Use of Acorn Leaves as a Natural Coagulant in a Drinking Water Treatment Plant
Abstract
1. Introduction
2. Materials and Methods
2.1. Analytical Methods
2.2. Raw Water
2.3. Preparation of Used Coagulants (Acorn Leaves)
Characterization of Acorn Leaves
2.4. Experiments (Jar Test Assays)
3. Results and Discussion
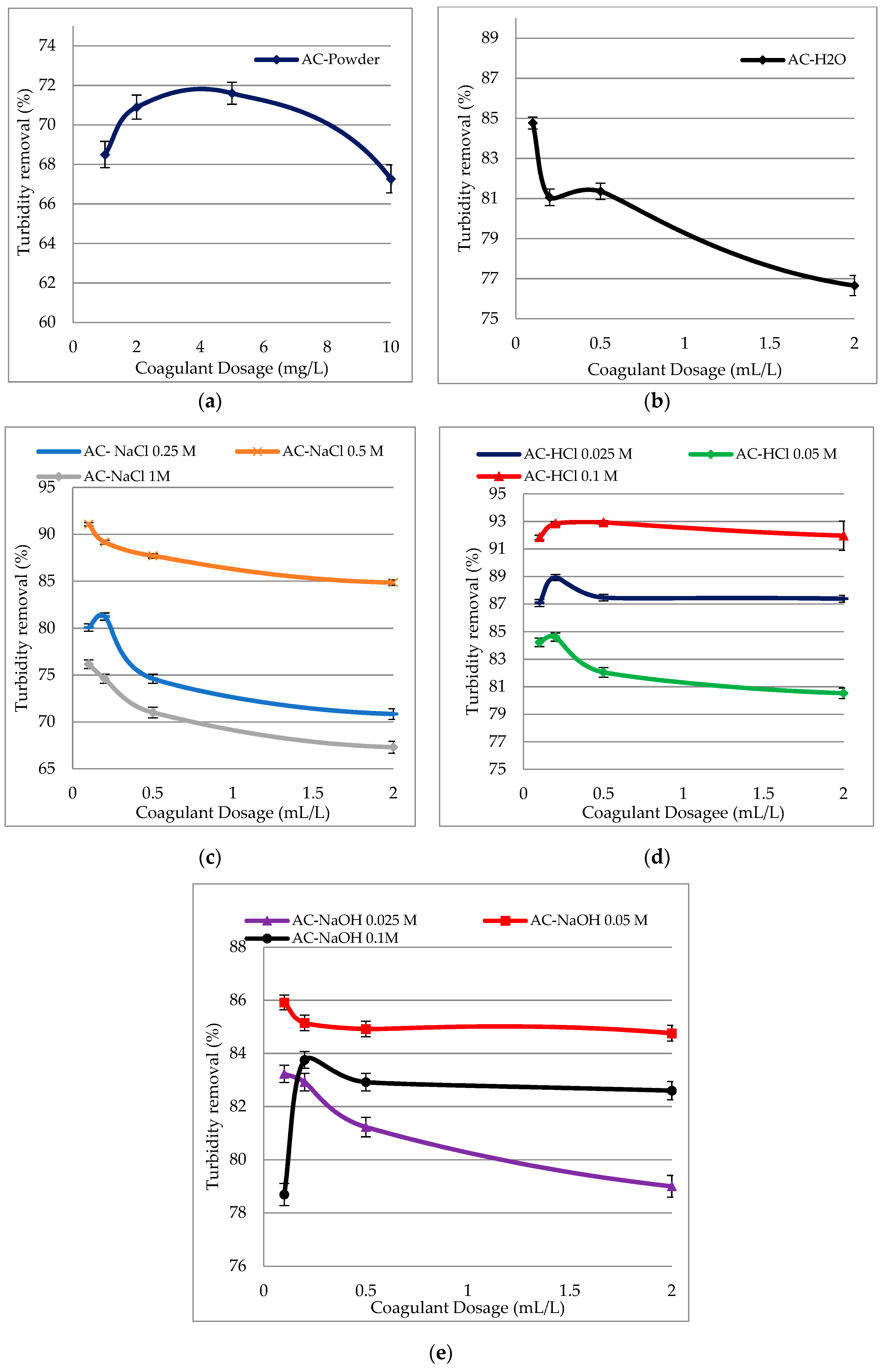
3.1. Effect of Using of Different Solvents to Extract the Coagulant
Effect of Coagulant Dosage on Turbidity Removal Efficiency for Different Solvents
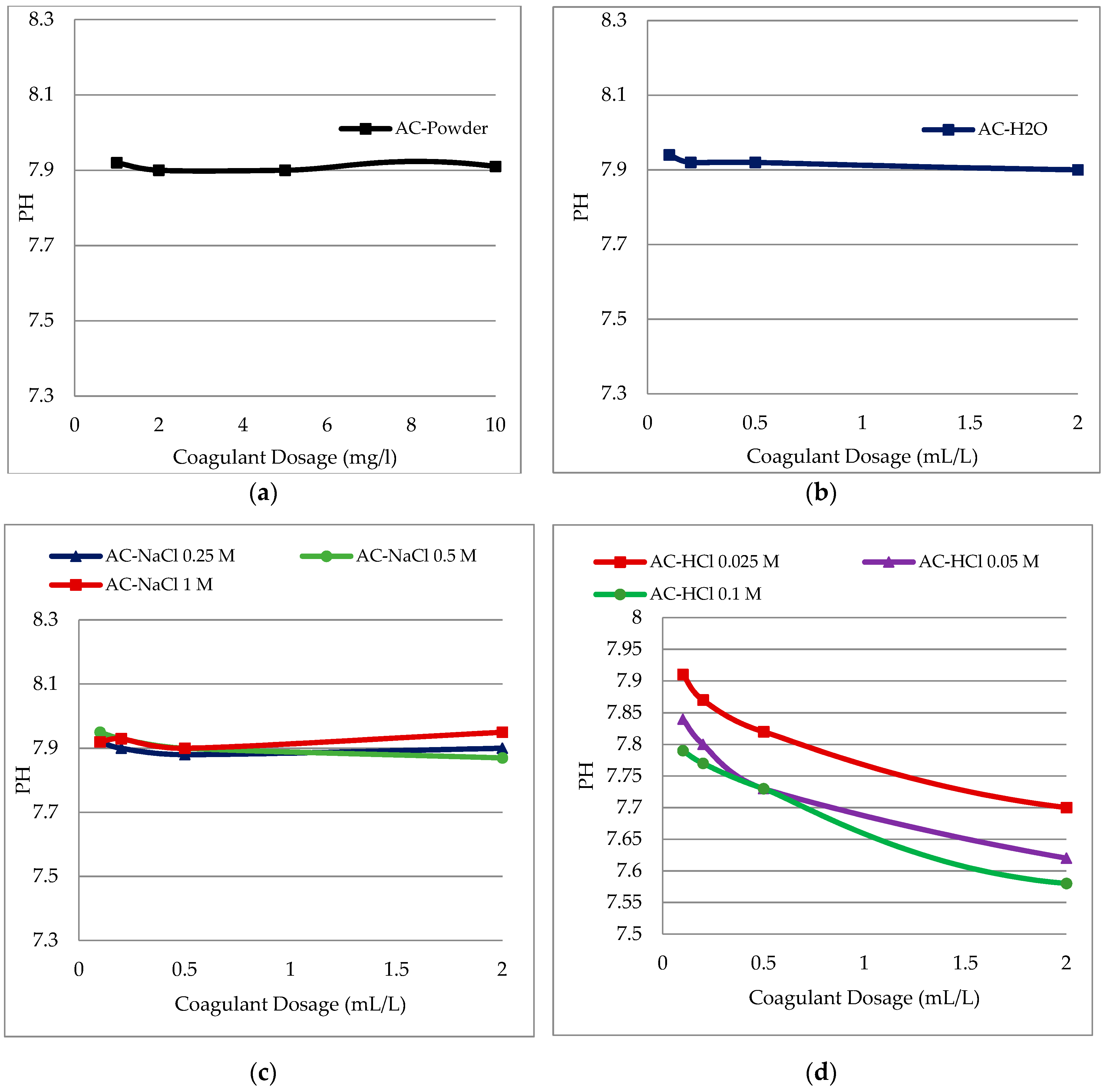
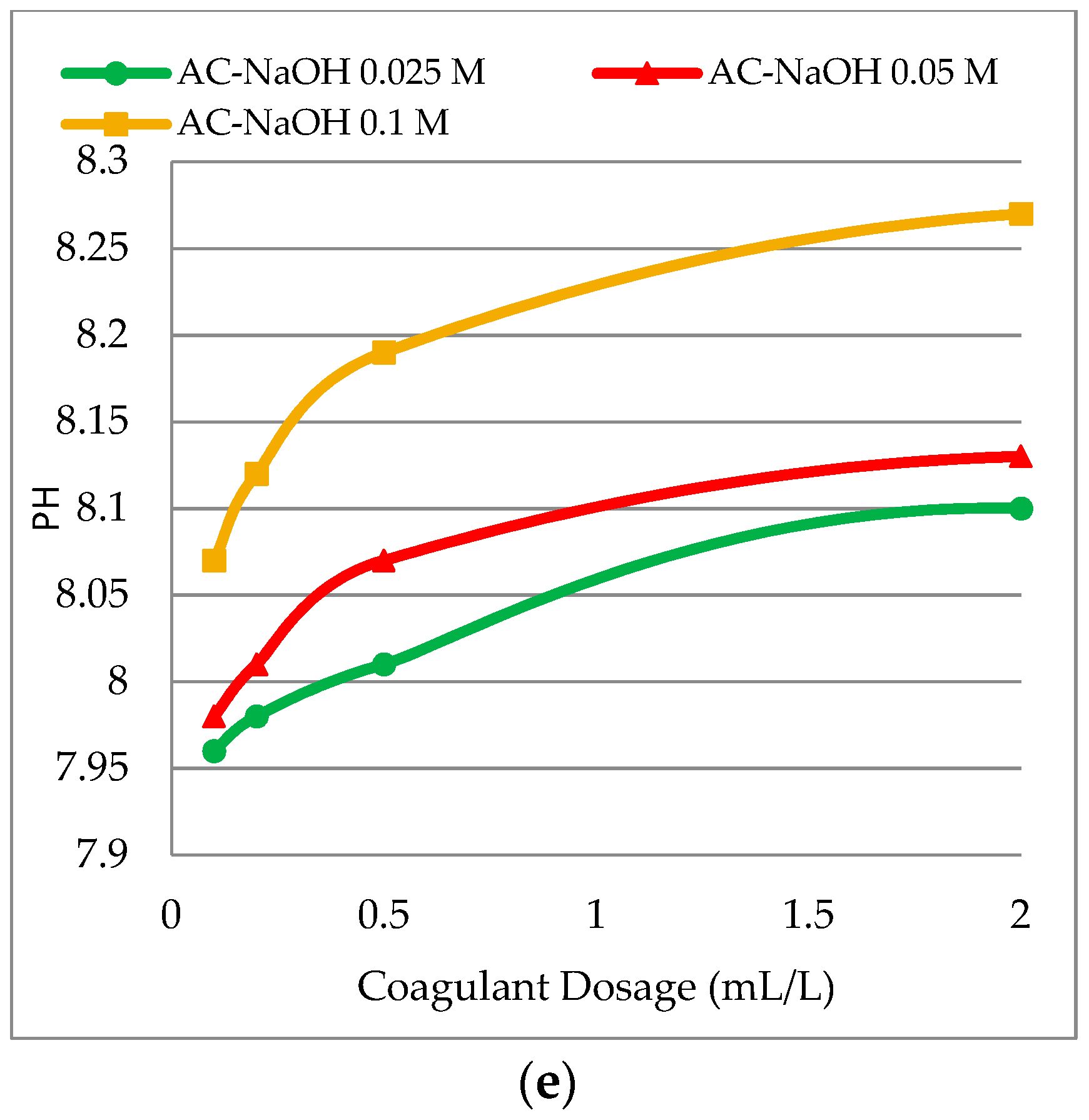
3.2. Effect of Coagulant Dosage on pH for Different Solvents
3.3. Effect of Coagulant Dosage on Total Alkalinity Hardness, Organic Matter Content and Electrical Conductivity
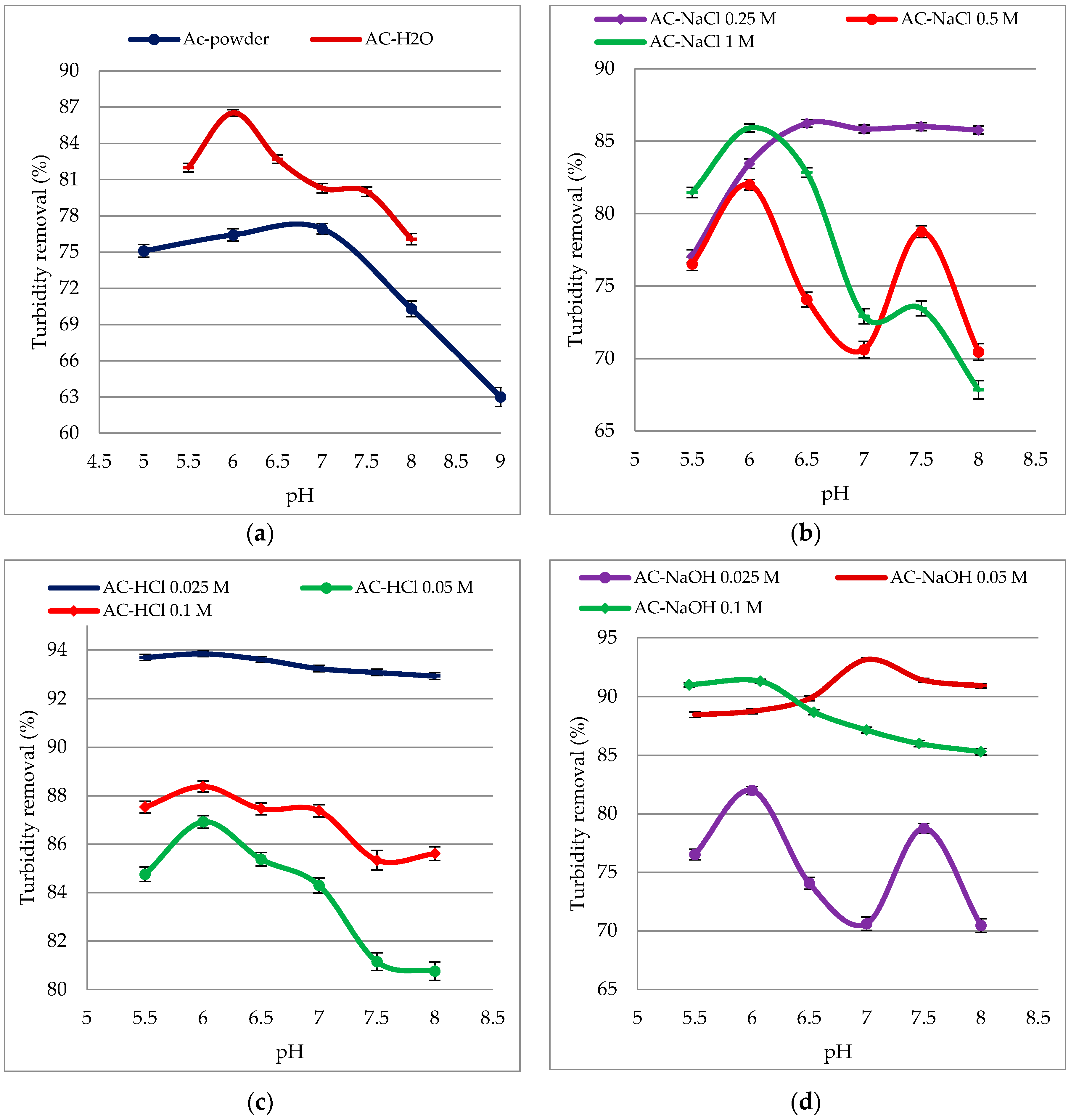
3.4. Effect of pH on the Turbidity Removal Efficiency
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Allouane, A. Caractérisation D’une Argile (Bentonite) de la Région de Maghnia pour L’adsorption du Phénol; Mémoire d’Ingéniera; Université d’Ouargla: Ouargla, Algérie, 2005. (In French) [Google Scholar]
- Chaib, I. Contribution au Dimensionnement D’une Station D’épuration pour la Station de Haoud El Hamra; Mémoire d’Ingéniera; Université d’Ouargla: Ouargla, Algerie, 2003. (In French) [Google Scholar]
- Race, M. Applicability of alkaline precipitation for the recovery of EDDS spent solution. J. Environ. Manag. 2017, 203, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Ciniglia, C.; Sansone, C.; Panzella, L.; Napolitano, A.; d’Ischia, M. Effects of walnut husk washing waters and their phenolic constituents on horticultural species. Environ. Sci. Pollut. Res. 2012, 19, 3299–3306. [Google Scholar] [CrossRef] [PubMed]
- Petriccione, M.; Forte, V.; Valente, D.; Ciniglia, C. DNA integrity of onion root cells under catechol influence. Environ. Sci. Pollut. Res. 2013, 20, 4859–4871. [Google Scholar] [CrossRef] [PubMed]
- Marsa, Z. Etude de L’effet de Différents Coagulants et Adsorbants sur la Qualité de L’eau Potable de la Station. Ph.D. Thesis, Université de Skikda, Skikda, Algérie, 2015. (In French). [Google Scholar]
- David, A.R.; Laura, S.; Julian, T.; Rocky, N.; Nicholas, A.P. Bioremediation of Aluminum from the Waste Water of a Conventional Water Treatment Plant Using the Freshwater Macroalga Oedogonium. Water 2018, 10, 626. [Google Scholar] [CrossRef]
- Alexander, K.; Gerald, H.; Kenneth, M.P.; Stephan, J.K. Combined Coagulation and Ultrafiltration Process to Counteract Increasing NOM in Brown Surface Water. Water 2017, 9, 697. [Google Scholar] [CrossRef]
- Kim, H.N.; Walker, S.L.; Bradford, S.A. Macromolecule mediated transport and retention of Escherichia coli O157:H7 in saturated porous media. Water Res. 2010, 44, 1082–1093. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.N.; Walker, S.L. Escherichia coli transport in porous media: Influence of cell strain, solution chemistry, and temperature. Colloids Surf. B Biointerfaces 2009, 71, 160–167. [Google Scholar] [CrossRef]
- Kim, H.N.; Bradford, S.A.; Walker, S.L. Escherichia coli O157:H7 Transport in Saturated PorousMedia: Role of Solution Chemistry and Surface Macromolecules. Environ. Sci. Technol. 2009, 43, 4340–4347. [Google Scholar] [CrossRef]
- Han, Y.; Kim, H. Surface Modification of Calcium Carbonate with Cationic Polymer and Their Dispersibility. Mater. Trans. 2012, 53, 2195–2199. [Google Scholar] [CrossRef]
- Kim, H.N.; Hong, Y.; Lee, I.; Bradford, S.A.; Walker, S.L. Surface Characteristics and Adhesion Behavior of Escherichia coli O157:H7: Role of Extracellular macromolecules. Biomacromolecules 2009, 10, 2556–2564. [Google Scholar] [CrossRef]
- Tong, M.; Long, G.; Jiang, X.; Kim, H.N. Contribution of Extracellular Polymeric Substances on Representative Gram Negative and Gram Positive Bacterial Deposition in Porous Media. Environ. Sci. Technol. 2010, 44, 2393–2399. [Google Scholar] [CrossRef] [PubMed]
- Benalia, A.; Derbal, K. Etude Expérimentale et Modélisation du Processus de la Coagulation Floculation: Application aux eaux Destinée a la Consommation. Master’s Thesis, Université de Constantine 3, Mendjeli, Algérie, 2015. (In French). [Google Scholar]
- Lefebvre, E.; Legube, B. Coagulation-floculation par le chlorure ferrique de quelques acides et phénols en solution aqueuse. Water 1993, 27, 433–447. [Google Scholar] [CrossRef]
- Benalia, A.; Derbal, K. Comparative study between Aluminum sulfate and Ferric Chloride. J. Dr. 2015, 1, 4–9. [Google Scholar]
- Colin, L.; Julien, F.; Lynda, K.; Mourad, K. Évaluation d’un Procédé de Coagulation-Floculation au Chitosane pour L’enlèvement du Phosphore dans les Effluents Piscicoles; Ecole de Technologie Supérieure: Montreal, QC, USA, 2008. (In French) [Google Scholar]
- Achouri, O.; Panico, A.; Bencheikh-Lehocine, M.; Derbal, K.; Pirozzi, F. Effect of chemical coagulation pretreatment on anaerobic digestion of tannery wastewater. J. Environ. Eng. 2017, 143, 04017039. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, F.; Luo, Y.; Yang, H. A preliminary study on the cactus coagulant in water treatment. Bioprocess Chem. 2006, 41, 730–733. [Google Scholar] [CrossRef]
- Vishali, S.; Karthikeyan, R. Cactus opuntia (ficus-indica): An eco-friendly alternative coagulant in the treatment of paint effluent. Desalin. Water Treat. 2014, 56, 1489–1497. [Google Scholar] [CrossRef]
- Sánchez-Martín, J.; Beltrán-Heredia, J.; Solera-Hernández, C. Surface water and wastewater treatment using a new tannin-based coagulant: Pilot plant trials. J. Environ. Manag. 2010, 9, 2051–2058. [Google Scholar] [CrossRef]
- Muyibi, S.A.; Noor, M.J.M.; Leong, T.K.; Loon, L.H. Effects of oil extraction from Moringa oleifera seeds on coagulation of turbid water. Int. J. Environ. Stud. 2002, 59, 243–254. [Google Scholar] [CrossRef]
- Lea, M. Bioremediation of turbid surface water using seed extract from the Moringa oleifera lam. (drumstick) tree. Curr. Protoc. Microbiol. 2014, 33, 1G.2.1–1G.2.8. [Google Scholar] [CrossRef]
- Sćiban, M.; Klasnja, M.; Antov, M.; Skrbić, B. Removal of water turbidity by natural coagulants obtained from chestnut and acorn. Bioresour. Technol. 2009, 100, 6639–6643. [Google Scholar] [CrossRef]
- Savi, B.; Lee, D.W. Investigation of Self-Assembly Processes for Chitosan-Based Coagulant-Flocculant systems: A Mini-Review. Int. J. Mol. Sci. 2016, 17, 1662. [Google Scholar] [CrossRef]
- Sofía, E.G.; Martha, A.F.; Ramírez, G.A.; Celia, G.F.; Saúl, C.Z.; Hayron, V.O. Comparing Two Operating Configurations in a Full-Scale Arsenic Removal Plant. Case Study: Guatemala. Water 2013, 5, 834–851. [Google Scholar] [CrossRef]
- Félix, L.J.; Olivier, L.; Ulysse, V.G.; Christian, T.; Caetano, C.D. Laboratory Efficacy and Disinfection by-Product Formation of a Coagulant/Disinfectant Tablet for Point-of-Use Water Treatment. Water 2018, 10, 1567. [Google Scholar] [CrossRef]
- Rice, E.W.; Baird, R.B.; Eaton, A.D.; Clesceri, L.S. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association (APHA), American Water Works Association (AWWA) and Water Environment Federation (WEF): Washington, DC, USA, 2012; ISBN 978-087553-0130. [Google Scholar]
- Zurina, Z.A.; Nur, S.M.S.; Norhafizah, M.; Shafreeza, S. Optimization of a method to extract the active coagulant agent from Jatropha curcas seeds for use in turbidity removal. Ind. Crops Prod. 2013, 41, 319–323. [Google Scholar] [CrossRef]
- Subramonian, W.; Wu, T.Y.; Chai, S.P. A comprehensive study on coagulant performance and floc characterization of natural Cassia obtusifolia seed gum in treatment of raw pulp and paper mill effluent. Ind. Crops Prod. 2014, 61, 317–324. [Google Scholar] [CrossRef]
- Kaplan, M.; Davidson, G.; Poliakoff, M. Capillary SFC-FTIR study of triglycerides. J. Chromatogr. 1994, A673, 231–237. [Google Scholar] [CrossRef]
- Dave, N.; Troullier, A.; Mus-Veteau, I.; Duñach, M.; Leblanc, G.; Padrós, E. Secondary structure components and properties of the melibiosepermease from Escherichia coli: A Fourier transform infrared spectroscopy analysis. Biophys. J. 2000, 79, 747–755. [Google Scholar] [CrossRef]
- Sanghi, R.; Bhattacharya, B.; Singh, V. Use of Cassia javahikai seed gum and gum-gpolyacrylamide as coagulant aid for the decolorization of textile dye solutions. Bioresour. Technol. 2006, 97, 1259–1264. [Google Scholar] [CrossRef]
- Nelson, D.L.; Lehninger, A.L.; Cox, M.M. Lehninger Principles of Biochemistry, 5th ed.; W.H. Freeman: New York, NY, USA, 2008. [Google Scholar]
- Voet, D.; Voet, J.G. Biochemistry; Wiley: New York, NY, USA, 1990; ISBN 10 0471512877; 13 9780471512875. [Google Scholar]
- Tong, M.; Zhu, P.; Jiang, X.; Kim, H. Influence of natural organic matter on the deposition kinetics of extracellular. Colloids Surf. B Biointerfaces 2011, 87, 160–167. [Google Scholar] [CrossRef]
- Han, P.; Shen, X.; Yang, H.; Kim, H.; Tong, M. Influence of nutrient conditions on the transport of bacteria in saturated porous media. Colloids Surf. B Biointerfaces 2013, 102, 752–758. [Google Scholar] [CrossRef]
- Choi, J.; Choi, S.Q.; Park, K.; Han, Y.; Kim, H. Flotation behaviour of malachite in mono- and di-valent salt solutions using sodium oleate as a collector. Int. J. Miner. Process. 2016, 146, 38–45. [Google Scholar] [CrossRef]
- Lestari, D.; Mulder, W.; Sanders, J. Improving Jatrophacurcas seed protein recovery by using counter current multistage extraction. Biochem. Eng. J. 2010, 50, 16–23. [Google Scholar] [CrossRef]
- Kim, H.N.; Walker, S.L.; Bradford, S.A. Coupled factors influencing the transport and retention of Cryptosporidium parvum oocysts in saturated porous media. Water Res. 2010, 40, 1213–1223. [Google Scholar] [CrossRef]
- Nacoulma, O.G.; Piro, J.; Bayane, A. Etude de l’activité floculant d’un complexe protéine-micilage végétale dans la clarification des eaux brutes. J. Soc. Ouest Afr. Chim. 2000, 9, 43–57. [Google Scholar]


| Parameters | Values | Algerian Standard |
|---|---|---|
| pH | 7.94 ± 0.01 | 6.5–9 |
| Turbidity (NTU) | 13.0 ± 0.1 | 5 |
| Total alkalinity hardness (F°) | 16 ± 1 | 20 |
| Electrical conductivity (μs/cm) | 1244 ± 10 | 2800 |
| Organic matter (mg O2/L) | 2.1 ± 0.2 | 5 |
| Coagulant | Optimal Dosage | Turbidity (NTU) | Total Alkalinity Hardness (F°) | Organic Matter (mg O2/L) | Electrical Conductivity (μs/cm) |
|---|---|---|---|---|---|
| AC-powder (mg/L) | 5.0 | 3.69 ± 0.0 | 16.1 ± 0.2 | 3.8 ± 0.4 | 1244 ± 12 |
| AC-H2O (mL/L) | 0.1 | 1.97 ± 0.03 | 16.2 ± 0.1 | 2.2 ± 0.2 | 1246 ± 24 |
| AC-NaCl 0.25 M (mL/L) | 0.2 | 2.44 ± 0.07 | 16.2 ± 0.1 | 2.1 ± 0.2 | 1260 ± 16 |
| AC-NaCl 0.5 M (mL/L) | 0.1 | 1.16 ± 0.05 | 16.3 ± 0.2 | 2.0 ± 0.1 | 1260 ± 18 |
| AC-NaCl 1 M (mL/L) | 0.1 | 3.10 ± 0.16 | 16.3 ± 0.3 | 2.4 ± 0.2 | 1269 ± 15 |
| AC-HCl 0.025 M (mL/L) | 0.2 | 1.44 ± 0.03 | 15.4 ± 0.2 | 2.8 ± 0.1 | 1264 ± 16 |
| AC-HCl 0.05 M (mL/L) | 0.2 | 2.00 ± 0.08 | 15.0 ± 0.1 | 2.8 ± 0.5 | 1272 ± 17 |
| AC-HCl 0.1 M (mL/L) | 0.5 | 0.92 ± 0.02 | 13.4 ± 0.1 | 3.0 ± 0.3 | 1280 ± 15 |
| AC-NaOH 0.025 M (mL/L) | 0.1 | 2.18 ± 0.09 | 16.4 ± 0.2 | 1.7 ± 0.3 | 1264 ± 28 |
| AC-NaOH 0.05 M (mL/L) | 0.1 | 1.83 ± 0.13 | 16.6 ± 0.2 | 2.3 ± 0.2 | 1267 ± 16 |
| AC-NaOH 0.1 M (mL/L) | 0.2 | 2.11 ± 0.08 | 17.0 ± 0.1 | 3.0 ± 0.4 | 1274 ± 13 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benalia, A.; Derbal, K.; Panico, A.; Pirozzi, F. Use of Acorn Leaves as a Natural Coagulant in a Drinking Water Treatment Plant. Water 2019, 11, 57. https://doi.org/10.3390/w11010057
Benalia A, Derbal K, Panico A, Pirozzi F. Use of Acorn Leaves as a Natural Coagulant in a Drinking Water Treatment Plant. Water. 2019; 11(1):57. https://doi.org/10.3390/w11010057
Chicago/Turabian StyleBenalia, Abderrezzaq, Kerroum Derbal, Antonio Panico, and Francesco Pirozzi. 2019. "Use of Acorn Leaves as a Natural Coagulant in a Drinking Water Treatment Plant" Water 11, no. 1: 57. https://doi.org/10.3390/w11010057
APA StyleBenalia, A., Derbal, K., Panico, A., & Pirozzi, F. (2019). Use of Acorn Leaves as a Natural Coagulant in a Drinking Water Treatment Plant. Water, 11(1), 57. https://doi.org/10.3390/w11010057







