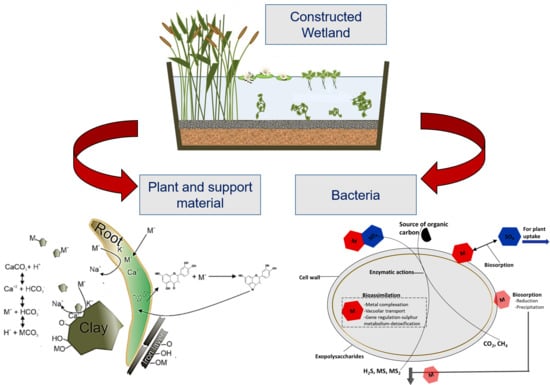
Review of Constructed Wetlands for Acid Mine Drainage Treatment
Abstract
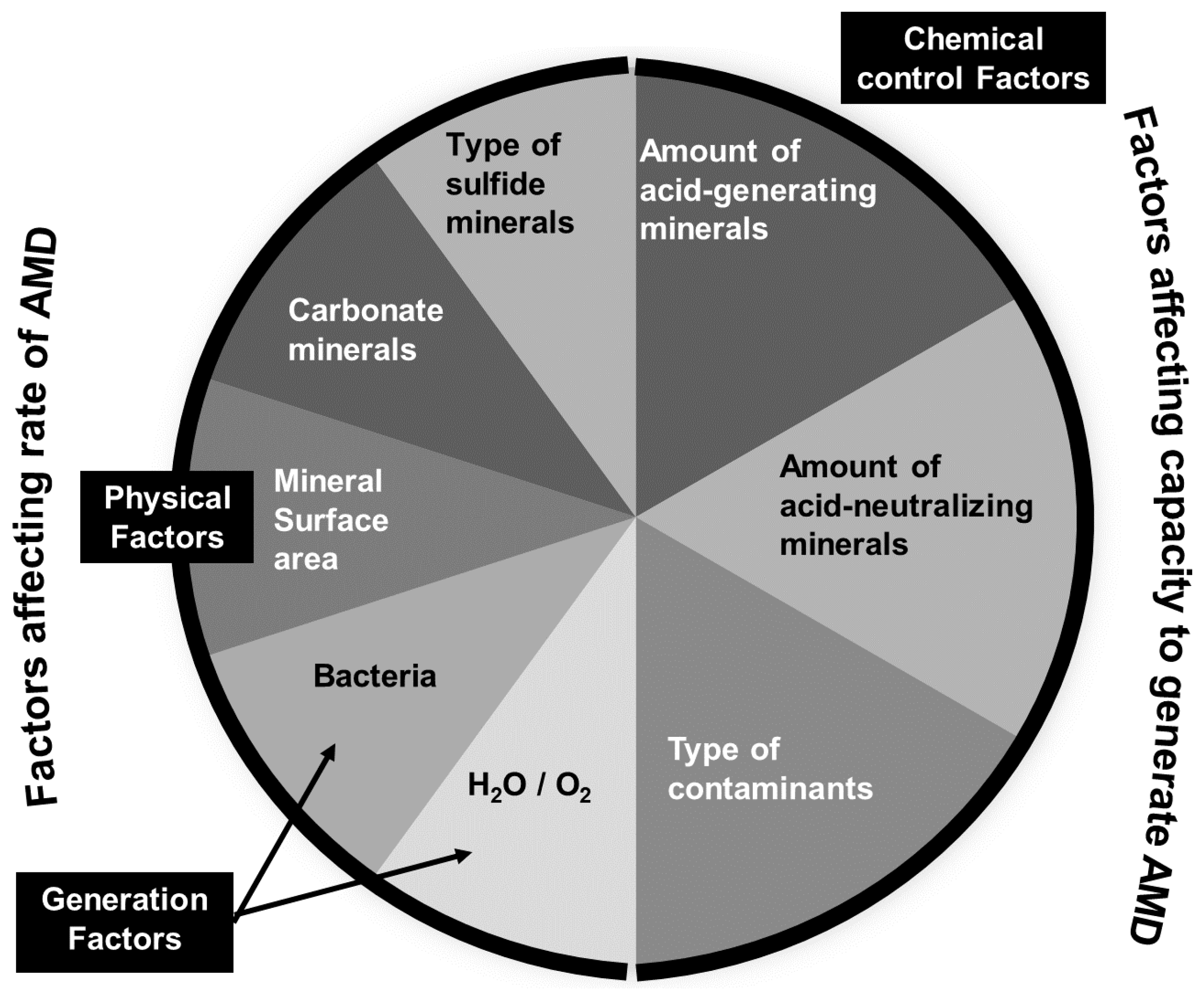
1. Physical-Chemical and Toxicological Characterization of Acid Mine Drainage
2. Constructed Wetlands Applied to AMD Treatment: Types, Operational Parameters, and Efficiency
3. Heavy Metal Uptake Mechanisms in Constructed Wetlands
3.1. Role of Vegetation on the Removal of Heavy Metals
3.1.1. Factors Involved in Metal Uptake Efficiencies by Plants
3.1.2. Accumulation and Detoxification Mechanisms
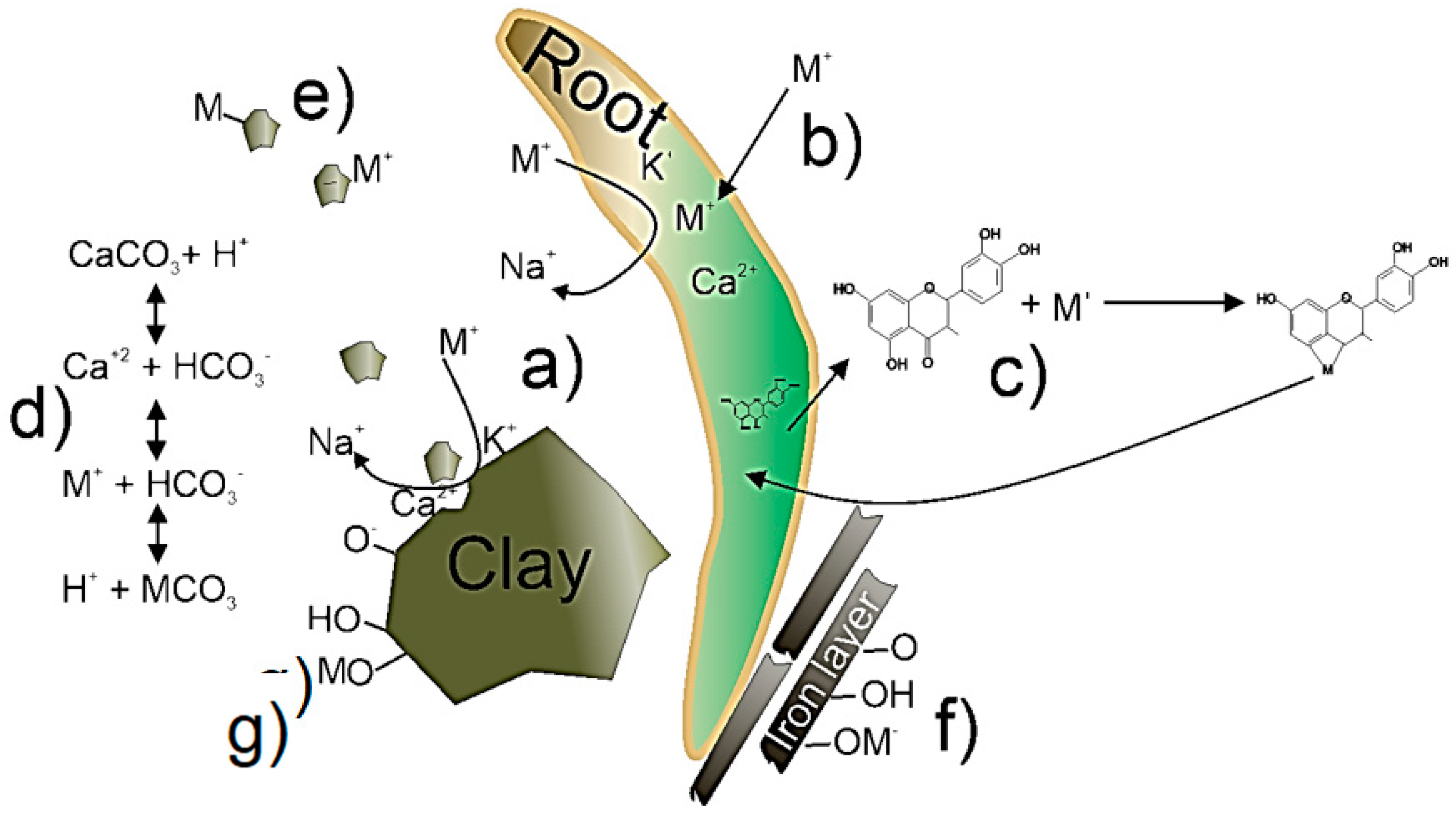
3.2. Contributions from Support Material to Metal Removal
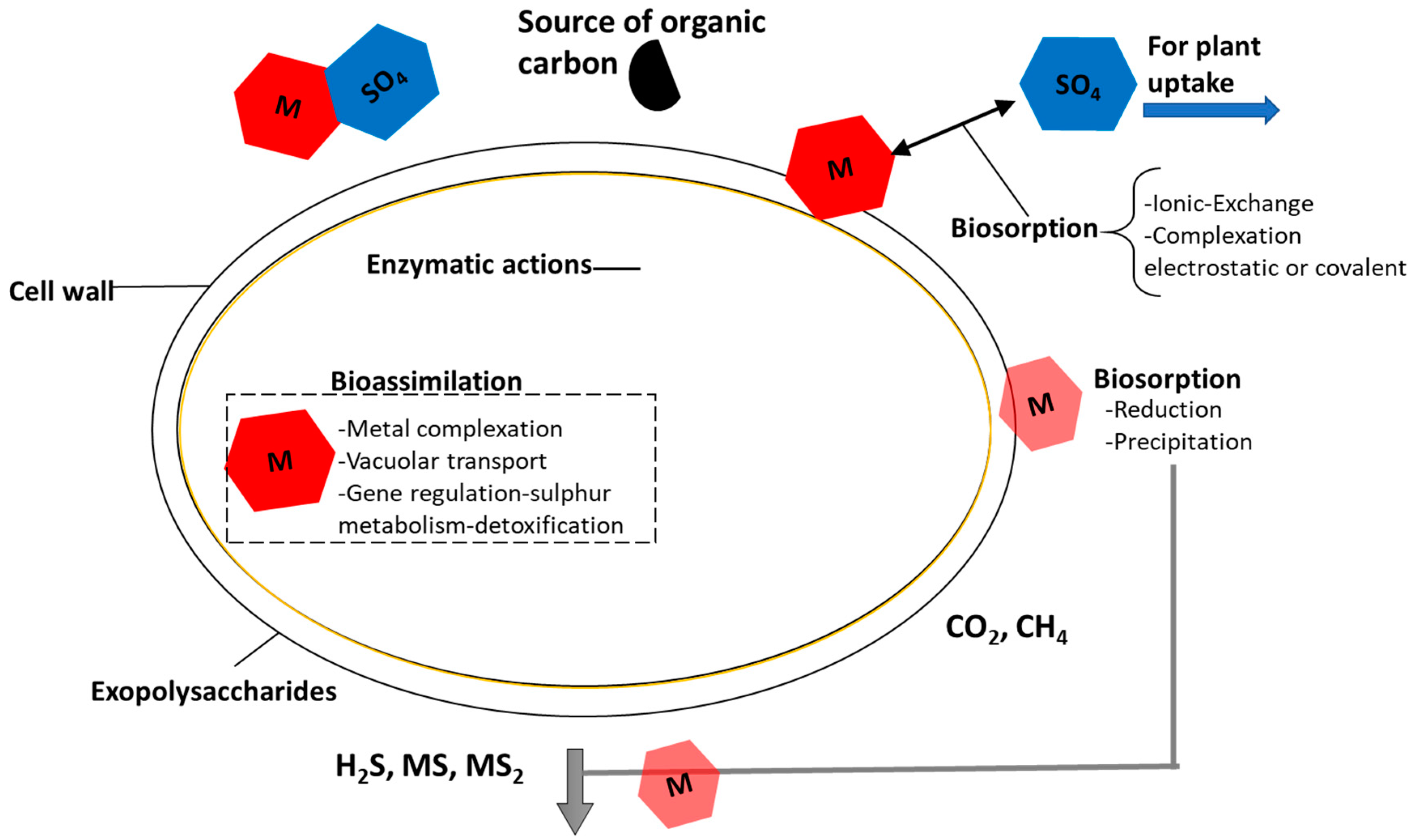
3.3. Role of Microorganisms in CW Treating Acid Mine Drainage Containing Heavy Metals
3.3.1. Microorganism Response to Heavy Metals Presence and Other Conditions of AMD
3.3.2. Plant Growth Promoting Bacterium
3.3.3. Isolated Consortium from Acid Mine Drains
4. Implementation of Constructed Wetlands (CW) for Mine Water Remediation: Scaling and Residues Generated
5. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Skousen, J.; Sexstone, A.; Ziemkiewicz, P. Acid mine drainage control and treatment. In Reclamation of Drastically Disturbed Lands; West Virginia University: Morgantown, WV, USA, 2000. [Google Scholar]
- Akcil, A.; Koldas, S. Acid Mine Drainage (AMD): Causes, treatment and case studies. J. Clean. Prod. 2006, 14, 1139–1145. [Google Scholar] [CrossRef]
- Johnson, D.B. Chemical and microbiological characteristics of mineral spoils and drainage waters at abandoned coal and metal mines. Wat Air Soil Pollut. Focus 2003, 3, 47–66. [Google Scholar] [CrossRef]
- Dold, B. Aguas Ácidas: Formación, predicción, control y prevención. Rev. Miner. 2003, 309, 29–37. [Google Scholar]
- Strosnider, W.; Llanos, F.; Marcillo, C.; Callapa, R.; Nairn, R. Impact on tributaries of the Pilcomayo River by additional pollutants from mine acid drainage from Cerro Rico de Potosí-Bolivia. Adv. Sci. Eng. 2014, 5, 1–17. [Google Scholar]
- Calabrese, E.J.; Kostecki, P.T.; Dragun, J. Contaminated Soils, Sediments and Water Volume 10: Successes and Challenges; Springer Science & Business Media: Berlin, Germany, 2006; ISBN 978-0-387-28324. [Google Scholar]
- Hutchinson, I.P.G.; Ellison, R.D. Mine Waste Management; International Atomic Energy Agency: Vienna, Austria, 1992. [Google Scholar]
- USEPA Technical Document: Acid Mine Drainage Prediction 1994. Available online: http://www.epa.gov/sites/production/file/2015-09/documents/amd.pdf (accessed on 13 September 2018).
- Blowes, D.W.; Ptacek, C.J.; Jambor, J.L.; Weisener, C.G. The geochemistry of acid mine drainage. Treatise Geochem. 2003, 9, 612. [Google Scholar]
- ITRC. Biochemical Reactors for Treating Mining- Influenced Water. BCR-1; Interstate Technology & Regulatory Council, Biochemical Reactors for Mining-Influenced Waste Team: Washington, DC, USA, 2013. [Google Scholar]
- Lopes, I.; Gonçalves, F.; Soares, A.; Ribeiro, R. Ecotoxicological tools in the remediation of acid mine drainage. Toxicol. Environ. Chem. 1999, 70, 441–460. [Google Scholar] [CrossRef]
- Rügner, H.; Finkel, M.; Kaschl, A.; Bittens, M. Application of monitored natural attenuation in contaminated land management—A review and recommended approach for Europe. Environ. Sci. Policy 2006, 9, 568–576. [Google Scholar] [CrossRef]
- Yim, J.; Kim, K.; Kim, S. Effect of hardness on acute toxicity of metal mixtures using Daphnia magna: Prediction of acid mine drainage toxicity. J. Hazard. Mater. B 2006, 138, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Xavier, C.; Chamorro, S.; Vidal, G. Chronic effects of Kraft mill effluents and endocrine active chemical on Daphnia magna. Bull. Environ. Contam. Toxicol. 2005, 75, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Chamorro, S.; López, D.; Brito, P.; Jarpa, P.; Piña, B.; Vidal, G. Sublethal effects of chlorine-free kraft mill effluents on Daphnia magna. Bull. Environ. Contam. Toxicol. 2016, 97, 843–847. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Kennaway, G.; Croteau, M.; Dybowska, A.; Smith, B.; Nogueira, A.; Rainbow, P.; Luoma, S.; Valsami-Jones, E. In vivo retention of ingested Au NPs by Daphnia magna: No evidence for trans-epithelial alimentary uptake. Chemosphere 2014, 100, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Allen, I.; Calow, P.; Baird, D. A mechanistic model of contaminant-induced feeding inhibition in Daphnia magna. Environ. Toxicol. Chem. 1995, 14, 1625–1630. [Google Scholar] [CrossRef]
- Yang, Y.; Wei, X.; Lu, J.; You, J.; Wang, W.; Shi, R. Lead-induced phytotoxicity mechanism involved in seed germination and seedling growth of wheat (Triticum aestivum L.). Ecotoxicol. Environ. Safe 2010, 73, 1982–1987. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, T.; Islam, K.; Muhammad, S. Toxic effects of heavy metals on early growth and tolerance of cereal crops. Pak. J. Bot. 2007, 39, 451–462. [Google Scholar]
- Bagur-González, M.; Estepa-Molina, C.; Martín-Peinado, F.; Morales-Ruano, S. Toxicity assessment using Lactuca sativa L. bioassay of the metal(loid)s As, Cu, Mn, Pb and Zn in soluble in water saturated soil extracts from an abandoned mining site. J. Soil Sediment 2011, 11, 281–289. [Google Scholar] [CrossRef]
- Chamorro, S.; Barata, C.; Piña, B.; Casado, M.; Schwarz, A.; Sáez, K.; Vidal, G. Toxicological analysis of acid mine drainage by water quality and land use bioassays. Mine Water Environ. 2018, 37, 88–97. [Google Scholar] [CrossRef]
- Biehler, D.; Falck, W. Simulation of the effects of geochemical reactions on groundwater quality during planned flooding of the Königstein uranium mine, Saxony, Germany. Hydrogeol. J. 1999, 7, 284–293. [Google Scholar] [CrossRef]
- Nordstrom, D.; Alpers, C.; Ptacek, C.; Blowes, D. Negative pH and extremely acid mine waters from Iron Montain, California. Environ. Sci. Technol. 2000, 34, 254–258. [Google Scholar] [CrossRef]
- Eary, L.; Runnells, D.; Esposito, K. Geochemical controls on ground water composition at the cripple creek mining district, Cripple Creek, Colorado. Appl. Geochem. 2003, 18, 1–24. [Google Scholar] [CrossRef]
- Kimura, S.; Bryan, C.; Hallberg, K.; Johnson, D. Biodiversity and geochemistry of an extremely acidic, low temperature subterranean environment sustained by chemolithotrophy. Environ. Microbiol. 2011, 13, 2092–2104. [Google Scholar] [CrossRef] [PubMed]
- Williams, M. Arsenic in mine waters: An international study. Environ. Geol. 2001, 267–278. [Google Scholar] [CrossRef]
- Younger, P.L.; Banwart, S.A.; Hedin, R.S. Passive Treatment of Polluted Mine Waters. In Mine Water; Environmental Pollution; Springer: Dordrecht, The Netherlands, 2002; pp. 311–396. ISBN 978-1-4020-0138-3. [Google Scholar]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Kalin, M. Passive mine water treatment: The correct approach? Ecol. Eng. 2004, 22, 299–304. [Google Scholar] [CrossRef]
- Johnson, D.; Hallberg, K. Pitfalls of passive mine water treatment. Rev. Environ. Sci. Biotechnol 2002, 1, 335–343. [Google Scholar] [CrossRef]
- Mine Environment Neutral Drainage, (MEND). Review of Passive Systems for Treatment of Acid Mine Drainage; MEND Report 3.14.1; MEND Program: Toronto, ON, Canada, 1999. [Google Scholar]
- US EPA, Constructed Wetlands. Available online: https://www.epa.gov/wetlands/constructed-wetlands (accessed on 13 March 2018).
- Reed, S.C.; Brown, D.S. Constructed wetland design—The first generation. Water Environ. Res. 1992, 64, 776–781. [Google Scholar] [CrossRef]
- Vymazal, J.; Kröpfelová, L. Wastewater Treatment in Constructed Wetlands with Horizontal Sub-Surface Flow; Springer Science & Business Media: Berlin, Germany, 2008; ISBN 978-1-4020-8580-2. [Google Scholar]
- Sheoran, A.S. Management of acidic mine waste water by constructed wetland treatment system: A bench scale study. Eur. J. Sustain. Dev. 2017, 6, 245–255. [Google Scholar] [CrossRef]
- Sheoran, A.S.; Sheoran, V. Heavy metal removal mechanism of acid mine drainage in wetlands: A critical review. Miner. Eng. 2006, 19, 105–116. [Google Scholar] [CrossRef]
- Sheridan, C.; Akcil, A.; Kappelmeyer, U.; Moodley, I. A Review on the Use of Constructed Wetlands for the Treatment of Acid Mine Drainage. In Constructed Wetlands for Industrial Wastewater Treatment; Wiley-Blackwell: Hoboken, NJ, USA, 2018; pp. 249–262. ISBN 978-1-119-26837-6. [Google Scholar]
- Nyquist, J.; Greger, M. A field study of constructed wetlands for preventing and treating acid mine drainage. Ecol. Eng. 2009, 35, 630–642. [Google Scholar] [CrossRef]
- Woulds, C.; Ngwenya, B.T. Geochemical processes governing the performance of a constructed wetland treating acid mine drainage, Central Scotland. Appl. Geochem. 2004, 19, 1773–1783. [Google Scholar] [CrossRef]
- Karathanasis, A.D.; Johnson, C.M. Metal removal potential by three aquatic plants in an acid mine drainage wetland. Mine Water Environ. 2003, 22, 22–30. [Google Scholar] [CrossRef]
- Türker, O.C.; Böcük, H.; Yakar, A. The phytoremediation ability of a polyculture constructed wetland to treat boron from mine effluent. J. Hazard. Mater. 2013, 252, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Younger, P.L.; Henderson, R. Synergistic wetland treatment of sewage and mine water: Pollutant removal performance of the first full-scale system. Water Res. 2014, 55, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Dufresne, K.; Neculita, C.M.; Brisson, J.; Genty, T. Metal retention mechanisms in pilot-scale constructed wetlands receiving acid mine drainage. In Proceedings of the 10th ICARD (International Conference on Acid Rock Drainage)-IMWA (International Mine Water Association), Santiago, Chile, 21–24 April 2015. [Google Scholar]
- Stottmeister, U.; Buddhawong, S.; Kuschk, P.; Wiessner, A.; Mattusch, J. Constructed wetlands and their performance for treatment of water contaminated with arsenic and heavy metals. In Soil and Water Pollution Monitoring, Protection and Remediation; Springer: Berlin, Germany, 2006; pp. 417–432. [Google Scholar]
- Mitsch, W.J.; Wise, K.M. Water quality, fate of metals, and predictive model validation of a constructed wetland treating acid mine drainage. Water Res. 1998, 32, 1888–1900. [Google Scholar] [CrossRef]
- Marchand, L.; Mench, M.; Jacob, D.L.; Otte, M.L. Metal and metalloid removal in constructed wetlands, with emphasis on the importance of plants and standardized measurements: A review. Environ. Pollut. 2010, 158, 3447–3461. [Google Scholar] [CrossRef] [PubMed]
- Leung, H.M.; Duzgoren-Aydin, N.S.; Au, C.K.; Krupanidhi, S.; Fung, K.Y.; Cheung, K.C.; Wong, Y.K.; Peng, X.L.; Ye, Z.H.; Yung, K.K.L.; et al. Monitoring and assessment of heavy metal contamination in a constructed wetland in Shaoguan (Guangdong Province, China): Bioaccumulation of Pb, Zn, Cu and Cd in aquatic and terrestrial components. Environ. Sci. Pollut. Res. Int. 2017, 24, 9079–9088. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Gannon, S.M.; Nevin, K.P.; Franks, A.E.; Lovley, D.R. Stimulating the anaerobic degradation of aromatic hydrocarbons in contaminated sediments by providing an electrode as the electron acceptor. Environ. Microbiol. 2010, 12, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Sarma, H. Metal hyperaccumulation in plants: A review focusing on phytoremediation technology. J Environ. Sci. Technol. 2011, 4, 118–138. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E.; Sajad, M.A. Phytoremediation of heavy metals—Concepts and applications. Chemosphere 2013, 91, 869–881. [Google Scholar] [CrossRef] [PubMed]
- Girdhar, M.; Sharma, N.R.; Rehman, H.; Kumar, A.; Mohan, A. Comparative assessment for hyperaccumulatory and phytoremediation capability of three wild weeds. 3 Biotech 2014, 4, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, G.; Harding, K.; Koller, E.; Pretto, A.D. A comparison of charcoal- and slag-based constructed wetlands for acid mine drainage remediation. Water SA 2013, 39, 369–374. [Google Scholar] [CrossRef]
- Batty, L.C.; Younger, P.L. Growth of Phragmites australis (Cav.) Trin ex. Steudel in mine water treatment wetlands: Effects of metal and nutrient uptake. Environ. Pollut. 2004, 132, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Stein, O.R.; Borden-Stewart, D.; Hook, P.; Jones, W. Seasonal influence on sulfate reduction and zinc sequestration in subsurface treatment wetlands. Water Res. 2007, 41, 3440–3448. [Google Scholar] [CrossRef] [PubMed]
- Brisson, J.; Chazarenc, F. Maximizing pollutant removal in constructed wetlands: Should we pay more attention to macrophyte species selection? Sci. Total Environ. 2009, 13, 3923–3930. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wen, Y.; Zhou, Q.; Huang, J.; Vymazal, J.; Kuschk, P. Sulfate removal and sulfur transformation in constructed wetlands: The roles of filling material and plant biomass. Water Res. 2016, 102, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Oertli, J.J.; Grgurevic, E. Effect of pH on the Absorption of Boron by Excised Barley Roots. Agron. J. 1975, 67, 278–280. [Google Scholar] [CrossRef]
- Dhir, B.; Sharmila, P.; Saradhi, P.P. Potential of aquatic macrophytes for removing contaminants from the environment. Crit. Rev. Environ. Sci. Technol. 2009, 39, 754–781. [Google Scholar] [CrossRef]
- Fleming, E.J.; Cetinić, I.; Chan, C.S.; Whitney King, D.; Emerson, D. Ecological succession among iron-oxidizing bacteria. ISME J. 2014, 8, 804–815. [Google Scholar] [CrossRef] [PubMed]
- Yeh, T. Removal of metals in constructed wetlands: Review. Pract. Period. Hazard. Toxic Radioact. Waste Manage 2008, 12, 96–101. [Google Scholar] [CrossRef]
- He, H.; Veneklaas, E.J.; Kuo, J.; Lambers, H. Physiological and ecological significance of biomineralization in plants. Trends Plant Sci. 2014, 19, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, R.D.; Tripathi, P.; Dwivedi, S.; Kumar, A.; Mishra, A.; Chauhan, P.S.; Norton, G.J.; Nautiyal, C.S. Roles for root iron plaque in sequestration and uptake of heavy metals and metalloids in aquatic and wetland plants. Metallomics 2014, 6, 1789–1800. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.M. Toxicity of glutathione-binding metals: A review of targets and mechanisms. Toxics 2015, 3, 20–62. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.J.K.; Ahmaruzzaman, M. A review on potential usage of industrial waste materials for binding heavy metal ions from aqueous solutions. J. Water Process Eng. 2016, 10, 39–47. [Google Scholar] [CrossRef]
- Nayek, S.; Gupta, S.; Saha, R. Effects of metal stress on biochemical response of some aquatic macrophytes growing along an industrial waste discharge channel. J. Plant Interact. 2010, 5, 91–99. [Google Scholar] [CrossRef]
- Lyubenova, L.; Kuhn, A.J.; Höltkemeier, A.; Schröder, P. Root exudation pattern of Typha latifolia L. plants after copper exposure. Plant Soil 2013, 370, 187–195. [Google Scholar] [CrossRef]
- Montiel-Rozas, M.M.; Madejón, E.; Madejón, P. Effect of heavy metals and organic matter on root exudates (low molecular weight organic acids) of herbaceous species: An assessment in sand and soil conditions under different levels of contamination. Environ. Pollut. 2016, 216, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Brodie, G.A.; Britt, C.R.; Tomaszewski, T.M.; Taylor, H.N. Anoxic Limestone Drains to Enhance Performance of Aerobic Acid Drainage Treatment Wetlands the Tennessee Valley Authority; CRC Press: Boca Raton, FL, USA, 1992. [Google Scholar]
- Bai, J.; Yang, Z.; Cui, B.; Gao, H.; Ding, Q. Some heavy metals distribution in wetland soils under different land use types along a typical plateau lake, China. Soil Tillage Res. 2010, 106, 344–348. [Google Scholar] [CrossRef]
- Than, K. Principles of Soil Chemistry, 4th ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2011. [Google Scholar]
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, G.; Ng, W.J.; Tan, S.K. A review on removing pharmaceutical contaminants from wastewater by constructed wetlands: Design, performance and mechanism. Sci. Total Environ. 2014, 468–469, 908–932. [Google Scholar] [CrossRef] [PubMed]
- Adrados, B.; Sánchez, O.; Arias, C.A.; Becares, E.; Garrido, L.; Mas, J.; Brix, H.; Morató, J. Microbial communities from different types of natural wastewater treatment systems: Vertical and horizontal flow constructed wetlands and biofilters. Water Res. 2014, 55, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Kuschk, P.; Brix, H.; Vymazal, J.; Dong, R. Development of constructed wetlands in performance intensifications for wastewater treatment: A nitrogen and organic matter targeted review. Water Res. 2014, 57, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Haydar, S.; Haider, H.; Nadeem, O.; Hussain, G.; Zahra, S. Proposed model for wastewater treatment in Lahore using constructed wetlands. J. Fac. Eng. Technol. 2015, 22, 9–19. [Google Scholar]
- Martín-González, A.; Borniquel, S.; Díaz, S.; Ortega, R.; Gutiérrez, J.C. Ultrastructural alterations in ciliated protozoa under heavy metal exposure. Cell Biol. Int. 2005, 29, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Amaro, F.; Turkewitz, A.P.; Martín-González, A.; Gutiérrez, J.-C. Whole-cell biosensors for detection of heavy metal ions in environmental samples based on metallothionein promoters from Tetrahymena thermophila. Microb. Biotechnol. 2011, 4, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Kamika, I.; Momba, M.N.B. Assessing the resistance and bioremediation ability of selected bacterial and protozoan species to heavy metals in metal-rich industrial wastewater. BMC Microbiol. 2013, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Motlagh, A.M.; Bhattacharjee, A.S.; Goel, R. Microbiological study of bacteriophage induction in the presence of chemical stress factors in enhanced biological phosphorus removal (EBPR). Water Res. 2015, 81, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska-Niczyporuk, A.; Bajguz, A.; Zambrzycka, E.; Godlewska-Żyłkiewicz, B. Phytohormones as regulators of heavy metal biosorption and toxicity in green alga Chlorella vulgaris (Chlorophyceae). Plant Physiol. Biochem. 2012, 52, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, H.; Kong, X.; He, W.; Qin, N.; He, Q.; Wang, Y.; Wang, R.; Xu, F. Effects of five heavy metals at sub-lethal concentrations on the growth and photosynthesis of Chlorella vulgaris. Chin. Sci. Bull. 2012, 57, 3363–3370. [Google Scholar] [CrossRef]
- Kumari, A.; Lal, B.; Rai, U.N. Assessment of native plant species for phytoremediation of heavy metals growing in the vicinity of NTPC sites, Kahalgaon, India. Int. J. Phytoremediation 2016, 18, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Thakur, S.; Singh, L.; Wahid, Z.A.; Siddiqui, M.F.; Atnaw, S.M.; Din, M.F.M. Plant-driven removal of heavy metals from soil: Uptake, translocation, tolerance mechanism, challenges, and future perspectives. Environ. Monit. Assess. 2016, 188, 206. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Fan, J.; Zhang, J.; Ngo, H.H.; Guo, W.; Hu, Z.; Liang, S. Decentralized domestic wastewater treatment using intermittently aerated vertical flow constructed wetlands: Impact of influent strengths. Bioresour. Technol. 2015, 176, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, H.; Chen, Z.-S.; Chen, G. Biosynthesis of Nanoparticles by Microorganisms and Their Applications. J. Nanomater. 2011, 2011, 1–16. [Google Scholar] [CrossRef]
- Hafeznezami, S.; Kim, J.L.; Redman, J. Redman Jeremy Evaluating Removal Efficiency of Heavy Metals in Constructed Wetlands. J. Environ. Eng. 2012, 138, 475–482. [Google Scholar] [CrossRef]
- Moreau, J.W.; Fournelle, J.H.; Banfield, J.F. Quantifying Heavy Metals Sequestration by Sulfate-Reducing Bacteria in an Acid Mine Drainage-Contaminated Natural Wetland. Front. Microbiol. 2013, 4, 43. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, K.B.; González-Toril, E.; Johnson, D.B. Acidithiobacillus ferrivorans, sp. nov.; facultatively anaerobic, psychrotolerant iron-, and sulfur-oxidizing acidophiles isolated from metal mine-impacted environments. Extremophiles 2010, 14, 9–19. [Google Scholar] [CrossRef] [PubMed]
- White, C.; Gadd, G.M. An internal sedimentation bioreactor for laboratory-scale removal of toxic metals from soil leachates using biogenic sulphide precipitation. J. Ind. Microbiol. Biotechnol. 1997, 18, 414–421. [Google Scholar] [CrossRef]
- Wu, S.; Kuschk, P.; Wiessner, A.; Müller, J.; Saad, R.A.B.; Dong, R. Sulphur transformations in constructed wetlands for wastewater treatment: A review. Ecol. Eng. 2013, 52, 278–289. [Google Scholar] [CrossRef]
- Vymazal, J. Plants used in constructed wetlands with horizontal subsurface flow: A review. Hydrobiologia 2011, 674, 133–156. [Google Scholar] [CrossRef]
- Piccirillo, C.; Pereira, S.I.A.; Marques, A.P.G.C.; Pullar, R.C.; Tobaldi, D.M.; Pintado, M.E.; Castro, P.M.L. Bacteria immobilisation on hydroxyapatite surface for heavy metals removal. J. Environ. Manag. 2013, 121, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, Q.; Li, M.-M.; Chen, T.-H.; Zhou, Y.-F.; Yue, Z.-B. Competitive adsorption of heavy metal by extracellular polymeric substances (EPS) extracted from sulfate reducing bacteria. Bioresour. Technol. 2014, 163, 374–376. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Lyu, T.; Bai, S.; Li, Z.; Ding, H.; You, S.; Xie, Q. Effect of multilayer substrate configuration in horizontal subsurface flow constructed wetlands: Assessment of treatment performance, biofilm development, and solids accumulation. Environ. Sci. Pollut. Res. Int. 2018, 25, 1883–1891. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Díaz, M.I.; Riveros-Rosas, H.; Campos-García, J. Reducción bacteriana de cromo hexavalente: Mecanismos y aplicaciones. Rev. Educ. Bioquím. 2009, 28, 73–79. [Google Scholar]
- Amabilis-Sosa, L.E.; Siebe, C.; Moeller-Chávez, G.; Durán-Domínguez-de-Bazúa, M.D.C. Accumulation and distribution of lead and chromium in laboratory-scale constructed wetlands inoculated with metal-tolerant bacteria. Int. J. Phytoremediation 2015, 17, 1090–1096. [Google Scholar] [CrossRef] [PubMed]
- Patten, C.L.; Glick, B.R. Regulation of indoleacetic acid production in Pseudomonas putida GR12-2 by tryptophan and the stationary-phase sigma factor RpoS. Can. J. Microbiol. 2002, 48, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Nester, E.W. Indoleacetic acid, a product of transferred DNA, inhibits vir gene expression and growth of Agrobacterium tumefaciens C58. Proc. Natl. Acad. Sci. USA 2006, 103, 4658–4662. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Oliveira, R.S.; Freitas, H.; Zhang, C. Biochemical and molecular mechanisms of plant-microbe-metal interactions: Relevance for phytoremediation. Front. Plant Sci. 2016, 7, 918. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.A.; Hussain, I.; Rasheed, R.; Iqbal, M.; Riaz, M.; Arif, M.S. Advances in microbe-assisted reclamation of heavy metal contaminated soils over the last decade: A review. J. Environ. Manag. 2017, 198, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.J.; Kjellerup, B.V. Applications of biofilms in bioremediation and biotransformation of persistent organic pollutants, pharmaceuticals/personal care products, and heavy metals. Appl. Microbiol. Biotechnol. 2013, 97, 9909–9921. [Google Scholar] [CrossRef] [PubMed]
- Carpio, I.E.M.; Machado-Santelli, G.; Sakata, S.K.; Ferreira Filho, S.S.; Rodrigues, D.F. Copper removal using a heavy-metal resistant microbial consortium in a fixed-bed reactor. Water Res. 2014, 62, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Neu, T.R.; Lawrence, J.R. Advanced techniques for in situ analysis of the biofilm matrix (structure, composition, dynamics) by means of laser scanning microscopy. Methods Mol. Biol. 2014, 1147, 43–64. [Google Scholar] [PubMed]
- Kelly, J.J.; Minalt, N.; Culotti, A.; Pryor, M.; Packman, A. Temporal variations in the abundance and composition of biofilm communities colonizing drinking water distribution pipes. PLoS ONE 2014, 9, e98542. [Google Scholar] [CrossRef] [PubMed]
- Silver, S. Bacterial Heavy Metal Detoxification and Resistance Systems. In Biotechnology and Environmental Science: Molecular Approaches; Mongkolsuk, S., Lovett, P.S., Trempy, J.E., Eds.; Springer: Boston, MA, USA, 1992; pp. 109–129. ISBN 978-0-585-32386-2. [Google Scholar]
- Syed, S.; Chinthala, P. Heavy Metal detoxification by different Bacillus species isolated from solar sSalterns. Scientifica 2015, 2015, 319760. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Shi, W.; Zeng, X.-C.; Yang, Y.; Zhou, L.; Mu, Y.; Liu, Y. Draft genome sequence of Arthrobacter sp. strain B6 isolated from the high-arsenic sediments in Datong Basin, China. Stand. Genom. Sci. 2017, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Samanta, A.; Bera, P.; Khatun, M.; Sinha, C.; Pal, P.; Lalee, A.; Mandal, A. An investigation on heavy metal tolerance and antibiotic resistance properties of bacterial strain Bacillus sp. isolated from municipal waste. J. Microbiol. Biotechnol. Res. 2017, 2, 178–189. [Google Scholar]
- Diels, L.; Mergeay, M. DNA Probe-mediated detection of resistant bacteria from soils highly polluted by heavy metals. Appl Environ. Microbiol. 1990, 56, 1485–1491. [Google Scholar] [PubMed]
- Simmons, S.L.; DiBartolo, G.; Denef, V.J.; Goltsman, D.S.A.; Thelen, M.P.; Banfield, J.F. Population genomic analysis of strain variation in Leptospirillum Group II bacteria involved in acid mine drainage formation. PLoS Biol. 2008, 6, e177. [Google Scholar] [CrossRef] [PubMed]
- Kuppusamy, S.; Thavamani, P.; Megharaj, M.; Lee, Y.B.; Naidu, R. Kinetics of PAH degradation by a new acid-metal-tolerant Trabulsiella isolated from the MGP site soil and identification of its potential to fix nitrogen and solubilize phosphorous. J. Hazard. Mater. 2016, 307, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Zhang, B.; Zhang, J.; Ngo, H.H.; Guo, W.; Liu, F.; Guo, Y.; Wu, H. Intermittent aeration strategy to enhance organics and nitrogen removal in subsurface flow constructed wetlands. Bioresour. Technol. 2013, 141, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Skousen, J.; Zipper, C.E.; Rose, A.; Ziemkiewicz, P.F.; Nairn, R.; McDonald, L.M.; Kleinmann, R.L. Review of Passive Systems for Acid Mine Drainage Treatment. Mine Water Environ. 2017, 36, 133–153. [Google Scholar] [CrossRef]
- Wildeman, T.R.; Duggan, L.; Phillips, P.; Rodriguez-Eaton, S.; Simms, R.; Bender, J. Passive treatment methods for manganese: Preliminary results from two pilot sites. The Challenge of Integrating Diverse Perspectives in Reclamation. In Proceedings of the 10th Annual National Meeting of the American Society of Surface Mining and Reclamation, Spokane, WA, USA, 16–19 May 1993; pp. 16–19. [Google Scholar]
- Hedin, R.S.; Watzlaf, G.R.; Nairn, R.W. Passive treatment of acid mine drainage with limestone. J. Environ. Qual. 1994, 23, 1338–1345. [Google Scholar] [CrossRef]
- Rose, A.W.; Dietz, J.M. Case studies of passive treatment systems: Vertical flow systems. In Proceedings of the 2002 National Meeting of the American Society of Mining and Reclamation, Lexington, KY, USA, 9–13 June 2002; pp. 776–797. [Google Scholar]
- Younger, P.L. The adoption and adaptation of passive treatment technologies for mine waters in the United Kingdom. Mine Water Environ. 2000, 19, 84–97. [Google Scholar] [CrossRef]
- Skousen, J.; Ziemkiewicz, P. Performance of 116 passive treatment systems for acid mine drainage. J. Am. Soc. Min. Reclam. 2005, 1100–1133. [Google Scholar] [CrossRef]
- ITRC. The Interstate Technology & Regulatory Council. Technical and Regulatory Guidance Document for Constructed Treatment Wetlands. 2003. The Interstate Technology & Regulatory Council Wetland Team. Available online: https://www.itrcweb.org/GuidanceDocuments/WTLND-1.pdf (accessed on 30 July 2018).
- Sullivan, C.; Tyrer, M.; Cheeseman, C.R.; Graham, N.J.D. Disposal of water treatment wastes containing arsenic—A review. Sci. Total Environ. 2010, 408, 1770–1778. [Google Scholar] [CrossRef] [PubMed]
- Swash, P.M.; Monhemius, A.J. Characteristics and stabilities of residues from the Wheal Jane constructed wetlands. Sci. Total Environ. 2005, 338, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Gadepalle, V.; Ouki, S.; Herwijnen, R. Immobilization of heavy metals in soil using natural and waste materials for vegetation establishment on contaminated sites. Soil Sediment Contam. 2007, 16, 233–251. [Google Scholar] [CrossRef]
| Description | pH | SO42− | As | Cd | Cr | Cu | Fe | Zn | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Underground water near Königstein mine, Germany # | 5.9 | 33 | <0.02 | 1.5 | <0.01 | [22] | |||
| Unsaturated area near the Königstein mine, Germany * | 1.9 | 12,322 | 0.97 | 1171 | 132 | [22] | |||
| Aquifer near the Richmond mine, USA | 0.5–1 | 20,000–108,000 | 3–222 | 4.0–19.0 | 120–650 | 13,000–19,000 | 700–2600 | [23] | |
| Aquifer affected by Carlton mine, USA | 7 | 1292 | 0.01 | 0.04 | [24] | ||||
| Groundwater from different locations in the Cae mine, Canada | 1.9–2.3 | 19–265 | 0.04–37 | 31,570–1100 | 1.3–120.0 | [25] | |||
| Mine Mamut, Malaysia | 2.6 | 14.8 | 299 | 443 | 90 | [26] | |||
| Groundwater close to iron mine, Zimbabwe | 1.5 | 355,425 | 72 | 3.7 | 18 | 20 | 132,909 | 55 | [26] |
| Type of CW | Influent Characteristics | Operational Parameters | Type of Vegetation | Removal Efficiency of Metal (%) | Reference | |
|---|---|---|---|---|---|---|
| Metal (mg/L) | (a) SO42− (mg L−1) (b) pH (c) Alkalinity (mg/L CaCO3) | (a) HRT (d) (b) Hydraulic load (m3/m2·d) (c) Scale | ||||
| Surface Flow | Fe (260.5 ± 23.7) Zn (5.9 ± 0.3) Cu (1.44 ± 0.1) Cd (0.006 ± 0.0) | (a) 1336 ± 5.0 (b) 2.65 ± 0.02 (c) 0 | (a) - (b) 0.033 (c) small-scale | Carex rostrata Eriophorum angustifolium Phragmites australis | −0.8–0.4 −3.4–8.5 50–57 | [38] |
| Al (30–100) Ca ( - ) Fe (200–250) Mg (30–100) Mn (30–100) S ( - ) | (a) 500–1000 (b) 2.6 (c) - | (a) - (b) - (c) full-scale | Typhia latifolia | 23–30 5–8 25–31 17 18–19 17–18 | [39] | |
| Fe (12.0) Al (11.3) Zn (0.385) Ni (0.388) Cu (0.03) Pb (0.01) | (a) 2610 (b) 2.54–2.99 (c) - | (a) - (b) - (c) lab-scale | Chrysopogon zizanioides | - | [41] | |
| Fe (44–205/1.3) Mn (5.9–7.4/0.2) Al (0.02–0.29/0.1) Zn (<0.009–0.03/<0.009) Cd (<0.006–0.02/<0.006) B (0.01–1.17/<0.006) As (0.0009–0.1/0.0004) Pb (<0.002–0.0022–<0.002) | (a) - (b) 6.3–7.2/5.7 (c) - | (a) - (b) 113.68/302.69/928.8 (c) full-scale | Scirpus cyperinus Typha latifolia Juncus effusus | 97–98/10 47–79/40 - 33/- 100/- 52/- 99–100/- 26/- | [42] | |
| Fe (17.861) Cu (14.620) Zn (29.367) Pb (1.753) Co (0.323) Ni (0.388) Mn (2.143) | (a) 395 (b) 2.93–3.31 (c) 0 | (a) 1,2,3,4,7 (b) - (c) bench-scale | Desmostachya bipinnata | 95–96 89–92 77–78 89–90 68–72 30–64 36–76 | [43] | |
| Subsurface flow | Al (12.6 ± 4.1) Fe (787 ± 121) Mn (10.9 ± 2.1) | (a) - (b) 3.38 ± 0.45 (c) - | (a) - (b) - (c)full-scale | Typha latifolia Scirpus validus Bidens aristosa | 95.8 99.9 98.4 | [40] |
| B (187) Ca (54.9) Mn (19.6) Na (318) | (a) - (b) 8.96 (c) - | (a) 15 (b) 0.00078 (c) small-scale | Typha latifolia Phragmites australis | 30–37 20–25 30–34 30–33.5 | [38] | |
| Fe (1–191) Al (<1–48) Ca (64–170) | (a) 312–1603 (b) 3.4–6.1 (c) 0–54 | (a) 2.4–27 (b) 0.12–0.75 (c) full-scale | - | 0–92 0–90 (−8)–(−57) | [36] | |
| Fe (38.1) Mn (2.6) Ni (0.4) Zn (9.0) | (a) 292–377 (b) 4.2 (c) - | (a) 5 (b) 0.01 (c) small-scale | Typhia latifolia | 98.6/89.8 75.5/−20.3 88.5/58.1 96.7/96.3 | [43] | |
| Hybrid (surface + subsurface flow) | Zn (1.8) As (0.5) | (a) 2000–3000 (b) 3.0 (c) - | (a) - (b) Batch system (c) small-scale | Juncus effusus | 67 98 | [44] |
| Fe (166) Al (83) Mn (250) | (a) 1672 (b) 2.73–3.08 (c) 48-63.1 | (a) 12 (b) 4.6 (c) full-scale | Typha sp. Typha latifolia | 82.35 61.25 94.9 | [45] | |
| Genus | Phenotype | Mine Water Type |
|---|---|---|
| Leptospirillum spp. | iron-oxidizer, mesophile | Tin, Cooper, cooper with pH ≤ 1.0 |
| Ferroplasma acidiphillum | iron-reducing heterotroph | Tin, cooper and copper wit pH ≤ 1.0 |
| Acidocella spp. | iron-reducing heterotroph | Tin |
| Ferrimicrobium spp. | iron-oxidizing/reducing heterotroph, mesophile | Tin and copper wit pH ≤ 1.0 |
| Acidimicrobium ferroxidans | Fe ox./red. Heterotroph, moderate thermophile | Copper with pH ≤ 1.0 |
| Ferrovum myxofaciens | iron-oxidizer, psychrotolerant | Copper with pH ≤ 1.0 |
| Thiomonas spp. | Iron-and sulfur-oxidizer, moderate acidophile | Coal, tin and copper |
| Halothiobacillus spp. | Iron-and sulfur-oxidizer, moderate acidophile | Tin |
| Acidobacterium-like spp. | iron-reducing heterotroph | Tin and copper |
| Ferroplasma spp. | iron-oxidizing/reducing heterotroph | Copper with pH ≤ 1.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pat-Espadas, A.M.; Loredo Portales, R.; Amabilis-Sosa, L.E.; Gómez, G.; Vidal, G. Review of Constructed Wetlands for Acid Mine Drainage Treatment. Water 2018, 10, 1685. https://doi.org/10.3390/w10111685
Pat-Espadas AM, Loredo Portales R, Amabilis-Sosa LE, Gómez G, Vidal G. Review of Constructed Wetlands for Acid Mine Drainage Treatment. Water. 2018; 10(11):1685. https://doi.org/10.3390/w10111685
Chicago/Turabian StylePat-Espadas, Aurora M., Rene Loredo Portales, Leonel E. Amabilis-Sosa, Gloria Gómez, and Gladys Vidal. 2018. "Review of Constructed Wetlands for Acid Mine Drainage Treatment" Water 10, no. 11: 1685. https://doi.org/10.3390/w10111685
APA StylePat-Espadas, A. M., Loredo Portales, R., Amabilis-Sosa, L. E., Gómez, G., & Vidal, G. (2018). Review of Constructed Wetlands for Acid Mine Drainage Treatment. Water, 10(11), 1685. https://doi.org/10.3390/w10111685