Climate Change Impact on the Evolution of the Saline Lakes of the Soan-Sakaser Valley (Central Salt Range; Pakistan): Evidences from Hydrochemistry and Water (δD, δ18O) and Chlorine (δ37Cl) Stable Isotopes
Abstract
1. Introduction
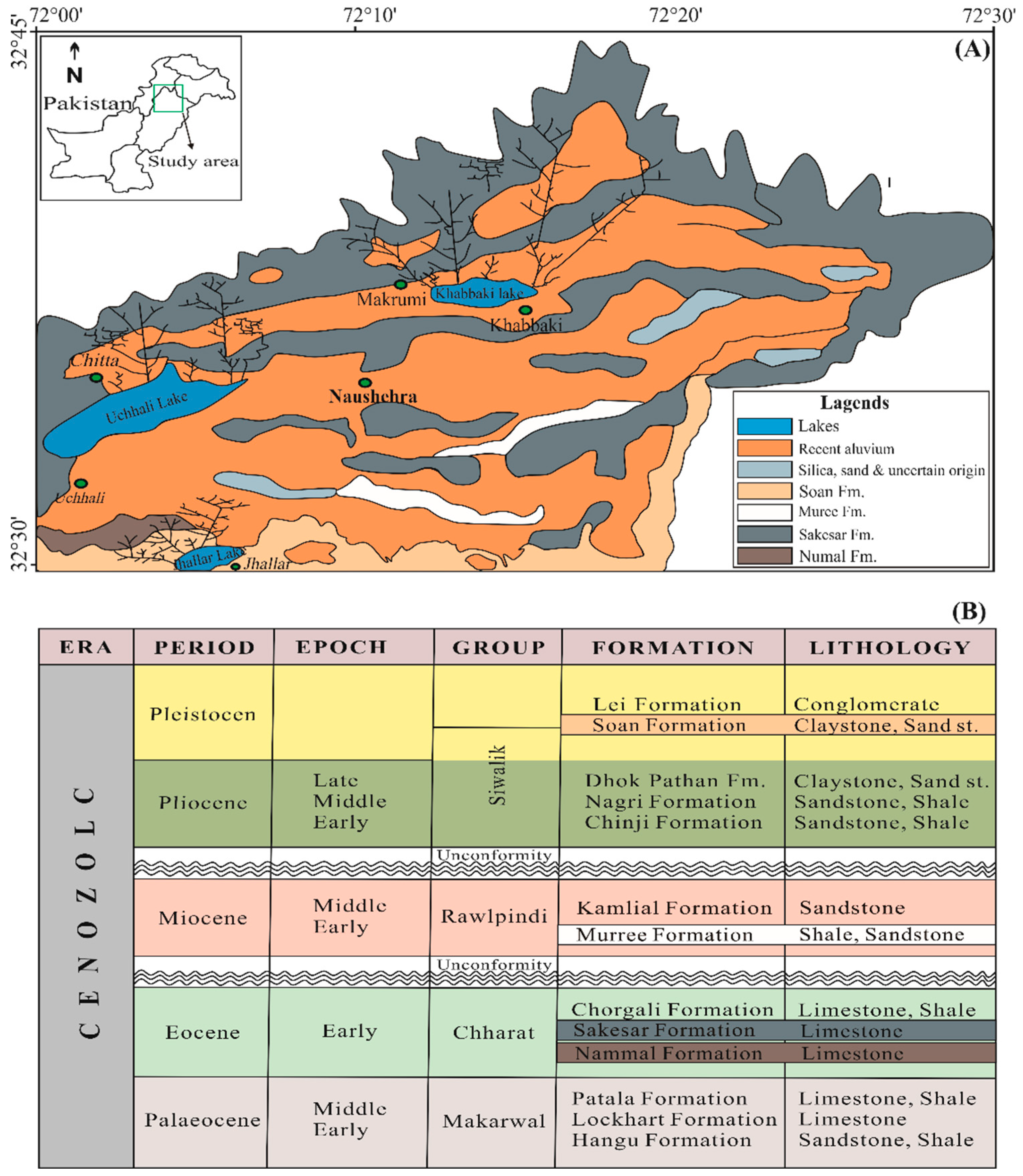
2. Study Site
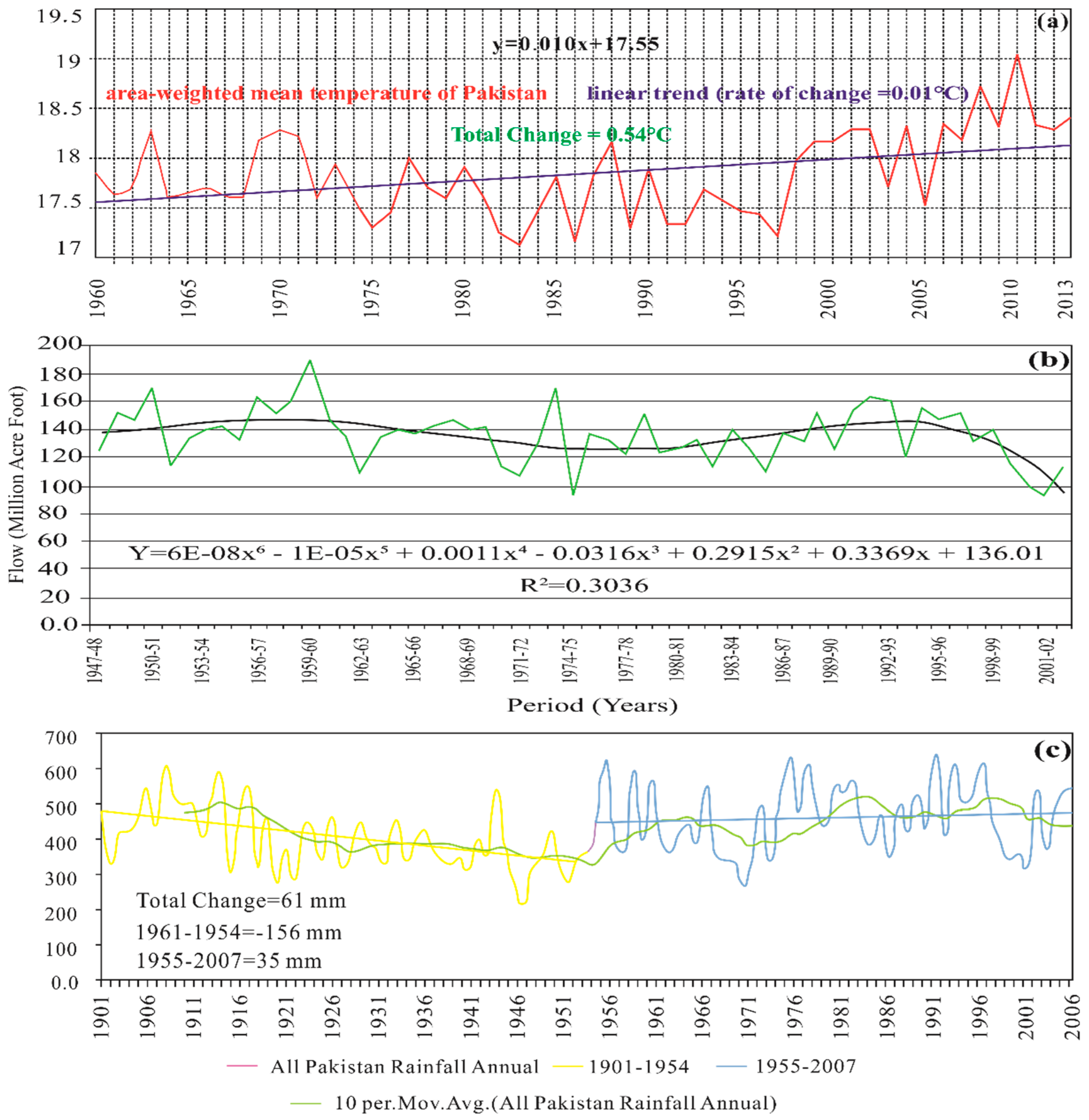
Hydrogeology and Climate Conditions
3. Materials and Methods
3.1. Sampling
3.2. Sample Preparation (δ37Cl)
4. Results and Discussion
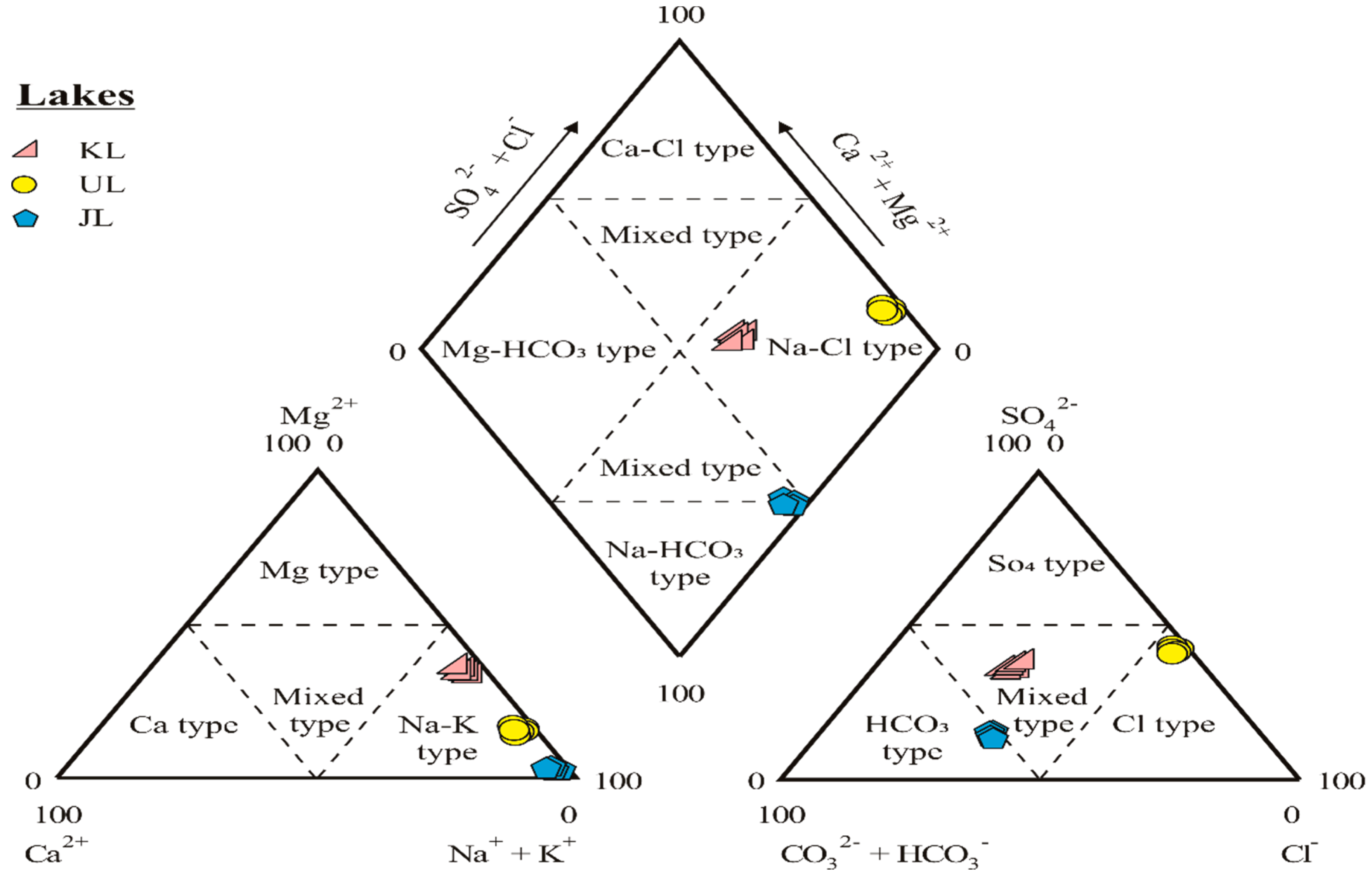
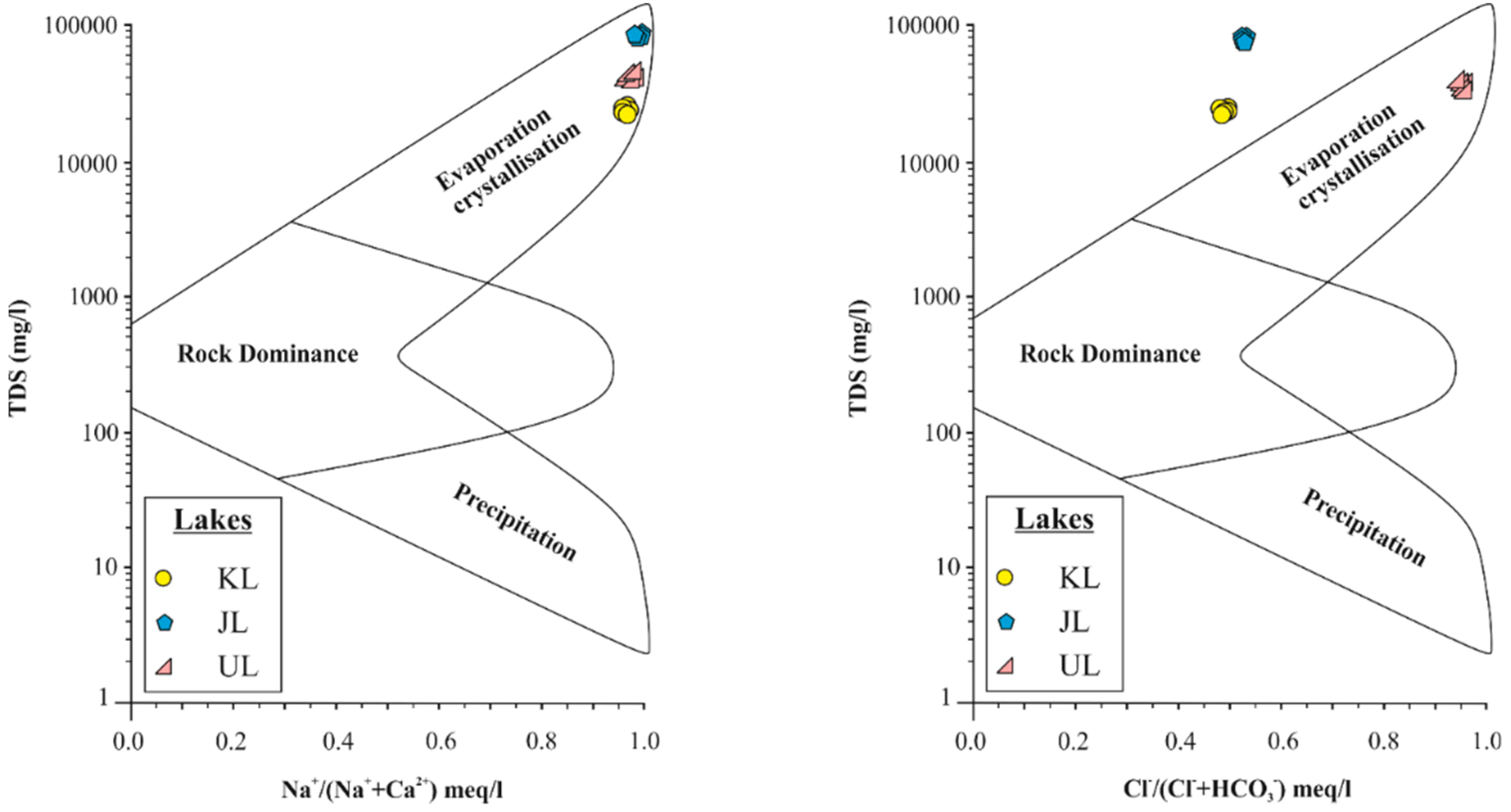
4.1. Salinity and Hydrofacies
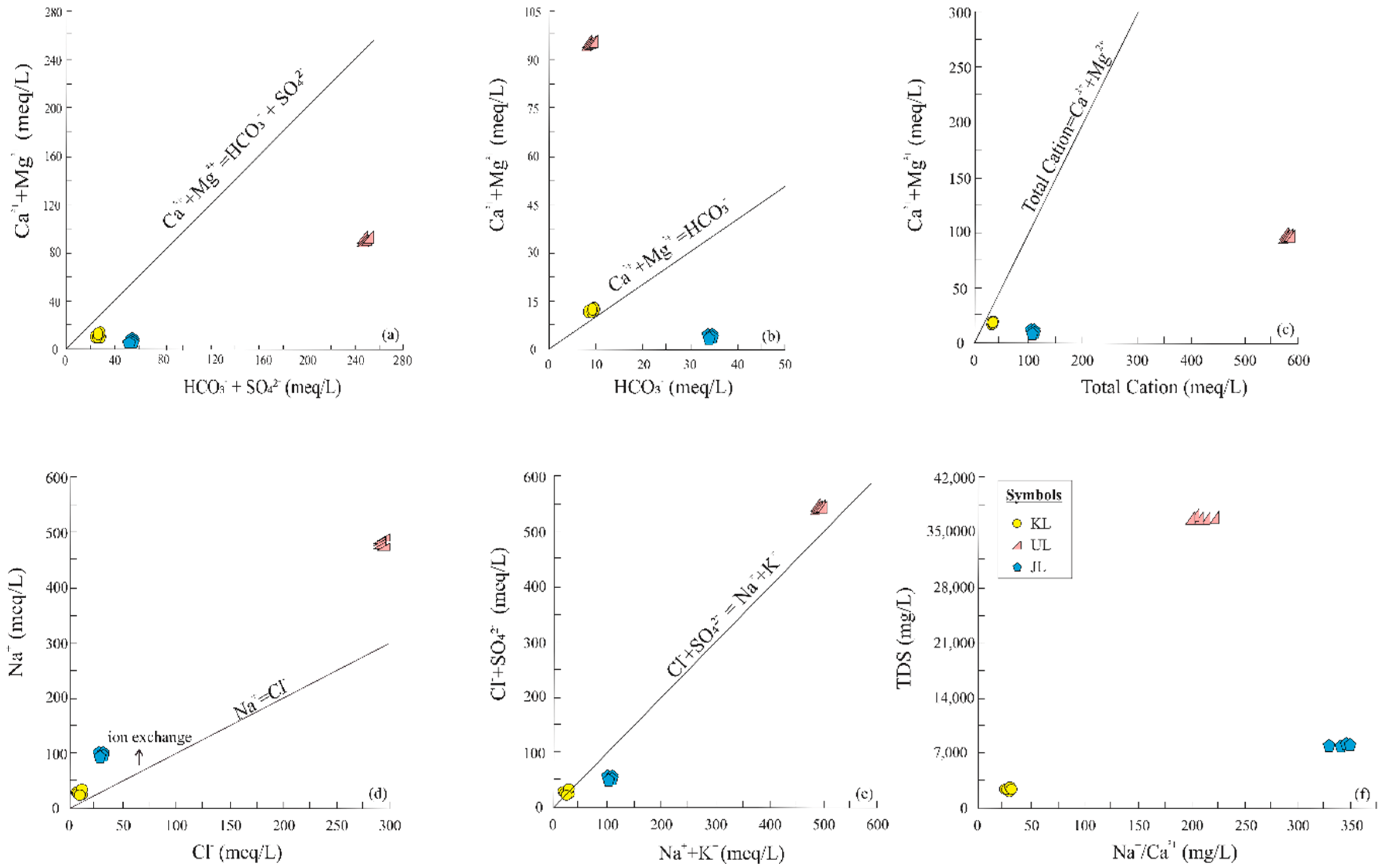
4.2. Sources and Processes Controlling Ion Compositions of the Lakes
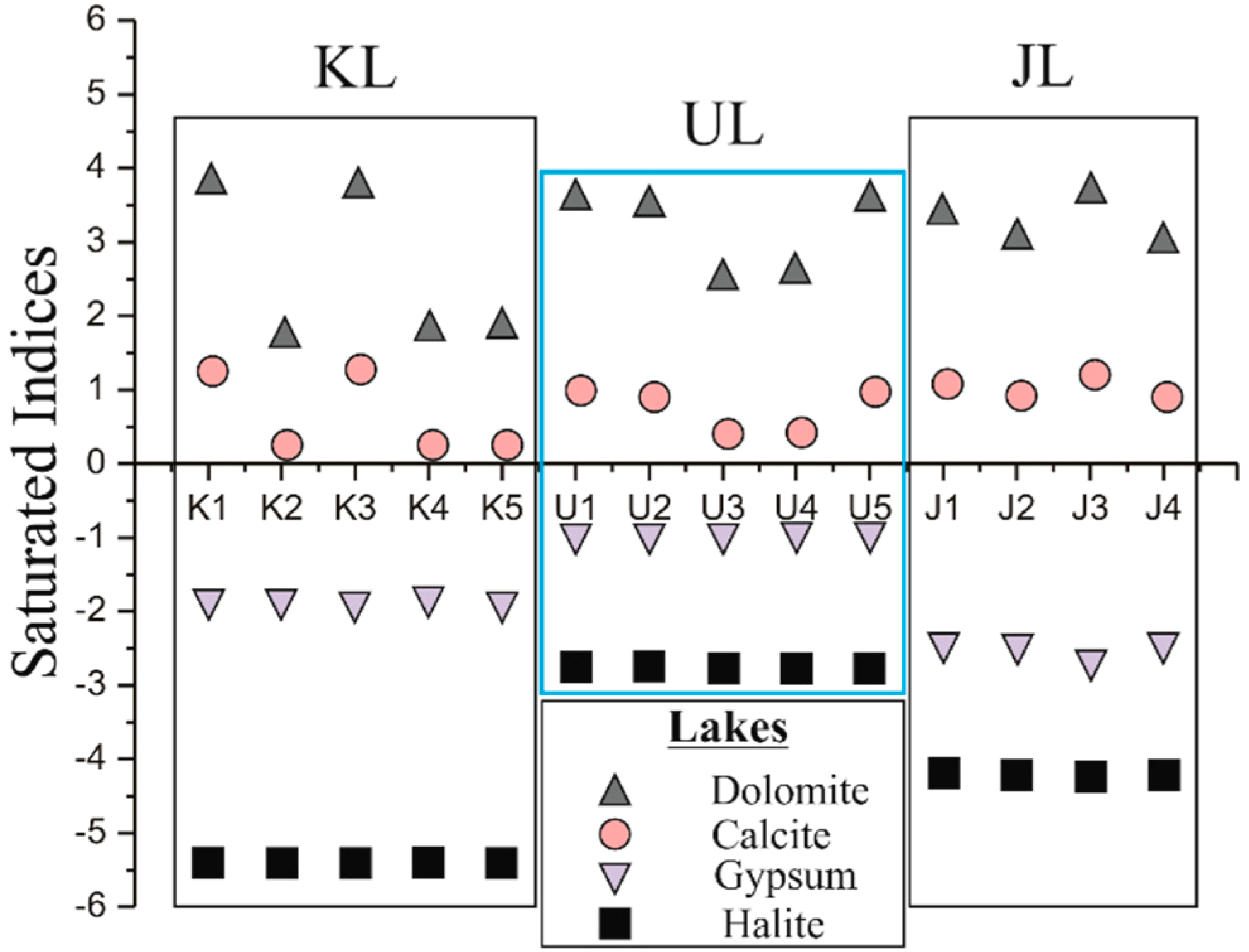
4.3. Dissolution and Deposition
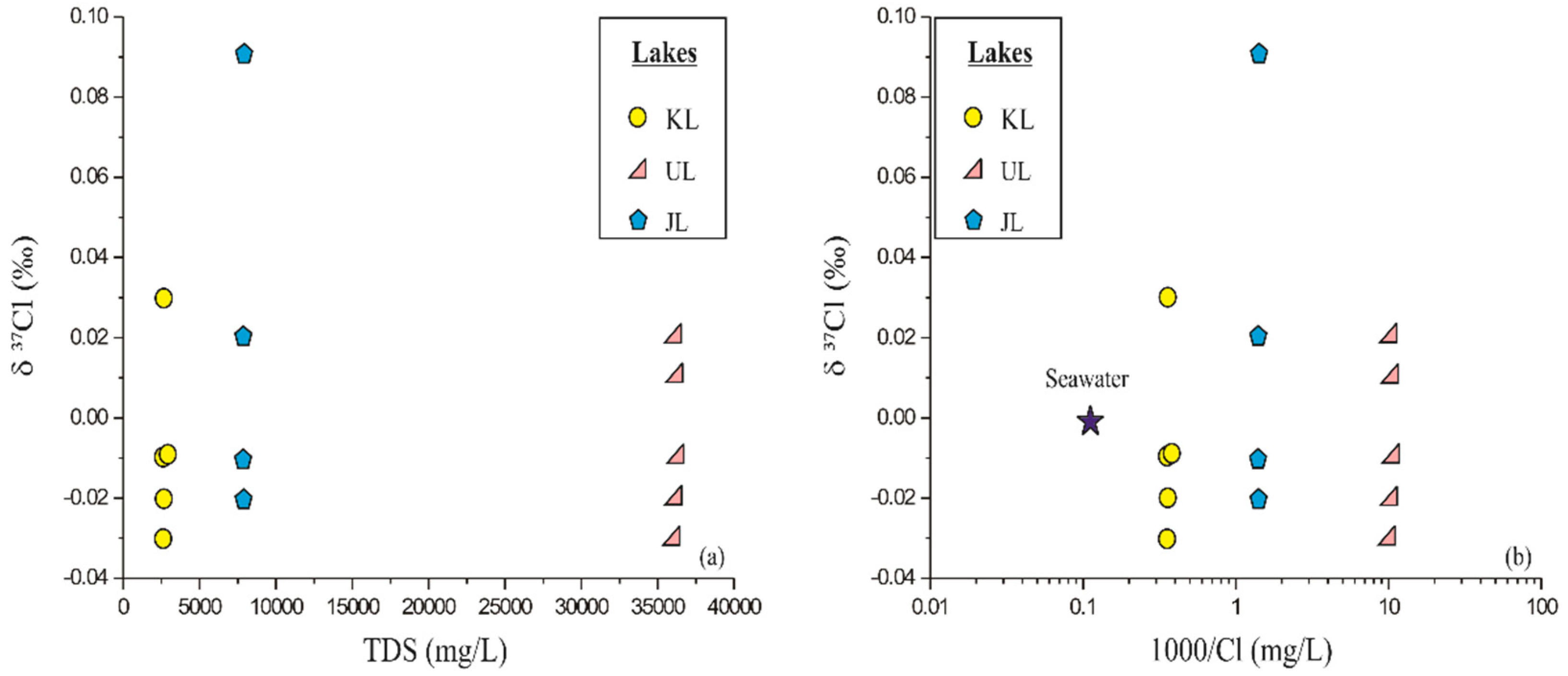
4.4. Chlorine Isotope Compositions
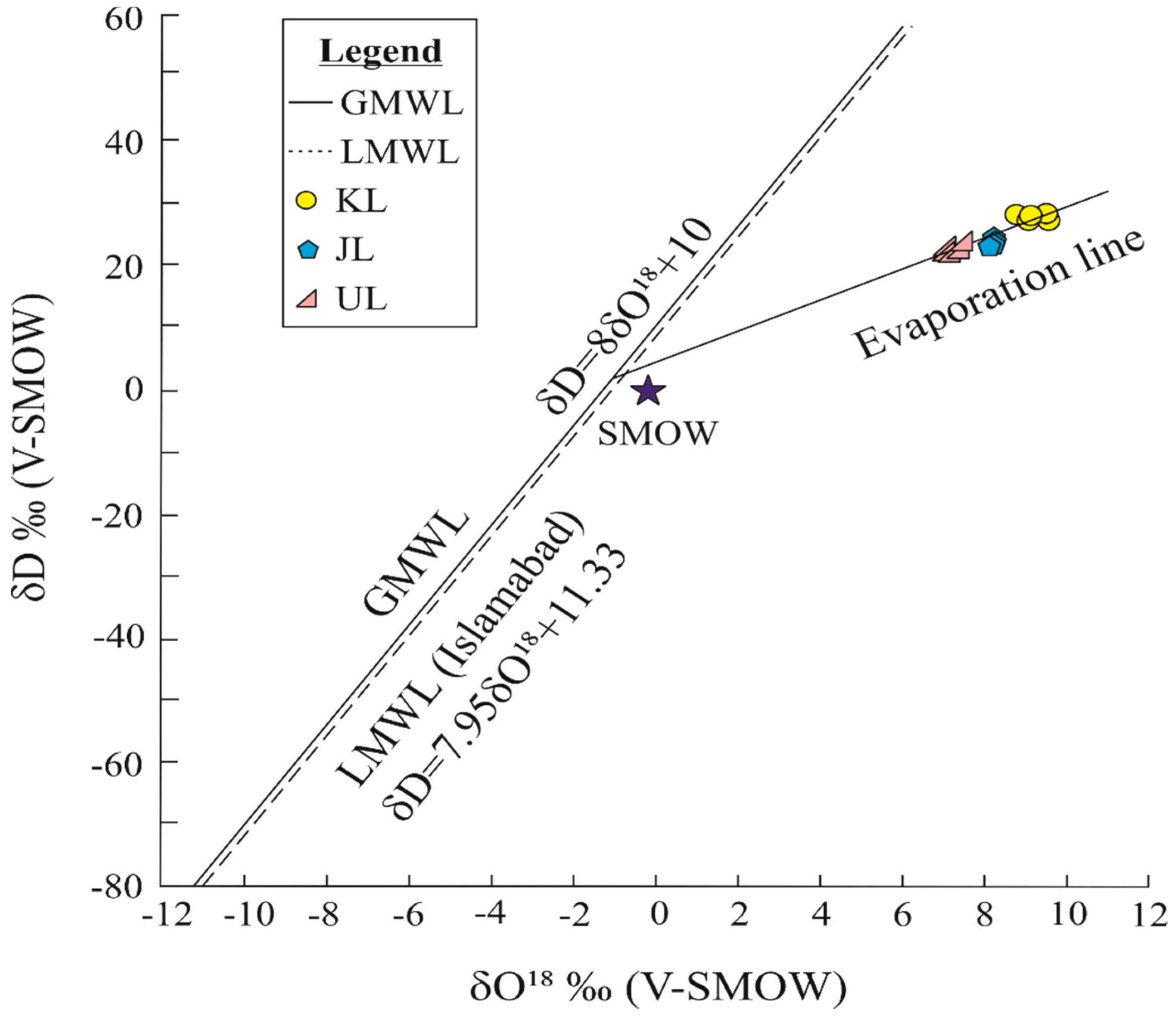
4.5. Water Stable Isotope Compositions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wen, X.; Wu, Y.; Su, J.; Zhang, Y.; Liu, F. Hydrochemical characteristics and salinity of groundwater in the Ejina Basin, Northwestern China. Environ. Geol. 2005, 48, 665–675. [Google Scholar] [CrossRef]
- Hussain, S.A.; Hussain, A.; Fatima, U.; Ali, W.; Hussain, A.; Hussain, N. Evaluation of drinking water quality in urban areas of Pakistan: A case study of Gulshan-e-Iqbal Karachi, Pakistan. J. Biodivers. Environ. Sci. 2016, 8, 64–76. [Google Scholar]
- Messager, M.L.; Lehner, B.; Grill, G.; Nedeva, I.; Schmitt, O. Estimating the volume and age of water stored in global lakes using a geo-statistical approach. Nat. Commun. 2016, 7, 13603. [Google Scholar] [CrossRef] [PubMed]
- MEA, M.E.A. Ecosystems and Human Well-Being: Wetlands and Water; World Resources Institute: Washington, DC, USA, 2005. [Google Scholar]
- Williams, W.D. What future for saline lakes? Environ. Sci. Policy Sustain. Dev. 1996, 38, 12–39. [Google Scholar] [CrossRef]
- Wurtsbaugh, W.A.; Miller, C.; Null, S.E.; DeRose, R.J.; Wilcock, P.; Hahnenberger, M.; Howe, F.; Moore, J. Decline of the world’s saline lakes. Nat. Geosci. 2017, 10, 816. [Google Scholar] [CrossRef]
- Fisher, R.S.; Mullican, W.F., III. Hydrochemical evolution of sodium-sulfate and sodium-chloride groundwater beneath the northern Chihuahuan Desert, Trans-Pecos, Texas, USA. Hydrogeol. J. 1997, 5, 4–16. [Google Scholar] [CrossRef]
- Kraft, G.J.; Stites, W.; Mechenich, D. Impacts of irrigated vegetable agriculture on a humid north-central US sand plain aquifer. Groundwater 1999, 37, 572–580. [Google Scholar] [CrossRef]
- Chaudhry, Q.U.Z. Climate Change Profile of Pakistan; Asian Development Bank: Mandaluyong, Philippines, 2017. [Google Scholar]
- Chaudhry, Q.-U.-Z.; Mahmood, A.; Rasul, G.; Afzaal, M. Climate Change Indicators of Pakistan; Pakistan Meteorological Department: Islamabad, Pakistan, 2009.
- Farooqi, A.B.; Khan, A.H.; Mir, H. Climate change perspective in Pakistan. Pak. J. Meteorol. 2005, 2, 11–21. [Google Scholar]
- Ahmad, S. Water Resources and Management in Pakistan–Challenges and Future Vision. Course on Integrated Water Resources Management at the National University. 2009. [Google Scholar]
- Rasul, G.; Afzal, M.; Zahid, M.; Ahsan, A.; Bukhari, A. Climate Change in Pakistan, Focused on Sindh Province; Technical Report No. PMD-25/2012; Pakistan Meteorological Department: Islamabad, Pakistan, 2012.
- Kahlown, M.A.; Majeed, A. Water-resources situation in Pakistan: Challenges and future strategies. In Water Resources in the South: Present Scenario and Future Prospects; Commission on Science and Technology for Sustainable Development in the South: Islamabad, Pakistan, 2003; pp. 20–39. [Google Scholar]
- Ahmad, M.; Farooq, U. The state of food security in Pakistan: Future challenges and coping strategies. Pak. Dev. Rev. 2010, 49, 903–923. [Google Scholar] [CrossRef]
- Rafique, T.; Naseem, S.; Usmani, T.H.; Bashir, E.; Khan, F.A.; Bhanger, M.I. Geochemical factors controlling the occurrence of high fluoride groundwater in the Nagar Parkar area, Sindh, Pakistan. J. Hazard. Mater. 2009, 171, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Naseem, S.; Rafique, T.; Bashir, E.; Bhanger, M.I.; Laghari, A.; Usmani, T.H. Lithological influences on occurrence of high-fluoride groundwater in Nagar Parkar area, Thar Desert, Pakistan. Chemosphere 2010, 78, 1313–1321. [Google Scholar] [CrossRef]
- Bashir, E.; Huda, S.N.-u.; Naseem, S.; Hamza, S.; Kaleem, M. Geochemistry and quality parameters of dug and tube well water of Khipro, District Sanghar, Sindh, Pakistan. Appl. Water Sci. 2017, 7, 1645–1655. [Google Scholar] [CrossRef]
- Bashir, E.; Naseem, S.; Hanif, H.; Pirzada, T. Geochemical study of groundwater of Uthal and Bela areas, Balochistan and its appraisal for drinking and irrigation water quality. Int. J. Agric. Environ. 2013, 2, 1–13. [Google Scholar]
- Sher, H.; Aldosari, A. Strategic program for biodiversity and water resource management and climate change adaptation in Pakistan. In Proceedings of the EGU General Assembly 2014, Vienna, Austria, 27 April–2 May 2014. [Google Scholar]
- Khoso, S.; Wagan, H.F.; Tunio, H.A.; Ansari, A.A. An overview on emerging water scarcity in Pakistan, its causes, impacts and remedial measures. J. Appl. Eng. Sci. 2015, 13, 35–44. [Google Scholar] [CrossRef]
- Ghalib, S.A.; Rais, M.; Abbas, D.; Tabassum, F.; Begum, A.; Jabeen, T. An overview of the status of shorebirds and internationally important sites in Pakistan. Pak. J. Zool. 2009, 41, 165–172. [Google Scholar]
- Arshad, M.; Mehmood, N.; Hira Muqadas, I.M.; Chaudhry, J.I.; Iram, A.; Batool, F.Z.; Malik, A. Avifauna census, assesment of heavy metal pollution and its effects on aquatic biota in relation with alteration in precipitation at jahlar lake, punjab, pakistan. Int. J. Agric. Appl. Sci. 2015, 7, 193–201. [Google Scholar]
- Arshad, M.; Mehmood, N.; Muqadas, H.; Chaudhry, J.; Mustafa, I.; Khan, M.R.; Malik, I.U.; Ahmed, H. Avifauna Studies in Co-Relation with Alteration in Climatic Patterns and Hydrology of Uchalli Lake, Punjab, Pakistan. Pak. J. Zool. 2014, 46, 503–515. [Google Scholar]
- Afzal, S.; Younas, M.; Ali, K. Selenium speciation studies from Soan-Sakesar Valley, salt range, Pakistan. Water Int. 2000, 25, 425–436. [Google Scholar] [CrossRef]
- Vengosh, A.; Kloppmann, W.; Marei, A.; Livshitz, Y.; Gutierrez, A.; Banna, M.; Guerrot, C.; Pankratov, I.; Raanan, H. Sources of salinity and boron in the Gaza strip: Natural contaminant flow in the southern Mediterranean coastal aquifer. Water Resour. Res. 2005, 41, W01013. [Google Scholar] [CrossRef]
- Han, D.; Song, X.; Currell, M.J.; Yang, J.; Xiao, G. Chemical and isotopic constraints on evolution of groundwater salinization in the coastal plain aquifer of Laizhou Bay, China. J. Hydrol. 2014, 508, 12–27. [Google Scholar] [CrossRef]
- Lapworth, D.; MacDonald, A.; Tijani, M.; Darling, W.; Gooddy, D.; Bonsor, H.; Araguás-Araguás, L. Residence times of shallow groundwater in West Africa: Implications for hydrogeology and resilience to future changes in climate. Hydrogeol. J. 2013, 21, 673–686. [Google Scholar] [CrossRef]
- Vanderzalm, J.; Jeuken, B.; Wischusen, J.; Pavelic, P.; La Salle, C.L.G.; Knapton, A.; Dillon, P. Recharge sources and hydrogeochemical evolution of groundwater in alluvial basins in arid central Australia. J. Hydrol. 2011, 397, 71–82. [Google Scholar] [CrossRef]
- Amundson, R.; Barnes, J.D.; Ewing, S.; Heimsath, A.; Chong, G. The stable isotope composition of halite and sulfate of hyperarid soils and its relation to aqueous transport. Geochim. Cosmochim. Acta 2012, 99, 271–286. [Google Scholar] [CrossRef]
- Ala-Aho, P.; Soulsby, C.; Pokrovsky, O.S.; Kirpotin, S.N.; Karlsson, J.; Serikova, S.; Vorobyev, S.N.; Manasypov, R.M.; Loiko, S.; Tetzlaff, D. Using stable isotopes to assess surface water source dynamics and hydrological connectivity in a high-latitude wetland and permafrost influenced landscape. J. Hydrol. 2018, 556, 279–293. [Google Scholar] [CrossRef]
- Kumar, S.; Prakash, S.; Logan, A.; Singh, Y. Tracing groundwater recharge sources in the northwestern Indian alluvial aquifer using water isotopes (δ18O, δ2H and 3H). J. Hydrol. 2018, 559, 835–847. [Google Scholar]
- Kaufmann, R.S.; Frape, S.; McNutt, R.; Eastoe, C. Chlorine stable isotope distribution of Michigan Basin formation waters. Appl. Geochem. 1993, 8, 403–407. [Google Scholar] [CrossRef]
- Eastoe, C.; Long, A.; Land, L.S.; Kyle, J.R. Stable chlorine isotopes in halite and brine from the Gulf Coast Basin: Brine genesis and evolution. Chem. Geol. 2001, 176, 343–360. [Google Scholar] [CrossRef]
- Berry Lyons, W.; Frape, S.K.; Welch, K.A. History of McMurdo Dry Valley lakes, Antarctica, from stable chlorine isotope data. Geology 1999, 27, 527–530. [Google Scholar] [CrossRef]
- Ying-kai, X.; Wei-guo, L.; Yin-min, Z.; Yun-hui, W.; Shirodkar, P. Variations in isotopic compositions of chlorine in evaporation-controlled salt lake brines of Qaidam Basin, China. Chin. J. Oceanol. Limnol. 2000, 18, 169–177. [Google Scholar] [CrossRef]
- Eastoe, C. Stable chlorine isotopes in arid non-marine basins: Instances and possible fractionation mechanisms. Appl. Geochem. 2016, 74, 1–12. [Google Scholar] [CrossRef][Green Version]
- Eggenkamp, H. The Geochemistry of Stable Chlorine and Bromine Isotopes; Springer: Berlin, Germany, 2014. [Google Scholar]
- Banks, D.; Green, R.; Cliff, R.; Yardley, B. Chlorine isotopes in fluid inclusions: Determination of the origins of salinity in magmatic fluids. Geochim. Cosmochim. Acta 2000, 64, 1785–1789. [Google Scholar] [CrossRef]
- Luo, C.; Xiao, Y.; Wen, H.; Ma, H.; Ma, Y.; Zhang, Y.; Zhang, Y.; He, M. Stable isotope fractionation of chlorine during the precipitation of single chloride minerals. Appl. Geochem. 2014, 47, 141–149. [Google Scholar] [CrossRef]
- Richards, L.A.; Magnone, D.; Boyce, A.J.; Casanueva-Marenco, M.J.; van Dongen, B.E.; Ballentine, C.J.; Polya, D.A. Delineating sources of groundwater recharge in an arsenic-affected Holocene aquifer in Cambodia using stable isotope-based mixing models. J. Hydrol. 2018, 557, 321–334. [Google Scholar] [CrossRef]
- Kamtchueng, B.T.; Fantong, W.Y.; Wirmvem, M.J.; Tiodjio, R.E.; Takounjou, A.F.; Asai, K.; Djomou, S.L.B.; Kusakabe, M.; Ohba, T.; Tanyileke, G. A multi-tracer approach for assessing the origin, apparent age and recharge mechanism of shallow groundwater in the Lake Nyos catchment, Northwest, Cameroon. J. Hydrol. 2015, 523, 790–803. [Google Scholar] [CrossRef]
- Chaudhry, A.A. Wetlands in Pakistan: What is happening to them? World Environment Day, 5 June 2010. [Google Scholar]
- Afzal, S.; Younas, M.; Hussain, K. Physical and chemical characterisation of the agricultural lands of the Soan–Sakesar Valley, Salt Range, Pakistan. Soil Res. 1999, 37, 1035–1046. [Google Scholar] [CrossRef]
- Afzal, S.; Younas, M.; Ali, K. Temporal variability of the water quality of saline lakes from the Soan-Sakesar Valley, Salt Range, Pakistan. Water Qual. Res. J. 1998, 33, 331–346. [Google Scholar] [CrossRef]
- Ahmad, I.; Hussain, M.; Ahmad, M.S.A.; Ashraf, M.Y.; Ahmad, R.; Ali, A. Spatio-temporal variations in physiochemical attributes of Adiantum capillus-veneris from Soone Valley of salt range (Pakistan). Pak. J. Bot. 2008, 40, 1387–1398. [Google Scholar]
- Arshad, M. Management Plan Uchalli Wetlands Complex; The Ministry of Environment’s Pakistan Wetlands Programme: Islamabad, Pakistan, 2011.
- Qinghai Institute of Salt Lakes, Chinese Academy of Sciences. Analytical Method of Brines and Salts; Science Press Beijing: Beijing, China, 1988; pp. 29–368. (In Chinese) [Google Scholar]
- Magenheim, A.J.; Spivack, A.J.; Michael, P.J.; Gieskes, J.M. Chlorine stable isotope composition of the oceanic crust: Implications for Earth’s distribution of chlorine. Earth Planet. Sci. Lett. 1995, 131, 427–432. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhou, Y.; Liu, W. Precise Measurement of Chlorine Isotopes Based on Cs2Cl2 by Thermal Ionization Mass Spectrometry. Anal. Lett. 1995, 28, 1295–1304. [Google Scholar] [CrossRef]
- Xiao, Y.; Yinming, Z.; Qingzhong, W.; Haizhen, W.; Weiguo, L.; Eastoe, C. A secondary isotopic reference material of chlorine from selected seawater. Chem. Geol. 2002, 182, 655–661. [Google Scholar] [CrossRef]
- Wan, H.; Liu, W. An isotope study (δ18O and δD) of water movements on the Loess Plateau of China in arid and semiarid climates. Ecol. Eng. 2016, 93, 226–233. [Google Scholar] [CrossRef]
- Liu, W.; Yang, H.; Wang, H.; Yao, Y.; Wang, Z.; Cao, Y. Influence of aquatic plants on the hydrogen isotope composition of sedimentary long-chain n-alkanes in the Lake Qinghai region, Qinghai-Tibet Plateau. Sci. China Earth Sci. 2016, 59, 1368–1377. [Google Scholar] [CrossRef]
- Stallard, R.; Edmond, J. Geochemistry of the Amazon: 2. the influence of geology and weathering environment on the dissolved load. J. Geophys. Res. Ocean. 1983, 88, 9671–9688. [Google Scholar] [CrossRef]
- Sami, K. Recharge mechanisms and geochemical processes in a semi-arid sedimentary basin, Eastern Cape, South Africa. J. Hydrol. 1992, 139, 27–48. [Google Scholar] [CrossRef]
- Spears, D. Mineralogical control of the chemical evolution of groundwater. In Solute Processes; Wiley: Chichester, UK, 1986; p. 512. [Google Scholar]
- Hounslow, A. Water Quality Data: Analysis and Interpretation; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Singh, A.K.; Hasnain, S. Environmental geochemistry of Damodar River basin, east coast of India. Environ. Geol. 1999, 37, 124–136. [Google Scholar] [CrossRef]
- Chidambaram, S. Recent Trends in Water Research: Hydrogeochemical and Hydrological Perspectives; I.K. International Publishing House Pvt. Ltd.: New Delhi, India, 2010. [Google Scholar]
- Okiongbo, K.; Douglas, R. Evaluation of major factors influencing the geochemistry of groundwater using graphical and multivariate statistical methods in Yenagoa city, Southern Nigeria. Appl. Water Sci. 2015, 5, 27–37. [Google Scholar] [CrossRef]
- Chebotarev, I. Metamorphism of natural waters in the crust of weathering—1. Geochim. Cosmochim. Acta 1955, 8, 22–48. [Google Scholar] [CrossRef]
- Qian, H.; Ma, Z.; Li, P. Hydrogeochemistry; Geological Publishing House: Beijing, China, 2005. [Google Scholar]
- Eggenkamp, H.; Kreulen, R.; Van Groos, A.K. Chlorine stable isotope fractionation in evaporites. Geochim. Cosmochim. Acta 1995, 59, 5169–5175. [Google Scholar] [CrossRef]
- Vengosh, A.; Chivas, A.R.; McCulloch, M.T. Direct determination of boron and chlorine isotopic compositions in geological materials by negative thermal-ionization mass spectrometry. Chem. Geol. Isot. Geosci. Sect. 1989, 79, 333–343. [Google Scholar] [CrossRef]
- Sherif, M.I.; Sultan, M.; Sturchio, N.C. Chlorine isotopes as tracers of solute origin and age of groundwaters from the Eastern Desert of Egypt. Earth Planet. Sci. Lett. 2019, 510, 37–44. [Google Scholar] [CrossRef]
- Zhang, M.; Frape, S.K.; Love, A.J.; Herczeg, A.L.; Lehmann, B.; Beyerle, U.; Purtschert, R. Chlorine stable isotope studies of old groundwater, southwestern Great Artesian Basin, Australia. Appl. Geochem. 2007, 22, 557–574. [Google Scholar] [CrossRef]
- Frape, S.; Bryant, G.; Blomqvist, R.; Ruskeeniemi, T. Evidence from stable chlorine isotopes for multiple sources of chloride in groundwaters from crystalline shield environments. In Proceedings of the Symposium on Isotopes in Water Resources Management, Vienna, Austria, 20–24 March 1995. [Google Scholar]
- Eggenkamp, H.G.M. δ37Cl: The Geochemistry of Chlorine Isotopes. Ph.D. Thesis, Utrecht University, Utrecht, The Netherlands, 1994. [Google Scholar]
- Santoni, S.; Huneau, F.; Garel, E.; Vergnaud-Ayraud, V.; Labasque, T.; Aquilina, L.; Jaunat, J.; Celle-Jeanton, H. Residence time, mineralization processes and groundwater origin within a carbonate coastal aquifer with a thick unsaturated zone. J. Hydrol. 2016, 540, 50–63. [Google Scholar] [CrossRef]
- Craig, H. Isotopic variations in meteoric waters. Science 1961, 133, 1702–1703. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.; Xianfang, S.; Hussain, I.; Jianrong, L.; Dong Mei, H.; Li Hu, Y.; Huang, W. Controlling factors of the stable isotope composition in the precipitation of Islamabad, Pakistan. Adv. Meteorol. 2015, 2015, 817513. [Google Scholar] [CrossRef]
| Sample | D | K+ | Na+ | Mg2+ | Ca2+ | SO42− | Cl− | CO3− | HCO3− | B3+ | δ18O | δD | δ37Cl | TDS | Density | pH |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (m) | (mg/L) | (‰) | (g/L) | g/cm3 | ||||||||||||
| K-1 | 0 | 27.66 | 500 | 167.94 | 17.77 | 660.2 | 357.9 | 140 | 589.7 | 2.3 | 9.24 | 27.89 | −0.01 | 2.60 | 1.001 | 8.66 |
| K-2 | 4 | 31.08 | 498.2 | 167.68 | 15.61 | 643.6 | 355.1 | 128 | 592 | 2.15 | 8.87 | 29.31 | 0.03 | 2.63 | 1.001 | 8.63 |
| K-3 | 0 | 31 | 502.1 | 165 | 16.12 | 655.5 | 357.2 | 133 | 590.1 | 2.25 | 9.01 | 28 | −0.03 | 2.61 | 1.001 | 8.63 |
| K-4 | 2 | 29.9 | 499 | 167.2 | 17 | 645 | 356.9 | 127.8 | 590.3 | 2.18 | 9.23 | 28.9 | −0.01 | 2.6 | 1.001 | 8.65 |
| K-5 | 1 | 29.91 | 500.1 | 166.13 | 16.44 | 554 | 356.12 | 133.21 | 590.55 | 2.23 | 9.05 | 28.4 | −0.02 | 2.61 | 1.003 | 8.84 |
| Mean | - | 29.91 | 499.88 | 166.79 | 16.58 | 631.66 | 356.64 | 132.40 | 590.53 | 2.22 | 2.61 | 1.001 | 8.68 | |||
| 1986 | - | 82.11 | 770.5 | 130.1 | 5.81 | 964.1 | 478.7 | - | - | - | - | - | - | 2.65 | - | 8.5 |
| 1994 | - | 32.45 | 508.2 | 149.57 | 37.07 | 417.9 | 484.03 | - | - | 0.64 | - | - | - | 2.4 | - | 8.9 |
| 2007 | - | - | - | - | - | 605.66 | 208.8 | - | - | - | - | - | - | 1.43 | - | 9.5 |
| U-1 | 0 | 151.3 | 11,180.1 | 1062 | 53.64 | 11,590 | 10,536 | 190.6 | 522.7 | 8.06 | 7.73 | 21.08 | −0.03 | 36.10 | 1.027 | 8.84 |
| U-2 | 3.5 | 158.4 | 11,470.34 | 1085 | 51.02 | 11,610 | 10,532 | 162.6 | 550.5 | 8.03 | 7.75 | 22.95 | 0.01 | 36.21 | 1.034 | 8.84 |
| U-3 | 1 | 155.1 | 11,442.9 | 1077.1 | 52 | 11,601.12 | 10,514 | 172.9 | 543.7 | 8.03 | 7.7 | 22.24 | 0.02 | 36.13 | 1.028 | 8.81 |
| U-4 | 0 | 157 | 11,278 | 1072 | 53.65 | 11,699 | 10,525 | 190 | 523 | 8 | 7.71 | 21.98 | −0.01 | 36.20 | 1.024 | 8.83 |
| U-5 | 2 | 155.1 | 11,334.51 | 1074.1 | 53 | 11,580 | 10,528.1 | 181.12 | 539.41 | 8.01 | 7.7 | 22.19 | −0.02 | 36.0 | 1.02 | 8.82 |
| Mean | - | 1.3 | 155.38 | 11,341.17 | 1074 | 52.66 | 11,616 | 10,527. | 179.4 | 535.8 | 1.026 | 8.82 | ||||
| 1986 | - | 344.08 | 11,707 | 680.96 | traces | 16,272 | 8120.34 | - | - | - | - | - | - | 26.83 | - | >11 |
| 1994 | - | 253.98 | 9890 | 1770.5 | 158.92 | 14,288.9 | 9528.1 | - | - | 0.96 | - | - | - | 36.5 | - | 8.81 |
| 2007 | - | - | - | - | - | 7208.5 | 9175 | - | - | ND | - | - | 29.06 | - | 9.21 | |
| J-1 | 0 | 115.02 | 2386.71 | 36.36 | 6.78 | 890.2 | 1414 | 789.3 | 2123 | 3.06 | 8.11 | 23.82 | 0.09 | 7.97 | 1.004 | 9.35 |
| J-2 | 0 | 114 | 2380.09 | 36.59 | 7.2 | 883 | 1419.3 | 788 | 2133.02 | 3.05 | 8.2 | 23.7 | −0.02 | 7.95 | 1.005 | 9.36 |
| J-3 | 0 | 114.3 | 2383.1 | 37 | 6.8 | 889.7 | 1415.1 | 777.12 | 2140.22 | 3 | 8.1 | 23.7 | 0.02 | 7.97 | 1.004 | 9.32 |
| J-4 | 0 | 115.29 | 2382.87 | 35.12 | 6.91 | 886.01 | 1415.2 | 785.1 | 2132.1 | 3.05 | 8.15 | 23.76 | −0.01 | 7.92 | 1.004 | 9.33 |
| Mean | - | 114.65 | 2383.19 | 36.26 | 6.9225 | 887.22 | 1415.9 | 784.88 | 2132.08 | 3.04 | 1.004 | 9.34 | ||||
| 1986 | - | 207.2 | 1897.5 | 52.29 | 12.02 | 984 | 900.7 | - | - | - | - | - | - | 4.96 | - | 9 |
| 1994 | - | 43.0 | 1807.8 | 53.4 | 9.82 | 835.7 | 1035.4 | - | - | 1.23 | - | - | - | 8.75 | - | 9.24 |
| 2007 | - | - | - | - | - | 624.3 | 990 | - | - | ND | - | - | - | 4.2 | - | 9.69 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, S.A.; Han, F.-Q.; Han, W.; Rodríguez, A.; Han, J.-L.; Han, J.; Nian, X.-Q.; Yi, L.; Ma, Z.; Widory, D. Climate Change Impact on the Evolution of the Saline Lakes of the Soan-Sakaser Valley (Central Salt Range; Pakistan): Evidences from Hydrochemistry and Water (δD, δ18O) and Chlorine (δ37Cl) Stable Isotopes. Water 2019, 11, 912. https://doi.org/10.3390/w11050912
Hussain SA, Han F-Q, Han W, Rodríguez A, Han J-L, Han J, Nian X-Q, Yi L, Ma Z, Widory D. Climate Change Impact on the Evolution of the Saline Lakes of the Soan-Sakaser Valley (Central Salt Range; Pakistan): Evidences from Hydrochemistry and Water (δD, δ18O) and Chlorine (δ37Cl) Stable Isotopes. Water. 2019; 11(5):912. https://doi.org/10.3390/w11050912
Chicago/Turabian StyleHussain, Syed Asim, Feng-Qing Han, Wenxia Han, Alejandro Rodríguez, Ji-Long Han, Jibin Han, Xiu-Qing Nian, Lei Yi, Zhe Ma, and David Widory. 2019. "Climate Change Impact on the Evolution of the Saline Lakes of the Soan-Sakaser Valley (Central Salt Range; Pakistan): Evidences from Hydrochemistry and Water (δD, δ18O) and Chlorine (δ37Cl) Stable Isotopes" Water 11, no. 5: 912. https://doi.org/10.3390/w11050912
APA StyleHussain, S. A., Han, F.-Q., Han, W., Rodríguez, A., Han, J.-L., Han, J., Nian, X.-Q., Yi, L., Ma, Z., & Widory, D. (2019). Climate Change Impact on the Evolution of the Saline Lakes of the Soan-Sakaser Valley (Central Salt Range; Pakistan): Evidences from Hydrochemistry and Water (δD, δ18O) and Chlorine (δ37Cl) Stable Isotopes. Water, 11(5), 912. https://doi.org/10.3390/w11050912






