Quantifying Averted Disability-Adjusted Life Years as a Performance Indicator for Water Quality Interventions: A Review of Current Methodologies and Challenges
Abstract
1. Background
2. Incentivizing Health Gains over Infrastructure Coverage
2.1. Current Delivery Approaches
2.2. Pay-for-Performance Financing
3. Measuring Disease Burden as a Performance Indicator
3.1. Health Impact Performance Indicators
3.2. Estimating Averted Disability-Adjusted Life Years
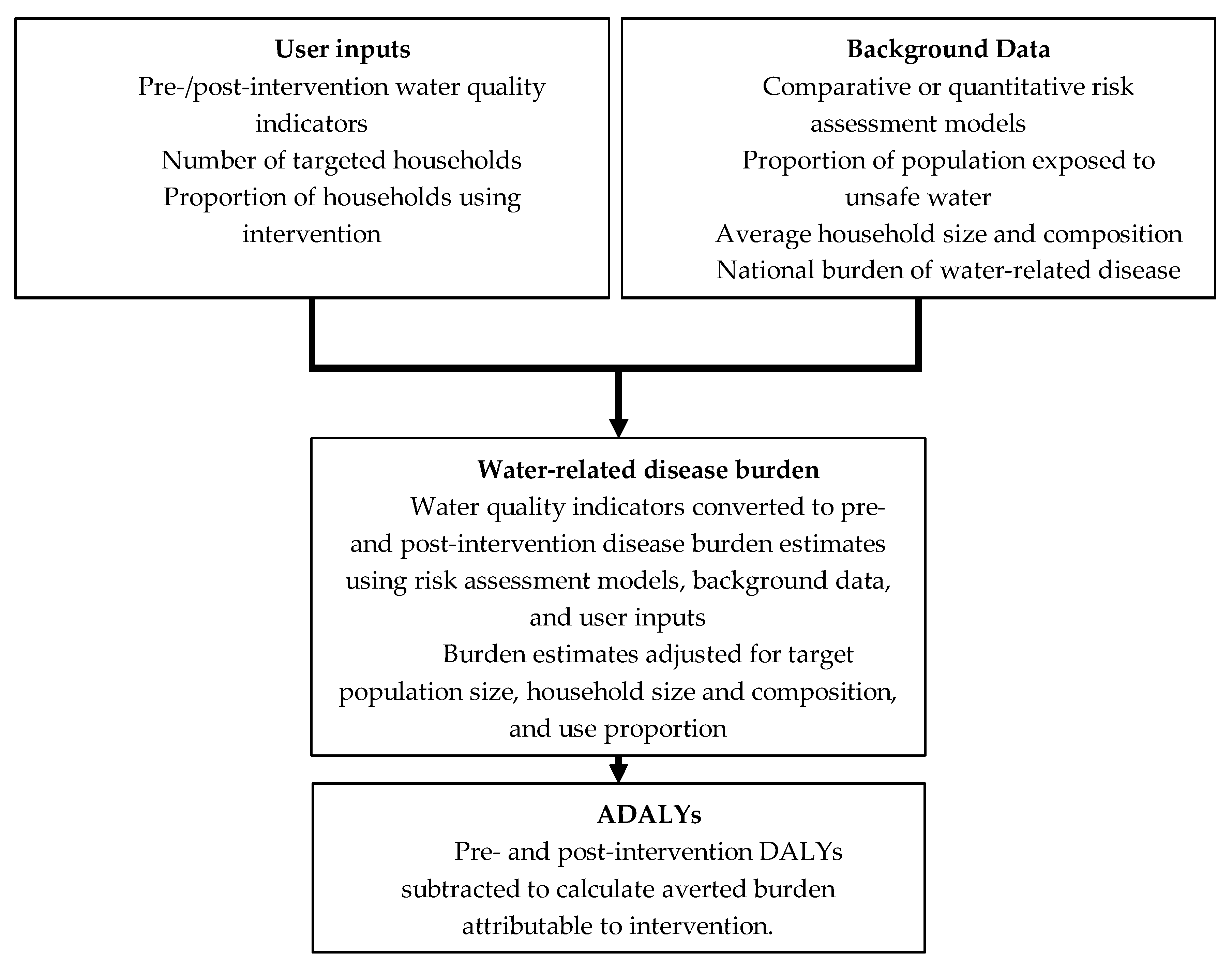
3.3. Quantifying ADALYs through Comparative Risk Assessment
3.4. Quantifying ADALYs through Quantitative Microbial Risk Assessment
3.5. Challenges of Exposure Assessment
3.6. Challenges of Service Delivery Measurement
3.7. Evaluating Health Impacts beyond Diarrheal Disease
4. Discussion
Author Contributions
Conflicts of Interest
References
- Bartram, J.; Cairncross, S. Hygiene, sanitation, and water: Forgotten foundations of health. PLoS Med. 2010, 7, e1000367. [Google Scholar] [CrossRef] [PubMed]
- Ngure, F.M.; Reid, B.M.; Humphrey, J.H.; Mbuya, M.N.; Pelto, G.; Stoltzfus, R.J. Water, sanitation, and hygiene (WASH), environmental enteropathy, nutrition, and early child development: Making the links. Ann. N. Y. Acad. Sci. 2014, 1308, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Stelmach, R.D.; Clasen, T. Household water quantity and health: A systematic review. Int. J. Environ. Res. Public Health 2015, 12, 5954–5974. [Google Scholar] [CrossRef] [PubMed]
- Rabie, T.; Curtis, V. Handwashing and risk of respiratory infections: A quantitative systematic review. Trop. Med. Int. Health 2006, 11, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Pickering, A.J.; Davis, J. Freshwater availability and water fetching distance affect child health in sub-Saharan Africa. Environ. Sci. Technol. 2012, 46, 2391–2397. [Google Scholar] [CrossRef] [PubMed]
- Hemson, D. ‘The toughest of chores’: Policy and practice in children collecting water in South Africa. Policy Futures Educ. 2007, 5, 315–326. [Google Scholar] [CrossRef]
- Graham, J.P.; Hirai, M.; Kim, S.S. An analysis of water collection labor among women and children in 24 sub-Saharan African countries. PLoS ONE 2016, 11, e0155981. [Google Scholar] [CrossRef] [PubMed]
- WHO/UNICEF Joint Monitoring Programme for Water Supply and Sanitation. Progress on Sanitation and Drinking-Water: 2015 Update; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Bartram, J.; Brocklehurst, C.; Fisher, M.B.; Luyendijk, R.; Hossain, R.; Wardlaw, T.; Gordon, B. Global monitoring of water supply and sanitation: History, methods and future challenges. Int. J. Environ. Res. Public Health 2014, 11, 8137–8165. [Google Scholar] [CrossRef] [PubMed]
- WHO/UNICEF. WHO/UNICEF Joint Monitoring Programme (JMP) for Water Supply and Sanitation. Available online: http://www.wssinfo.org/ (accessed on 5 January 2017).
- WHO/UNICEF Joint Monitoring Programme for Water Supply and Sanitation. Progress on Sanitation and Drinking-Water: 2012 Update; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Cumming, O.; Elliott, M.; Overbo, A.; Bartram, J. Does global progress on sanitation really lag behind water? An analysis of global progress on community- and household-level access to safe water and sanitation. PLoS ONE 2014, 9, e114699. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Schwab, K.J. Deficiencies in drinking water distribution systems in developing countries. J. Water Health 2005, 3, 109–127. [Google Scholar] [CrossRef] [PubMed]
- Foster, T. Predictors of sustainability for community-managed handpumps in sub-Saharan Africa: Evidence from Liberia, sierra Leone, and Uganda. Environ. Sci. Technol. 2013, 47, 12037–12046. [Google Scholar] [CrossRef] [PubMed]
- Yuthas, K.; Thomas, E. Performance over promises. In Broken Pumps and Promises: Incentivizing Impact in Environmental Health; Thomas, E., Ed.; Springer International Publishing: New York, NY, USA, 2016; pp. 5–17. [Google Scholar]
- Rural Water Supply Network. Sustainable Rural Water Supplies; Rural Water Supply Network: St. Gallen, Switzerland, 2012. [Google Scholar]
- Parry-Jones, S.; Reed, R.; Skinner, B.H. Sustainable Handpump Projects in Africa: A Literature Review; Water, Engineering, and Development Center, Loughborough University: Leicestershire, UK, 2001. [Google Scholar]
- Hope, R.; Rouse, M. Risks and responses to universal drinking water security. Philos. Trans. A Math. Phys. Eng. Sci. 2013, 371, 20120417. [Google Scholar] [CrossRef] [PubMed]
- Crocker, J.; Bartram, J. Comparison and cost analysis of drinking water quality monitoring requirements versus practice in seven developing countries. Int. J. Environ. Res. Public Health 2014, 11, 7333–7346. [Google Scholar] [CrossRef] [PubMed]
- Harvey, P.A.; Reed, R.A. Community-managed water supplies in Africa: Sustainable or dispensable? Community Dev. J. 2007, 42, 365–378. [Google Scholar] [CrossRef]
- Thomas, E. Introduction. In Broken Pumps and Promises: Incentivizing Impact in Environmental Health; Thomas, E., Ed.; Springer International Publishing: New York, NY, USA, 2016; pp. 1–4. [Google Scholar]
- Marks, S.J.; Komives, K.; Davis, J. Community participation and water supply sustainability: Evidence from handpump projects in rural Ghana. J. Plan. Educ. Res. 2014, 34, 276–286. [Google Scholar] [CrossRef]
- Komives, K.; Akanbang, B.; Thorsetn, R.; Tuffuour, B.; Wakeman, W.; Labri, E.; Bakalian, A.; Whittington, D. Post-Construction Support and the Sustainability or Fural Water Projects in Ghana. In Proceedings of the 33rd 2008 WEDC International Conference, Accra, Ghana, 7–11 April 2008; The Water, Engineering, and Development Center: Accra, Ghana, 2008; pp. 147–150. [Google Scholar]
- Whittington, D.; Davis, J.; Prokopy, L.; Komives, K.; Thorsten, R.; Lukacs, H.; Bakalian, A.; Wakeman, W. How well is the demand-driven, community management model for rural water supply systems doing? Evidence from Bolivia, Peru and Ghana. Water Policy 2009, 11, 696–718. [Google Scholar] [CrossRef]
- Montgomery, M.A.; Bartram, J.; Elimelech, M. Increasing functional sustainability of water and sanitation supplies in rural sub-Saharan Africa. Environ. Eng. Sci. 2009, 26, 1017–1023. [Google Scholar] [CrossRef]
- Rodriguez, D.; Suardi, M.; Ham, M.; Mimmi, L.; Goksu, A. Applying Results-Based Financing in Water Investments; World Bank: Washington, DC, USA, 2014. [Google Scholar]
- Meessen, B.; Soucat, A.; Sekabaraga, C. Performance-based financing: Just a donor fad or a catalyst towards comprehensive health-care reform? Bull. World Health Org. 2011, 89, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Department for International Development (DFID). DFID Evaluation Framework for Payment by Results; DFID: London, UK, 2015. [Google Scholar]
- Project, A.-T. The PBF Handbook: Designing and Implementing Effective Performance-Based Financing Programs; Management Sciences for Health: Cambridge, MA, USA, 2011. [Google Scholar]
- Eldridge, C.; Palmer, N. Performance-based payment: Some reflections on the discourse, evidence and unanswered questions. Health Policy Plan. 2009, 24, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Oxman, A.D.; Fretheim, A. An Overview of Research on the Effects of Results-Based Financing; Norwegian, Knowledge Centre for the Health Services: Oslo, Norway, 2008. [Google Scholar]
- Daniell, K.A.; White, I.; Ferrand, N.; Ribarova, I.; Coad, P.; Rougier, J.E.; Hare, M.; Jones, N.; Popov, A.; Popova, D.; Rollin, P.; et al. Co-engineering participatory water management processes: Theory and insights from Australian and Bulgarian interventions. Ecol. Soc. 2010, 15, 11. [Google Scholar] [CrossRef]
- Oxman, A.D.; Fretheim, A. Can paying for results help to achieve the millennium development goals? A critical review of selected evaluations of results-based financing. J. Evid. Based Med. 2009, 2, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Review of Results-Based Financing Schemes in WASH: Report to Bill & Melinda Gates Foundation. 2015. Available online: https://www.ircwash.org/sites/default/files/rbf_in_wash_final_report_full_jan_2015.pdf (accessed on 21 May 2018).
- Program for Results Financing. Available online: http://www.worldbank.org/en/programs/program-for-results-financing (accessed on 16 April 2017).
- World Bank. Socialist Republic of Vietnam: Results-Based Rural Water Supply and Sanitation under the National Target Program: Impact Evaluation; World Bank: Washington, DC, USA, 2016. [Google Scholar]
- World Bank. Vietnam Red River Delta Rural Water Supply and Sanitation Project; World Bank: Washington, DC, USA, 2015. [Google Scholar]
- Hurrell, A.; Colin, J.; Lucrezia, T. Evaluation of the Wash Results Programme: Mide-Term Evaluation Report; UK Department for International Development: London, UK, 2017. [Google Scholar]
- Chase, C.; Coville, A. Trade-offs and risks in results-based approaches. In Broken Pumps and Promises: Incentivizing Impact in Environmental Health; Thomas, E., Ed.; Springer International Publishing: New York, NY, USA, 2016; pp. 19–29. [Google Scholar]
- Kohn, L.B.; Bruysten, S. Mobilizing commercial investment for social good: The social success note. In Broken Pumps and Promises: Incentivizing Impact in Environmental Health; Thomas, E., Ed.; Springer International Publishing: New York, NY, USA, 2016; pp. 231–238. [Google Scholar]
- Koehler, J.; Thomson, P.; Hope, R. Pump-priming payments for sustainable water services in rural Africa. World Dev. 2015, 74, 397–411. [Google Scholar] [CrossRef]
- Oxford/RFL. From Rights to Results in Rural Water Services—Evidence from Kyuso, Kenya; Oxford/RFL: Oxford, UK, 2014. [Google Scholar]
- Hodge, J.M.; Clasen, T.F. Carbon financing of household water treatment: Background, operation and recommendations to improve potential for health gains. Environ. Sci. Technol. 2014, 48, 12509–12515. [Google Scholar] [CrossRef] [PubMed]
- World Bank. Program. Appraisal Document on a Proposed Credit to the Socialist Repbulic of Vietnam for the Results-Based Scaling up Rural Sanitation and Water Supply Program; World Bank: Washington, DC, USA, 2015. [Google Scholar]
- AusAID. Midterm Review and Verification Survey for Water and Sanitation Hibah; AusAID: Canberra, Australia, 2011. [Google Scholar]
- Pillarisetti, A.; Sumi, M.; Smith, K. Hapit, the household air pollution intervention tool, to evaluate the health benefits and cost-effectiveness of clean cooking interventions. In Broken Pumps and Promises: Incentivizing Impact in Environmental Health; Thomas, E., Ed.; Springer International Publishing: New York, NY, USA, 2016; pp. 147–169. [Google Scholar]
- Anenberg, S.; Kinney, P.; Newcombe, K.; Talyan, V.; Goyal, A.; Hewlett, O. Gold Standard Methodology to Estimate and Verify Averted Mortality and Disability Adjusted Life Years (Adalys) from Cleaner Household Air; Gold Standard Foundation: Berkeley, CA, USA, 2017. [Google Scholar]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the global burden of disease study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Mathers, C.; Fat, D.M.; Boerma, J.T. The Global Burden of Disease: 2004 Update; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet 2013, 380, 2224–2260. [Google Scholar] [CrossRef]
- Forouzanfar, M.H.; Alexander, L.; Anderson, H.R.; Bachman, V.F.; Biryukov, S.; Brauer, M.; Burnett, R.; Casey, D.; Coates, M.M.; Cohen, A. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks in 188 countries, 1990–2013: A systematic analysis for the global burden of disease study 2013. Lancet 2015, 386, 2287–2323. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality, 4 ed.; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Haas, C.N.; Rose, J.B.; Gerba, C.P. Quantitative Microbial Risk Assessment; John Wiley & Sons: New York, NY, USA, 1999. [Google Scholar]
- Brown, J.; Clasen, T. High adherence is necessary to realize health gains from water quality interventions. PLoS ONE 2012, 7, e36735. [Google Scholar] [CrossRef] [PubMed]
- Pruss-Ustun, A.; Bartram, J.; Clasen, T.; Colford, J.M., Jr.; Cumming, O.; Curtis, V.; Bonjour, S.; Dangour, A.D.; De France, J.; Fewtrell, L.; et al. Burden of disease from inadequate water, sanitation and hygiene in low- and middle-income settings: A retrospective analysis of data from 145 countries. Trop. Med. Int. Health 2014, 19, 894–905. [Google Scholar] [CrossRef] [PubMed]
- Pruss-Ustun, A.; Bonjour, S.; Corvalan, C. The impact of the environment on health by country: A meta-synthesis. Environ. Health 2008, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Pruss-Ustun, A.; Corvalan, C. How much disease burden can be prevented by environmental interventions? Epidemiology 2007, 18, 167–178. [Google Scholar] [PubMed]
- Wolf, J.; Pruss-Ustun, A.; Cumming, O.; Bartram, J.; Bonjour, S.; Cairncross, S.; Clasen, T.; Colford, J.M., Jr.; Curtis, V.; De France, J.; et al. Assessing the impact of drinking water and sanitation on diarrhoeal disease in low- and middle-income settings: Systematic review and meta-regression. Trop. Med. Int. Health 2014, 19, 928–942. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, L.; Kaufmann, R.B.; Kay, D.; Enanoria, W.; Haller, L.; Colford, J.M., Jr. Water, sanitation, and hygiene interventions to reduce diarrhoea in less developed countries: A systematic review and meta-analysis. Lancet Infect. Dis. 2005, 5, 42–52. [Google Scholar] [CrossRef]
- Waddington, H.; Snilstveit, B. Effectiveness and sustainability of water, sanitation, and hygiene interventions in combating diarrhoea. J. Dev. Effect. 2009, 1, 295–335. [Google Scholar] [CrossRef]
- Hunter, P.R. Household water treatment in developing countries: Comparing different intervention types using meta-regression. Environ. Sci. Technol. 2009, 43, 8991–8997. [Google Scholar] [CrossRef] [PubMed]
- Clasen, T.F.; Alexander, K.T.; Sinclair, D.; Boisson, S.; Peletz, R.; Chang, H.H.; Majorin, F.; Cairncross, S. Interventions to improve water quality for preventing diarrhoea. Cochrane Database Syst. Rev. 2015, 19, CD004794. [Google Scholar] [CrossRef] [PubMed]
- Engell, R.E.; Lim, S.S. Does clean water matter? An updated meta-analysis of water supply and sanitation interventions and diarrhoeal diseases. Lancet 2013, 381, S44. [Google Scholar] [CrossRef]
- Bain, R.; Cronk, R.; Wright, J.; Yang, H.; Slaymaker, T.; Bartram, J. Fecal contamination of drinking-water in low- and middle-income countries: A systematic review and meta-analysis. PLoS Med. 2014, 11, e1001644. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, W.P.; Cairncross, S. Household water treatment in poor populations: Is there enough evidence for scaling up now? Environ. Sci. Technol. 2009, 43, 986–992. [Google Scholar] [CrossRef] [PubMed]
- Clasen, T.; Pruss-Ustun, A.; Mathers, C.D.; Cumming, O.; Cairncross, S.; Colford, J.M., Jr. Estimating the impact of unsafe water, sanitation and hygiene on the global burden of disease: Evolving and alternative methods. Trop. Med. Int. Health 2014, 19, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Clasen, T.; Boisson, S. Assessing the health impact of water quality interventions in low-income settings: Concerns associated with blinded trials and the need for objective outcomes. Environ. Health Perspect. 2016, 124, 886–889. [Google Scholar] [CrossRef] [PubMed]
- Savovic, J.; Jones, H.; Altman, D.; Harris, R.; Juni, P.; Pildal, J.; Als-Nielsen, B.; Balk, E.; Gluud, C.; Gluud, L.; et al. Influence of reported study design characteristics on intervention effect estimates from randomised controlled trials: Combined analysis of meta-epidemiological studies. Health Technol. Assess. 2012, 16, 1–82. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Proum, S.; Sobsey, M.D. Sustained use of a household-scale water filtration device in rural cambodia. J. Water Health 2009, 7, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Boisson, S.; Kiyombo, M.; Sthreshley, L.; Tumba, S.; Makambo, J.; Clasen, T. Field assessment of a novel household-based water filtration device: A randomised, placebo-controlled trial in the democratic Republic of Congo. PLoS ONE 2010, 5, e12613. [Google Scholar] [CrossRef] [PubMed]
- Boisson, S.; Schmidt, W.P.; Berhanu, T.; Gezahegn, H.; Clasen, T. Randomized controlled trial in rural Ethiopia to assess a portable water treatment device. Environ. Sci. Technol. 2009, 43, 5934–5939. [Google Scholar] [CrossRef] [PubMed]
- Boisson, S.; Stevenson, M.; Shapiro, L.; Kumar, V.; Singh, L.P.; Ward, D.; Clasen, T. Effect of household-based drinking water chlorination on diarrhoea among children under five in Orissa, India: A double-blind randomised placebo-controlled trial. PLoS Med. 2013, 10, e1001497. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Sobsey, M.D.; Loomis, D. Local drinking water filters reduce diarrheal disease in Cambodia: A randomized, controlled trial of the ceramic water purifier. Am. J. Trop. Med. Hyg. 2008, 79, 394–400. [Google Scholar] [PubMed]
- Colindres, R.E.; Jain, S.; Bowen, A.; Mintz, E.; Domond, P. After the flood: An evaluation of in-home drinking water treatment with combined flocculent-disinfectant following tropical storm Jeanne—Gonaives, Haiti, 2004. J. Water Health 2007, 5, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Luby, S.; Agboatwalla, M.; Raza, A.; Sobel, J.; Mintz, E.; Baier, K.; Rahbar, M.; Qureshi, S.; Hassan, R.; Ghouri, F.; et al. A low-cost intervention for cleaner drinking water in Karachi, Pakistan. Int. J. Infect. Dis. 2001, 5, 144–150. [Google Scholar] [CrossRef]
- Mausezahl, D.; Christen, A.; Pacheco, G.D.; Tellez, F.A.; Iriarte, M.; Zapata, M.E.; Cevallos, M.; Hattendorf, J.; Cattaneo, M.D.; Arnold, B.; et al. Solar drinking water disinfection (Sodis) to reduce childhood diarrhoea in rural Bolivia: A cluster-randomized, controlled trial. PLoS Med. 2009, 6, e1000125. [Google Scholar] [CrossRef] [PubMed]
- Mong, Y.; Kaiser, R.; Ibrahim, D.; Rasoatiana; Razafimbololona, L.; Quick, R.E. Impact of the safe water system on water quality in cyclone-affected communities in Madagascar. Am. J. Public Health 2001, 91, 1577–1579. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations; World Health Organization. Hazard. Characterization for Pathogens in Food and Water: Guidelines; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Howard, G.; Pedley, S.; Tibatemwa, S. Quantitative microbial risk assessment to estimate health risks attributable to water supply: Can the technique be applied in developing countries with limited data? J. Water Health 2006, 4, 49–65. [Google Scholar] [CrossRef] [PubMed]
- Kotloff, K.L.; Nataro, J.P.; Blackwelder, W.C.; Nasrin, D.; Farag, T.H.; Panchalingam, S.; Wu, Y.; Sow, S.O.; Sur, D.; Breiman, R.F.; et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the global enteric multicenter study, gems): A prospective, case-control study. Lancet 2013, 382, 209–222. [Google Scholar] [CrossRef]
- Casadevall, A.; Pirofski, L.A. Host-pathogen interactions: Redefining the basic concepts of virulence and pathogenicity. Infect. Immun. 1999, 67, 3703–3713. [Google Scholar] [PubMed]
- Nwachuku, N.; Gerba, C.P. Microbial risk assessment: Don’t forget the children. Curr. Opin. Microbiol. 2004, 7, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L.; Rudan, I.; Liu, L.; Nair, H.; Theodoratou, E.; Bhutta, Z.A.; O’Brien, K.L.; Campbell, H.; Black, R.E. Global burden of childhood pneumonia and diarrhoea. Lancet 2013, 381, 1405–1416. [Google Scholar] [CrossRef]
- Gerba, C.P.; Rose, J.B.; Haas, C.N. Sensitive populations: Who is at the greatest risk? Int. J. Food Microbiol. 1996, 30, 113–123. [Google Scholar] [CrossRef]
- Hunter, P.R.; Payment, P.; Ashbolt, N.; Bartram, J. Assessment of risk. In Assessing Microbiological Safety of Drinking Water: Improving Approaches and Methods; Ronchi, E., Bartram, J., Eds.; Organization for Economic Co-operation and Development: Paris, France, 2003; pp. 79–109. [Google Scholar]
- Okhuysen, P.C.; Chappell, C.L.; Crabb, J.H.; Sterling, C.R.; DuPont, H.L. Virulence of three distinct cryptosporidium parvum isolates for healthy adults. J. Infect. Dis. 1999, 180, 1275–1281. [Google Scholar] [CrossRef] [PubMed]
- Kothary, M.H.; Babu, U.S. Infective dose of foodborne pathogens in volunteers: A review. J. Food Saf. 2001, 21, 49–68. [Google Scholar] [CrossRef]
- Mara, D.; Lane, J.; Scott, B.; Trouba, D. Sanitation and health. PLoS Med. 2010, 7, e1000363. [Google Scholar] [CrossRef] [PubMed]
- Wagner, E.G.; Lanoix, J.N. Excreta Disposal for Rural Areas and Small Communities; World Health Organization: Geneva, Switzerland, 1958. [Google Scholar]
- Eisenberg, J.N.; Scott, J.C.; Porco, T. Integrating disease control strategies: Balancing water sanitation and hygiene interventions to reduce diarrheal disease burden. Am. J. Public Health 2007, 97, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Barstow, C.K.; Nagel, C.L.; Clasen, T.F.; Thomas, E.A. Process evaluation and assessment of use of a large scale water filter and cookstove program in Rwanda. BMC Public Health 2016, 16, 584. [Google Scholar] [CrossRef] [PubMed]
- Barstow, C.K.; Ngabo, F.; Rosa, G.; Majorin, F.; Boisson, S.; Clasen, T.; Thomas, E.A. Designing and piloting a program to provide water filters and improved cookstoves in Rwanda. PLoS ONE 2014, 9, e92403. [Google Scholar] [CrossRef] [PubMed]
- Rosa, G.; Majorin, F.; Boisson, S.; Barstow, C.; Johnson, M.; Kirby, M.; Ngabo, F.; Thomas, E.; Clasen, T. Assessing the impact of water filters and improved cook stoves on drinking water quality and household air pollution: A randomised controlled trial in Rwanda. PLoS ONE 2014, 9, e91011. [Google Scholar] [CrossRef] [PubMed]
- Clark, P.A.; Pinedo, C.A.; Fadus, M.; Capuzzi, S. Slow-sand water filter: Design, implementation, accessibility and sustainability in developing countries. Med. Sci. Monit. 2012, 18, RA105–RA117. [Google Scholar] [CrossRef] [PubMed]
- Misati, A.G.; Ogendi, G.; Peletz, R.; Khush, R.; Kumpel, E. Can sanitary surveys replace water quality testing? Evidence from Kisii, Kenya. Int. J. Environ. Res. Public Health 2017, 14, 152. [Google Scholar] [CrossRef] [PubMed]
- Snoad, C.; Nagel, C.; Bhattacharya, A.; Thomas, E. The effectiveness of sanitary inspections as a risk assessment tool for thermotolerant coliform bacteria contamination of rural drinking water: A review of data from west Bengal, India. Am. J. Trop. Med. Hyg. 2017, 96, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.; Andres, L.A.; Borja-Vega, C.; Sturzenegger, G. Innovations in Wash Impact Measures: Water and Sanitation Measurement Technologies and Practices to Inform the Sustainable Development Goals; World Bank: Washington, DC, USA, 2017. [Google Scholar]
- Thomas, E.A.; Zumr, Z.; Graf, J.; Wick, C.A.; McCellan, J.H.; Imam, Z.; Barstow, C.; Spiller, K.; Fleming, M. Remotely accessible instrumented monitoring of global development programs: Technology development and validation. Sustainability (Basel) 2013, 5, 3288–3301. [Google Scholar] [CrossRef]
- Howard, G.; Bartram, J. Domestic Water Quality, Service Level, and Health; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Sorenson, S.B.; Morssink, C.; Campos, P.A. Safe access to safe water in low income countries: Water fetching in current times. Soc. Sci. Med. 2011, 72, 1522–1526. [Google Scholar] [CrossRef] [PubMed]
- Whittington, D.; Mu, X.; Roche, R. Calculating the value of time spent collecting water: Some estimates for Ukunda, Kenya. World Dev. 1990, 18, 269–280. [Google Scholar] [CrossRef]
- Independent Commission for Aid Impact. Assessing Dfid’s Results in Water, Sanitation, and Hygiene: An Impact Review; Independent Commission for Aid Impact: London, UK, 2016. [Google Scholar]
- World Health Organization. Safely Managed Drinking Water: Thematic Report on Drinking Water 2017; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Yu, W.; Wardrop, N.A.; Bain, R.E.; Lin, Y.; Zhang, C.; Wright, J.A. A global perspective on drinking-water and sanitation classification: An evaluation of census content. PLoS ONE 2016, 11, e0151645. [Google Scholar] [CrossRef] [PubMed]
| Program | Objective(s) | Performance Indicators | Payment Mechanism |
|---|---|---|---|
| World Bank-funded Results-Based Rural Water and Sanitation Under The National Target Program in the Red River Delta of Vietnam [36,44] | To improve hygiene behavior, as well as increase and sustain access to rural sanitation and water supply in rural areas in the participating provinces |
| Payments disbursed annually as a proportion of the indicator target achieved, with independent verification of indicators |
| United Kingdom Department for International Development-funded Water, Sanitation, and Hygiene Results Programme in 12 countries in sub-Saharan Africa and South Asia [38] | To bring equitable and sustainable water and sanitation services and hygiene practices to 4.5 million people in 12 countries, and thus to improve health by reducing diarrhea morbidity and child mortality |
| Payments disbursed annually as a proportion of the indicator target achieved, with the independent verification of indicators |
| Australian Department for Foreign Affairs and Trade-funded Water and Sanitation Hibah in Indonesia [45] | To increase local government investment in water infrastructure, with an emphasis on low-income populations, and to improve the governance of the water sector at the local government level by increasing their accountability to adhere to an agreed water investment program and a level of incremental improvements to services |
| Local governments requested audits of eligible connections from central government, funding disbursed to local government per eligible connection upon verification |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anderson, D.M.; Thomas, E.A.; Clasen, T.F. Quantifying Averted Disability-Adjusted Life Years as a Performance Indicator for Water Quality Interventions: A Review of Current Methodologies and Challenges. Water 2018, 10, 744. https://doi.org/10.3390/w10060744
Anderson DM, Thomas EA, Clasen TF. Quantifying Averted Disability-Adjusted Life Years as a Performance Indicator for Water Quality Interventions: A Review of Current Methodologies and Challenges. Water. 2018; 10(6):744. https://doi.org/10.3390/w10060744
Chicago/Turabian StyleAnderson, Darcy M., Evan A. Thomas, and Thomas F. Clasen. 2018. "Quantifying Averted Disability-Adjusted Life Years as a Performance Indicator for Water Quality Interventions: A Review of Current Methodologies and Challenges" Water 10, no. 6: 744. https://doi.org/10.3390/w10060744
APA StyleAnderson, D. M., Thomas, E. A., & Clasen, T. F. (2018). Quantifying Averted Disability-Adjusted Life Years as a Performance Indicator for Water Quality Interventions: A Review of Current Methodologies and Challenges. Water, 10(6), 744. https://doi.org/10.3390/w10060744






