Bioremediation of Aluminium from the Waste Water of a Conventional Water Treatment Plant Using the Freshwater Macroalga Oedogonium
Abstract
:1. Introduction
2. Methods
2.1. Study Site and Water Collection
2.2. Productivity of Oedogonium in Giru Waste Water and Sequestration of Al
2.3. The Effect of CO2 on Oedogonium Productivity and Al Sequestration from Giru Waste Water
3. Results and Discussion
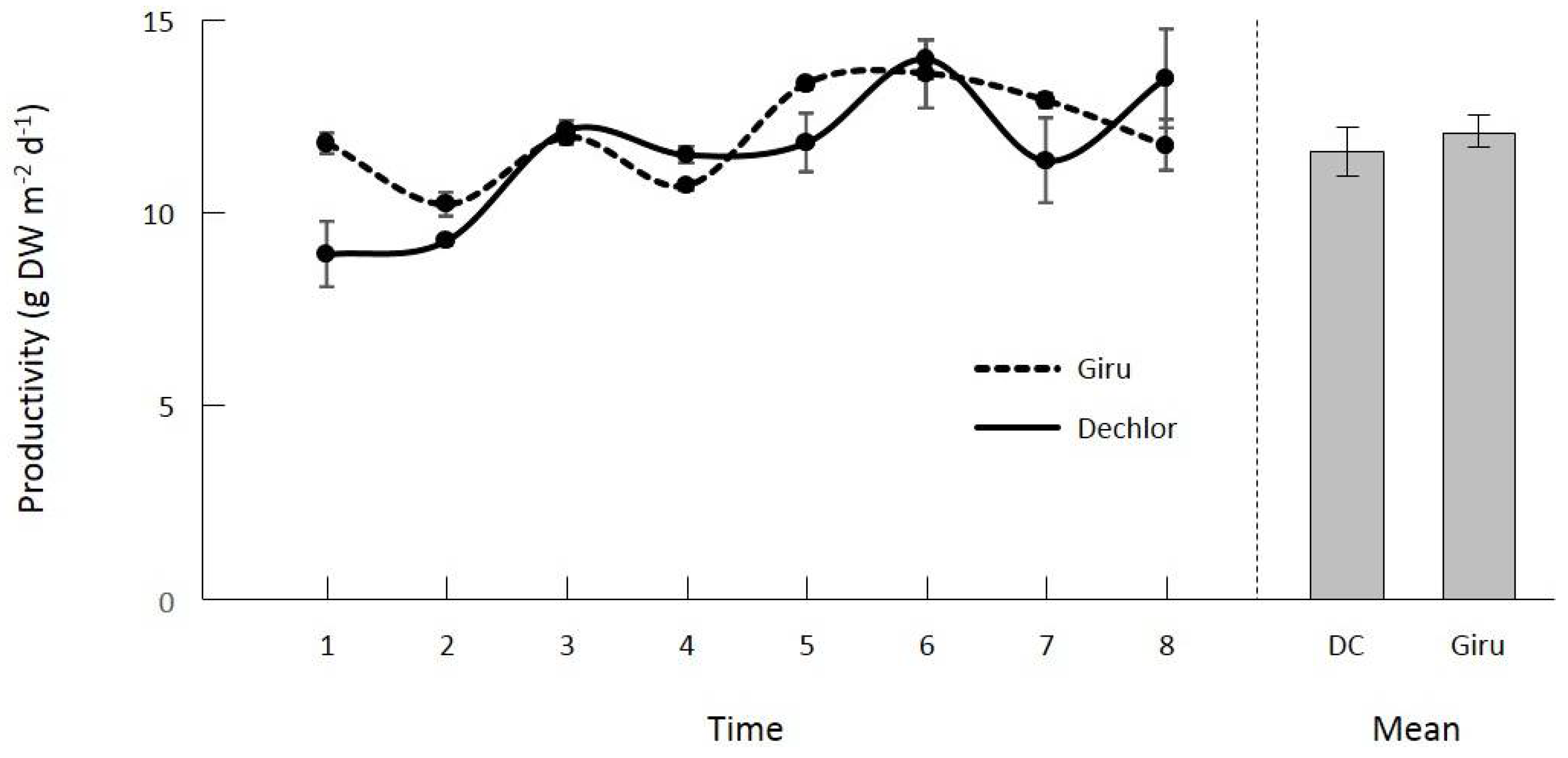
3.1. Productivity of Oedogonium and Al Sequestration in Giru Waste Water
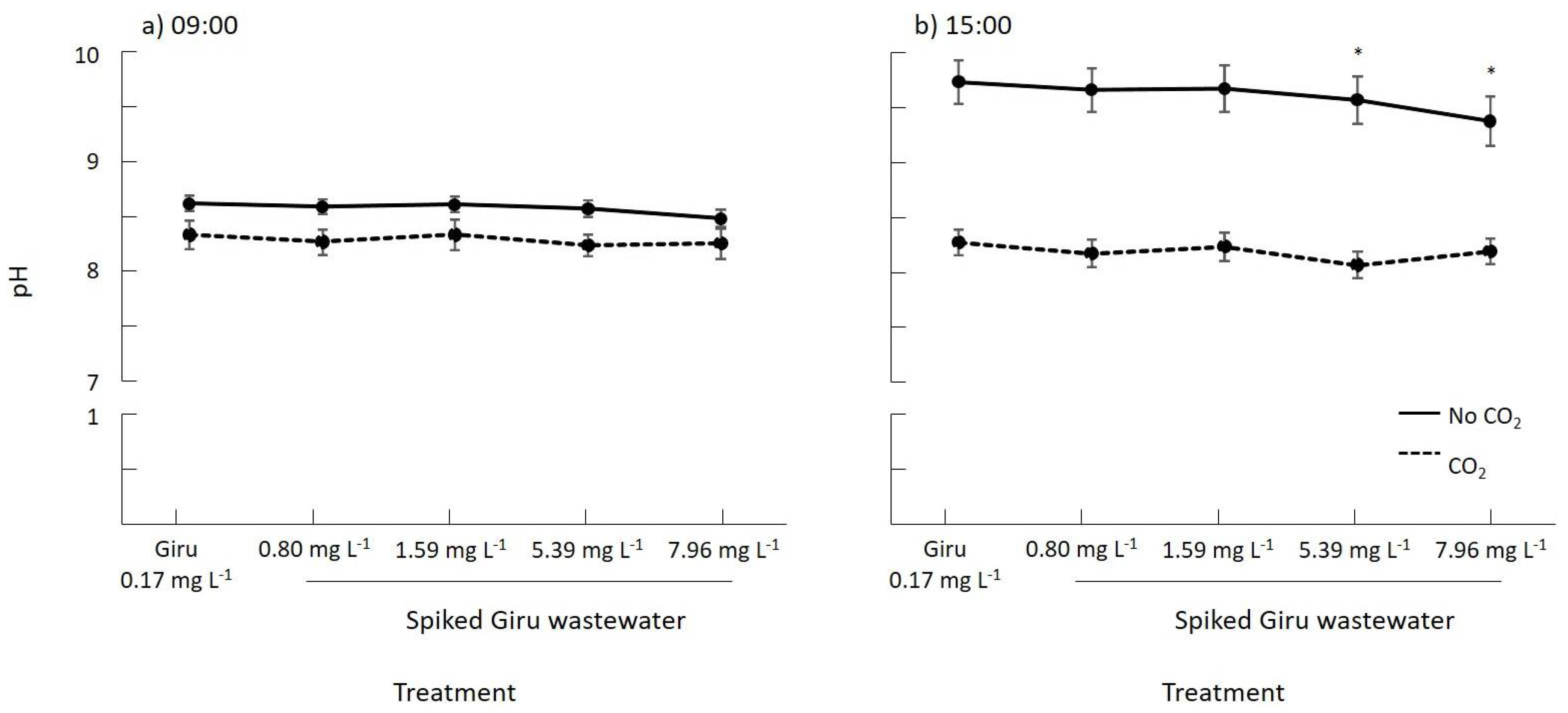
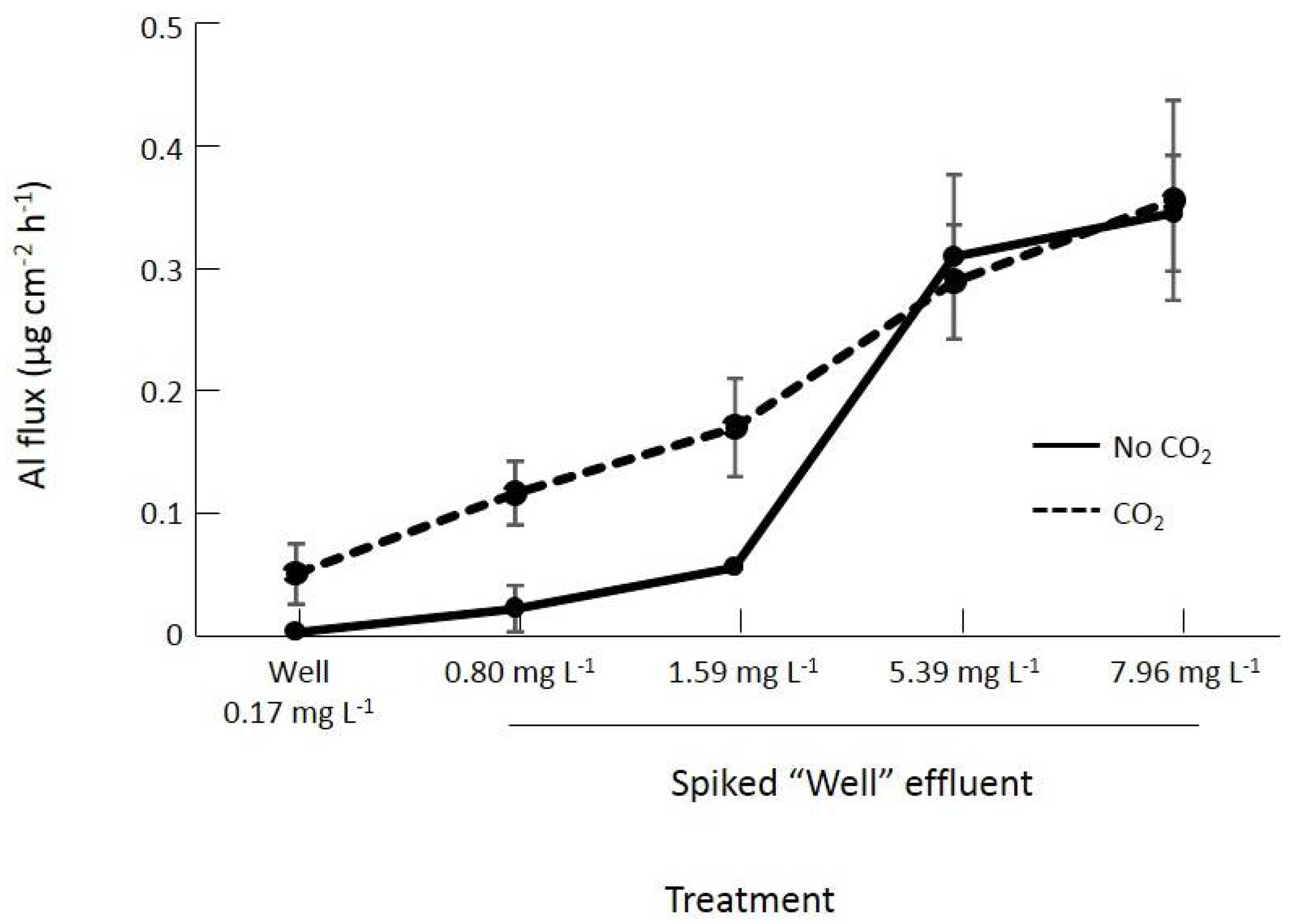
3.2. The Effect of CO2 on the Productivity of Oedogonium and the Bioremediation of Al
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Betancourt, W.Q.; Rose, J.B. Drinking water treatment processes for removal for Cyrptosporidium and Giardia. Vet. Parasitol. 2004, 126, 219–234. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.Y.; Hahn, H.H.; Hoffmann, E. Evaluation of aluminium-silicate polymer composite as a coagulant for water treatment. Water Res. 2002, 36, 3573–3581. [Google Scholar] [CrossRef]
- Sobsey, M.D. Managing Water in the Home: Accelerated Health Gains from Improved Water Supply; World Health Organisation: Geneva, Switzerland, 2002. [Google Scholar]
- Lee, L.Y.; Wang, B.; Guo, H.; Hu, J.Y.; Ong, S.L. Aluminum-based water treatment residue reuse for phosphorus removal. Water 2015, 7, 1480–1496. [Google Scholar] [CrossRef]
- (United States Environmental Protection Agency) USEPA. Drinking Water Treatment Plant Residuals Management; USEPA: Washington, DC, USA, 2011.
- Gensemer, R.; Playle, R. The bioavailability and toxicity of aluminium in aquatic environments. Crit. Rev. Environ. Sci. Technol. 1999, 29, 315–450. [Google Scholar] [CrossRef]
- Roberts, D.A.; de Nys, R.; Paul, N.A. The effect of CO2 on algal growth in industrial waste water for bioenergy and bioremediation applications. PLoS ONE 2013, 8, e81631. [Google Scholar] [CrossRef] [PubMed]
- Roberts, D.A.; Paul, N.A.; Bird, M.I.; de Nys, R. Bioremediation for coal-fired power stations using macroalgae. J. Environ. Manag. 2015, 153, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Cole, A.J.; Mata, L.; Paul, N.; de Nys, R. Using CO2 to enhance carbon capture and biomass applications of freshwater macroalgae. Glob. Chang. Biol. Bioenergy 2014, 6, 637–645. [Google Scholar] [CrossRef]
- Rickman, M.; Pellegrino, J.; Hock, J.; Shaw, S.; Freeman, B. Life-cycle techno-economic analysis of utility-connected algae systems. Algal Res. 2013, 2, 59–65. [Google Scholar] [CrossRef]
- Zhang, H. DGT–for Measurements in Waters, Soils, and Sediments; DGT Research Ltd.: Lancaster, UK, 2013. [Google Scholar]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002; p. 556. [Google Scholar]
- Golding, L.A.; Angel, B.M.; Batley, G.E.; Apte, S.C.; Krassol, R.; Doyle, C.J. Derivation of a water quality guideline for aluminium in marine waters. Environ. Toxicol. Chem. 2015, 34, 141–151. [Google Scholar] [CrossRef] [PubMed]
- ANZECC. An Introduction to the Australian and New Zealand Guidelines for Fresh and Marine Water Quality; National Water Quality Management Strategy: Canberra, Australia, 2000. [Google Scholar]
- National Research Council. Mineral Tolerance of Animals, 2nd ed.; National Acadamy of Sciences: Washington, DC, USA, 2005. [Google Scholar]
- Lawton, R.J.; Cole, A.J.; Roberts, D.A.; Paul, N.A.; de Nys, R. The industrial ecology of freshwater macroalgae for biomass applications. Algal Res. 2017, 24, 486–491. [Google Scholar] [CrossRef]
- Cole, A.J.; Angell, A.R.; de Nys, R.; Paul, N.A. Cyclical changes in biomass productivity and amino acid content of freshwater macroalgae following nitrogen manipulation. Algal Res. 2015, 12, 477–486. [Google Scholar] [CrossRef]
- Neveux, N.; Magnusson, M.; Maschmeyer, T.; de Nys, R.; Paul, N.A. Comparing the potential production and value of high-energy liquid fuels and protein from marine and freshwater macroalgae. Glob. Chang. Biol. Bioenergy 2015, 7, 673–689. [Google Scholar] [CrossRef]
- Neveux, N.; Yuen, A.K.L.; Jazrawi, C.; Magnusson, M.; Haynes, B.S.; Masters, A.F.; Montoya, A.; Paul, N.A.; Maschmeyer, T.; de Nys, R. Biocrude yield and productivity from the hydrothermal liquefaction of marine and freshwater green macroalgae. Bioresour. Technol. 2014, 155, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.J.; van Eyk, P.J.; de Nys, R.; Roberts, D.A.; Cole, A.J.; Ashman, P.J. Mobilisation of trace elements during thermal conversion of algae cultivated in ash dam water. Biomass Bioenergy 2015, 83, 183–195. [Google Scholar] [CrossRef]
- Roberts, D.A.; Paul, N.A.; Cole, A.J.; de Nys, R. From waste water treatment to land management: Conversion of biomass to biochar for soil amelioration and the fortification of crops with essential trace elements. J. Environ. Manag. 2015, 157, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Neveux, N.; Magnusson, M.; Mata, L.; Whelan, A.; de Nys, R.; Paul, N.A. The treatment of municipal wastewater by the macroalga Oedogonium sp. and its potential for the production of biocrude. Algal Res. 2016, 13, 284–292. [Google Scholar] [CrossRef]
- Ge, S.J.; Champagne, P. Cultivation of the marine macroalgae Chaetomorpha linum in municipal wastewater. Environ. Sci. Technol. 2017, 51, 3558–3566. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.J.; Madill, M.; Champagne, P. Use of freshwater macroalgae Spirogyra sp. for the treatment of municipal wastewaters and biomass production for biofuel applications. Biomass Bioenergy 2018, 111, 213–223. [Google Scholar] [CrossRef]
- Wang, N.; Manabe, Y.; Sugawara, T.; Paul, N.A.; Zhao, J. Identification and biological activities of carotenoids from the freshwater alga Oedogonium intermedium. Food Chem. 2018, 242, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.Y.; Kao, C.Y.; Chen, T.Y.; Chang, Y.B.; Kuo, C.M.; Lin, C.S. Cultivation of microalgal Chlorella for biomass and lipid production using wastewater as nutrient resource. Bioresour. Technol. 2015, 184, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Mayhead, E.; Silkina, A.; Llewellyn, C.A.; Fuentes-Grünewald, C. Comparing nutrient removal from membrane filtered and unfiltered domestic wastewater using Chlorella vulgaris. Biology 2018, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Cole, A.J.; Vucko, M.J.; de Nys, R. A comparative assessment on how molasses and CO2 gas prevent carbon limitation in the large-scale culture of freshwater macroalgae. Algal Res. 2017, 27, 215–222. [Google Scholar] [CrossRef]
- Iasimone, F.; De Felice, V.; Panico, A.; Pirozzi, F. Experimental study for the reduction of CO2 emissions in wastewater treatment plant using microalgal cultivation. J. CO2 Util. 2017, 22, 1–8. [Google Scholar] [CrossRef]

| Variable | Units | Value |
|---|---|---|
| Initial [Al] in waste water | mg L−1 | 0.260 ± 0.020 |
| Final [Al] in waste water | mg L−1 | 0.068 ± 0.007 |
| Mass Al lost from waste water | Mg | 2.68 ± 0.11 |
| [Al] in harvested Oedogonium | mg kg−1 | 467 ± 25 |
| Mass harvested Oedogonium | g DW | 5.40 ± 0.13 |
| Mass Al sequestered in Oedogonium | Mg | 2.48 ± 0.09 |
| Proportion of Al in Oedogonium | % | 89.5 ± 6.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roberts, D.A.; Shiels, L.; Tickle, J.; De Nys, R.; Paul, N.A. Bioremediation of Aluminium from the Waste Water of a Conventional Water Treatment Plant Using the Freshwater Macroalga Oedogonium. Water 2018, 10, 626. https://doi.org/10.3390/w10050626
Roberts DA, Shiels L, Tickle J, De Nys R, Paul NA. Bioremediation of Aluminium from the Waste Water of a Conventional Water Treatment Plant Using the Freshwater Macroalga Oedogonium. Water. 2018; 10(5):626. https://doi.org/10.3390/w10050626
Chicago/Turabian StyleRoberts, David A., Laura Shiels, Julian Tickle, Rocky De Nys, and Nicholas A. Paul. 2018. "Bioremediation of Aluminium from the Waste Water of a Conventional Water Treatment Plant Using the Freshwater Macroalga Oedogonium" Water 10, no. 5: 626. https://doi.org/10.3390/w10050626
APA StyleRoberts, D. A., Shiels, L., Tickle, J., De Nys, R., & Paul, N. A. (2018). Bioremediation of Aluminium from the Waste Water of a Conventional Water Treatment Plant Using the Freshwater Macroalga Oedogonium. Water, 10(5), 626. https://doi.org/10.3390/w10050626






