Rapid and Effective Isolation of Dissolved Organic Matter Using Solid-Phase Extraction Cartridges Packed with Amberlite XAD 8/4 Resins
Abstract
1. Introduction
2. Materials and Methods
2.1. Water Sample Preparation
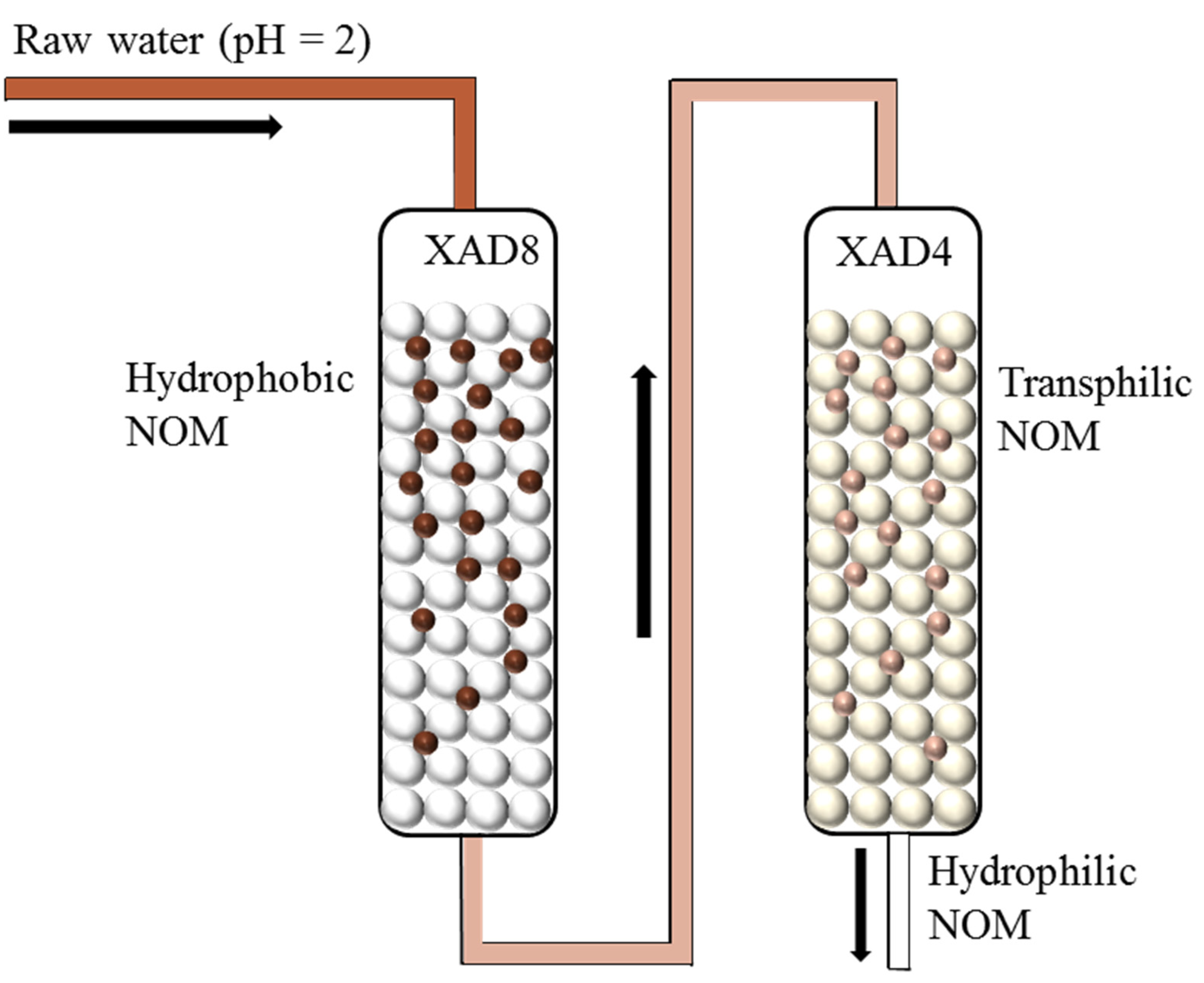
2.2. Procedure of Combined XAD 8/4 Resins Isolation with General Columns
2.3. Procedure for Combined XAD 8/4 Resin Isolation with SPE Cartridges
2.4. Analytical Methods
3. Results and Discussion
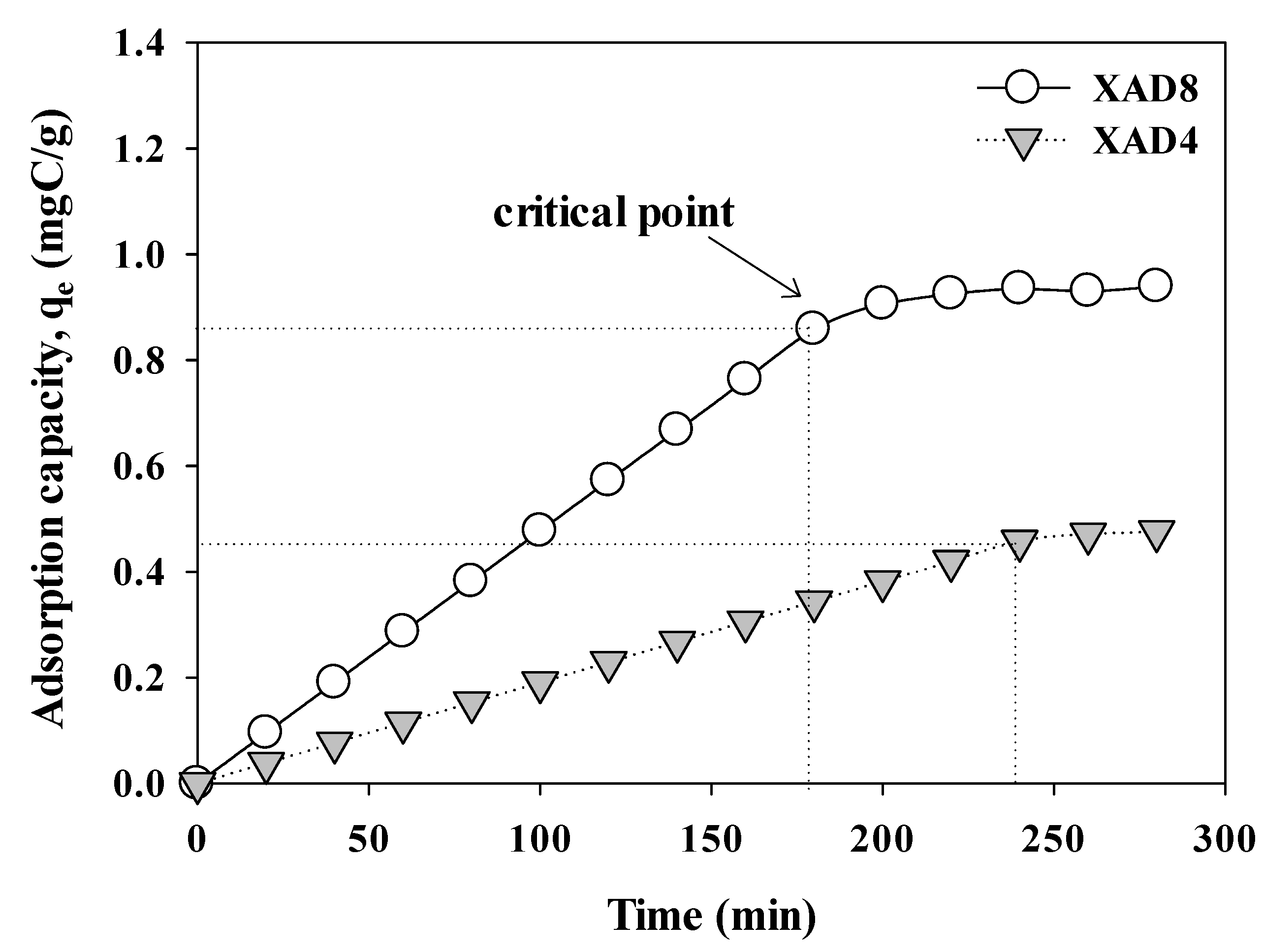
3.1. Comparison of Time for Organic Matter Combined Isolation with XAD 8/4 Resins
3.2. Adsorption Capacity (qe) of Combined XAD 8/4 Resins for AHA
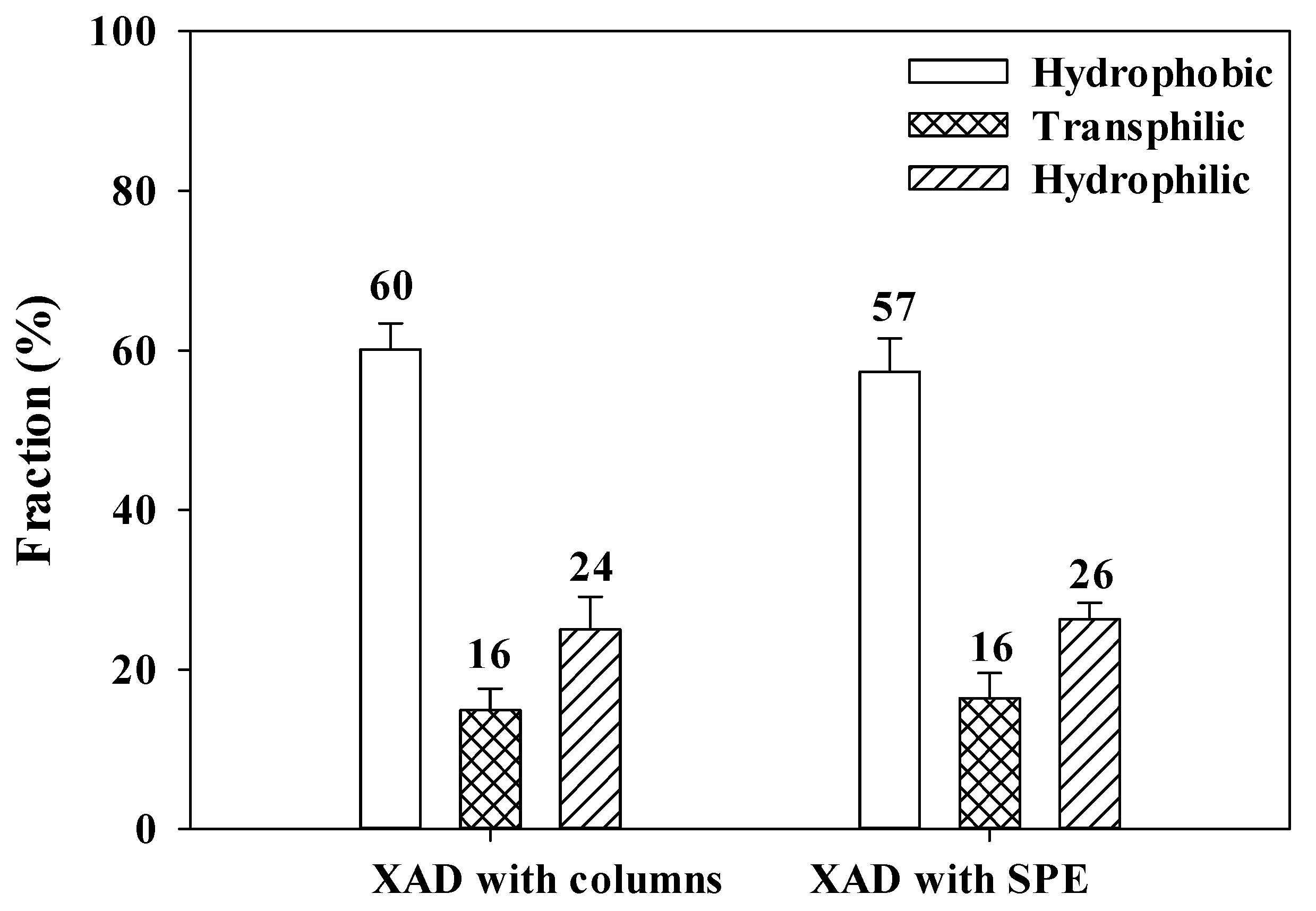
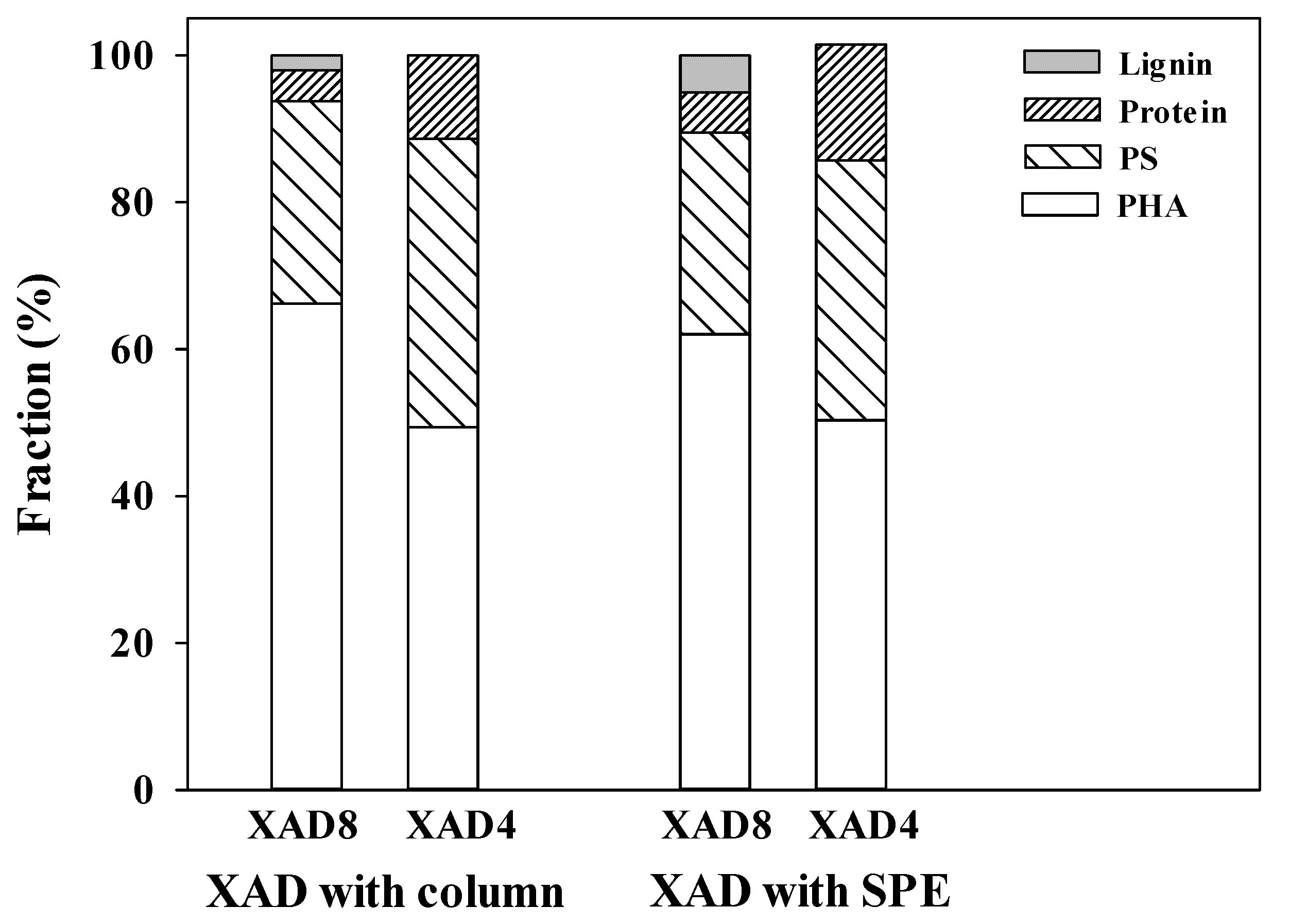
3.3. Relative Fraction of AHA and Mass Balance Determined by Combined XAD 8/4 Isolation with Columns and SPE Cartridges
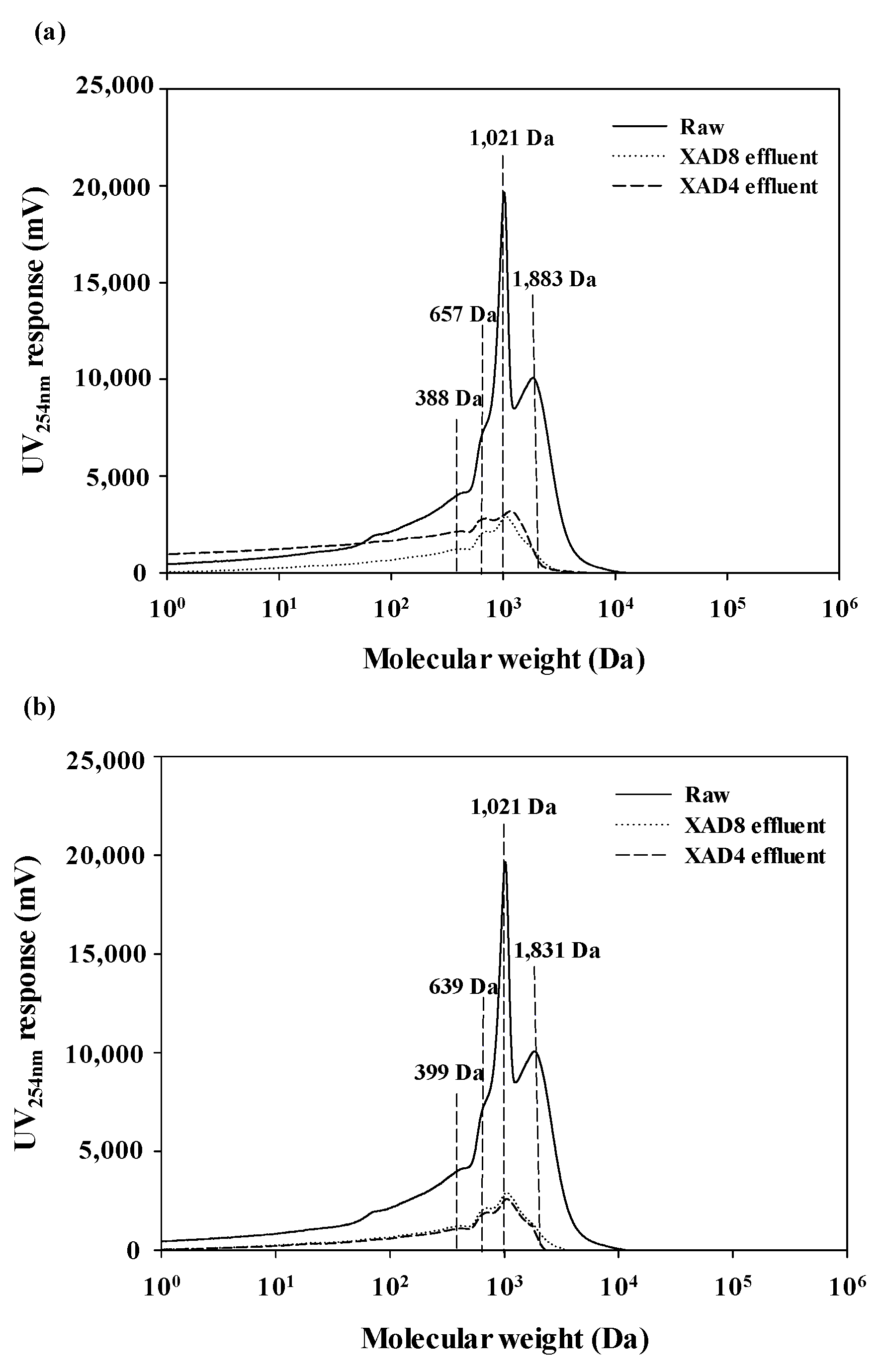
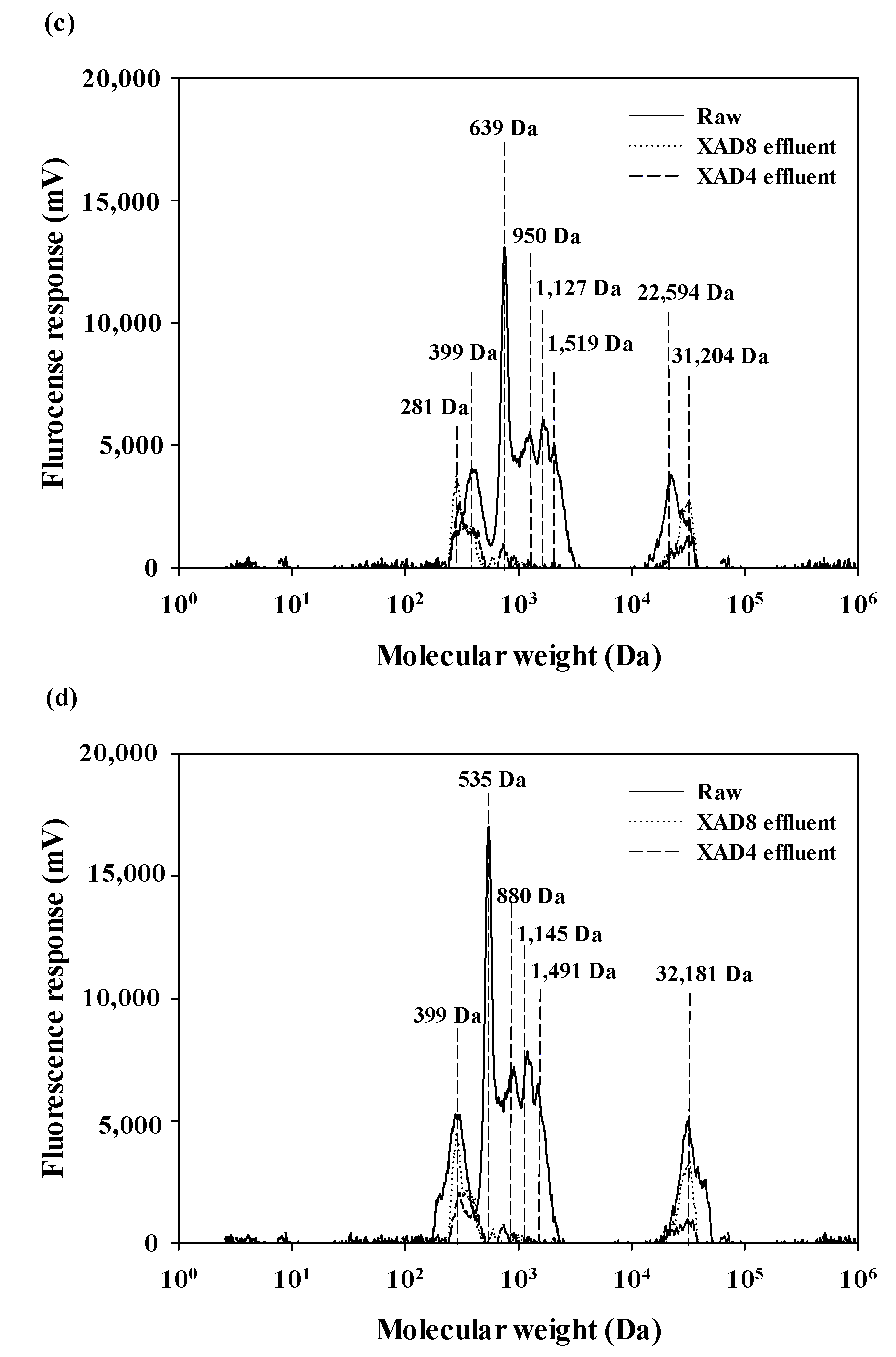
3.4. Physicochemical Properties of Isolated AHA by Combined XAD 8/4 with Columns and with SPE Cartridges
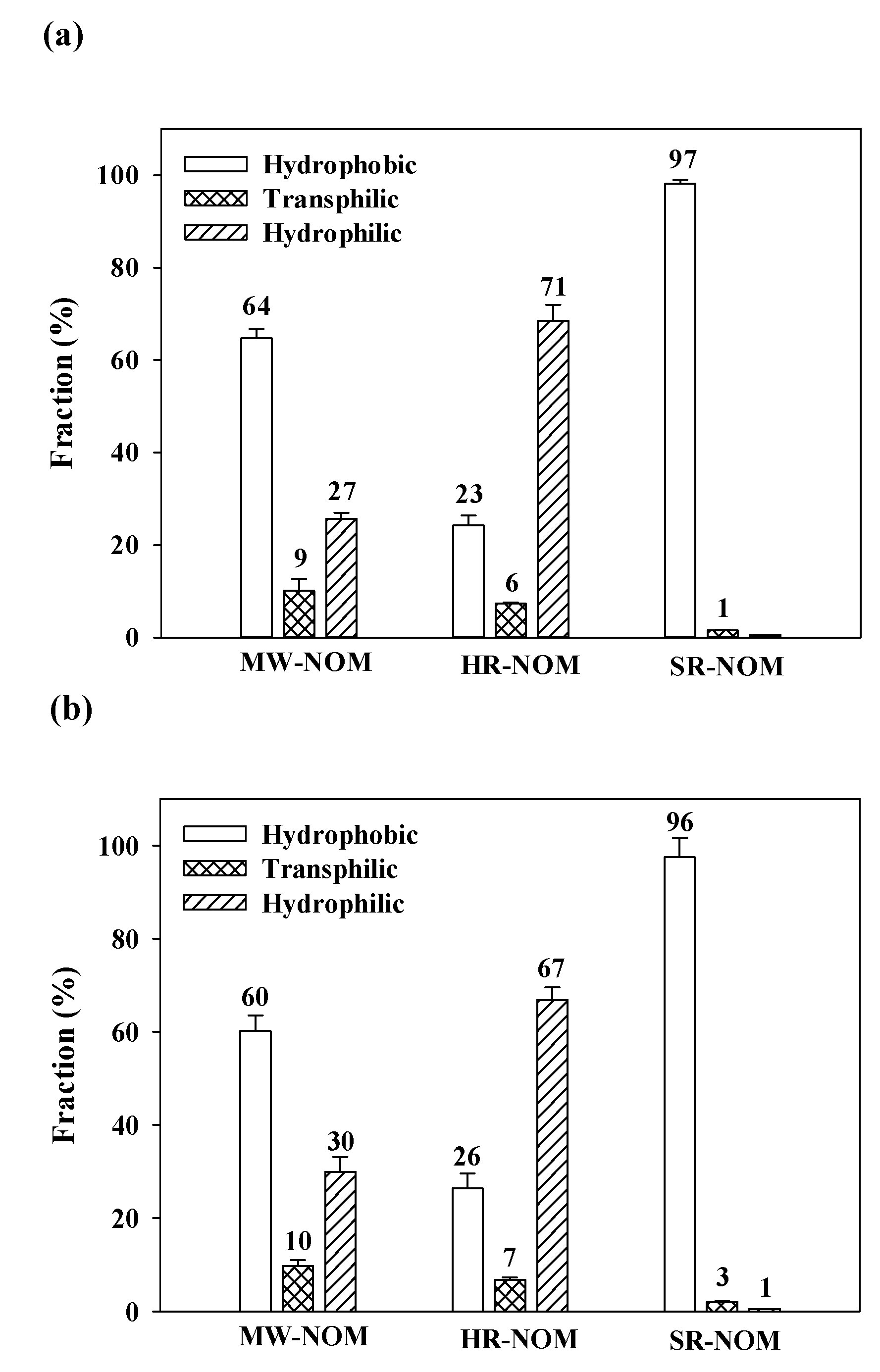
3.5. Comparison of NOM Fractionation with Environmental Samples by the XAD 8/4 Resins with Columns and with SPE Cartridges
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dietrich, A.M. Aesthetic issues for drinking water. J. Water Health 2006, 4, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Nyström, M.; Ruohomäki, K.; Kaipia, L. Humic acid as a fouling agent in filtration. Desalination 1996, 106, 79–87. [Google Scholar] [CrossRef]
- Yuan, W.; Zydney, A.L. Effects of solution environment on humic acid fouling during microfiltration. Desalination 1999, 122, 63–76. [Google Scholar] [CrossRef]
- Daignault, S.A.; Noot, D.K.; Williams, D.T.; Huck, P.M. A review of the use of XAD resins to concentrate organic compounds in water. Water Res. 1988, 22, 803–813. [Google Scholar] [CrossRef]
- Mantoura, R.F.C.; Riley, J.P. The analytical concentration of humic substances from natural waters. Anal. Chim. Acta 1975, 76, 97–106. [Google Scholar] [CrossRef]
- Thurman, E.M. Trace Compounds. In Organic Geochemistry of Natural Waters; Springer: Berlin, Germany, 1985; pp. 243–271. [Google Scholar]
- Leenheer, J.A. Comprehensive approach to preparative isolation and fractionation of dissolved organic carbon from natural waters and wastewaters. Environ. Sci. Technol. 1981, 15, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Aiken, G.R.; Thurman, E.M.; Malcolm, R.L.; Walton, H.F. Comparison of XAD macroporous resins for the concentration of fulvic acid from aqueous solution. Anal. Chem. 1979, 51, 1799–1803. [Google Scholar] [CrossRef]
- Thurman, E.M.; Malcolm, R.L.; Aiken, G.R. Prediction of capacity factors for aqueous organic solutes adsorbed on a porous acrylic resin. Anal. Chem. 1978, 50, 775–779. [Google Scholar] [CrossRef]
- Aiken, G.R.; McKnight, D.M.; Thorn, K.A.; Thurman, E.M. Isolation of hydrophilic organic acids from water using nonionic macroporous resins. Org. Geochem. 1992, 18, 567–573. [Google Scholar] [CrossRef]
- Malcolm, R.L.; MacCarthy, P. Quantitative evaluation of XAD-8 and XAD-4 resins used in tandem for removing organic solutes from water. Environ. Int. 1992, 18, 597–607. [Google Scholar] [CrossRef]
- Poole, C.F. New trends in solid-phase extraction. TrAC Trends Anal. Chem. 2003, 22, 362–373. [Google Scholar] [CrossRef]
- Mallet, A.I. Solid-phase extraction: Principles and practice E. M THURMAN and M. S MILLS: vol 147 in series chemical analysis, ed. J. D. Winefordner, John Wiley and Sons, New York, 1998 ISBN 0471 61422X Price £50. Rapid Commun. Mass Spectrom. 1998, 12, 988. [Google Scholar] [CrossRef]
- Rendle, D. Analytical Solid-Phase Extraction, by J.S. Fritz, Wiley-VCH, New York, 1999, xiii+209 pp, ISBN 0-471-24667-0; £41.95. Talanta 2000, 51, 1235. [Google Scholar] [CrossRef]
- Simpson, N. Solid-Phase Extraction; CRC Press: Boca Raton, FL, USA, 2000; ISBN 9780824700218. [Google Scholar]
- Pawliszyn, J. Chapter 9 Unified theory of extraction. Sampl. Sample Prep. Field Lab. 2002, 37, 253–278. [Google Scholar]
- Her, N.; Amy, G.; McKnight, D.; Sohn, J.; Yoon, Y. Characterization of DOM as a function of MW by fluorescence EEM and HPLC-SEC using UVA, DOC, and fluorescence detection. Water Res. 2003, 37, 4295–4303. [Google Scholar] [CrossRef]
- Her, N.; Amy, G.; Foss, D.; Cho, J.; Yoon, Y.; Kosenka, P. Optimization of Method for Detecting and Characterizing NOM by HPLC−Size Exclusion Chromatography with UV and On-Line DOC Detection. Environ. Sci. Technol. 2002, 36, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Bruchet, A.; Rousseau, C.; Mallevialle, J. Pyrolysis-GC-MS for Investigating High-Molecular-Weight THM Precursors and Other Refractory Organics. J. Am. Water Works Assoc. 1990, 82, 66–74. [Google Scholar] [CrossRef]
- Rodrı́guez, J.; Castrillón, L.; Marañón, E.; Sastre, H.; Fernández, E. Removal of non-biodegradable organic matter from landfill leachates by adsorption. Water Res. 2004, 38, 3297–3303. [Google Scholar] [CrossRef]
- Reinsberg, J.; Ackermann, D.; van der Ven, H. Pitfalls in assessment of progesterone production by granulosa cells cultured in contact with silicone rubber or paraffin oil. Arch. Gynecol. Obstet. 2003, 270, 174–178. [Google Scholar] [CrossRef]
- Valencia, S.; Marín, J.M.; Restrepo, G.; Frimmel, F.H. Application of excitation–emission fluorescence matrices and UV/Vis absorption to monitoring the photocatalytic degradation of commercial humic acid. Sci. Total Environ. 2013, 442, 207–214. [Google Scholar] [CrossRef]
- Fuentes, M.; Baigorri, R.; González-Vila, F.J.; González-Gaitano, G.; García-Mina, J.M. Pyrolysis–Gas Chromatography/Mass Spectrometry Identification of Distinctive Structures Providing Humic Character to Organic Materials. J. Environ. Qual. 2010, 39, 1486. [Google Scholar] [CrossRef] [PubMed]
- Leenheer, J.A.; Croué, J.-P. Peer Reviewed: Characterizing Aquatic Dissolved Organic Matter. Environ. Sci. Technol. 2003, 37, 18A–26A. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.A.; Philp, R.P.; Gillam, A.H.; Gilbert, T.D.; Tate, K.R. Comparison of the structures of humic substances from aquatic and terrestrial sources by pyrolysis gas chromatography-mass spectrometry. Geochim. Cosmochim. Acta 1983, 47, 497–502. [Google Scholar] [CrossRef]
- Cho, J.; Amy, G.; Pellegrino, J. Membrane filtration of natural organic matter: Factors and mechanisms affecting rejection and flux decline with charged ultrafiltration (UF) membrane. J. Memb. Sci. 2000, 164, 89–110. [Google Scholar] [CrossRef]
- Ferry, J.D.; Carritt, D.E. Action of Antifouling Paints. Ind. Eng. Chem. 1946, 38, 612–617. [Google Scholar] [CrossRef]
| Procedure | Solution | Flow Rate | XAD with Columns | XAD with SPE | ||
|---|---|---|---|---|---|---|
| (mL/min) | Amount (mL) | Duration (min) | Amount (mL) | Duration (min) | ||
| Clean | DI water | 2 | 250 | 125 | 83 | 42 |
| 0.1 N NaOH | 2 | 40 | 20 | 13 | 7 | |
| DI water | 2 | 40 | 20 | 13 | 7 | |
| 0.1 N HCl | 2 | 40 | 20 | 13 | 7 | |
| DI water | 2 | 40 | 20 | 13 | 7 | |
| 0.1 N NaOH | 2 | 40 | 20 | 13 | 7 | |
| DI water | 2 | 40 | 20 | 13 | 7 | |
| 0.1 N HCl | 2 | 40 | 20 | 13 | 7 | |
| DI water | 2 | 40 | 20 | 13 | 7 | |
| 0.1 N NaOH | 2 | 40 | 20 | 13 | 7 | |
| DI water | 2 | 40 | 20 | 13 | 7 | |
| 0.1 N HCl | 2 | 40 | 20 | 13 | 7 | |
| DI water | 2 | 40 | 20 | 13 | 7 | |
| Acidify | 0.1 N HCl | 2 | 40 | 20 | 13 | 7 |
| Load | Sample | 2 | 600 | 300 | 200 | 100 |
| Wash | DI water | 2 | 8 | 4 | 3 | 2 |
| Elute | 0.1 N NaOH | 1 | 40 | 40 | 13 | 13 |
| DI | 1 | 20 | 20 | 7 | 7 | |
| 0.1 N HCl | 1 | 30 | 30 | 10 | 10 | |
| Total | 1468 | 779 | 485 | 265 | ||
| Recovery (%) | 88.9 | 95.9 | ||||
| XAD with Columns | XAD with SPE | ||
|---|---|---|---|
| Carbon mass (mgC) | Raw AHA | 5.05 ± 0 | 5.05 ± 0 |
| XAD 8 isolate | 2.65 ± 0.02 | 2.74 ± 0.02 | |
| XAD 4 isolate | 0.63 ± 0.01 | 0.79 ± 0.01 | |
| XAD 4 effluent | 1.21 ± 0.01 | 1.31 ± 0.01 | |
| Total | 4.49 ± 0.04 | 4.84 ± 0.04 | |
| Recovery (%) | 88.9 ± 0.8 | 95.9 ± 0.6 | |
| Muryeongari Wetland (MW-NOM) | Han River (HR-NOM) | Suwannee River (SR-NOM) | |
|---|---|---|---|
| pH | 6.5 | 6.8 | 7.3 |
| DOC (mg-C L-1) | 7.6 ± 0.5 | 2.1 ± 0.3 | 10.1 ± 0.2 |
| UV254nm | 0.2123 ± 0.002 | 0.0358 ± 0.001 | 0.4341 ± 0.001 |
| SUVA (L/mg-m) | 2.8 ± 0.4 | 1.7 ± 0.3 | 4.3 ± 0.5 |
| TN (mg-N L-1) | 0.4 ± 0.1 | 4.6 ± 0.3 | 0.2 ± 0.1 |
| Conductivity (μS cm-1) | 42.9 ± 0.5 | 321.0 ± 2.4 | 12.1 ± 1.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rho, H.; Chon, K.; Park, J.; Cho, J. Rapid and Effective Isolation of Dissolved Organic Matter Using Solid-Phase Extraction Cartridges Packed with Amberlite XAD 8/4 Resins. Water 2019, 11, 67. https://doi.org/10.3390/w11010067
Rho H, Chon K, Park J, Cho J. Rapid and Effective Isolation of Dissolved Organic Matter Using Solid-Phase Extraction Cartridges Packed with Amberlite XAD 8/4 Resins. Water. 2019; 11(1):67. https://doi.org/10.3390/w11010067
Chicago/Turabian StyleRho, Hojung, Kangmin Chon, Jongkwan Park, and Jaeweon Cho. 2019. "Rapid and Effective Isolation of Dissolved Organic Matter Using Solid-Phase Extraction Cartridges Packed with Amberlite XAD 8/4 Resins" Water 11, no. 1: 67. https://doi.org/10.3390/w11010067
APA StyleRho, H., Chon, K., Park, J., & Cho, J. (2019). Rapid and Effective Isolation of Dissolved Organic Matter Using Solid-Phase Extraction Cartridges Packed with Amberlite XAD 8/4 Resins. Water, 11(1), 67. https://doi.org/10.3390/w11010067






