Value-Added Products Derived from Waste Activated Sludge: A Biorefinery Perspective
Abstract
:1. Introduction
2. Amino Acids and Proteins
3. Short-Chain Fatty Acids
4. Enzymes
5. Bio-Pesticides
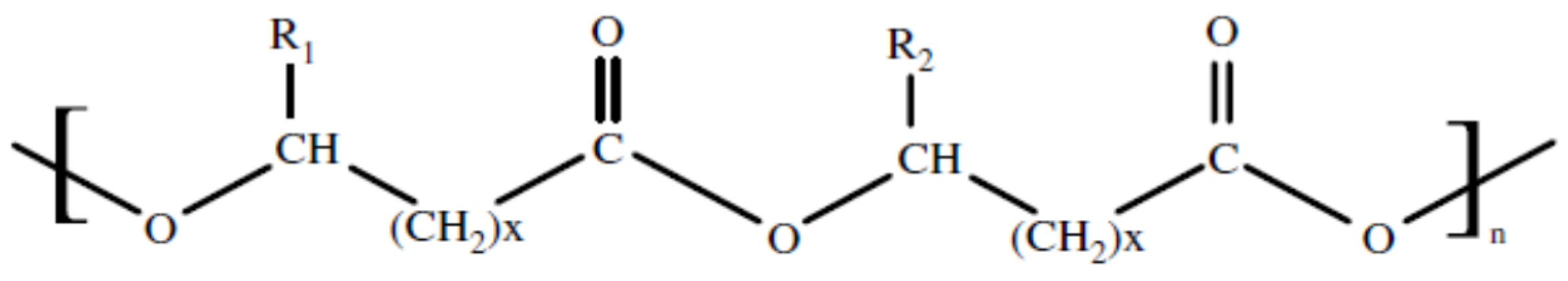
6. Bio-Plastics
7. Bio-Flocculants and Bio-Surfactants
8. Current Challenges
- (1)
- Biorefinery production of protein and enzymes are still considered to be higher cost routes compared with agro-industry residues and animal health in terms of pathogenic and metals toxicity, which has effectively stifled developments in this area [139].
- (2)
- Recovery of metabolic WAS products (e.g., bio-plastics, bio-pesticides, bio-flocculants and bio-surfactants) remains in its infancy and further optimisation of operational parameters and selection of non-toxic strains are needed to progress this research area.
- (3)
- Important factors such as optimum growth environment or wastewater matrix for harvesting WAS with the highest concentration of specific bio-products need to be further investigated and refined.
- (4)
- WAS bio-product production through biological processes such as fermentation, bioleaching or enzymatic extraction, while an evolving area of research, still requires further optimisation.
- (5)
- Anaerobic digestion as a potential option for enhancing the production of SCFAs and their extraction from WAS through fermentation in the presence of key chemical surfactants deserves further exploration.
- (6)
- Simple and efficient purification procedures for certain bio-products recovered from WAS (in line with the required purity for specific applications) need further development and would contribute to improving the overall economics of biorefinery approaches.
- (7)
- Scale of WAS-based biorefinery production is an important future challenge, since much of the research so far has been at laboratory scale. To scale up the biorefinery approach using WAS, further research must be carried out at larger scales (pilot- and ultimately full-scale) to optimize each biorefinery process. Only then can the full techno-economic performance of these processes be fully investigated and understood.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Drecht, G.; Bouwman, A.; Harrison, J.; Knoop, J. Global nitrogen and phosphate in urban wastewater for the period 1970 to 2050. Glob. Biogeochem. Cycles 2009, 23, GB0A03. [Google Scholar] [CrossRef]
- Peccia, J.; Westerhoff, P. We Should Expect More out of Our Sewage Sludge. Environ. Sci. Technol. 2015, 49, 8271–8276. [Google Scholar] [CrossRef] [PubMed]
- Spinosa, L. Wastewater Sludge: A Global Overview of the Current Status and Future Prospects, 2nd ed.; IWA Publishing: London, UK, 2011; ISBN 9781780401195. [Google Scholar]
- UNICEF; World Health Organization (WHO). Progress on Sanitation and Drinking Water—2015 Update and MDG Assessment; WHO/UNICEF Joint Monitoring Programme for Water Supply and Sanitation; UNICEF: New York, NY, USA, 2015; ISBN 978 92 4 150914 5. [Google Scholar]
- Egan, M. Biosolds management strategies: An evaluation of energy production as an alterntive to land application. Environ. Sci. Pollut. Res. 2013, 20, 4299–4310. [Google Scholar] [CrossRef] [PubMed]
- Lundin, M.; Olofsson, M.; Pettersson, G.; Zetterlund, H. Environmental and economic assessment of sewage sludge handling options. Resources. Conserv. Recycl. 2004, 41, 255–278. [Google Scholar] [CrossRef]
- Campbell, H. Sludge management–future issues and trends. Water Sci. Technol. 2000, 41, 1–8. [Google Scholar]
- Rulkens, W. Sewage sludge as a biomass resource for the production of energy: Overview and assessment of the various options. Energy Fuels 2007, 22, 9–15. [Google Scholar] [CrossRef]
- Wang, H.; Brown, S.L.; Magesan, G.N.; Slade, A.H.; Quintern, M.; Clinton, P.W. Technological options for the management of biosolids. Environ. Sci. Pollut. Res. 2008, 15, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Hadi, P.; Xu, M.; Ning, C.; Lin, C.S.K.; McKay, G. A critical review on preparation, characterization and utilization of sludge-derived activated carbons for wastewater treatment. Chem. Eng. J. 2015, 260, 895–906. [Google Scholar] [CrossRef]
- Ukwatta, A.; Mohajerani, A.; Setunge, S.; Eshtiaghi, N. Possible use of biosolids in fired-clay bricks. Constr. Build. Mater. 2015, 91, 86–93. [Google Scholar] [CrossRef]
- Raunkjaer, K.; Hvitvedjacobsen, T.; Nielsen, P.H. Measurement of pools of protein, carbohydrate and lipid in domestic wastewater. Water Res. 1994, 28, 251–262. [Google Scholar] [CrossRef]
- Adebayo, O.; Fagbenro, O.; Jegede, T. Evaluation of Cassia fistula meal as a replacement for soybean meal in practical diets of Oreochromis niloticus fingerlings. Aquac. Nutr. 2004, 10, 99–104. [Google Scholar] [CrossRef]
- Shier, W.T.; Purwono, S.K. Extraction of single-cell protein from activated sewage sludge: Thermal solubilization of protein. Bioresour. Technol. 1994, 49, 157–162. [Google Scholar] [CrossRef]
- Vriens, L.; Nihoul, R.; Verachtert, H. Activated sludges as animal feed: A review. Biol. Wastes 1989, 27, 161–207. [Google Scholar] [CrossRef]
- Chishti, S.S.; Hasnain, S.N.; Khan, M.A. Studies on the recovery of sludge protein. Water Res. 1992, 26, 241–248. [Google Scholar] [CrossRef]
- Hwang, J.; Zhang, L.; Seo, S.; Lee, Y.W.; Jahng, D. Protein recovery from excess sludge for its use as animal feed. Bioresour. Technol. 2008, 99, 8949–8954. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.F.; Su, R.J.; Guo, G.L.; Li, D.X. Experimental Research on Protein Extraction from Excess Activated Sludge by Papain Hydrolysis. Adv. Mater. Res. 2012, 518–523, 3367–3370. [Google Scholar] [CrossRef]
- Ras, M.; Girbal-Neuhauser, E.; Paul, E.; Sperandio, M.; Lefebvre, D. Protein extraction from activated sludge: An analytical approach. Water Res. 2008, 42, 1867–1878. [Google Scholar] [CrossRef] [PubMed]
- Kroiss, H. What is the potential for utilizing the resources in sludge? Water Sci. Technol. 2004, 49, 1–10. [Google Scholar] [PubMed]
- Lu, J.; Zhao, Y.; Gong, H.; Li, D.; Liu, J. New technology for production of the amino acid chelated trace metal fertilizers. Phosphate Compd. Fertil. 2007, 6, 49–52. [Google Scholar]
- Liu, Y.; Kong, S.; Li, Y.; Zeng, H. Novel technology for sewage sludge utilization: Preparation of amino acids chelated trace elements (AACTE) fertilizer. J. Hazard. Mater. 2009, 171, 1159–1167. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Q.; Cai, Q.R.; He, X.M.; Gao, L.X.; Zhou, G.D. Inhibition effect of some amino acids on copper corrosion in HCl solution. Mater. Chem. Phys. 2008, 112, 353–358. [Google Scholar] [CrossRef]
- Gece, G.; Bilgiç, S. A theoretical study on the inhibition efficiencies of some amino acids as corrosion inhibitors of nickel. Corros. Sci. 2010, 52, 3435–3443. [Google Scholar] [CrossRef]
- Khaled, K. Corrosion control of copper in nitric acid solutions using some amino acids—A combined experimental and theoretical study. Corros. Sci. 2010, 52, 3225–3234. [Google Scholar] [CrossRef]
- Eddy, N.O. Experimental and theoretical studies on some amino acids and their potential activity as inhibitors for the corrosion of mild steel, part 2. J. Adv. Res. 2011, 2, 35–47. [Google Scholar] [CrossRef]
- Su, W.; Tang, B.; Fu, F.; Huang, S.; Zhao, S.; Bin, L. A new insight into resource recovery of excess sewage sludge: Feasibility of extracting mixed amino acids as an environment-friendly corrosion inhibitor for industrial pickling. J. Hazard. Mater. 2014, 279, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Pervaiz, M.; Sain, M. Protein extraction from secondary sludge of paper mill wastewater and its utilization as a wood adhesive. Bioresources 2011, 6, 961–970. [Google Scholar]
- Rulkens, W. Sustainable sludge management—What are the challenges for the future? Water Sci. Technol. 2004, 49, 11–19. [Google Scholar] [PubMed]
- Yuan, H.; Chen, Y.; Zhang, H.; Jiang, S.; Zhou, Q.; Gu, G. Improved bioproduction of short-chain fatty acids (SCFAs) from excess sludge under alkaline conditions. Environ. Sci. Technol. 2006, 40, 2025–2029. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jiang, S.; Yuan, H.; Zhou, Q.; Gu, G. Hydrolysis and acidification of waste activated sludge at different pHs. Water Res. 2007, 41, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.H.; He, P.J.; Shao, L.M.; He, P.P. Toward understanding the mechanism of improving the production of volatile fatty acids from activated sludge at pH 10.0. Water Res. 2008, 42, 4637–4644. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, X.; Kang, X.; Yuan, Y.; Du, M. Short chain fatty acids accumulation and microbial community succession during ultrasonic-pretreated sludge anaerobic fermentation process: Effect of alkaline adjustment. Int. Biodeterior. Biodegradation 2014, 94, 128–133. [Google Scholar] [CrossRef]
- Rughoonundun, H.; Granda, C.; Mohee, R.; Holtzapple, M.T. Effect of thermochemical pretreatment on sewage sludge and its impact on carboxylic acids production. Waste Manag. 2010, 30, 1614–1621. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, J.; Zhang, A.; Wang, L. Enhancing the quantity and quality of short-chain fatty acids production from waste activated sludge using CaO2 as an additive. Water Res. 2015, 83, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Wang, J.; Li, Y. Performance of calcium peroxide for removal of endocrine-disrupting compounds in waste activated sludge and promotion of sludge solubilization. Water Res. 2015, 71, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Chen, Y.; Zhou, Q.; Gu, G. Biological short-chain fatty acids (SCFAs) production from waste-activated sludge affected by surfactant. Water Res. 2007, 41, 3112–3120. [Google Scholar] [CrossRef] [PubMed]
- Shanableh, A.; Jones, S. Production and transformation of volatile fatty acids from sludge subjected to hydrothermal treatment. Water Sci. Technol. 2001, 44, 129–135. [Google Scholar] [PubMed]
- Liao, P.H.; Lo, K.V.; Chan, W.I.; Wong, W.T. Sludge reduction and volatile fatty acid recovery using microwave advanced oxidation process. J. Environ. Sci. Health Part A 2007, 42, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Yu, L.; Huang, S.; Luo, J.; Zhuo, Y. Energy efficiency of pre-treating excess sewage sludge with microwave irradiation. Bioresour. Technol. 2010, 101, 5092–5097. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Lei, H.; Li, Z.; Li, H.; Chen, K.; Zhang, X. Physical and chemical properties of waste-activated sludge after microwave treatment. Water Res. 2010, 44, 2841–2849. [Google Scholar] [CrossRef] [PubMed]
- Qiao, W.; Wang, W.; Xun, R.; Lu, W.; Yin, K. Sewage sludge hydrothermal treatment by microwave irradiation combined with alkali addition. J. Mater. Sci. 2008, 43, 2431–2436. [Google Scholar] [CrossRef]
- Hong, S.M.; Park, J.K.; Teeradej, N.; Lee, Y.; Cho, Y.; Park, C. Pretreatment of sludge with microwaves for pathogen destruction and improved anaerobic digestion performance. Water Environ. Res. 2006, 78, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Dufreche, S.; Hernandez, R.; French, T.; Sparks, D.; Zappi, M.; Alley, E. Extraction of lipids from municipal wastewater plant microorganisms for production of biodiesel. J. Am. Oil Chem. Soc. 2007, 84, 181–187. [Google Scholar] [CrossRef]
- Huynh, L.H.; Kasim, N.S.; Ju, Y.H. Extraction and analysis of neutral lipids from activated sludge with and without sub-critical water pre-treatment. Bioresour. Technol. 2010, 101, 8891–8896. [Google Scholar] [CrossRef] [PubMed]
- Kwon, E.; Kim, S.; Jeon, Y.; Yi, H. Biodiesel production from sewage sludge: New paradigm for mining enegry from municipal hazardous material. Environ. Sci. Technol. 2012, 46, 10222–10228. [Google Scholar] [CrossRef] [PubMed]
- Kargbo, D.M. Biodiesel production from municipal sewage sludges. Energy Fuels 2010, 24, 2791–2794. [Google Scholar] [CrossRef]
- Feng, L.; Wang, H.; Chen, Y.; Wang, Q. Effect of solids retention time and temperature on waste activated sludge hydrolysis and short-chain fatty acids accumulation under alkaline conditions in continuous-flow reactors. Bioresour. Technol. 2009, 100, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Peng, Y.; Ren, N.; Li, B.; Chai, T.; Zhang, L. Effect of temperature on short chain fatty acids (SCFAs) accumulation and microbiological transformation in sludge alkaline fermentation with Ca(OH)2 adjustment. Water Res. 2014, 61, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Jia, Q.; Xiong, H.; Wang, H.; Shi, H.; Sheng, X.; Sun, R. Production of polyhydroxyalkanoates (PHA) by bacterial consortium from excess sludge fermentation liquid at laboratory and pilot scales. Bioresour. Technol. 2014, 171, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Elefsiniotis, P.; Wareham, D.G. Utilization patterns of volatile fatty acids in the denitrification reaction. Enzym. Microb. Technol. 2007, 41, 92–97. [Google Scholar] [CrossRef]
- Levine, A.D.; Tchobanoglous, G.; Asano, T. Characterization of the size distribution of contaminants in wastewater: Treatment and reuse implications. J. Water Pollut. Control Fed. 1985, 57, 805–816. [Google Scholar]
- Tyagi, R.D.; Surampalli, R.Y.; Yan, S. (Eds.) Sustainable Sludge Management: Production of Value Added Products; American Society of Civil Engineers: Reston, VA, USA, 2009. [Google Scholar]
- Juwon, A.D.; Emmanuel, O.F. Experimental investigations on the effects of carbon and nitrogen sources on concomitant amylase and polygalacturonase production by Trichoderma viride BITRS-1001 in submerged fermentation. Biotechnol. Res. Int. 2012, 2012, 904763. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.H.; He, P.J.; Shao, L.M.; Lee, D.J. Enzyme activities in activated sludge flocs. Appl. Microbiol. Biotechnol. 2007, 77, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.H.; He, P.; Shao, L.; Zhu, Y. Enzyme extraction by ultrasound from sludge flocs. J. Environ. Sci. 2009, 21, 204–210. [Google Scholar] [CrossRef]
- Nabarlatz, D.; Vondrysova, J.; Jenicek, P.; Stüber, F.; Font, J.; Fortuny, A. Hydrolytic enzymes in activated sludge: Extraction of protease and lipase by stirring and ultrasonication. Ultrason. Sonochemistry 2010, 17, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Nabarlatz, D.; Stüber, F.; Font, J.; Fortuny, A.; Fabregat, A.; Bengoa, C. Extraction and purification of hydrolytic enzymes from activated sludge. Resour. Conserv. Recycl. 2012, 59, 9–13. [Google Scholar] [CrossRef]
- Cortes-Lorenzo, C.; Rodriguez-Diaz, M.; Lopez-Lopez, C.; Sanchez-Peinado, M.; Rodelas, B.; Gonzalez-Lopez, J. Effect of salinity on enzymatic activities in a submerged fixed bed biofilm reactor for municipal sewage treatment. Bioresour. Technol. 2012, 121, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Gessesse, A.; Petersen, S.B.; Nielsen, P.H. Lipase and protease extraction from activated sludge. Water Res. 2003, 37, 3652–3657. [Google Scholar] [CrossRef]
- Jung, J.; Yu, K.O.; Ramzi, A.B.; Choe, S.H.; Kim, S.W.; Han, S.O. Improvement of surfactin production in Bacillus subtilis using synthetic wastewater by overexpression of specific extracellular signaling peptides, comX and phrC. Biotechnol. Bioeng. 2012, 109, 2349–2356. [Google Scholar] [CrossRef] [PubMed]
- Bravo, A.; Likitvivatanavong, S.; Gill, S.S.; Soberón, M. Bacillus thuringiensis: A story of a successful bioinsecticide. Insect Biochem. Mol. Biol. 2001, 41, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Lisansky, S.; Quinlan, R.; Tassoni, G. Bacillus Thuringiensis Production Handbook: Laboratory Methods, Manufacturing, Formulation, Quality Control, Registration; CPL Scientific Ltd.: Berkshire, UK, 1993. [Google Scholar]
- Flores, E.R.; Pérez, F.; de la Torre, M. Scale-up of Bacillus thuringiensis fermentation based on oxygen transfer. J. Ferment. Bioeng. 1997, 83, 561–564. [Google Scholar] [CrossRef]
- Jégou, S.; Douliez, J.P.; Mollé, D.; Boivin, P.; Marion, D. Evidence of the glycation and denaturation of LTP1 during the malting and brewing process. J. Agric. Food Chem. 2001, 49, 4942–4949. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, V.; Tyagi, R.; Valero, J. Production of biopesticides as a novel method of wastewater sludge utilization/disposal. Water Sci. Technol. 2000, 42, 211–216. [Google Scholar]
- Lachhab, K.; Tyagi, R.; Valero, J. Production of Bacillus thuringiensis biopesticides using wastewater sludge as a raw material: Effect of inoculum and sludge solids concentration. Process Biochem. 2001, 37, 197–208. [Google Scholar] [CrossRef]
- Vidyarthi, A.; Tyagi, R.; Valero, J.; Surampalli, R. Studies on the production of B. thuringiensis based biopesticides using wastewater sludge as a raw material. Water Res. 2002, 36, 4850–4860. [Google Scholar] [CrossRef]
- Montiel, M.T.; Tyagi, R.; Valero, J.; Surampalli, R. Production of biopesticides using wastewater sludge as a raw material-effect of process parameters. Water Sci. Technol. 2003, 48, 239–246. [Google Scholar]
- Vidyarthi, A.; Desrosiers, M.; Tyagi, R.; Valero, J. Foam control in biopesticide production from sewage sludge. J. Ind. Microbiol. Biotechnol. 2000, 25, 86–92. [Google Scholar] [CrossRef]
- Barnabe, S.; Verma, M.; Tyagi, R.D.; Valero, J.R. Culture Media for Increasing Biopesticide Producing Microorganisms’ Pesticidal Activity, Methods of Producing Same, Biopesticide Producing Microorganisms So Produced. Google Patents WO2006089388A1, 31 August 2006. [Google Scholar]
- Montiel, M.D.L.T.; Tyagi, R.; Valero, J. Wastewater treatment sludge as a raw material for the production of Bacillus thuringiensis based biopesticides. Water Res. 2001, 35, 3807–3816. [Google Scholar] [CrossRef]
- Verma, M.; Brar, S.K.; Tyagi, R.; Valéro, J.; Surampalli, R. Wastewater sludge as a potential raw material for antagonistic fungus (Trichoderma sp.): Role of pre-treatment and solids concentration. Water Res. 2005, 39, 3587–3596. [Google Scholar] [CrossRef] [PubMed]
- Akaraonye, E.; Keshavarz, T.; Roy, I. Production of polyhydroxyalkanoates: The future green materials of choice. J. Chem. Technol. Biotechnol. 2010, 85, 732–743. [Google Scholar] [CrossRef]
- Thomson, N.; Roy, I.; Summers, D.; Sivaniah, E. In vitro production of polyhydroxyalkanoates: Achievements and applications. J. Chem. Technol. Biotechnol. 2010, 85, 760–767. [Google Scholar] [CrossRef]
- Philip, S.; Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Biodegradable polymers with a range of applications. J. Chem. Technol. Biotechnol. 2007, 82, 233–247. [Google Scholar] [CrossRef]
- Wu, Q.; Wang, Y.; Chen, G.Q. Medical application of microbial biopolyesters polyhydroxyalkanoates. Artif. Cells Blood Substit. Immobil. Biotechnol. 2009, 37, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Chua, H.; Peter, H.; Ma, C.K. Accumulation of Biopolymers in Activated Sludge Biomass; Springer: Berlin, Germany, 1999; pp. 389–399. [Google Scholar]
- Lee, S.Y. Bacterial polyhydroxyalkanoates. Biotechnol. Bioeng. 1996, 49, 1–14. [Google Scholar] [CrossRef]
- Law, K.H.; Leung, Y.C.; Lawford, H.; Chua, H.; Lo, W.H.; Yu, P.H. Production of Polyhydroxybutyrate by Bacillus Species Isolated from Municipal Activated Sludge; Springer: Berlin, Germany, 2001; pp. 515–524. [Google Scholar]
- SUCDEN. World Ethanol Market. Available online: http://www.sucden.com/statistics/10_world-ethanol-market (accessed on 18 October 2016).
- Baetens, D.; Aurola, A.M.; Foglia, A.; Dionisi, D.; Van Loosdrecht, M. Gas chromatographic analysis of polyhydroxybutyrate in activated sludge: A round-robin test. Water Sci. Technol. 2002, 46, 357–361. [Google Scholar] [PubMed]
- Satoh, H.; Mino, T.; Matsuo, T. PHA production by activated sludge. Int. J. Biol. Macromol. 1999, 25, 105–109. [Google Scholar] [CrossRef]
- Chua, A.S.; Takabatake, H.; Satoh, H.; Mino, T. Production of polyhydroxyalkanoates (PHA) by activated sludge treating municipal wastewater: Effect of pH, sludge retention time (SRT), and acetate concentration in influent. Water Res. 2003, 37, 3602–3611. [Google Scholar] [CrossRef]
- Krishna, C.; Van Loosdrecht, M.C. Effect of temperature on storage polymers and settleability of activated sludge. Water Res. 1999, 33, 2374–2382. [Google Scholar] [CrossRef]
- Ma, C.; Chua, H.; Yu, P.; Hong, K. Optimal production of polyhydroxyalkanoates in activated sludge biomass. Appl. Biochem. Biotechnol. 2000, 84, 981–989. [Google Scholar] [CrossRef]
- Kumar, M.S.; Mudliar, S.; Reddy, K.; Chakrabarti, T. Production of biodegradable plastics from activated sludge generated from a food processing industrial wastewater treatment plant. Bioresour. Technol. 2004, 95, 327–330. [Google Scholar]
- Satoh, H.; Iwamoto, Y.; Mino, T.; Matsuo, T. Activated sludge as a possible source of biodegradable plastic. Water Sci. Technol. 1998, 38, 103–109. [Google Scholar]
- Satoh, H.; Mino, T.; Matsuo, T. Anaerobic uptake of glutamate and aspartate by enhanced biological phosphorus removal activated sludge. Water Sci. Technol. 1998, 37, 579–582. [Google Scholar]
- Takabatake, H.; Satoh, H.; Mino, T.; Matsuo, T. Recovery of biodegradable plastics from activated sludge process. Water Sci. Technol. 2000, 42, 351–356. [Google Scholar]
- Yan, S.; Subramanian, S.B.; Tyagi, R.; Surampalli, R. Bioplastics from waste activated sludge-batch process. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2008, 12, 239–248. [Google Scholar] [CrossRef]
- Khardenavis, A.A.; Kumar, M.S.; Mudliar, S.N.; Chakrabarti, T. Biotechnological conversion of agro-industrial wastewaters into biodegradable plastic, poly β-hydroxybutyrate. Bioresour. Technol. 2007, 98, 3579–3584. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Yu, J. Production of biodegradable thermoplastics from municipal solids by a two-stage bioprocess. Resour. Conserv. Recycl. 1997, 19, 151–164. [Google Scholar] [CrossRef]
- Pleissner, D.; Lam, W.C.; Han, W.; Lau, K.Y.; Cheung, L.C.; Lee, M.W.; Lei, H.M.; Lo, K.Y.; Ng, W.Y.; Sun, Z.; et al. Fermentative Polyhydroxybutyrate Production from a Novel Feedstock Derived from Bakery Waste. BioMed Res. Int. 2014, 2014, 819474. [Google Scholar] [CrossRef] [PubMed]
- Moralejo-Gárate, H.; Palmeiro-Sánchez, T.; Kleerebezem, R.; Mosquera-Corral, A.; Campos, J.L.; van Loosdrecht, M.C.M. Influence of the cycel length on the production of PHA and polyglucose from glycerol by bacterial enrichments in sequencing batch reactors. Biotechnol. Bioeng. 2013, 110, 3148–3155. [Google Scholar] [CrossRef] [PubMed]
- Valentino, F.; Morgan-Sagastume, F.; Fraraccio, S.; Corsi, G.; Zanaroli, G.; Werker, A.; Majone, M. Sludge minization in municipal wastewater treatment by polyhydroxyalkanoate (PHA) production. Environ. Sci. Pollut. Res. 2015, 22, 7281–7294. [Google Scholar] [CrossRef] [PubMed]
- Brar, S.K.; Verma, M.; Tyagi, R.; Surampalli, R. Value addition of wastewater sludge: Future course in sludge reutilization. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2009, 13, 59–74. [Google Scholar] [CrossRef]
- He, N.; Li, Y.; Chen, J.; Lun, S.Y. Identification of a novel bioflocculant from a newly isolated Corynebacterium glutamicum. Biochem. Eng. J. 2002, 11, 137–148. [Google Scholar] [CrossRef]
- He, N.; Li, Y.; Chen, J. Production of a novel polygalacturonic acid bioflocculant REA-11 by Corynebacterium glutamicum. Bioresour. Technol. 2004, 94, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Kurane, R.; Hatamochi, K.; Kakuno, T.; Kiyohara, M.; Hirano, M.; Taniguchi, Y. Production of a bioflocculant by Rhodococcus erythropolis S-1 grown on alcohols. Biosci. Biotechnol. Biochem. 1994, 58, 428–429. [Google Scholar] [CrossRef]
- Lee, S.H.; Lee, S.O.; Jang, K.L.; Lee, T.H. Microbial flocculant from Arcuadendron sp. TS-49. Biotechnol. Lett. 1995, 17, 95–100. [Google Scholar] [CrossRef]
- Yokoi, H.; Shiraki, M.; Hirose, J.; Hayashi, S.; Takasaki, Y. Flocculation properties of xanthan produced by Xanthomonas campestris. Biotechnol. Tech. 1996, 10, 789–792. [Google Scholar] [CrossRef]
- Morgan, J.; Forster, C.; Evison, L. A comparative study of the nature of biopolymers extracted from anaerobic and activated sludges. Water Res. 1990, 24, 743–750. [Google Scholar] [CrossRef]
- More, T.; Yan, S.; Hoang, N.; Tyagi, R.; Surampalli, R. Bacterial polymer production using pre-treated sludge as raw material and its flocculation and dewatering potential. Bioresour. Technol. 2012, 121, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Frølund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res. 1996, 30, 1749–1758. [Google Scholar] [CrossRef]
- Sesay, M.L.; Özcengiz, G.; Sanin, F.D. Enzymatic extraction of activated sludge extracellular polymers and implications on bioflocculation. Water Res. 2006, 40, 1359–1366. [Google Scholar] [CrossRef] [PubMed]
- Batta, N.; Subudhi, S.; Lal, B.; Devi, A. Isolation of a lead tolerant novel bacterial species, Achromobacter sp. TL-3: Assessment of bioflocculant activity. Indian J. Exp. Biol. 2013, 51, 1004–1011. [Google Scholar] [PubMed]
- Guo, J.; Yang, C.; Zeng, G. Treatment of swine wastewater using chemically modified zeolite and bioflocculant from activated sludge. Bioresour. Technol. 2013, 143, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Zhao, X.Q.; Guo, S.L.; Alam, M.A.; Bai, F.W. Bioflocculant production from Solibacillus silvestris W01 and its application in cost-effective harvest of marine microalga Nannochloropsis oceanica by flocculation. Bioresour. Technol. 2013, 135, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ma, F.; Lee, D.J.; Wang, A.; Ren, N. Bioflocculants from hydrolysates of corn stover using isolated strain Ochrobactium ciceri W2. Bioresour. Technol. 2013, 145, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lee, D.J.; Ma, F.; Wang, A.; Ren, N. Bioflocculants from isolated strain or mixed culture: Role of phosphate salts and Ca2+ ions. J. Taiwan Inst. Chem. Eng. 2014, 45, 527–532. [Google Scholar] [CrossRef]
- Fujita, M.; Ike, M.; Jang, J.H.; Kim, S.M.; Hirao, T. Bioflocculation production from lower-molecular fatty acids as a novel strategy for utilization of sludge digestion liquor. Water Sci. Technol. 2001, 44, 237–243. [Google Scholar] [PubMed]
- Sponza, D.T. Investigation of extracellular polymer substances (EPS) and physicochemical properties of different activated sludge flocs under steady-state conditions. Enzym. Microb. Technol. 2003, 32, 375–385. [Google Scholar] [CrossRef]
- Ye, F.; Peng, G.; Li, Y. Influences of influent carbon source on extracellular polymeric substances (EPS) and physicochemical properties of activated sludge. Chemosphere 2011, 84, 1250–1255. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.G.; Gong, W.X.; Liu, X.W.; Tian, L.; Yue, Q.Y.; Gao, B.Y. Production of a novel bioflocculant by culture of Klebsiella mobilis using dairy wastewater. Biochem. Eng. J. 2007, 36, 81–86. [Google Scholar] [CrossRef]
- Zhang, Z.Q.; Bo, L.; Xia, S.Q.; Wang, X.J.; Yang, A.M. Production and application of a novel bioflocculant by multiple-microorganism consortia using brewery wastewater as carbon source. J. Environ. Sci. 2007, 19, 667–673. [Google Scholar] [CrossRef]
- Peng, L.; Yang, C.; Zeng, G.; Wang, L.; Dai, C.; Long, Z. Characterization and application of bioflocculant prepared by Rhodococcus erythropolis using sludge and livestock wastewater as cheap culture media. Appl. Microbiol. Biotechnol. 2014, 98, 6847–6858. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Ren, N.; Wang, A.; Ma, F.; Gao, L.; Peng, Y. Use of waste fermenting liquor to produce bioflocculants with isolated strains. Int. J. Hydrogen Energy 2008, 33, 3295–3301. [Google Scholar] [CrossRef]
- Pagilla, K.; Sood, A.; Kim, H. Gordonia (Nocardia) amarae foaming due to biosurfactant production. Water Sci. Technol. 2002, 46, 519–524. [Google Scholar] [PubMed]
- Desai, J.D.; Banat, I.M. Microbial production of surfactants and their commercial potential. Microbiol. Mol. Biol. Rev. 1997, 61, 47–64. [Google Scholar] [PubMed]
- Cameotra, S.; Makkar, R. Synthesis of biosurfactants in extreme conditions. Appl. Microbiol. Biotechnol. 1998, 50, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Kosaric, N. Biosurfactants in industry. Pure Appl. Chem. 1992, 64, 1731–1737. [Google Scholar] [CrossRef]
- Rosenberg, E.; Ron, E. High-and low-molecular-mass microbial surfactants. Appl. Microbiol. Biotechnol. 1999, 52, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Deleu, M.; Paquot, M. From renewable vegetables resources to microorganisms: New trends in surfactants. Comptes Rendus Chimie 2004, 7, 641–646. [Google Scholar] [CrossRef]
- Makkar, R.; Cameotra, S. An update on the use of unconventional substrates for biosurfactant production and their new applications. Appl. Microbiol. Biotechnol. 2002, 58, 428–434. [Google Scholar] [PubMed]
- Gallert, C.; Winter, J. Solid and liquid residues as raw materials for biotechnology. Naturwissenschaften 2002, 89, 483–496. [Google Scholar] [CrossRef] [PubMed]
- Becerra, F.Y.G.; Acosta, E.J.; Allen, D.G. Alkaline extraction of wastewater activated sludge biosolids. Bioresour. Technol. 2010, 101, 6972–6980. [Google Scholar] [CrossRef] [PubMed]
- Becerra, F.Y.G.; Allen, D.G.; Acosta, E.J. Surfactant-like properties of alkaline extracts from wastewater biosolids. J. Surfactants Deterg. 2010, 13, 261–271. [Google Scholar] [CrossRef]
- Baxter, M.D.; Acosta, E.; Montoneri, E.; Tabasso, S. Waste biomass-extracted surfactants for heavy oil removal. Ind. Eng. Chem. Res. 2014, 53, 3612–3621. [Google Scholar] [CrossRef]
- Jardine, I.; Scanlan, G.; McNeil, M.; Brennan, P.J. Plasma desorption mass spectrometric analysis of mycobacterial glycolipids. Anal. Chem. 1989, 61, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T.; Kato, I. Novel methyl rhamnolipids from Pseudomonas aeruginosa. FEBS Lett. 1982, 139, 81–85. [Google Scholar] [CrossRef]
- Nitschke, M.; Pastore, G.M. Cassava flour wastewater as a substrate for biosurfactant production. Appl. Biochem. Biotechnol. 2003, 106, 295–301. [Google Scholar] [CrossRef]
- Manivasagan, P.; Kang, K.H.; Kim, D.G.; Kim, S.K. Production of polysaccharide-based bioflocculant for the synthesis of silver nanoparticles by Streptomyces sp. Int. J. Biol. Macromol. 2015, 77, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Comte, S.; Guibaud, G.; Baudu, M. Relations between extraction protocols for activated sludge extracellular polymeric substances (EPS) and EPS complexation properties: Part I. Comparison of the efficiency of eight EPS extraction methods. Enzym. Microb. Technol. 2006, 38, 237–245. [Google Scholar] [CrossRef]
- Sheng, G.P.; Yu, H.Q.; Li, X.Y. Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: A review. Biotechnol. Adv. 2010, 28, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Wingender, J.; Neu, T.R.; Flemming, H.C. Microbial Extracellular Polymeric Substances: Characterization, Structure and Function; Springer Science & Business Media: Berlin, Germany, 2012. [Google Scholar]
- Adav, S.S.; Lee, D.J. Extraction of extracellular polymeric substances from aerobic granule with compact interior structure. J. Hazard. Mater. 2008, 154, 1120–1126. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, N.S.; Rohani, S. Lipid extraction and biodiesel production from municipal sewage sludges: A review. Renew. Sustain. Energy Rev. 2011, 15, 1067–1072. [Google Scholar]
- Kalogo, Y.; Monteith, H. State of Science Report: Energy and Resource Recovery from Sludge; Water Environment Research Foundation: Alexandria, VA, USA, 2008. [Google Scholar]

| Amino Acid | WAS | Soybean Meal | White Fish Meal | FAO Ref. Protein | Wheat | Whole Egg |
|---|---|---|---|---|---|---|
| Alanine | 7.3 | - | - | - | - | - |
| Glycine | 4.9 | - | - | - | - | - |
| Valine | 4.1 | 5.2 | 4.7 | 4.2 | 4.4 | 7.3 |
| Threonine | 4.2 | 4.4 | 3.8 | 2.8 | 2.9 | 5.1 |
| Serine | 3.4 | - | - | - | - | - |
| Leucine | 5.6 | 7.6 | 6.5 | 4.8 | 6.7 | 8.9 |
| Isoleucine | 2.7 | 5.8 | 3.9 | 4.2 | 3.3 | 6.7 |
| Proline | 3.1 | - | - | - | - | - |
| Methionine | 1.45 | 1.3 | 2.9 | 2.2 | 1.5 | 3.2 |
| Aspatic acid | 8.3 | - | - | - | - | - |
| Phenylalanine | 3.1 | 5.3 | 3.5 | 2.8 | 4.5 | 5.8 |
| Glutamic acid | 8.1 | - | - | - | - | - |
| Lysine | 3.3 | 6.6 | 7.6 | 4.2 | 2.8 | 6.5 |
| Tyrosine | 2.4 | 4.1 | 3 | 2.8 | - | - |
| Arginine | 2.9 | 7.3 | 6.8 | - | - | - |
| Histidine | 0.6 | 2.7 | 2 | - | - | - |
| Cystine | 2.1 | 1.2 | 0.7 | 2 | 2.5 | 2.4 |
| Tryptophan | 0.8 | 1.3 | 0.9 | 1.4 | 1.1 | 1.6 |
| SCFAs Composition | Yield | Fermentation Conditions | Medium | References |
|---|---|---|---|---|
| Acetic, butyric, valeric and propionic acid | Max 520.1 mg/g VSS | 21 ± 1 °C, pH 8, 8 days | Wastewater sludge | [48] |
| Acetic, butyric, valeric and propionic acid | Max 2599.1 mg/L | 21 ± 1 °C, pH 7.2, 6 days | Wastewater sludge | [37] |
| Acetic, butyric, valeric and propionic acid | Max 319.8 mg/g VSS | 15–55 °C, pH 10 ± 0.3, 48 days | Wastewater sludge | [49] |
| Acetic, butyric, valeric and propionic acid | 450–790 mg/g VSS | 55 °C, pH 7, 1 rpm, 28 days | Wastewater and bagasse | [34] |
| Acetic, butyric, valeric and propionic acid | Max 284 mg/g VSS | 35 ± 1 °C, pH 7, 200 rpm, 7 days | Wastewater sludge | [35] |
| Acetic, butyric, valeric and propionic acid | 345.49–2708.02 mg/L | 21 ± 1 °C, pH 4–11, 80 rpm, 20 days | Wastewater sludge | [31] |
| Acetic, butyric, valeric and propionic acid | Max 256.2 mg/g VSS | 20–22 °C, pH 4–11, 20 days | Wastewater sludge | [30] |
| Acetic, butyric, valeric and propionic acid | Max 368.71 g/kg TS | 35 ± 1 °C, C/N ratio 26 to 7, 24 days | Wastewater sludge and perennial ryegrass | [50] |
| Acetic, butyric, valeric and propionic acid | 704.9–1994.7 mg/L | 20 ± 2 °C, pH 7–10, 70 rpm, 15 days | Wastewater sludge | [33] |
| Acetic, butyric, valeric and propionic acid | 18.1–539.4 mg/L | 21 ± 2°C, 2 days | Wastewater sludge and potato-processing wastewater | [51] |
| Enzymes Type and Activities | Extraction Techniques and Conditions | Medium | References |
|---|---|---|---|
| Protease: max. ≈3 μmol min−1 g−1 VSS α-amylase: max. ≈20 μmol min−1 g−1 VSS α-glucosidase: max. >1 μmol min−1 g−1 VSS Alkaline phosphatase: max. ≈9 μmol min−1 g−1 VSS Acid phosphatase: max. ≈3 μmol min−1 g−1 VSS | Ultrasound 20–40 kHz, 2–20 min, 138–690 W/g VSS, pH 7 and EDTA 2% at 4 °C for 3 h | Wastewater sludge | [56] |
| Protease: 1.06–28.2 μmol min-1 g-1 VSS α-amylase: 10.2–14.9 μmol min−1 g−1 VSS α-glucosidase: 188–319 μmol min−1 g−1 VSS | Ultrasound 40 kHz, 120 W, 2 min, 0–15 kW/L, pH 8 and 2% EDTA, 36.5% formaldehyde, CER 70 g/g VSS at 4 °C for 1 h | Wastewater sludge | [55] |
| Protease: max. ≈37 μmol min−1 g−1 VSS α-amylase: max. ≈34 μmol min−1 g−1 VSS α-glucosidase: max. ≈3 μmol min−1 g-1 VSS Alkaline phosphatase: max. ≈16 μmol min−1 g−1 VSS Acid phosphatase: max. ≈16 μmol min−1 g−1 VSS | Ultrasound 20 kHz, 0–20 min, 0–15 kW/L, pH 7, 4 °C | Wastewater sludge | [32] |
| Protease: max 52 units/g VSS Lipase: max 22.9 ± 0.8 units/g VSS | Ultrasound 24 kHz, 3.9 W/cm2, 20 min, 5 ± 1 °C, and Triton X100 0.1 to 2% (v/v) | Wastewater sludge | [58] |
| Protease: max 57.4 units/g VSS Lipase: max 21.4 units/g VSS | Ultrasound 20–24 kHz, 3–8 W/cm2, 1–60 min, pH 7–8, 5 ± 1 °C, TX 100 0.1 to 10% (v/v), CER 0.24–0.8 g/mL | Wastewater sludge | [57] |
| Protease: max. ≈4,000 units/g VSS Lipase: 108.4–335 units/g VSS | 1 h 900 rpm stirring followed by 30 min sonication (200 W + 15 kHz), pH 8, 0–2% Triton X-100 and EDTA, CER 60–70 g/g VSS | Wastewater sludge | [60] |
| Protease: 3450 ± 124 units/g mixed liquor suspended solids (MLSS) Amylase: 111 ± 5 units/g MLSS Glucosidase: 59.5 ± 2.9 units/g MLSS Lipase: 8.8 ± 1.4 × 10−2 units/g MLSS Dehydrogenase: 36.6 ± 3.8 × 10−3 units/g MLSS | Agitation 200–4200 rpm, 4 °C, 1–10 min and ammonium sulphate | Wastewater and laboratory-cultivated sludge | [61] |
| Type | Entomotoxicity | Fermentation Conditions | Medium | References |
|---|---|---|---|---|
| Bacillus thuringiensis | 9534–13,020 IU/μL | 30 °C, pH 6.9 ± 0.1, 400 rpm, inoculum concentration 2% (v/v) | Tryptic soy yeast broth and sludge | [70] |
| Bacillus thuringiensis | 8300–10,813 IU/μL | 30 °C, pH 7.0 ± 0.1, 300 rpm, inoculum concentration 2% (v/v), C/N ratio 7.9–9.9, total solids up to 33 g/L | Tryptic soy yeast broth and sludge | [68] |
| Bacillus thuringiensis | 8115–12,970 IU/μL | 31 °C, 250 rpm, inoculum concentration 1–5% (v/v), total solids 10–46 g/L | Tryptic soy yeast broth and sludge | [67] |
| Bacillus thuringiensis | 900–4100 IU/μL | 30 °C, pH 7.0, 250 rpm | Wastewater sludge | [72] |
| Bacillus thuringiensis | 3400–10,000 IU/μL | 30–36 °C, pH 7.0, 50–350 rpm, inoculum concentration 5% (v/v), total solids up to 13 g/L | Wastewater sludge | [69] |
| Bacillus thuringiensis | 4500–10,840 IU/μL | 31 °C, pH 7.0, 250 rpm, inoculum concentration 1% and 5% (v/v) | Wastewater sludge | [66] |
| Trichoderma sp. | 6278–15,036 SBU/μL | 28 ± 1 °C, pH 6.0 ± 0.01, 250 ± 5 rpm, total solids 10–50 g/L | Wastewater sludge | [73] |
| Organism | PHA Yield (mg/g Biomass) | Fermentation Conditions | Medium | Additional Carbon Source | References |
|---|---|---|---|---|---|
| Unspecified | - | pH 7, 25 °C, solids concentration 15 g/L, 0.3–0.5 vvm aeration, 60 h SRT | Paper and pulp wastewater | Acetate | [91] |
| Unspecified | 111 | pH 7, 30 °C, C/N ratio 20–140, 300 rpm shaking, 48 h SRT | Domestic WAS | Valeric acid | [78,86] |
| Bacillus | - | 37 °C, 250 rpm shaking, 18 h SRT | Beer brewery WAS | None | [80] |
| Unspecified | 792 | Solids concentration 3 g/L, C/N ratio 144, 30 °C, 150 rpm shaking, 48 and 96 h SRT | Dairy and food processing WAS | Acetic acids | [87,92] |
| Alcaligenes eutrophus | - | pH 7, 30 °C, 50 rpm shaking, 0.15 vvm aeration | Domestic WAS | None | [93] |
| Unspecified | - | pH 7.5, 15–35 °C, 450 rpm shaking, 0.29 vvm aeration, 60 h SRT | Domestic WAS | Acetate | [85] |
| Halomonas boliviensis | - | pH 7.5, inoculum size 2%, 35 °C, 850–1300 rpm stirring | Bakery WAS and seawater | None | [94] |
| Bio-Flocculants Composition and Yield | Organisms | Medium | Fermentation Conditions | Reference |
|---|---|---|---|---|
| Polysaccharide, 2.58 g/L | Klebsiella mobilis | Dairy wastewater | Inoculum size 5% (v/v), pH 6.0, 30 °C | [115] |
| Polysaccharide and protein, 15 g/L | Staphylococcus sp. and Pseudomonas sp. | Brewery wastewater | Inoculum size 2% (v/v), pH 6.0, 30 °C and shaking speed 160 rpm | [116] |
| 91.2% polysaccharide, 7.6% protein and 1.2% DNA, 1.6 g/L | Rhodococcus erythropolis | Livestock wastewater | Inoculum size 2% (v/v), pH 7, Na2HPO4 0.5 g/L | [117] |
| Unspecified, 1.5–3.4 g/L | Serratia sp. | Pretreated WAS | Inoculum size 3% (v/v), 25 °C, shaking speeding 250 rpm, 72 h | [104] |
| Carbohydrates (91% w/w), 2.1 g/L | Bacillus subtilis, Bacillus fusiformis, Bacillus flexus | WAS digestion liquor | Inoculum size 10% (v/v), pH 4–5, 24 h | [118] |
| Chitosan-like polymer, 4.6 g/L | Citrobacter sp. | WAS digestion liquor | pH 8.5, 30 °C, shaking speed 120 rpm | [112] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, W.; Alvarez-Gaitan, J.P.; Dastyar, W.; Saint, C.P.; Zhao, M.; Short, M.D. Value-Added Products Derived from Waste Activated Sludge: A Biorefinery Perspective. Water 2018, 10, 545. https://doi.org/10.3390/w10050545
Zhang W, Alvarez-Gaitan JP, Dastyar W, Saint CP, Zhao M, Short MD. Value-Added Products Derived from Waste Activated Sludge: A Biorefinery Perspective. Water. 2018; 10(5):545. https://doi.org/10.3390/w10050545
Chicago/Turabian StyleZhang, Wei, Juan Pablo Alvarez-Gaitan, Wafa Dastyar, Christopher P. Saint, Ming Zhao, and Michael D. Short. 2018. "Value-Added Products Derived from Waste Activated Sludge: A Biorefinery Perspective" Water 10, no. 5: 545. https://doi.org/10.3390/w10050545
APA StyleZhang, W., Alvarez-Gaitan, J. P., Dastyar, W., Saint, C. P., Zhao, M., & Short, M. D. (2018). Value-Added Products Derived from Waste Activated Sludge: A Biorefinery Perspective. Water, 10(5), 545. https://doi.org/10.3390/w10050545






