Influence of Temperature and De-Icing Salt on the Sedimentation of Particulate Matter in Traffic Area Runoff
Abstract
1. Introduction
2. Materials and Methods
2.1. Monitoring of Full-Scale Sedimentation Tank
2.2. Calculations
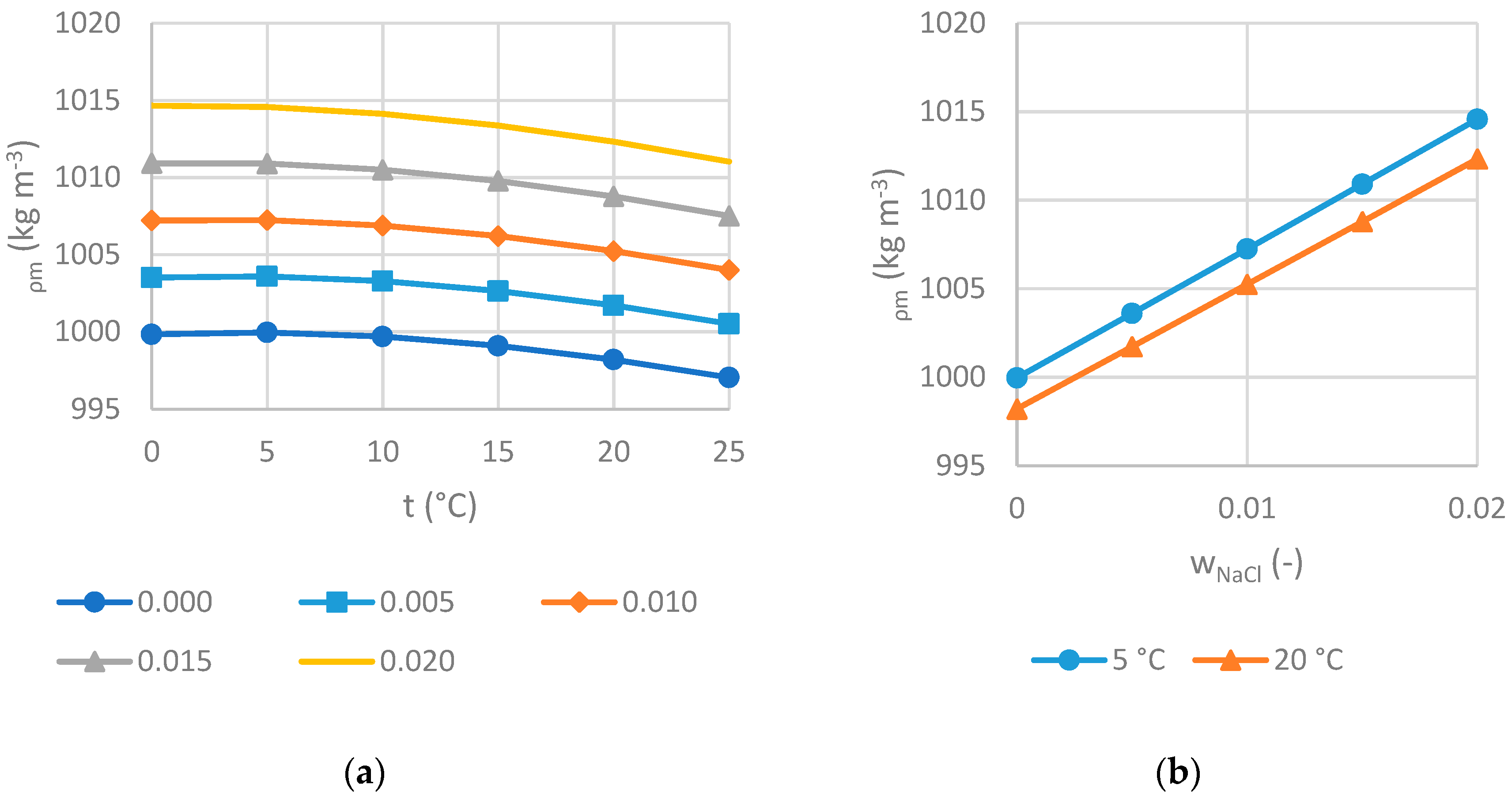
2.2.1. Density
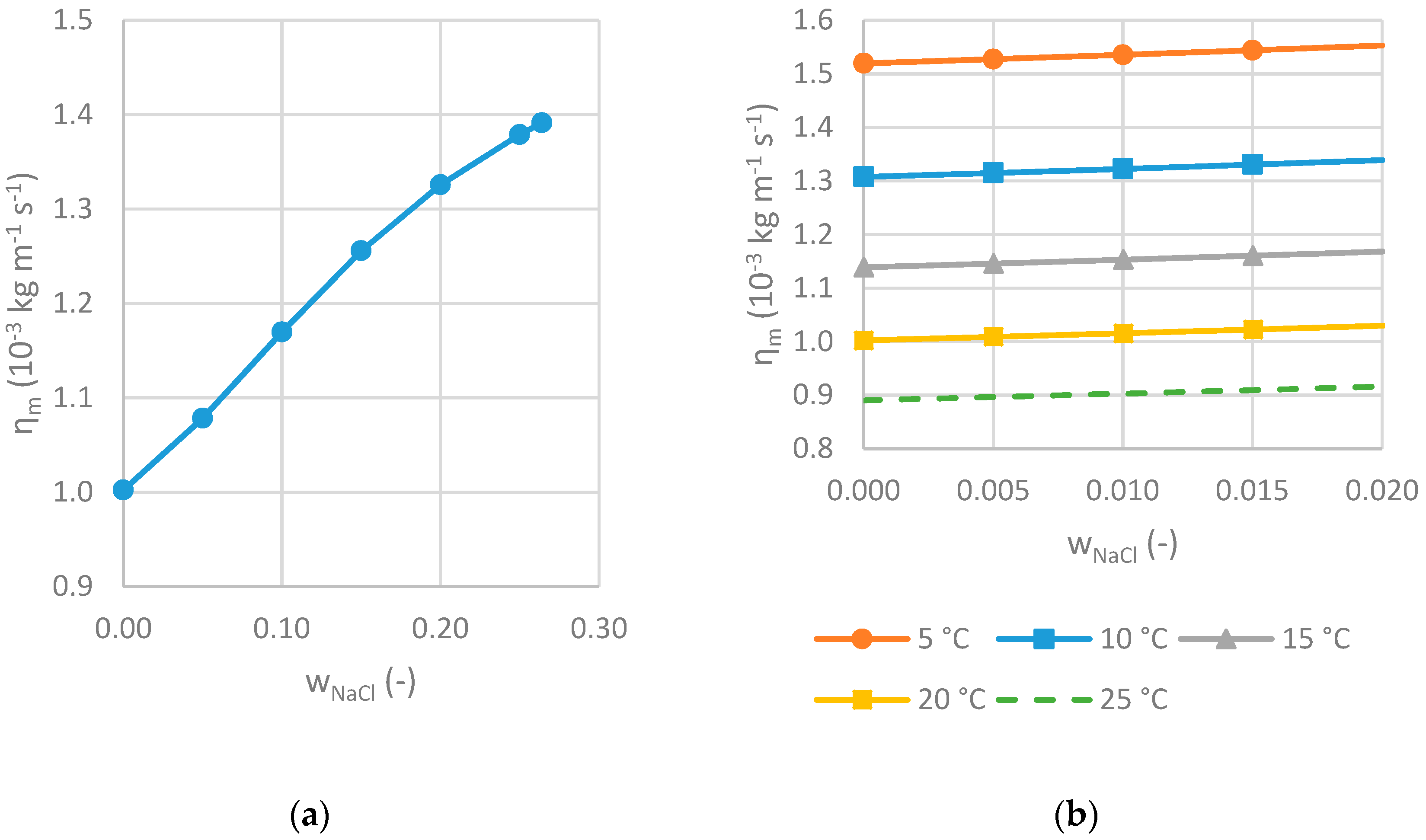
2.2.2. Viscosity
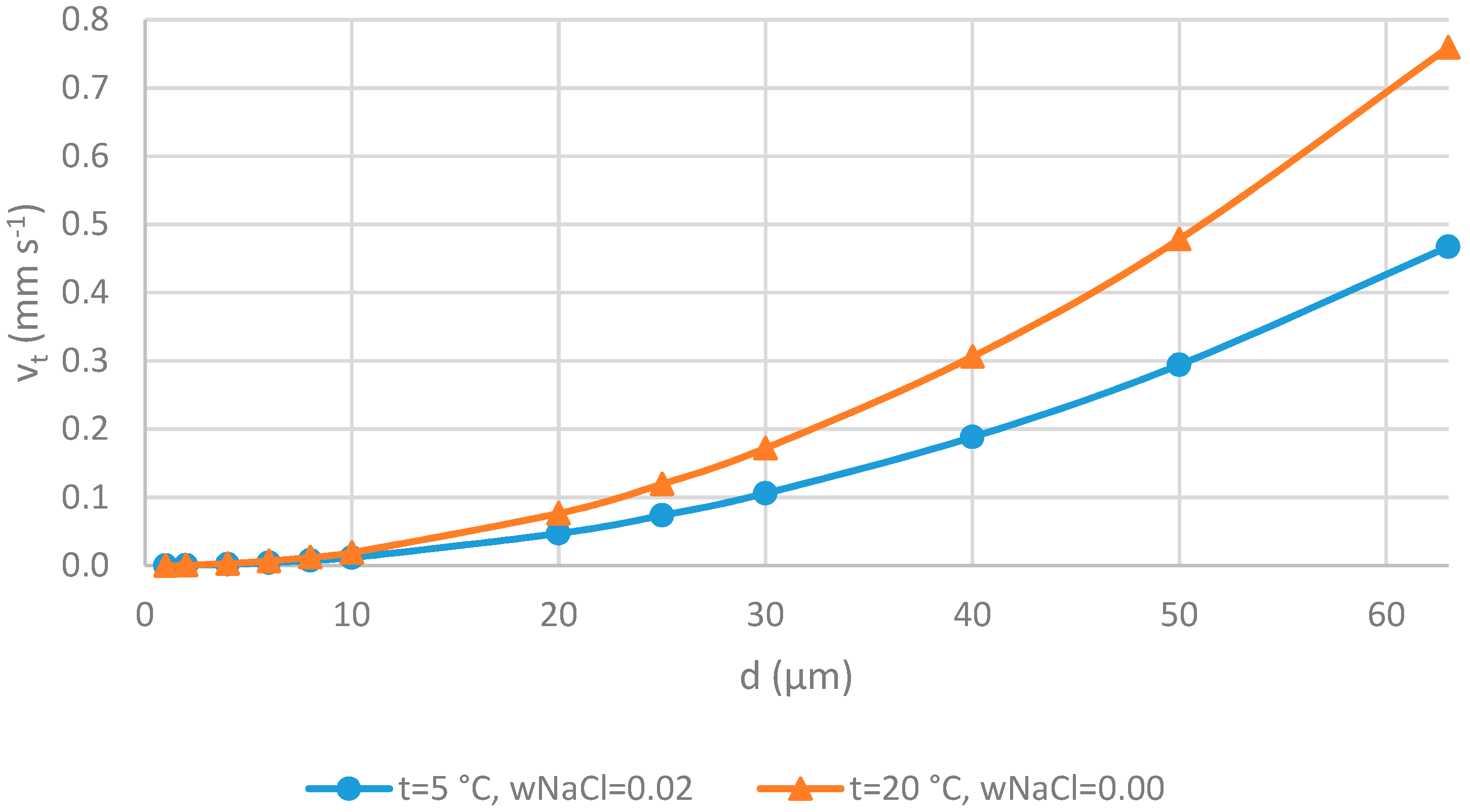
2.2.3. Settling Velocity
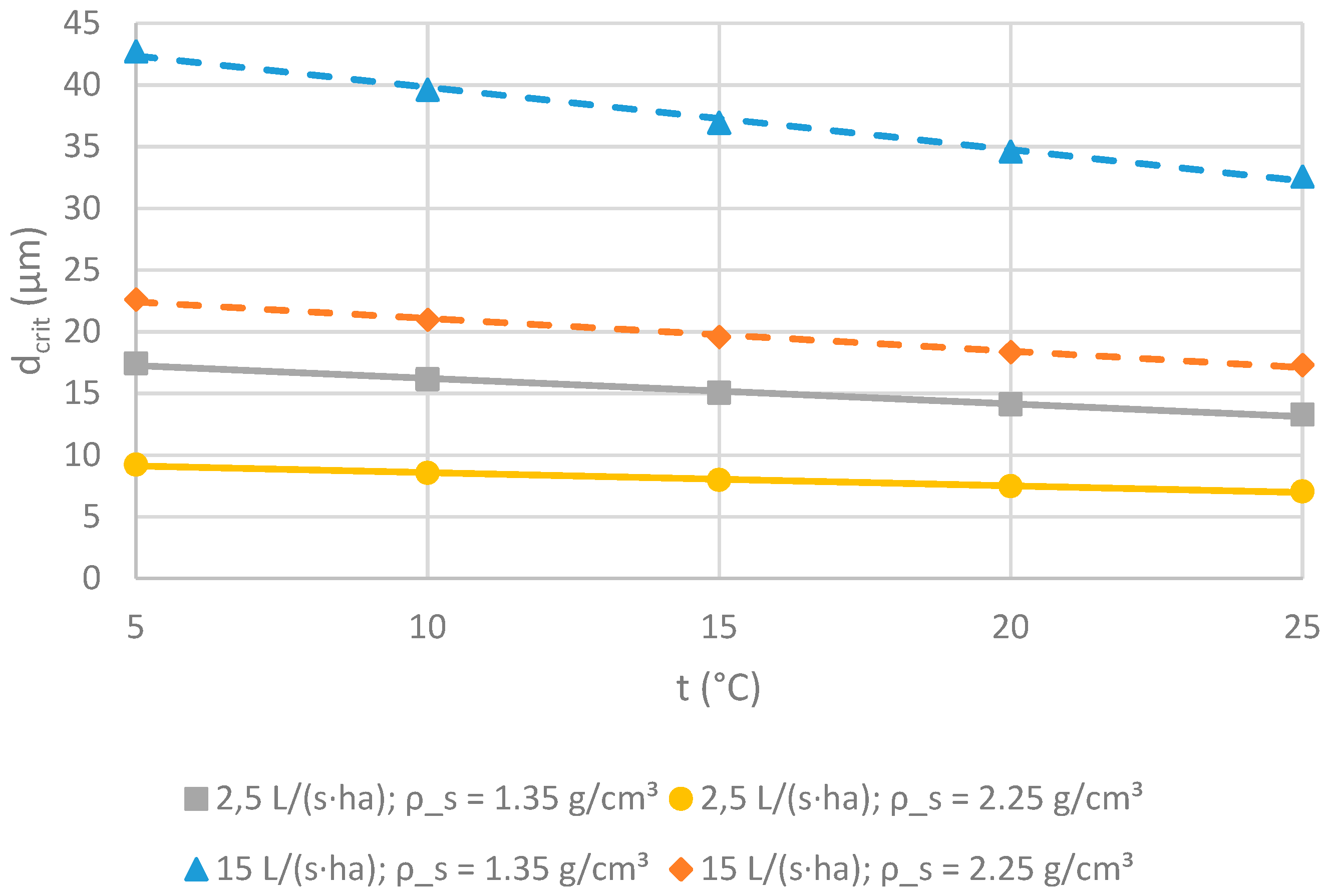
2.2.4. Retention of Suspended Solids
3. Results and Discussion
3.1. Monitoring of the Full-Scale Sedimentation Tank
3.2. Calculations
3.2.1. Density
3.2.2. Viscosity
3.2.3. Settling Velocity
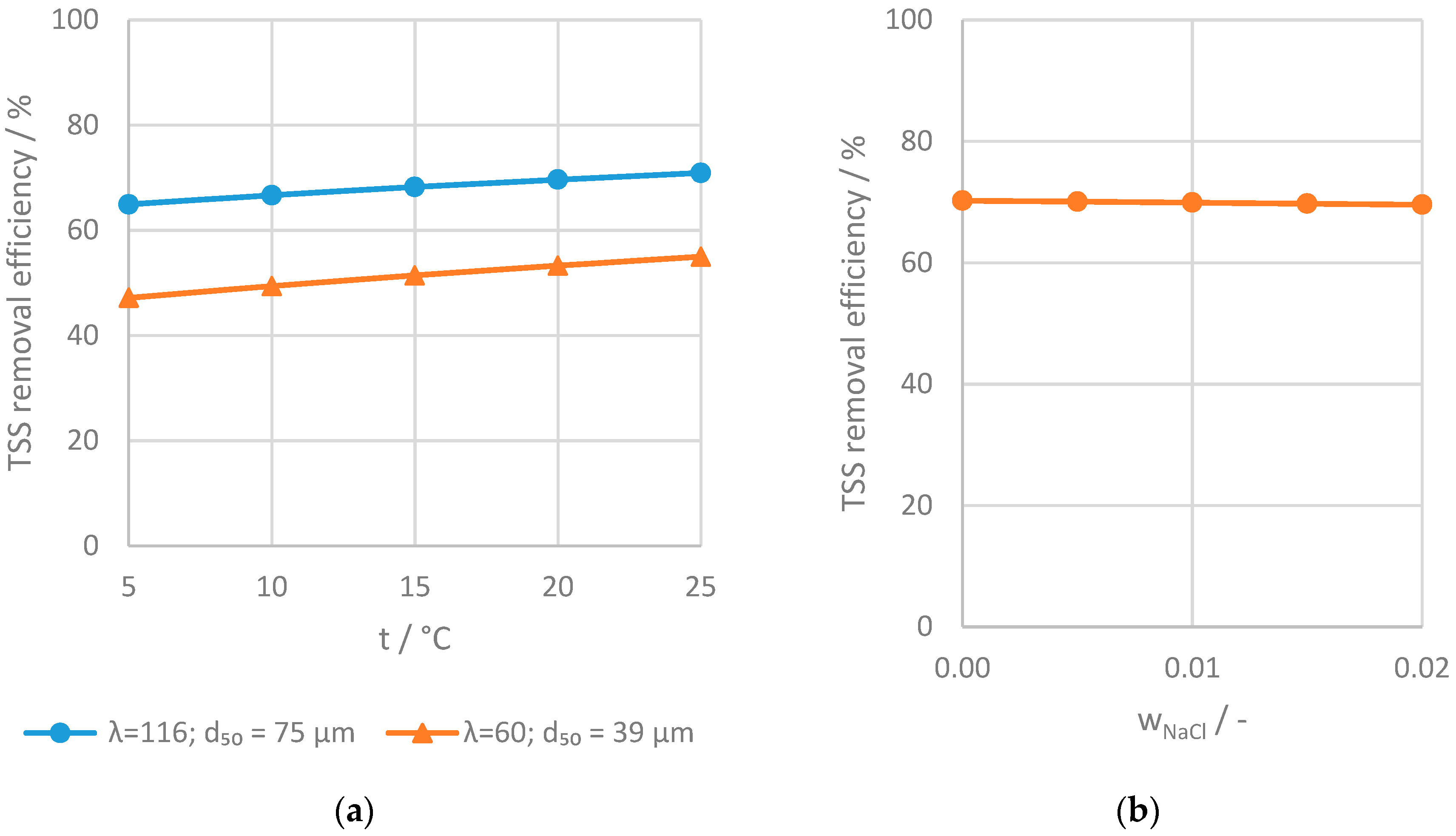
3.2.4. Retention of Suspended Solids
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| A | Area |
| AADT | Annual average daily traffic |
| CDF | Cumulative distribution function |
| d | Particle diameter |
| d50 | Mass-median-diameter of particles |
| DIBt | Deutsches Insitut für Bautechnik |
| DWD | German Weather Service |
| EC | Electric conductivity |
| Eff | Effluent |
| g | Gravitational acceleration |
| Inf | Influent |
| NaCl | Sodium chloride |
| PM | Particulate matter |
| PSD | Particle size distribution |
| Q | Flow rate |
| q | Discharge rate, mean inflow |
| QEff | Outflow |
| QIn | Inflow |
| RE | Removal efficiency |
| Re | Reynold’s number |
| rs | Spearman’s rank correlation coefficients |
| SQID | Stormwater quality improvement device |
| SS > 63 | Suspended solids with particle diameter >63 µm |
| SS63 | Suspended solids with particle diameter between 0.45 µm and 63 µm |
| SUDS | Sustainable urban drainage systems |
| t | Temperature |
| TSS | Total suspended solids |
| NaCl specific volume | |
| vt | Terminal settling velocity |
| wH2O | Mass fraction of water in the solution |
| wNaCl | Mass fraction of the NaCl in the solution |
| ηm | Dynamic viscosity of the NaCl solution |
| κ | Scale parameter of CDF |
| λ | Shape parameter of CDF |
| νm | Kinematic viscosity of the solution |
| ρs | Particle density |
| ρw | Water density |
| ψ | Discharge coefficient |
References
- Ball, J.E.; Jenks, R.; Aubourg, D. An assessment of the availability of pollutant constituents on road surfaces. Sci. Total Environ. 1998, 209, 243–254. [Google Scholar] [CrossRef]
- Legret, M.; Pagotto, C. Evaluation of pollutant loadings in the runoff waters from a major rural highway. Sci. Total Environ. 1999, 235, 143–150. [Google Scholar] [CrossRef]
- McKenzie, E.R.; Money, J.E.; Green, P.G.; Young, T.M. Metals associated with stormwater-relevant brake and tire samples. Sci. Total Environ. 2009, 407, 5855–5860. [Google Scholar] [CrossRef] [PubMed]
- Wik, A.; Dave, G. Occurrence and effects of tire wear particles in the environment—A critical review and an initial risk assessment. Environ. Pollut. 2009, 157, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.; Welker, A.; Helmreich, B. Critical review of heavy metal pollution of traffic area runoff: Occurrence, influencing factors, and partitioning. Sci. Total Environ. 2016, 541, 895–919. [Google Scholar] [CrossRef] [PubMed]
- Kayhanian, M.; Fruchtman, B.D.; Gulliver, J.S.; Montanaro, C.; Ranieri, E.; Wuertz, S. Review of highway runoff characteristics: Comparative analysis and universal implications. Water Res. 2012, 46, 6609–6624. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.; Welker, A.; Drewes, J.E.; Helmreich, B. Auftausalze im Straßenwinterdienst—Aufkommen und Bedeutung für dezentrale Behandlungsanlagen von Verkehrsflächenabflüssen zur Versickerung (De-icing salts in road maintenance—Occurrence and impact on decentralized systems treating traffic area runoff for infiltration). GWF Wasser/Abwasser 2015, 156, 1138–1152. [Google Scholar]
- Fay, L.; Shi, X. Environmental impacts of chemicals for snow and ice control: State of the knowledge. Water Air Soil Pollut. 2012, 223, 2751–2770. [Google Scholar] [CrossRef]
- Murray, D.C.; Ernst, U.F.W. An Economic Analysis of the Environmental Impact of Highway Deicing; USEPA 600/2-76-033; US EPA: Washington, DC, USA, 1976.
- Helmreich, B.; Hilliges, R.; Schriewer, A.; Horn, H. Runoff pollutants of a highly trafficked urban road—Correlation analysis and seasonal influences. Chemosphere 2010, 80, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Akin, M.; Pan, T.; Fay, L.; Liu, Y.; Yang, Z. Deicer impacts on pavement materials: Introduction and recent developments. Open Civ. Eng. J. 2009, 3, 16–27. [Google Scholar] [CrossRef]
- Dierkes, C.; Lucke, T.; Helmreich, B. General technical approvals for decentralised sustainable urban drainage systems (suds)—The current situation in germany. Sustainability 2015, 7, 3031–3051. [Google Scholar] [CrossRef]
- Huber, M.; Helmreich, B.; Welker, A. Einführung in Die Dezentrale Niederschlagswasserbehandlung für Verkehrsflächen- und Metalldachabflüsse: Schacht-/Kompaktsysteme, Rinnensysteme, Straßeneinläufe und Flächenbeläge (Introduction on Decentralized Stormwater Treatment of Traffic Area and Metal Roof Runoff); Technische Universität München Garching: München, Germany, 2015; Volume 213, p. 96. ISSN 0942-914X. [Google Scholar]
- Rommel, S.H.; Helmreich, B. Feinpartikuläre Stoffe (AFS63) in Verkehrsflächenabflüssen—Vorkommen und Relevanz für Dezentrale Behandlungsanlagen (Fine Particulate Matter (SS63) in Traffic Area Runoff—Occurrence and Relevance for Decentralized Treatment Systems). In Proceedings of the Aqua Urbanica Trifft RegenwasserTage, Landau (Pfalz), Germany, 18–19 June 2018; Schmitt, T.G., Ed.; Technische Universität Kaiserslautern: Kaiserslautern, Germany, 2018; pp. 159–178. [Google Scholar]
- Semadeni–Davies, A. Winter performance of an urban stormwater pond in southern sweden. Hydrol. Process. 2006, 20, 165–182. [Google Scholar] [CrossRef]
- Hendi, E.; Shamseldin, A.Y.; Melville, B.W.; Norris, S.E. Experimental investigation of the effect of temperature differentials on hydraulic performance and flow pattern of a sediment retention pond. Water Sci. Technol. 2018, 77, 2896–2906. [Google Scholar] [CrossRef] [PubMed]
- Adamsson, Å.; Bergdahl, L. Simulation of temperature influence on flow pattern and residence time in a detention tank. Hydrol. Res. 2006, 37, 53–68. [Google Scholar] [CrossRef]
- Marsalek, J.; Oberts, G.; Exall, K.; Viklander, M. Review of operation of urban drainage systems in cold weather: Water quality considerations. Water Sci. Technol. 2003, 48, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D. Wastewater Engineering: Treatment and Resource Recovery; McGraw-Hill Education: Berkshire, UK, 2014. [Google Scholar]
- Winkler, M.K.H.; Bassin, J.P.; Kleerebezem, R.; van der Lans, R.G.J.M.; van Loosdrecht, M.C.M. Temperature and salt effects on settling velocity in granular sludge technology. Water Res. 2012, 46, 5445–5451. [Google Scholar] [CrossRef] [PubMed]
- Krishnappan, B.G.; Marsalek, J.; Watt, W.E.; Anderson, B.C. Seasonal size distributions of suspended solids in a stormwater management pond. Water Sci. Technol. 1999, 39, 127–134. [Google Scholar] [CrossRef]
- Krishnappan, B.G.; Marsalek, J. Modelling of flocculation and transport of cohesive sediment from an on-stream stormwater detention pond. Water Res. 2002, 36, 3849–3859. [Google Scholar] [CrossRef]
- Lau, Y.L. Temperature effect on settling velocity and deposition of cohesive sediments. J. Hydraul. Res. 1994, 32, 41–51. [Google Scholar] [CrossRef]
- Spelman, D.; Sansalone, J.J. Is the treatment response of manufactured bmps to urban drainage pm loads portable? J. Environ. Eng. 2018, 144. [Google Scholar] [CrossRef]
- Laliberté, M.; Cooper, W.E. Model for calculating the density of aqueous electrolyte solutions. J. Chem. Eng. Data 2004, 49, 1141–1151. [Google Scholar] [CrossRef]
- Laliberté, M. Model for calculating the viscosity of aqueous solutions. J. Chem. Eng. Data 2007, 52, 321–335. [Google Scholar] [CrossRef]
- Baird, R.; Eaton, A.D.; Rice, E.W.; Bridgewater, L.; American Public Health Association; American Water Works, Association; Water Environment Federation. Standard Methods for the Examination of Water and Wastewater; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2017; ISBN 978-087553-287-5. [Google Scholar]
- Dierschke, M.; Welker, A. Bestimmung von Feststoffen in Niederschlagsabflüssen (Method for measuring suspended solids in runoffs). GWF Wasser/Abwasser 2015, 156, 440–446. [Google Scholar]
- Sansalone, J.J.; Glenn, D.W. Accretion of pollutants in snow exposed to urban traffic and winter storm maintenance activities. I. J. Environ. Eng. 2002, 128, 151–166. [Google Scholar] [CrossRef]
- Hilliges, R.; Endres, M.; Tiffert, A.; Brenner, E.; Marks, T. Characterization of road runoff with regard to seasonal variations, particle size distribution and the correlation of fine particles and pollutants. Water Sci. Technol. 2017, 75, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Sansalone, J.; Lin, H.; Ying, G. Experimental and field studies of type I settling for particulate matter transported by urban runoff. J. Environ. Eng. 2009, 135, 953–963. [Google Scholar] [CrossRef]
- Sansalone, J.J.; Koran, J.M.; Smithson, J.A.; Buchberger, S.G. Physical characteristics of urban roadway solids transported during rain events. J. Environ. Eng. 1998, 124, 427–440. [Google Scholar] [CrossRef]
- Kayhanian, M.; McKenzie, E.R.; Leatherbarrow, J.E.; Young, T.M. Characteristics of road sediment fractionated particles captured from paved surfaces, surface run-off and detention basins. Sci. Total Environ. 2012, 439, 172–186. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kang, J.-H.; Lau, S.-L.; Kayhanian, M.; Stenstrom, M.K. Optimization of settling tank design to remove particles and metals. J. Environ. Eng. 2008, 134, 885–894. [Google Scholar] [CrossRef]
- Li, Y.; Lau, S.; Kayhanian, M.; Stenstrom, M. Dynamic characteristics of particle size distribution in highway runoff: Implications for settling tank design. J. Environ. Eng. 2006, 132, 852–861. [Google Scholar] [CrossRef]
- Gunawardana, C.; Egodawatta, P.; Goonetilleke, A. Role of particle size and composition in metal adsorption by solids deposited on urban road surfaces. Environ. Pollut. 2014, 184, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Charters, F.J.; Cochrane, T.A.; O’Sullivan, A.D. Particle size distribution variance in untreated urban runoff and its implication on treatment selection. Water Res. 2015, 85, 337–345. [Google Scholar] [CrossRef] [PubMed]
- DIBt. Teil 1: Anlagen zur dezentralen Behandlung des Abwassers von Kfz-Verkehrsflächen zur anschließenden Versickerung in Boden und Grundwasser (Devices treating traffic area runoff for percolation into soil and groundwater). In Zulassungsgrundsätze Niederschlagswasserbehandlungsanlagen (Approval Principles for Stormwater Quality Improvement Devices); Deutsches Insitut für Bautechnik: Berlin, Germany, 2017. [Google Scholar]
- DWA-A 102 (Draft). Grundsätze zur Bewirtschaftung und Behandlung von Regenwetterabflüssen zur Einleitung in Oberflächengewässer (Principles for Management and Treatment of Runoff for Discharge into Surface Waters); DWA Deutsche Vereinigung für Wasserwirtschaft, Abwasser und Abfall e. V.: Hennef, Germany, 2016. [Google Scholar]
- Selbig, W.R.; Fienen, M.N. Regression modeling of particle size distributions in urban storm water: Advancements through improved sample collection methods. J. Environ. Eng. 2012, 138, 1186–1193. [Google Scholar] [CrossRef]
- Lee, D.H.; Min, K.S.; Kang, J.-H. Performance evaluation and a sizing method for hydrodynamic separators treating urban stormwater runoff. Water Sci. 2014, 69, 2122–2131. [Google Scholar] [CrossRef] [PubMed]
- Bardin, J.P.; Barraud, S.; Chocat, B. Uncertainty in measuring the event pollutant removal performance of online detention tanks with permanent outflow. Urban Water 2001, 3, 91–106. [Google Scholar] [CrossRef]
- Kell, G.S. Density, thermal expansivity, and compressibility of liquid water from 0 to 150 °C: Correlations and tables for atmospheric pressure and saturation reviewed and expressed on 1968 temperature scale. J. Chem. Eng. Data 1975, 20, 97–105. [Google Scholar] [CrossRef]
- Corridoni, T.; Mancinelli, R.; Ricci, M.A.; Bruni, F. Viscosity of aqueous solutions and local microscopic structure. J. Phys. Chem. B 2011, 115, 14008–14013. [Google Scholar] [CrossRef] [PubMed]
- Faram, M.G.; Iwugo, K.O.; Andoh, R.Y.G. Characteristics of urban run-off derived sediments captured by proprietary flow-through stormwater interceptors. Water Sci. Technol. 2007, 56, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Gunawardana, C.; Goonetilleke, A.; Egodawatta, P.; Dawes, L.; Kokot, S. Source characterisation of road dust based on chemical and mineralogical composition. Chemosphere 2012, 87, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Gunawardena, J.M.A.; Liu, A.; Egodawatta, P.; Ayoko, G.A.; Goonetilleke, A. Influence of traffic and land use on pollutant transport pathways. In Influence of Traffic and Land Use on Urban Stormwater Quality: Implications for Urban Stormwater Treatment Design; Springer: Singapore, 2018; pp. 27–54. [Google Scholar]
- Ferreira, M.; Stenstrom, M.K. The importance of particle characterization in stormwater runoff. Water Environ. Res. 2013, 85, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Selbig, W.R.; Bannerman, R.T. Characterizing the Size Distribution of Particles in Urban Stormwater by Use of Fixed-Point Sample-Collection Methods; US Geological Survey: Reston, VA, USA, 2011.
- Li, Y.; Lau, S.; Kayhanian, M.; Stenstrom, M. Particle size distribution in highway runoff. J. Environ. Eng. 2005, 131, 1267–1276. [Google Scholar] [CrossRef]
- Faltermaier, S.; Krause, S.; Günther, F.W. Einfluss von Streusalz auf die Flockung Partikulärer Stoffe in Regenbecken an Autobahnen (Influence of De-Icing Salt on the Flocculation of Particulate Matter in Stormwater Tanks on Motorways); Aqua Urbanica 2017; Muschalla, D., Gruber, G., Eds.; TU Graz, Institut für Siedlungswasserwirtschaft und Landschaftswasserbau: Graz, Austria, 2017. [Google Scholar]
- Portela, L.I.; Ramos, S.; Teixeira, A.T. Effect of salinity on the settling velocity of fine sediments of a harbour basin. J. Coast. Res. 2013, 65, 1188–1193. [Google Scholar] [CrossRef]
- Sutherland, B.R.; Barrett, K.J.; Gingras, M.K. Clay settling in fresh and salt water. Environ. Fluid Mech. 2015, 15, 147–160. [Google Scholar] [CrossRef]
- Duzgoren-Aydin, N.S.; Wong, C.S.C.; Song, Z.G.; Aydin, A.; Li, X.D.; You, M. Fate of heavy metal contaminants in road dusts and gully sediments in guangzhou, se china: A chemical and mineralogical assessment. Hum. Ecol. Risk Assess. Int. J. 2006, 12, 374–389. [Google Scholar] [CrossRef]
- Bäckström, M. Sediment transport in grassed swales during simulated runoff events. Water Sci. Technol. 2002, 45, 41–49. [Google Scholar] [CrossRef] [PubMed]
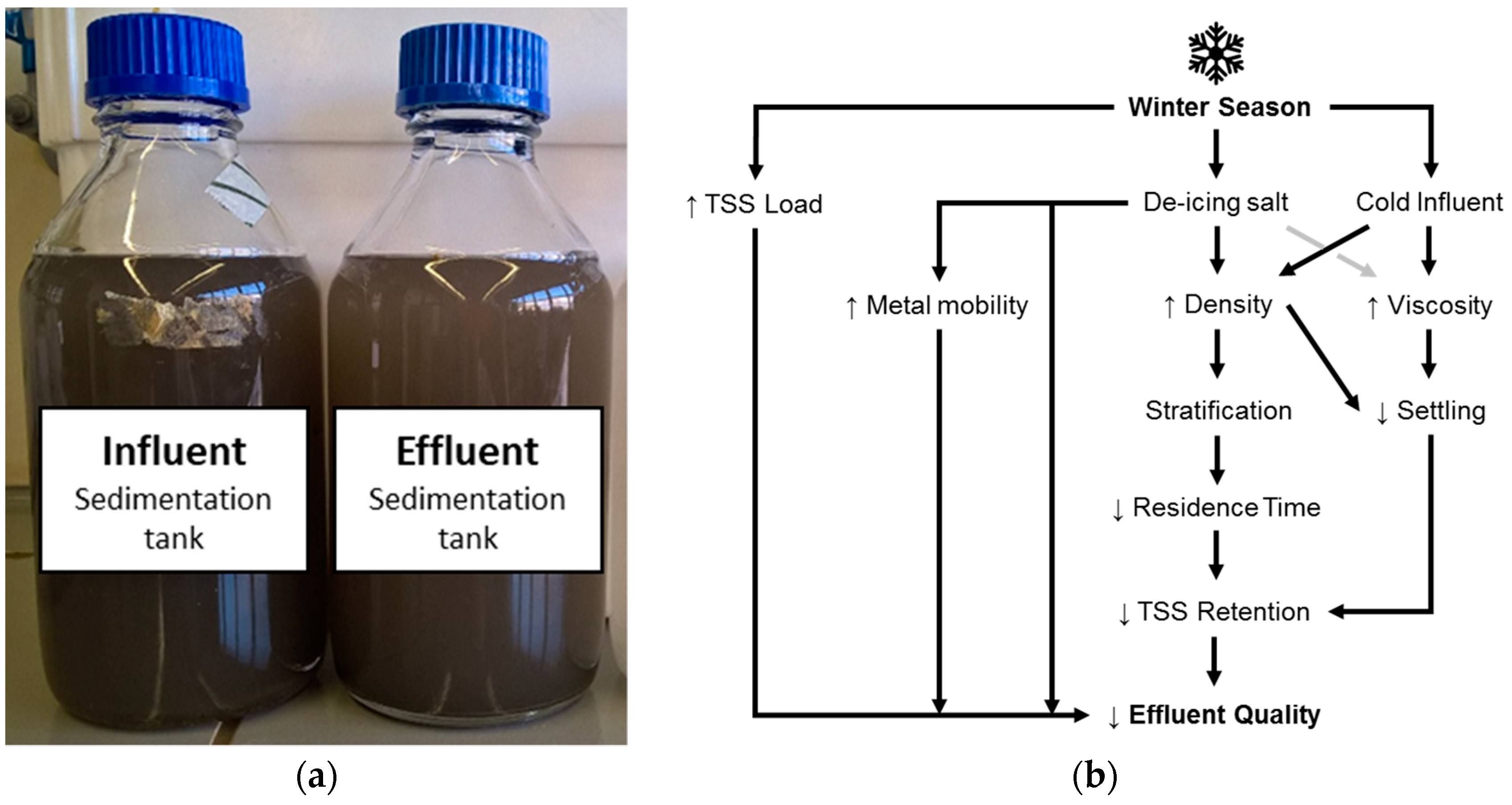
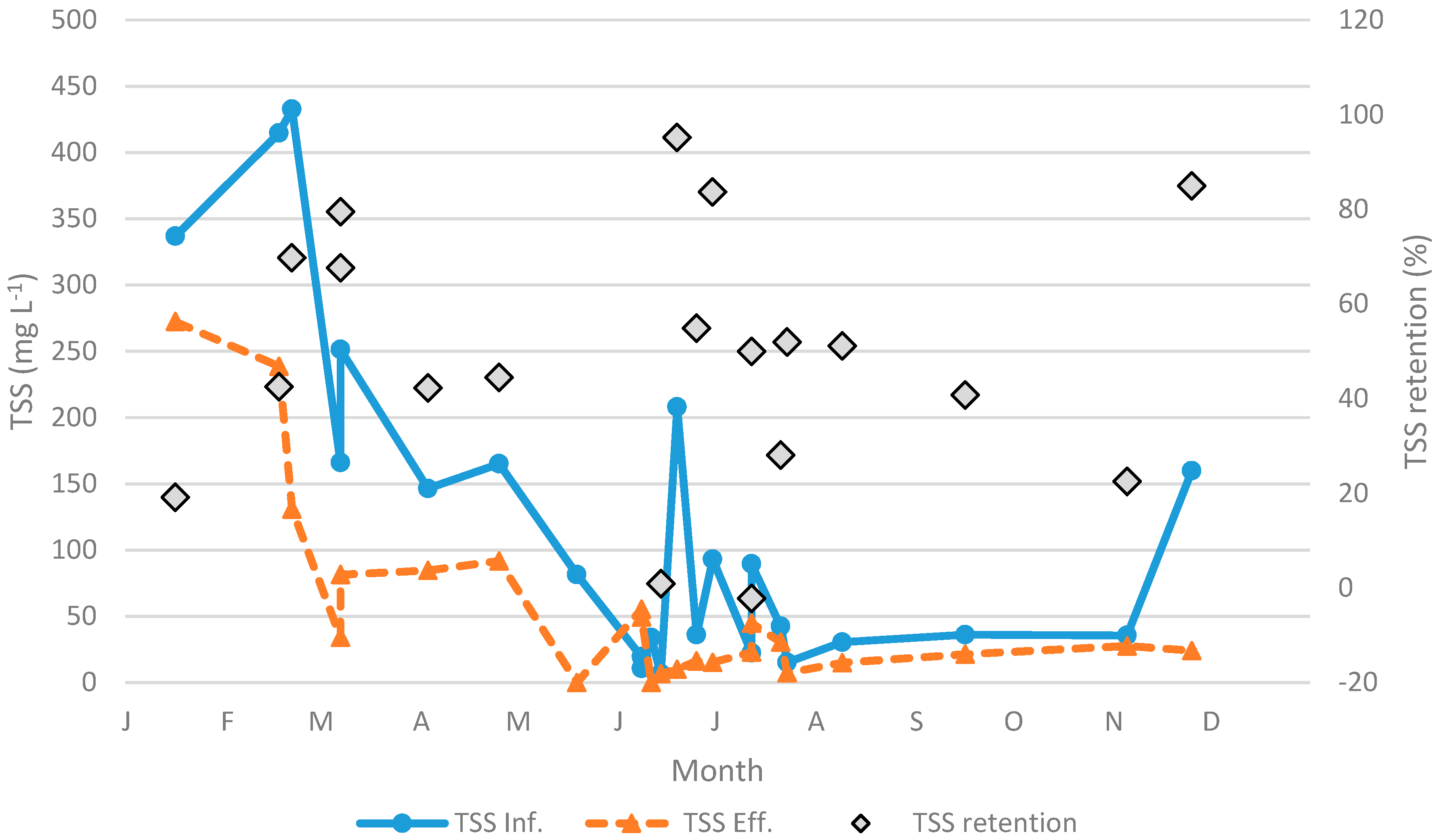
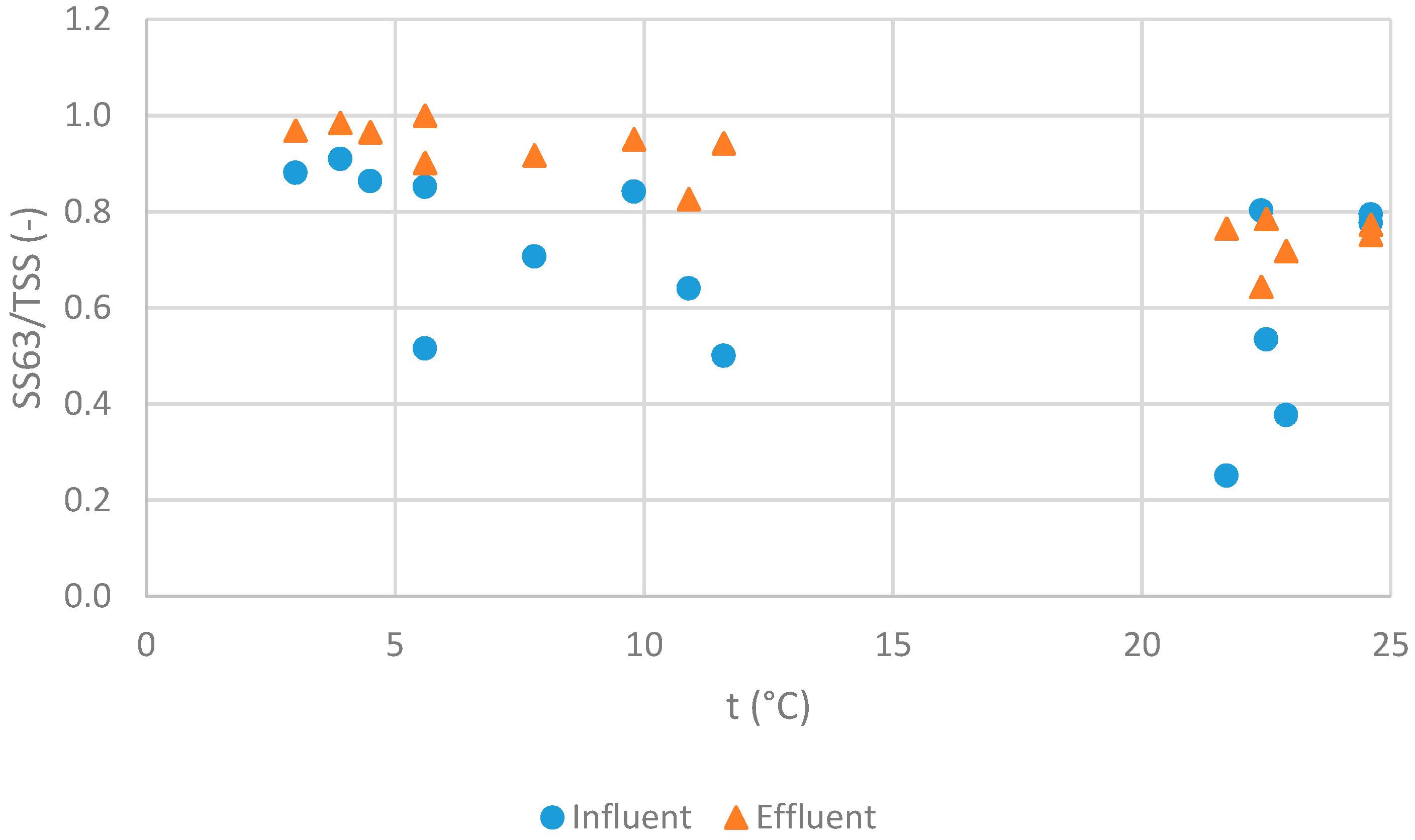
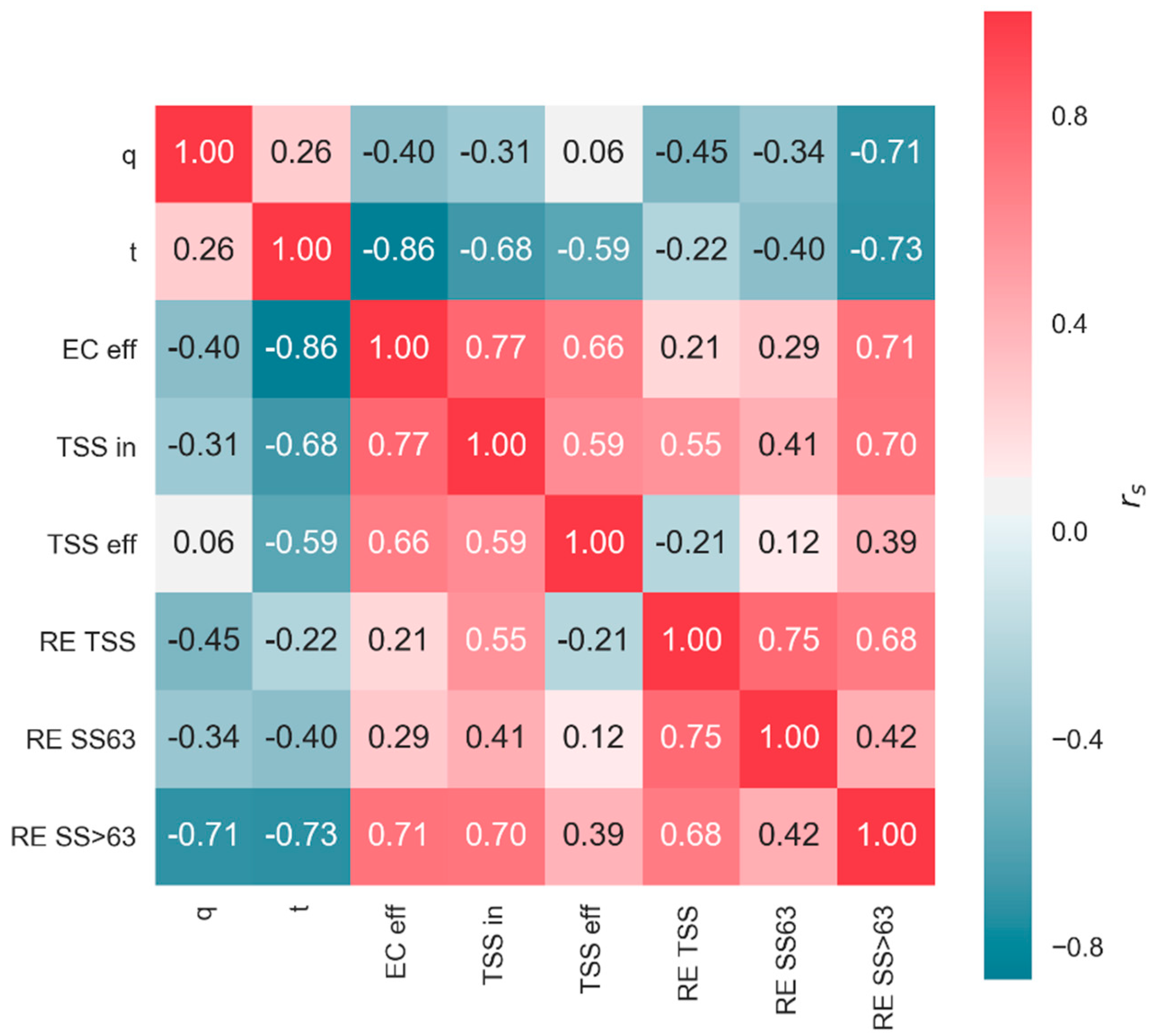
| Parameter | Unit | n | Min | 25th Percentile | Median | Mean | 75th Percentile | Max | SD |
|---|---|---|---|---|---|---|---|---|---|
| q | L s−1 ha−1 | 23 | 1.6 | 3.4 | 5.7 | 8.1 | 7.2 | 26.1 | 7.5 |
| t | °C | 23 | 3.0 | 8.8 | 17.5 | 17.5 | 21.7 | 24.6 | 7.3 |
| TSS Inf. | mg L−1 | 23 | 7 | 32 | 82 | 123 | 166 | 433 | 129 |
| SS > 63 Inf. | mg L−1 | 15 | 3 | 18 | 26 | 35 | 46 | 122 | 30 |
| SS63 Inf. | mg L−1 | 15 | 8 | 18 | 73.3 | 121 | 140 | 394 | 130 |
| EC Eff. | µS cm−1 | 22 | 59 | 95 | 119 | 119 | 867 | 4790 | 1225 |
| TSS retention | % | 21 | −365 | 22 | 44 | 18 | 68 | 95 | 105 |
| SS > 63 retention | % | 15 | −4 | 65 | 83 | 71 | 93 | 100 | 31 |
| SS63 retention | % | 15 | −49 | −1 | 37 | 25 | 57 | 80 | 40 |
| t | ρs | d50 | q | |
|---|---|---|---|---|
| TSS removal efficiency | 0.07 | 0.22 | 0.85 | −0.32 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rommel, S.H.; Helmreich, B. Influence of Temperature and De-Icing Salt on the Sedimentation of Particulate Matter in Traffic Area Runoff. Water 2018, 10, 1738. https://doi.org/10.3390/w10121738
Rommel SH, Helmreich B. Influence of Temperature and De-Icing Salt on the Sedimentation of Particulate Matter in Traffic Area Runoff. Water. 2018; 10(12):1738. https://doi.org/10.3390/w10121738
Chicago/Turabian StyleRommel, Steffen H., and Brigitte Helmreich. 2018. "Influence of Temperature and De-Icing Salt on the Sedimentation of Particulate Matter in Traffic Area Runoff" Water 10, no. 12: 1738. https://doi.org/10.3390/w10121738
APA StyleRommel, S. H., & Helmreich, B. (2018). Influence of Temperature and De-Icing Salt on the Sedimentation of Particulate Matter in Traffic Area Runoff. Water, 10(12), 1738. https://doi.org/10.3390/w10121738






