Harmless Treatment of Phenylhydrazine Hydrochloride Production Effluent: From Lab Scale to Pilot Scale
Abstract
1. Introduction
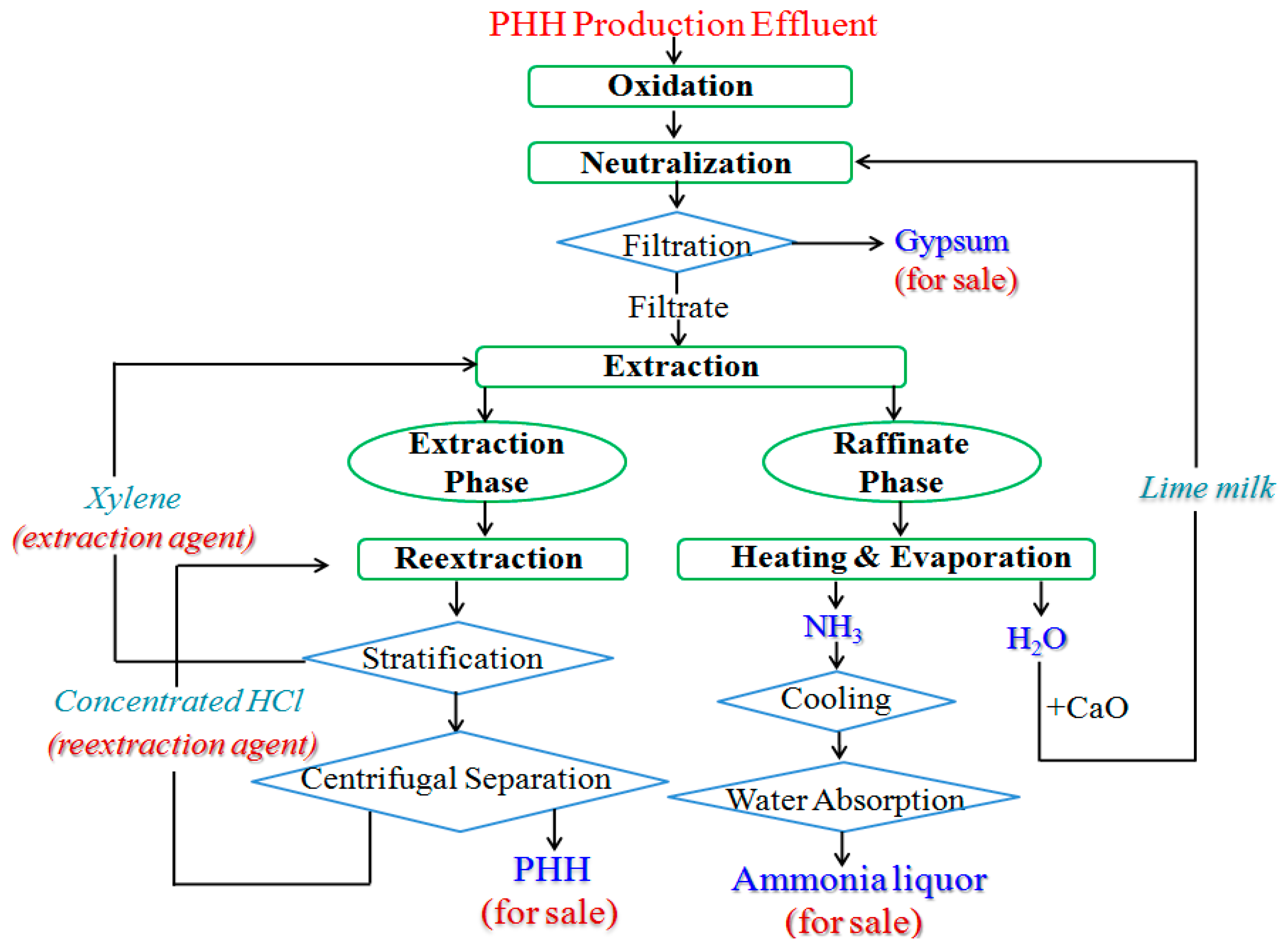
2. Materials and Methods
2.1. Sulfite Oxidation
2.2. Lime Milk Neutralization
2.3. Xylene Extraction and HCl Re-Extraction
2.4. Ammonia Evaporation
3. Results and Discussion
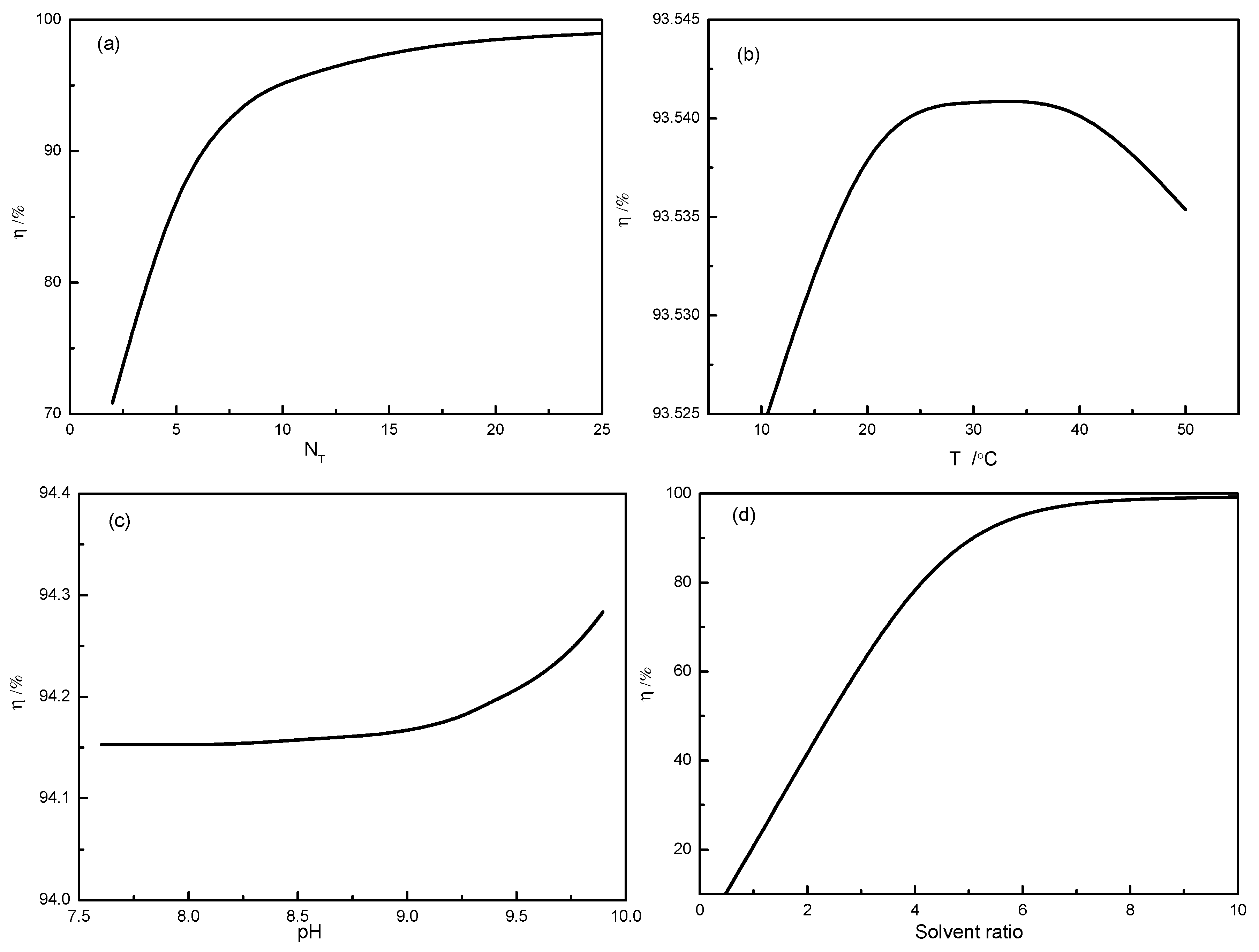
3.1. Lab-Scale Test
3.2. Pilot-Scale Test
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Larsson, D.G.J.; De Pedro, C.; Paxeus, N. Effluent from drug manufactures contains extremely high levels of pharmaceuticals. J. Hazard. Mater. 2007, 148, 751–755. [Google Scholar] [CrossRef] [PubMed]
- Oller, I.; Malato, S.; Sánchez-Pérez, J.A. Combination of advanced oxidation processes and biological treatments for wastewater decontamination—A review. Sci. Total Environ. 2011, 409, 4141–4166. [Google Scholar] [CrossRef] [PubMed]
- Gadipelly, C.; Pérez-González, A.; Yadav, G.D.; Ortiz, I.; Ibáñez, R.; Rathod, V.K.; Marathe, K.V. Pharmaceutical industry wastewater: Review of the technologies for water treatment and reuse. Ind. Eng. Chem. Res. 2014, 53, 11571–11592. [Google Scholar] [CrossRef]
- Wu, P.; Jiang, L.Y.; He, Z.; Song, Y. Treatment of metallurgical industry wastewater for organic contaminant removal in China: Status, challenges, and perspectives. Environ. Sci. Water Res. Technol. 2015, 3, 1015–1031. [Google Scholar] [CrossRef]
- Hung, Y.-T.; Aziz, H.A.; Ramli, S.F.; Yeh, R.Y.-L.; Liu, L.-H.; Huhnke, C.R. Chemical waste and allied products. Water Environ. Res. 2016, 88, 1374–1394. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Maken, S.; Jang, J.H.; Park, K.; Park, J.W. Development of physicochemical nitrogen removal process for high strength industrial wastewater. Water Res. 2006, 40, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Aloui, F.; Kchaou, S.; Sayadi, S. Physicochemical treatments of anionic surfactants wastewater: Effect on aerobic biodegradability. J. Hazard. Mater. 2009, 164, 353–359. [Google Scholar] [CrossRef]
- Shi, S.; Wang, C.; Fang, S.; Jia, M.; Li, X. Removal performance and water quality analysis of paper machine white water in a full-scale wastewater treatment plant. Environ. Technol. 2017, 38, 1443–1451. [Google Scholar] [CrossRef]
- Kim, V.K.; Ihm, S. Heterogeneous catalytic wet air oxidation of refractory organic pollutants in industrial wastewaters: a review. J. Hazard. Mater. 2011, 186, 16–34. [Google Scholar] [CrossRef]
- Kumari, M.; Saroha, A.K. Performance of various catalysts on treatment of refractory pollutants in industrial wastewater by catalytic wet air oxidation: A review. J. Environ. Manag. 2018, 228, 169–188. [Google Scholar]
- Comninellis, C.; Kapalka, A.; Malato, S.; Parsons, S.A.; Poulios, L.; Mantzavinos, D. Advanced oxidation processes for water treatment: Advances and trends for R&D. J. Chem. Technol. Biotechnol. 2008, 83, 769–776. [Google Scholar]
- Balcioglu, I.A.; Alaton, I.A.; Otker, M.; Bahar, R.; Bakar, N.; Ikiz, M. Application of advanced oxidation processes to different industrial wastewaters. J. Environ. Sci. Health. Part Atoxic/Hazard. Subst. Environ. Eng. 2003, 38, 1587–1596. [Google Scholar] [CrossRef]
- Ghatak, H.R. Advanced xxidation processes for the treatment of biorecalcitrant organics in wastewater. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1167–1219. [Google Scholar] [CrossRef]
- Krysa, J.; Mantzavinos, D.; Pichat, P.; Poulios, I. Advanced oxidation processes for water/wastewater treatment. Environ. Sci. Pollut. Res. 2018, 25, 34799–34800. [Google Scholar] [CrossRef]
- Gupta, V.K.; Ali, I.; Saleh, T.A.; Nayak, A.; Agarwal, S. Chemical treatment technologies for waste-water recycling—An overview. RSC Adv. 2012, 2, 6380–6388. [Google Scholar] [CrossRef]
- Rojas, M.R.; Leung, C.; Bonk, F.; Zhu, Y.; Edwards, L.; Arnold, R.G.; Sáez, A.E.; Klečka, G. Assessment of the effectiveness of secondary wastewater treatment technologies to remove trace chemicals of emerging concern. Crit. Rev. Environ. Sci. Technol. 2013, 43, 1281–1314. [Google Scholar] [CrossRef]
- Li, X.; Chen, S.; Angelidaki, I.; Zhang, Y. Bio-electro-Fenton processes for wastewater treatment: Advances and prospects. Chem. Eng. J. 2018, 354, 492–506. [Google Scholar] [CrossRef]
- Zhong, C.; Ye, W.; Cui, Y. Improvement on technology for synthesizing triazophos. Mod. Chem. Ind. 1997, 17, 19–20. [Google Scholar]
- Banerjee, M.; Ray, A.K. The role of thyroid hormone on phenylhydrazine hydrochloride mediated inhibitory effects on blood acetylcholinesterase: An in vivo and in vitro study. J. Biochem. Mol. Toxicol. 2002, 16, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Ramyashree, D.; Raghavendra, K.R.; Kumar, A.D.; Vagish, C.B.; Kumar, K.A. Synthesis, characterization and antimicrobial activities of chalcones and their post transformation to pyrazole derivatives. Asian J. Chem. 2017, 29, 1538–1542. [Google Scholar] [CrossRef]
- Zheng, J.; Huang, F.; Li, Y.; Xu, T.; Xu, H.; Jia, J.; Ye, Q.; Gao, J. The aggregation-induced emission enhancement properties of BF2 complex isatin-phenylhydrazone: Synthesis and fluorescence characteristics. Dyes Pigments 2015, 113, 502–509. [Google Scholar] [CrossRef]
- ChemicalBook, CAS Database List, Phenylhydrazine Hydrochloride. Available online: https://www.chemicalbook.com/ChemicalProductProperty_EN_CB0101763.htm (accessed on 28 February 2019).
- Heinz, H. Process for the Production of Phenylhydrazine Hydrochloride. U.S. Patent 3203989, 31 August 1965. [Google Scholar]
- Zhang, R.; Wu, Y.-Z.; Zheng, D.-M. The synthesis of phenylhydrazine hydrochloride. J. Nanchang Univ. (Nat. Sci.) 2002, 26, 394–396. [Google Scholar]
- Liu, X.; Zhou, D.; Zhang, H. Research on the treatment and comprehensive utilization of phenylhydrazine hydrochloride effluent. Water 2018, 10, 438. [Google Scholar]
- Brillas, E.; Sirés, I.; Oturan, M.A. Electro-Fenton process and related electrochemical technologies based on Fenton’s reaction chemistry. Chem. Rev. 2009, 109, 6570–6631. [Google Scholar] [CrossRef]
- Zazo, J.A.; Casas, J.A.; Mohedano, A.F.; Gilarranz, M.A.; Rodriaguez, J.J. Chemical pathway and kinetics of phenol oxidation by Fenton’s reagent. Environ. Sci. Technol. 2005, 39, 9295–9302. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.-C.; Wei, D.; Bao, X.-Z. Research on treatment of phenylhydrazine hydrochloride wastewater. Technol. Dev. Chem. Ind. 2013, 42, 39–44. [Google Scholar]
- Amaral-Silva, N.; Martins, R.C.; Nunes, P.; Castro-Silvac, S.; Quinta-Ferreira, R.M. From a lab test to industrial application: scale-up of Fenton process for real olive mill wastewater treatment. J. Chem. Technol. Biotechnol. 2017, 92, 1336–1344. [Google Scholar] [CrossRef]
- Mu, J.; Li, H. Determination of distillation-titration method for the ammonia nitrogen in waste water. J. Henan Univ. (Nat. Sci.) 2006, 36, 54–57. [Google Scholar]
- Zhao, X.; Shao, Y.; Lu, Z.; Yao, J.; Cao, H.; Fan, J. Determination of phenylhydrazine hydrochloride in industrial wastewater by HPLC. Ind. Water Treat. 2012, 32, 80–82. [Google Scholar]

| Component | PHH | NaCl | HCl | NH4Cl | NH4HSO4 | NH4HSO3 | H2O | Other Organics |
|---|---|---|---|---|---|---|---|---|
| Wt % | 1.15 | 4.20 | 0.90 | 9.10 | 16.80 | 1.70 | 66.00 | 0.15 |
| pH | Vxylene | T | mPHH | η |
|---|---|---|---|---|
| /mL | /°C | /g | /% | |
| 7.30 | 100.0 | 35.0 | 0.5244 | 45.6 |
| 8.02 | 100.0 | 35.0 | 0.5888 | 51.2 |
| 8.34 | 100.0 | 35.0 | 0.6820 | 59.3 |
| 8.58 | 100.0 | 35.0 | 0.7383 | 64.2 |
| 9.00 | 100.0 | 35.0 | 0.7495 | 65.2 |
| 9.30 | 100.0 | 35.0 | 0.7544 | 65.6 |
| 9.56 | 100.0 | 35.0 | 0.7552 | 65.7 |
| 10.00 | 100.0 | 35.0 | 0.7561 | 65.8 |
| 9.28 | 100.0 | 20.0 | 0.6130 | 53.3 |
| 9.30 | 100.0 | 25.0 | 0.7303 | 63.5 |
| 9.30 | 100.0 | 30.0 | 0.7441 | 64.7 |
| 9.32 | 100.0 | 35.0 | 0.7544 | 65.6 |
| 9.26 | 100.0 | 40.0 | 0.7395 | 64.3 |
| 9.31 | 100.0 | 45.0 | 0.7153 | 62.2 |
| 9.30 | 100.0 | 50.0 | 0.6774 | 58.9 |
| 9.29 | 100.0 | 55.0 | 0.6509 | 56.6 |
| 9.30 | 20.0 | 35.0 | 0.3473 | 30.2 |
| 9.32 | 40.0 | 35.0 | 0.5256 | 45.7 |
| 9.31 | 60.0 | 35.0 | 0.6268 | 54.5 |
| 9.30 | 100.0 | 35.0 | 0.7740 | 67.3 |
| 9.30 | 200.0 | 35.0 | 0.9453 | 82.2 |
| 9.32 | 300.0 | 35.0 | 0.9948 | 86.5 |
| 9.30 | 400.0 | 35.0 | 1.0316 | 89.7 |
| 9.30 | 500.0 | 35.0 | 1.0730 | 93.3 |
| Material Name | Mass/kg |
|---|---|
| Aniline | 300 |
| NaNO2 | 232 |
| 30% HCl (diazotization reaction) | 835 |
| 56% NH4HSO3 | 1400 |
| 22% NH3·H2O | 210 |
| 30% HCl (acid precipitation analysis) | 870 |
| PHH | 370 |
| Material Name | Mass/kg |
|---|---|
| Total wastewater | 3500 |
| CaO (neutralization reaction) | 520 |
| Dissolved PHH | 42 |
| Recycled PHH | 39 |
| Recycled ammonia | 190 |
| Gypsum | 960 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, D.; Liu, X.; Chen, C. Harmless Treatment of Phenylhydrazine Hydrochloride Production Effluent: From Lab Scale to Pilot Scale. Water 2019, 11, 608. https://doi.org/10.3390/w11030608
Zhou D, Liu X, Chen C. Harmless Treatment of Phenylhydrazine Hydrochloride Production Effluent: From Lab Scale to Pilot Scale. Water. 2019; 11(3):608. https://doi.org/10.3390/w11030608
Chicago/Turabian StyleZhou, Di, Xide Liu, and Chuangqi Chen. 2019. "Harmless Treatment of Phenylhydrazine Hydrochloride Production Effluent: From Lab Scale to Pilot Scale" Water 11, no. 3: 608. https://doi.org/10.3390/w11030608
APA StyleZhou, D., Liu, X., & Chen, C. (2019). Harmless Treatment of Phenylhydrazine Hydrochloride Production Effluent: From Lab Scale to Pilot Scale. Water, 11(3), 608. https://doi.org/10.3390/w11030608





