Long-Term Changes in the Zooplankton Community of Lake Maggiore in Response to Multiple Stressors: A Functional Principal Components Analysis
Abstract
1. Introduction
2. Material and Methods
2.1. Study Area
2.2. Data Collection
2.3. Smoothing Data through Penalized B-Spline Expansions
2.4. Functional Principal Components Analysis (FPCA)
2.5. Describing Lake Maggiore’s Dynamic by Functional Data Analysis
3. Results
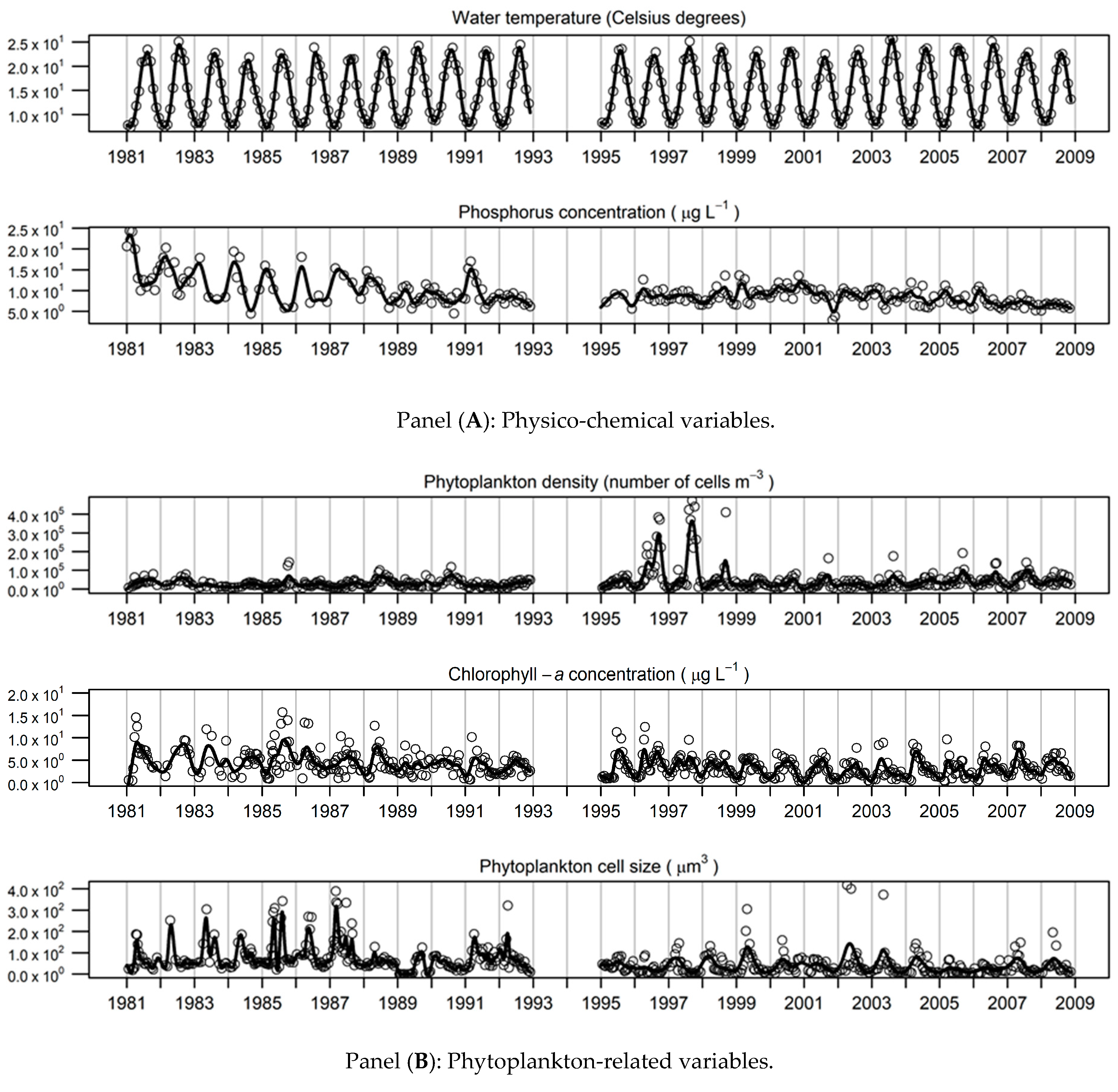
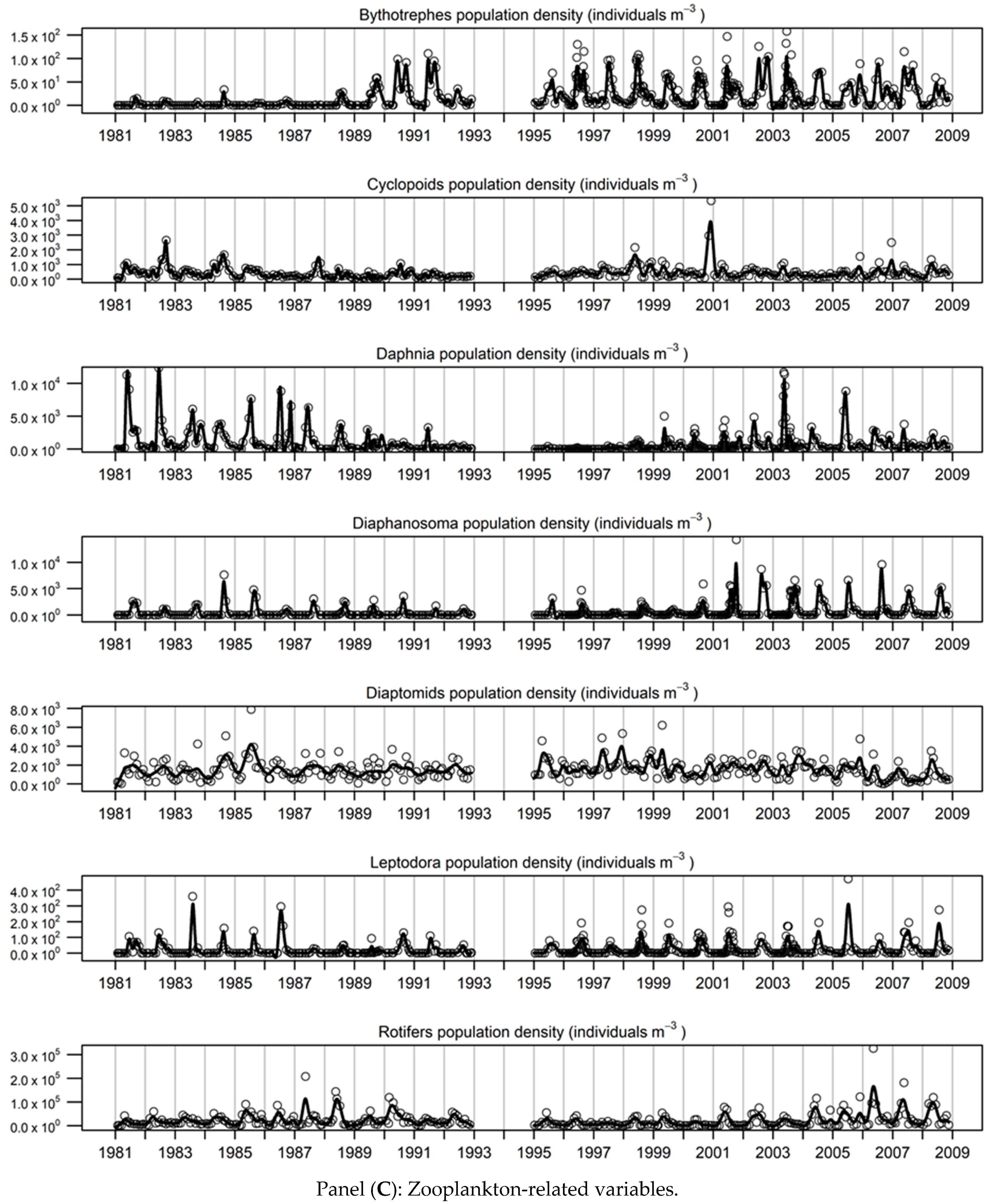
3.1. Long-Term Limnological Change
3.2. Description of the Smoothed Time-Series
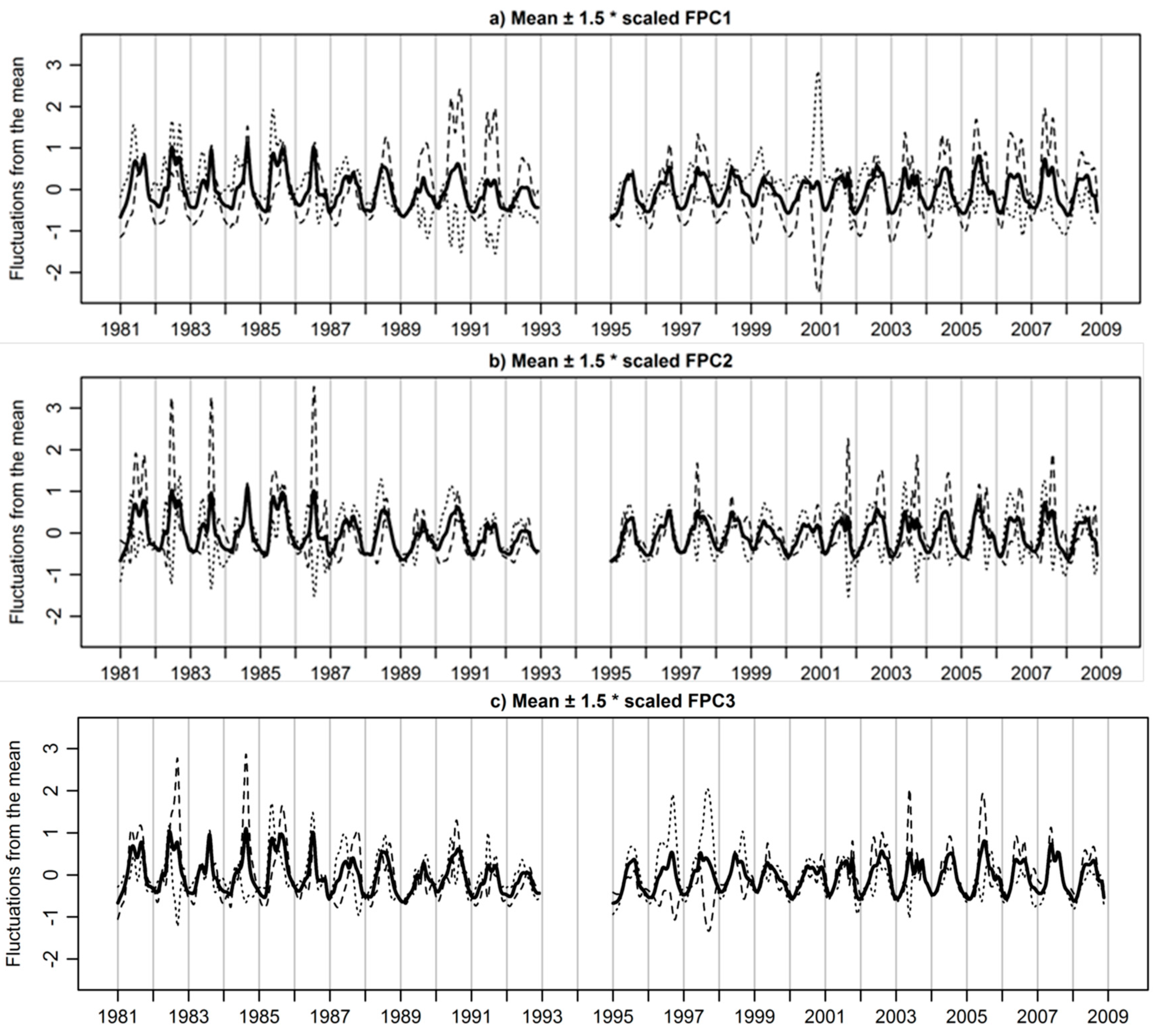
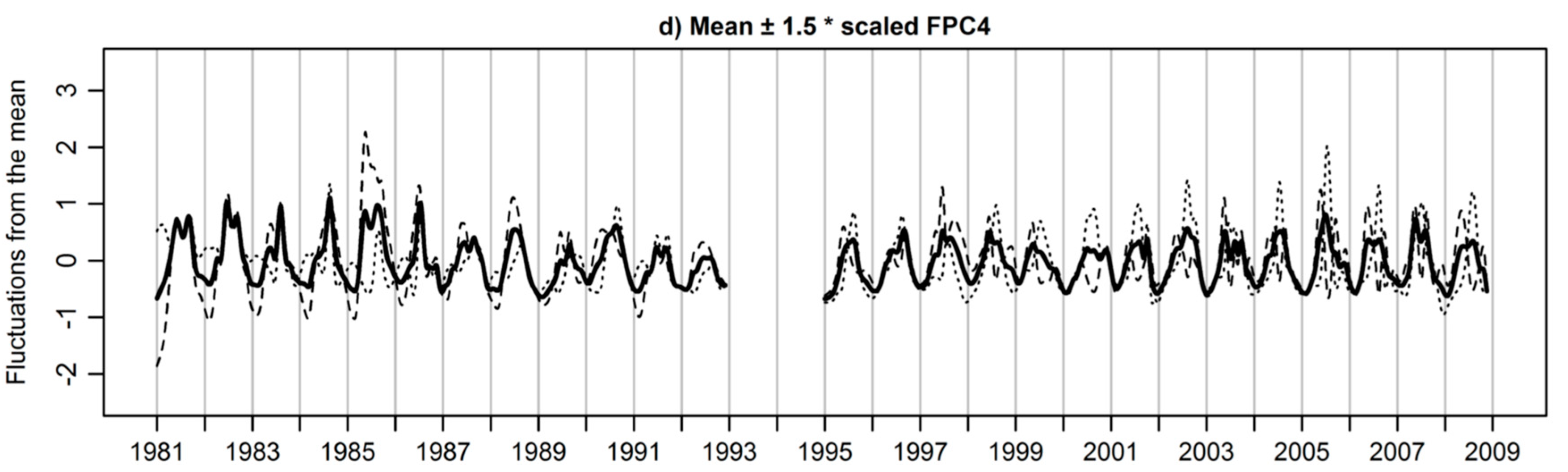
3.3. Extracted Functional Principal Components
3.4. Functional Principal Components Scores
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Technical Details
References
- Binelli, A.; Provini, A. DDT is still a problem in developed countries: The heavy pollution of Lake Maggiore. Chemosphere 2003, 52, 717–723. [Google Scholar] [CrossRef]
- Vinebrooke, R.D.; Cottingham, K.L.; Norberg, J.; Scheffer, M.; Dodson, S.I.; Maberly, S.C.; Sommer, U. Impacts of multiple stressors on biodiversity and ecosystem functioning: The role of species co-tolerance. Oikos 2004, 104, 451–457. [Google Scholar] [CrossRef]
- Yan, N.D.; Girard, R.; Heneberry, J.H.; Keller, W.B.; Gunn, J.M.; Dillon, P.J. Recovery of copepod, but not cladoceran, zooplankton from severe and chronic effects of multiple stressors. Ecol. Lett. 2004, 7, 452–460. [Google Scholar] [CrossRef]
- Schindler, D.W. Recent advances in the understanding and management of eutrophication. Limnol. Oceanogr. 2006, 51, 356–363. [Google Scholar] [CrossRef]
- Jeziorski, A.; Yan, N.D.; Paterson, A.M.; DeSellas, A.M.; Turner, M.A.; Jeffries, D.S.; Keller, B.; Weeber, R.C.; McNicol, D.K.; Palmer, M.E.; et al. The widespread threat of calcium decline in fresh waters. Science 2008, 322, 1374–1377. [Google Scholar] [CrossRef] [PubMed]
- Ormerod, S.J.; Dobson, M.; Hildrew, A.G.; Townsend, C.R. Multiple stressors in freshwater ecosystems. Freshw. Biol. 2010, 55 (Suppl. 1), 1–4. [Google Scholar] [CrossRef]
- Strayer, D.L. Alien species in fresh waters: Ecological effects, interactions with other stressors, and prospects for the future. Freshw. Biol. 2010, 55 (Suppl. 1), 152–174. [Google Scholar] [CrossRef]
- Maberly, S.C.; Elliott, J.A. Insights from long-term studies in the Windermere catchment: External stressors, internal interactions and the structure and function of lake ecosystems. Freshw. Biol. 2012, 57, 233–243. [Google Scholar] [CrossRef][Green Version]
- Guilizzoni, P.; Levine, S.N.; Manca, M.; Marchetto, A.; Lami, A.; Ambrosetti, W.; Brauer, A.; Gerli, S.; Carrara, E.A.; Rolla, A.; et al. Ecological effects of multiple stressors on a deep lake (Lago Maggiore, Italy) integrating neo and paleolimnological approaches. J. Limnol. 2012, 71, 1–22. [Google Scholar] [CrossRef]
- Allan, A.J.D.; McIntyre, P.B.; Smitha, S.D.P.; Halpern, B.S.; Boyer, G.L.; Buchsbaum, A.; Burton, G.A., Jr.; Campbell, L.M.; Chadderton, W.L.; Ciborowski, J.J.H.; et al. Joint analysis of stressors and ecosystem services to enhance restoration effectiveness. Proc. Nat. Acad. Sci. USA 2013, 110, 372–377. [Google Scholar] [CrossRef]
- Verdonschot, P.F.M.; Spears, B.M.; Feld, C.K.; Brucet, S.; Keizer-Vlek, H.; Borja, A.; Elliott, M.; Kernan, M.; Johnson, R.F. A comparative review of recovery processes in rivers, lakes, estuarine and coastal waters. Hydrobiologia 2013, 704, 453–474. [Google Scholar] [CrossRef]
- Smol, J.P.; Wolfe, A.P.; Birks, H.J.B.; Douglas, M.S.V.; Jones, V.J.; Korhola, A.; Pienitz, R.; Rühland, K.; Sorvari, S.; Antoniades, D.; et al. Climate-driven regime shifts in the biological communities of arctic lakes. Proc. Nat. Acad. Sci. USA 2005, 102, 4397–4402. [Google Scholar] [CrossRef]
- Moss, B. Climate change, nutrient pollution and the bargain of Dr Faustus. Freshw. Biol. 2010, 55 (Suppl. 1), 175–187. [Google Scholar] [CrossRef]
- Thompson, P.L.; Shurin, J.B. Regional zooplankton biodiversity provides limited buffering of pond ecosystems against climate change. J. Anim. Ecol. 2012, 81, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.C.; Loewen, C.J.G.; Vinebrooke, R.D.; Chimimba, C.T. Net effects of multiple stressors in freshwater ecosystems: A meta-analysis. Glob. Chang. Biol. 2016, 22, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Hering, D.; Borja, A.; Carvalho, L.; Feld, C.K. Assessment and recovery of European water bodies: Key messages from the WISER project. Hydrobiologia 2013, 704, 1–9. [Google Scholar] [CrossRef]
- Jeppesen, E.; Sondergaard, M.; Jensen, J.P.; Havens, K.E.; Anneville, O.; Carvalho, L.; Coveney, M.F.; Deneke, R.; Dokulil, M.; Foy, B.; et al. Lake responses to reduced nutrient loading—An analysis of contemporary long-term data from 35 case studies. Freshw. Biol. 2005, 50, 1747–1771. [Google Scholar] [CrossRef]
- Schindler, D.W. The dilemma of controlling eutrophication of lakes. Proc. R. Soc. B 2012, 279, 4322–4333. [Google Scholar] [CrossRef]
- Yan, N.D.; Pawson, T.W. Changes in the crustacean zooplankton community of Harp Lake, Canada, following invasion by Bythotrephes cederstrœmi. Freshw. Biol. 1997, 37, 409–425. [Google Scholar] [CrossRef]
- Straile, D.; Geller, W. The response of Daphnia to changes in trophic status and weather patterns: A case study from Lake Constance. ICES J. Mar. Sci. 1998, 55, 775–782. [Google Scholar] [CrossRef]
- May, L.; Spears, B.M. A history of scientific research at Loch Leven, Kinross, Scotland. Hydrobiologia 2012, 681, 3–9. [Google Scholar] [CrossRef]
- Lottig, N.R.; Carpenter, S.R. Interpolating and forecasting lake characteristics using long-term monitoring data. Limnol. Oceanogr. 2012, 57, 1113–1125. [Google Scholar] [CrossRef]
- Ramsay, J.; Silverman, B.W. Functional Data Analysis, 2nd ed.; Springer: New York, NY, USA, 2005. [Google Scholar]
- de Bernardi, R.; Giussani, G.; Manca, M.; Ruggiu, D. Long-term dynamics of plankton communities in Lago Maggiore (N. Italy). Internationale Vereinigung für Theoretische und Angewandte Limnologie: Verhandlungen 1988, 23, 729–733. [Google Scholar] [CrossRef]
- de Bernardi, R.; Giussani, G.; Manca, M.; Ruggiu, D. Trophic status and the pelagic system in Lake Maggiore. Hydrobiologia 1990, 191, 1–8. [Google Scholar] [CrossRef]
- Manca, M.; Ruggiu, D. Consequences of pelagic food-web changes during a long-term lake oligotrophication process. Limnol. Oceanogr. 1998, 43, 1368–1373. [Google Scholar] [CrossRef]
- Sas, H. Lake Restoration by Reduction of Nutrient Loading: Expectations, Experiences, Extrapolation; Academic Verlag: St. Augustin, FL, USA, 1989. [Google Scholar]
- de Bernardi, R.; Canale, C. Ricerche pluriennali (1948–1992) sull’ecologia dello zooplancton del Lago Maggiore. Doc Ist. Ital. Hydrobiol. 1995, 55, 1–68. [Google Scholar]
- Manca, M.; Calderoni, A.; Mosello, R. Limnological research in Lago Maggiore: Studies on hydrochemistry and plankton. Memorie Istituto Italiano di Idrobiologia 1992, 50, 171–200. [Google Scholar]
- Tonolli, L. Holomixy and oligomixy in Lake Maggiore: Inference on the vertical distribution of zooplankton. Verh. Int. Ver. Limnol. 1969, 17, 231–236. [Google Scholar] [CrossRef]
- Manca, M.; DeMott, W.R. Response of the invertebrate predator Bythotrephes to a climate-linked increase in the duration of a refuge from fish predation. Limnol. Oceanogr. 2009, 54 Pt 2, 2506–2512. [Google Scholar] [CrossRef]
- Arca, G.; Barbanti, L. Annuario dell’Osservatorio Meteorologico di Pallanza; Documenta Ist. Ital. Idrobiol; Consiglio Nazionale delle Ricerche, Istituto Italiano di Idrobiologia: Pallanza, Italy, 1991; pp. 1–83. (In Italian) [Google Scholar]
- Barbanti, L. Annuario dell’Osservatorio Meteorologico di Pallanza; Documenta Ist. Ital. Idrobiol; Consiglio Nazionale delle Ricerche, Istituto Italiano di Idrobiologia: Pallanza, Italy, 1997; pp. 1–81. (In Italian) [Google Scholar]
- Ambrosetti, W.; Barbanti, L.; Rolla, A. Annuario dell’Osservatorio Meteorologico di Pallanza. 1997–2003; CNR Istituto per lo Studio degli Ecosistemi: Verbania Pallanza, Italy, 2006; pp. 1–38. [Google Scholar]
- Ambrosetti, W.; Barbanti, L.; Rolla, A. Annuario dell’Osservatorio Meteorologico di Pallanza. 2004–2005; CNR Istituto per lo Studio degli Ecosistemi: Verbania Pallanza, Italy, 2006; pp. 1–70. [Google Scholar]
- Ambrosetti, W.; Barbanti, L.; Rolla, A. Annuario dell’Osservatorio Meteorologico di Pallanza. 2006; CNR Istituto per lo Studio degli Ecosistemi: Verbania Pallanza, Italy, 2007; pp. 1–51. [Google Scholar]
- Ambrosetti, W.; Barbanti, L.; Rolla, A. Annuario dell’Osservatorio Meteorologico di Pallanza. 2007; CNR Istituto per lo Studio degli Ecosistemi: Verbania Pallanza, Italy, 2008; pp. 1–56. [Google Scholar]
- Lorenzen, C.J. Determination of chlorophyll and pheo-pigments: Spectrophotometric equations. Limnol. Oceanogr. 1967, 12, 343–346. [Google Scholar] [CrossRef]
- Rott, E. Some results from phytoplankton counting intercalibrations. Schweiz. Z. Hydrol. 1981, 43, 34–63. [Google Scholar] [CrossRef]
- Lund, J.W.G.; Kipling, C.; Le Cren, E.D. The inverted microscope method of estimating algal numbers and the statistical basis of estimations by counting. Hydrobiologia 1958, 11, 143–170. [Google Scholar] [CrossRef]
- Wacklin, P.; Hoffmann, L.; Komárek, J. Nomenclatural validation of the genetically revised cyanobacterial genus. Fottea 2009, 9, 59–64. [Google Scholar] [CrossRef]
- Morabito, G.; Manca, M. Eutrophication and recovery of the large and deep subalpine Lake Maggiore: Patterns, trends and interactions of planktonic organisms between trophic and climatic forcings. In Chapter 6 in Eutrophication; Lambert, A., Roux, C., Eds.; Nova Science Publishers: New York, NY, USA, 2013; ISBN 978-1-62808-498-6. [Google Scholar]
- Carrara, E.; Ambrosetti, W.; Barbanti, L. The management of Lake Maggiore water levels: A study of low water episodes. In Proceedings of the Symposium on the Role of Hydrology in Water Resources Management, Capri, Italy, 29–31 October 2008; Volume 327, pp. 114–123. [Google Scholar]
- Ambrosetti, W.; Barbanti, L.; Carrara, E. Mechanisms of hypolimnion erosion in a deep lake (Lago Maggiore, N. Italy). J. Limnol. 2010, 69, 3–14. [Google Scholar] [CrossRef]
- Ruggiu, D.; Morabito, G.; Panzani, P.; Pugnetti, A. Trends and relations among basic phytoplankton characteristics in the course of the long-term oligotrophication of Lago Maggiore (Italy). Hydrobiologia 1998, 369–370, 243–257. [Google Scholar] [CrossRef]
- Obertegger, U.; Manca, M. Response of rotifer functional groups to changing trophic state and crustacean community. J. Limnol. 2011, 70, 231–238. [Google Scholar] [CrossRef]
- Manca, M.; Ramoni, C.; Comoli, P. The decline of Daphnia hyalina galeata in Lago Maggiore: A comparison of the population dynamics before and after oligotrophication. Aquat. Sci. 2000, 62, 142–153. [Google Scholar] [CrossRef]
- Manca, M.; Portogallo, M.; Brown, M. Shifts in phenology of Bythotrephes longimanus and its modern success in Lake Maggiore as a result of changes in climate and trophy. J. Plankton Res. 2007, 29, 515–525. [Google Scholar] [CrossRef]
- MacIsaac, H.J.; Ketelaars, H.A.M.; Girgorovich, I.A.; Ramcharan, C.W.; Yan, N.D. Modeling Bythotrephes longimanus invasions in the Great Lakes basin based on its European distribution. Arch. Hydrobiol. 2000, 141, 1–21. [Google Scholar] [CrossRef]
- Boudreau, S.A.; Yan, N.D. The differing crustacean zooplankton communities of Canadian Shield lakes with and without the nonindigenous zooplanktivore Bythotrephes longimanus. Can. J. Fish. Aquat. Sci. 2003, 60, 1307–1313. [Google Scholar] [CrossRef]
- Weisz, E.J.; Yan, N.D. Shifting invertebrate zooplanktivores: Watershed-level replacement of the native Leptodora by the non-indigenous Bythotrephes in Canadian Shield lakes. Biol. Invasions 2011, 13, 115–123. [Google Scholar] [CrossRef]
- Manca, M. Considerazioni generali sull’evoluzione a lungo termine dei popolamenti planctonici. In Ecologia delle Acque Interne. Ricerche sull’evoluzione del Lago Maggiore. Aspetti limnologici. Programma quinquennale; Campagna 2002; Commissione Internazionale per la protezione delle acque italo-svizzere, sezione di Idrobiologia, C.N.R.-I.S.E.: Verbania, Italy, 2014; pp. 134–136. (In Italian) [Google Scholar]
- de Bernardi, R.; Giussani, G.; Manca, M. Cladocera: Predators and Prey; Springer: Dordrecht, The Netherlands, 1987; pp. 225–243. [Google Scholar]
- Visconti, A.; Volta, P.; Fadda, A.; Di Guardo, A.; Manca, M. Seasonality, littoral versus pelagic carbon sources, and stepwise δ15N-enrichment of pelagic food web in a deep subalpine lake: The role of planktivorous fish. Can. J. Fish. Aquat. Sci. 2013, 71, 436–446. [Google Scholar] [CrossRef]
- Coulas, R.A.; MacIsaac, H.J.; Dunlop, W. Selective predation by lake herring (Coregonus artedi) on exotic zooplankton (Bythotrephes cederstroemi) in Harp Lake, Ontario. Freshw. Biol. 1998, 40, 343–356. [Google Scholar] [CrossRef]
- Visconti, A.; Manca, M.; De Bernardi, R. Eutrophication-like response to climate warming: an analysis of Lago Maggiore (N. Italy) zooplankton in contrasting years. J. Limnol. 2008, 67, 87–92. [Google Scholar] [CrossRef]
- Gillooly, J.F.; Charnov, E.L.; West, G.B.; Savage, V.M.; Brown, J. Effects of size and temperature on developmental time. Nature 2003, 417, 70–73. [Google Scholar] [CrossRef]
- Manca, M.; Sonvico, D. Seasonal variations in population density and size structure of Conochilus in Lago Maggiore: A biannual study. Mem. Ist. Ital. Idrobiol. 1996, 54, 97–108. [Google Scholar]
- Obertegger, U.; Smith, H.A.; Flaim, G.; Wallace, R.L. Using the guild ratio to characterize pelagic rotifer communities. Hydrobiologia 2011, 662, 157–162. [Google Scholar] [CrossRef]
- Pourriot, R. Food and feeding habits of Rotifera. Arch. Hydrobiol. Beih. Ergebn. Limnol. 1977, 8, 243–260. [Google Scholar]
- Wallace, R.L.; Snell, T.W.; Ricci C Nogrady, T. Rotifera Volume 1: Biology, Ecology and Systematics. In Guides to the Identification of the Microinvertebrates of the Continental Waters of the World, 2nd ed.; Dumont, H.J.F., Ed.; Kenobi Productions: Ghent, Belgium; Backhuys Publishers: Leiden, The Netherlands, 2006; Volume 23. [Google Scholar]
- temberger, R.S.; Gilbert, J.J. Rotifer threshold food concentrations and the size-efficiency hypothesis. Ecology 1987, 68, 181–187. [Google Scholar] [CrossRef]
- Manca, M. Invasions and re-emergences: An analysis of the success of Bythotrephes in Lago Maggiore (Italy). J. Limnol. 2011, 70, 76–82. [Google Scholar] [CrossRef]
- Hovius, J.T.; Beisner, B.E.; McCann, K.S.; Yan, N.D. Indirect food web effects of Bythotrephes invasion: Responses by the rotifer Conochilus in Harp Lake, Canada. Biol. Invasions 2007, 9, 233–243. [Google Scholar] [CrossRef]
- Manca, M.; Torretta, B.; Comoli, P.; Amsinck, S.L.; Jeppesen, E. Major changes in trophic dynamics in large, deep sub-alpine Lake Maggiore from 1940s to 2002: A high resolution comparative paleo-neolimnological study. Freshw. Biol. 2007, 52, 2256–2269. [Google Scholar] [CrossRef]
- Manca, M.; Cavicchioni, N.; Morabito, G. First observations on the effect of a complete, exceptional overturn of Lake Maggiore on plankton and primary productivity. Int. Rev. Hydrobiol. 2000, 85, 209–222. [Google Scholar] [CrossRef]
- Salmaso, N.; Buzzi, F.; Garibaldi, L.; Morabito, G.; Simona, M. Effects of nutrient availability and temperature on phytoplankton development: A case study from large lakes south of the Alps. Aquat. Sci. 2012, 74, 555–570. [Google Scholar] [CrossRef]
- Manca, M.; Rogora, M.; Salmaso, N. Inter-annual climate variability and zooplankton: Applying teleconnection indices to two deep subalpine lakes in Italy. J. Limnol. 2014, 74. [Google Scholar] [CrossRef]
- Visconti, A.; Caroni, R.; Rawcliffe, R.; Fadda, A.; Piscia, R.; Manca, M. Defining Seasonal Functional Traits of a Freshwater Zooplankton Community Using δ13C and δ15N Stable Isotope Analysis. Water 2018, 10, 108. [Google Scholar] [CrossRef]
- Visconti, A.; Manca, M. Seasonal changes in the δ13C and δ15N signatures of the Lago Maggiore pelagic food web. J. Limnol. 2011, 70, 263–271. [Google Scholar] [CrossRef]
| Functional Principal Components (FPCs) Scores | ||||||||
|---|---|---|---|---|---|---|---|---|
| Calendar period | 1981–1992 | 1995–2008 | ||||||
| Variables | FPC1 | FPC2 | FPC3 | FPC4 | FPC1 | FPC2 | FPC3 | FPC4 |
| Physico-Chemical | ||||||||
| Water temperature | 30.5 | 11.9 | 16.7 | 13.1 | 30.2 | 10.9 | −12.6 | −25.7 |
| Phosphorus concentration | −37.8 | −16.6 | −25.9 | −46.9 | −45.2 | −15.2 | −14.2 | −2.0 |
| Phytoplankton-related | ||||||||
| Phytoplankton pop. dens. | 13.7 | −10.6 | 12.6 | −3.8 | 8.6 | 1.1 | −39.6 | 5.5 |
| Chlorophyll-a concentration | −11.2 | −9.7 | 3.8 | 8.6 | 12.6 | −29.1 | −16.5 | 1.9 |
| Phytoplankton cell size | −20.8 | −18.7 | −21.0 | 3.9 | −17.0 | −19.5 | 12.1 | 10.4 |
| Zooplankton-related | ||||||||
| Bosmina pop. dens. | −13.0 | 37.5 | −17.7 | 26.0 | 20.6 | 46.1 | 1.4 | 20.0 |
| Bythotrephes pop. dens. | 48.2 | −17.2 | −14.7 | −17.1 | 19.8 | 1.0 | −1.8 | −16.6 |
| Cyclopoid pop. dens. | −16.0 | −10.6 | 41.8 | 0.1 | −38.5 | 1.4 | 7.6 | 15.9 |
| Daphnia pop. dens. | −14.0 | 34.6 | −4.6 | 7.9 | 9.2 | −15.0 | 33.8 | 17.9 |
| Diaphanosoma pop. dens. | 12.4 | −2.1 | 24.8 | −9.7 | 4.3 | 34.0 | 10.2 | −32.9 |
| Diaptomid pop. dens. | −1.3 | −9.1 | 2.9 | 13.8 | −28.1 | 5.4 | −3.0 | 24.7 |
| Leptodora pop. dens. | 13.3 | 31.8 | 4.6 | −12.2 | 11.4 | 0.7 | 4.7 | −33.5 |
| Rotifer pop. dens. | −4.1 | −21.1 | −23.2 | 16.3 | 12.0 | −21.8 | 18.0 | 14.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arfè, A.; Quatto, P.; Zambon, A.; MacIsaac, H.J.; Manca, M. Long-Term Changes in the Zooplankton Community of Lake Maggiore in Response to Multiple Stressors: A Functional Principal Components Analysis. Water 2019, 11, 962. https://doi.org/10.3390/w11050962
Arfè A, Quatto P, Zambon A, MacIsaac HJ, Manca M. Long-Term Changes in the Zooplankton Community of Lake Maggiore in Response to Multiple Stressors: A Functional Principal Components Analysis. Water. 2019; 11(5):962. https://doi.org/10.3390/w11050962
Chicago/Turabian StyleArfè, Andrea, Piero Quatto, Antonella Zambon, Hugh J. MacIsaac, and Marina Manca. 2019. "Long-Term Changes in the Zooplankton Community of Lake Maggiore in Response to Multiple Stressors: A Functional Principal Components Analysis" Water 11, no. 5: 962. https://doi.org/10.3390/w11050962
APA StyleArfè, A., Quatto, P., Zambon, A., MacIsaac, H. J., & Manca, M. (2019). Long-Term Changes in the Zooplankton Community of Lake Maggiore in Response to Multiple Stressors: A Functional Principal Components Analysis. Water, 11(5), 962. https://doi.org/10.3390/w11050962







