Removal of Emulsified Oil from Aqueous Environment by Using Polyvinylpyrrolidone-Coated Magnetic Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Fe3O4 MNPs
2.3. Characterization
2.4. Demulsification Test
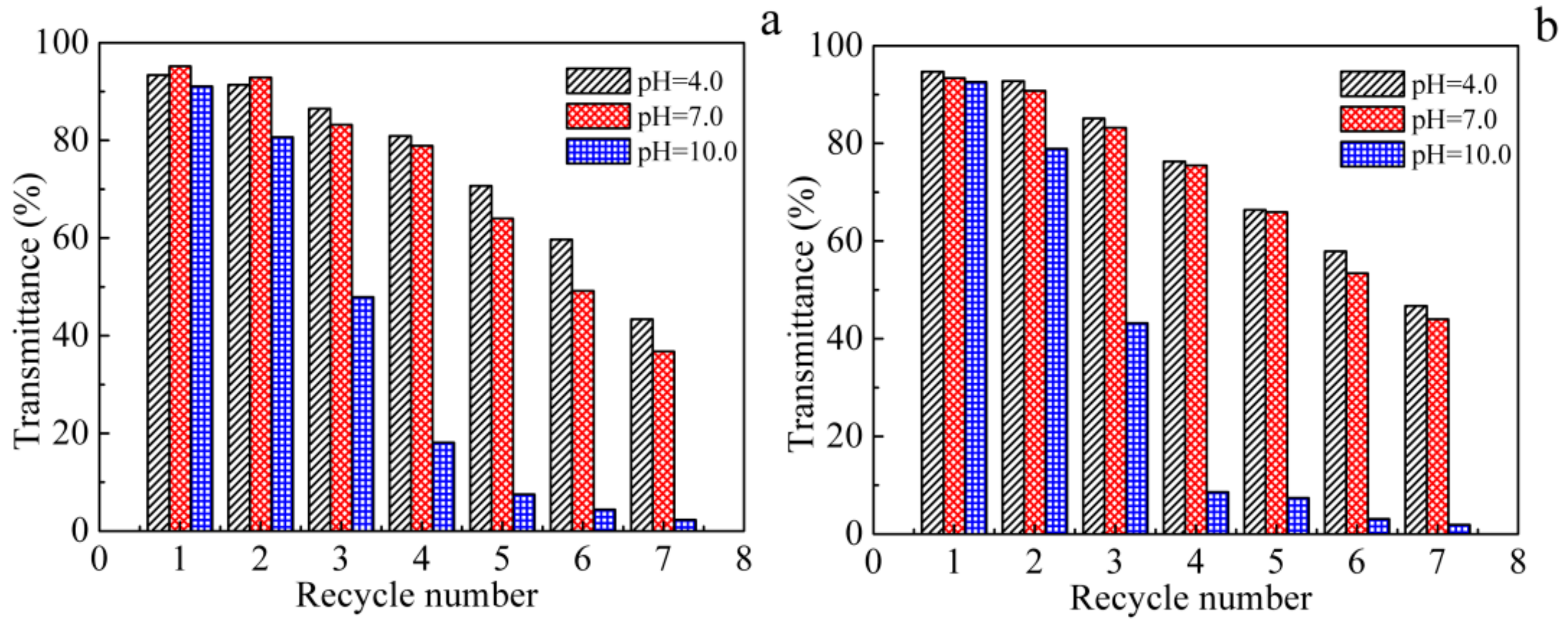
2.5. Recycle Tests
3. Results and Discussion
3.1. Characterizations
3.2. Demulsification Performance
3.3. Recyclability of MNPs
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bratskaya, S.; Avramenko, V.; Schwarz, S.; Philippov, A. Enhanced flocculation of oil-in-water emulsions by hydrophobically modified chitosan derivatives. Colloids Surf. A Physicochem. Eng. Asp. 2006, 275, 168–176. [Google Scholar] [CrossRef]
- Liang, J.; Du, N.; Song, S.; Hou, W. Magnetic demulsification of diluted crude oil-in-water nanoemulsions using oleic acid-coated magnetite nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2015, 466, 197–202. [Google Scholar] [CrossRef]
- Rajak, V.K.; Relish, K.K.; Kumar, S.; Mandal, A. Mechanism and kinetics of separation of oil from oil-in-water emulsion by air flotation. Petrol. Sci. Technol. 2016, 33, 1861–1868. [Google Scholar] [CrossRef]
- Lü, T.; Luo, C.; Qi, D.; Zhang, D.; Zhao, H. Efficient treatment of emulsified oily wastewater by using amphipathic chitosan-based flocculant. React. Funct. Polym. 2019, 139, 133–141. [Google Scholar] [CrossRef]
- Zhang, H.; Duan, M.; Fang, S.; Zhai, L.; Wang, X. Preparation of a novel flocculant and its performance for treating acidic oily wastewater. RSC Adv. 2016, 6, 106102–106108. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, M.; Wang, Z. Underwater superoleophobic membrane with enhanced oil-water separation, antimicrobial, and antifouling activities. Adv. Mater. Interfaces 2016, 3, 1500664. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, G.; Huang, S. In situ generated janus fabrics for the rapid and efficient separation of oil from oil-in-water emulsions. Angew. Chem. Int. Ed. 2016, 55, 14610–14613. [Google Scholar] [CrossRef]
- Lv, W.; Mei, Q.; Xiao, J.; Du, M.; Zheng, Q. 3D multiscale superhydrophilic sponges with delicately designed pore size for ultrafast oil/water separation. Adv. Funct. Mater. 2017, 27, 1704293. [Google Scholar] [CrossRef]
- Xu, H.; Jia, W.; Ren, S.; Wang, J. Novel and recyclable demulsifier of expanded perlite grafted by magnetic nanoparticles for oil separation from emulsified oil wastewaters. Chem. Eng. J. 2018, 337, 10–18. [Google Scholar] [CrossRef]
- Mirshahghassemi, S.; Ebner, A.D.; Cai, B.; Lead, J.R. Application of high gradient magnetic separation for oil remediation using polymer-coated magnetic nanoparticles. Sep. Purif. Technol. 2017, 179, 328–334. [Google Scholar] [CrossRef]
- Liang, J.; Li, H.; Yan, J.; Hou, W. Demulsification of oleic-acid-coated magnetite nanoparticles for cyclohexane-in-water nanoemulsions. Energy Fuels 2014, 28, 6172–6178. [Google Scholar] [CrossRef]
- Lü, T.; Qi, D.; Zhang, D.; Lü, Y.; Zhao, H. A facile method for emulsified oil-water separation by using polyethylenimine-coated magnetic nanoparticles. J. Nanopart. Res. 2018, 20, 88. [Google Scholar] [CrossRef]
- Liang, C.; He, X.; Liu, Q.; Xu, Z. Adsorption-based synthesis of magnetically responsive and interfacially active composite nanoparticles for dewatering of water-in-diluted bitumen emulsions. Energy Fuels 2018, 32, 8078–8089. [Google Scholar] [CrossRef]
- Lü, T.; Zhang, S.; Qi, D.; Zhang, D.; Vancec, G.F.; Zhao, H. Synthesis of pH-sensitive and recyclable magnetic nanoparticles for efficient separation of emulsified oil from aqueous environments. Appl. Surf. Sci. 2017, 396, 1604–1612. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Y.; Bao, M.; Yang, X.; Wang, Z. Facile fabrication of cyclodextrin-modified magnetic particles for effective demulsification from various types of emulsions. Environ. Sci. Technol. 2016, 50, 8809–8816. [Google Scholar] [CrossRef]
- Lü, T.; Zhang, S.; Qi, D.; Zhang, D.; Zhao, H. Thermosensitive poly(N-isopropylacrylamide)-grafted magnetic nanoparticles for efficient treatment of emulsified oily wastewater. J. Alloys Compd. 2016, 688, 513–520. [Google Scholar] [CrossRef]
- Chen, Y.; Bai, Y.; Chen, S.; Ju, J.; Li, Y.; Wang, T.; Wang, Q. Stimuli-responsive composite particles as solid-stabilizers for effective oil harvesting. ACS Appl. Mater. Interfaces 2014, 6, 13334–13338. [Google Scholar] [CrossRef]
- Wang, X.; Shi, Y.; Graff, R.W.; Lee, D.; Gao, H. Developing recyclable pH-responsive magnetic nanoparticles for oil-water separation. Polymer 2015, 72, 361–367. [Google Scholar] [CrossRef]
- Lü, T.; Zhang, S.; Qi, D.; Zhang, D.; Zhao, H. Enhanced demulsification from aqueous media by using magnetic chitosan-based flocculant. J. Colloid Interfaces Sci. 2018, 518, 76–83. [Google Scholar] [CrossRef]
- Lü, T.; Chen, Y.; Qi, D.; Cao, Z.; Zhang, D.; Zhao, H. Treatment of emulsified oil wastewaters by using chitosan grafted magnetic nanoparticles. J. Alloys Compd. 2017, 696, 1205–1212. [Google Scholar] [CrossRef]
- Zhang, S.; Lü, T.; Qi, D.; Cao, Z.; Zhang, D.; Zhao, H. Synthesis of quaternized chitosan-coated magnetic nanoparticles for oil-water separation. Mater. Lett. 2017, 191, 128–131. [Google Scholar] [CrossRef]
- Mirshahghassemi, S.; Lead, J.R. Oil recovery from water under environmentally relevant conditions using magnetic nanoparticles. Environ. Sci. Technol. 2015, 49, 11729–11736. [Google Scholar] [CrossRef]
- Mirshahghassemi, S.; Cai, B.; Lead, J.R. Evaluation of polymer-coated magnetic nanoparticles for oil separation under environmentally relevant conditions: Effect of ionic strength and natural organic macromolecules. Environ. Sci. Nano 2016, 3, 780–787. [Google Scholar] [CrossRef]
- Mirshahghassemi, S.; Lead, J.R. A facile and cost-effective method for separation of oil-water mixtures using polymer-coated iron oxide nanoparticles. Environ. Sci. Technol. 2014, 48, 14558–14563. [Google Scholar]
- Song, H.; Liu, M.; Li, S.; Chen, L.; Lin, C.; Zhang, L. Polyvinyl pyrrolidone-assisted solvothermal synthesis of Fe3O4 vesicular nanospheres. J. Nanosci. Nanotechnol. 2015, 15, 3998–4002. [Google Scholar] [CrossRef]
- Li, L.; Nicholas, R.J.; Chen, C.; Darton, R.C.; Baker, S.C. Comparative study of photoluminescence of single-walled carbon nanotubes wrapped with sodium dodecyl sulfate, surfactin and polyvinylpyrrolidone. Nanotechnology 2005, 16, S202–S205. [Google Scholar] [CrossRef]
- Shen, Q.; Wei, H.; Wang, L.; Zhou, Y.; Zhao, Y.; Zhang, Z.; Wang, D.; Xu, G.; Xu, D. Crystallization and aggregation behaviors of calcium carbonate in the presence of poly(vinylpyrrolidone) and sodium dodecyl sulfate. J. Phys. Chem. B 2005, 109, 18342–18347. [Google Scholar] [CrossRef]






| Sample Number | FeCl3∙6H2O (g) | NaAc (g) | PVP (g) | EG (mL) |
|---|---|---|---|---|
| S0 | 2.0 | 6.0 | 0 | 65 |
| S1 | 2.0 | 6.0 | 1.5 (10 k) | 65 |
| S2 | 2.0 | 6.0 | 3.0 (10 k) | 65 |
| S3 | 2.0 | 6.0 | 4.5 (10 k) | 65 |
| S4 | 2.0 | 6.0 | 1.5 (1300 k) | 65 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shao, S.; Li, Y.; Lü, T.; Qi, D.; Zhang, D.; Zhao, H. Removal of Emulsified Oil from Aqueous Environment by Using Polyvinylpyrrolidone-Coated Magnetic Nanoparticles. Water 2019, 11, 1993. https://doi.org/10.3390/w11101993
Shao S, Li Y, Lü T, Qi D, Zhang D, Zhao H. Removal of Emulsified Oil from Aqueous Environment by Using Polyvinylpyrrolidone-Coated Magnetic Nanoparticles. Water. 2019; 11(10):1993. https://doi.org/10.3390/w11101993
Chicago/Turabian StyleShao, Shimin, Yan Li, Ting Lü, Dongming Qi, Dong Zhang, and Hongting Zhao. 2019. "Removal of Emulsified Oil from Aqueous Environment by Using Polyvinylpyrrolidone-Coated Magnetic Nanoparticles" Water 11, no. 10: 1993. https://doi.org/10.3390/w11101993
APA StyleShao, S., Li, Y., Lü, T., Qi, D., Zhang, D., & Zhao, H. (2019). Removal of Emulsified Oil from Aqueous Environment by Using Polyvinylpyrrolidone-Coated Magnetic Nanoparticles. Water, 11(10), 1993. https://doi.org/10.3390/w11101993





