The Combination of Coagulation and Adsorption for Controlling Ultra-Filtration Membrane Fouling in Water Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Model Water
2.2. Coagulant and Adsorbent Preparation
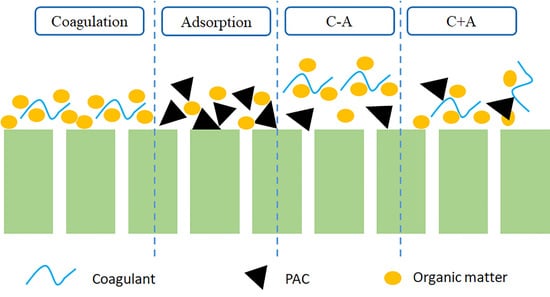
2.3. Comparison of the Four Pre-Treatments
2.3.1. Jar Tests
2.3.2. The Dynamic Ultra-Filtration Processes
2.4. Determination of Disinfection By-Products
2.5. Other Analytical Methods
3. Results
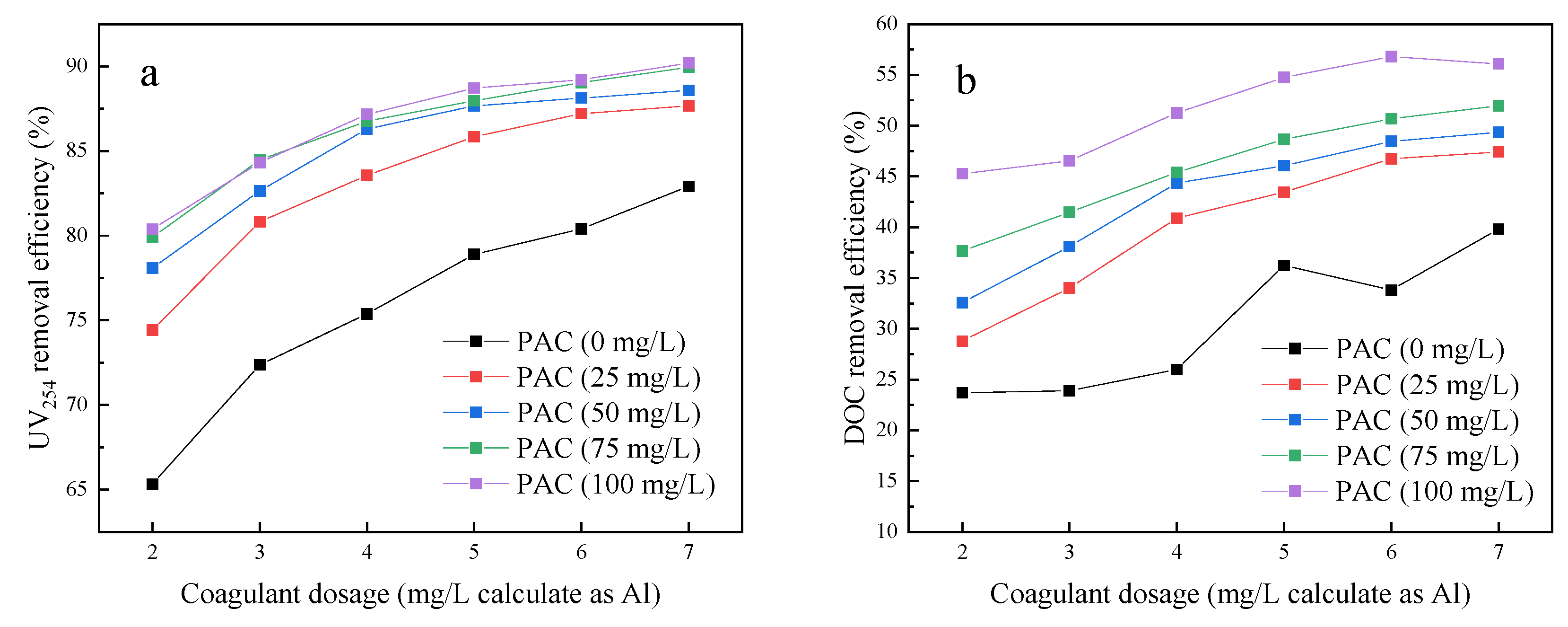
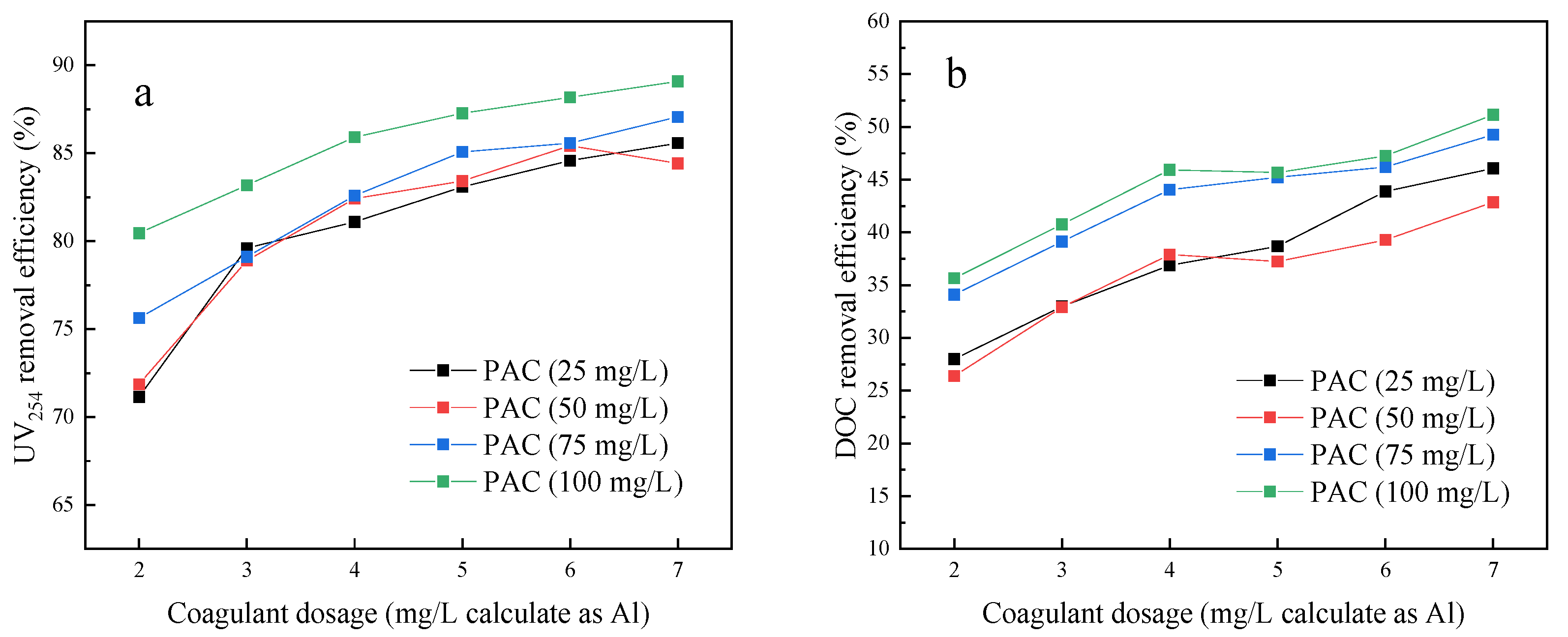
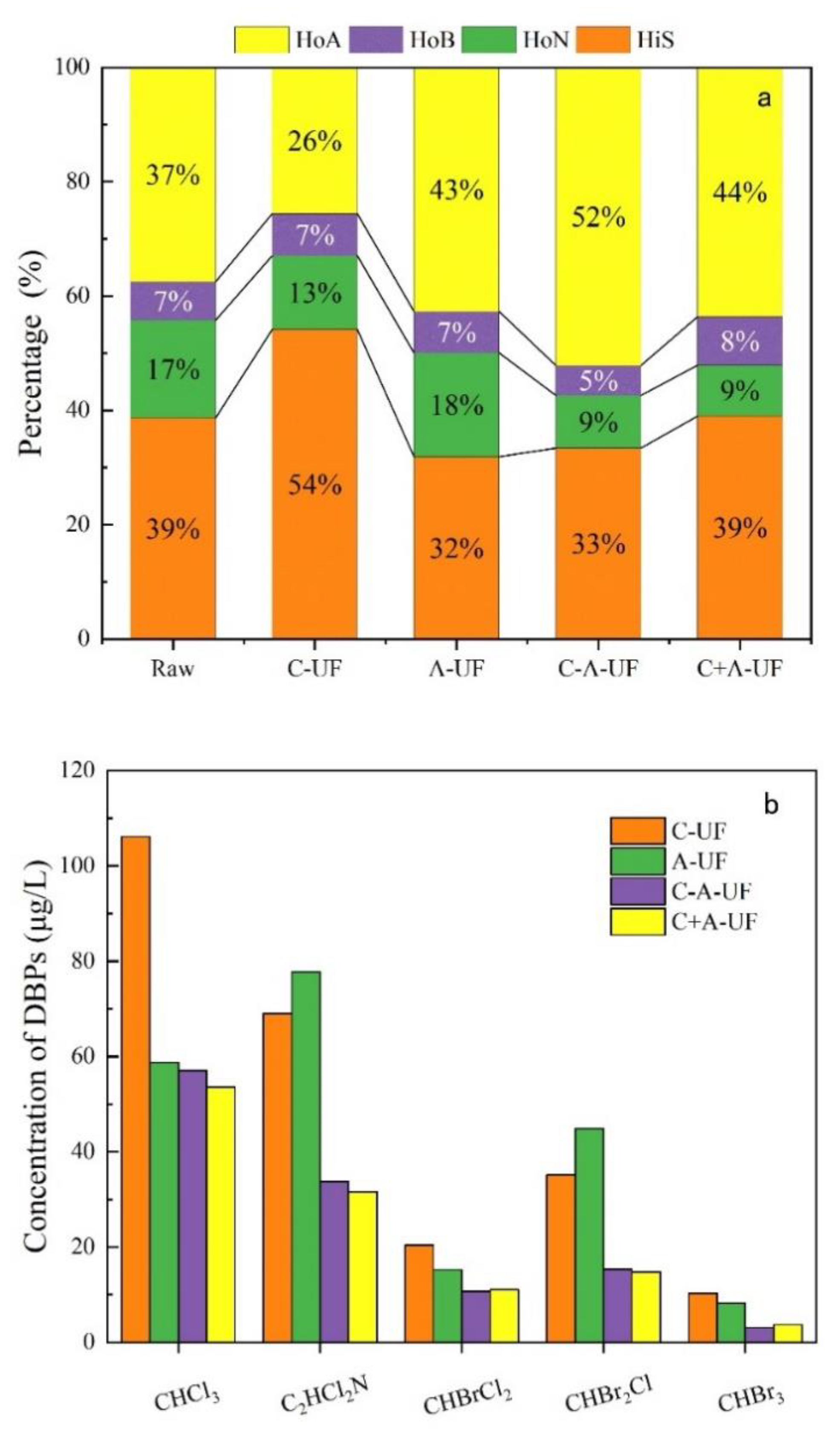
3.1. Jar Test Results Comparing NOM Removal by Four Different Pre-Treatments
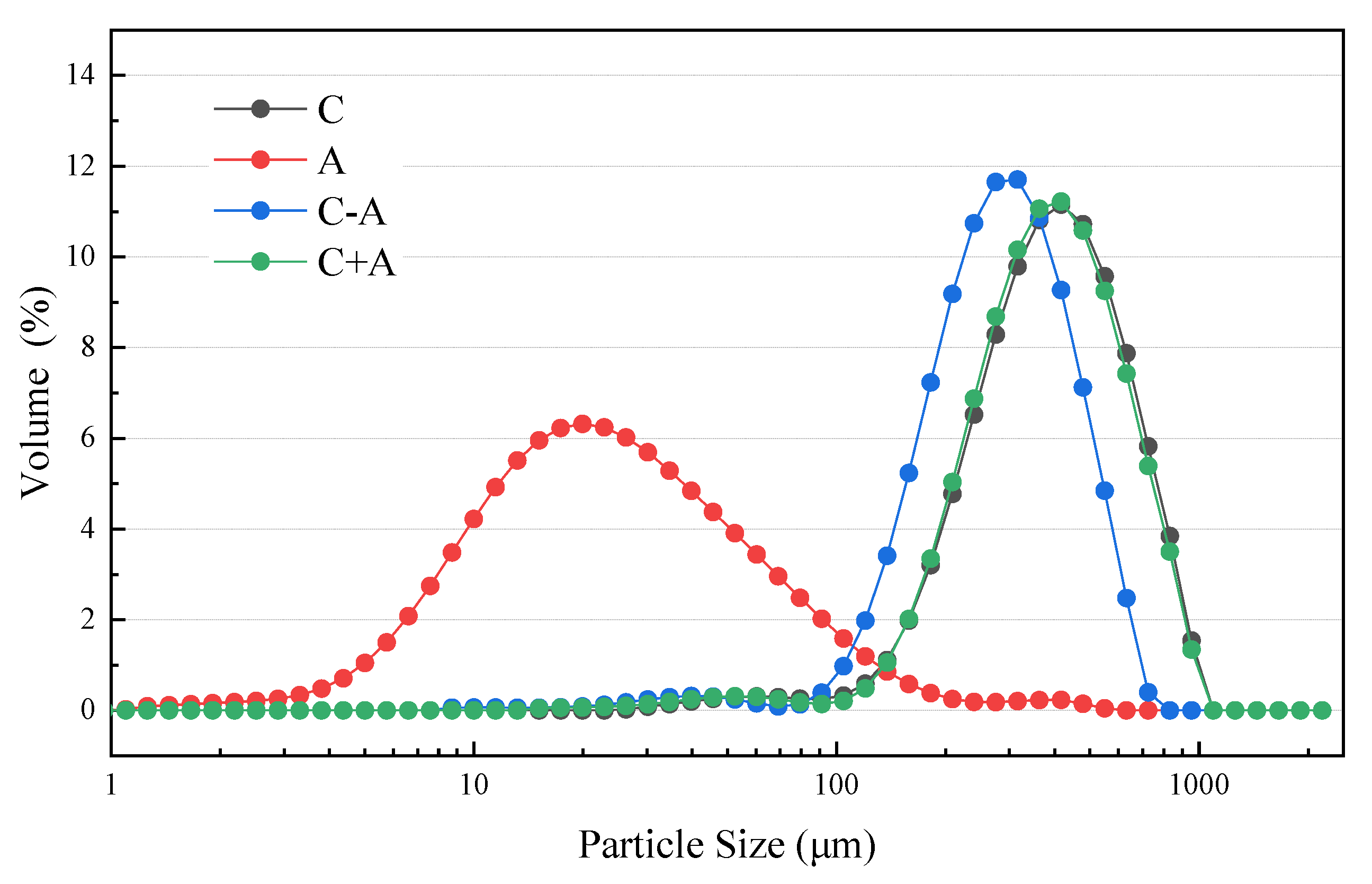
3.2. The Particle Size Distribution in the Effluents of Different Pre-Treatments
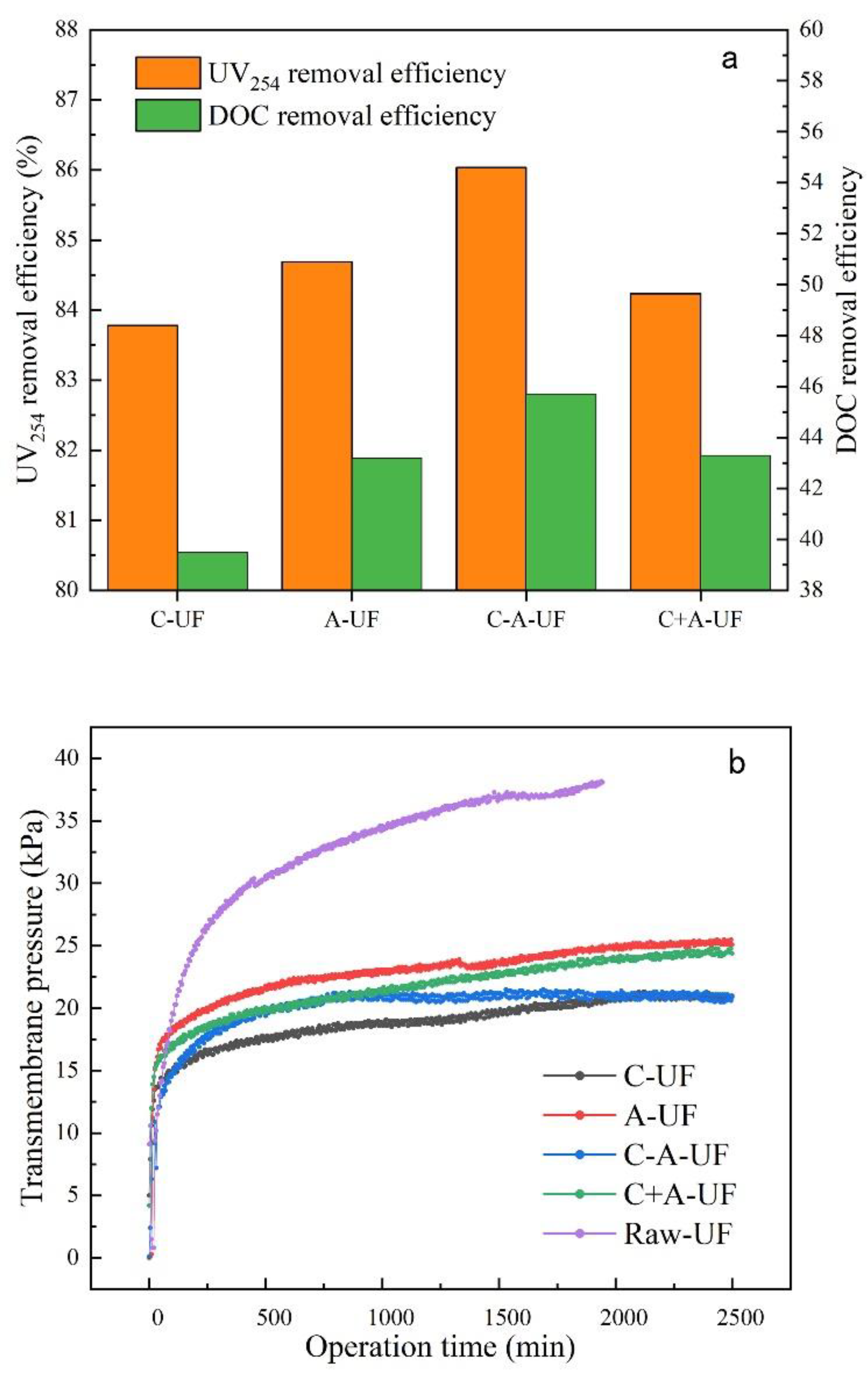
3.3. The Comparison of the Ultra-Filtration Effluents
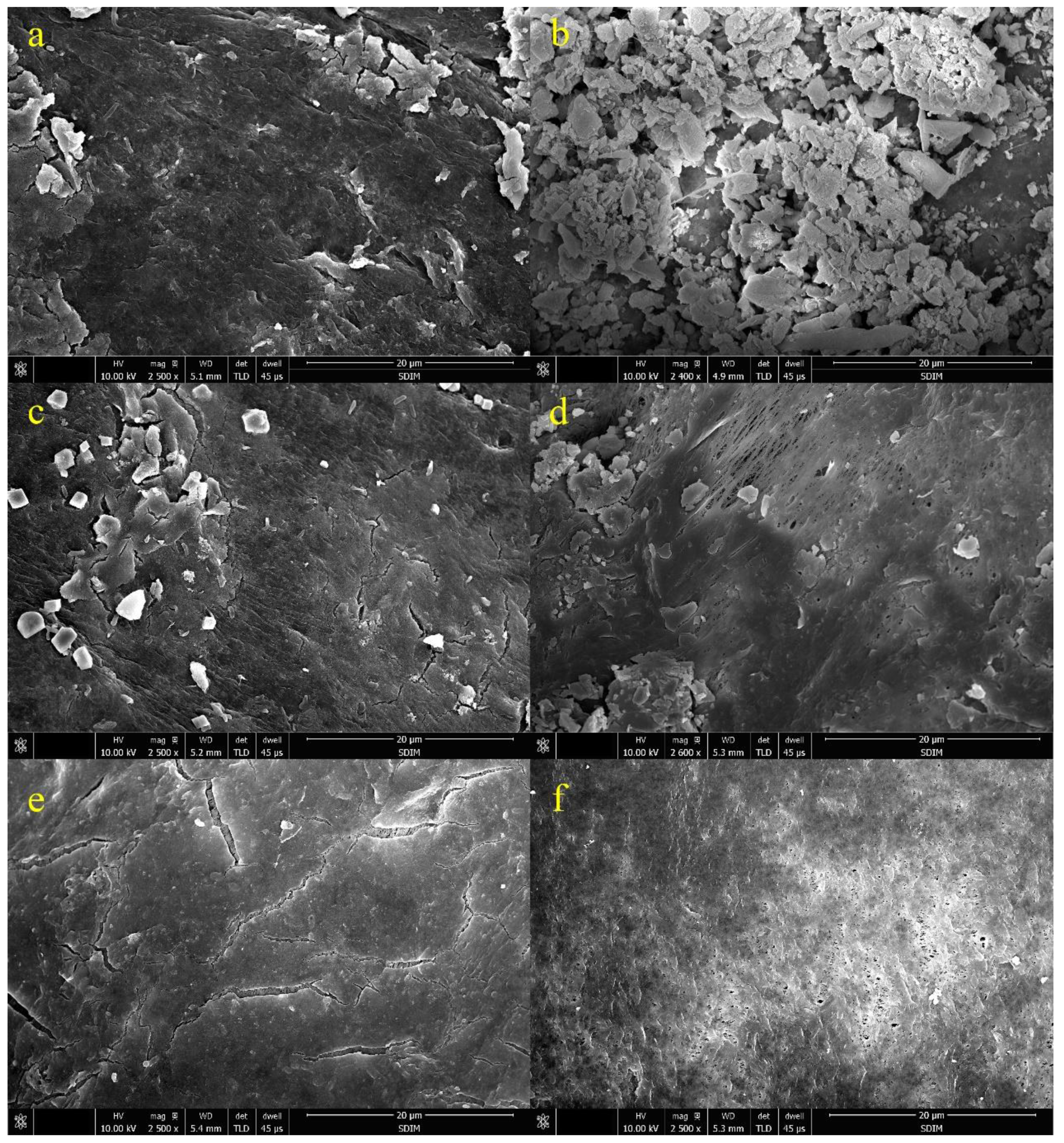
3.4. Characterization of the Fouled Membrane
4. Conclusions
- The addition of PAC enhanced the DOC and UV254 removal efficiencies of coagulation. The coagulation followed by adsorption pre-treatment (C-A-UF) process achieved the best water quality of the four processes.
- The C-A-UF process achieved a higher removal of hydrophilic substances than the C+A-UF process. As for the DBPs, the C-A-UF process showed better performance on the removal of CHBrCl2 and CHBr3, while the C+A-UF process was more effective in the removal of CHCl3, C2HCl2N and CHBr2Cl.
- The TMP of C-UF and C-A-UF were the lowest of all processes. However, the addition of PAC can reduce the accumulation of particles on the membrane surface because PAC removes the organic matter from the filter cake. The C-A-UF process was the best choice for long term operation of ultra-filtration.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Boudaud, N.; Machinal, C.; David, F.; Frévalle, B.A.; Jossent, J.; Bakanga, F.; Arnal, C.; Jaffrezic, M.P.; Oberti, S.; Gantzer, C. Removal of ms2, qβ and ga bacteriophages during drinking water treatment at pilot scale. Water Res. 2012, 46, 2651–2664. [Google Scholar] [CrossRef] [PubMed]
- Jacangelo, J.G.; Trussell, R.R.; Watson, M. Role of membrane technology in drinking water treatment in the united states ☆. Desalination 1997, 113, 119–127. [Google Scholar] [CrossRef]
- Iritani, E.; Katagiri, N.; Masuda, H. Evaluation of variable pressure dead-end ultrafiltration behaviors of nanocolloids based on filter cake properties. Chem. Eng. Res. Des. 2018, 134, 528–539. [Google Scholar] [CrossRef]
- Loginov, M.; Samper, F.; Gésan-Guiziou, G.; Sobisch, T.; Lerche, D.; Vorobiev, E. Centrifugal ultrafiltration for determination of filter cake properties of colloids. J. Membr. Sci. 2017, 536, 59–75. [Google Scholar] [CrossRef]
- Sutzkover-Gutman, I.; Hasson, D.; Semiat, R. Humic substances fouling in ultrafiltration processes. Desalination 2010, 261, 218–231. [Google Scholar] [CrossRef]
- Al Aani, S.; Wright, C.J.; Hilal, N. Investigation of uf membranes fouling and potentials as pre-treatment step in desalination and surface water applications. Desalination 2018, 432, 115–127. [Google Scholar] [CrossRef]
- Amy, G. Fundamental understanding of organic matter fouling of membranes. Desalination 2008, 231, 44–51. [Google Scholar] [CrossRef]
- Gao, W.; Liang, H.; Ma, J.; Han, M.; Chen, Z.L.; Han, Z.S.; Li, G.B. Membrane fouling control in ultrafiltration technology for drinking water production: A review. Desalination 2011, 272, 1–8. [Google Scholar] [CrossRef]
- Zularisam, A.W.; Ismail, A.F.; Salim, M.R.; Sakinah, M.; Ozaki, H. The effects of natural organic matter (nom) fractions on fouling characteristics and flux recovery of ultrafiltration membranes. Desalination 2007, 212, 191–208. [Google Scholar] [CrossRef]
- Tomaszewska, M.; Mozia, S. Removal of organic matter from water by pac/uf system. Water Res. 2002, 36, 4137–4143. [Google Scholar] [CrossRef]
- Stoquart, C.; Servais, P.; Bérubé, P.R.; Barbeau, B. Hybrid membrane processes using activated carbon treatment for drinking water: A review. J. Membr. Sci. 2012, 411–412, 1–12. [Google Scholar] [CrossRef]
- Lee, S.J.; Choo, K.H.; Lee, C.H. Conjunctive use of ultrafiltration with powdered activated carbon adsorption for removal of synthetic and natural organic matter. Neuro-Ophthalmology 2000, 6, 357–364. [Google Scholar]
- Lin, C.F.; Hao, O.J.; Huang, Y.J. Ultrafiltration processes for removing humic substances: Effect of molecular weight fractions and pac treatment. Water Res. 1999, 33, 1252–1264. [Google Scholar] [CrossRef]
- Dong, B.Z.; Wang, H.W.; Feng, J.; Li, W.Y. Influence of coagulation pretreatment on uf membrane flux. Huan Jing Ke Xue 2008, 29, 2783. [Google Scholar] [PubMed]
- Yu, W.Z.; Liu, H.J.; Xu, L.; Qu, J.H.; Graham, N. The pre-treatment of submerged ultrafiltration membrane by coagulation—Effect of polyacrylamide as a coagulant aid. J. Membr. Sci. 2013, 446, 50–58. [Google Scholar] [CrossRef]
- Korshin, G.; Chow, C.W.K.; Fabris, R.; Drikas, M. Absorbance spectroscopy-based examination of effects of coagulation on the reactivity of fractions of natural organic matter with varying apparent molecular weights. Water Res. 2009, 43, 1541–1548. [Google Scholar] [CrossRef] [PubMed]
- Altmann, J.; Zietzschmann, F.; Geiling, E.L.; Ruhl, A.S.; Sperlich, A.; Jekel, M. Impacts of coagulation on the adsorption of organic micropollutants onto powdered activated carbon in treated domestic wastewater. Chemosphere 2015, 125, 198–204. [Google Scholar] [CrossRef]
- Zhang, X.; Minear, R.A. Formation, adsorption and separation of high molecular weight disinfection byproducts resulting from chlorination of aquatic humic substances. Water Res. 2006, 40, 221–230. [Google Scholar] [CrossRef]
- Jiang, J.; Zhang, X. A smart strategy for controlling disinfection byproducts by reversing the sequence of activated carbon adsorption and chlorine disinfection. Sci. Bull. 2018, 64, 1167–1169. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Y.; Zhao, X.; Gao, N.; Fu, T. The characteristics of sludge from enhanced coagulation processes using pac/pdmdaac composite coagulants in treatment of micro-polluted raw water. Sep. Purif. Technol. 2015, 147, 125–131. [Google Scholar] [CrossRef]
- Xu, W.; Yue, Q.; Gao, B.; Du, B. Impacts of organic coagulant aid on purification performance and membrane fouling of coagulation/ultrafiltration hybrid process with different al-based coagulants. Desalination 2015, 363, 126–133. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, B.; Yue, Q.; Wei, J.; Li, Q. The characterization and flocculation efficiency of composite flocculant iron salts–polydimethyldiallylammonium chloride. Chem. Eng. J. 2008, 142, 175–181. [Google Scholar] [CrossRef]
- Ma, D.; Gao, B.; Sun, S.; Wang, Y.; Yue, Q.; Li, Q. Effects of dissolved organic matter size fractions on trihalomethanes formation in mbr effluents during chlorine disinfection. Bioresour. Technol. 2013, 136, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Edzwald, J.K. Coagulation in drinking water treatment: Particles, organics and coagulants. Waterence Technol. 1993, 27, 21–35. [Google Scholar] [CrossRef]
- Benschoten, J.E.V.; Edzwald, J.K. Chemical aspects of coagulation using aluminum salts—I. Hydrolytic reactions of alum and polyaluminum chloride. Water Res. 1990, 24, 1519–1526. [Google Scholar] [CrossRef]
- Li, K.; Liang, H.; Qu, F.; Shao, S.; Yu, H.; Han, Z.S.; Du, X.; Li, G. Control of natural organic matter fouling of ultrafiltration membrane by adsorption pretreatment: Comparison of mesoporous adsorbent resin and powdered activated carbon. J. Membr. Sci. 2014, 471, 94–102. [Google Scholar] [CrossRef]
- Kovalova, L.; Knappe, D.R.U.; Lehnberg, K.; Kazner, C.; Hollender, J. Removal of highly polar micropollutants from wastewater by powdered activated carbon. Environ. Sci. Pollut. Res. 2013, 20, 3607–3615. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Jin, Z.; Wang, Q.; Zuo, J.; Wu, J.; Wang, K. Effects of adsorbent cake layer on membrane fouling during hybrid coagulation/adsorption microfiltration for sewage organic recovery. Chem. Eng. J. 2017, 317, 751–757. [Google Scholar] [CrossRef]
- Alexander, J.T.; Hai, F.I.; Al-Aboud, T.M. Chemical coagulation-based processes for trace organic contaminant removal: Current state and future potential. J. Environ. Manag. 2012, 111, 195–207. [Google Scholar] [CrossRef]
- Richardson, S.D.; Plewa, M.J.; Wagner, E.D.; Schoeny, R.; Demarini, D.M. Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research. Mutat. Res.-Rev. Mutat. Res. 2007, 636, 178–242. [Google Scholar] [CrossRef]
- Yang, L.; Kim, D.; Uzun, H.; Karanfil, T.; Hur, J. Assessing trihalomethanes (thms) and n-nitrosodimethylamine (ndma) formation potentials in drinking water treatment plants using fluorescence spectroscopy and parallel factor analysis. Chemosphere 2015, 121, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, S.; Kawai, Y.; Sugita, M. Infrared spectrum of poly(vinylidene fluoride). J. Polym. Sci. Part A Polym. Chem. 2010, 6, 861–869. [Google Scholar] [CrossRef]
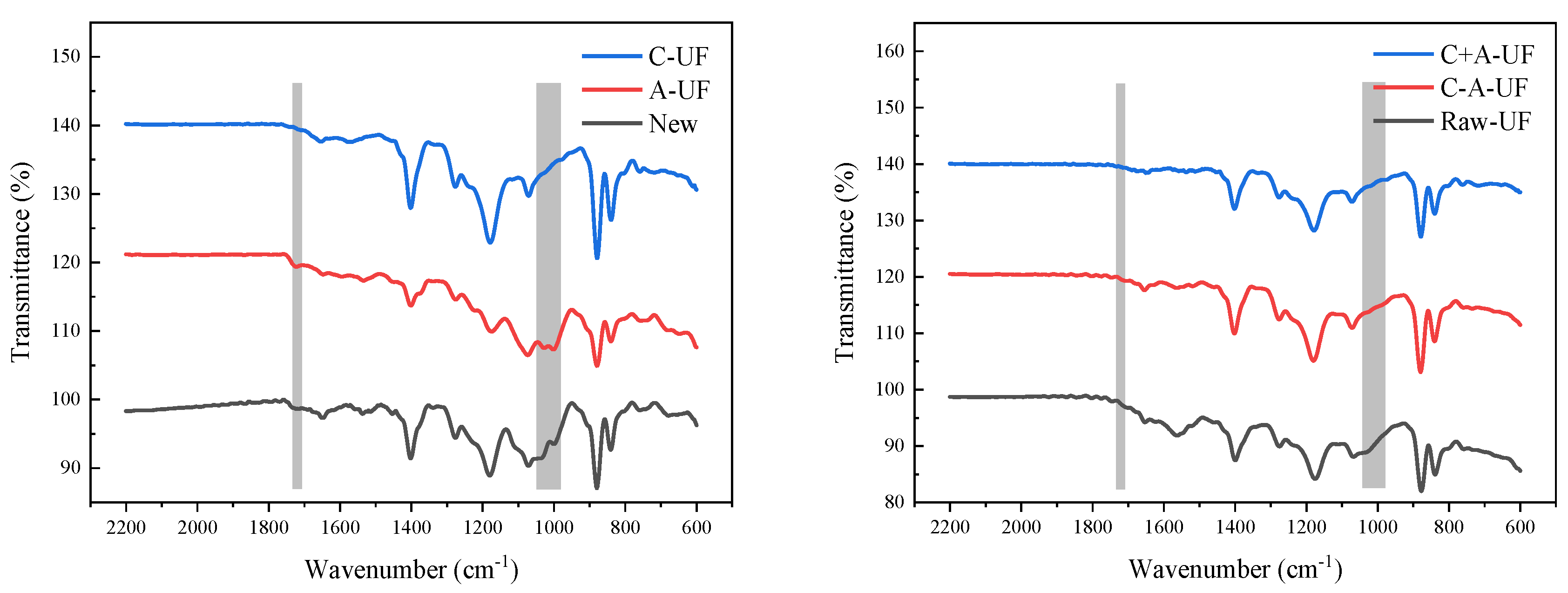
| Absorption Peaks (cm−1) | Assignments |
|---|---|
| 830—520 | F-C-F bending vibration |
| 1174 | CF2 stretching vibration |
| 1250—1220, 1016 | C-O stretching vibration |
| 1380 | C-H stretching vibration |
| 1402 | C-F stretching vibration |
| 1675—1500 | C=C stretching vibration |
| 1720 | C=O stretching vibration |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bu, F.; Gao, B.; Yue, Q.; Liu, C.; Wang, W.; Shen, X. The Combination of Coagulation and Adsorption for Controlling Ultra-Filtration Membrane Fouling in Water Treatment. Water 2019, 11, 90. https://doi.org/10.3390/w11010090
Bu F, Gao B, Yue Q, Liu C, Wang W, Shen X. The Combination of Coagulation and Adsorption for Controlling Ultra-Filtration Membrane Fouling in Water Treatment. Water. 2019; 11(1):90. https://doi.org/10.3390/w11010090
Chicago/Turabian StyleBu, Fan, Baoyu Gao, Qinyan Yue, Caiyu Liu, Wenyu Wang, and Xue Shen. 2019. "The Combination of Coagulation and Adsorption for Controlling Ultra-Filtration Membrane Fouling in Water Treatment" Water 11, no. 1: 90. https://doi.org/10.3390/w11010090
APA StyleBu, F., Gao, B., Yue, Q., Liu, C., Wang, W., & Shen, X. (2019). The Combination of Coagulation and Adsorption for Controlling Ultra-Filtration Membrane Fouling in Water Treatment. Water, 11(1), 90. https://doi.org/10.3390/w11010090