Chemical and Light Extinction Characteristics of Atmospheric Aerosols in Suburban Nanjing, China
Abstract
:1. Introduction
2. Methodology
2.1. Aerosol Sampling
2.2. Chemical Analysis
2.3. Data Analysis
2.3.1. IMPROVE Formula
2.3.2. Koschmieder’s Formula
2.3.3. Backward Trajectory and Clustering Analysis
3. Results and Discussion
3.1. Chemical Characteristics of Atmospheric Aerosol
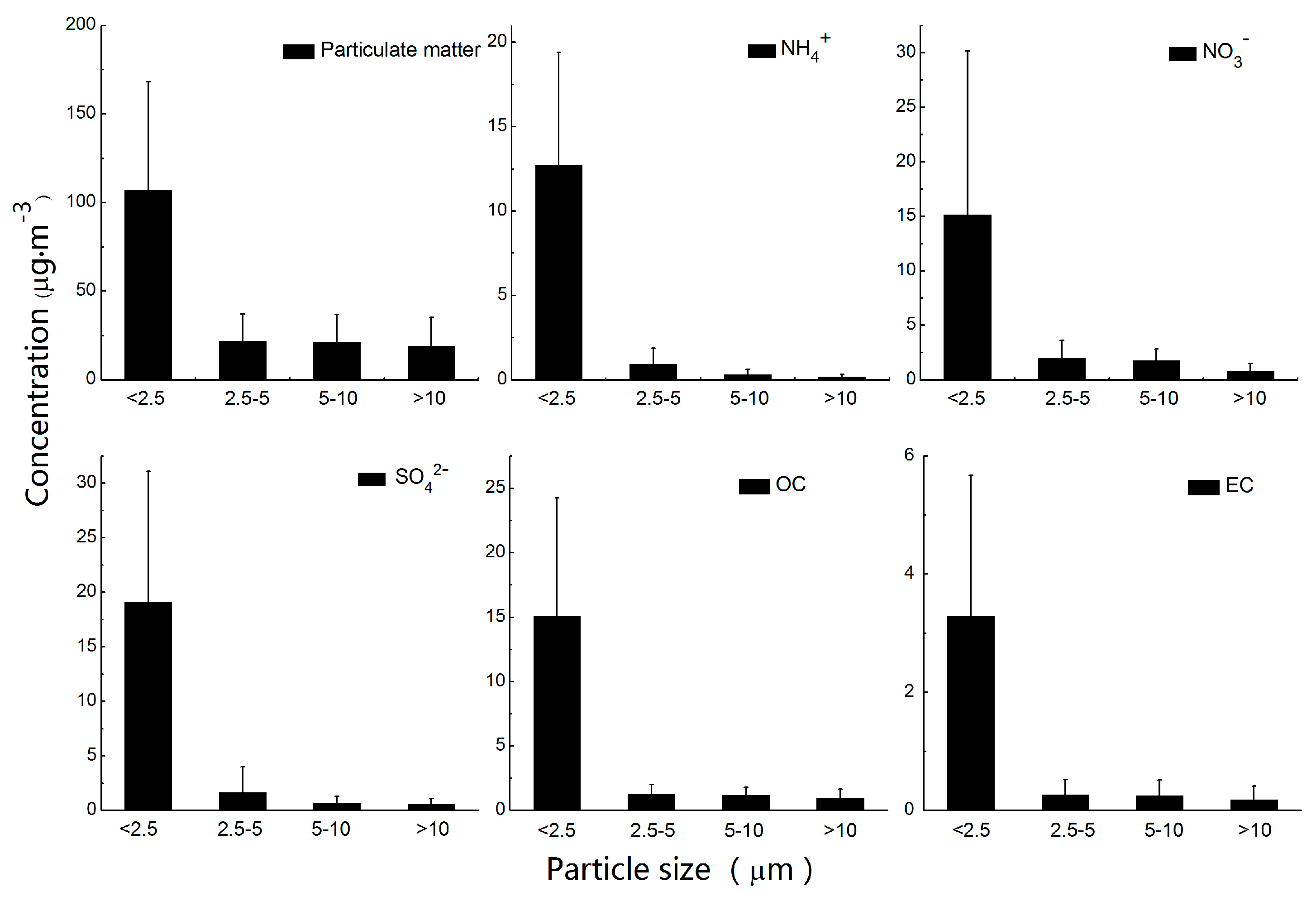
3.1.1. Size-Segregated Mass Concentrations
3.1.2. Seasonal Differences
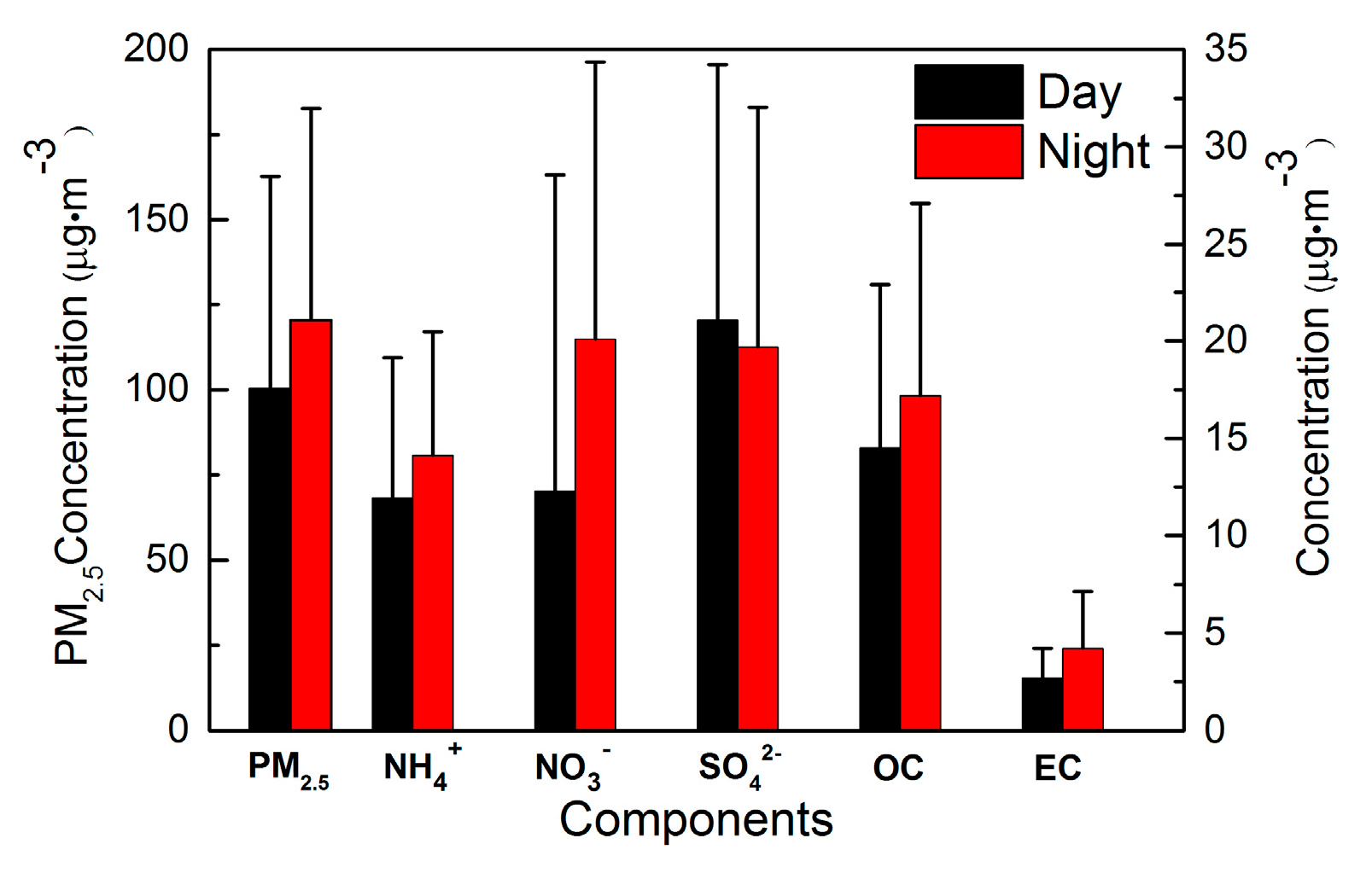
3.1.3. Day–Night Differences
3.2. Light Extinction Estimated by the IMPROVE Formula
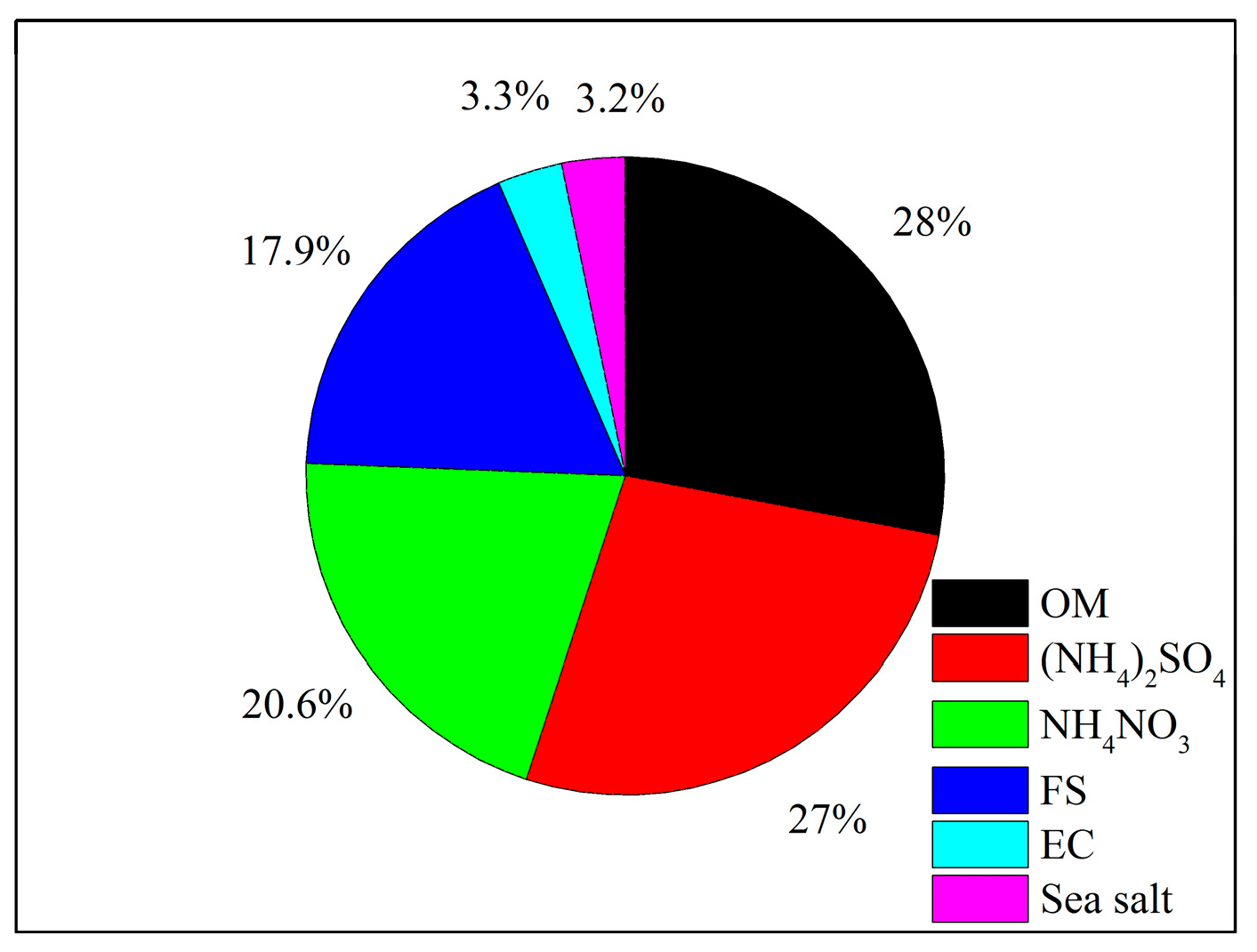
3.2.1. Reconstruction of PM2.5 Mass
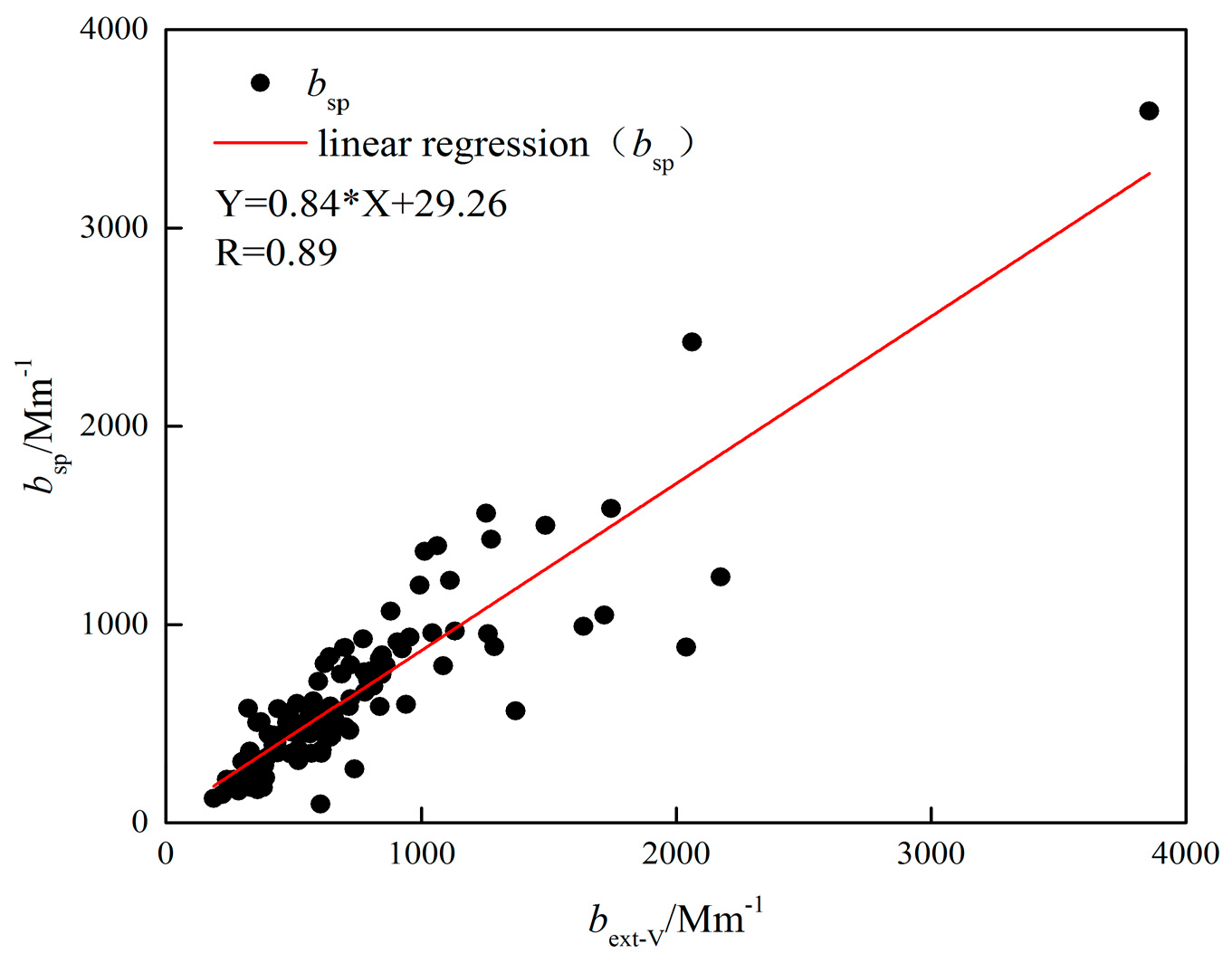
3.2.2. Estimation of Extinction Coefficients
3.3. Variations of Chemical and Light Extinction Characteristics under Different Circumstances
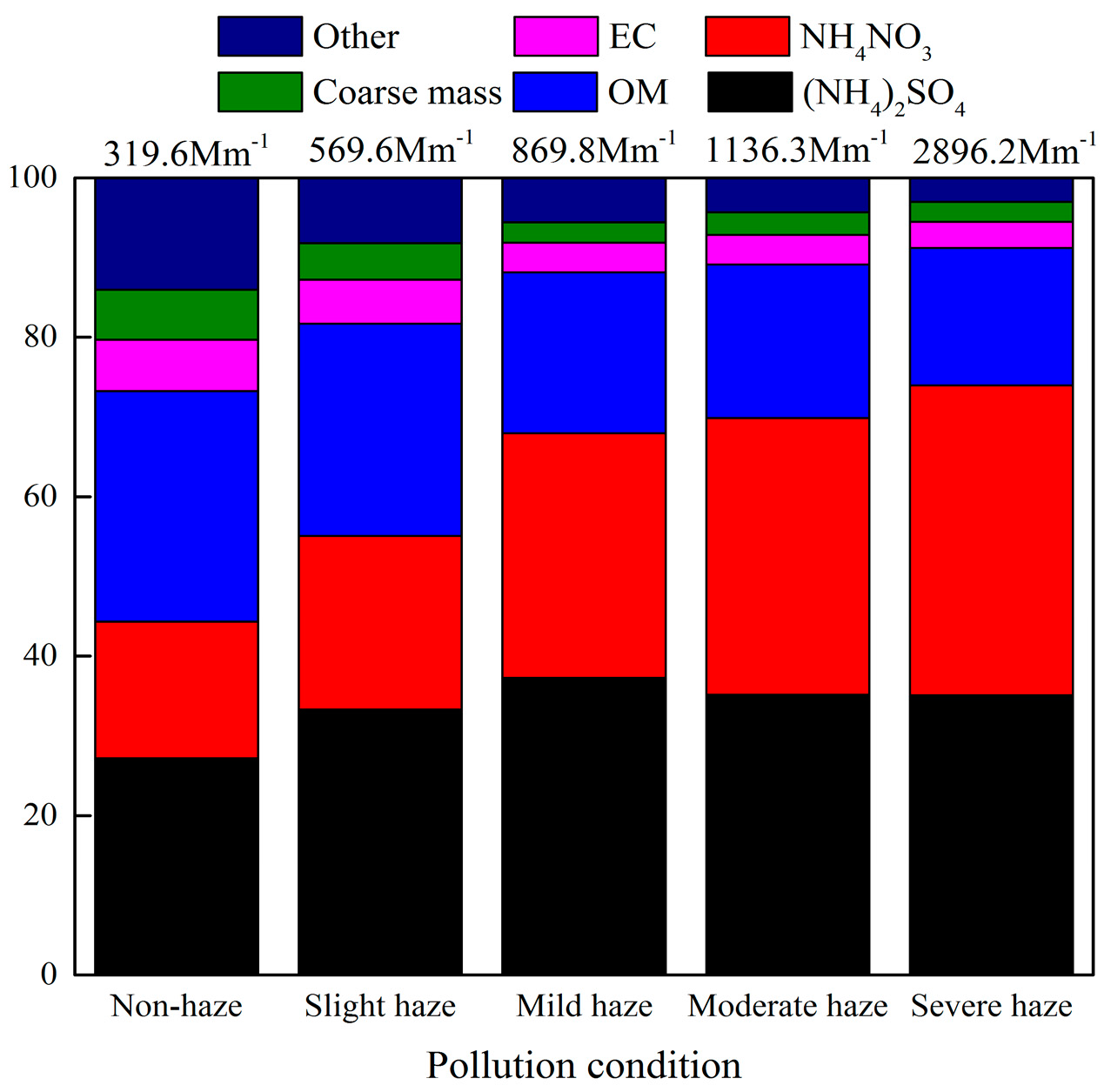
3.3.1. Influences of Different Pollution Levels
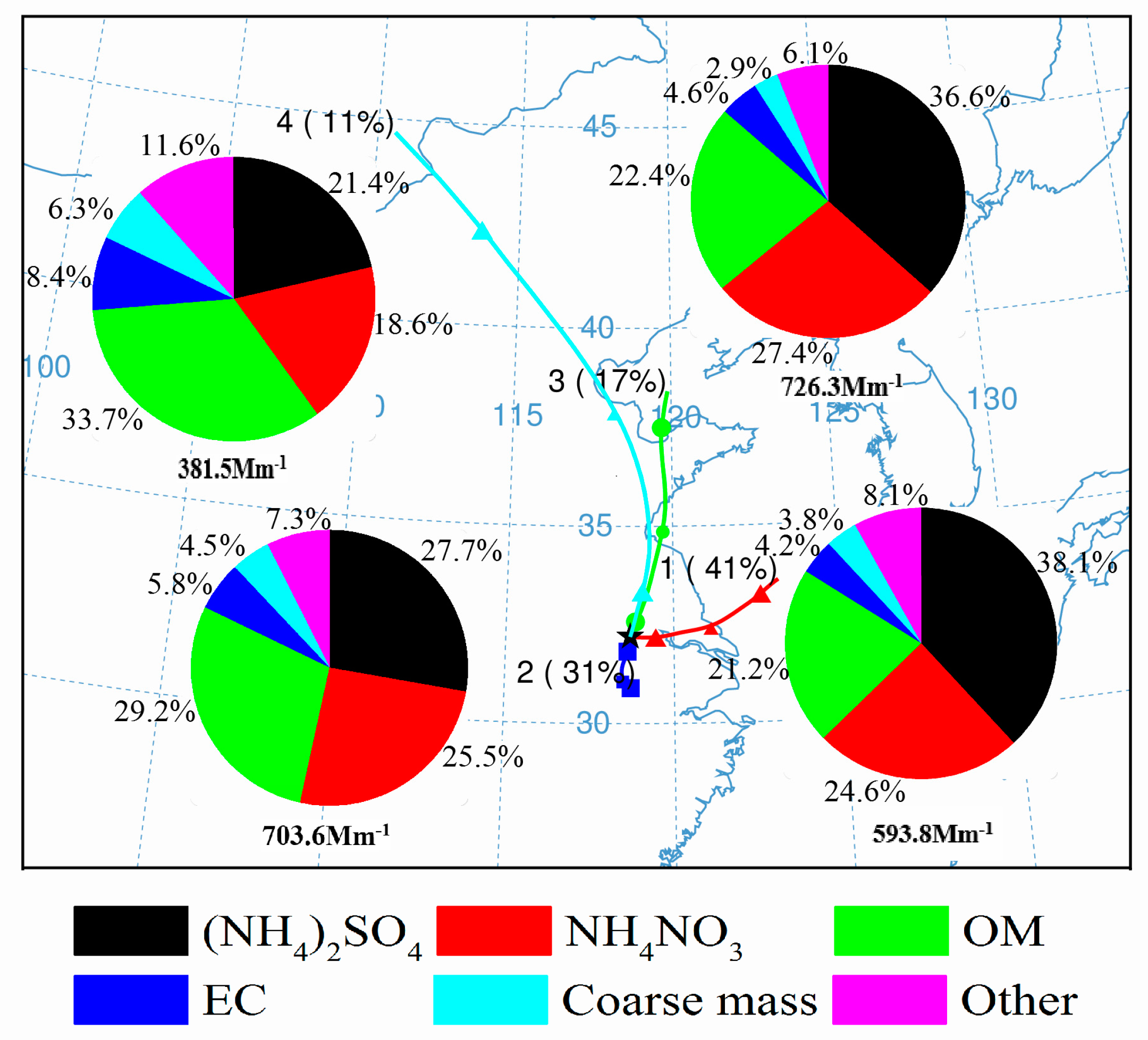
3.3.2. Influences of Different Air Masses
4. Conclusions
- PM2.5 dominated the aerosol pollution, with its highest mass concentration in winter, followed by summer, spring, and autumn. Concentrations of most chemical components were highest in winter too, except that the highest SO42− concentration occurred in summer, K+ and Mg2+ peaked in spring while Ca2+ reached its maximum in autumn. Mass concentrations of PM2.5 and most major species were higher during nighttime than daytime due to unfavorable conditions for pollutant diffusion. For NO3−, its loading was much lower during daytime, owing to its semi-volatile behavior and/or possible nocturnal heterogeneous production. However, SO42− concentration was higher during daytime, indicating the significant photochemical production.
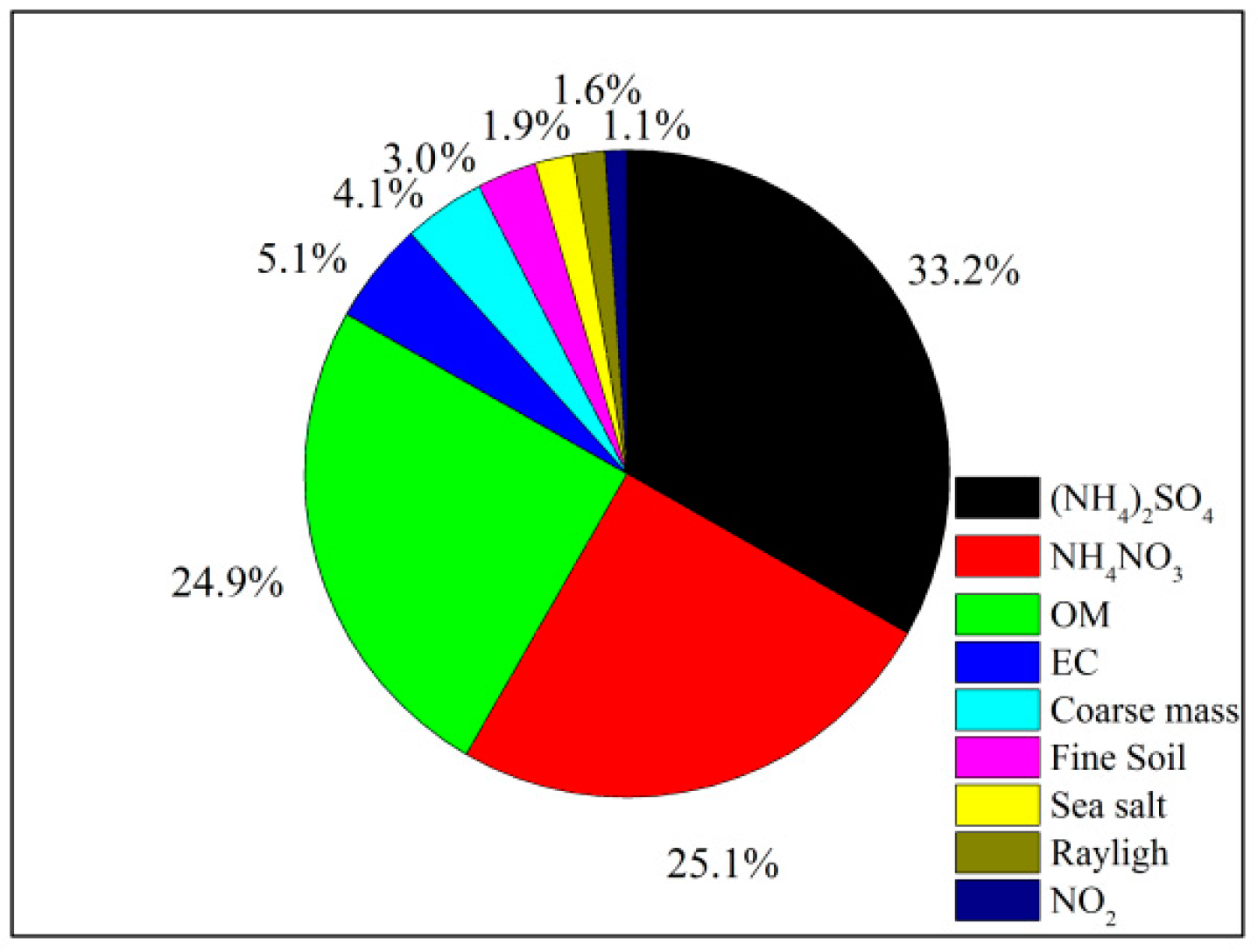
- The measured species can reconstruct the total PM2.5 mass well, and OM, (NH4)2SO4, and NH4NO3 comprised the majority of PM2.5 (75.6%) with another significant component of FS contributing ~18%, and two minor components of SS and EC both occupying ~3%. The IMPROVE formula could estimate the aerosol light extinction reasonably well, with (NH4)2SO4, NH4NO3, and OM contributing 33.2%, 25.1%, and 24.9%, respectively. The light extinction was dominated by the SNA, indicating a significant role of secondary ions in visibility degradation.
- Mass concentrations of PM2.5 and all species increased gradually with the increase of haze pollution levels. The increase of SNA was particularly remarkable, and NO3− appeared to arise most rapidly among all species. Regarding the light extinction, the contribution from OM continuously decreased, while SNA, in particular nitrate contribution, increased with the increase of pollution levels, indicating a significant role of secondary species in haze formation and also underscoring the priority of reduction of vehicular NO2 emissions in Nanjing.
- Four clusters of air masses were identified, with differing chemical and light extinction characteristics. The local air mass and air mass originated from Bohai, but travelled through Shandong Province and north of Jiangsu Province appeared to be heavily polluted. The local air mass had high mass loadings of OC and EC and, correspondingly with OM, contributing most to the light extinction. The air mass from Bohai was contributed mainly by the SNA in terms of both mass concentration and light extinction. The air parcel from Huanghai was relatively clean, but with significant light extinction contributed by SO42−. The air mass starting from the northwest was the cleanest and clearest, and carbonaceous aerosols seemed to contribute most to its light extinction.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, Y.L.; Cao, F. Fine particulate matter (PM2.5) in China at a city level. Sci. Rep. 2015, 5, 14884. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.J.; Zhang, Y.; Bozzetti, C.; Ho, K.F.; Cao, J.; Han, Y.M.; Daellenbach, K.; Slowik, J.; Platt, S.; Canonaco, F.; et al. High secondary aerosol contribution to particulate pollution during haze events in China. Nature 2014, 514, 218–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, M.; Guttikunda, S.K.; Carmichael, G.R.; Wang, Y.; Liu, Z.; Stanier, C.O.; Saide, P.E.; Yu, M. Health impacts and economic losses assessment of the 2013 severe haze event in Beijing area. Sci. Total Environ. 2015, 511, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y. Aerosol over China and their climate effect. Adv. Earth Sci. 2007, 22, 12–16. (In Chinese) [Google Scholar]
- Cheng, M.C.; You, C.F.; Cao, J.; Jin, Z.D. Spatial and seasonal variability of water-soluble ions in PM2.5, aerosols in 14 major cities in China. Atmos. Environ. 2012, 60, 182–192. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Wang, Y.Q.; Niu, T.J.; Zhang, X.C.; Gong, S.L.; Zhang, Y.M.; Sun, J.Y. Atmospheric aerosol compositions in China: Spatial/temporal variability, chemical signature, regional haze distribution and comparisons with global aerosols. Atmos. Chem. Phys. 2012, 12, 771–799. [Google Scholar] [CrossRef] [Green Version]
- Zheng, J.; Hu, M.; Peng, J.F.; Wu, Z.J.; Kumar, P.; Li, M.R.; Wang, Y.J.; Guo, S. Spatial distributions and chemical properties of PM2.5 based on 21 field campaigns at 17 sites in China. Chemosphere 2016, 159, 480–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, F.; Tan, J.; Zhao, Q.; Du, Z.; He, K.; Ma, Y.; Duan, F.; Chen, G.; Zhao, Q. Characteristics of PM2.5 speciation in representative megacities and across China. Atmos. Chem. Phys. 2011, 11, 5207–5219. [Google Scholar] [CrossRef]
- Sisler, J.F.; Malm, W.C. Interpretation of trends of PM2.5 and reconstructed visibility from the IMPROVE network. J. Air Waste Manag. 2000, 50, 775–789. [Google Scholar] [CrossRef] [PubMed]
- Pitchford, M.; Malm, W.; Schichtel, B.; Kumar, N.; Lowenthal, D.; Hand, J. Revised algorithm for estimating light extinction from IMPROVE particle speciation data. J. Air Waste Manag. 2007, 57, 1326–1336. [Google Scholar]
- Li, X.H.; He, K.B.; Li, C.C.; Yang, F.M.; Zhao, Q.; Ma, Y.L.; Chen, Y.; Ouyang, W.J.; Chen, G.C. PM2.5 mass, chemical composition, and light extinction before and during the 2008 Beijing Olympics. J. Geophys. Res. Atmos. 2013, 118, 12158–12167. [Google Scholar] [CrossRef]
- Wang, H.; Shi, G.; Tian, M.; Zhang, L.; Chen, Y.; Yang, F.M.; Gao, X.Y. Aerosol optical properties and chemical composition apportionment in Sichuan Basin, China. Sci. Total Environ. 2017, 577, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.F.; Huang, K.; Zhuang, G.S.; Fu, J.S.; Wang, Q.Z.; Liu, T.N.; Deng, C.R.; Fu, Q.Y. A multi-year evolution of aerosol chemistry impacting visibility and haze formation over an Eastern Asia megacity, Shanghai. Atmos. Environ. 2014, 92, 76–86. [Google Scholar] [CrossRef]
- Deng, J.J.; Zhang, Y.R.; Hong, Y.W.; Xu, L.L.; Chen, Y.T.; Du, W.J.; Chen, J.S. Optical properties of PM2.5 and the impacts of chemical compositions in the coastal city Xiamen in China. Sci. Total Environ. 2016, 557–558, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Zhang, L.M.; Cao, J.J.; Hsu, S.; Xia, X.J.; Zhang, Z.S.; Lin, Z.J.; Chen, T.T.; Zhang, R.J. Characterization and source apportionment of aerosol light extinction in Chengdu, southwest China. Atmos. Environ. 2014, 95, 552–562. [Google Scholar] [CrossRef]
- Cao, J.J.; Wang, Q.Y.; Chow, J.C.; Waston, J.G.; Tie, X.X.; Shen, Z.X.; Wang, P.; An, Z.S. Impacts of aerosol compositions on visibility impairment in Xi’an, China. Atmos. Environ. 2012, 59, 559–566. [Google Scholar] [CrossRef]
- Tao, J.; Zhang, L.M.; Gao, J.; Wang, H.; Chai, F.H.; Wang, S.L. Aerosol chemical composition and light scattering during a winter season in Beijing. Atmos. Environ. 2015, 110, 36–44. [Google Scholar] [CrossRef]
- Tian, M.; Wang, H.B.; Chen, Y.; Yang, F.M.; Zhang, X.H.; Zou, Q.; Zhang, R.Q.; Ma, Y.L.; He, K.B. Characteristics of aerosol pollution during heavy haze events in Suzhou, China. Atmos. Chem. Phys. 2016, 16, 7357–7371. [Google Scholar] [CrossRef]
- Tao, J.; Zhang, L.M.; Ho, K.F.; Zhang, R.J.; Lin, Z.J.; Zhang, Z.S.; Lin, M.; Cao, J.J.; Liu, S.X.; Wang, G.H. Impact of PM2.5 chemical compositions on aerosol light scattering in Guangzhou—The largest megacity in South China. Atmos. Res. 2014, 135–136, 48–58. [Google Scholar] [CrossRef]
- Li, B.; Zhang, J.; Zhao, Y.; Yuan, S.; Zhao, Q.; Shen, G.; Wu, H. Seasonal variation of urban carbonaceous aerosols in a typical city Nanjing in Yangtze River Delta, China. Atmos. Environ. 2015, 106, 223–231. [Google Scholar] [CrossRef]
- Wu, D.; Cao, S.; Tang, L.L.; Xia, J.R.; Lu, J.G.; Liu, G.; Yang, M.; Li, F.Y.; Ge, X.L. Variation of size distribution and the influencing factors of aerosol in Northern Suburbs of Nanjing. Environ. Sci. 2016, 37, 3268–3279. (In Chinese) [Google Scholar]
- Zhuang, B.L.; Wang, T.J.; Liu, J.; Li, S.; Xie, M.; Han, Y.; Cheng, P.L.; Hu, Q.; Yang, X.Q.; Fu, C.B.; et al. The surface aerosol optical properties in the urban area of Nanjing, west Yangtze River Delta, China. Atmos. Chem. Phys. 2017, 17, 1143–1160. [Google Scholar] [CrossRef]
- Wang, H.L.; An, J.L.; Cheng, M.T.; Shen, L.J.; Zhu, B.; Li, Y.; Wang, Y.S.; Duan, Q.; Sullivan, A.; Xia, L. One year online measurements of water-soluble ions at the industrially polluted town of Nanjing, China: Sources, seasonal and diurnal variations. Chemosphere 2016, 148, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.L.; An, J.L.; Zhu, B.; Shen, L.J.; Duan, Q.; Shi, Y.Z. Characteristics of carbonaceous aerosol in a typical industrial city—Nanjing in Yangtze River Delta, China: Size distributions, seasonal variations, and sources. Atmosphere 2017, 8, 73. [Google Scholar] [CrossRef]
- Li, H.M.; Wang, J.H.; Wang, Q.; Qian, X.; Qian, Y.; Yang, M.; Li, F.; Lu, H.; Wang, C. Chemical fractionation of arsenic and heavy metals in fine particle matter and its implications for risk assessment: A case study in Nanjing, China. Atmos. Environ. 2015, 103, 339–346. [Google Scholar] [CrossRef]
- Ge, X.; He, Y.; Sun, Y.; Xu, J.Z.; Wang, J.; Shen, Y.; Chen, M.D. Characteristics and formation mechanisms of fine particulate nitrate in typical urban areas in China. Atmosphere 2017, 8, 62. [Google Scholar] [CrossRef]
- Ming, L.; Jin, L.; Li, J.; Fu, P.; Yang, W.; Liu, D.; Zhang, G.; Wang, Z.; Li, X. PM2.5 in the Yangtze River Delta, China: Chemical compositions, seasonal variations, and regional pollution events. Environ. Pollut. 2017, 223, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Xue, M.; Yuan, S.; Zhang, J.; Zhao, Q.; Li, B.; Wu, H.; Ding, A. Chemical compositions and reconstructed light extinction coefficients of particulate matter in a mega-city in the western Yangtze River Delta, China. Atmos. Environ. 2014, 83, 14–20. [Google Scholar] [CrossRef]
- Yu, X.; Ma, J.; An, J.; Yuan, L.; Zhu, B.; Liu, D.; Wang, J.; Yang, Y.; Cui, H. Impacts of meteorological condition and aerosol chemical compositions on visibility impairment in Nanjing, China. J. Clean. Prod. 2016, 131, 112–120. [Google Scholar] [CrossRef]
- Hua, Y.; Cheng, Z.; Wang, S.; Jiang, J.; Chen, D.; Cai, Y.; Fu, X.; Fu, Q.Y.; Chen, C.; Xu, B.; et al. Characteristics and source apportionment of PM2.5 during a fall heavy haze episode in the Yangtze River Delta of China. Atmos. Environ. 2015, 123, 380–391. [Google Scholar] [CrossRef]
- Wei, L.F.; Yang, F.M.; Tan, J.H.; Ma, Y.L.; He, K.B. Research progress on aerosol extinction properties. Environ. Chem. 2014, 33, 705–715. (In Chinese) [Google Scholar]
- Chen, Z. Relationship between Haze Pollution and Aerosol Properties in the Yangtze River Delta of China. Ph.D. Dissertation, Tsinghua University, Beijing, China, November 2013. (In Chinese). [Google Scholar]
- Wang, Q. Study of Air Pollution Transportation Source in Shanghai Using Trajectory Model. Res. Environ. Sci. 2013, 26, 357–363. (In Chinese) [Google Scholar]
- Chen, W.H.; Wang, X.M.; Cohen, J.B.; Zhou, S.Z.S.; Chang, M.; Chan, C.Y. Properties of aerosols and formation mechanisms over southern China during the monsoon season. Atmos. Chem. Phys. 2016, 16, 13271–13289. [Google Scholar] [CrossRef]
- Li, L.; An, J.Y.; Zhou, M.; Yan, R.S.; Huang, C.; Lu, Q.; Lin, L.; Wang, Y.J.; Tao, S.K.; Qiao, L.P.; et al. Source apportionment of fine particles and its chemical components over the Yangtze River Delta, China during a heavy haze pollution episode. Atmos. Environ. 2015, 123, 415–429. [Google Scholar] [CrossRef]
- Zhao, J.P.; Zhang, F.W.; Xu, Y.; Chen, J.S. Characterization of water-soluble inorganic ions in size-segregated aerosols in coastal city, Xiamen. Atmos. Res. 2011, 99, 546–562. [Google Scholar] [CrossRef]
- Sun, Y.; Zhuang, G.; Tang, A.A.; Wang, Y.; An, Z. Chemical characteristics of PM2.5 and PM10 in haze−fog episodes in Beijing. Environ. Sci. Technol. 2006, 15, 3148–3155. [Google Scholar] [CrossRef]
- Huang, Y.M.; Liu, Z.R.; Chen, H.; Wang, Y.S. Characteristics of mass size distributions of Water-Soluble inorganic ions during summer and winter haze days of Beijing. Environ. Sci. 2013, 34, 1236–1244. (In Chinese) [Google Scholar]
- Waheed, A.; Lin, X.L.; Tan, M.G.; Bao, L.M.; Liu, J.F.; Zhang, Y.X.; Zhang, G.L.; Li, Y. Size Distribution and Sources of trace metals in ultrafine/fine/coarse airborne particles in the atmosphere of Shanghai. Aerosol Sci. Technol. 2011, 45, 163–171. [Google Scholar] [CrossRef]
- Luo, X.S.; Yu, S.; Li, X.D. Distribution, availability, and sources of trace metals in different particle size fractions of urban soils in Hong Kong: Implications for assessing the risk to human health. Environ. Pollut. 2011, 159, 1317–1326. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.L.; Zhu, B.; Shen, L.J.; Xu, H.H.; An, J.L.; Xue, G.Q.; Cao, L.F. Water-soluble ions in atmospheric aerosols measured in five sites in the Yangtze River Delta, China: Size-fractionated, seasonal variations and sources. Atmos. Environ. 2015, 123, 370–379. [Google Scholar] [CrossRef]
- Huang, X.J.; Liu, Z.R.; Zhang, J.K.; Wen, T.X.; Ji, D.S.; Wang, Y.S. Seasonal variations and size distributions of water-soluble ions in atmospheric aerosols in Beijing. Atmos. Res. 2016, 168, 70–79. [Google Scholar] [CrossRef]
- Liu, G.; Li, J.; Wu, D.; Xu, H. Chemical composition and source apportionment of the ambient PM2.5 in Hangzhou, China. Particuology 2015, 18, 135–143. [Google Scholar] [CrossRef]
- Zhang, T.; Cao, J.J.; Tie, X.X.; Shen, Z.X.; Liu, S.X.; Ding, H.; Han, Y.M.; Wang, G.H.; Ho, K.F.; Qiang, J.; et al. Water-soluble ions in atmospheric aerosols measured in Xi’an, China seasonal variations and sources. Atmos. Res. 2011, 102, 110–119. [Google Scholar] [CrossRef]
- Ge, X.L.; Li, L.; Chen, Y.F.; Chen, H.; Wu, D.; Wang, J.F.; Xie, X.C.; Ge, S.; Ye, Z.L.; Xu, J.Z.; et al. Aerosol characteristics and sources in Yangzhou, China resolved by offline aerosol mass spectrometry and other techniques. Environ. Pollut. 2017, 225, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Niu, H.Y.; Zhao, X.; Dai, Z.X.; Wang, G.H.; Wang, L.S. Characterization, source apportionment of particulate matter and n-Alk in atmospheric aerosols in Nanjing City. Environ. Pollut. Control 2005, 27, 363–366. (In Chinese) [Google Scholar]
- Liu, J.; Zhu, L.; Wang, H.; Yang, Y.; Liu, J.T.; Qiu, D.D.; Ma, W.; Zhang, Z.M.; Liu, J.L. Dry deposition of particulate matter at an urban forest, wetland and lake surface in Beijing. Atmos. Environ. 2016, 125, 178–187. [Google Scholar] [CrossRef]
- Wang, D.F.; Zhou, B.; Fu, Q.Y.; Zhao, Q.B.; Zhang, Q.; Chen, J.M.; Yang, X.; Duan, Y.S.; Li, J. Intense secondary aerosol formation due to strong atmospheric photochemical reactions in summer observations at a rural site in eastern Yangtze River Delta of China. Sci. Total Environ. 2016, 571, 1454–1466. [Google Scholar] [CrossRef] [PubMed]
- Squizzato, S.; Masiol, M.; Brunelli, A.; Pistollato, S.; Tarabotti, E.; Rampazzo, G.; Pavoni, B. Factors determining the formation of secondary inorganic aerosol: A case study in the Po Valley (Italy). Atmos. Chem. Phys. 2013, 13, 1927–1939. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.B.; Li, X.H.; Shi, G.M.; Cao, J.J.; Yang, F.M.; Ma, Y.L.; He, K.B. PM2.5 Chemical compositions and aerosol optical properties in Beijing during the late fall. Atmosphere 2015, 6, 164–182. [Google Scholar] [CrossRef]
- Ye, Z.L.; Liu, J.S.; Gu, A.J.; Feng, F.F.; Liu, Y.H.; Bi, C.L.; Xu, J.Z.; Chen, H.; Chen, Y.F.; Dai, L.; et al. Chemical characterization of fine particular matter in Changzhou, China and source apportionment with offline aerosol mass spectrometry. Atmos. Chem. Phys. 2017, 17, 2573–2592. [Google Scholar] [CrossRef]
- Wu, D.; Lin, S.L.; Yang, H.Q.; Du, R.G.; Xia, J.R.; Qi, B.; Liu, G.; Li, F.Y.; Yang, M.; Ge, X.L. Pollution characteristics and light extinction contribution of water-soluble ions of PM2.5 in Hangzhou. Atmos. Environ. 2017, 38, 2656–2666. (In Chinese) [Google Scholar]
- Huang, K.; Zhuang, G.; Lin, Y.; Fu, J.S.; Wang, Q.; Liu, T.; Zhang, R.; Jiang, Y.; Deng, C.; Fi, Q.; et al. Typical types and formation mechanisms of haze in an Eastern Asia megacity, Shanghai. Atmos. Chem. Phys. 2012, 12, 105–124. [Google Scholar] [CrossRef]
- Cheng, Z.; Wang, S.X.; Jiang, J.K.; Fu, Q.Y.; Chen, C.H.; Xu, B.Y.; Yu, J.Q.; Fu, X.; Hao, J.M. Long-term trend of haze pollution and impact of particulate matter in the Yangtze River Delta, China. Environ. Pollut. 2013, 182, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.H.; Zhang, R.Y.; Gomez, M.E.; Yang, L.X.; Zamora, M.L.; Hu, M.; Lin, Y.; Peng, J.F.; Guo, S.; Meng, J.J.; et al. Persistent sulfate formation from London Fog to Chinese haze. Proc. Natl. Aacd. Sci. USA 2016, 113, 13630–13635. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.W.; Zhang, X.Y.; Zhang, Y.M.; Shen, X.J.; Sun, J.Y.; Ma, Q.L.; Yu, X.M.; Zhu, J.L.; Zhang, L.; Che, H.C. Significant concentration changes of chemical components of PM1 in the Yangtze River Delta area of China and the implications for the formation mechanism of heavy haze–fog pollution. Sci. Total Environ. 2015, 538, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.J.; Sun, J.Y.; Zhang, X.Y.; Zhang, Y.M.; Zhang, L.; Che, H.C.; Ma, Q.L.; Yu, X.M.; Yue, Y.; Zhang, Y.W. Characterization of submicron aerosols and effect on visibility during a severe haze-fog episode in Yangtze River Delta, China. Atmos. Environ. 2015, 120, 307–316. [Google Scholar] [CrossRef]
| Components | Mean ± SD | |||
|---|---|---|---|---|
| Winter (Number = 79) | Spring (Number = 108) | Summer (Number = 123) | Autumn (Number = 132) | |
| PM2.5 | 166.0 ± 96.3 | 97.4 ± 35.9 | 100.4 ± 40.8 | 85.3 ± 44.4 |
| SO42− | 23.3 ± 19.6 | 15.7 ± 8.32 | 24.3 ± 9.51 | 14.5 ± 7.89 |
| NO3− | 29.0 ± 22.8 | 10.9 ± 7.85 | 12.2 ± 11.3 | 13.3 ± 12.0 |
| NH4+ | 16.7 ± 8.37 | 9.18 ± 4.12 | 13.1 ± 5.36 | 12.5 ± 7.45 |
| Na+ | 0.73 ± 0.30 | 0.55 ± 0.26 | 0.59 ± 0.78 | 0.36 ± 0.11 |
| K+ | 1.99 ± 1.02 | 2.03 ± 2.17 | 1.60 ± 1.40 | 1.01 ± 0.43 |
| Mg2+ | 0.09 ± 0.04 | 0.10 ± 0.07 | 0.06 ± 0.05 | 0.08 ± 0.04 |
| Ca2+ | 0.75 ± 0.60 | 1.06 ± 0.55 | 0.50 ± 0.28 | 1.43 ± 0.75 |
| F- | 0.11 ± 0.08 | 0.08 ± 0.09 | 0.07 ± 0.06 | 0.06 ± 0.03 |
| Cl− | 3.55 ± 2.15 | 1.36 ± 1.41 | 1.15 ± 1.59 | 1.39 ± 1.44 |
| OC | 21.5 ± 12.9 | 15.0 ± 7.54 | 13.6 ± 8.24 | 12.7 ± 6.87 |
| EC | 5.10 ± 3.30 | 3.95 ± 2.72 | 2.88 ± 1.50 | 2.06 ± 1.00 |
| SOC | 6.66 ± 6.61 | 4.23 ± 4.38 | 5.06 ± 4.82 | 4.44 ± 4.16 |
| SOC/OC | 28.9 ± 16.1% | 27.4 ± 19.2% | 33.7 ± 18.4% | 32.8 ± 16.0% |
| Season | Time | Temperature/°C | Relative Humidity (%) | Wind Velocity (m·s−1) | Mixed Layer Height (m) 1 | Rainfall (mm) 2 |
|---|---|---|---|---|---|---|
| Winter | Day | 6.3 | 49.0 | 1.8 | 962.3 | 116.8 |
| Night | 2.4 | 66.8 | 1.0 | 153.3 | ||
| Spring | Day | 22.2 | 36.3 | 2.0 | 1384.7 | 136.4 |
| Night | 17.3 | 51.4 | 1.7 | 135.2 | ||
| Summer | Day | 27.8 | 59.8 | 1.4 | 1361.2 | 405.5 |
| Night | 24.0 | 78.9 | 1.1 | 147.3 | ||
| Autumn | Day | 20.1 | 56.2 | 1.7 | 1194.1 | 170.6 |
| Night | 15.9 | 78.5 | 1.2 | 155.3 |
| Haze Grades | Distinguishing Criteria | Day (Proportion, %) | PM2.5 (μg·m−3) | NH4+ (μg·m−3) | NO3− (μg·m−3) | SO42− (μg·m−3) | OC (μg·m−3) | EC (μg·m−3) | RH 1 (%) | WS (m·s−1) | MLH (m) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Non_haze | V > 10.0 2 | 18 (27.7) | 69.9 | 7.25 | 6.58 | 9.66 | 9.27 | 2.06 | 56.3 | 2.29 | 1082.2 |
| Slight | 5.0 ≤ V < 10.0 | 32 (49.2) | 98.0 | 11.9 | 12.4 | 17.9 | 14.5 | 3.16 | 62.3 | 1.35 | 1279.9 |
| Mild | 3.0 ≤ V < 5.0 | 11 (16.9) | 132.8 | 19.2 | 22.3 | 25.5 | 16.6 | 3.31 | 69.7 | 1.37 | 1041.0 |
| Moderate | 2.0 ≤ V < 3.0 | 3 (4.6) | 165.4 | 23.2 | 34.6 | 30.9 | 20.4 | 4.25 | 66.6 | 1.68 | 841.7 |
| Severe | V < 2.0 | 1 (1.5) | 383.7 | 23.8 | 80.3 | 72.7 | 45.5 | 9.44 | 75.4 | 0.38 | 772.0 |
| Mass Source | PM2.5 (μg·m−3) | NH4+ (μg·m−3) | NO3− (μg·m−3) | SO42− (μg·m−3) | OC (μg·m−3) | EC (μg·m−3) | RH (%) | WS (m·s−1) | MLH (m) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 94.3 | 12.4 | 12.7 | 19.1 | 12.1 | 2.52 | 68.0 | 1.48 | 1227.1 |
| 2 | 119.7 | 14.3 | 17.9 | 19.4 | 18.8 | 4.06 | 56.3 | 1.39 | 1079.4 |
| 3 | 120.1 | 13.4 | 17.9 | 21.5 | 15.6 | 3.37 | 65.1 | 1.39 | 1147.5 |
| 4 | 86.2 | 7.90 | 8.41 | 10.2 | 13.2 | 3.30 | 48.3 | 2.48 | 1391.4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, D.; Zhang, F.; Ge, X.; Yang, M.; Xia, J.; Liu, G.; Li, F. Chemical and Light Extinction Characteristics of Atmospheric Aerosols in Suburban Nanjing, China. Atmosphere 2017, 8, 149. https://doi.org/10.3390/atmos8080149
Wu D, Zhang F, Ge X, Yang M, Xia J, Liu G, Li F. Chemical and Light Extinction Characteristics of Atmospheric Aerosols in Suburban Nanjing, China. Atmosphere. 2017; 8(8):149. https://doi.org/10.3390/atmos8080149
Chicago/Turabian StyleWu, Dan, Fan Zhang, Xinlei Ge, Meng Yang, Junrong Xia, Gang Liu, and Fengying Li. 2017. "Chemical and Light Extinction Characteristics of Atmospheric Aerosols in Suburban Nanjing, China" Atmosphere 8, no. 8: 149. https://doi.org/10.3390/atmos8080149




