Distributive Characteristics of Riverine Nutrients in the Mun River, Northeast Thailand: Implications for Anthropogenic Inputs
Abstract
1. Introduction
2. Study Area and Method
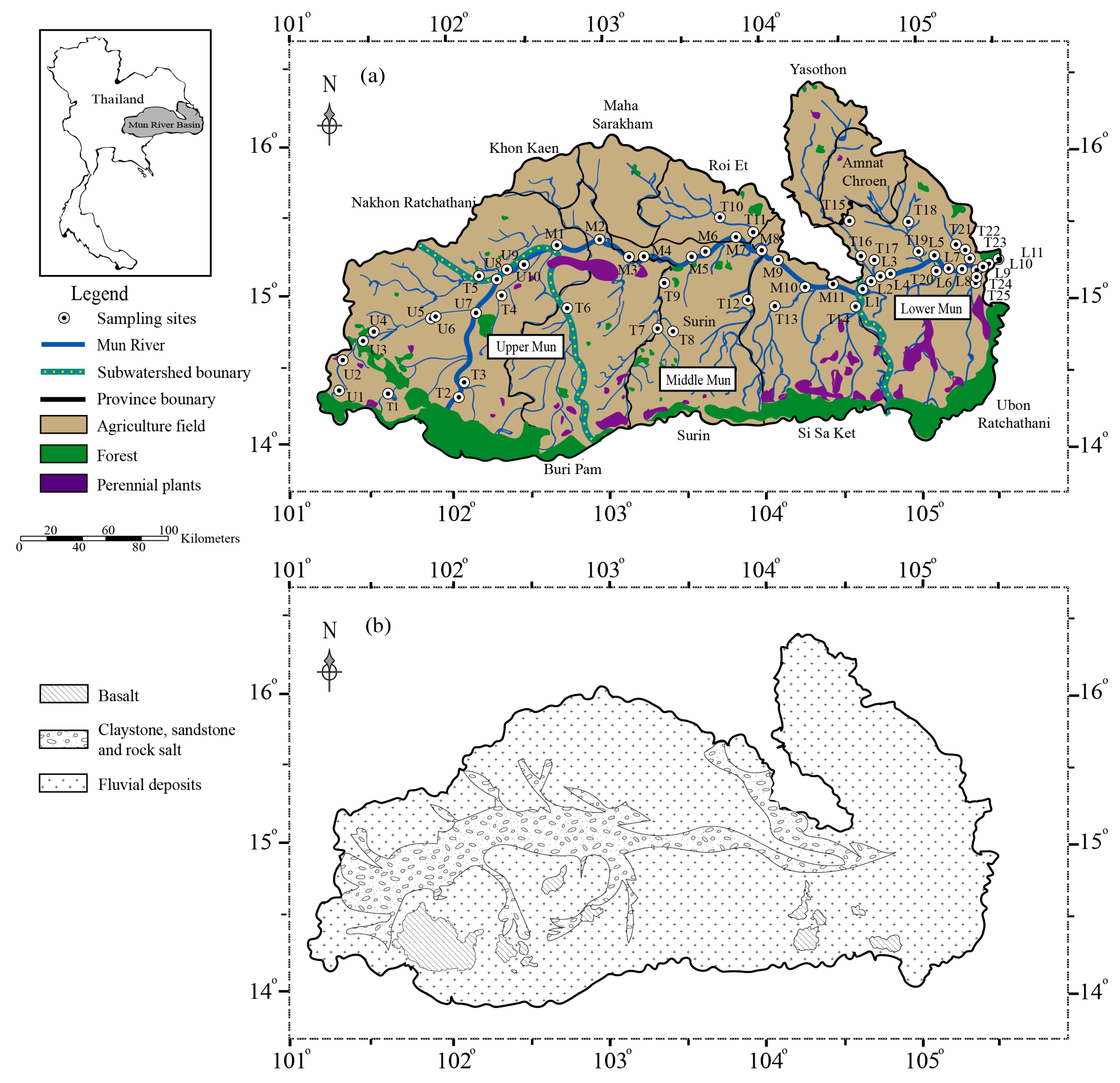
2.1. Study Area Location
2.2. Sampling Procedure and Analytical Methods
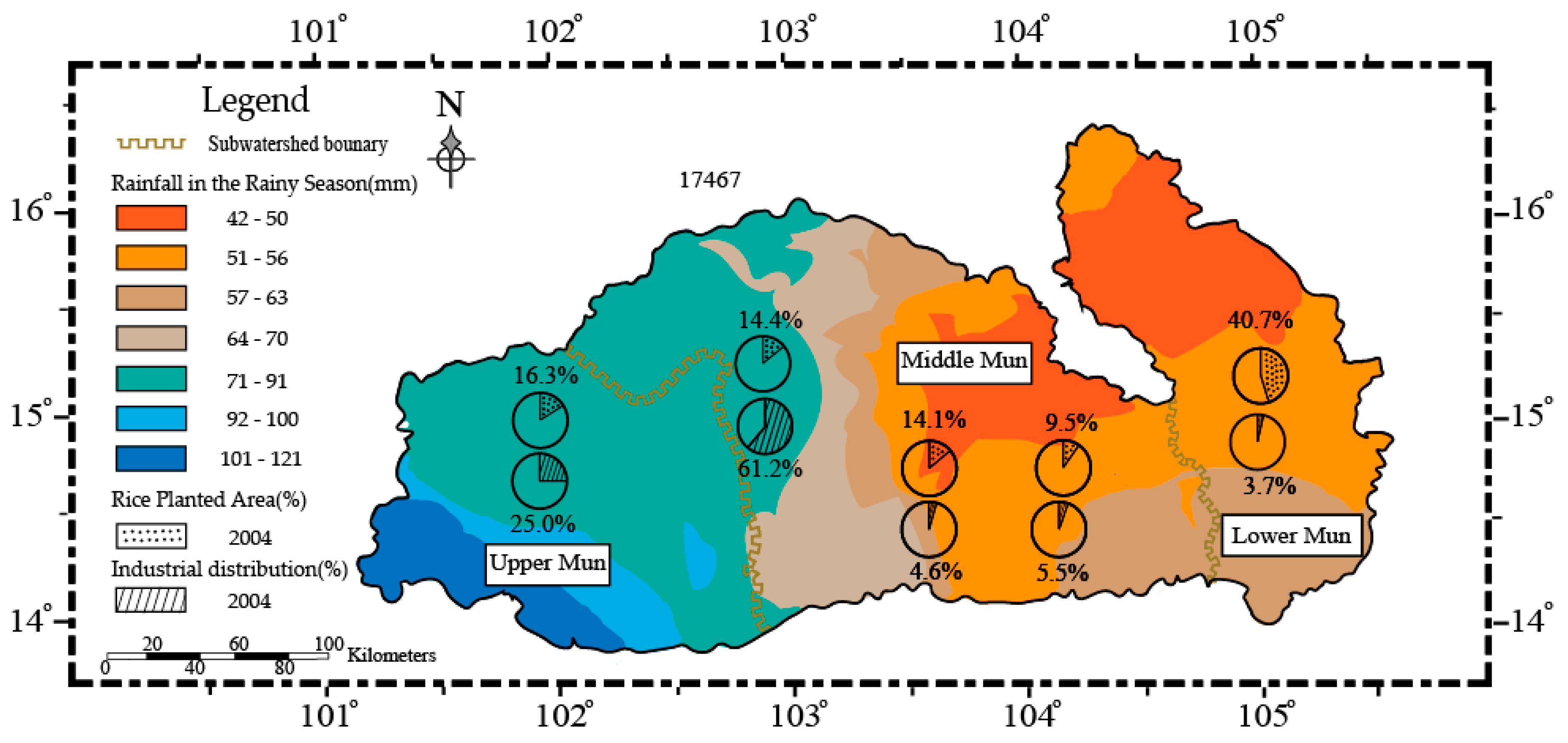
2.3. Land-Use Type and Precipitation Distributions
3. Results and Discussion
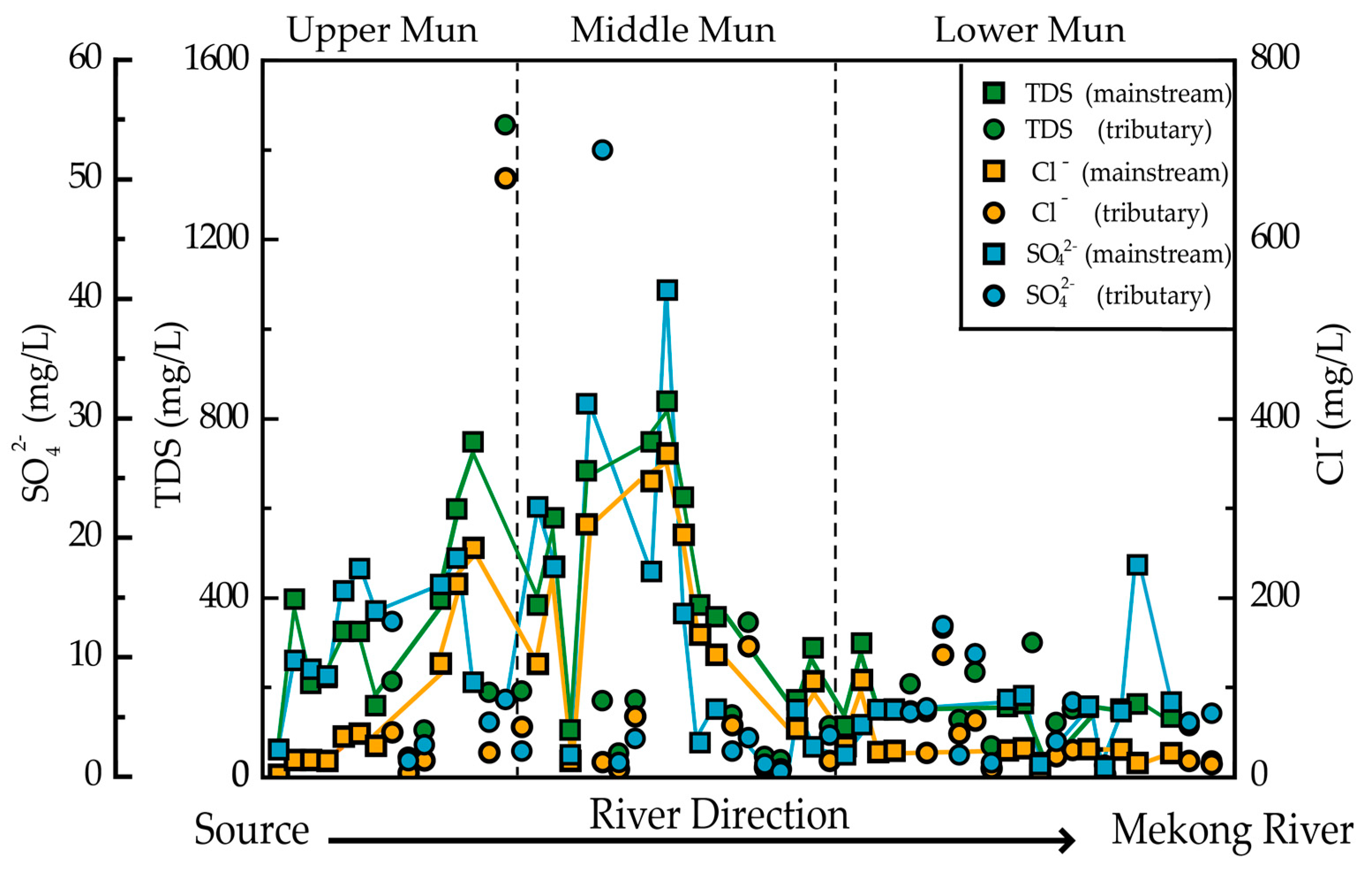
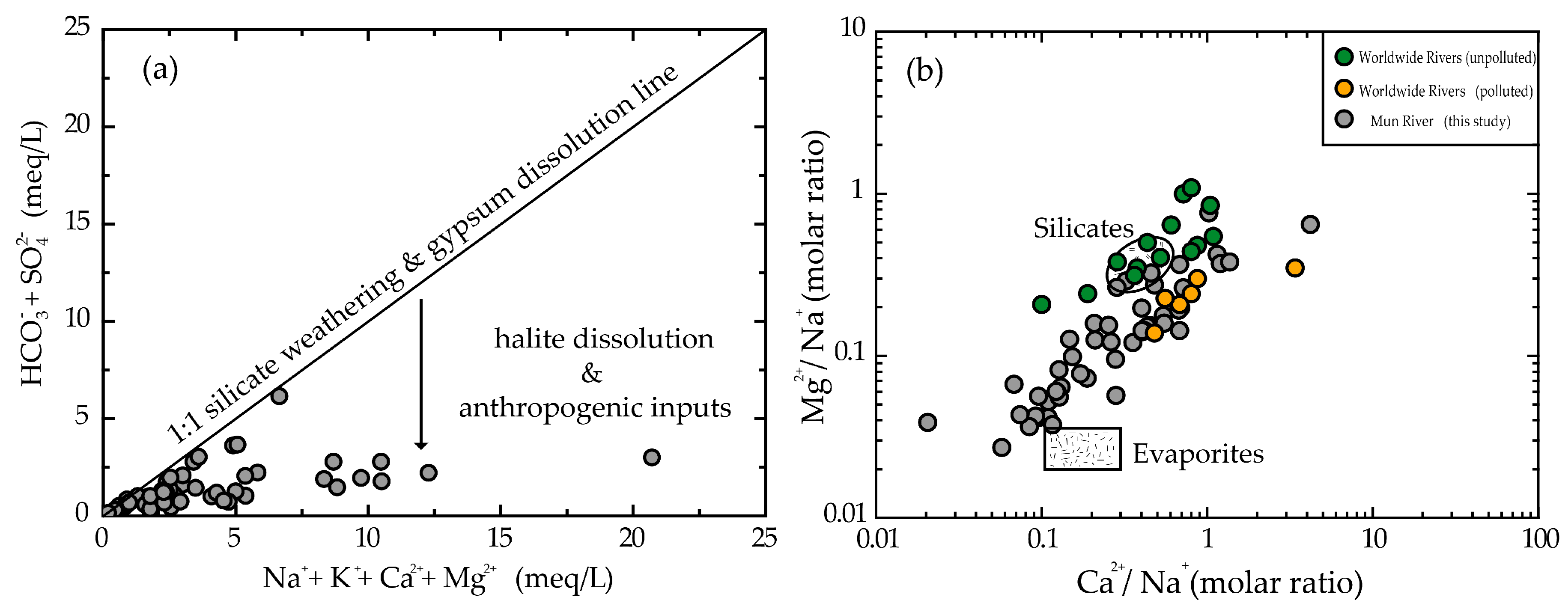
3.1. Sources of Major Anions
3.2. Weathering Processes
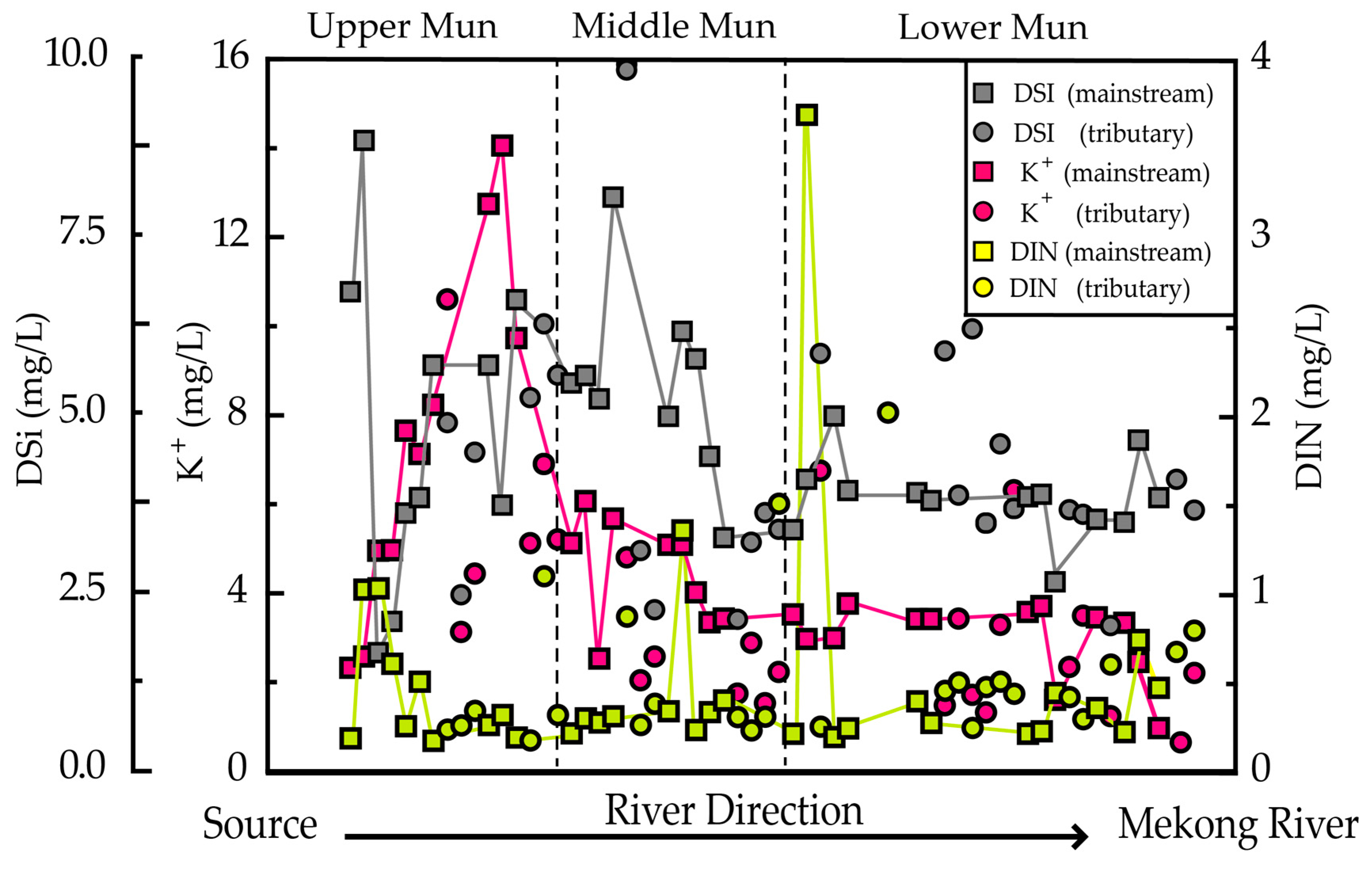
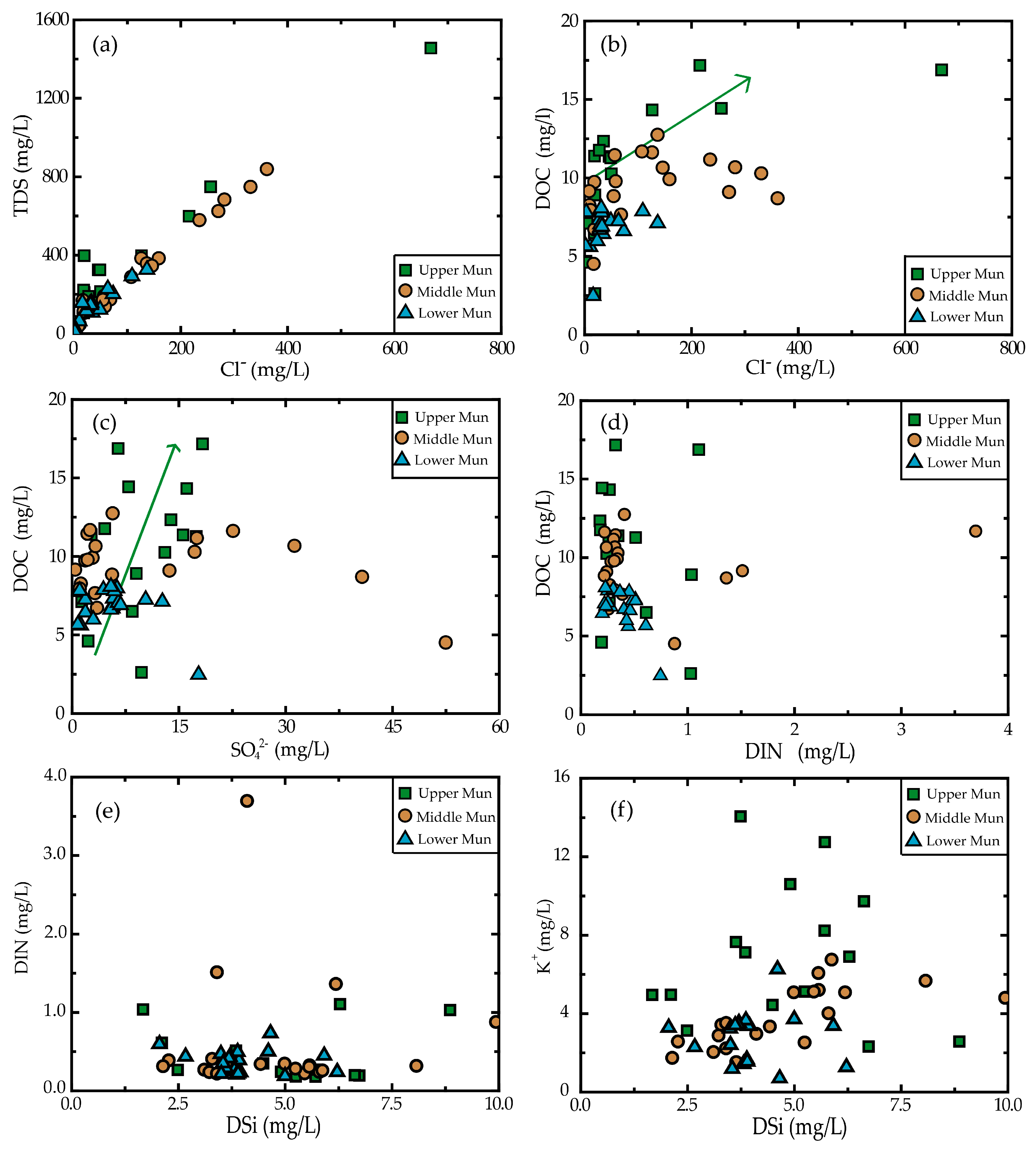
3.3. Relationship between Dissolved Loads
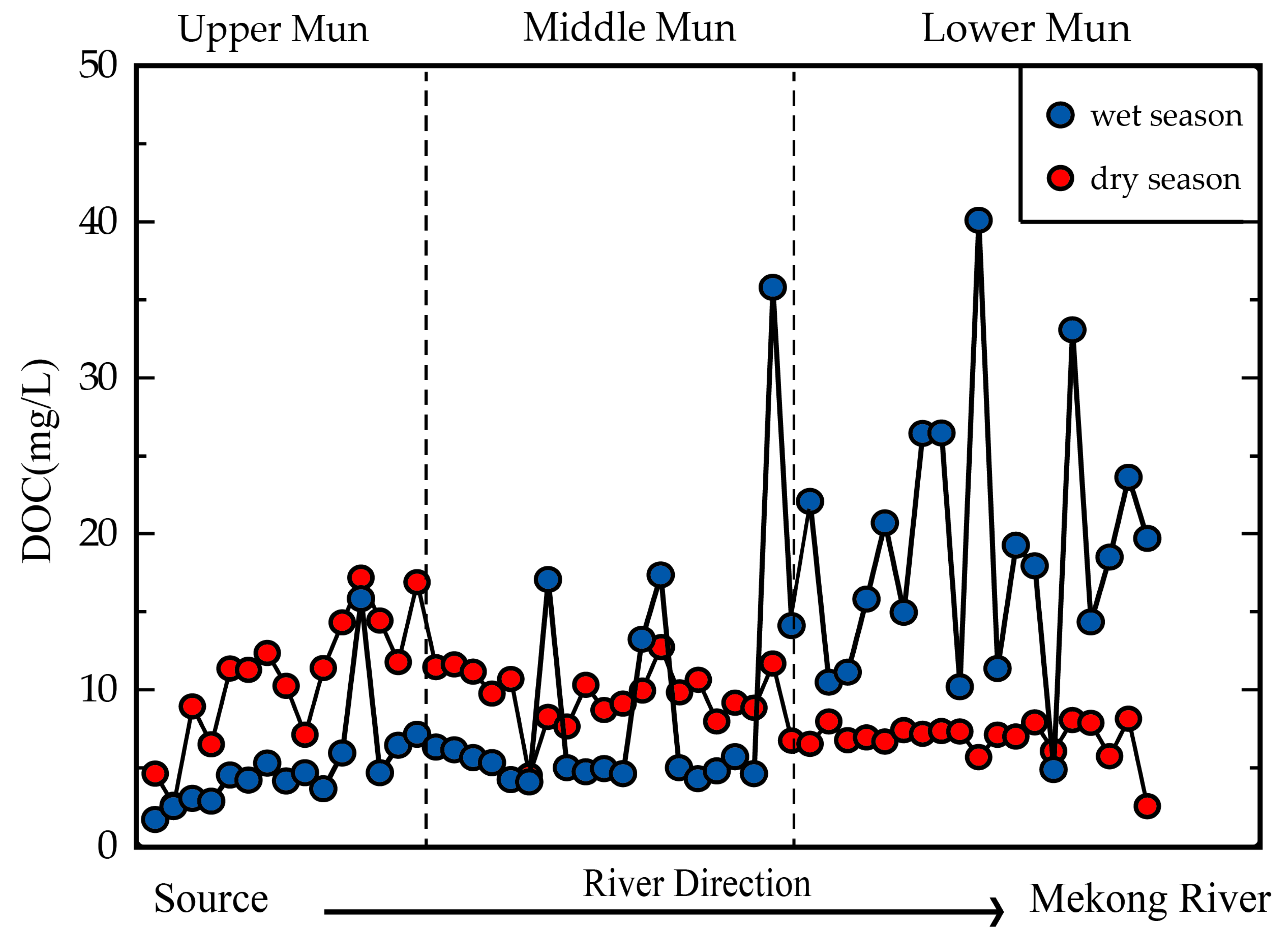
3.4. The Impact of Precipitation and Soil Erosion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Meybeck, M. Global geochemical weathering of surficial rocks estimates from river dissolved loads. Am. J. Sci. 1987, 287, 401–428. [Google Scholar] [CrossRef]
- Suchet, P.A.; Probst, J.L.; Ludwig, W. Worldwide distribution of continental rock lithology: Implications for the atmospheric/soil CO2 uptake for continental weathering and alkalinity river transport to the oceans. Glob. Biogeochem. Cycles 2003, 17. [Google Scholar] [CrossRef]
- Sharples, J.; Middelburg, J.J.; Fennel, K.; Jickells, T.D. What proportion of riverine nutrients reaches the open ocean? Glob. Biogeochem. Cycles 2017, 31, 39–58. [Google Scholar] [CrossRef]
- Teng, F.Z. Magnesium Isotope Geochemistry. Rev. Mineral. Geochem. 2017, 82, 219–287. [Google Scholar] [CrossRef]
- Leach, J.A.; Larsson, A.; Wallin, M.B.; Nilsson, M.B.; Laudon, H. Twelve year interannual and seasonal variability of stream carbon export from a boreal peatland catchment. J. Geophys. Res. Biogeosci. 2016, 121, 1851–1866. [Google Scholar] [CrossRef]
- Chetelat, B.; Liu, C.Q.; Zhao, Z.Q.; Wang, Q.L.; Li, S.L.; Li, J.; Wang, B.L. Geochemistry of the dissolved load of the Changjiang Basin rivers: Anthropogenic impacts and chemical weathering. Geochim. Cosmochim. Acta 2008, 72, 4254–4277. [Google Scholar] [CrossRef]
- Blum, J.D.; Gazis, C.A.; Jacobson, A.D.; Chamberlain, C.P. Carbonate versus silicate weathering in the Raikhot watershed within the High Himalayan Crystalline Series. Geology 1998, 26, 411–414. [Google Scholar] [CrossRef]
- West, A.J.; Galy, A.; Bickle, M. Tectonic and climatic controls on silicate weathering. Earth Planet. Sci. Lett. 2005, 235, 211–228. [Google Scholar] [CrossRef]
- White, A.F.; Blum, A.E. Effects of climate on chemical weathering in watersheds. Geochim. Cosmochim. Act 1995, 59, 1729–1747. [Google Scholar] [CrossRef]
- Riebe, C.S.; Kirchner, J.W.; Finkel, R. Erosional and climatic effects on long-term chemical weathering rates in granitic land-scapes spanning diverse climate regimes. Earth Planet. Sci. Lett. 2004, 224, 547–562. [Google Scholar] [CrossRef]
- Gaillardet, J.; Dupré, B.; Louvat, P.; Allegre, C.J. Global silicate weathering and CO2 consumption rates deduced from the chemistry of large rivers. Chem. Geol. 1999, 159, 3–30. [Google Scholar] [CrossRef]
- Tipper, E.T.; Galy, A.; Bickle, M.J. Riverine evidence for a fractionated reservoir of Ca and Mg on the continents: Implications for the oceanic Ca cycle. Earth Planet. Sci. Lett. 2006, 247, 267–279. [Google Scholar] [CrossRef]
- Wu, Y.; Bao, H.Y.; Unger, D.; Herbeck, L.S.; Zhu, Z.Y.; Zhang, J.; Jennerjahn, T.C. Biogeochemical behavior of organic carbon in a small tropical river and estuary, Hainan, China. Cont. Shelf Res. 2013, 57, 32–43. [Google Scholar] [CrossRef]
- Christophe, R.; Fred, T.M.; Leah, M.V. Influence of the human perturbation on carbon, nitrogen, and oxygen biogeochemical cycles in the global coastal ocean. Geochim. Cosmochim. Act 2001, 65, 3615–3641. [Google Scholar] [CrossRef]
- Zhang, S.; Lu, X.X.; Sun, H.; Han, J.; Laurence, D. Major ion chemistry and dissolved inorganic carbon cycling in a human-disturbed mountainous river (the Luodingjiang River) of the Zhujiang (Pearl River), China. Sci. Total Environ. 2009, 407, 2796–2807. [Google Scholar] [CrossRef] [PubMed]
- Kaushal, S.S.; Duan, S.; Doody, T.R.; Haq, S.; Smith, R.M.; Newcomer, T.A.; Delaney, K.; Gorman, J.; Bowman, N.; Mayer, P.M.; et al. Human-accelerated weathering increases salinization, major ions, and alkalinization in fresh water across land use. Appl. Geochem. 2017, 83, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Li, F.; Tan, Q. Effects of land use on water chemistry in a river draining karst terrain, southwest China. Hydrol. Sci. J. 2014, 59, 1063–1073. [Google Scholar] [CrossRef]
- Kronvang, B.; Hoffmann, C.C.; Svendsen, L.M.; Windolf, J.; Jensen, J.P.; Dørge, J. Retention of nutrients in river basins. Aquat. Ecol. 1999, 33, 29–40. [Google Scholar] [CrossRef]
- Lorite-Herrera, M.; Hiscock, K.; Jiménez-Espinosa, R. Distribution of dissolved inorganic and organic nitrogen in river water and groundwater in an agriculturally-dominated catchment, south-East Spain. Water. Air. Soil Pollut. 2009, 198, 335–346. [Google Scholar] [CrossRef]
- Evans, C.D.; Monteith, D.T.; Cooper, D.M. Long-term increases in surface water dissolved organic carbon: Observations, possible causes and environmental impacts. Environ. Pollut. 2005, 137, 55–71. [Google Scholar] [CrossRef]
- Gorham, E.; Underwood, J.K.; Janssens, J.A.; Freedman, B.; Maass, W.; Waller, D.H.; Ogden, J.G. The chemistry of streams in southwestern and central Nova Scotia, with particular reference to catchment vegetation and the influence of dissolved organic carbon primarily from wetlands. Wetlands 1998, 8, 115–132. [Google Scholar] [CrossRef]
- Shang, F.; Ren, S.; Yang, P.; Li, C.; Ma, N. Effects of different fertilizer and irrigation water types, and dissolved organic matter on soil C and N mineralization in crop rotation farmland. Water. Air. Soil Pollut. 2015, 226, 396. [Google Scholar] [CrossRef]
- Eshleman, K.N.; Hemond, H.F. The role of organic acids in the acid-base status of surface waters at Bickford Watershed, Massachusetts. Water Resour. Res. 1985, 21, 1503–1510. [Google Scholar] [CrossRef]
- Roy, R.L.; Campbell, P.G.C. Decreased toxicity of Al to juvenile atlantic salmon (Salmo salar) in acidic soft water containing natural organic matter: A test of the free-ion model. Environ. Toxicol. Chem. 1997, 16, 1962–1969. [Google Scholar] [CrossRef]
- Lawlor, A.J.; Tipping, E. Metals in bulk deposition and surface waters at two upland locations in northern England. Environ. Pollut. 2003, 121, 153–168. [Google Scholar] [CrossRef]
- McCartney, A.G.; Harriman, R.; Watt, A.W.; Moore, D.W.; Taylor, E.M.; Collen, P.; Keay, E.J. Long-term trends in pH, aluminium and dissolved organic carbon in Scottish fresh waters; implications for brown trout (Salmo trutta) survival. Sci. Total Environ. 2003, 310, 133–141. [Google Scholar] [CrossRef]
- Thomas, G.H.; William, B.; George, R.A.; Justin, S.; Li, F.L.; Collin, S.R.; Philip, C. Climate change and dissolved organic carbon export to the Gulf of Mainecarbon export to the Gulf of Maine. J. Geophys. Res.-Biogeosci. 2016, 2700–2716. [Google Scholar] [CrossRef]
- Barth, J.A.C.; Cronin, A.A.; Dunlop, J.; Kalin, R.M. Influence of carbonates on the riverine carbon cycle in an anthropogenically dominated catchment basin: Evidence from major elements and stable carbon isotopes in the Lagan River (N. Irel.). Chem. Geol. 2003, 200, 203–216. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, C. Chemical weathering in the upper reaches of Xijiang River draining the Yunnan–Guizhou Plateau, Southwest China. Chem. Geol. 2007, 239, 83–95. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, G.; Liu, Q.; Huang, C.; Li, H. Studies on the spatiotemporal variability of river water quality and its relationships with soil and precipitation: a case study of the Mun River Basin in Thailand. Int. J. Environ. Res. Public Health 2018, 15, 2466. [Google Scholar] [CrossRef]
- Liu, J.; Han, G.; Liu, X.; Liu, M.; Song, C.; Zhang, Q. Impacts of anthropogenic changes on the Mun River water: insight from spatio-distributions and relationship of C and N species in Northeast Thailand. Int. J. Environ. Res. Public Health 2019, 16, 659. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.; Han, G.; Man, L.; Xiaoqiang, L. Spatial and temporal variation of dissolved heavy metals in the Mun River, Northeast Thailand. Water 2019, 11, 380. [Google Scholar] [CrossRef]
- Prabnakorn, S.; Maskey, S.; Suryadi, F.X.; de Fraiture, C. Rice yield in response to climate trends and drought index in the Mun River Basin, Thailand. Sci. Total Environ. 2018, 621, 108–119. [Google Scholar] [CrossRef]
- Akter, A.; Babel, M.S. Hydrological modeling of the Mun River basin in Thailand. J. Hydrol. 2012, 452–453. [Google Scholar] [CrossRef]
- Ahearn, D.S.; Sheibley, R.W.; Dahlgren, R.A.; Anderson, M.L.; Johnson, J.; Tate, K.W. Land use and land cover influence on water quality in the last free-flowing river draining the western Sierra Nevada, California. J. Hydrol. 2005, 313, 234–247. [Google Scholar] [CrossRef]
- Jiang, Y. The contribution of human activities to dissolved inorganic carbon fluxes in a karst underground river system: Evidence from major elements and δ 13C-DIC in Nandong, Southwest China. J. Contam. Hydrol. 2013, 152, 1–11. [Google Scholar] [CrossRef]
- Moon, S.; Huh, Y. Chemical weathering in the Hong (Red) River basin: Rates of silicate weathering and their controlling factors. Geochim. Cosmochim. Acta 2007, 71, 1411–1430. [Google Scholar] [CrossRef]
- Han, G.; Tang, Y.; Xu, Z. Fluvial geochemistry of rivers draining karst terrain in southwest China. J. Asian Earth Sci. 2010, 38, 65–75. [Google Scholar] [CrossRef]
- Tipper, E.T.; Bickle, M.J.; Galy, A.; West, A.J.; Pomiès, C.; Chapman, H.J. The short-term climatic sensitivity of carbonate and silicate weathering fluxes: Insight from seasonal variations in river chemistry. Geochim. Cosmochim. Acta 2006, 70, 2737–2754. [Google Scholar] [CrossRef]
- Galy, A.; France-Lanord, C. Weathering processes in the Ganges–Brahmaputra basin and the riverine alkalinity budget. Chem. Geol. 1999, 159, 31–60. [Google Scholar] [CrossRef]
- Conley, D.J. Terrestrial ecosystems and the global biogeochemical silica cycle. Glob. Biogeochem. 2002, 16, 61–68. [Google Scholar] [CrossRef]
- Beusen, A.H.W.; Bouwman, A.F.; Durr, H.H.; Dekkers, A.L.M.; Hartmann, J. Global patterns of dissolved silica export to the coastal zone: Results from a spatially explicit global model. Glob. Biogeochem. 2009, 23. [Google Scholar] [CrossRef]
- Conley, D.J. Riverine contribution of biogenic silica to the oceanic silica budget. Limnol. Oceanogr. 1997, 42, 774–777. [Google Scholar] [CrossRef]
- Admiraal, W.; Breugem, P.; Jacobs, D.; Steveninck, E. Fixation of dissolved silicate and sedimentation of biogenic silicate in the lower river Rhine during diatom blooms. Biogeochemistry 1990, 9, 175–185. [Google Scholar] [CrossRef]
- Dokkum, H.P.; Hulskotte, J.H.J.; Kramer, K.J.M.; Wilmot, J. Emission, fate and effects of soluble silicates (waterglass) in the aquatic environment. Environ. Sci. Technol. 2004, 38, 515–521. [Google Scholar] [CrossRef]
- Yan, W.; Mayorga, E.; Li, X.; Seitzinger, S.P.; Bouwman, A.F. Increasing anthropogenic nitrogen inputs and riverine DIN exports from the Changjiang River basin under changing human pressures. Glob. Biogeochem. Cycles 2010, 24. [Google Scholar] [CrossRef]
- Keller, D.P.; Hood, R.R. Modeling the seasonal autochthonous sources of dissolved organic carbon and nitrogen in the upper Chesapeake Bay. Ecol. Modell. 2011, 222, 1139–1162. [Google Scholar] [CrossRef]
- Alkhatib, M.; Del Giorgio, P.A.; Gelinas, Y.; Lehmann, M.F. Benthic fluxes of dissolved organic nitrogen in the lower St. Lawrence estuary and implications for selective organic matter degradation. Biogeosciences 2013, 10, 7609–7622. [Google Scholar] [CrossRef]
- Kerner, M.; Spitzy, A. Nitrate regeneration coupled to degradation of different size fractions of DON by the picoplankton in the Elbe estuary. Microb. Ecol. 2001, 41, 69–81. [Google Scholar] [CrossRef]
- Hartmann, J.; Levy, J.; Kempe, S. Increasing dissolved silica trends in the rhine river: An effect of recovery from high Ploads? Limnology 2011, 12, 63–73. [Google Scholar] [CrossRef]
- Keesstra, S.; Nunes, J.P.; Saco, P.; Parsons, T.; Poeppl, R.; Masselink, R.; Cerdà, A. The way forward: Can connectivity be useful to design better measuring and modelling schemes for water and sediment dynamics? Sci. Total Environ. 2018, 644, 1557–1572. [Google Scholar] [CrossRef] [PubMed]
- Keesstra, S.D.; Davis, J.; Masselink, R.H.; Casalí, J.; Peeters, E.T.; Dijksma, R. Coupling hysteresis analysis with sediment and hydrological connectivity in three agricultural catchments in Navarre, Spain. J. Soil Sediment. 2019, 19, 1598–1612. [Google Scholar] [CrossRef]
- Masselink, R.J.; Heckmann, T.; Temme, A.J.; Anders, N.S.; Gooren, H.P.; Keesstra, S.D. A network theory approach for a better understanding of overland flow connectivity. Hydrol. Process. 2017, 31, 207–220. [Google Scholar] [CrossRef]
- Górski, J.; Dragon, K.; Kaczmarek, P.M.J. Nitrate pollution in the Warta River (Poland) between 1958 and 2016: Trends and causes. Environ. Sci. Pollut. Res. 2019, 26, 2038–2046. [Google Scholar] [CrossRef] [PubMed]
- Keller, D.P.; Hood, R.R. Comparative simulations of dissolved organic matter cycling in idealized oceanic, coastal, and estuarine surface waters. J. Mar. Syst. 2013, 109–110, 109–128. [Google Scholar] [CrossRef]
- Meybeck, M. Carbon, nitrogen, and phosphorus transport by world rivers. Am. J. Sci. 1982, 282, 401–450. [Google Scholar] [CrossRef]
- Wiegner, T.N.; Seitzinger, S.P. Seasonal bioavailability of dissolved organic carbon and nitrogen from pristine and polluted freshwater wetlands. Limnol. Oceanogr. 2004, 49, 1703–1712. [Google Scholar] [CrossRef]
- Aitkenhead-Peterson, J.A.; Steele, M.K.; Nahar, N.; Santhy, K. Dissolved organic carbon and nitrogen in urban and rural watersheds of south-central Texas: Land use and land management influences. Biogeochemistry 2009, 96, 119–129. [Google Scholar] [CrossRef]
- Panizza, M.; Cerisola, G. Removal of organic pollutants from industrial wastewater by electrogenerated Fenton’s reagent. Water Res. 2001, 35, 3987–3992. [Google Scholar] [CrossRef]
| Dissolved Load | The Upper Mun | The Middle Mun | The Lower Mun | |||
|---|---|---|---|---|---|---|
| Range | Mean Value | Range | Mean Value | Range | Mean Value | |
| mg/L | mg/L | mg/L | ||||
| TDS | 44–1456 | 363 | 38–839 | 321 | 14–332 | 154 |
| DOC | 2.6–17.1 | 10.7 | 4.5–12.7 | 9.5 | 2.5–8.1 | 6.8 |
| Cl− | 2.7–668.5 | 103.7 | 8.7–361.5 | 123.6 | 1.7–136.8 | 39.9 |
| K+ | 2.3–14.1 | 6.9 | 1.7–6.8 | 3.8 | 1.2–6.3 | 2.9 |
| SO42− | 1.4–18.3 | 9.8 | 0.5–52.5 | 11.5 | 0.8–12.7 | 5.7 |
| DIN-N | 0.12–1.11 | 0.45 | 0.24–3.69 | 0.61 | 0.20–0.74 | 0.39 |
| DSi | 1.7–8.9 | 4.8 | 2.3–9.9 | 4.9 | 2.1–6.2 | 4.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Han, G.; Liu, X.; Liu, M.; Song, C.; Yang, K.; Li, X.; Zhang, Q. Distributive Characteristics of Riverine Nutrients in the Mun River, Northeast Thailand: Implications for Anthropogenic Inputs. Water 2019, 11, 954. https://doi.org/10.3390/w11050954
Liu J, Han G, Liu X, Liu M, Song C, Yang K, Li X, Zhang Q. Distributive Characteristics of Riverine Nutrients in the Mun River, Northeast Thailand: Implications for Anthropogenic Inputs. Water. 2019; 11(5):954. https://doi.org/10.3390/w11050954
Chicago/Turabian StyleLiu, Jinke, Guilin Han, Xiaolong Liu, Man Liu, Chao Song, Kunhua Yang, Xiaoqiang Li, and Qian Zhang. 2019. "Distributive Characteristics of Riverine Nutrients in the Mun River, Northeast Thailand: Implications for Anthropogenic Inputs" Water 11, no. 5: 954. https://doi.org/10.3390/w11050954
APA StyleLiu, J., Han, G., Liu, X., Liu, M., Song, C., Yang, K., Li, X., & Zhang, Q. (2019). Distributive Characteristics of Riverine Nutrients in the Mun River, Northeast Thailand: Implications for Anthropogenic Inputs. Water, 11(5), 954. https://doi.org/10.3390/w11050954







