Environmental Factors and the Microbial Quality of Urban Drinking Water in a Low-Income Country: The Case of Madagascar
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Strategy and Microbial Analysis
2.2. Samples Selected for the Analysis
2.3. Parameters Selected for the Description of the Water Supply Systems
2.4. Statistical Analysis
3. Results
3.1. Spatio-Temporal Variation of Water Contamination
3.2. Definition of Profiles of Water Contamination Based on Temporal Variation and Most Relevant Contamination
3.3. Impact of Rainfall Pattern on Contamination
3.4. Main Factors Influencing Water Quality (Environmental Clustering)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Byles, D.B. Water supply and health. Nature 1944, 154, 660–662. [Google Scholar] [CrossRef]
- UNICEF; WHO. Launch Version July 12 Main Report Progress on Drinking Water, Sanitation and Hygiene 2017. Available online: http://www.who.int/mediacentre/news/releases/2017/launch-version-report-jmp-water-sanitation-hygiene.pdf (accessed on 15 June 2018).
- Cohen, B. Urbanization in developing countries: Current trends, future projections, and key challenges for sustainability. Technol. Soc. 2006, 28, 63–80. [Google Scholar] [CrossRef]
- Christopher, C.; Estol-Peixoto, R.; Hartjes, E. Urban Environments in Low-Income and Lower Middle-Income Countries: Policy Performance Indicators at the Subnational Level. Available online: http://www.lafollette.wisc.edu/images/publications/workshops/2012-MCC.pdf (accessed on 15 June 2018).
- Hardoy, J.; Mitlin, D.; Satterthwaite, D. Environmental Problems in an Urbanizing World: Finding Solutions in Cities in Africa, Asia and Latin America, 2nd ed.; Routledge: London, UK, 2001. [Google Scholar]
- Anonimous. State of the World’s Cities 2012/2013: Prosperity of Cities; UN Habitat: Nairobi, Kenya, 2013. [Google Scholar]
- Jamwal, P.; Mittal, A.; Mouchel, J. Point and non-point microbial source pollution: A case study of Delhi. Phys. Chem. Earth Parts A/B/C 2011, 36, 490–499. [Google Scholar] [CrossRef]
- Katukiza, A.Y.; Ronteltap, M.; van der Steen, P.; Foppen, J.W.A.; Lens, P.N.L. Quantification of microbial risks to human health caused by waterborne viruses and bacteria in an urban slum. J. Appl. Microbiol. 2014, 116, 447–463. [Google Scholar] [CrossRef] [PubMed]
- Mara, D.; Evans, E. Sanitation & Water Supply in Low-Income Countries; Ventus Publishing; The University of Leeds: Leeds, UK, 2016; ISBN 8776818667. [Google Scholar]
- WHO. Water Supply, Sanitation and Hygiene Monitoring. Available online: http://www.who.int/water_sanitation_health/monitoring/coverage/en/ (accessed on 12 March 2018).
- WHO/UNICEF. Progress on Sanitation and Drinking Water: 2015 Update and MDG; World Health Organization: Geneva, Switzerland, 2015; p. 90. [Google Scholar]
- Anonymous. ONU-Habitat Madagascar: Profil Urbain d’Antananarivo; ONU-Habitat: Nairobi, Kenya, 2012; ISBN 9789211320237. [Google Scholar]
- Population of Cities in Madagascar 2018. Available online: http://worldpopulationreview.com/countries/madagascar-population/cities/ (accessed on 13 March 2018).
- Szabó, A.I.; Raveloson, A.; Székely, B. Landscape evolution and climate in Madagascar: Lavakization in the light of archive precipitation data. Cuad. Investig. Geogr. 2015, 41, 181. [Google Scholar] [CrossRef]
- Gade, D.W. Deforestation and its effects in highland Madagascar. Mt. Res. Dev. 1996, 16, 101. [Google Scholar] [CrossRef]
- Clark, M. Deforestation in Madagascar: Consequences of Population Growth and Unsustainable Agricultural Processes. Glob. Major. E-J. 2012, 3, 61–71. [Google Scholar]
- Heath, T.; Parker, A.; Weatherhead, E. How to Climate Proof Water and Sanitation Services in the Informal Areas and Peri-Urban Areas in Antananarivo; WSUP, Cranfield University: London, UK, 2010. [Google Scholar]
- WHO. Guidelines for Drinking-Water Quality, 4th ed. Incorporating the 1st Addendum. Available online: http://www.who.int/water_sanitation_health/publications/2011/9789241548151_toc.pdf (accessed on 15 June 2018).
- Bain, R.; Cronk, R.; Hossain, R.; Bonjour, S.; Onda, K.; Wright, J.; Yang, H.; Slaymaker, T.; Hunter, P.; Prüss-Ustün, A.; Bartram, J. Global assessment of exposure to faecal contamination through drinking water based on a systematic review. Trop. Med. Int. Heal. 2014, 19, 917–927. [Google Scholar] [CrossRef] [PubMed]
- Smedley, P. Groundwater Quality: Madagascar; British Geological Survey: Nottingham, UK, 2002; pp. 1–4. [Google Scholar]
- Les Données Climatiques Pour les Villes du Monde Entier—Climate-Data.org. Available online: https://fr.climate-data.org/ (accessed on 13 March 2018).
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 2007. [Google Scholar] [CrossRef]
- Institut National de la Statistique: Ministère de l’Économie et de l’Industrie INSTAT Madagascar. Available online: http://www.madacamp.com/INSTAT (accessed on 13 March 2018).
- Eau-Sites Eaux. Available online: http://www.jirama.mg/index.php?w=scripts&f=Jirama-page.php&act=siteeau (accessed on 13 March 2018).
- Strauss, M.E.; Smith, G.T. NIH Public Access Construct Validity: Advances in Theory and Methodology. Int. J. Forensic Mental Health 2009, 1–25. [Google Scholar] [CrossRef]
- Tian, H.; Lu, C.; Yang, J.; Banger, K.; Huntzinger, D.N.; Schwalm, C.R.; Michalak, A.M.; Cook, R.; Ciais, P.; Hayes, D.; et al. Global patterns and controls of soil organic carbon dynamics as simulated by multiple terrestrial biosphere models: Current status and future directions. Glob. Biogeochem. Cycles 2015, 29, 775–792. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.J.; Ellingsen, K.E.; McArdle, B.H. Multivariate dispersion as a measure of beta diversity. Ecol. Lett. 2006, 9, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Tao, F.; Du, J.; Shi, P.; Yu, D.; Meng, Y.; Sun, Y. Surface water quality and its control in a river with intensive human impacts-a case study of the Xiangjiang River, China. J. Environ. Manag. 2010, 91, 2483–2490. [Google Scholar] [CrossRef] [PubMed]
- Arnold, B.F.; Colford, J.M. Treating water with chlorine at point-of-use to improve water quality and reduce child diarrhea in developing countries: A systematic review and meta-analysis. Am. J. Trop. Med. Hyg. 2007, 76, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Bain, R.; Cronk, R.; Wright, J.; Yang, H.; Slaymaker, T.; Bartram, J. Fecal contamination of drinking-water in low- and middle-income countries: A systematic review and meta-analysis. PLoS Med. 2014, 11, e1001644. [Google Scholar] [CrossRef] [PubMed]
- Boehm, A.B.; Sassoubre, L.M. Enterococci as Indicators of Environmental Fecal Contamination. In Enterococci: From Commensals to Leading Causes of Drug Resistant Infection; Massachusetts Eye and Ear Infirmary: Boston, MA, USA, 2014; pp. 1–19. [Google Scholar]
- Lechevallier, M.W.; Cawthon, C.D.; Lee, R.G. Factors Promoting Survival of Bacteria in Chlorinated Water Supplies. Appl. Environ. Microbiol. 1988, 54, 649–654. [Google Scholar] [PubMed]
- Lechevallier, M.W.; Welch, N.J.; Smith, D.B.; Chevallier, M.W.L.E. Full-scale studies of factors related to coliform regrowth in drinking water. Appl. Environ. Microbiol. 1996, 62, 2201–2211. [Google Scholar] [PubMed]
- Stanfield, G.; Lechevallier, M.; Snozzi, M. Treatment Efficiency. In Assessing Microbial Safety of Drinking Water; IWA publishing: London, UK, 2003; pp. 159–178. [Google Scholar]
- Payment, P.; Franco, E. Clostridium perfringens and somatic coliphages as indicators of the efficiency of drinking water treatment for viruses and protozoan cysts. Appl. Environ. Microbiol. 1993, 59, 2418–2424. [Google Scholar] [PubMed]
- Brown, K.A.; Parks, K.E.; Bethell, C.A.; Johnson, S.E.; Mulligan, M. Predicting plant diversity patterns in madagascar: understanding the effects of climate and land cover change in a biodiversity hotspot. PLoS ONE 2015, 10, e0122721. [Google Scholar] [CrossRef] [PubMed]
- Payet, R.; Obura, D. The negative impacts of human activities in the eastern African region: an international waters perspective. Ambio 2004, 33, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.J.; Bootsma, M.J.; Morrison, H.G.; Sogin, M.L.; McLellan, S.L. A microbial signature approach to identify fecal pollution in the waters off an urbanized coast of lake michigan. Microb. Ecol. 2013, 65, 1011–1023. [Google Scholar] [CrossRef] [PubMed]
- Hynds, P.D.; Thomas, M.K.; Pintar, K.D.M. Contamination of groundwater systems in the US and Canada by enteric pathogens, 1990–2013: A review and pooled-analysis. PLoS ONE 2014, 9, e93301. [Google Scholar] [CrossRef] [PubMed]
- Christoffels, E. The importance of soil erosion for surface waters in the case of Rotbach Creek. In WIT Transactions on Ecology and the Environment; WIT Press: Ashurst, UK, 2016; Volume 209, pp. 1–14. [Google Scholar]
- Ishikawa, T.; Zhang, Y. Impact of fine sediment discharge from paddy fields on river water quality: A case study focusing on the rice farming calendar. In WIT Transactions on Ecology and the Environment; WIT Press: Ashurst, UK, 2016; pp. 15–26. [Google Scholar]
- Cox, R.; Bierman, P.; Jungers, M.C.; Rakotondrazafy, A.F.M. Erosion rates and sediment sources in Madagascar inferred from 10 Be analysis of lavaka, slope, and river sediment. J. Geol. 2009, 117, 363–376. [Google Scholar] [CrossRef]
- Farnleitner, A.H.; Ryzinska-Paier, G.; Reischer, G.H.; Burtscher, M.M.; Knetsch, S.; Kirschner, A.K.T.; Dirnböck, T.; Kuschnig, G.; Mach, R.L.; Sommer, R. Escherichia coli and enterococci are sensitive and reliable indicators for human, livestock and wildlife faecal pollution in alpine mountainous water resources. J. Appl. Microbiol. 2010, 109, 1599–1608. [Google Scholar] [CrossRef] [PubMed]
- Topalcengiz, Z.; Strawn, L.K.; Danyluk, M.D. Microbial quality of agricultural water in Central Florida. PLoS ONE 2017, 12, e0174889. [Google Scholar] [CrossRef] [PubMed]
- Ramos, M.C.; Quinton, J.N.; Tyrrel, S.F. Effects of cattle manure on erosion rates and runoff water pollution by faecal coliforms. J. Environ. Manag. 2006, 78, 97–101. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, M.C.; Juran, L.; Jose, J.; Srinivasan, S.; Ali, S.I.; Aronson, K.J.; Hall, K. The impact of rainfall and seasonal variability on the removal of bacteria by a point-of-use drinking water treatment intervention in Chennai, India. Int. J. Environ. Health Res. 2016, 26, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.; Zhang, Y.; Li, Z.; Li, P.; Xu, G. Influence of land use and land cover patterns on seasonal water quality at multi-spatial scales. Catena 2017, 151, 182–190. [Google Scholar] [CrossRef]
- Tornevi, A.; Bergstedt, O.; Forsberg, B. Precipitation effects on microbial pollution in a river: Lag structures and seasonal effect modification. PLoS ONE 2014, 9, e98546. [Google Scholar] [CrossRef] [PubMed]
- Gleason, J.A.; Fagliano, J.A. Effect of drinking water source on associations between gastrointestinal illness and heavy rainfall in New Jersey. PLoS ONE 2017, 12, e0173794. [Google Scholar] [CrossRef] [PubMed]

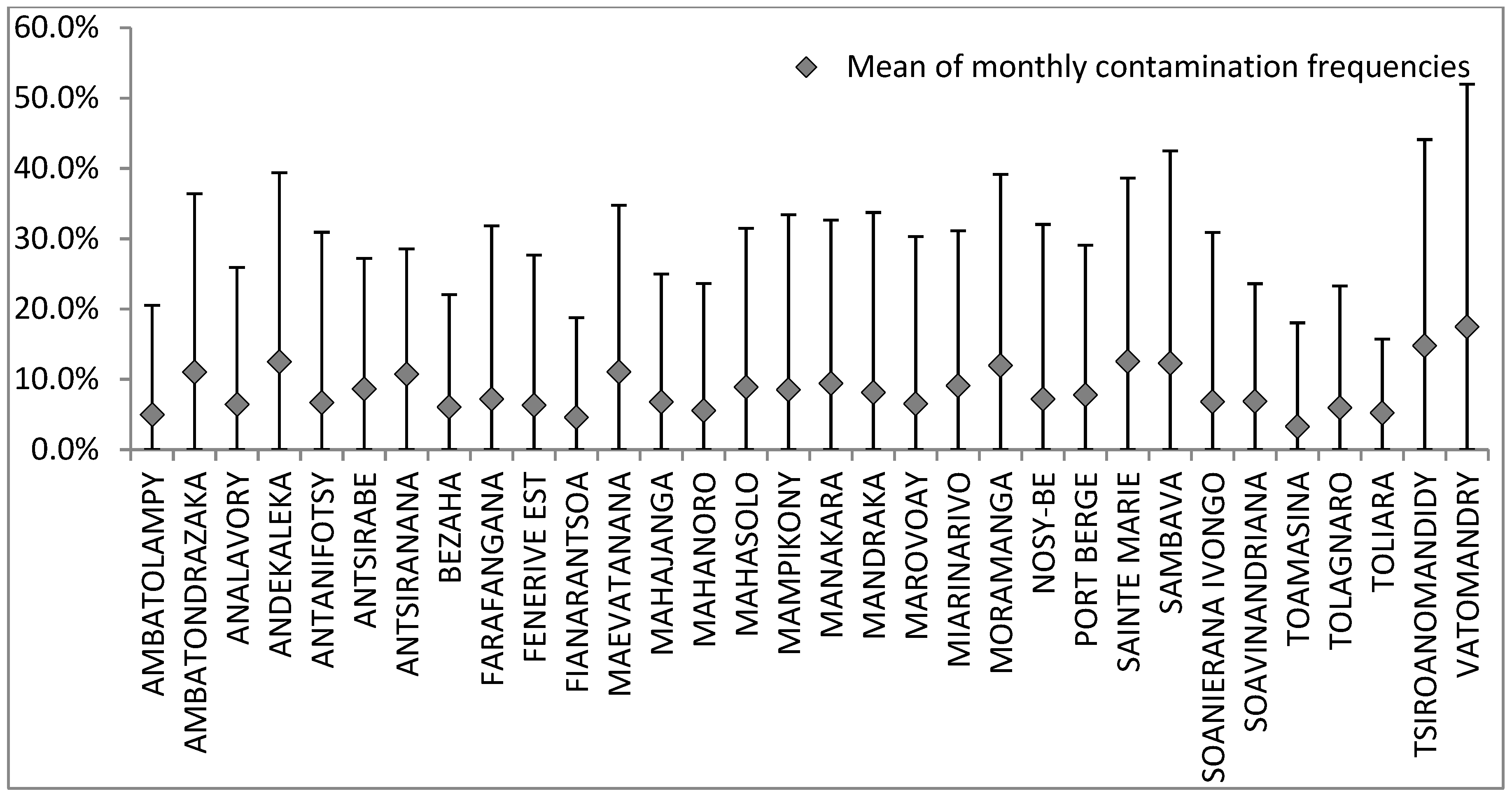
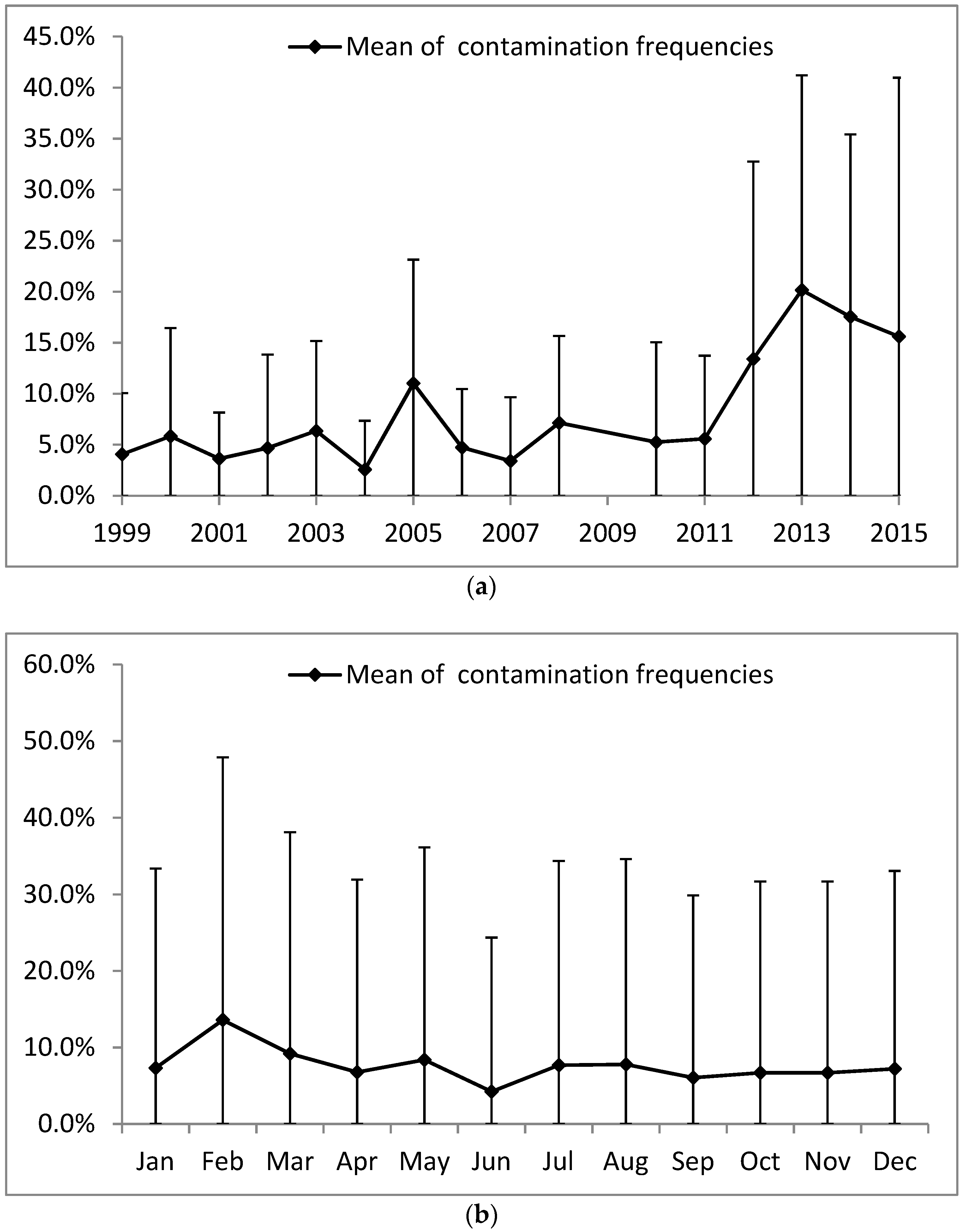

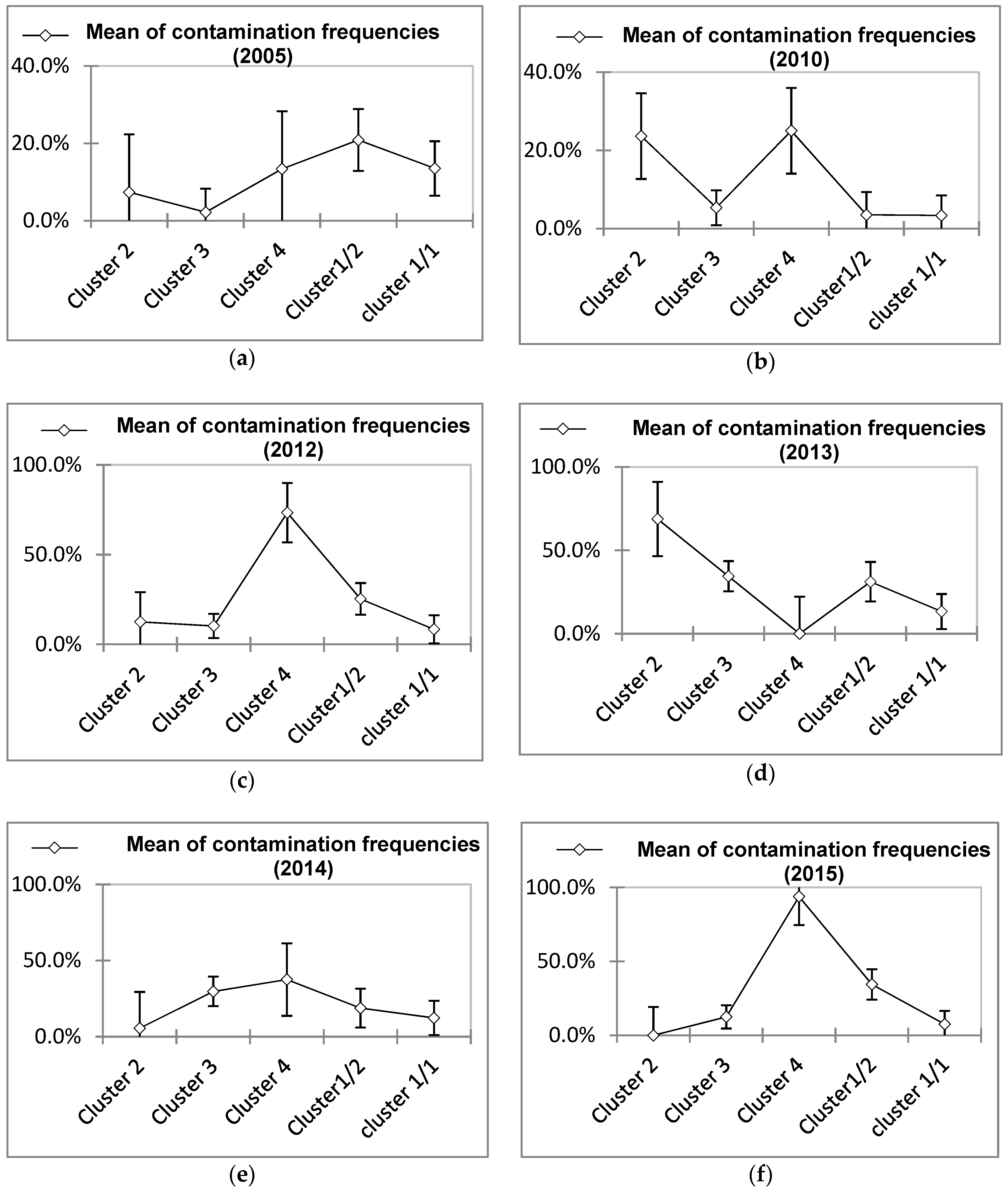

 ); mean of specific contamination frequencies (%) in spores of sulphite-reducing clostridia (SSRC), in intestinal enterococci (IE), in total coliforms (TC), and in Escherichia coli (EC); and means of overall contamination frequencies (line
); mean of specific contamination frequencies (%) in spores of sulphite-reducing clostridia (SSRC), in intestinal enterococci (IE), in total coliforms (TC), and in Escherichia coli (EC); and means of overall contamination frequencies (line  ). For the legend of each cluster see Figure 4: (a) Cluster 1/1; (b) Cluster 1/2; (c) Cluster 2; (d) Cluster 3; (e) Cluster 4.
). For the legend of each cluster see Figure 4: (a) Cluster 1/1; (b) Cluster 1/2; (c) Cluster 2; (d) Cluster 3; (e) Cluster 4.
 ); mean of specific contamination frequencies (%) in spores of sulphite-reducing clostridia (SSRC), in intestinal enterococci (IE), in total coliforms (TC), and in Escherichia coli (EC); and means of overall contamination frequencies (line
); mean of specific contamination frequencies (%) in spores of sulphite-reducing clostridia (SSRC), in intestinal enterococci (IE), in total coliforms (TC), and in Escherichia coli (EC); and means of overall contamination frequencies (line  ). For the legend of each cluster see Figure 4: (a) Cluster 1/1; (b) Cluster 1/2; (c) Cluster 2; (d) Cluster 3; (e) Cluster 4.
). For the legend of each cluster see Figure 4: (a) Cluster 1/1; (b) Cluster 1/2; (c) Cluster 2; (d) Cluster 3; (e) Cluster 4.

 active variable;
active variable;  supplementary data;
supplementary data;  observations). Four groups (circles) can be discriminated according to the type of water source, the treatment implemented, and the type of threats on the water source.
observations). Four groups (circles) can be discriminated according to the type of water source, the treatment implemented, and the type of threats on the water source.
 active variable;
active variable;  supplementary data;
supplementary data;  observations). Four groups (circles) can be discriminated according to the type of water source, the treatment implemented, and the type of threats on the water source.
observations). Four groups (circles) can be discriminated according to the type of water source, the treatment implemented, and the type of threats on the water source.

| Conventional Treatment | Unconventional Treatment | |||||
|---|---|---|---|---|---|---|
| Classic | Classic + Neutralization | Chlorination | Filtration + Chlorination | N of Stations | Total (%) | |
| Surface Water | ||||||
| Lakes | 1113 | 1419 | 0 | 0 | 6 | 2532 (20%) |
| Rivers | 3487 | 1436 | 0 | 0 | 11 | 4923 (39%) |
| Total | 4600 | 2855 | 0 | 0 | 7455 2 (60%) | |
| Groundwater | ||||||
| Boreholes | 0 | 1335 | 1458 | 0 | 5 | 2793 (22%) |
| Wells | 144 | 1014 | 0 | 0 | 6 | 1158 (9%) |
| Water springs | 0 | 533 | 248 | 308 | 4 | 1089 (9%) |
| Total | 144 | 2882 | 1706 | 308 | 5040 (40%) | |
| Total number of stations | 11 | 16 3 | 4 | 1 | 32 | |
| Total (%) | 4744 (38%) | 5737 (46%) | 1706 (14%) | 308 (2%) | 12,495 1 (100%) | |
| Division | Sites | FIB 1 | IE 2 | EC 3 | TC 4 | SSRC 5 |
|---|---|---|---|---|---|---|
| Toamasina | Vatomandry Bcn | 0.290 | 0.290 | 0.144 | 0.357 | 0.876 |
| Sainte Marie Rc | 0.844 | 0.816 | 0.442 | 0.526 | 0.394 | |
| Soaniera Ivongo Rc | 0.744 | 0.744 | 0.000 | 0.641 | 0.259 | |
| Fenerive Est Wcn | 0.620 | 0.620 | 0.000 | 0.436 | 0.671 | |
| Mahanoro Wc | 0.489 | 0.489 | 0.000 | 0.345 | 0.860 | |
| Toamasina Rc | 0.535 | 0.535 | 0.308 | 0.578 | 0.656 | |
| Antananarivo | Tsiroanomandidy Rcn | 0.411 | 0.411 | 0.316 | 0.489 | 0.785 |
| Andekaleka Rc | 0.723 | 0.575 | 0.475 | 0.387 | 0.552 | |
| Moramanga Rc | 0.553 | 0.419 | 0.387 | 0.576 | 0.724 | |
| Ambatondrazaka Su | 0.642 | 0.596 | 0.326 | 0.520 | 0.768 | |
| Miarinarivo Scn | 0.717 | 0.662 | 0.326 | 0.570 | 0.536 | |
| Mahasolo Wcn | 0.734 | 0.541 | 0.541 | 0.817 | 0.483 | |
| Mandraka Lc | 0.596 | 0.596 | 0.000 | 0.341 | 0.778 | |
| Soavandriana Scn | 0.628 | 0.628 | 0.235 | 0.756 | 0.628 | |
| Analavory Su | 0.781 | 0.695 | 0.488 | 0.649 | 0.600 | |
| Ambatolampy Lcn | 0.588 | 0.454 | 0.370 | 0.588 | 0.698 | |
| Antsirabe | Antsirabe Lcn | 0.677 | 0.629 | 0.359 | 0.619 | 0.609 |
| Antanifotsy Lc | 0.727 | 0.727 | 0.414 | 0.590 | 0.661 | |
| Fianarantsoa | Manakara Rc | 0.674 | 0.448 | 0.492 | 0.674 | 0.607 |
| Faranfangana Rcn | 0.694 | 0.694 | 0.342 | 0.647 | 0.420 | |
| Fianarantsoa Rc | 0.660 | 0.537 | 0.378 | 0.574 | 0.706 | |
| Toliara | Bezaha Bu | 0.859 | 0.754 | 0.398 | 0.398 | 0.634 |
| Tolagnaro Lc | 0.220 | 0.220 | 0.000 | 0.382 | 0.915 | |
| Toliara Bu | 0.598 | 0.534 | 0.421 | 0.583 | 0.671 | |
| Mahajanga | Maevatanana Wcn | 0.426 | 0.000 | 0.426 | 0.543 | 0.884 |
| Mampikony Wcn | 0.431 | 0.431 | 0.000 | 0.500 | 0.804 | |
| Port Berge Rcn | 0.751 | 0.562 | 0.562 | 0.751 | 0.630 | |
| Mahajanga Bcn | 0.772 | 0.764 | 0.336 | 0.587 | 0.598 | |
| Marovoay Bu | 0.848 | 0.719 | 0.504 | 0.671 | 0.504 | |
| Antsiranana | Sambava Bu | 0.476 | 0.386 | 0.386 | 0.515 | 0.798 |
| Antsiranana Rcn | 0.427 | 0.338 | 0.275 | 0.492 | 0.832 | |
| Nosy Be Lcn | 0.738 | 0.598 | 0.545 | 0.647 | 0.694 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bastaraud, A.; Rakotondramanga, J.M.; Mahazosaotra, J.; Ravaonindrina, N.; Jambou, R. Environmental Factors and the Microbial Quality of Urban Drinking Water in a Low-Income Country: The Case of Madagascar. Water 2018, 10, 1450. https://doi.org/10.3390/w10101450
Bastaraud A, Rakotondramanga JM, Mahazosaotra J, Ravaonindrina N, Jambou R. Environmental Factors and the Microbial Quality of Urban Drinking Water in a Low-Income Country: The Case of Madagascar. Water. 2018; 10(10):1450. https://doi.org/10.3390/w10101450
Chicago/Turabian StyleBastaraud, Alexandra, Jean Marius Rakotondramanga, Jackson Mahazosaotra, Noror Ravaonindrina, and Ronan Jambou. 2018. "Environmental Factors and the Microbial Quality of Urban Drinking Water in a Low-Income Country: The Case of Madagascar" Water 10, no. 10: 1450. https://doi.org/10.3390/w10101450
APA StyleBastaraud, A., Rakotondramanga, J. M., Mahazosaotra, J., Ravaonindrina, N., & Jambou, R. (2018). Environmental Factors and the Microbial Quality of Urban Drinking Water in a Low-Income Country: The Case of Madagascar. Water, 10(10), 1450. https://doi.org/10.3390/w10101450






