A Novel System for Water Disinfection with UV Radiation
Abstract
1. Introduction
2. Materials and Methods
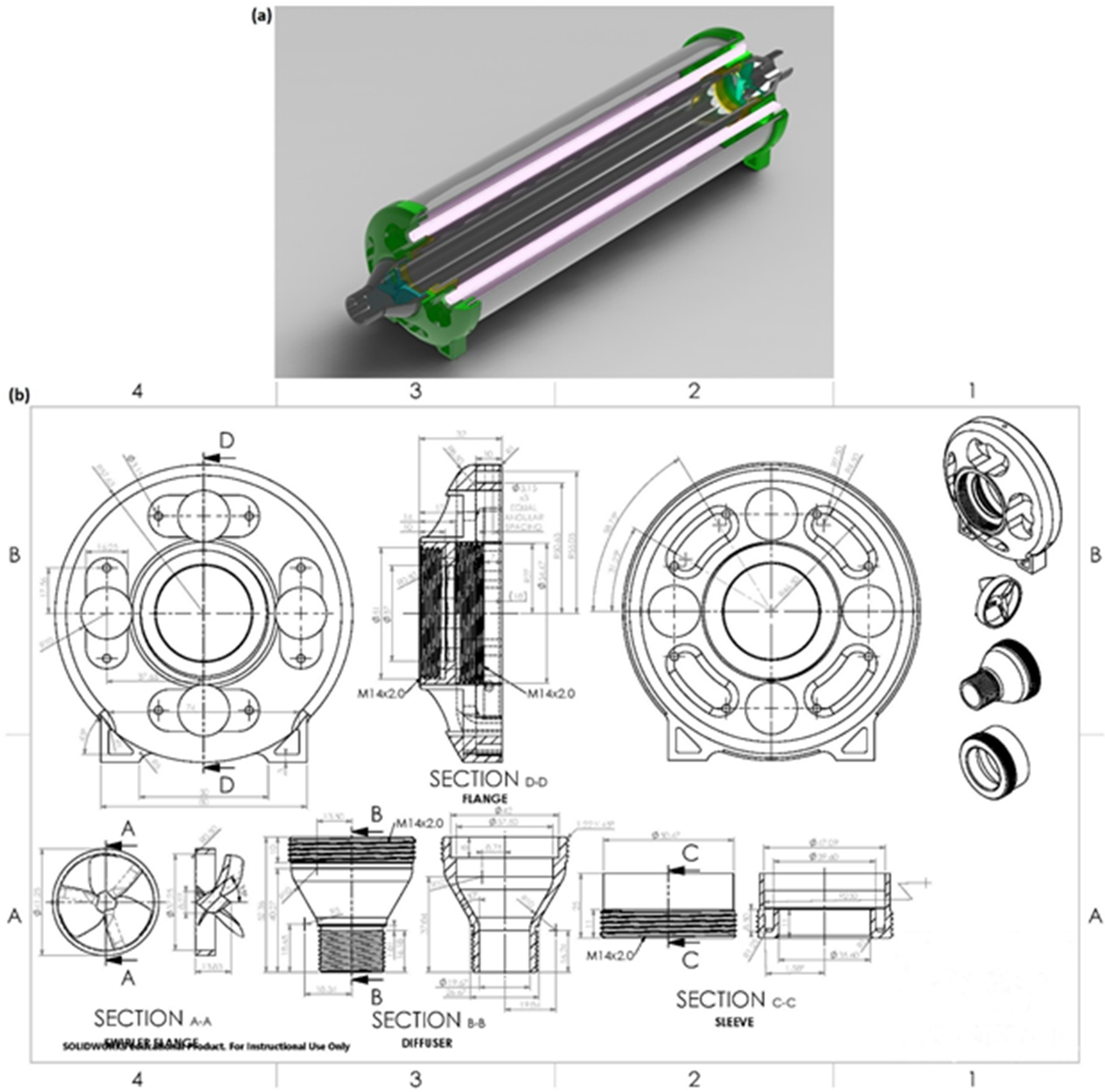
2.1. System Development
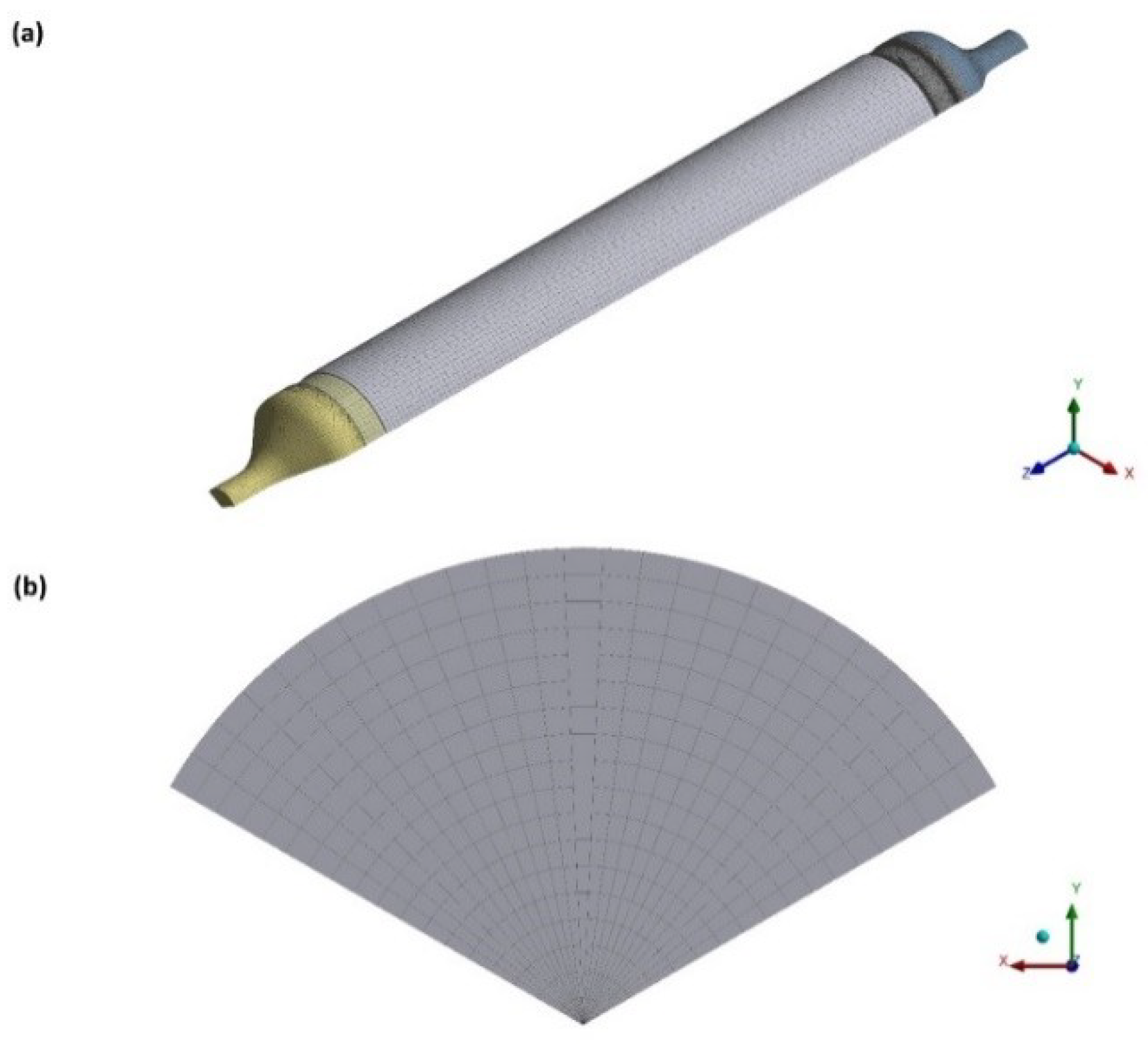
2.2. Computer Modeling
2.3. UV System Setup
2.4. Microbial Propagation
2.5. Germicidal Effectiveness
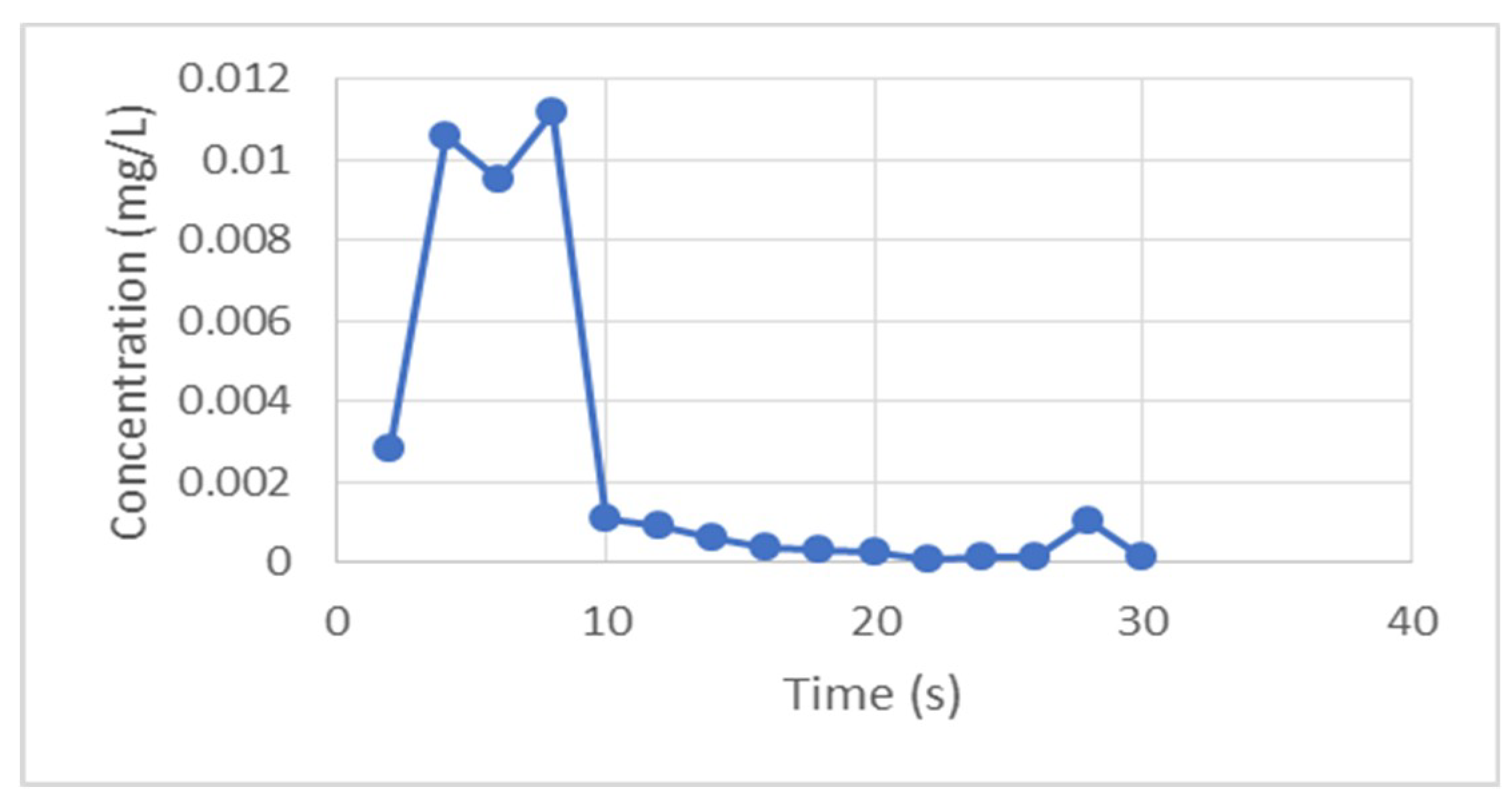
2.6. Hydraulic Tracer Test
2.7. Analysis of Scaling
2.8. Analysis of the Impact of Turbidity
3. Results and Discussion
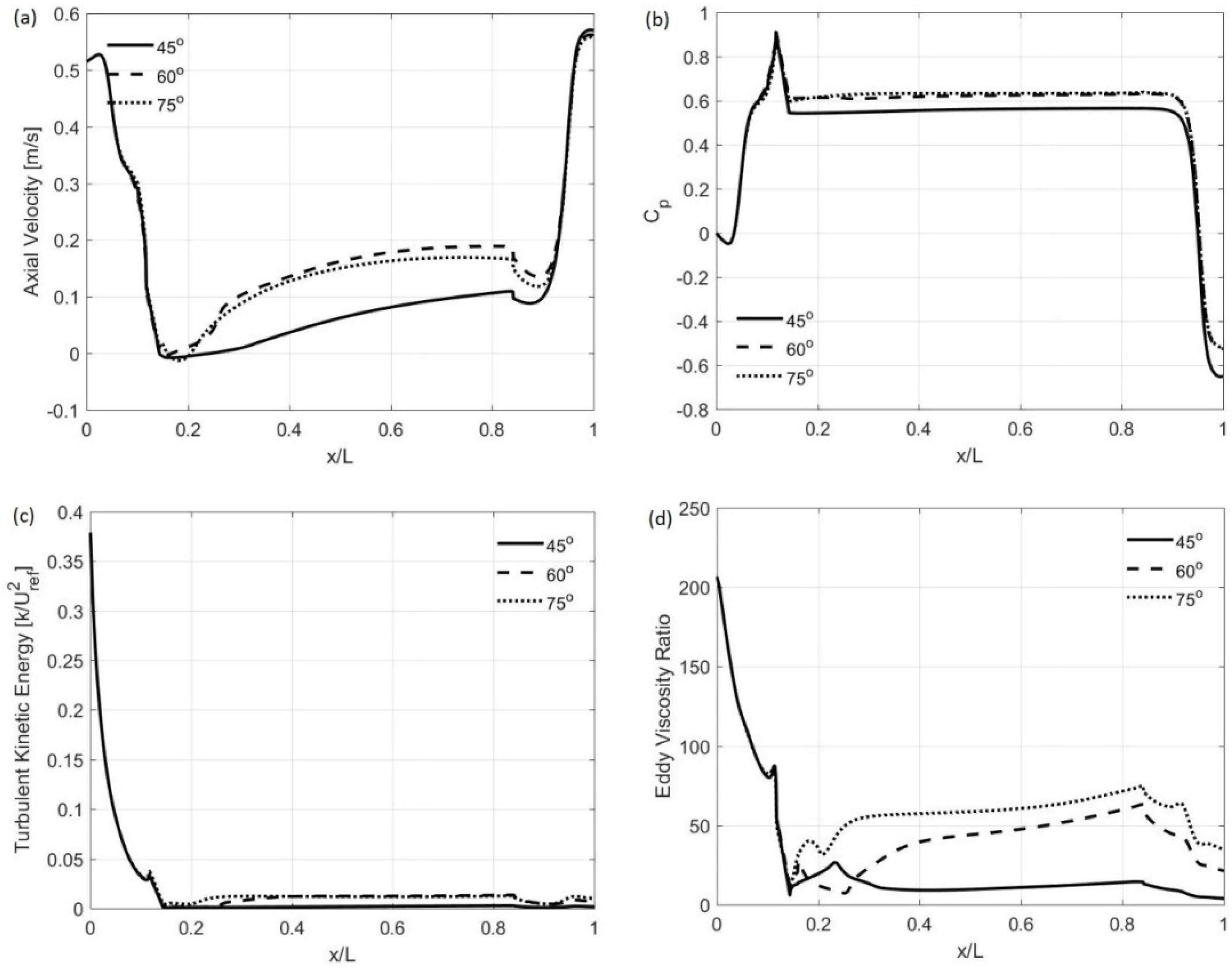
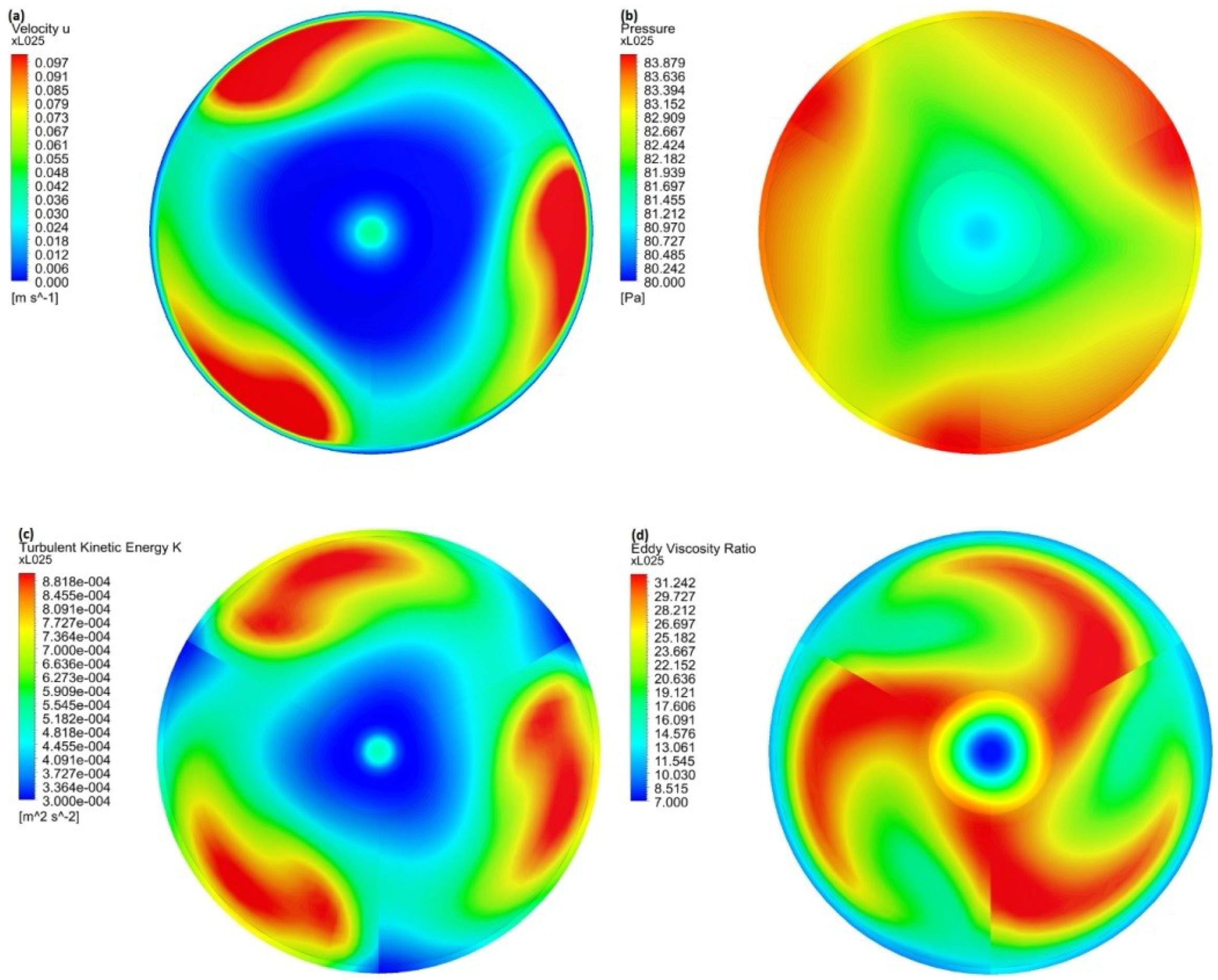
3.1. Computer Simulations
3.2. Hydraulic Tracer Results
3.3. Germicidal Effectiveness
3.4. Analysis of Scaling
3.5. Impact of Turbidity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Berndt, E.C.; Creighton, H.M. Method for irradiating substances with active rays. US Patent 2072417A, 2 March 1934. [Google Scholar]
- Bolton, J.R.; Linden, K.G. Standardization of methods for fluence (UV dose) determination in bench-scale UV experiments. J. Environ. Eng. 2003, 129, 209–215. [Google Scholar] [CrossRef]
- Gates, F.L. A study of the bactericidal action of ultra violet light: III. The absorption of ultra violet light by bacteria. J. Gen. Physiol. 1930, 14, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Linden, K.G.; Shin, G.; Sobsey, M.D. Comparative effectiveness of UV wavelengths for the inactivation of Cryptosporidium parvum oocysts in water. Water Sci. Technol. 2001, 43, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Tchobanoglous, G.; Stensel, H.; Tsuchihashi, R.; Burton, F.; Abu-Orf, M.; Bowden, G.; Pfrang, W. Wastewater Engineering: Treatment and Resource Recovery, 5th ed.; Metcalf & Eddy I AECOM; McGraw-Hill Book Company: New York, NY, USA, 2014. [Google Scholar]
- Oliver, M. Validation of Cleaning Methods for UV Disinfection Systems. Proc. Water Environ. Federation 2002, 1, 564–579. [Google Scholar] [CrossRef]
- Sultan, T. Numerical study of the effects of lamp configuration and reactor wall roughness in an open channel water disinfection UV reactor. Chemosphere 2016, 155, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Wols, B.A.; Hofman, J.A.M.H.; Beerendonk, E.F.; Uijttewaal, W.S.J.; Van Dijk, J.C. A systematic approach for the design of UV reactors using computational fluid dynamics. AIChE J. 2011, 57, 193–207. [Google Scholar] [CrossRef]
- Li, W.; Li, M.; Bolton, J.R.; Qiang, Z. Configuration optimization of UV reactors for water disinfection with computational fluid dynamics: Feasibility of using particle minimum UV dose as a performance indicator. Chem. Eng. J. 2016, 306, 1–8. [Google Scholar] [CrossRef]
- Brownell, S.A.; Chakrabarti, A.R.; Kaser, F.M.; Connelly, L.G.; Peletz, R.L.; Reygadas, F.; Lang, M.J.; Kammen, D.M.; Nelson, K.L. Assessment of a low-cost, point-of-use, ultraviolet water disinfection technology. J. Water Health 2008, 6, 53–65. [Google Scholar] [CrossRef] [PubMed]
- APHA. Standard Methods for the Examination of Water and Wastewater; Rice, E.W., Baird, R.B., Eaton, A.D., Clesceri, L.S., Eds.; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- NWRI. Ultraviolet Disinfection Guidelines for Drinking Water and Water Reuse, 3rd ed.; National Water Research Institute: Fountain Valley, CA, USA, 2012. [Google Scholar]
- Adams, M.H. Bacteriophages; Interscience Publishers: New York, NY, USA, 1959. [Google Scholar]
- Taborek, J.; Aoki, T.; Ritter, R.; Palen, J.; Knudsen, J. Fouling: the major unresolved problem in heat transfer. Chem. Eng. Prog. 1972, 68, 69–78. [Google Scholar]
- Hasson, D.; Bramson, D. Effectiveness of magnetic water treatment in suppressing calcium carbonate scale deposition. Ind. Eng. Chem. Process Design Dev. 1985, 24, 588–592. [Google Scholar] [CrossRef]
- Nessim, Y.; Gehr, R. Fouling Mechanisms in a Laboratory-Scale UV Disinfection System. Water Environ. Res. 2006, 78, 2311–2323. [Google Scholar] [CrossRef] [PubMed]
- Sheikholeslami, R.; Watkinson, A. Scaling of plain and externally finned heat exchanger tubes. J. Heat Trans. 1986, 108, 147–152. [Google Scholar] [CrossRef]
- Lin, L.-S.; Johnston, C.T.; Blatchley, E.R. Inorganic fouling at quartz: water interfaces in ultraviolet photoreactors: I. Chemical characterization. Water Res. 1999, 33, 3321–3329. [Google Scholar] [CrossRef]
- Lin, L.-S.; Johnston, C.T.; Blatchley, E.R. Inorganic fouling at quartz: water interfaces in ultraviolet photoreactors—II. Temporal and spatial distributions. Water Res. 1999, 33, 3330–3338. [Google Scholar] [CrossRef]
- Glaze, W.; Kang, J.-W.; Chapin, D.H. The Chemistry of water treatment processes involving ozone, hydrogen peroxide and ultraviolet radiation. Ozone Sci. Eng. 1987, 9, 335–352. [Google Scholar] [CrossRef]

| Type | Constituent | Concentration (mg/L) |
|---|---|---|
| 1 | Iron, as ferric chloride (Sigma-Aldrich) | 1.0 |
| Magnesium, as magnesium chloride (Sigma-Aldrich) | 200 | |
| Calcium, as calcium chloride (Sigma-Aldrich) | 300 | |
| 2 | Calcium, as calcium chloride (Sigma-Aldrich) | 300 |
| Carbonate, as sodium carbonate (Sigma-Aldrich) | 100 |
| Parameter | Value |
|---|---|
| Hydraulic Residence Time (s) | 1.6 |
| Variance (s2) | 0.86 |
| Standard Deviation (s) | 0.93 |
| t10 (s) | 2.3 |
| t50 (s) | 7.3 |
| t90 (s) | 13.6 |
| Morrill Dispersion Index | 1.4 |
| Volumetric Efficiency (%) | 73% |
| Flow (L/s) | UVT (%) | Log10 Influent MS2 Conc. | Log10 Effluent MS2 Conc. | Log10 Removal of MS2 | UV Dose (mJ/cm2) |
|---|---|---|---|---|---|
| 0.16 | 95 | 8.18 ± 0.10 | 1.60 ± 0.12 | 7.12 ± 0.93 | 215.6 * |
| 0.16 | 70 | 8.60 ± 0.05 | 5.59 ± 0.09 | 3.01 ± 0.09 | 93.9 |
| 0.22 | 95 | 8.18 ± 0.10 | 2.72 ± 0.08 | 5.46 ± 0.08 | 173.3 * |
| 0.22 | 70 | 8.60 ± 0.05 | 6.38 ± 0.13 | 2.22 ± 0.13 | 64.5 |
| Water Type | Number of Lamps | Flow Rate (L/s) | Transmittance Reduction (%) | Change in Alkalinity (%) |
|---|---|---|---|---|
| Feed (Ca+Fe +Mg) | 1 | 0 | 22 | 53 |
| 2 | 0.16 | 10 | 67 | |
| Feed (CaCl2+Na2CO3) | 0 | 0 | 63 | 49 |
| 0 | 0.16 | 12 | 18 | |
| 2 | 0 | 34 | 30 | |
| 2 | 0.16 | 43% | 22% |
| Sample | Log10 Influent E. coli Conc. | Log10 Effluent E. coli Conc. | Log10 Removal of E. coli |
|---|---|---|---|
| Pre-fouling | 7.32 ± 0.04 | 0.35 ± 0.28 | 6.9 ± 0.50 |
| Post-fouling | 6.97 ± 0.58 | −0.11 ± 0.34 | 6.9 ± 0.58 |
| Turbidity (NTU) | Log10 Influent E. coli Conc. | Log10 Effluent E. coli Conc. | Log10 Removal of E. coli |
|---|---|---|---|
| 0.16 ± 0.03 | 7.17 ± 0.12 | 1.45 ± 0.17 | 5.5 ± 0.3 |
| 3.53 ± 0.11 | 7.02 ± 0.16 | 1.55 ± 1.16 | 5.1 ± 1.0 |
| 6.62 ± 0.21 | 7.15 ± 0.12 | 1.24 ± 0.86 | 5.6 ± 1.0 |
| 13.30 ± 0.53 | 6.91 ± 0.42 | 0.35 ± 0.49 | 6.8 ± 0.9 |
| 17.83 ± 0.32 | 6.93 ± 0.06 | 1.80 ± 0.21 | 5.1 ± 0.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Younis, B.A.; Mahoney, L.; Palomo, N. A Novel System for Water Disinfection with UV Radiation. Water 2018, 10, 1275. https://doi.org/10.3390/w10091275
Younis BA, Mahoney L, Palomo N. A Novel System for Water Disinfection with UV Radiation. Water. 2018; 10(9):1275. https://doi.org/10.3390/w10091275
Chicago/Turabian StyleYounis, Bassam A., Laura Mahoney, and Nicholas Palomo. 2018. "A Novel System for Water Disinfection with UV Radiation" Water 10, no. 9: 1275. https://doi.org/10.3390/w10091275
APA StyleYounis, B. A., Mahoney, L., & Palomo, N. (2018). A Novel System for Water Disinfection with UV Radiation. Water, 10(9), 1275. https://doi.org/10.3390/w10091275




