Wastewater Treatment by Constructed Wetland Eco-Technology: Influence of Mineral and Plastic Materials as Filter Media and Tropical Ornamental Plants
Abstract
:1. Introduction
2. Materials and Methods
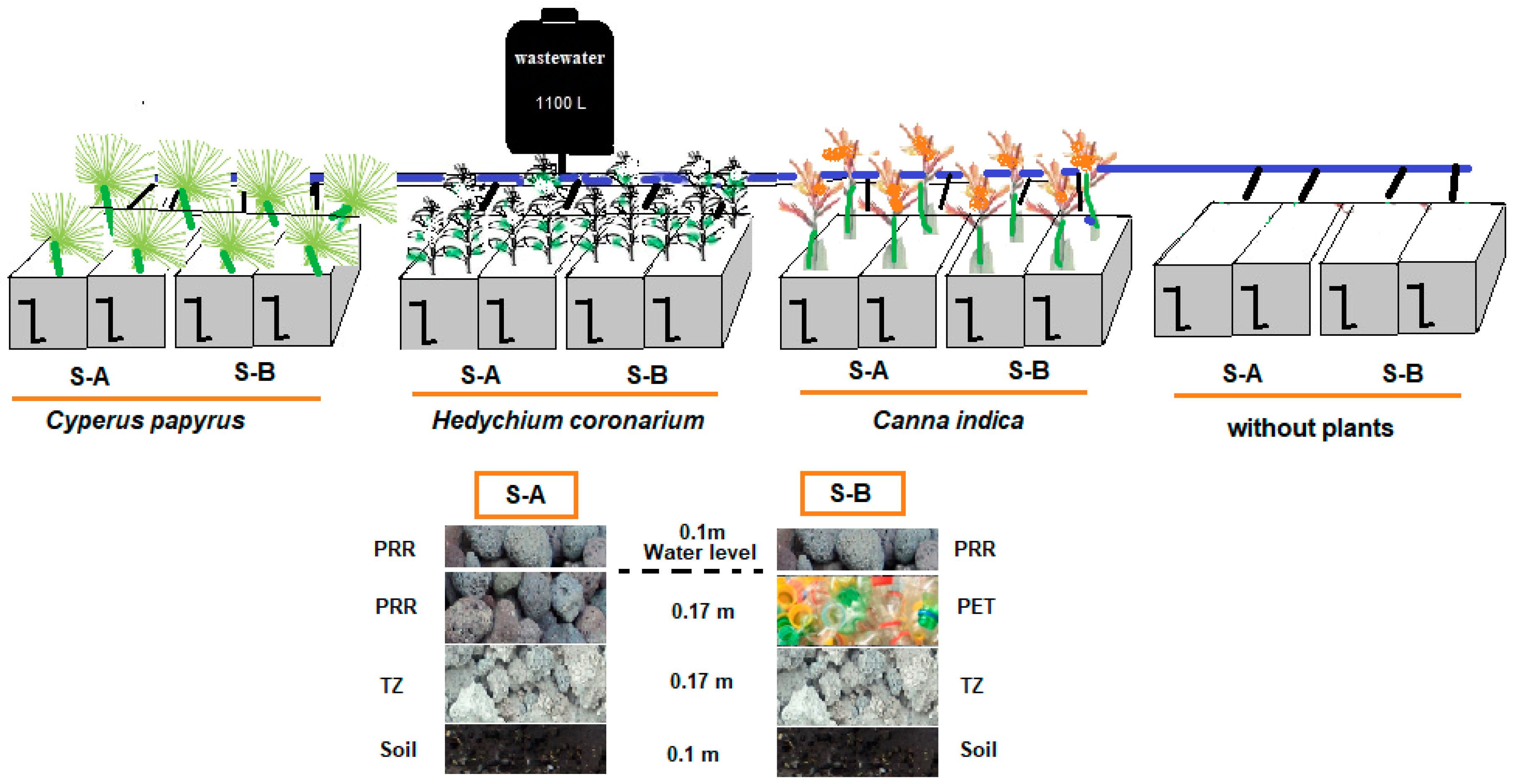
Design Features and Operating Criteria of Microcosms
3. Results
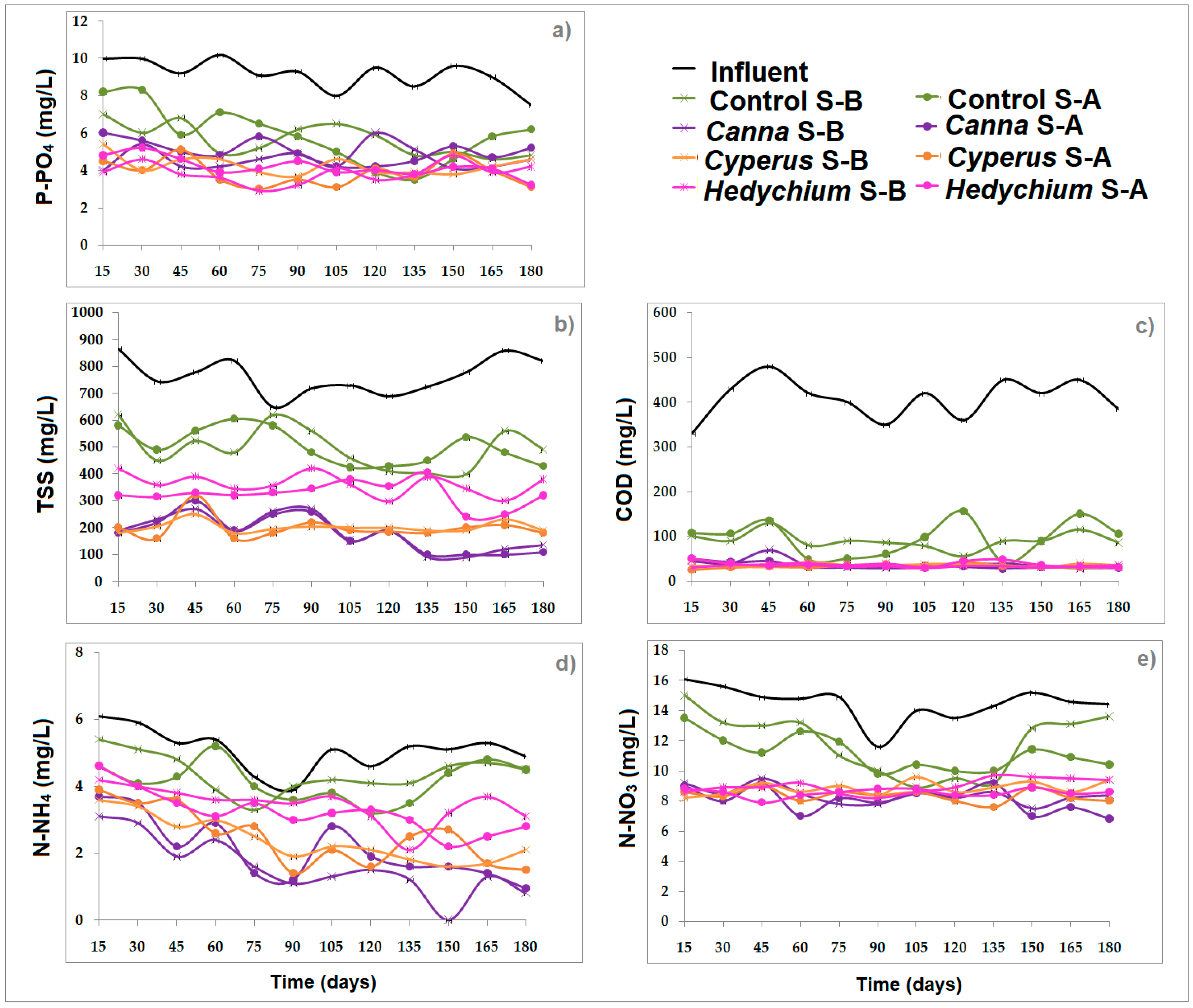
Concentration and Removal of Pollutants in CW Microcosms
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vymazal, J. Plants used in constructed wetlands with horizontal subsurface flow: A review. Hydrobiologia 2011, 674, 133–156. [Google Scholar] [CrossRef]
- Vidal, G.; Hormazábal, S. Constructed Wetland Operation and Design, 1st ed.; Universidad de Concepción: Concepción, Chile, 2018; ISBN 978-956-227-419-7. (In Spanish) [Google Scholar]
- Mitsch, W.J.; Gosselink, J. Wetlands; John Wiley and Sons Inc.: New York, NY, USA, 2015; Volume 5, p. 456. [Google Scholar]
- Marín-Muñiz, J.L. Humedales, Riñones del Planeta y Hábitat de Múltiples Especies; Editora de Gobierno del Estado de Veracruz: Xalapa, Veracruz, Mexico, 2018; (In Spanish). Available online: https://www.sev.gob.mx/servicios/publicaciones/serie_fueraseries/Humedales_Impresion.pdf (accessed on 5 November 2019).
- Garfí, M.; Flores, L.; Ferrer, I. Life cycle assessment of wastewater treatment system for small communities: Activated sludge, constructed wetlands and high rate algal ponds. J. Clean. Prod. 2017, 161, 211–219. [Google Scholar] [CrossRef]
- Molinos-Senante, M.; Gómez, T.; Garrido-Baserba, M.; Caballero, R.; Sala-Garrido, R. Assessing the sustainability of small wastewater treatment systems: A composite indicator approach. Sci. Total Environ. 2014, 497, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Marín-Muñiz, J.L. Humedales construidos para el tratamiento de aguas residuales, producción de plantas ornamentales y reuso del agua. Agroproductividad 2017, 10, 90–95. (In Spanish). Available online: http://revista-agroproductividad.org/index.php/agroproductividad/article/view/1028 (accessed on 5 November 2019).
- Yang, Y.; Zhao, Y.; Liu, R.; Morgan, D. Global development of various emerged substrates utilized in constructed wetlands. Bioresour. Technol. 2018, 261, 441–452. [Google Scholar] [CrossRef]
- Wang, R.; Korboulewsky, N.; Prudent, P.; Domeizel, M.; Rolando, C.; Bonin, G. Feasibility of using an organic substrate in a wetland system treating sewage sludge: Impact of plant species. Bioresour. Technol. 2010, 101, 51–57. [Google Scholar] [CrossRef]
- Zhu, W.L.; Cui, L.H.; Ouyang, Y.; Long, C.F.; Tang, X.D. Kinetic adsorption of ammonium nitrogen by substrate materials for constructed wetlands. Pedosphere 2011, 21, 454–463. [Google Scholar] [CrossRef]
- Marín-Muñiz, J.L.; García-González, M.C.; Ruelas-Monjardín, L.C.; Moreno-Casasola, P. Influence of different porous media and ornamental vegetation on wastewater pollutant removal in vertical subsurface flow wetland microcosms. Environ. Eng. Sci. 2018, 35, 88–94. [Google Scholar] [CrossRef]
- Marín-Muñiz, J.L. Removal of wastewater pollutant in artificial wetlands implemented in Actopan, Veracruz, Mexico. Rev. Mex. Ing. Química 2016, 15, 553–563. Available online: http://www.redalyc.org/articulo.oa?id=62046829021 (accessed on 30 July 2019).
- Dallas, S.; Ho, G. Subsurface flow reedbeds using alternative media for the treatment of domestic greywater in Monteverde, Costa Rica, Central America. Water Sci. Technol. 2005, 51, 119–128. Available online: https://www.ncbi.nlm.nih.gov/pubmed/16104413 (accessed on 5 November 2019). [CrossRef]
- Shiwei, C.; Zhaoqian, J.; Peng, Y.; Yue, W.; Yin, W. Performance of constructed wetlands with different substrate for the treated effluent form municipal sewage plants. J. Water Reuse Desalin. 2019. (In press) [Google Scholar] [CrossRef]
- Zamora, S.; Sandoval, L.; Marín-Muñiz, J.L.; Fernández-Lambert, G.; Hernánez-Orduña, G. Impact of ornamental vegetation type and different substrate layers on pollutant removal in constructed wetland mesocosms treating rural community wastewater. Processes 2019, 11, 531. [Google Scholar] [CrossRef]
- Ding, Y.; Luy, T.; Bail, S.; Li, Z.; Ding, H.; You, S.; Xie, Q. Effect of multilayer substrate configuration in horizontal subsurface flow constructed wetlands: Assessment of treatment performance, biofilm development, and solids accumulation. Environ. Sci. Pollut. Res. 2018, 25, 1883–1891. [Google Scholar] [CrossRef] [PubMed]
- Bail, S.; Lyu, T.; Ding, Y.; Li, Z.; Wang, D.; You, S.; Xie, Q. Campus sewage treatment in multilayer horizontal subsurface flow constructed wetlands: Nitrogen removal and microbial community distribution. Clean Soil Air Water 2017, 45, 1700254. [Google Scholar] [CrossRef]
- APHA-AWWA-WFF. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Liu, X.; Huang, S.; Tang, T.; Liu, X.; Scholz, M. Growth characteristic and nutrient removal capability of plants in subsurface vertical flow constructed wetlands. Ecol. Eng. 2012, 44, 189–198. [Google Scholar] [CrossRef]
- Vymazal, J. Removal of nutrients in various types of constructed wetlands. Sci. Total Environ. 2007, 380, 48–65. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Reddy, K.R. Temperature effects in treatment wetlands. Water Environ. Res. 2001, 73, 543–557. [Google Scholar] [CrossRef]
- Sandoval, L.; Zamora-Castro, S.A.; Vidal-Álvarez, M.; Marín-Muñiz, J.L. Role of Wetland Plants and Use of Ornamental Flowering Plants in Constructed Wetlands for Wastewater Treatment: A Review. Appl. Sci. 2019, 9, 685. [Google Scholar] [CrossRef]
- Cárdenas-López, D.; Baptiste, M.P.; Castaño, N. Familia Zingiberaceae Hedychium coronarium. Plantas Exóticas con Alto Potencial de Invasión en Colombia. 2010. (In Spanish). Available online: http://hdl.handle.net/20.500.11761/32935 (accessed on 25 February 2019).
- Cordeiro, C.A.M.M.; Costa, T.M. Evaluation of solid residues removed from a mangrove swamp in the São Vicente Estuary, SP, Brazil. Mar. Pollut. Bull. 2010, 60, 1762–1767. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, Y.; Xie, H.; Yang, Z. Constructed wetlands: A review on the role of radial oxygen loss in the rhizosphere by macrophytes. Water 2018, 10, 678. [Google Scholar] [CrossRef]
- Tian, C.; Wang, C.; Tian, Y.; Wu, X.; Xiao, B. Root oxygen loss and the effects of rhizosphere micro area of two submerged plants. Pol. J. Environ. Stud. 2015, 24, 1795–1802. [Google Scholar] [CrossRef]
- CONAGUA. Numeragua. Comisión Nacional del Agua. México. 2018. (In Spanish). Available online: http://sina.conagua.gob.mx/publicaciones/Numeragua_2018.pdf (accessed on 7 August 2019).
- Russell, D.L. Practical Wastewater Treatment; John Wiley& Sons: Hoboken, NJ, USA, 2019; Available online: https://books.google.com.mx/books?hl=es&lr=&id=F6mMDwAAQBAJ&oi=fnd&pg=PR17&dq=+in+Mexico,+monitors+water+bodies+have+established+that+an+acceptable+criterion+COD+discharge+biologically+treated+wastewater+must+not+exceed+40+mg/L+&ots=fGvreeyPSF&sig=t74EfHW3bcT7GLhmGNhjA6_BjHs#v=onepage&q&f=false (accessed on 15 June 2019).
- Abbasi, H.; Xie, J.; Hussain, S.; Lu, X. Nutrient removal in hybrid constructed wetlands: Spatial-seasonal variation and the effect of vegetation. Water Sci. Technol. 2019, 79, 1985–1994. [Google Scholar] [CrossRef] [PubMed]
- Zurita, F.; White, J.R. Comparative study of three two-stage hybrid ecological wastewater treatment systems for producing high nutrient reclaimed water for irrigation reuse in developing countries. Water 2014, 6, 213–228. [Google Scholar] [CrossRef]
- Martínez, N.B.; Tejeda, A.; Del Toro, A.; Sánchez, M.P.; Zurita, F. Nitrogen removal in pilot-scale partially saturated vertical wetlands with and without and internal source of carbon. Sci. Total Environ. 2018, 645, 524–532. [Google Scholar] [CrossRef]
- USEPA. United States Environmental Protection Agency, Superfund Public Health Evaluation Manual; EPA/5401/1-86/060; USEPA: Washington, DC, USA, 1986.
- Wang, G.; Gao, J.; Yang, R.; Zhang, J.; Guo, H.; Gao, J. Preparation of sustainable non-combustion filler substrate form waterworks sludge/aluminum slag/gypsum/silica/malfan stone for phosphorous immobilization in constructed wetlands. Water Sci. Technol. 2019, 80, 153–163. [Google Scholar] [CrossRef]
- Kizito, S.; Luo, H.; Wu, S.; Ajmal, Z.; Lv, T.; Dong, R. Phosphate recovery form liquid fraction of anaerobic digestate using four slow pyrolyzed biochars: Dynamics of adsorption, desorption and regeneration. J. Environ. Manag. 2017, 201, 260–267. [Google Scholar] [CrossRef]
- Hernández, M.E.; Galindo-Zetina, M.; Hernández-Hernández, J.C. Greenhouse gas emissions and pollutant removal in treatment wetlands with ornamental plants under subtropical conditions. Ecol. Eng. 2018, 114, 88–95. [Google Scholar] [CrossRef]
- Liang, M.; Zhang, C.; Peng, C.; Lai, Z.; Chen, D.; Chen, Z. Plant growth, community structure, and nutrient removal in monoculture and mixed constructed wetlands. Ecol. Eng. 2011, 37, 309–316. [Google Scholar] [CrossRef]
- Karathanasis, A.D.; Potter, C.L.; Coyne, M.S. Vegetation effects on fecal bacteria, BOD, and suspended solid removal in constructed wetlands treating domestic wastewater. Ecol. Eng. 2003, 20, 157–169. [Google Scholar] [CrossRef]
- Zhang, L.; Lyu, T.; Zhang, Y.; Button, M.; Arias, C.; Weber, K.; Brix, H.; Carvalho, P. Impacts of design configuration and plants on the functionality of the microbial community of mesocosm-scale constructed wetlands treating ibuprofen. Water Res. 2018, 131, 228–238. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Gao, T.; Kang, S.; Sillanpää. Importance of atmospheric transport for microplastics deposited in remote areas. Environ. Pollut. 2019, 254, 112953. [Google Scholar] [CrossRef]
| Parameter/Microcosms | Influent | C. papyrus | C. indica | H. coronarium | Control | ||||
|---|---|---|---|---|---|---|---|---|---|
| S-A | S-B | S-A | S-B | S-A | S-B | S-A | S-B | ||
| pH (pH units) | 7.6 ± 0.3 | 7.5 ± 0.9 | 7.4 ± 0.6 | 7.3 ± 0.6 | 7.5 ± 0.2 | 7.1 ± 0.9 | 7.0 ± 0.7 | 7.4 ± 0.2 | 7.4 ± 0.1 |
| DO (mg L−1) | 2.2 ± 0.3 | 5.1 ± 0.9 | 4.9 ± 0.8 | 4.8 ± 0.7 | 5.2 ± 0.7 | 4.7 ± 1.1 | 4.5 ± 0.5 | 2.6 ± 0.5 | 2.5 ± 0.4 |
| Temperature (°C) | 18.0 ± 0.9 | 18.6 ± 0.2 | 18.1 ± 1.3 | 18.5 ± 0.6 | 18.6 ± 0.8 | 17.8 ± 1.6 | 18.2 ± 0.7 | 20.0 ± 0.6 | 19.9 ± 0.7 |
| Ornamental Plant | Material Layer | Maximum Root Length (cm) | Maximum Root Volume (cm3) | Maximum Plant Height (cm) | Number of Flower Production | Number of Shoots | Plant Health |
|---|---|---|---|---|---|---|---|
| C. indica | S-A | 42.3 ± 11.6 a | 424 ± 29.6 a | 59.2 ± 16.2 a | 8 a | 5 a | Without wilting or pests |
| S-B | 40.6 ± 12.0 a | 408 ± 19.2 a | 61.8 ± 15.9 a | 7 a | 5 a | Without wilting or pests | |
| H. coronarium | S-A | 14.4 ± 2.6c | 60 ± 11.0 c | 40.8 ± 3.2 b | 0 c | 1 c | Without wilting or pests |
| S-B | 19.0 ± 6.9 c | 56 ± 11.1 c | 43.0 ± 6.3 b | 0 c | 0 c | Without wilting or pests | |
| C. papyrus | S-A | 28.6 ± 9.4 b | 106 ± 28.2 b | 30.3 ± 6.7 b | 4 b | 4 b | Without wilting or pests |
| S-B | 30.2 ± 9.1 b | 99 ± 16.4 b | 30.0 ± 5.7 b | 4 b | 4 b | Without wilting or pests |
| Parameter | Wetland Vegetation in Different Substrates | |||||||
|---|---|---|---|---|---|---|---|---|
| Canna indica | Cyperus papyrus | Hedychium coronarium | Control | |||||
| S-A | S-B | S-A | S-B | S-A | S-B | S-A | S-B | |
| COD | ||||||||
| EC | 408 ± 49 | |||||||
| CS | 35 ± 13 | 37 ± 11 | 33 ± 14 | 36 ± 14 | 37 ± 9.6 | 35 ± 14 | 96 ± 11 | 91 ± 7.4 |
| Removal (%) | 91.4 ± 16 a | 90.9 ± 09 a | 91.9 ± 11 a | 91.2 ± 13 a | 90.9 ± 14.3 a | 91.4 ± 10.2 a | 76.5 ± 14.4 b | 77.7 ± 11.2 b |
| TSS | ||||||||
| EC | 766 ± 85 | |||||||
| CS | 179 ± 18 | 182 ± 28 | 198 ± 32 | 201 ± 25 | 326 ± 56 | 364 ± 49 | 504 ± 61 | 498 ± 69 |
| Removal (%) | 76.7 ± 12 a | 76.2 ± 17 a | 74.2 ± 11.2 a | 73.8 ± 29 a | 57.4 ± 11 b | 52.5 ± 16 b | 34.2 ± 12 c | 35.1 ± 08 c |
| N-NO3 | ||||||||
| EC | 14.4 ± 1.6 | |||||||
| CS | 8.1 ± 1.2 | 8.5 ± 1.4 | 8.4 ± 0.72 | 8.9 ± 0.22 | 8.6 ± 0.95 | 9.0 ± 0.98 | 11.1 ± 0.13 | 11.9 ± 1.0 |
| Removal (%) | 44 ± 6.8 a | 41.0 ± 4.2 a | 41.7 ± 8.8 a | 38.2 ± 9.6 a | 40.3 ± 11.6 a | 37.5 ± 5.0 a | 20.8 ± 7.6 b | 17.4 ± 4.3 b |
| P-PO4 | ||||||||
| EC | 9.11 ± 1.4 | |||||||
| CS | 5.0 ± 0.6 | 4.6 ± 9.2 | 3.9 ± 0.2 | 4.2 ± 0.7 | 4.1 ± 0.6 | 3.9 ± 0.16 | 5.9 ± 1.0 | 5.6 ± 0.8 |
| Removal (%) | 45.1 ± 6.1 a | 49.5 ± 4.9 a | 57.2 ± 9.9 a | 53.9 ± 8.9 a | 55.0 ± 3.1 a | 57.2 ± 2.6 a | 35.2 ± 0.8 b | 38.5 ± 2.1 b |
| N-NH4 | ||||||||
| EC | 5.09 ± 1.0 | |||||||
| CS | 1.9 ± 0.7 | 1.6 ± 0.8 | 2.5 ± 0.5 | 2.3 ± 0.4 | 3.1 ± 0.14 | 3.5 ± 0.9 | 4.2 ± 1.2 | 4.6 ± 0.9 |
| Removal (%) | 62.7 ± 4.2 a | 68.6 ± 0.8 a | 50.9 ± 5.2 b | 54.8 ± 3.6 b | 39.1 ± 4.6 c | 31.2 ± 8.1 c | 17.5 ± 1.6 d | 20.2 ± 1.2 d |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zamora, S.; Marín-Muñíz, J.L.; Nakase-Rodríguez, C.; Fernández-Lambert, G.; Sandoval, L. Wastewater Treatment by Constructed Wetland Eco-Technology: Influence of Mineral and Plastic Materials as Filter Media and Tropical Ornamental Plants. Water 2019, 11, 2344. https://doi.org/10.3390/w11112344
Zamora S, Marín-Muñíz JL, Nakase-Rodríguez C, Fernández-Lambert G, Sandoval L. Wastewater Treatment by Constructed Wetland Eco-Technology: Influence of Mineral and Plastic Materials as Filter Media and Tropical Ornamental Plants. Water. 2019; 11(11):2344. https://doi.org/10.3390/w11112344
Chicago/Turabian StyleZamora, Sergio, J. Luis Marín-Muñíz, Carlos Nakase-Rodríguez, Gregorio Fernández-Lambert, and Luis Sandoval. 2019. "Wastewater Treatment by Constructed Wetland Eco-Technology: Influence of Mineral and Plastic Materials as Filter Media and Tropical Ornamental Plants" Water 11, no. 11: 2344. https://doi.org/10.3390/w11112344
APA StyleZamora, S., Marín-Muñíz, J. L., Nakase-Rodríguez, C., Fernández-Lambert, G., & Sandoval, L. (2019). Wastewater Treatment by Constructed Wetland Eco-Technology: Influence of Mineral and Plastic Materials as Filter Media and Tropical Ornamental Plants. Water, 11(11), 2344. https://doi.org/10.3390/w11112344








