Treatability of a Highly-Impaired, Saline Surface Water for Potential Urban Water Use
Abstract
:1. Introduction
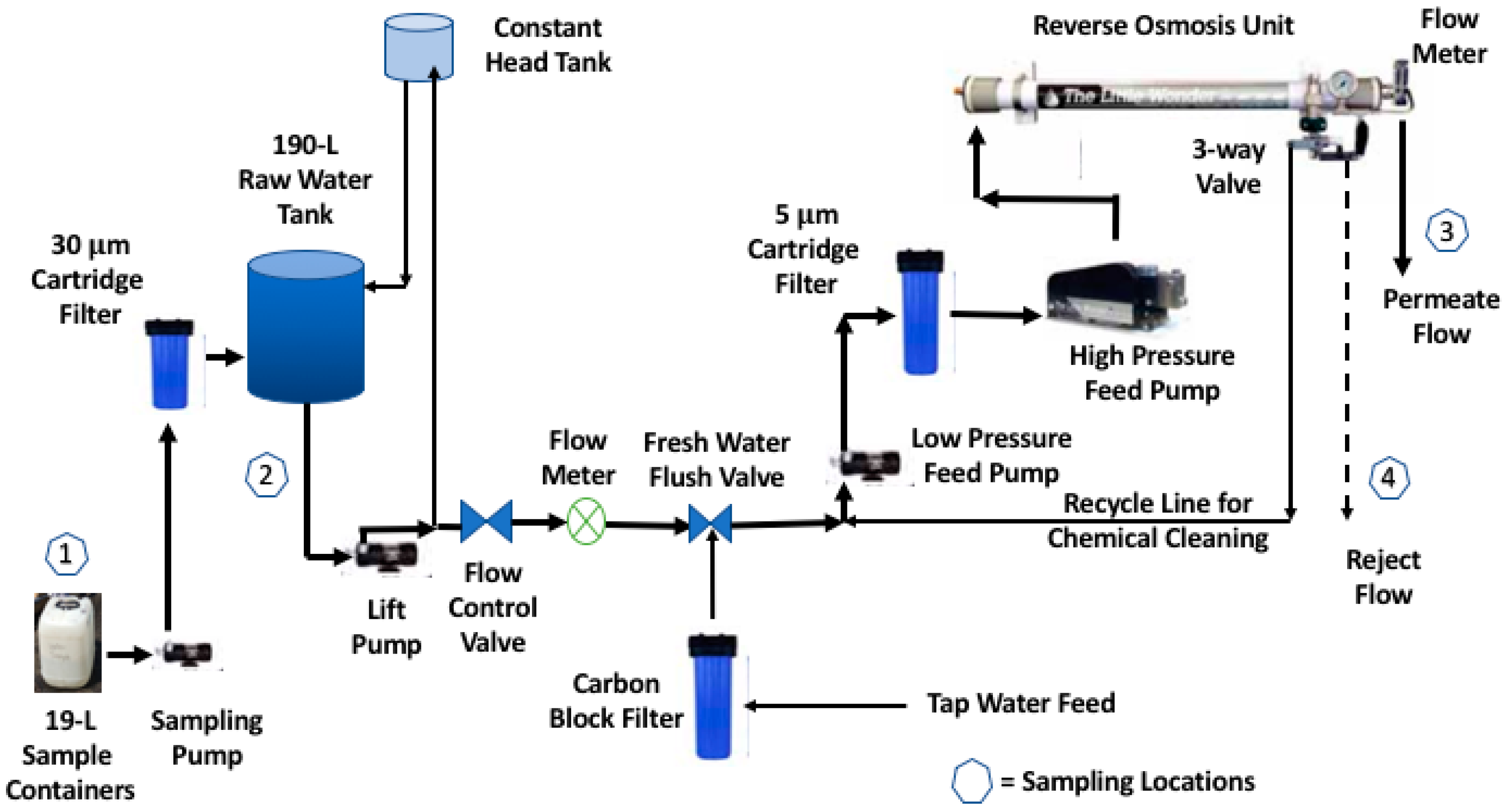
2. Materials and Methods
2.1. Cartridge Filtration
2.2 Coagulation
2.2.1. Jar Testing
2.2.2. Bench RO Treatment
3. Analytical Methods
4. Results and Discussion
4.1. Cartridge Filtration
4.2. Water Quality Test Results
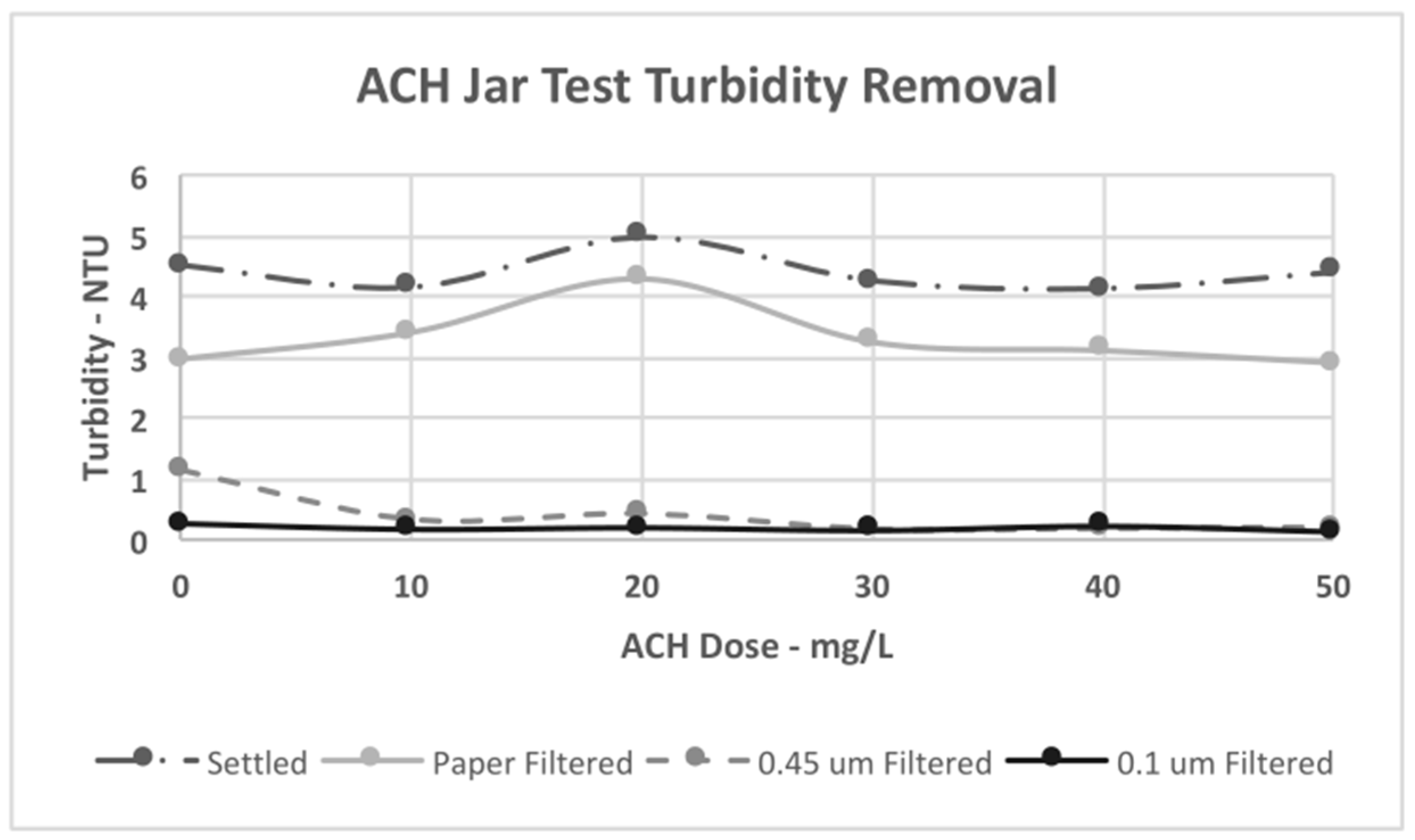
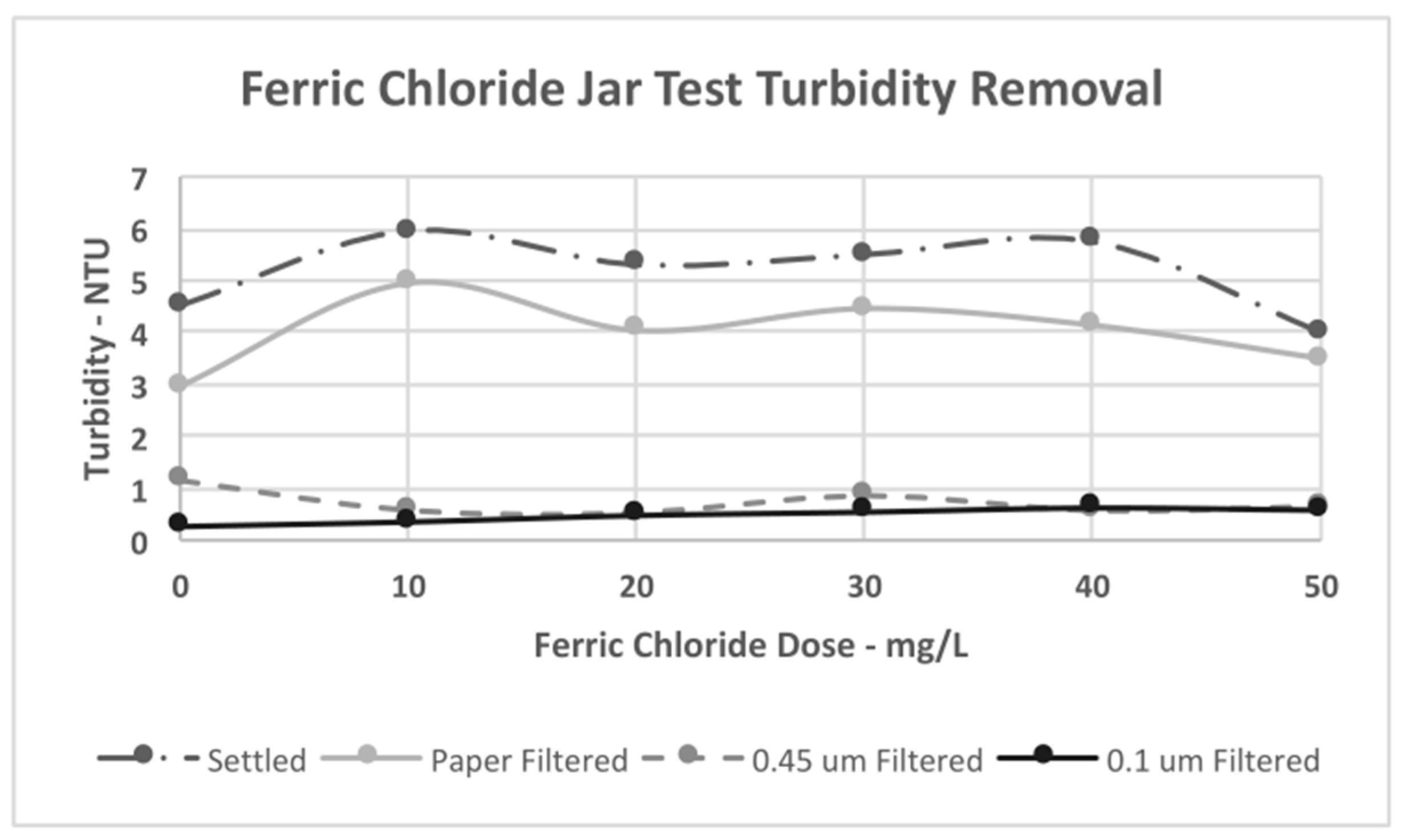
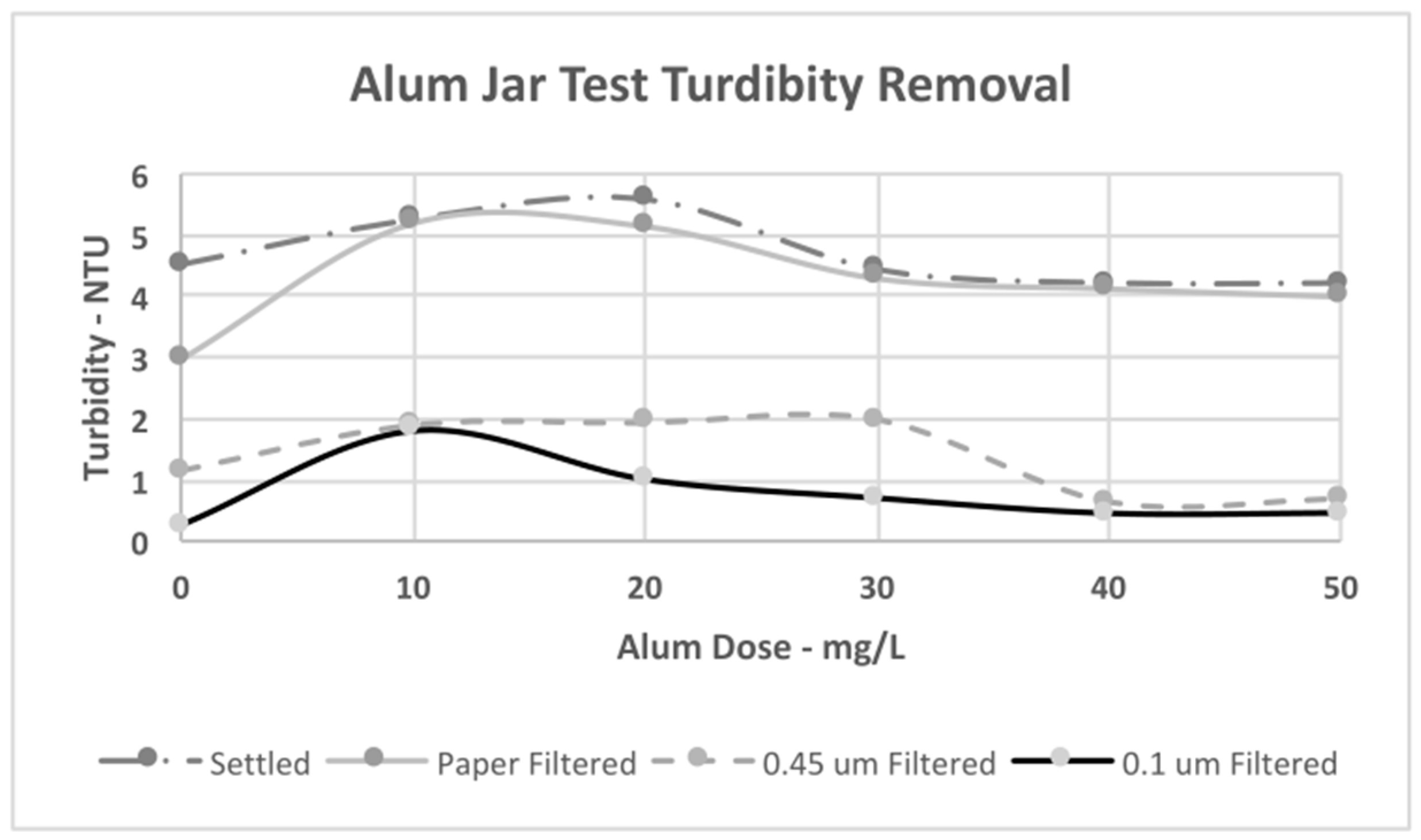
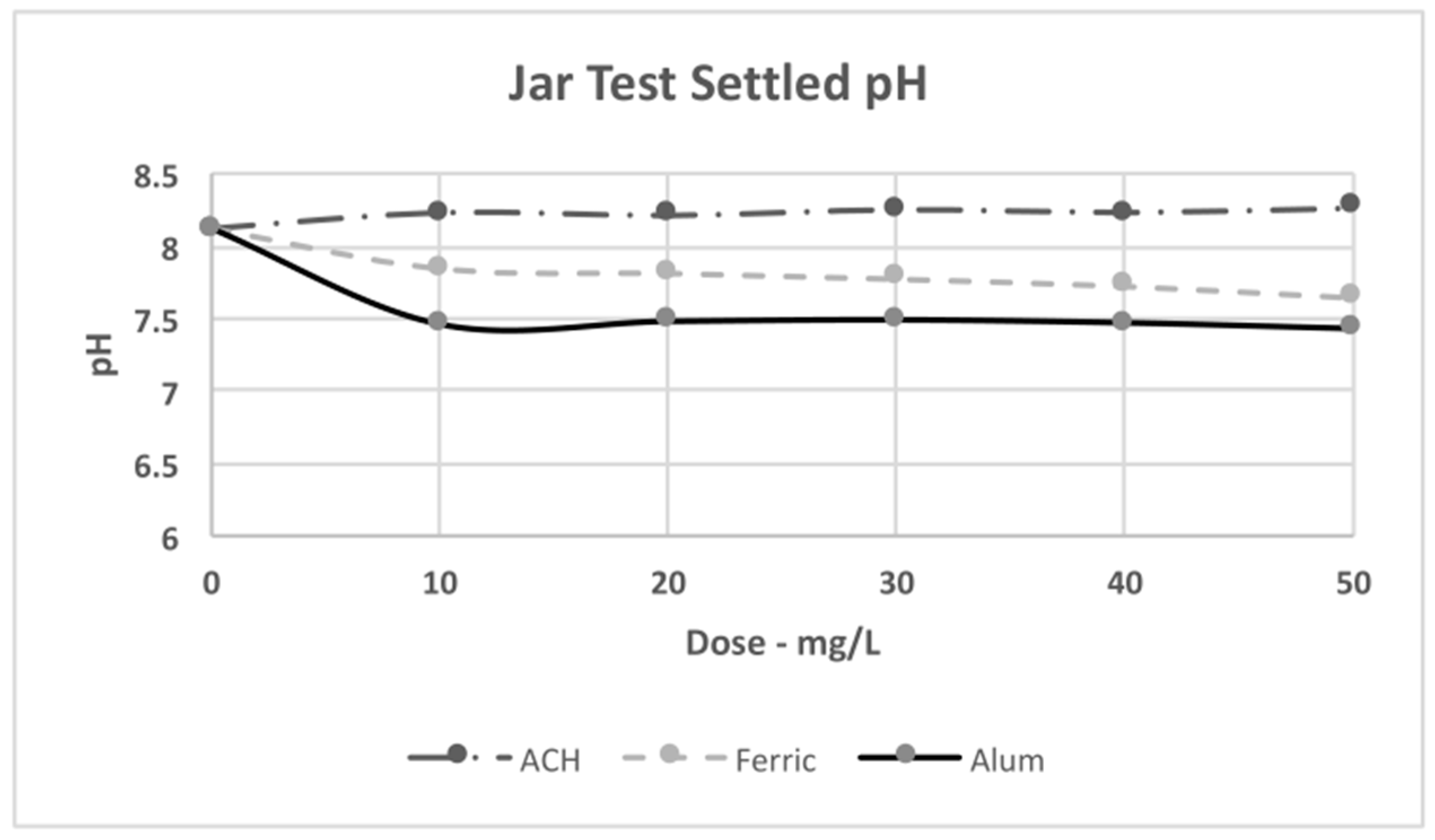
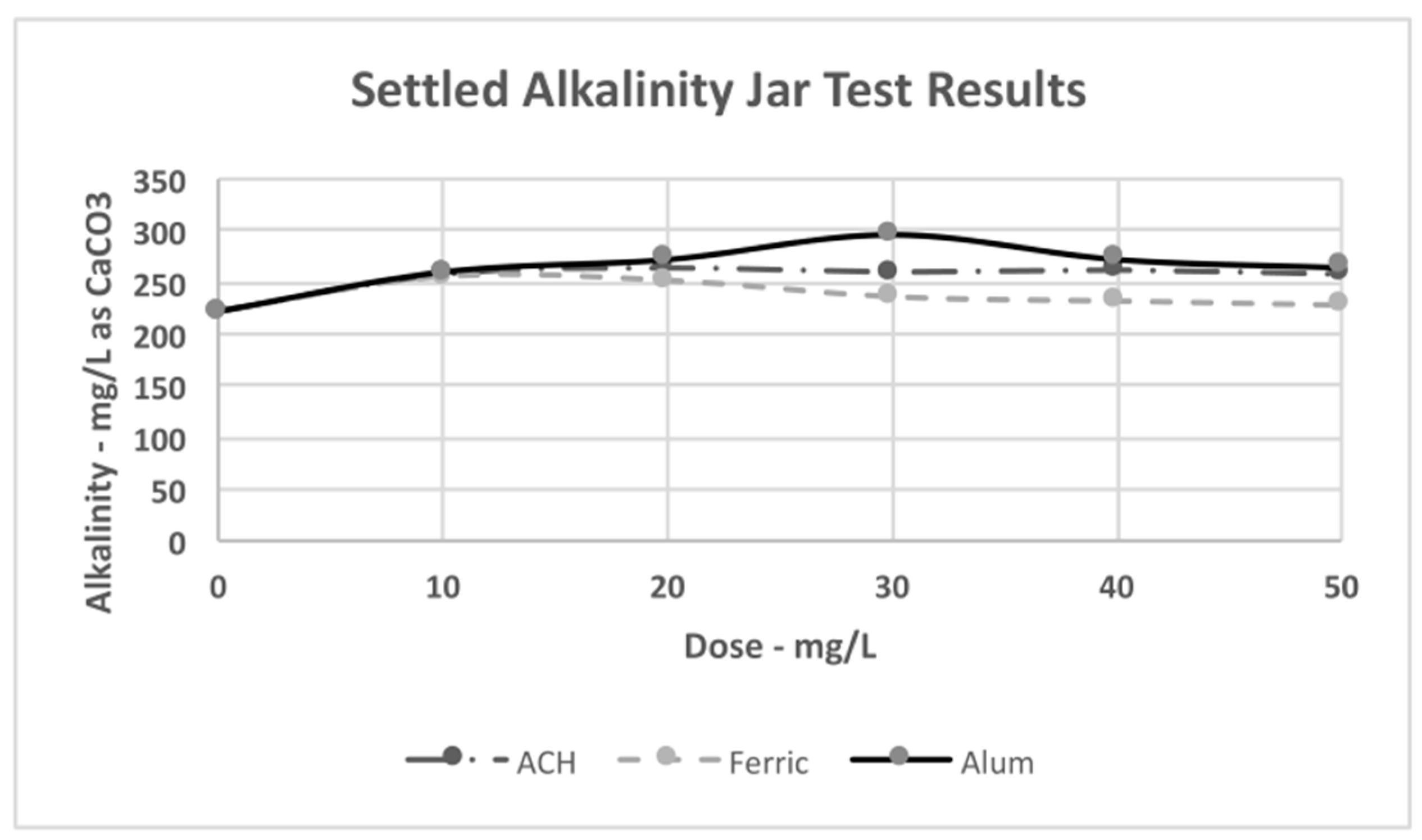
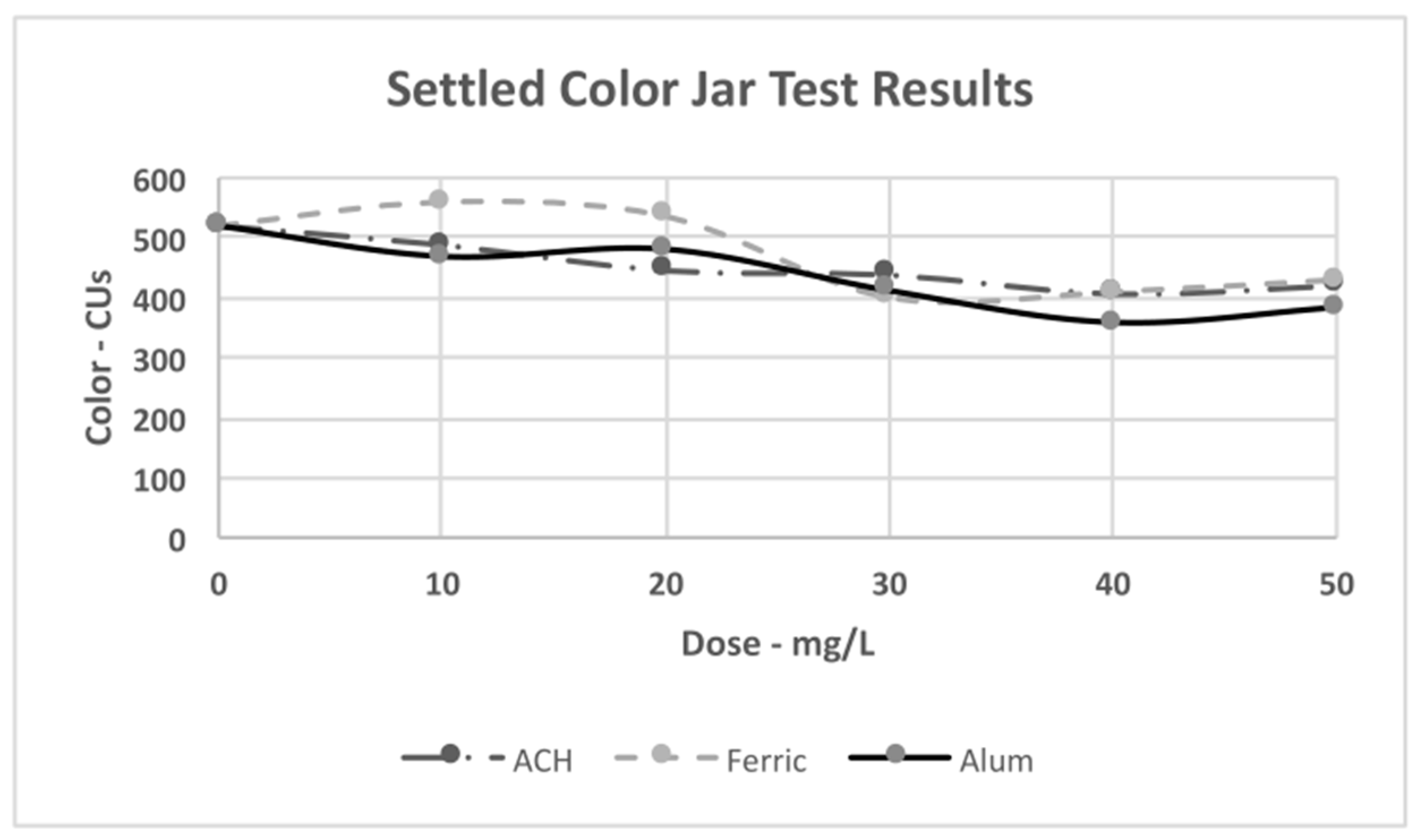
4.3. Jar Test Results
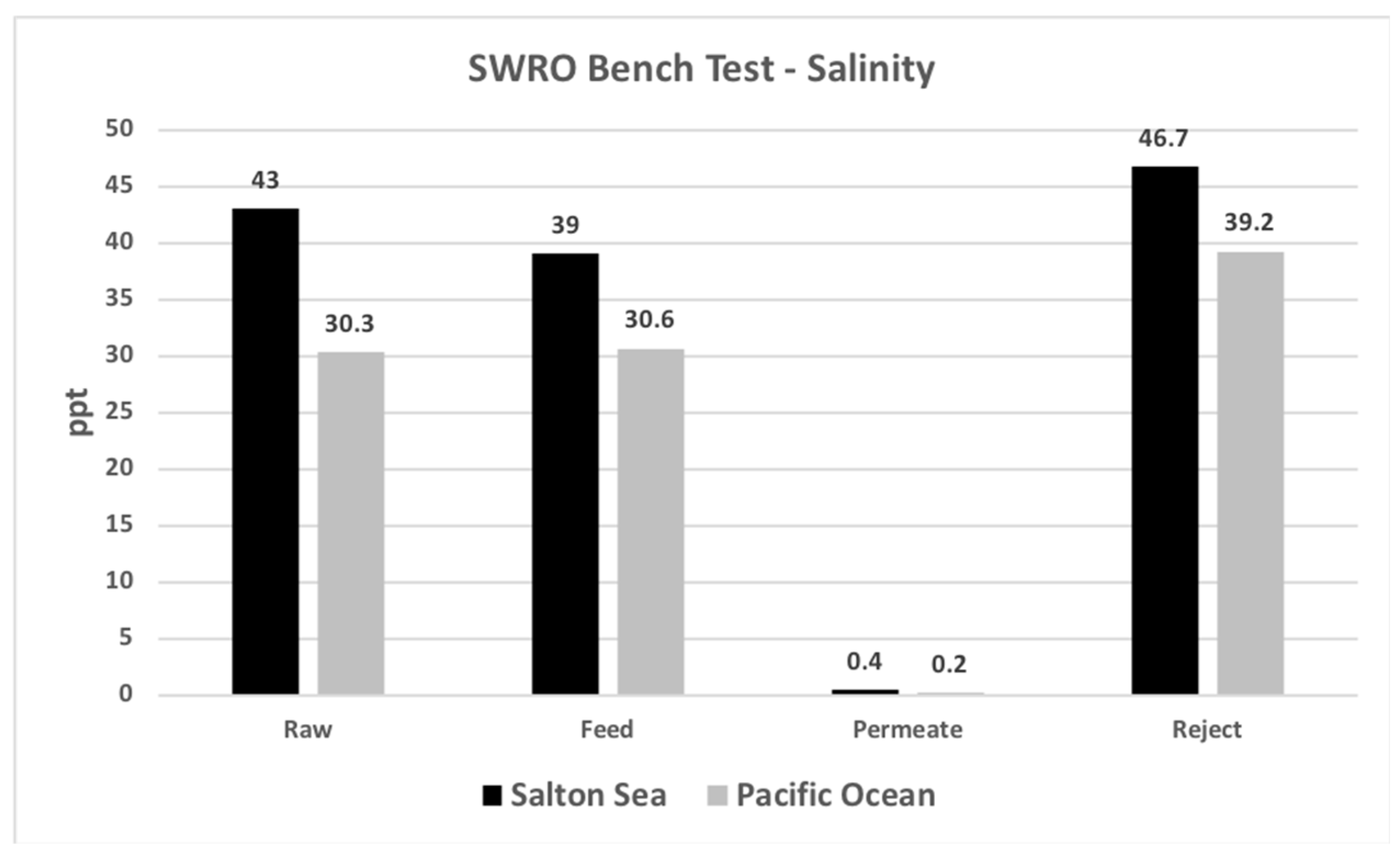
4.4. SWRO Treatability Results
5. Discussion
Acknowledgments
Conflicts of Interest
References
- Dugan, H.A.; Bartlett, S.L.; Burke, S.M.; Doubek, J.P.; Krivak-Tetley, F.E.; Skaff, N.K.; Summers, J.C.; Farrell, K.J.; McCullough, I.M.; Morales-Williams, A.M.; et al. Salting our freshwater lakes. Proc. Natl. Acad. Sci. USA 2017, 114, 4453–4458. [Google Scholar] [CrossRef] [PubMed]
- Ghaly, F.M. Role of natural vegetation in improving salt affected soil in northern Egypt. Soil Tillage Res. 2002, 64, 173–178. [Google Scholar] [CrossRef]
- Jenke, A.L. Evaluation of Salinity Created by Irrigation Return Flows; EPA 430/9-74-006; USEPA Office of Water Programs: Washington, DC, USA, 1974.
- Yihdego, Y.; Webb, J. Modelling of seasonal and long-term trends in lake salinity in southwestern Victoria, Australia. J. Environ. Manag. 2012, 112, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Tran, Q.K.; Jassby, D.; Schwabe, K.A. The implications of drought and water conservation on the reuse of municipal wastewater: Recognizing impacts and identifying mitigation possibilities. Water Res. 2017, 124, 472–481. [Google Scholar] [CrossRef] [PubMed]
- California Department of Water Resources. Salton Sea Ecosystem Restoration Program. Available online: www.water.ca.gov/saltonsea/ (accessed on 3 January 2018).
- James, I. California has a new $383 million plan for the shrinking Salton Sea. The Desert Sun. 16 March 2017. Available online: http://www.desertsun.com/story/news/environment/2017/03/16/california-has-new-383-million-plan-shrinking-salton-sea/99124850/ (accessed on 9 January 2018).
- Dillon, J.G.; McMath, L.M.; Trout, A.L. Seasonal Changes in bacterial diversity in the Salton Sea. Hydrobiologia 2009, 632, 49–64. [Google Scholar] [CrossRef]
- De Koff, J.P.; Anderson, M.A.; Amrhein, C. Geochemistry of iron in the Salton Sea, California. Hydrobiologia 2008, 604, 111–121. [Google Scholar] [CrossRef]
- LeBlanc, L.A.; Kuivila, K.M. Occurrence, distribution and transport of pesticides into the Salton Sea Basin, California, 2001–2002. Hydrobiologia 2008, 604, 151–172. [Google Scholar] [CrossRef]
- Reese, B.K.; Anderson, M.A.; Amrheim, C. Hydrogen sulfide production and volatilization in a polymictic eutrophic saline lake, Salton Sea, California. Sci. Total Environ. 2008, 406, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.J. Hazard’s Toll: The Costs of Inaction at the Salton Sea; Pacific Institute: Oakland, CA, USA, 2014; Available online: http://pacinst.org/publication/hazards-toll/ (accessed on 14 February 2018).
- US Bureau of Reclamation. Restoration of the Salton Sea; Summary Report; US Department of the Interior: Washington, DC, USA, 2007.
- Tetra Tech, Inc. Review of US Filter Corporation Salton River Proposal Final Report; TC 10138-41; Tetra Tech, Inc.: Pasadena, CA, USA, 2003. [Google Scholar]
- Solis, G. How Water from Mexico Can Save the Salton Sea. The Desert Sun, Undated. Available online: https://www.desertsun.com/story/news/environment/2016/03/23/how-water-mexico-can-save-salton-sea/82163024/ (accessed on 16 February 2018).
- California Water Boards. “State Water Board Adopts Order on Long-Term Management of Salton Sea.” Press Release. 7 November 2017. Available online: https://www.waterboards.ca.gov/press_room/press_releases/2017/pr110717_salton_sea.pdf (accessed on 16 February 2018).
- Global Water Intelligence. Desalination Markets 2016; Media Analytics Ltd.: Oxford, UK, 2016; Available online: https://www.desaldata.com/desalination-markets-2016 (accessed on 9 January 2018).
- UNESCO. Energy Requirements of Desalination Processes. Encyclopedia of Desalination and Water Resources (DESWARE). Available online: http://www.desware.net/Energy-Requirements-Desalination-Processes.aspx (accessed on 15 February 2018).
- US Bureau of Reclamation. Salton Sea Projects Map. 16 April 2015. Available online: https://usbr.gov/lc/region/programs/SaltonSeaProjectsMap.pdf (accessed on 15 February 2018).
- Younker, L.W.; Kasameyer, P.W.; Tewhey, J.D. Geological, Geophysical, and Thermal Characteristics of the Salton Sea Geothermal Field, California. J. Vulcanol. Geotherm. Res. 1982, 12, 221–258. [Google Scholar] [CrossRef]
- McGuire, J.J.; Lohman, R.B.; Catchings, R.D.; Rymer, M.J.; Goldman, M.R. Relationships among seismic velocity, metamorphism, and seismic and aseismic fault slip in the Salton Sea Geothermal Field region. J. Geophys. Res. Solid Earth 2015, 120, 2600–2615. [Google Scholar] [CrossRef]
- Penn, I. Australian Firm Plans Nation’s Largest Geothermal Plant in Imperial Valley. Los Angeles Times. 14 January 2017. Available online: http://www.latimes.com/business/la-fi-geothermal-salton-sea-20170114-story.html (accessed on 15 February 2018).
- ASTM D 2035-08. Standard Practice for Coagulation-Flocculation Jar Test of Water; American Society of Testing and Materials: West Conshohocken, PA, USA, 2008. [Google Scholar]
- Parker Hannifin Corporation, Racor Division/Village Marine Tec., Carson, California. Available online: www.villagemarine.com (accessed on 9 January 2018).
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Methods Approved to Analyze Drinking Water Samples to Ensure Compliance with Regulations. Available online: https://www.epa.gov/dwanalyticalmethods (accessed on 9 January 2018).
- Hach. Loveland, Colorado. Available online: www.hach.com/wateranalysis (accessed on 9 January 2018).
- Micrology Laboratories. Goshen, Indiana. Available online: www.micrologylabs.com (accessed on 9 January 2018).
- Rodriguez, I.R.; Amrhein, C.; Anderson, M.A. Reducing dissolved phosphorus loading to the Salton Sea with aluminum sulfate. Hydrobiologia 2008, 604, 37–44. [Google Scholar] [CrossRef]
- Holdren, G.C.; Montaño, A. Chemical and physical characteristics of the Salton Sea, California. Hydrobiologia 2002, 473, 1–21. [Google Scholar] [CrossRef]
- Rodriguez, I.R.; Amrhein, C.; Anderson, M.A. Laboratory studies on the coprecipitation of phosphate with calcium carbonate in the Salton Sea, California. Hydrobiologia 2008, 604, 45–55. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA). Edition of the Drinking Water Standards and Health Advisories; EPA 822-S-12-001; Office of Water: Washington, DC, USA, 2012.
- Pontius, F.W. Regulatory Compliance Planning to Ensure Water Supply Safety. J. AWWA 2002, 94, 52–64. [Google Scholar] [CrossRef]
- Morillo, J.U. Comparative study of brine management technologies for desalination plants. Desalination 2014, 336, 32–49. [Google Scholar] [CrossRef]
- Voutchkov, N. Overview of seawater concentrate disposal alternatives. Desalination 2011, 273, 205–219. [Google Scholar] [CrossRef]
- Global Water Intelligence. IDA Desalination Yearbook Water Desalination Report 2017–2018; Media Analytics Ltd.: Oxford, UK, 2017. [Google Scholar]
- San Diego County Water Authority. Fact Sheet: Seawater Desalination; The Claude “Bud” Lewis Desalination Plant and Related Facilities. October 2017. Available online: http://www.sdcwa.org/sites/default/files/desal-carlsbad-fs-single.pdf (accessed on 9 January 2018).
| Constituent | Salton Sea Water | Pacific Ocean Water | ||||||
|---|---|---|---|---|---|---|---|---|
| Raw | Filt | Perm | Reject | Raw | Filt | Perm | Reject | |
| Alkalinity | X | X | X | X | X | X | X | X |
| Aeromonas | X | - | - | - | X | - | - | - |
| Ca2+ Hardness | X | X | X | X | X | X | X | X |
| Chloride | X | - | X | X | X | - | X | X |
| Color | X | - | X | X | X | - | X | X |
| Conductivity | X | - | X | X | X | - | X | X |
| E. coli | X | X | X | X | X | X | X | X |
| HPC 1 | X | X | X | X | X | X | X | X |
| pH | X | - | X | X | X | - | X | X |
| Salinity | X | - | X | X | X | - | X | X |
| Sulfate | X | - | X | X | X | - | X | X |
| Suspended Solids | X | - | X | X | X | - | X | X |
| Total Coliform | X | - | - | - | X | - | - | - |
| Total Hardness | X | - | X | X | X | - | X | X |
| Total Solids | X | - | X | X | X | - | X | X |
| Turbidity | X | X | X | X | X | X | X | X |
| UV254 | X | - | X | X | X | - | X | X |
| Item | Design Criteria |
|---|---|
| Manufacturer | Parker Hannifin Corp./Village Marine |
| Model No. | LWM-200 |
| No. Modules | 1 |
| Module Diameter | 4-inch |
| Module Length | 40-inch |
| No. Elements | 1 (Aqua Pro® Sea Water RO Membrane (Parker Hannifin Corp., Gardena, California, CA, USA) |
| Membrane Type | Thin-Film Composite |
| Membrane Surface Area | 1 m2 (estimated) |
| Pre-filter | Pentek® 5 μm polypropylene (Pentair) (Parker Hannifin Corp., Gardena, California, CA, USA) |
| High-Pressure Pump | 708 Titan Series (Aqua Pro Pumps, Gardena, California, CA, USA) |
| Max. Operating Pressure | 1000 psi |
| Max. Operating Temp. | 45 °C |
| Design Flux | 30 Lmh (estimated) |
| Design Product Flow | 0.8 m3/day (210 gpd) |
| Max. Feed Turbidity | NTU 1 |
| Free Chlorine Tolerance | 0 ppm (5 μm carbon block filter provided) |
| Max. Feed SDI 1 | SDI 5 |
| Typical Salt Rejection | 99.0 percent |
| pH range | 4 to 11 (2.5 to 11 during short-term cleaning) |
| Analyte | Technique | Analytical Method |
|---|---|---|
| Alkalinity | Titrimetric, pH 4.5 | EPA Method 310.1 |
| Aeromonas | Easygel ECA Check 1 | Standard Methods 9223 * |
| Ca2+ Hardness | Titrimetric, EDTA | Hach Method 8204 |
| Chloride | Mercuric Nitrate Titration | Hach Method 8206 |
| Color | Platinum-Cobalt | Standard Methods 2120 |
| Conductivity | Conductivity Cell | Standard Methods 2510 |
| HPC | Easygel Total Count T-salt 1 | Standard Methods 9215B * |
| pH | Electrometric | EPA Method 150.1 |
| Salinity | Mercuric Nitrate Titration | Hach Method 10073 |
| Sulfate | Turbidimetric | Hach Method 10227 |
| Suspended Solids | Gravimetric | EPA Method 160.1 |
| Total Coliform | Easygel ECA Check 1 | Standard Methods 9223 * |
| Total Hardness | Titrimetric, EDTA | Hach Method 8213 |
| Total Solids | Gravimetric | EPA Method 160.1 |
| Turbidity | Nephelometer | EPA Method 180.1 |
| UV254 | UVA at 254 nm | EPA Method 415.3 |
| Constituent | Units | Salton Sea Water | |||
|---|---|---|---|---|---|
| Raw | Feed | Permeate | Reject | ||
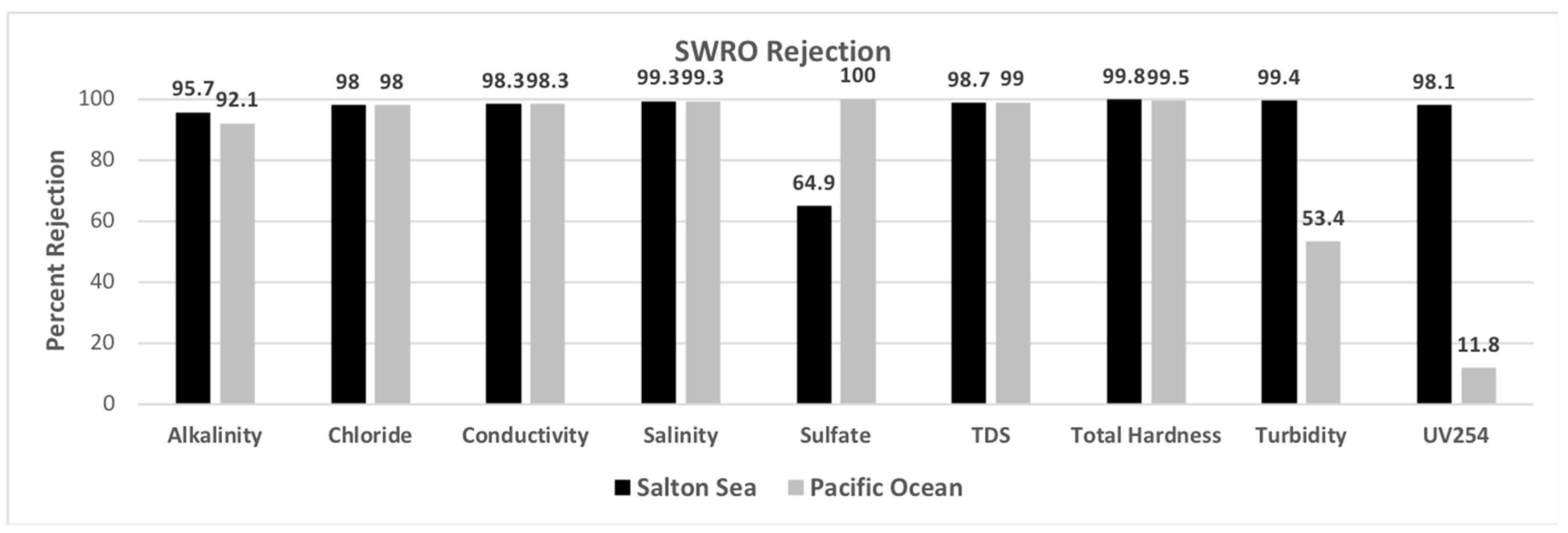
| Alkalinity | mg/L as CaCO3 | 276 | 268 | 12 | 288 |
| Aeromonas | CFU/mL | 33 | - | - | - |
| Ca2+ Hardness | mg/L as CaCO3 | 2200 | 2050 | 14 | 2200 |
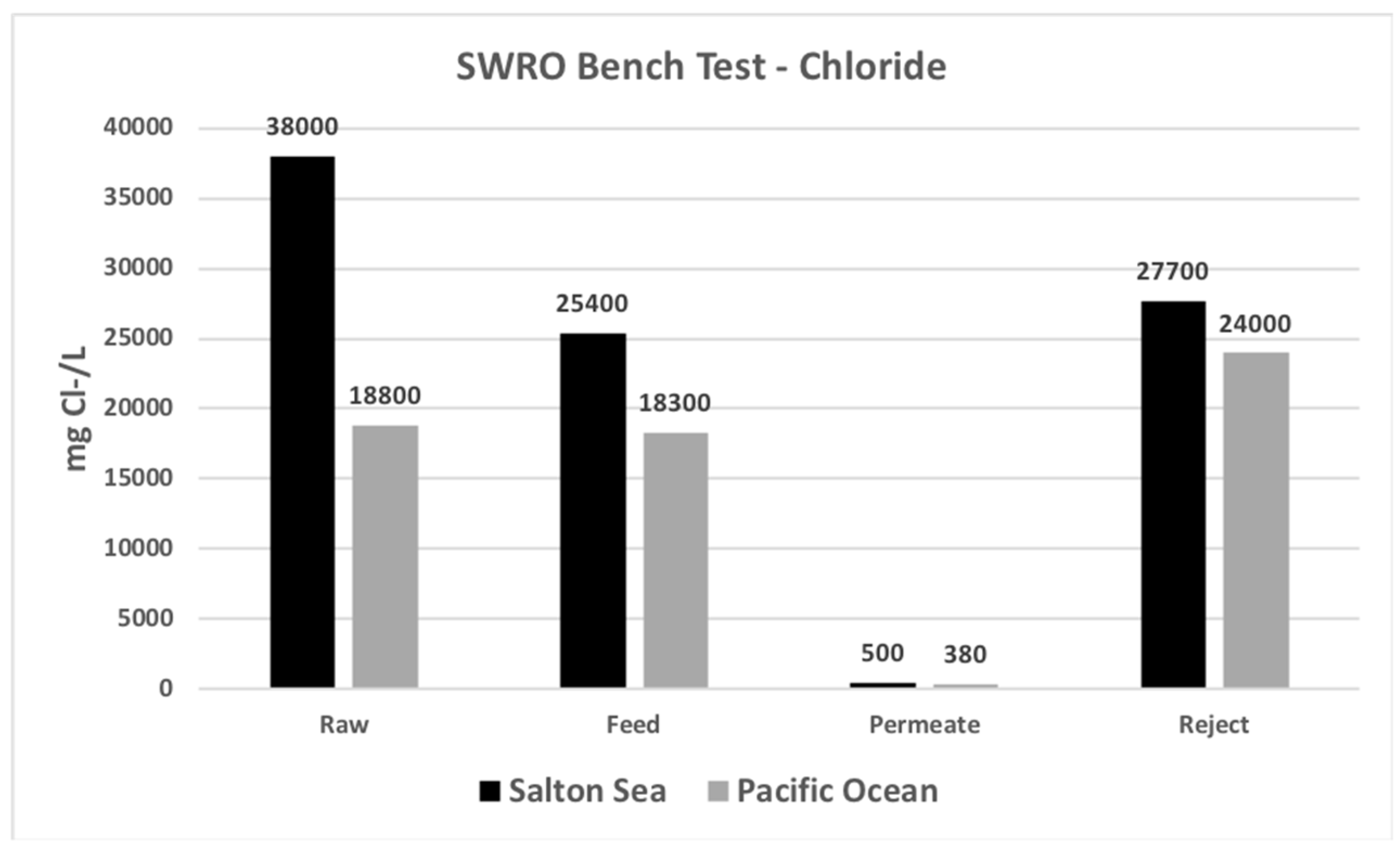
| Chloride | mg Cl−/L | 38,000 | 25,400 | 500 | 27,700 |
| Color | CU | 1440 | 1300 | 58 | 127 |
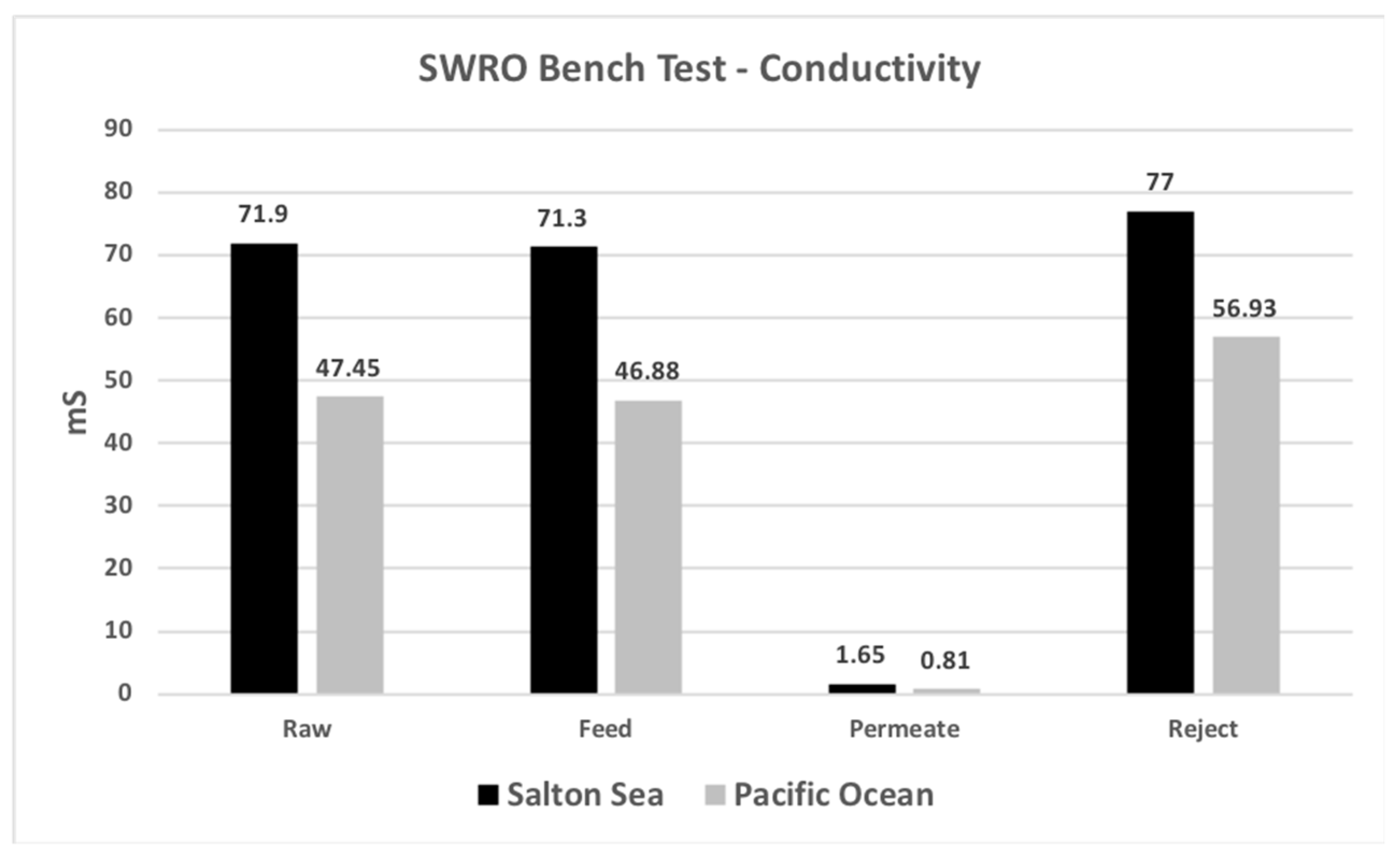
| Conductivity | mS/m | 71.9 | 71.3 | 1.65 | 77.0 |
| HPC 1 | CFU/mL | 66 | 32 | 14 | 122 |
| pH | units | 8.1 | - | 7.9 | 8.06 |
| Salinity | ppt | 43 | 39 | 0.4 | 46.7 |
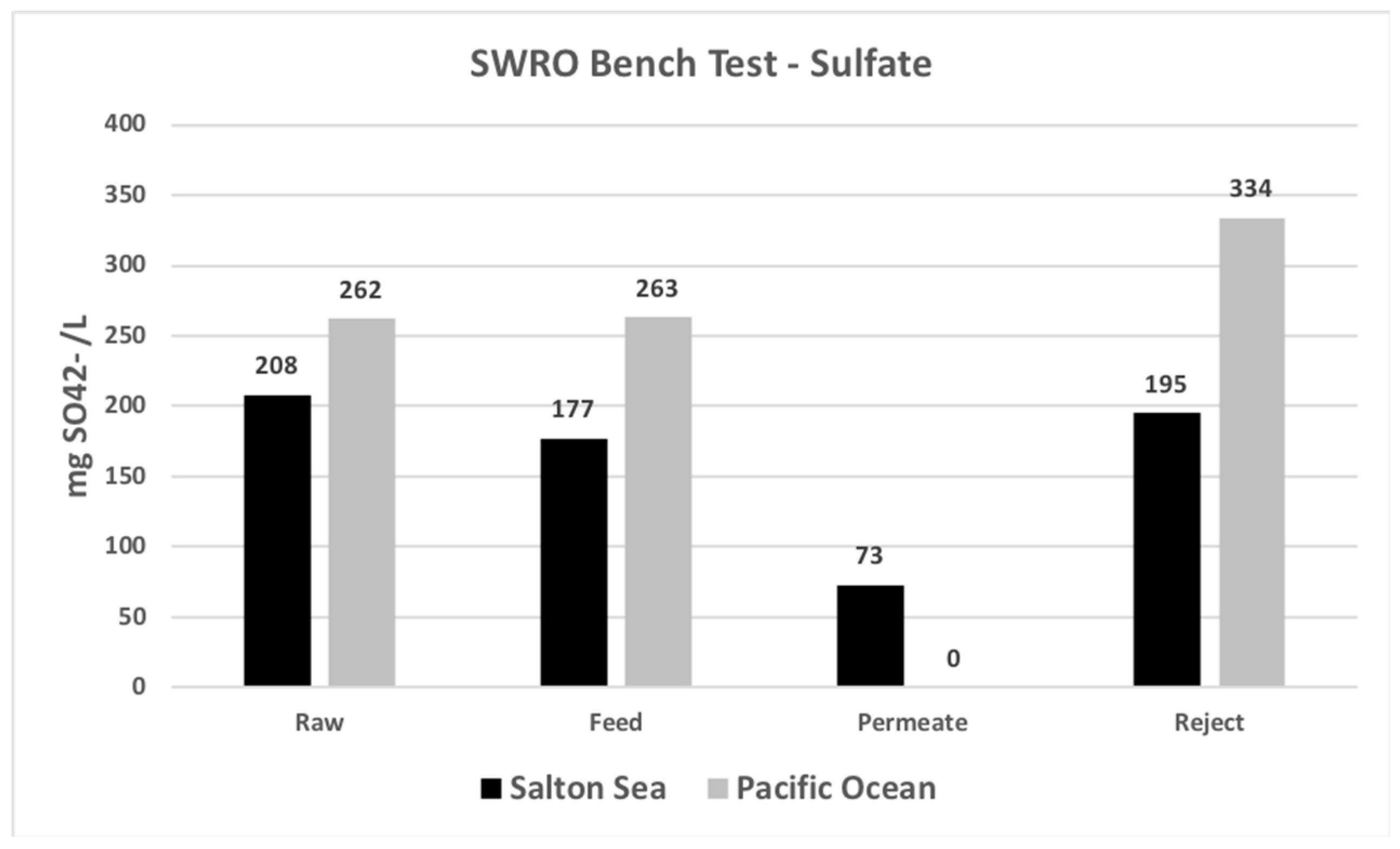
| Sulfate | mg SO42−/L | 20,800 | 17,700 | ND | 19,500 |
| Suspended Solids | mg/L | 44 | - | ND | 162 |
| Total Coliform | CFU/mL | 37 | - | - | - |
| Total Hardness | mg/L as CaCO3 | 17,500 | 9300 | 38 | 10,900 |
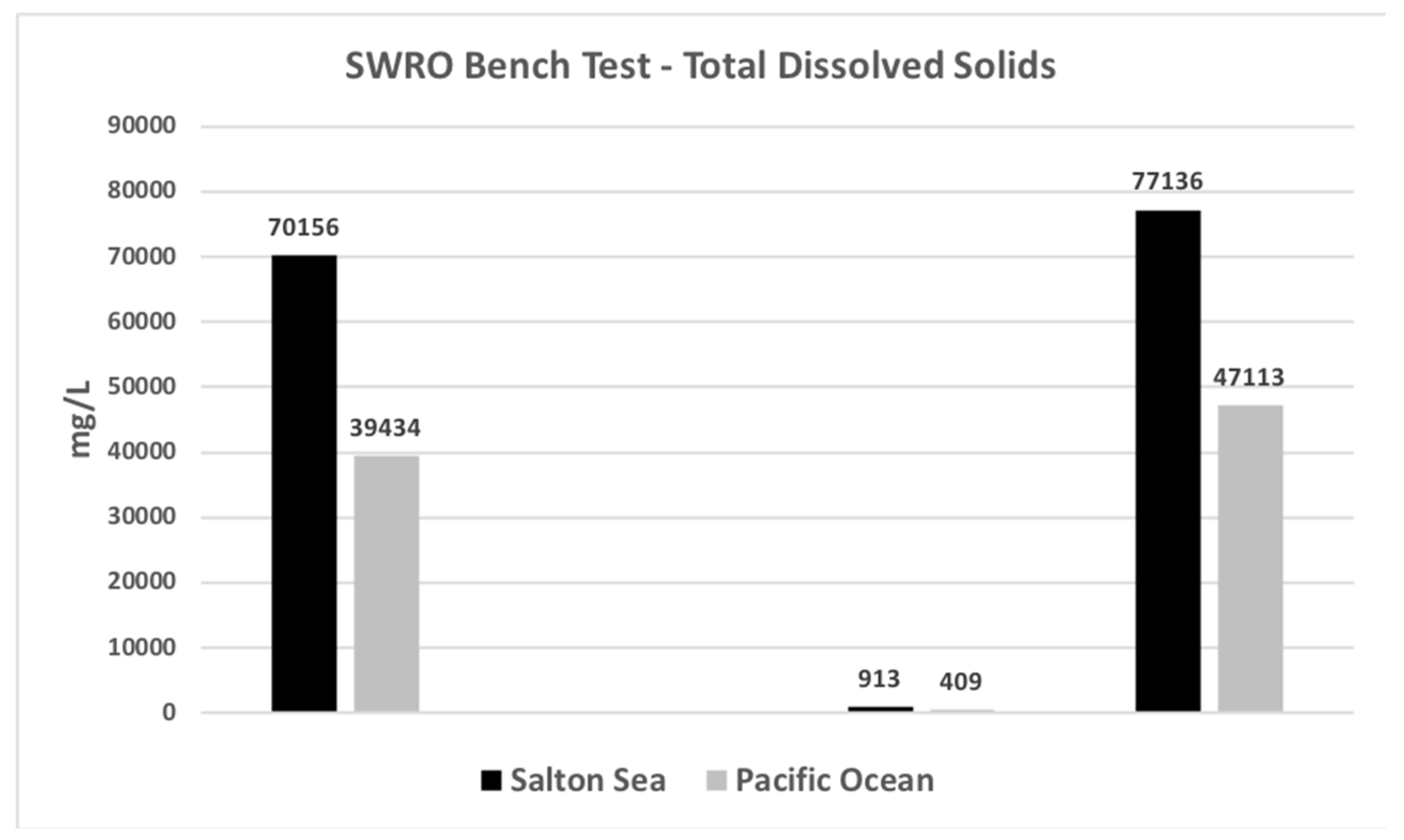
| Total Solids | mg/L | 70,200 | - | 913 | 77,136 |
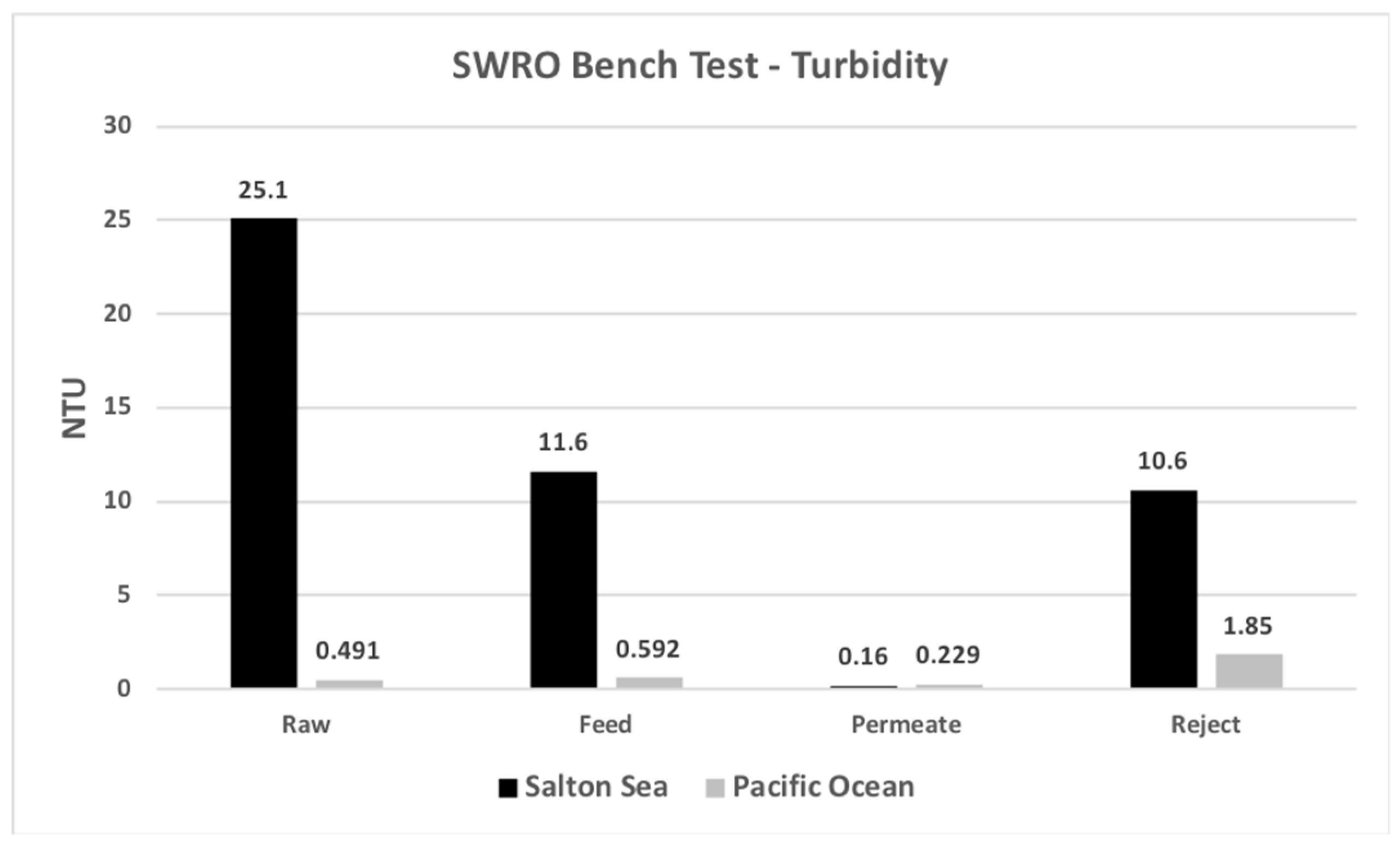
| Turbidity | NTU | 25.1 | 11.6 | 0.16 | 10.6 |
| UV254 | cm−1 | 0.696 | 0.69 | 0.013 | 0.815 |
| Constituent | Units | Pacific Ocean Water | |||
|---|---|---|---|---|---|
| Raw | Feed | Permeate | Reject | ||
| Alkalinity | mg/L as CaCO3 | 126 | 124 | 10 | 168 |
| Aeromonas | CFU/mL | None | - | - | - |
| Ca2+ Hardness | mg/L as CaCO3 | 900 | 875 | 4 | 1250 |
| Chloride | mg Cl−/L | 18,800 | 18,300 | 380 | 24,000 |
| Color | CU | ND | ND | ND | 1 |
| Conductivity | mS | 48 | 47 | 0.82 | 60 |
| HPC 1 | CFU/mL | 2047 | 243 | 1 | 3470 |
| pH | units | 8.0 | - | 7.5 | 8.0 |
| Salinity | ppt | 30.3 | 30.6 | 0.2 | 39.2 |
| Sulfate | mg SO42−/L | 262 | 263 | ND | 334 |
| Suspended Solids | mg/L | 9 | - | 1 | 8 |
| Total Coliform | CFU/mL | None | - | - | - |
| Total Hardness | mg/L as CaCO3 | 4700 | 4900 | 24 | 8800 |
| Total Solids | mg/L | 39,443 | - | 410 | 47,121 |
| Turbidity | NTU | 0.491 | 0.5 | 0.229 | 1.85 |
| UV254 | cm−1 | 0.017 | 0.016 | 0.015 | 0.13 |
| Constituent | Units | Goal 2 | Salton Sea | Pacific Ocean |
|---|---|---|---|---|
| Alkalinity | mg/L as CaCO3 | NA 3 | 12 | 10 |
| Aeromonas | CFU/mL | None | None | None |
| Ca2+ Hardness | mg/L as CaCO3 | NA 3 | 14 | 4 |
| Chloride | mg Cl−/L | <250 | 500 | 380 |
| Color | CU | ND | 58 | ND |
| Conductivity | mS | NA | 1.65 | 0.82 |
| HPC1 | CFU/mL | <500 | 14 | 1 |
| pH | units | 7.0–8.5 3 | 7.9 | 7.5 |
| Salinity | ppt | NA | 0.4 | 0.2 |
| Sulfate | mg SO42−/L | <250 | ND | ND |
| Suspended Solids | mg/L | None | ND | 1 |
| Total Coliform | CFU/mL | None | - | - |
| Total Hardness | mg/L as CaCO3 | 50–100 | 38 | 24 |
| Total Solids | mg/L | <500 | 913 | 410 |
| Turbidity | NTU | <0.3 | 0.16 | 0.229 |
| UV254 | cm−1 | NA | 0.013 | 0.015 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pontius, F. Treatability of a Highly-Impaired, Saline Surface Water for Potential Urban Water Use. Water 2018, 10, 324. https://doi.org/10.3390/w10030324
Pontius F. Treatability of a Highly-Impaired, Saline Surface Water for Potential Urban Water Use. Water. 2018; 10(3):324. https://doi.org/10.3390/w10030324
Chicago/Turabian StylePontius, Frederick. 2018. "Treatability of a Highly-Impaired, Saline Surface Water for Potential Urban Water Use" Water 10, no. 3: 324. https://doi.org/10.3390/w10030324
APA StylePontius, F. (2018). Treatability of a Highly-Impaired, Saline Surface Water for Potential Urban Water Use. Water, 10(3), 324. https://doi.org/10.3390/w10030324




