environ. Radioact. 2009, 100, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Dhaoui, Z.; Chkir, N.; Zouari, K.; Ammar, F.H.; Agoune, A. Investigation of uranium geochemistry along groundwater flow path in the Continental Intercalaire aquifer (Southern Tunisia). J. Environ. Radioact. 2016, 157, 67–76. [Google Scholar] [CrossRef]
- Priestley, S.C.; Payne, T.E.; Harrison, J.J.; Post, V.E.A.; Shand, P.; Love, A.J.; Wohling, D.L. Use of U-isotopes in exploring groundwater flow and inter-aquifer leakage in the south-western margin of the Great Artesian Basin and Arckaringa Basin, central Australia. Appl. Geochem. 2018, 98, 331–344. [Google Scholar] [CrossRef]
- Suksi, J.; Rasilainen, K.; Marcos, N. U isotopic fractionation—A process characterising groundwater systems. In Uranium in the Environment; Merkel, B.J., Hasche-Berger, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 683–690. [Google Scholar]
- Chabaux, F.; Bourdon, B.; Riotte, J. U-series geochemistry in weathering profiles, riverwaters and lakes. In U/Th Series Radionuclides in Aquatic Systems, Radioactivity in the Environment; Krishnaswami, S., Cochran, J.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; Volume 13, pp. 49–104. [Google Scholar]
- Porcelli, D. Investigating groundwater processes using U- and Th-series nuclides. Radioact. Environ. 2008, 13, 105–153. [Google Scholar]
- Maher, K.; DePaolo, D.J.; Lin, J.C.F. Rates of silicate dissolution in deep-sea sediment: In situ measurement using U-234/U-238 of pore fluids. Geochim. Cosmochim. Acta 2004, 68, 4629–4648. [Google Scholar] [CrossRef]
- Ivanovich, M.; Fröhlich, K.; Hendry, M.J. Uranium-series radio nuclides in fluids and solids, Milk River aquifer, Alberta, Canada. Appl. Geochem. 1991, 6, 405–418. [Google Scholar] [CrossRef]
- Chabaux, F.; Dequincey, O.; Leveque, J.J.; Leprun, J.C.; Clauer, N.; Riotte, J.; Paquet, H. Tracing and dating recent chemical transfers in weathering profiles by trace-element geochemistry and U-238-U-234-Th-230 disequilibria: The example of the Kaya lateritic toposequence (Burkina-Faso). Comptes Rendus Geosci. 2003, 335, 1219–1231. [Google Scholar] [CrossRef]
- Dosseto, A.; Bourdon, B.; Turner, S. Uranium-series isotopes in river materials: Insights into the timescales of erosion and sediment transport. Earth Planet. Sci. Lett. 2008, 265, 1–17. [Google Scholar] [CrossRef]
- Vigier, N.; Bourdon, B. Chapter 27: Constraining Rates of Chemical and Physical Erosion Using U-Series Radionuclides. In Handbook of Environmental Isotope Geochemistry, Advances in Isotope Geochemistry; Baskaran, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 553–571. [Google Scholar]
- Duff, M.C.; Coughlin, J.U.; Hunter, D.B. Uranium co-precipitation with iron oxide minerals. Geochim. Cosmochim. Acta 2002, 66, 3533–3547. [Google Scholar] [CrossRef]
- Giammar, D.E.; Hering, J.G. Timescale for sorption-desorption and surface precipitation of uranyl on goethite. Environ. Sci. Technol. 2001, 35, 3332–3337. [Google Scholar] [CrossRef]
- Singer, D.M.; Maher, K.; Brown, G.E., Jr. Uranyl-chlorite sorption/desorption: Evaluation of different U (VI) sequestration processes. Geochim. Cosmochim. Acta 2009, 73, 5989–6007. [Google Scholar] [CrossRef]
- Tricca, A.; Wasserburg, G.J.; Porcelli, D.; Baskaran, M. The transport of U- and Th-series nuclides in a sandy unconfined aquifer. Geochim. Cosmochim. Acta 2001, 65, 1187–1210. [Google Scholar] [CrossRef]
- Lowson, R.T.; Short, S.A.; Davey, B.G.; Gray, D.J. 234U/238U and 230Th/234U activity ratios in mineral phases of a lateritic weathered zone. Geochim. Cosmochim. Acta 1986, 50, 1697–1702. [Google Scholar] [CrossRef]
- Payne, T.E.; Edis, R.; Fenton, B.R.; Waite, T.D. Comparison of laboratory uranium sorption data with ‘in situ distribution coefficients’ at the Koongarra uranium deposit, Northern Australia. J. Environ. Radioact. 2001, 57, 35–55. [Google Scholar] [CrossRef]
- Dabous, A.A.; Osmond, J.K.; Dawood, Y.H. Uranium/thorium isotope evidence for groundwater history in the Eastern Desert of Egypt. J. Arid Environ. 2002, 50, 343–357. [Google Scholar] [CrossRef]
- Yanase, N.; Nightingale, T.; Payne, T.; Duerden, P. Uranium distribution in mineral phases of rock by sequential extraction procedure. Radiochim. Acta 1991, 52–53, 387–393. [Google Scholar] [CrossRef]
- Malov, A.I. Evolution of the uranium isotopic compositions of the groundwater and rock in the sandy-clayey aquifer. Water 2017, 9, 910. [Google Scholar] [CrossRef]
- DePaolo, D.J.; Maher, K.; Christensen, J.N.; McManus, J. Sediment transport time measured with U-series isotopes: Results from ODP North Atlantic drift site 984. Earth Planet. Sci. Lett. 2006, 248, 394–410. [Google Scholar] [CrossRef]
- Maher, K.; DePaolo, D.J.; Christensen, J.N. U-Sr isotopic speedometer: Fluid flow and chemical weathering rates in aquifers. Geochim. Cosmochim. Acta 2006, 70, 4417–4435. [Google Scholar] [CrossRef]
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 51, 848–851. [Google Scholar] [CrossRef]
- Hall, G.E.M.; Vaive, J.E.; Beer, R.; Hoashi, M. Selective leaches revisited, with emphasis on the amorphous Fe oxyhydroxide phase extraction. J. Geochem. Explor. 1996, 56, 59–78. [Google Scholar] [CrossRef]
- Quevauviller, P.H.; Rauret, G.; López-Sánchez, J.-F.; Rubio, R.; Ure, A.; Muntau, H. Certification of trace metal extractable contents in a sediment reference material (CRM601) following a three-step sequential extraction procedure. Sci. Total Environ. 1997, 205, 223–234. [Google Scholar] [CrossRef]
- Sutherland, R.A.; Tack, F.M.G.; Tolosa, C.A.; Verloo, M.G. Metal Extraction from Road Sediment using Different Strength Reagents: Impact on Anthropogenic Contaminant Signals. Environ. Monit. Assess. 2001, 71–73, 221–242. [Google Scholar] [CrossRef] [PubMed]
- Malov, A.I. Estimation of uranium migration parameters in sandstone aquifers. J. Environ. Radioact. 2016, 153, 61–67. [Google Scholar] [CrossRef]
- Malov, A.I. Evolution of the groundwater chemistry in the coastal aquifers of the south-eastern White Sea area (NW Russia) using 14C and 234U-238U dating. Sci. Total Environ. 2018, 616–617, 1208–1223. [Google Scholar] [CrossRef]
- Andreas, J.N.; Kay, R.L.F. The evolution of enhanced 234U/238U activity ratios for dissolved uranium and groundwater dating. In Proceedings of the 4th International Conference on Geochronology, Cosmochronology and Isotope Geology, Denver, Colorado, 20–25 August 1978; U.S. Geological Survey: Reston, VA, USA, 1978; Volume 78, pp. 11–13. [Google Scholar]
- Fröhlich, K.; Gellermann, R. On the potential use of uranium isotopes for groundwater dating. Chem. Geol. 1987, 65, 67–77. [Google Scholar] [CrossRef]
- Andreas, J.N.; Kay, R.L.F. The U content and 234U/238U activity ratios of dissolved uranium in groundwaters from Triassic sandstones in England. Chem. Geol. 1983, 41, 101–117. [Google Scholar] [CrossRef]
- Stankovsky, A.F.; Verichev, E.M.; Grib, V.P.; Dobeiko, I.P. The Vendian in the Southeastern White Sea Region. Izv. Akad. Nauk SSSR Ser. Geol. 1981, 2, 78–87. [Google Scholar] [CrossRef]
- Stankovsky, A.F.; Verichev, E.M.; Dobeiko, I.P. The Vendian in Southeastern White Sea Region. In The Vendian: Historical-Geological and Paleontological Evidence, Stratigraphy and Geological Processes; Nauka: Moscow, Russia, 1985; Volume 2, pp. 67–76. [Google Scholar]
- Malov, A.I. Water-Rock Interaction in Vendian Sandy-Clayey Rocks of the Mezen Syneclise. Lithol. Miner. Resour. 2004, 39, 345–356. [Google Scholar] [CrossRef]
- Grazhdankin, D.V.; Podkovyrov, V.N.; Maslov, A.V. Paleoclimatic Environments of the Formation of Upper Vendian Rocks on the Belomorian-Kuloi Plateau, Southeastern White Sea Region. Lithol. Miner. Resour. 2005, 40, 232. [Google Scholar] [CrossRef]
- Bonotto, D.M.; Andrews, J.N. The mechanism of 234U/238U activity ratio enhancement in karstic limestone groundwater. Chem. Geol. 1993, 103, 193–206. [Google Scholar] [CrossRef]
- Bonotto, D.M.; Andrews, J.N. The transfer of uranium isotopes 234U and 238U to the waters interacting with carbonates from Mendip Hills area (England). Appl. Radiat. Isot. 2000, 52, 965–983. [Google Scholar] [CrossRef]
- Singh, S.P.; Tack, F.M.G.; Verloo, M.G. Solid-phase distribution of heavy metals as affected by single reagent extraction in dredged sediment derived surface soils. Chem. Speciat. Bioavailab. 1996, 8, 37–43. [Google Scholar] [CrossRef]
- Malyshev, V.I.; Bakhur, A.E.; Manuylova, L.I. Methods for Measuring the Volumetric Activity of Uranium Isotopes (234, 238) in Natural Water Sample Alpha Spectrometry with Radiochemical Separation; VIMS: Moscow, Russia, 1999. [Google Scholar]
- Fröhlich, K. Dating of old groundwater using uranium isotopes—Principles and applications. In Isotope Methods for Dating Old Groundwater; IAEA: Vienna, Austria, 2013; pp. 153–178. [Google Scholar]
- Malyshev, V.I.; Bakhur, A.E.; Manuylova, L.I. Methods for Measuring the Specific Activity of Uranium Isotopes (234, 238) in Soils, Sediments, Rocks and Building Materials Alpha Spectrometry with Radiochemical Separation; VIMS: Moscow, Russia, 1999. [Google Scholar]
- Malov, A.I. Application of Geological Benchmarks for Determining Groundwater Residence Time in the Aquifer Based on Uranium Isotope Data: Evidence from the Severnaya Dvina Basin. Lithol. Miner. Resour. 2013, 48, 254–265. [Google Scholar] [CrossRef]
- Krishnaswami, S.; Graustein, W.C.; Turekian, K.K.; Dowd, J.F. Radium, thorium and radioactive lead isotopes in groundwaters: Application to the in situ determination of adsorption-desorption rate constants and retardation factors. Water Resour. Res. 1982, 18, 1663–1675. [Google Scholar] [CrossRef]
- Pearson, F.J.; Norowha, C.J.; Andrews, R.W. Mathematical modelling of the distribution of natural 14C, 234U, and 238U in a regional groundwater system. Radiocarbon 1983, 25, 291–300. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).