Post-Treatment of Nanofiltration Polyamide Membrane through Alkali-Catalyzed Hydrolysis to Treat Dyes in Model Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of the Post-Treated Membranes
2.3. Confirmation of Physicochemical Properties of the Membranes
2.3.1. Surface Characterizations
2.3.2. Membrane Performances
3. Results and Discussion
3.1. Intrinsic Properties of the NF 90 Membrane
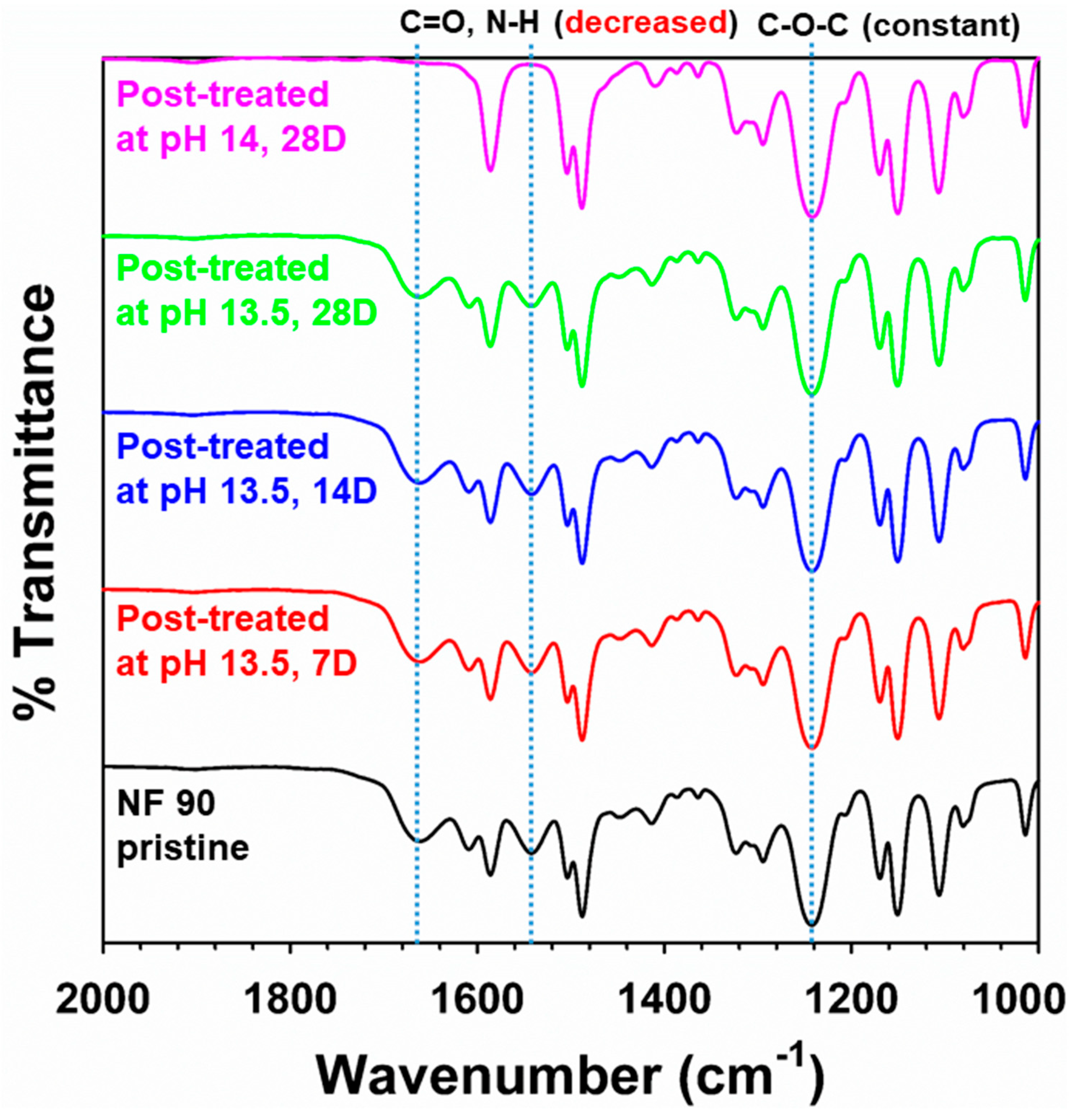
3.2. Effect of Alkali-Catalyzed Hydrolysis on the NF 90 Membrane
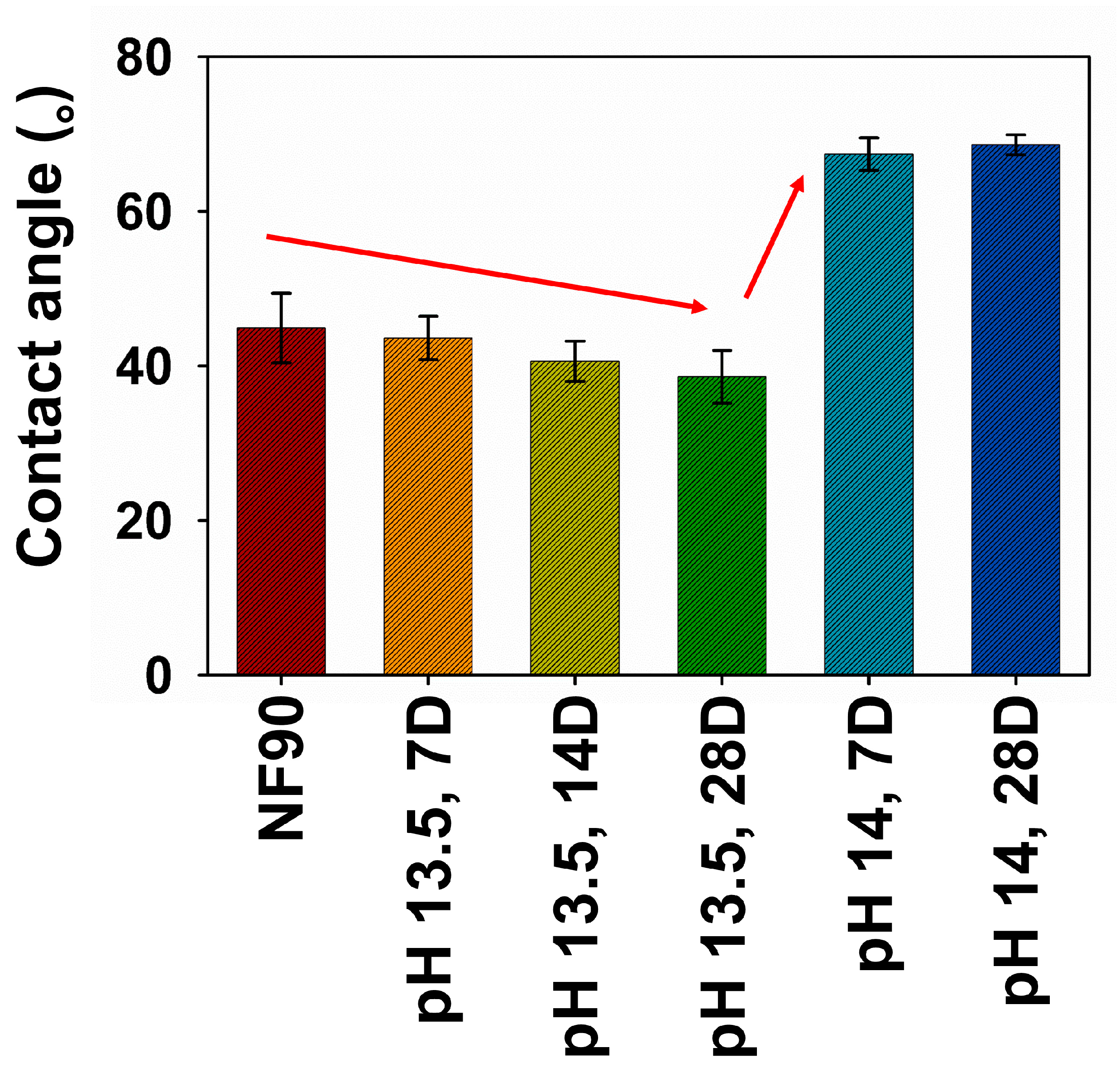
3.2.1. Change of Surface Properties
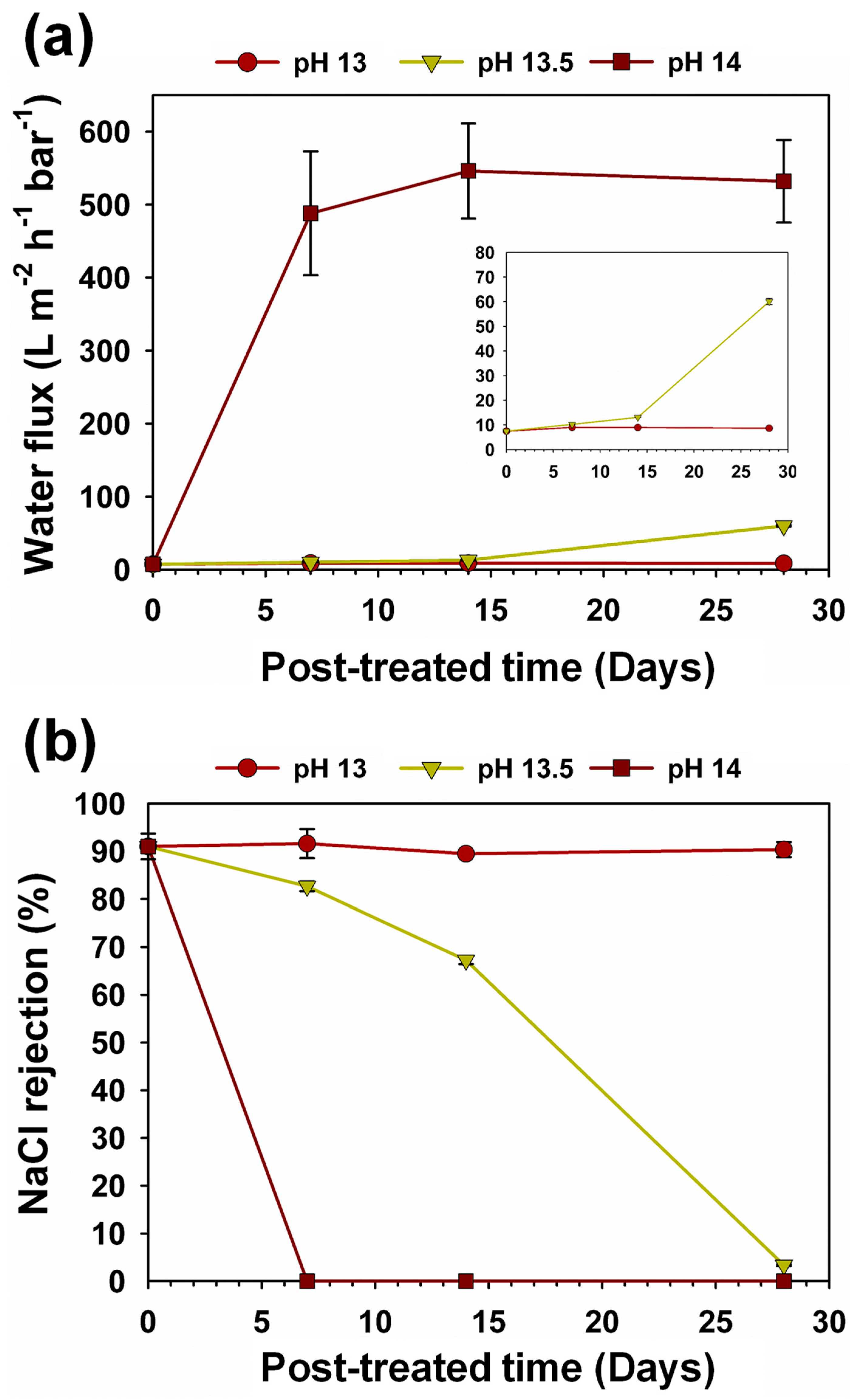
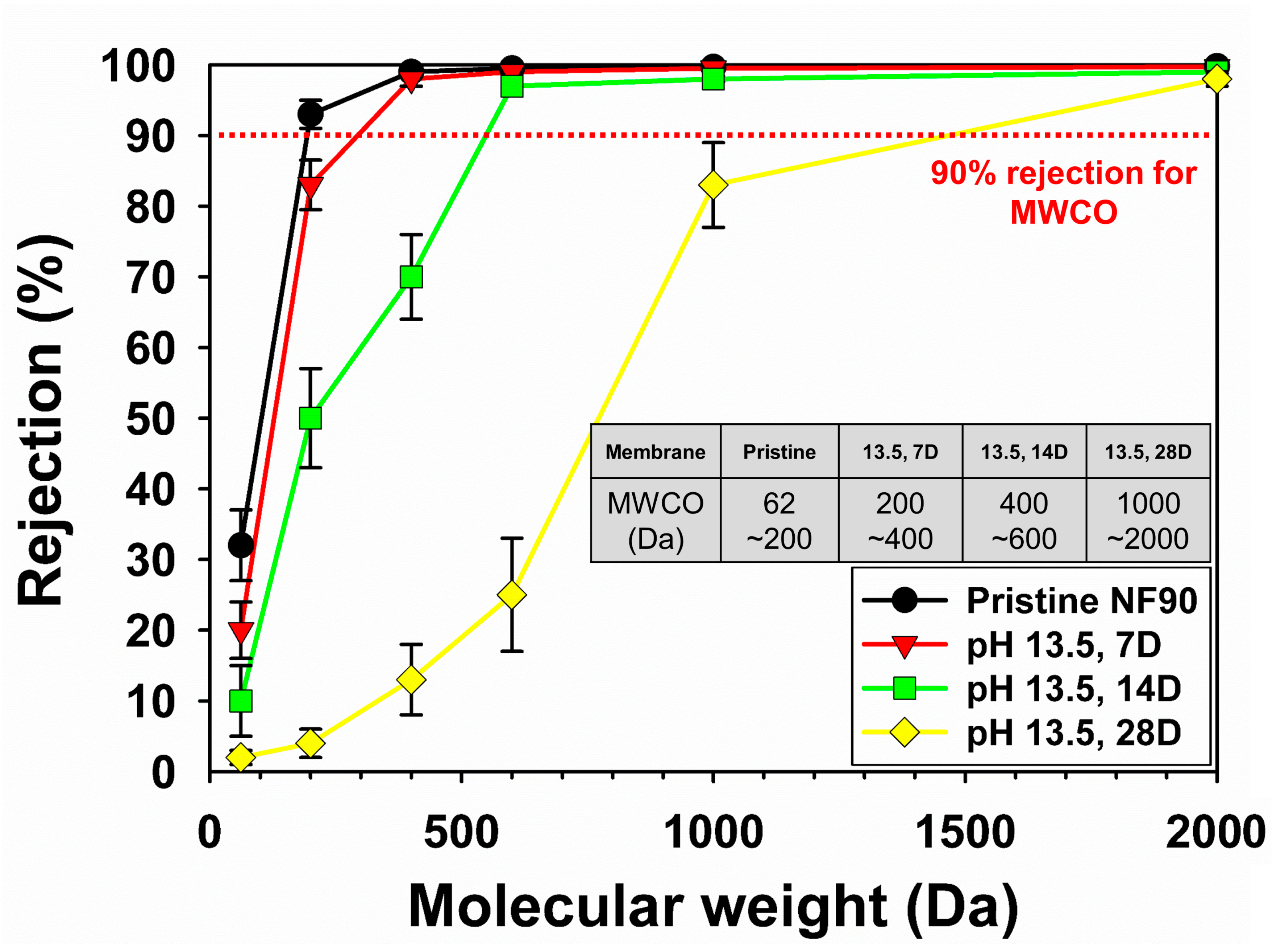
3.2.2. Change of Membrane Performances
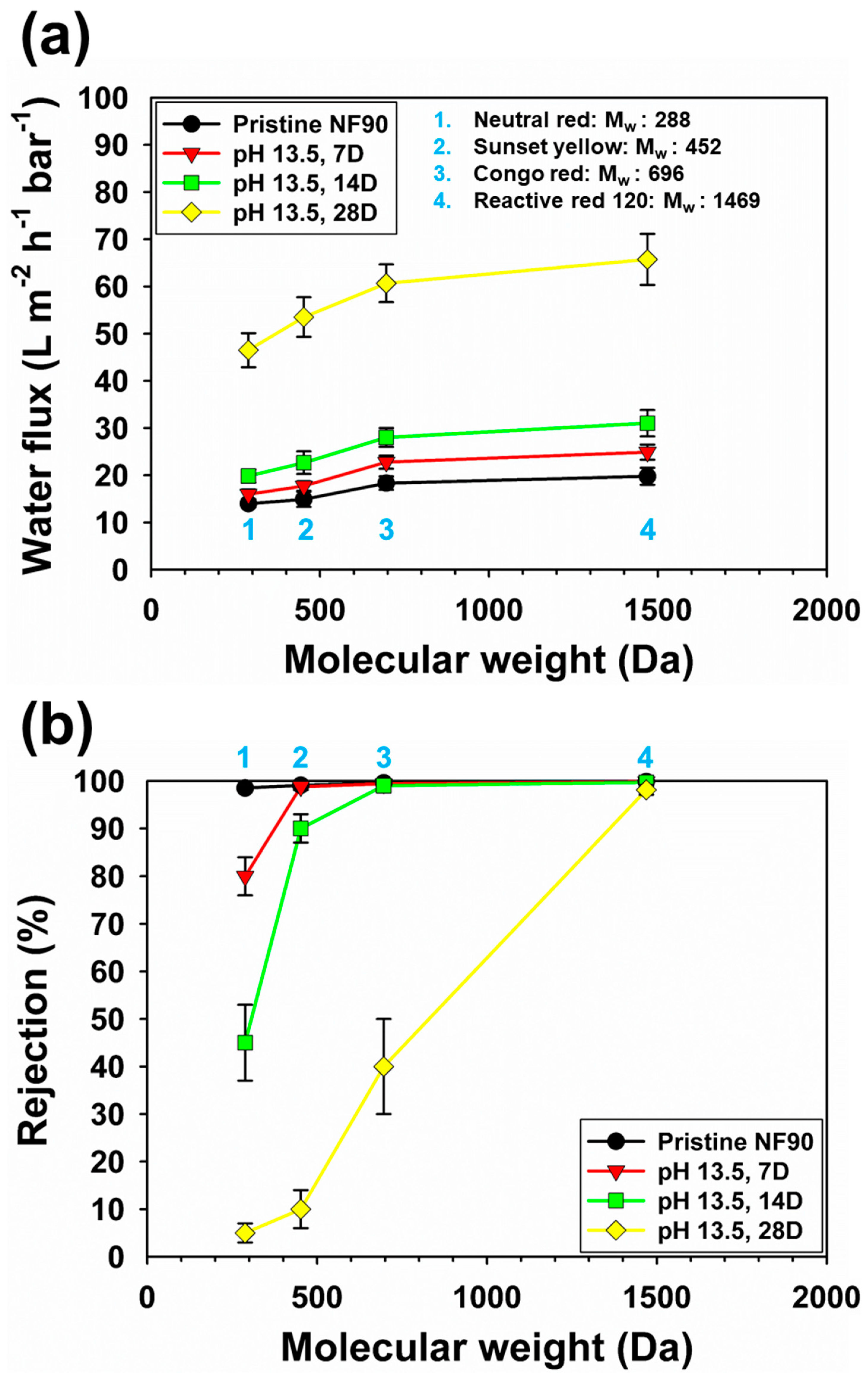
3.3. Removal of Dyes in Model Wastewater by the NF 90 and Post-Treated Membranes
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yu, S.; Chen, Z.; Cheng, Q.; Lü, Z.; Liu, M.; Gao, C. Application of thin-film composite hollow fiber membrane to submerged nanofiltration of anionic dye aqueous solutions. Sep. Purif. Technol. 2012, 88, 121–129. [Google Scholar] [CrossRef]
- Jun, B.-M.; Lee, H.K.; Park, Y.-I.; Kwon, Y.-N. Degradation of full aromatic polyamide NF membrane by sulfuric acid and hydrogen halides: Change of the surface/permeability properties. Polym. Degrad. Stab. 2019, 162, 1–11. [Google Scholar] [CrossRef]
- Zhou, C.; Shi, Y.; Sun, C.; Yu, S.; Liu, M.; Gao, C. Thin-film composite membranes formed by interfacial polymerization with natural material sericin and trimesoyl chloride for nanofiltration. J. Membr. Sci. 2014, 471, 381–391. [Google Scholar] [CrossRef]
- Jun, B.-M.; Nguyen, T.P.N.; Ahn, S.-H.; Kim, I.-C.; Kwon, Y.-N. The application of polyethyleneimine draw solution in a combined forward osmosis/nanofiltration system. J. Appl. Polym. Sci. 2015, 132, 42198–42206. [Google Scholar] [CrossRef]
- Mohammad, A.W.; Teow, Y.H.; Ang, W.L.; Chung, Y.T.; Oatley-Radcliffe, D.L.; Hilal, N. Nanofiltration membranes review: Recent advances and future prospects. Desalination 2015, 356, 226–254. [Google Scholar] [CrossRef]
- Rho, H.; Chon, K.; Cho, J. An autopsy study of a fouled reverse osmosis membrane used for ultrapure water production. Water 2019, 11, 1116. [Google Scholar] [CrossRef]
- Liu, M.; Chen, Q.; Lu, K.; Huang, W.; Lü, Z.; Zhou, C.; Yu, S.; Gao, C. High efficient removal of dyes from aqueous solution through nanofiltration using diethanolamine-modified polyamide thin-film composite membrane. Sep. Purif. Technol. 2017, 173, 135–143. [Google Scholar] [CrossRef]
- Lü, Z.; Hu, F.; Li, H.; Zhang, X.; Yu, S.; Liu, M.; Gao, C. Composite nanofiltration membrane with asymmetric selective separation layer for enhanced separation efficiency to anionic dye aqueous solution. J. Hazard. Mater. 2019, 368, 436–443. [Google Scholar] [CrossRef]
- Jun, B.-M.; Kim, S.; Heo, J.; Her, N.; Jang, M.; Park, C.M.; Yoon, Y. Enhanced sonocatalytic degradation of carbamazepine and salicylic acid using a metal-organic framework. Ultrason. Sonochem. 2019, 56, 174–182. [Google Scholar] [CrossRef]
- Jun, B.-M.; Kim, S.; Kim, Y.; Her, N.; Heo, J.; Han, J.; Jang, M.; Park, C.M.; Yoon, Y. Comprehensive evaluation on removal of lead by graphene oxide and metal organic framework. Chemosphere 2019, 231, 82–92. [Google Scholar] [CrossRef]
- Rho, H.; Chon, K.; Park, J.; Cho, J. Rapid and effective isolation of dissolved organic matter using solid-phase extraction cartridges packed with Amberlite XAD 8/4 resins. Water 2019, 11, 67. [Google Scholar] [CrossRef]
- Joseph, L.; Jun, B.-M.; Flora, J.R.V.; Park, C.M.; Yoon, Y. Removal of heavy metals from water sources in the developing world using low-cost materials: A review. Chemosphere 2019, 229, 142–159. [Google Scholar] [CrossRef] [PubMed]
- Jun, B.-M.; Kim, S.; Heo, J.; Park, C.M.; Her, N.; Jang, M.; Huang, Y.; Han, J.; Yoon, Y. Review of MXenes as new nanomaterials for energy storage/delivery and selected environmental applications. Nano Res. 2019, 12, 471–487. [Google Scholar] [CrossRef]
- Joseph, L.; Jun, B.-M.; Jang, M.; Park, C.M.; Muñoz-Senmache, J.C.; Hernández-Maldonado, A.J.; Heyden, A.; Yu, M.; Yoon, Y. Removal of contaminants of emerging concern by metal-organic framework nanoadsorbents: A review. Chem. Eng. J. 2019, 369, 928–946. [Google Scholar] [CrossRef]
- Dasgupta, J.; Sikder, J.; Mandal, T.; Adhikari, U. Reactive red 120 retention through ultrafiltration enhanced by synthetic and natural polyelectrolytes. J. Hazard. Mater. 2015, 299, 192–205. [Google Scholar] [CrossRef] [PubMed]
- Ayyavoo, J.; Nguyen, T.P.N.; Jun, B.-M.; Kim, I.-C.; Kwon, Y.-N. Protection of polymeric membranes with antifouling surfacing via surface modifications. Colloids Surf. A Physicochem. Eng. Asp. 2016, 506, 190–201. [Google Scholar] [CrossRef]
- Nguyen, T.P.N.; Jun, B.-M.; Lee, J.H.; Kwon, Y.-N. Comparison of integrally asymmetric and thin film composite structures for a desirable fashion of forward osmosis membranes. J. Membr. Sci. 2015, 495, 457–470. [Google Scholar] [CrossRef]
- Jun, B.-M.; Kim, S.H.; Kwak, S.K.; Kwon, Y.-N. Effect of acidic aqueous solution on chemical and physical properties of polyamide NF membranes. Appl. Surf. Sci. 2018, 444, 387–398. [Google Scholar] [CrossRef]
- Nguyen, T.P.N.; Jun, B.-M.; Kwon, Y.-N. The chlorination mechanism of integrally asymmetric cellulose triacetate (CTA)-based and thin film composite polyamide-based forward osmosis membrane. J. Membr. Sci. 2017, 523, 111–121. [Google Scholar] [CrossRef]
- Jun, B.-M.; Nguyen, T.P.N.; Kim, Y.-K.; Lee, H.K.; Kwon, Y.-N. Surface modification of TFC FO membrane using N-isopropylacrylamide (NIPAM) to enhance fouling resistance and cleaning efficiency. Desalin. Water Treat. 2017, 65, 11–21. [Google Scholar] [CrossRef]
- Guo, Y.-S.; Weng, X.-D.; Wu, B.; Mi, Y.-F.; Zhu, B.-K.; Ji, Y.-L.; An, Q.-F.; Gao, C.-J. Construction of nonfouling nanofiltration membrane via introducing uniformly tunable zwitterionic layer. J. Membr. Sci. 2019, 583, 152–162. [Google Scholar] [CrossRef]
- Jun, B.-M.; Lee, H.K.; Kwon, Y.-N. Acid-catalyzed hydrolysis of semi-aromatic polyamide NF membrane and its application to water softening and antibiotics enrichment. Chem. Eng. J. 2018, 332, 419–430. [Google Scholar] [CrossRef]
- Cohen, T.; Lipowitz, J. Acid-catalyzed amide hydrolysis assisted by a neighboring amide group. J. Am. Chem. Soc. 1964, 86, 5611–5616. [Google Scholar] [CrossRef]
- Ma, Q.; Shuler, P.J.; Aften, C.W.; Tang, Y. Theoretical studies of hydrolysis and stability of polyacrylamide polymers. Polym. Degrad. Stab. 2015, 121, 69–77. [Google Scholar] [CrossRef]
- Han, J.; Jun, B.-M.; Heo, J.; Lee, G.; Yoon, Y.; Park, C.M. Highly efficient organic dye removal from waters by magnetically recoverable La2O2CO3/ZnFe2O4-reduced graphene oxide nanohybrid. Ceram. Int. 2019, in press. [Google Scholar] [CrossRef]
- Jun, B.-M.; Heo, J.; Park, C.M.; Yoon, Y. Comprehensive evaluation of the removal mechanism of carbamazepine and ibuprofen by metal organic framework. Chemosphere 2019, 235, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Jun, B.-M.; Heo, J.; Kim, S.; Yoon, Y.; Park, C.M. Heterogeneous sonocatalytic degradation of an anionic dye in aqueous solution using a magnetic lanthanum dioxide carbonate-doped zinc ferrite-reduced graphene oxide nanostructure. Ecotox. Environ. Saf. 2019, 182, 109396–109404. [Google Scholar] [CrossRef]
- Jun, B.-M.; Hwang, H.S.; Heo, J.; Han, J.; Jang, M.; Sohn, J.; Park, C.M.; Yoon, Y. Removal of selected endocrine-disrupting compounds using Al-based metal organic framework: Performance and mechanism of competitive adsorption. J. Ind. Eng. Chem. 2019, in press. [Google Scholar] [CrossRef]
- Han, S.-W.; Kim, W.; Lee, Y.; Jun, B.-M.; Kwon, Y.-N. Investigation of Hydrate-induced Ice Desalination (HIID) and its application to a pretreatment of reverse osmosis (RO) process. Desalination 2016, 395, 8–16. [Google Scholar] [CrossRef]
- Nguyen, T.P.N.; Jun, B.-M.; Park, H.G.; Han, S.-W.; Kim, Y.-K.; Lee, H.K.; Kwon, Y.-N. Concentration polarization effect and preferable membrane configuration at pressure-retarded osmosis operation. Desalination 2016, 389, 58–67. [Google Scholar] [CrossRef]
- Tang, C.Y.; Kwon, Y.-N.; Leckie, J.O. Effect of membrane chemistry and coating layer on physiochemical properties of thin film composite polyamide RO and NF membranes: I. FTIR and XPS characterization of polyamide and coating layer chemistry. Desalination 2009, 242, 149–167. [Google Scholar] [CrossRef]
- Tu, K.L.; Nghiem, L.D.; Chivas, A.R. Coupling effects of feed solution pH and ionic strength on the rejection of boron by NF/RO membranes. Chem. Eng. J. 2011, 168, 700–706. [Google Scholar] [CrossRef]
- Mänttäri, M.; Pihlajamäki, A.; Nyström, M. Effect of pH on hydrophilicity and charge and their effect on the filtration efficiency of NF membranes at different pH. J. Membr. Sci. 2006, 280, 311–320. [Google Scholar] [CrossRef]
- Harrison Catherine, J.; Le Gouellec Yann, A.; Cheng Robert, C.; Childress Amy, E. Bench-scale testing of nanofiltration for seawater desalination. J. Environ. Eng. 2007, 133, 1004–1014. [Google Scholar] [CrossRef]
- Wamser, C.C.; Gilbert, M.I. Detection of surface functional group asymmetry in interfacially-polymerized films by contact angle titrations. Langmuir 1992, 8, 1608–1614. [Google Scholar] [CrossRef]
- Kim, K.S.; Lee, K.H.; Cho, K.; Park, C.E. Surface modification of polysulfone ultrafiltration membrane by oxygen plasma treatment. J. Membr. Sci. 2002, 199, 135–145. [Google Scholar] [CrossRef]
- Gu, J.-E.; Jun, B.-M.; Kwon, Y.-N. Effect of chlorination condition and permeability of chlorine species on the chlorination of a polyamide membrane. Water Res. 2012, 46, 5389–5400. [Google Scholar] [CrossRef]
- Nikooe, N.; Saljoughi, E. Preparation and characterization of novel PVDF nanofiltration membranes with hydrophilic property for filtration of dye aqueous solution. Appl. Surf. Sci. 2017, 413, 41–49. [Google Scholar] [CrossRef]
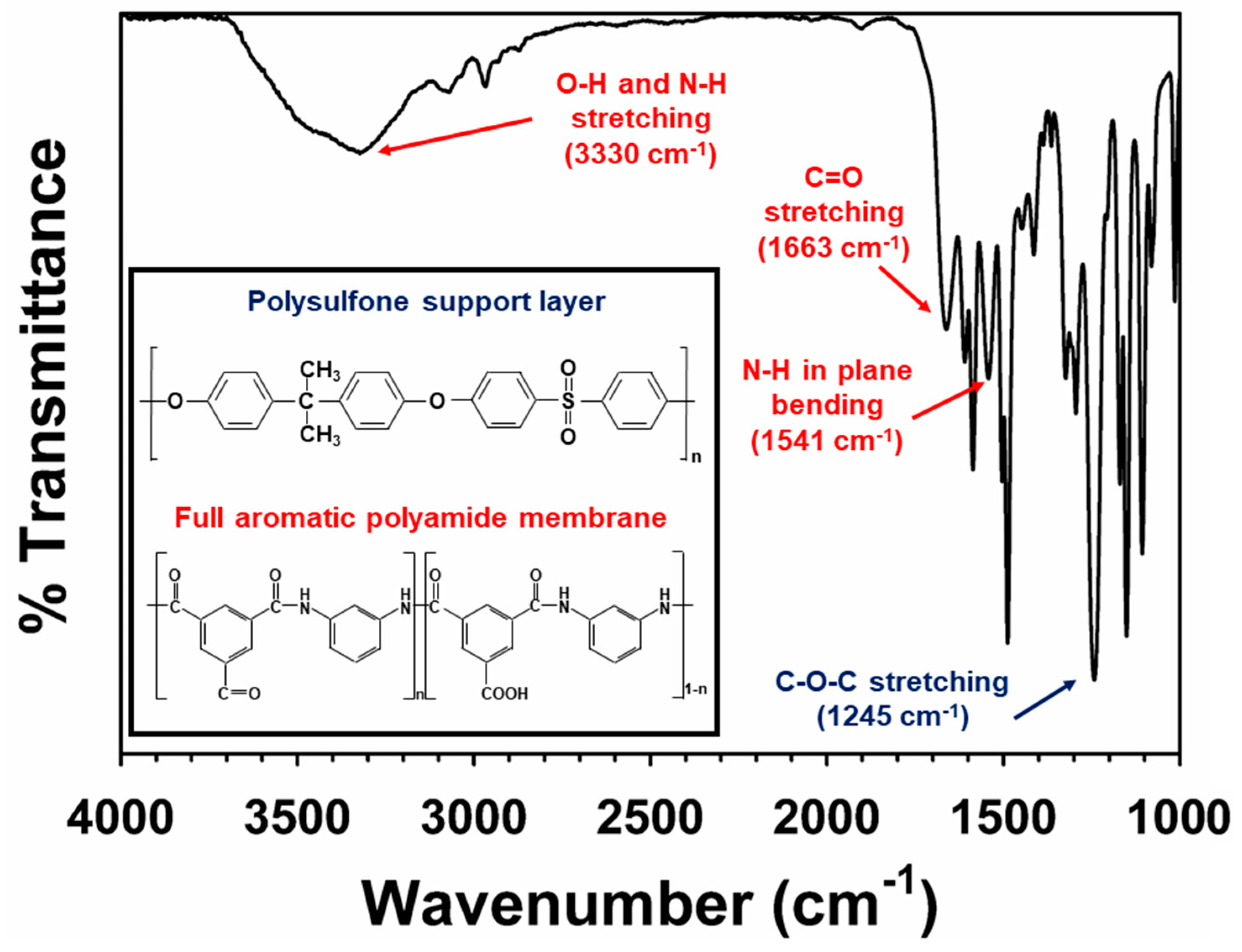

| NF 90 Membrane Property | Previous Study | Measured in This Study (n = 3) |
|---|---|---|
| Pure water permeability (L m2 h−1 bar−1) | 15.8 at 5 bar | 14.6 ± 0.9 at 5 bar |
| NaCl rejection (%) | 85–95 | 92.8 ± 1.3 |
| MgSO4 rejection (%) | >97 | 98.3 ± 0.6 |
| Zeta potential at pH 5.6 (mV) | −20.3 | −15.2 ± 2.1 |
| Contact angle (°) | 48.7 | 44.9 ± 4.5 |
| Membrane | NF 90 Pristine | Post-Treated at pH 13.5 for 7 D | Post-Treated at pH 13.5 for 14 D | Post-Treated at pH 13.5 for 28 D |
|---|---|---|---|---|
| Zeta potential (mV) | −15.2 ± 2.1 | −17.2 ± 1.6 | −18.2 ± 1.2 | −18.9 ± 1.4 |
| Membrane | Target Dye | Experimental Conditions | Water Flux (L m−2 h−1 bar−1) | Rejection (%) | Reference |
|---|---|---|---|---|---|
| NF 90 pristine | Sunset yellow | 50 mg L−1 dye at 5 bar | 14.9 | >99 | This work |
| Post-treated at pH 13.5, 7D | Sunset yellow | 50 mg L−1 dye at 5 bar | 16.0 | >99 | This work |
| DEA-modified PA-TFC | Sunset yellow | 50 mg L−1 dye at 5 bar | 14.8 | 97.5 | [7] |
| PA-TFC | Sunset yellow | 100 mg L−1 dye at 5 bar | 9.3 | >99 | [8] |
| CMCNa/PP composite membrane | Sunset yellow | 100 mg L−1 dye at 0.8 bar | 8.6 | 82.2 | [1] |
| Sericin-TMC membrane | Sunset yellow | 100 mg L−1 dye at 5 bar | 12.4 | 95.4 | [3] |
| NF 90 pristine | Congo red | 50 mg L−1 dye at 5 bar | 18.3 | >99 | This work |
| Post-treated at pH 13.5, 14D | Congo red | 50 mg L−1 dye at 5 bar | 28.0 | >99 | This work |
| NF 270 pristine | Congo red | 50 mg L−1 dye at 5 bar | 22.3 | >99 | This work |
| DEA-modified PA-TFC | Congo red | 50 mg L−1 dye at 5 bar | 15.7 | >99 | [7] |
| PA-TFC | Congo red | 100 mg L−1 dye at 5 bar | 9.2 | >99 | [8] |
| CMCNa/PP composite membrane | Congo red | 100 mg L−1 dye at 0.8 bar | 7.8 | >99 | [1] |
| Sericin-TMC membrane | Congo red | 100 mg L−1 dye at 5 bar | 12.6 | >99 | [3] |
| NF 90 pristine | Reactive red 120 | 50 mg L−1 dye at 5 bar | 19.8 | >99 | This work |
| Post-treated at pH 13.5, 28D | Reactive red 120 | 50 mg L−1 dye at 5 bar | 65.7 | 98.1 | This work |
| NF 270 pristine | Reactive red 120 | 50 mg L−1 dye at 5 bar | 25.8 | >99 | This work |
| Brij-58 contained PVDF membrane | Reactive red 141 | 15 mg L−1 dye at 6 bar | 5.2 (PWP) | 90.0 | [38] |
| PEI-enhanced UF membrane | Reactive red 120 | 50 mg L−1 dye at 4 bar | 37.0 | 99.0 | [15] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jun, B.-M.; Yoon, Y.; Park, C.M. Post-Treatment of Nanofiltration Polyamide Membrane through Alkali-Catalyzed Hydrolysis to Treat Dyes in Model Wastewater. Water 2019, 11, 1645. https://doi.org/10.3390/w11081645
Jun B-M, Yoon Y, Park CM. Post-Treatment of Nanofiltration Polyamide Membrane through Alkali-Catalyzed Hydrolysis to Treat Dyes in Model Wastewater. Water. 2019; 11(8):1645. https://doi.org/10.3390/w11081645
Chicago/Turabian StyleJun, Byung-Moon, Yeomin Yoon, and Chang Min Park. 2019. "Post-Treatment of Nanofiltration Polyamide Membrane through Alkali-Catalyzed Hydrolysis to Treat Dyes in Model Wastewater" Water 11, no. 8: 1645. https://doi.org/10.3390/w11081645
APA StyleJun, B.-M., Yoon, Y., & Park, C. M. (2019). Post-Treatment of Nanofiltration Polyamide Membrane through Alkali-Catalyzed Hydrolysis to Treat Dyes in Model Wastewater. Water, 11(8), 1645. https://doi.org/10.3390/w11081645






