Plant Harvesting Impacts on Soil Water Patterns and Phenology for Shrub-encroached Grassland
Abstract
1. Introduction
2. Field Experiments and Methods
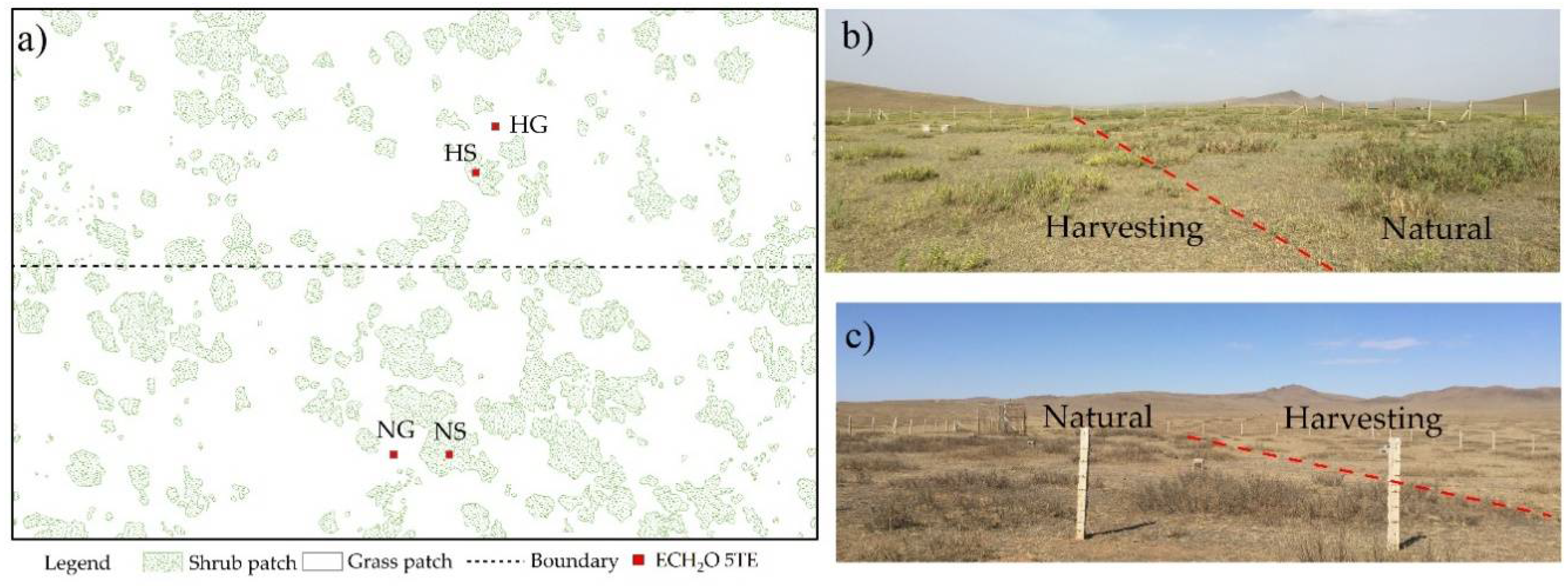
2.1. Study Area
2.2. Observation of Soil Water Dynamics
2.3. Shrub Pattern Analysis and Vegetation Investigation
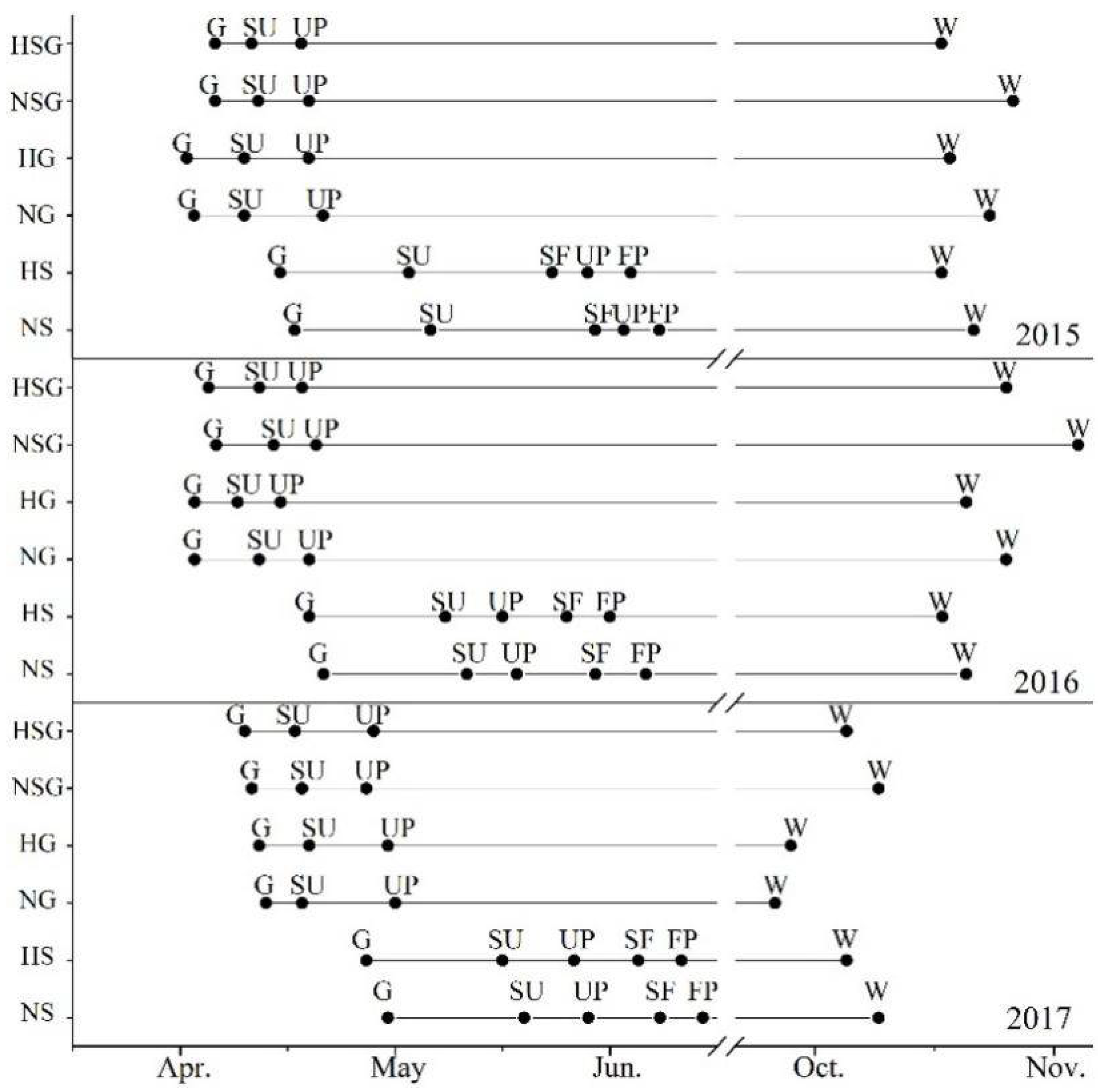
2.4. Plants Phenology Observation and Physiological Experiments
3. Results
3.1. Landscape Patterns and Plant Characteristics Change after Harvesting
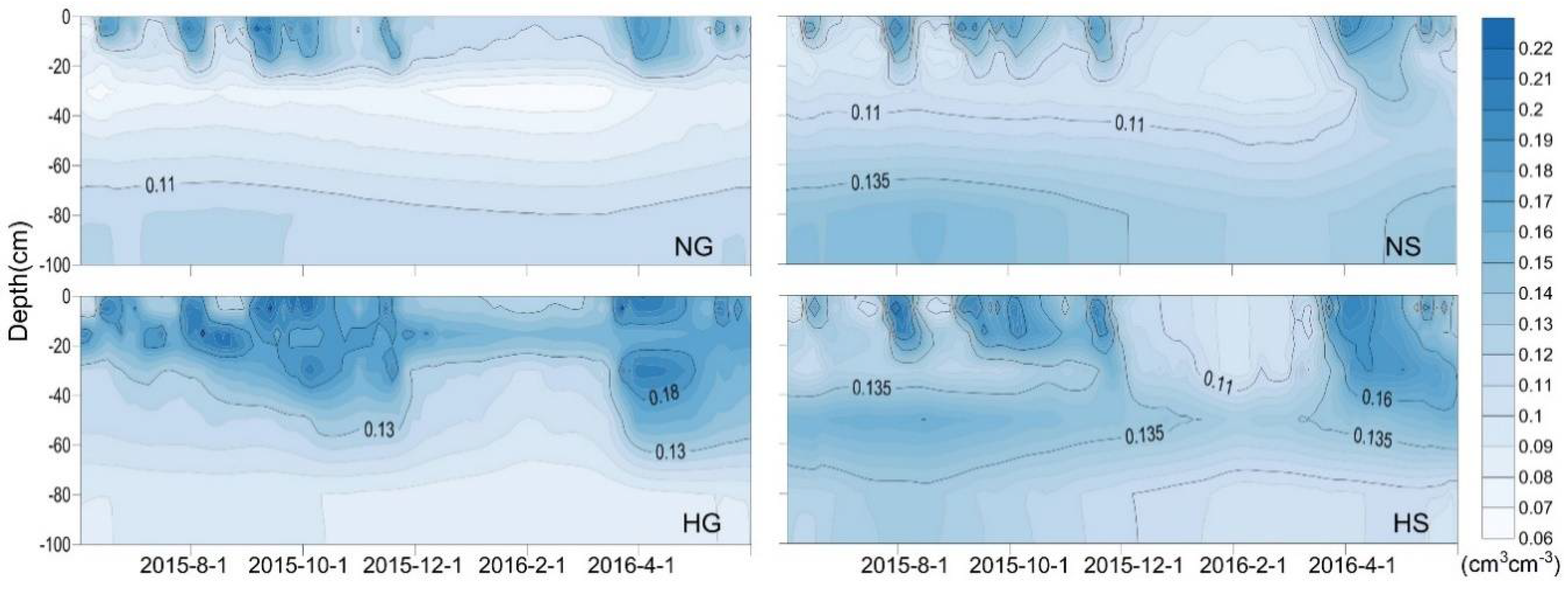
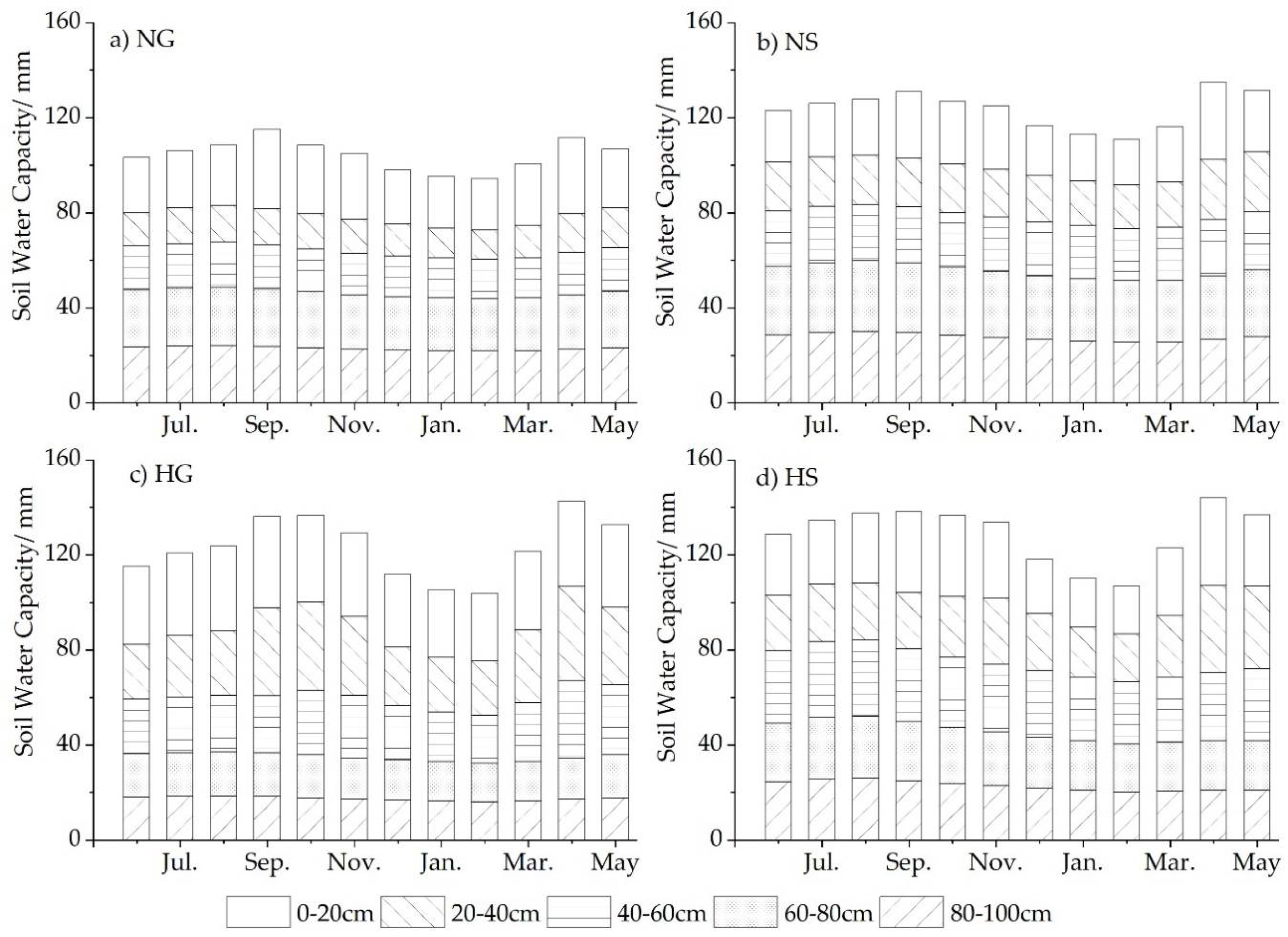
3.2. Annual Dynamics of Soil VWC in Four Sites
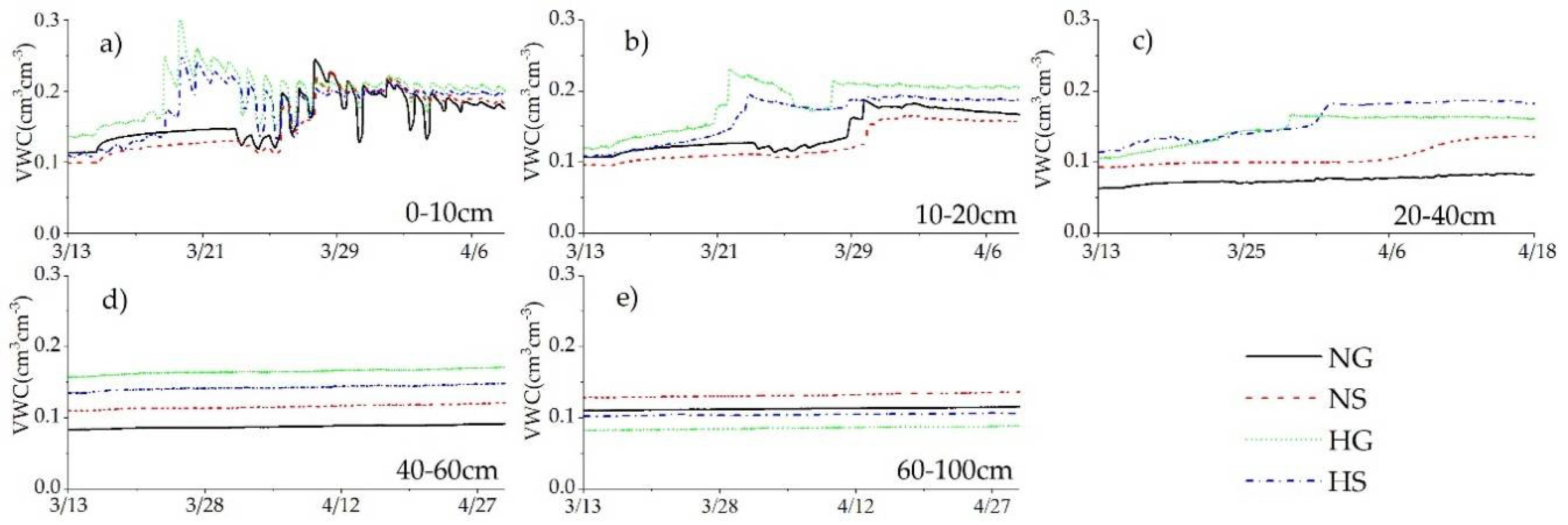
3.3. Impacts of Rainfall and Snowmelt Replenishment on Soil Moisture
3.4. Impacts of Harvesting on Phenology and WUE
4. Discussion
4.1. Effect of Precipitation on Soil Moisture
4.1.1. Rainfall
4.1.2. Snowfall
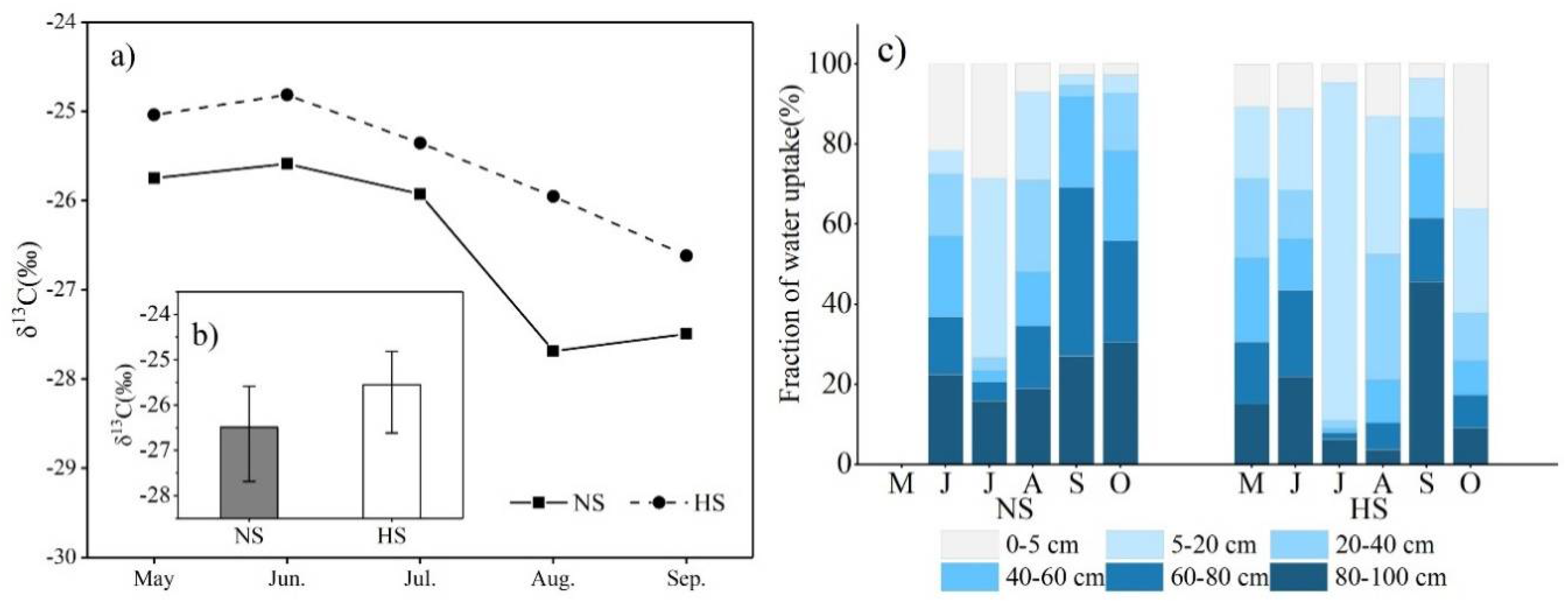
4.2. Water Consumption by Plants
4.3. Plant Physiological Responses
4.4. Predicted Future Scenario and Implications
5. Conclusions
- Shrub patches showed a slow regeneration process. After three years’ recovery, C. microphylla was still 40% shorter than natural ones and sparsely distributed with a 60% of NS in shrub coverage and 25% of NS in aboveground biomass.
- Harvesting has seemed to benefit the soil water recovery, especially under grass, impairing the “hydrologic island” effect of shrubs and promoting a balanced distribution of WS between shrub and grass.
- HS was seemed to be more water-saving than NS due to a higher WUE. The shorter growing season and less biomass in HS, HG, and HSG might reduce the water uptake time and consumption of soil moisture than the natural part.
- More snowmelt replenishment, lower water evapotranspiration, shorter growing season, higher WUE, and so forth could jointly contribute to the water recovery of harvesting soil, especially under HG.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aldous, A.E. Effect of Different Clipping Treatments on the Yield and Vigor of Prairie Grass Vegetation. Ecology 1930, 11, 752–759. [Google Scholar] [CrossRef]
- Armesto, J.J.; Pickett, S.T.A. Experiments on Disturbance in Old-Field Plant Communities: Impact on Species Richness and Abundance. Ecology 1985, 66, 230–240. [Google Scholar] [CrossRef]
- Kobayashi, T.; Hori, Y.; Nomoto, N. Effects of trampling and vegetation removal on species diversity and micro-environment under different shade conditions. J. Veg. Sci. 1997, 8, 873–880. [Google Scholar] [CrossRef]
- Vestergaard, P. Response to mowing of coastal brackish meadow plant communities along an elevational gradient. Nord. J. Bot. 2008, 14, 569–587. [Google Scholar] [CrossRef]
- Zoller, S.H.; Korner, C. Influences of mowing and grazing on plant species composition in calcareous grassland. Bot. Helv. 1998, 108, 57–67. [Google Scholar]
- Eldridge, D.J.; Wang, L.; Ruiz-Colmenero, M. Shrub encroachment alters the spatial patterns of infiltration. Ecohydrology 2014. [Google Scholar] [CrossRef]
- Cahoon, S.M.P.; Sullivan, P.F.; Post, E. Carbon and water relations of contrasting Arctic plants: Implications for shrub expansion in West Greenland. Ecosphere 2016, 7. [Google Scholar] [CrossRef]
- D’Odorico, P.; Okin, G.S.; Bestelmeyer, B.T. A synthetic review of feedbacks and drivers of shrub encroachment in arid grasslands. Ecohydrology 2012, 5, 520–530. [Google Scholar] [CrossRef]
- Maccherini, S.; Marignani, M.; Castagnini, P.; van den Brink, P.J. Multivariate analysis of the response of overgrown semi-natural calcareous grasslands to restorative shrub cutting. Basic Appl. Ecol. 2007, 8, 332–342. [Google Scholar] [CrossRef]
- Dzwonko, Z.; Loster, S. A functional analysis of vegetation dynamics in abandoned and restored limestone grasslands. J. Veg. Sci. 2007, 18, 203–212. [Google Scholar] [CrossRef]
- Papanastasis, V.P.; Platis, P.D.; Dini-Papanastasi, O. Effects of age and frequency of cutting on productivity of Mediterranean deciduous fodder tree and shrub plantations. For. Ecol. Manag. 1998, 110, 283–292. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Mooney, H. Vegetative regrowth following cutting in the shrub Baccharis pilularis ssp. consanguinea (DC) C.B. Wolf. Am. J. Bot. 1985, 72, 514–519. [Google Scholar] [CrossRef]
- Bechtold, H.; Inouye, R. Distribution of carbon and nitrogen in sagebrush steppe after six years of nitrogen addition and shrub removal. J. Arid Environ. 2007, 71, 122–132. [Google Scholar] [CrossRef]
- Fetcher, N. Effects of removal of neighboring species on growth, nutrients, and microclimate of Eriophorum vaginatum. Arct. Alp. Res. 1985, 7–17. [Google Scholar] [CrossRef]
- Lett, M.S.; Knapp, A.K. Consequences of shrub expansion in mesic grassland: resource alterations and graminoid responses. J. Veg. Sci. 2003, 14, 487–496. [Google Scholar] [CrossRef]
- Potts, D.L.; Scott, R.L.; Bayram, S.; Carbonara, J. Woody plants modulate the temporal dynamics of soil moisture in a semi-arid mesquite savanna. Ecohydrology 2010, 3, 20–27. [Google Scholar] [CrossRef]
- Facelli, J.M.; Temby, A.M. Multiple effects of shrubs on annual plant communities in arid lands of South Australia. Austral Ecol. 2002, 27, 422–432. [Google Scholar] [CrossRef]
- Heinrichs, S.; Schmidt, W. Short-term effects of selection and clear cutting on the shrub and herb layer vegetation during the conversion of even-aged Norway spruce stands into mixed stands. For. Ecol. Manag. 2009, 258, 667–678. [Google Scholar] [CrossRef]
- Rango, A.; Tartowski, S.L.; Laliberte, A.; Wainwright, J.; Parsons, A. Islands of hydrologically enhanced biotic productivity in natural and managed arid ecosystems. J. Arid Environ. 2006, 65, 235–252. [Google Scholar] [CrossRef]
- Li, X.Y.; Hu, X.; Zhang, Z.H.; Peng, H.Y.; Zhang, S.Y.; Li, G.Y.; Li, L.; Ma, Y.J. Shrub hydropedology: Preferential water availability to deep soil layer. Vadose Zone J. 2013, 12. [Google Scholar] [CrossRef]
- Peng, H.Y.; Li, X.Y.; Li, G.Y.; Zhang, Z.H.; Zhang, S.Y.; Li, L.; Zhao, G.Q.; Jiang, Z.Y.; Ma, Y.J. Shrub encroachment with increasing anthropogenic disturbance in the semiarid Inner Mongolian grasslands of China. Catena 2013, 109, 39–48. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Li, X.Y.; Peng, H.Y.; Zhang, Z.H. Infiltration pattern beneath shrub canopy and interspace grass patches in typical steppe ecosystems of Inner Mongolia, China. J. Earth Environ. 2012, 95, 1117–1125. [Google Scholar]
- Moore, G.W.; Barre, D.A.; Owens, M.K. Does Shrub Removal Increase Groundwater Recharge in Southwestern Texas Semiarid Rangelands? Rangel. Ecol. Manag. 2012, 65, 1–10. [Google Scholar] [CrossRef]
- Seyfried, M.S.; Wilcox, B.P. Soil water storage and rooting depth: key factors controlling recharge on rangelands. Hydrol. Process. 2006, 20, 3261–3275. [Google Scholar] [CrossRef]
- Sala, O.; Golluscio, R.; Lauenroth, W.; Soriano, A. Resource partitioning between shrubs and grasses in the Patagonian steppe. Oecologia 1989, 81, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Havstad, K.M.; Gibbens, R.P.; Knorr, C.A.; Murray, L.W. Long-term influences of shrub removal and lagomorph exclusion on Chihuahuan Desert vegetation dynamics. J. Arid Environ. 1999, 42, 155–166. [Google Scholar] [CrossRef]
- Rango, A.; Huenneke, L.; Buonopane, M.; Herrick, J.; Havstad, K. Using historic data to assess effectiveness of shrub removal in southern New Mexico. J. Arid Environ. 2005, 62, 75–91. [Google Scholar] [CrossRef]
- D'Odorico, P.; Porporato, A. Soil moisture dynamics in water-limited ecosystems. In Dryland Ecohydrology; Springer: Dordrecht, Netherlands, 2006; pp. 31–46. [Google Scholar]
- Bosch, J.M.; Hewlett, J.D. A review of catchment experiments to determine the effect of vegetation changes on water yield and evapotranspiration. J. Hydrol. 1982, 55, 3–23. [Google Scholar] [CrossRef]
- Mainguet, M. Aridity. In Droughts and Human Development, 1st ed.; Springer: Berlin, Germany, 1999. [Google Scholar] [CrossRef]
- Misson, L.; Tang, J.; Xu, M.; McKay, M.; Goldstein, A. Influences of recovery from clear-cut, climate variability, and thinning on the carbon balance of a young ponderosa pine plantation. Agric. For. Meteorol. 2005, 130, 207–222. [Google Scholar] [CrossRef]
- Wuzhesiguleng; Liu, S.M.; Song, B.Y.; Yang, J.Y. Characteristics of Photosynthesis and Water Physio-ecology of Stipa Grandis. J. Inner Mong. Univ. (Nat. Sci. Ed.) 2015, 46, 262–269. [Google Scholar]
- Kemp, P.R. Phenological patterns of Chihuahuan Desert plants in relation to the timing of water availability. J. Ecol. 1983, 71, 427–436. [Google Scholar] [CrossRef]
- Tissue, D.; Wright, S. Effect of seasonal water availability on phenology and the annual shoot carbohydrate cycle of tropical forest shrubs. Funct. Ecol. 1995, 9, 518–527. [Google Scholar] [CrossRef]
- Dreiss, L.M. The Role of Phenology in Invasive Plant Species Success in Temperate Forest Understories. Ph.D. Thesis, University of Connecticut, Storrs, CT, USA, August 2016. [Google Scholar]
- Li, X.Y.; Zhang, S.Y.; Peng, H.Y.; Hu, X.; Ma, Y.J. Soil water and temperature dynamics in shrub-encroached grasslands and climatic implications: Results from Inner Mongolia steppe ecosystem of North China. Agric. For. Meteorol. 2013, 171, 20–30. [Google Scholar] [CrossRef]
- Czarnomski, N.M.; Moore, G.W.; Pypker, T.G.; Licata, J.; Bond, B.J. Precision and accuracy of three alternative instruments for measuring soil water content in two forest soils of the Pacific Northwest. Can. J. For. Res. 2005, 35, 1867–1876. [Google Scholar] [CrossRef]
- McGarigal, K.; Cushman, S.; Ene, E. FRAGSTATS: Spatial Pattern Analysis Program for Categorical and Continuous Maps. Available online: https://www.umass.edu/landeco/research/fragstats/fragstats.html. (accessed on 23 January 2015).
- Wan, W.; Liu, X. The Observation Methods of Phenology in China; Science Press: Beijing, China, 1979; pp. 42–57. [Google Scholar]
- Phillips, D.L.; Gregg, J.W. Source partitioning using stable isotopes: coping with too many sources. Oecologia 2003, 136, 261–269. [Google Scholar] [CrossRef] [PubMed]
- West, A.G.; Patrickson, S.J.; Ehleringer, J.R. Water extraction times for plant and soil materials used in stable isotope analysis. Rapid Commun. Mass Spectrom. 2006, 20, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.L.; Gregg, J.W. IsoSource: Stable Isotope Mixing Model for Partitioning an Excess Number of Sources; Version 1.3.1; EPA: Washington, WA, USA, 2005. [Google Scholar]
- Farquhar, G.D.; Hubick, K.T.; Condon, A.G.; Richards, A.R. Carbon isotope fractionation and plant water-use efficiency. In Stable Isotopes in Ecological Research; Rundel, P.W., Ehleringer, J.R., Nagy, K.A., Eds.; Springer: Berlin, Germany, 1989; pp. 21–40. [Google Scholar]
- Yu, X.N.; Zhao, J.; Qi, Y.; Huang, Y.M.; Gong, J.R. Effects of soil moisture content on morphological and photosynthetic characteristics of Leymus chinensis and Medicago sativa. Acta Ecol. Sin. 2014, 32, 6067–6075. [Google Scholar]
- Elliott, A.; Carlson, W. Effects of sheep grazing episodes on sediment and nutrient loss in overland flow. Soil Res. 2004, 42, 213–220. [Google Scholar] [CrossRef]
- Chu, J.M. Study on Water Utility of Plant in Arid Area. Ph.D. Thesis, Chinese Academy of Forestry, Beijing, China, August 2007. [Google Scholar]
- Duniway, M.C.; Snyder, K.A.; Herrick, J.E. Spatial and temporal patterns of water availability in a grass-shrub ecotone and implications for grassland recovery in arid environments. Ecohydrology 2010, 3, 309–314. [Google Scholar] [CrossRef]
- Dodd, M.; Lauenroth, W.; Welker, J. Differential water resource use by herbaceous and woody plant life-forms in a shortgrass steppe community. Oecologia 1998, 117, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Zhao, W.Z.; Liu, H. The Response of Aboveground Net Primary Productivity of Desert Vegetation to Rainfall Pulse in the Temperate Desert Region of Northwest China. PLoS ONE 2013. [CrossRef] [PubMed]
- Fan, Y.; Li, X.Y.; Wu, X.C.; Li, L.; Li, W.; Huang, Y.M. Divergent responses of vegetation aboveground net primary productivity to rainfall pulses in the Inner Mongolian Plateau, China. J. Arid Environ. 2016, 129, 1–8. [Google Scholar] [CrossRef]
- Fang, Q.M.; Gong, D.Y.; Mao, R. Changes of Daily Precipitation of China in Wintertime during the Last 46 Years. Sci. Geogr. Sin. 2007, 27, 711–717. [Google Scholar]
- D’Odorico, P.; Fuentes, J.D.; Pockman, W.T.; Collins, S.L.; He, Y.; Medeiros, J.S.; DeWekker, S.; Litvak, M.E. Positive feedback between microclimate and shrub encroachment in the northern Chihuahuan desert. Ecosphere 2010. [Google Scholar] [CrossRef]
- Wepener, J.-P. The Control of Stoebe Vulgaris Encroachment in the Hartbeesfontein Area of the North West Province. Ph.D. Thesis, North-West University, Mahikeng, South Africa, 2007. [Google Scholar]

| Sites | Shrub Cover | Interspace Grass Cover | Shrub Biomass | Interspace Grass Biomass | Patch Size | HEIGHT | Patch Density |
|---|---|---|---|---|---|---|---|
| Natural shrub | 24.37% | 76.0% | 496.8 g/m2 | 72.5 g/m2 | 1.72 m2 | 0.71 m | 0.14 m−2 |
| Harvesting shrub | 14.58% | 79.3% | 123.4 g/m2 | 82.0 g/m2 | 1.06 m2 | 0.39 m | 0.14 m−2 |
| LPI | ENN | LSI | COHESION | DIVISION | SPLIT | AI | |
| Natural shrub | 5.53% | 0.46 | 16.79 | 95.53% | 97.87% | 46.91% | 92.11% |
| Harvesting shrub | 10.13% | 0.51 | 14.21 | 95.49% | 96.94% | 32.73% | 92.60% |
| Paired Sites | 0–10 cm | 10–20 cm | 20–40 cm | 40–60 cm | 60–100 cm |
|---|---|---|---|---|---|
| HG-NG | 0.026 ** | 0.051 ** | 0.076 ** | 0.035 ** | −0.028 ** |
| HS-NS | 0.011 ** | 0.031 ** | 0.026 ** | 0.030 ** | −0.025 ** |
| NG-NS | 0.006 * | 0.012 ** | −0.031 ** | −0.027 ** | −0.023 ** |
| HG-HS | 0.021 ** | 0.032 ** | 0.018 * | −0.021 ** | −0.026 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, Y.; Li, X.-Y.; Li, L.; Wei, J.-Q.; Shi, F.-Z.; Yao, H.-Y.; Liu, L. Plant Harvesting Impacts on Soil Water Patterns and Phenology for Shrub-encroached Grassland. Water 2018, 10, 736. https://doi.org/10.3390/w10060736
Fan Y, Li X-Y, Li L, Wei J-Q, Shi F-Z, Yao H-Y, Liu L. Plant Harvesting Impacts on Soil Water Patterns and Phenology for Shrub-encroached Grassland. Water. 2018; 10(6):736. https://doi.org/10.3390/w10060736
Chicago/Turabian StyleFan, Ying, Xiao-Yan Li, Liu Li, Jun-Qi Wei, Fang-Zhong Shi, Hong-Yun Yao, and Lei Liu. 2018. "Plant Harvesting Impacts on Soil Water Patterns and Phenology for Shrub-encroached Grassland" Water 10, no. 6: 736. https://doi.org/10.3390/w10060736
APA StyleFan, Y., Li, X.-Y., Li, L., Wei, J.-Q., Shi, F.-Z., Yao, H.-Y., & Liu, L. (2018). Plant Harvesting Impacts on Soil Water Patterns and Phenology for Shrub-encroached Grassland. Water, 10(6), 736. https://doi.org/10.3390/w10060736






