Effects of Backwashing on Granular Activated Carbon with Ammonium Removal Potential in a Full-Scale Drinking Water Purification Plant
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
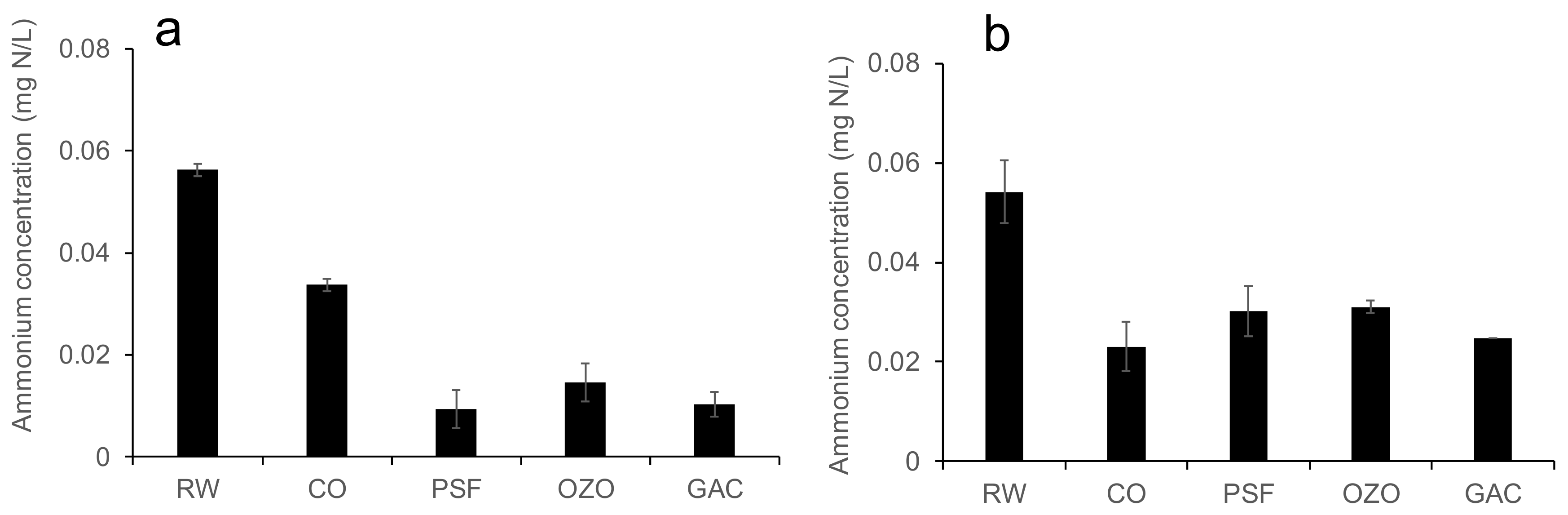
3.1. Ammonium Concentration in Raw Water and the Effluents of the DWPP
3.2. Effects of Backwashing on Ammonium Removal Potential of GAC
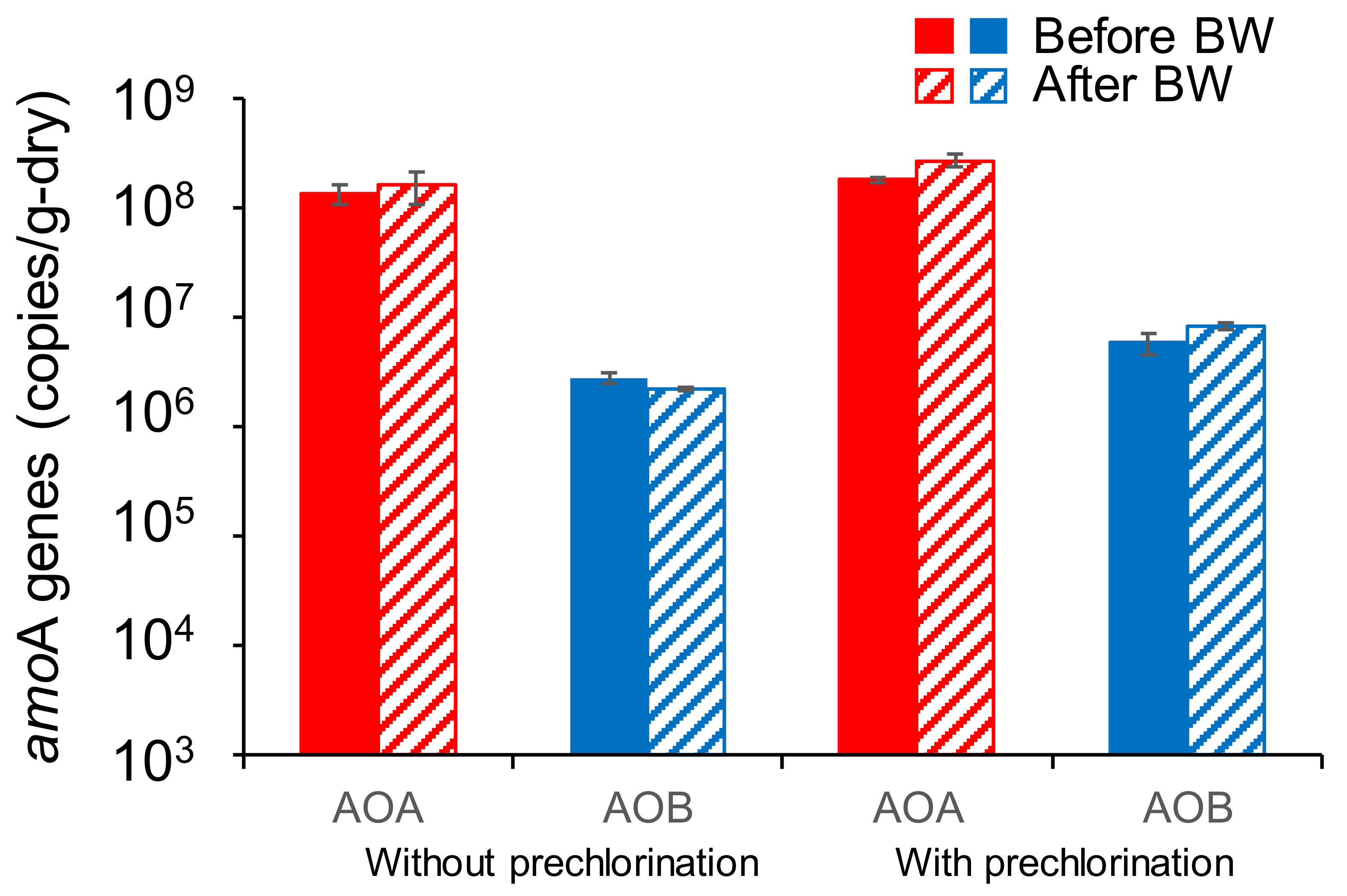
3.3. Effects of Backwashing on Abundances of AOA and AOB Associated with GAC
3.4. Analysis of AOA and AOB Dislodged from GAC by Backwashing
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yapsakli, K.; Mertoglu, B.; Çeçen, F. Identification of nitrifies and nitrification performance in drinking water biological activated carbon (BAC) filtration. Process Biochem. 2010, 45, 1543–1549. [Google Scholar] [CrossRef]
- Kasuga, I.; Nakagaki, H.; Kurisu, F.; Furumai, H. Predominance of ammonia-oxidizing archaea on granular activated carbon used in a full-scale advanced drinking water treatment plant. Water Res. 2010, 44, 5039–5049. [Google Scholar] [CrossRef]
- Seredyńska-Sobecka, B.; Tomaszewska, M.; Janus, M.; Morawski, A.W. Biological activation of carbon filters. Water Res. 2006, 40, 355–363. [Google Scholar] [CrossRef] [PubMed]
- LeChevallier, M.W.; McFeters, G.A. Microbiology of activated carbon. In Drinking Water Microbiology; McFeters, G.A., Ed.; Spinger: New York, NY, USA, 1990; pp. 104–119. [Google Scholar]
- Regan, J.M.; Harrington, G.W.; Noguera, D.R. Ammonia- and nitrite-oxidizing bacterial communities in a pilot-scale chloraminated drinking water distribution system. Appl. Environ. Microb. 2002, 68, 73–81. [Google Scholar] [CrossRef]
- Haas, C.N. Disinfection. In Water Quality and Treatment: A Handbook of Community Water Supplies, 5th ed.; Letterman, R.D., Ed.; McGraw-Hill, Inc.: New York, NY, USA, 1999; pp. 877–932. [Google Scholar]
- Laurent, P.; Andersson, A.; SERVAJS, P. Impact of backwashing on nitrification in the biological activated carbon filters used in drinking water treatment. Environ. Technol. 2003, 24, 277–287. [Google Scholar] [CrossRef]
- Andersson, A.; Laurent, P.; Kihn, A.; Prévost, M.; Servais, P. Impact of temperature on nitrification in biological activated carbon (BAC) filters used for drinking water treatment. Water Res. 2001, 35, 2923–2934. [Google Scholar] [CrossRef]
- Prévost, M.; Desjardins, R.; Lafrance, P.; Duchesne, D.; Coallier, J. Optimization of biological activated carbon (BAC) filtration of drinking water: Effect of filtration velocity, ozone dosage and position and of carbon type. In Proceedings of the AWWARF-Fifth National Conference on Drinking Water Proceedings, Winnipeg, MB, Canada, 13–15 September 1993; pp. 63–77. [Google Scholar]
- Limpiyakorn, T.; Fürhacker, M.; Haberl, R.; Chodanon, T.; Srithep, P.; Sonthiphand, P. amoA-encoding archaea in wastewater treatment plants: A review. Appl. Microbiol. Biotechnol. 2013, 97, 1425–1439. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, A.; Sihvonen, M.; Muñoz-Palazon, B.; Rodriguez-Sanchez, M.A.; Vahala, R. Microbial ecology of full-scale wastewater treatment systems in the Polar Arctic Circle: Archaea, Bacteria and Fungi. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Könneke, M.; Bernhard, A.E.; de la Torre, J.R.; Walker, C.B.; Waterbury, J.B.; Stahl, D.A. Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nature 2005, 437, 543–546. [Google Scholar] [CrossRef]
- Martens-Habbena, W.; Berube, P.M.; Urakawa, H.; de la Torre, J.R.; Stahl, D.A. Ammonia oxidation kinetics determine niche separation of nitrifying archaea and bacteria. Nature 2009, 461, 976–979. [Google Scholar] [CrossRef]
- Van der Wielen, P.W.; Voost, S.; van der Kooij, D. Ammonia-oxidizing bacteria and archaea in groundwater treatment and drinking water distribution systems. Appl. Environ. Microb. 2009, 75, 4687–4695. [Google Scholar] [CrossRef] [PubMed]
- Nicol, G.W.; Schleper, C. Ammonia-oxidising Crenarchaeota: Important players in the nitrogen cycle? Trends Microbiol. 2006, 14, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Sáez, L.; Waller, A.S.; Mende, D.R.; Bakker, K.; Farnelid, H.; Yager, P.L.; Lovejoy, C.; Tremblay, J.-É.; Potvin, M.; Heinrich, F.; et al. Role for urea in nitrification by polar marine Archaea. Proc. Natl. Acad. Sci. USA 2012, 109, 17989–17994. [Google Scholar] [CrossRef] [PubMed]
- Kasuga, I.; Nakagaki, H.; Kurisu, F.; Furumai, H. Abundance and diversity of ammonia-oxidizing archaea and bacteria on biological activated carbon in a pilot-scale drinking water treatment plant with different treatment processes. Water Sci. Technol. 2010, 61, 3070–3077. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Kasuga, I.; Kurisu, F.; Furuami, H.; Shigeeda, T.; Takahashi, K. Abundance and diversity of ammonia-oxidizing archaea and bacteria on granular activated carbon and their fates during drinking water purification process. Appl. Microbiol. Biotechnol. 2016, 100, 729–742. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Kasuga, I.; Kurisu, F.; Furumai, H.; Shigeeda, T. Evaluation of autotrophic growth of ammonia-oxidizers associated with granular activated carbon used for drinking water purification by DNA-stable isotope probing. Water Res. 2013, 47, 7053–7065. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.R. Biofilm processes in biologically activated carbon water purification. Water Res. 2008, 42, 2839–2848. [Google Scholar] [CrossRef]
- Morin, P.; Camper, A.K. Attachment and fate of carbon fines in simulated drinking water distribution system biofilms. Water Res. 1997, 31, 399–410. [Google Scholar] [CrossRef]
- Kasuga, I.; Shimazaki, D.; Kunikane, S. Influence of backwashing on the microbial community in a biofilm developed on biological activated carbon used in a drinking water treatment plant. Water Sci. Technol. 2007, 55, 173–180. [Google Scholar] [CrossRef]
- Liao, X.; Chen, C.; Zhang, J.; Dai, Y.; Zhang, X.; Xie, S. Operational performance, biomass and microbial community structure: Impacts of backwashing on drinking water biofilter. Environ. Sci. Pollut. Res. 2015, 22, 546–554. [Google Scholar] [CrossRef]
- Kim, T.G.; Yun, J.; Hong, S. Effects of water temperature and backwashing on bacterial population and community in a biological activated carbon process at a water treatment plant. Appl. Microbiol. Biotechnol. 2014, 98, 1417–1427. [Google Scholar] [CrossRef] [PubMed]
- Gray, N.F. Chapter Thirty-One-Free and Combined Chlorine. In Microbiology of Waterborne Diseases, 2nd ed.; Percival, S.L., Williams, D.W., Gray, N.F., Yates, M.V., Chalmers, R.M., Eds.; Academic Press: Cambridge, MA, USA, 2014; pp. 571–590. [Google Scholar]
- De Vet, W.W.J.M.; Kleerebezem, R.; van der Wielen, P.W.J.J.; Rietveld, L.C.; van Loosdrecht, M.C.M. Assessment of nitrification in groundwater filters for drinking water production by qPCR and activity measurement. Water Res. 2011, 45, 4008–4018. [Google Scholar] [CrossRef] [PubMed]
 ) in the sampling time of “without prechlorination” and “with prechlorination”. BW: backwashing, and error bar: the standard deviation.
) in the sampling time of “without prechlorination” and “with prechlorination”. BW: backwashing, and error bar: the standard deviation.
 ) in the sampling time of “without prechlorination” and “with prechlorination”. BW: backwashing, and error bar: the standard deviation.
) in the sampling time of “without prechlorination” and “with prechlorination”. BW: backwashing, and error bar: the standard deviation.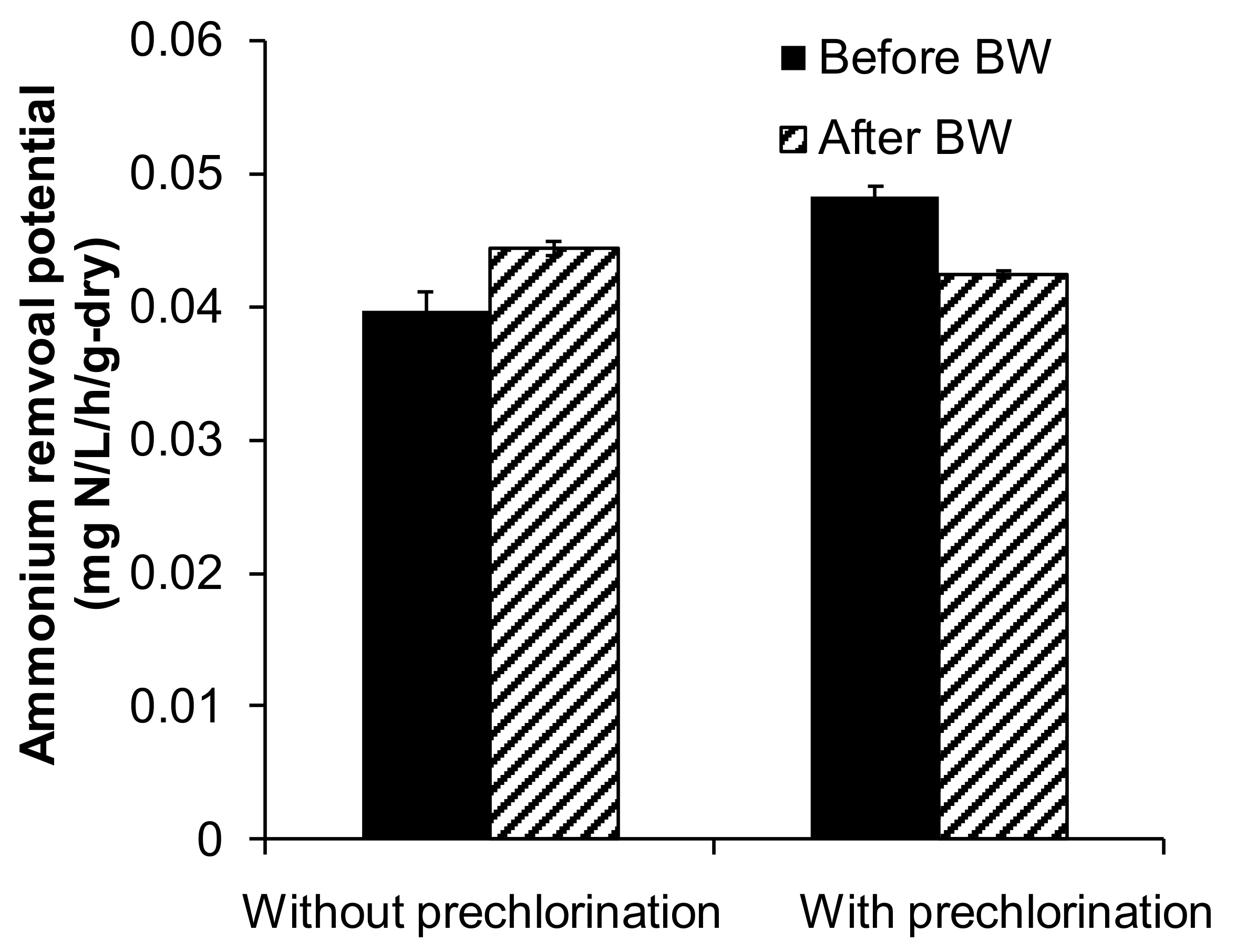
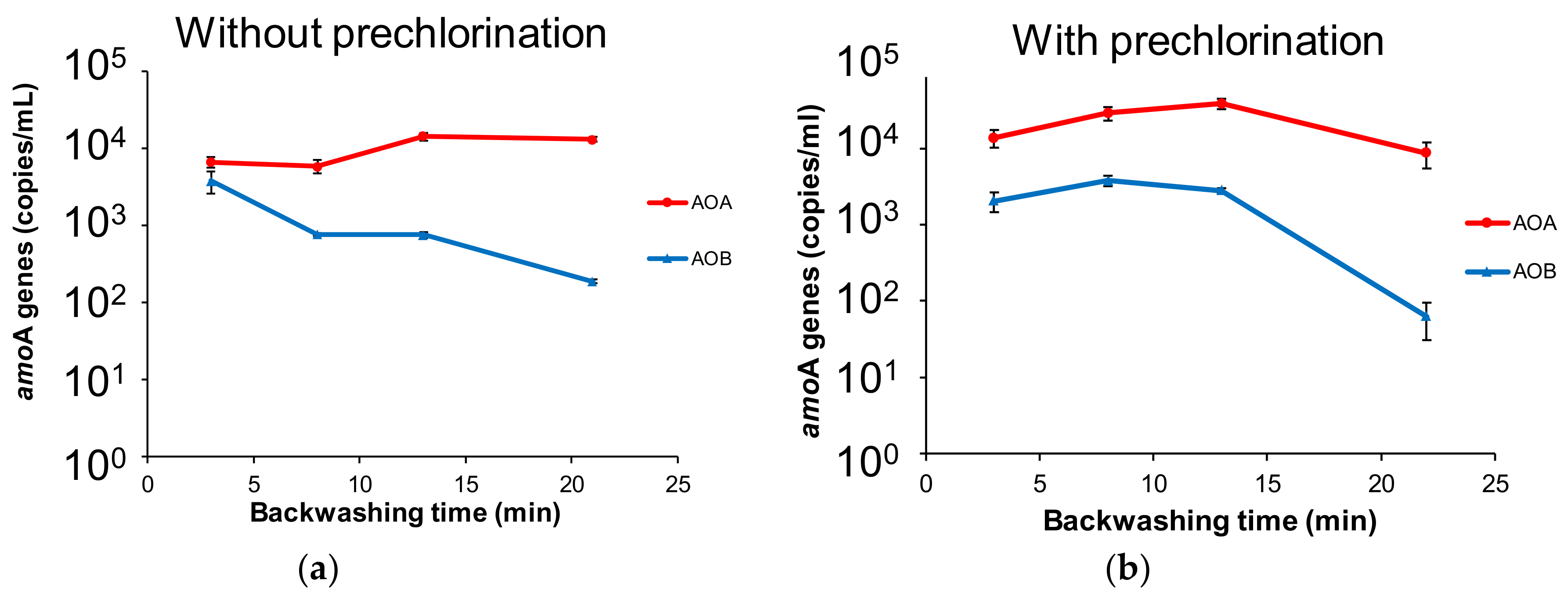
| Time (min) | 1–3 | 4–7 | 8–10 | 11–20 | 21–23 | 24–25 | |
|---|---|---|---|---|---|---|---|
| Program | |||||||
| Air scour (flow rate (m3/min)) | 60 | 60 | |||||
| Finished water with residual chlorine (flow rate (m3/min)) | 20 | 30 | 30 | 60 | 30 | 20 | |
| Sampling time of backwashing water (minutes from the beginning of the program) | 3 | 8 | 8 | 13 | 21 | ||
| Sampling Time | Percentage of Discharged AOA and AOB in Original Amount on GAC (%) | |
|---|---|---|
| AOA | AOB | |
| Without prechlorination | 0.08 | 0.23 |
| With prechlorination | 0.12 | 0.26 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niu, J.; Kasuga, I.; Kurisu, F.; Furumai, H. Effects of Backwashing on Granular Activated Carbon with Ammonium Removal Potential in a Full-Scale Drinking Water Purification Plant. Water 2018, 10, 1830. https://doi.org/10.3390/w10121830
Niu J, Kasuga I, Kurisu F, Furumai H. Effects of Backwashing on Granular Activated Carbon with Ammonium Removal Potential in a Full-Scale Drinking Water Purification Plant. Water. 2018; 10(12):1830. https://doi.org/10.3390/w10121830
Chicago/Turabian StyleNiu, Jia, Ikuro Kasuga, Futoshi Kurisu, and Hiroaki Furumai. 2018. "Effects of Backwashing on Granular Activated Carbon with Ammonium Removal Potential in a Full-Scale Drinking Water Purification Plant" Water 10, no. 12: 1830. https://doi.org/10.3390/w10121830
APA StyleNiu, J., Kasuga, I., Kurisu, F., & Furumai, H. (2018). Effects of Backwashing on Granular Activated Carbon with Ammonium Removal Potential in a Full-Scale Drinking Water Purification Plant. Water, 10(12), 1830. https://doi.org/10.3390/w10121830




