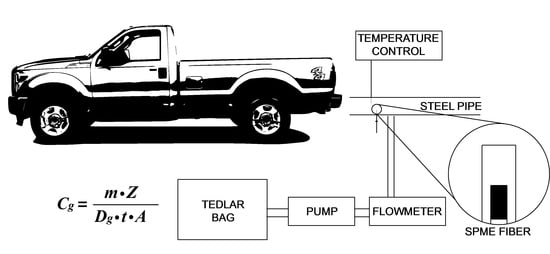
Passive Sampling and Analysis of Naphthalene in Internal Combustion Engine Exhaust with Retracted SPME Device and GC-MS
Abstract
1. Introduction
1.1. PAHs and Air Pollution
1.2. Naphthalene Toxicity
1.3. Source of PAHs and Fuel Content Regulations
1.4. Methods for PAHs Quantification
1.5. SPME for Sampling of Vehicle Exhaust Gases
1.6. Objectives
2. Theory
2.1. Passive TWA Naphthalene Gas Sampling with Retracted SPME Fiber
- m = naphthalene mass extracted by fiber coating (mg);
- Z = SPME fiber retraction depth (diffusion path length) (cm);
- Dg = gas-phase naphthalene diffusion coefficient (cm2·s−1);
- t = extraction (sampling) time (s);
- A = cross-sectional area of SPME needle opening (cm2).
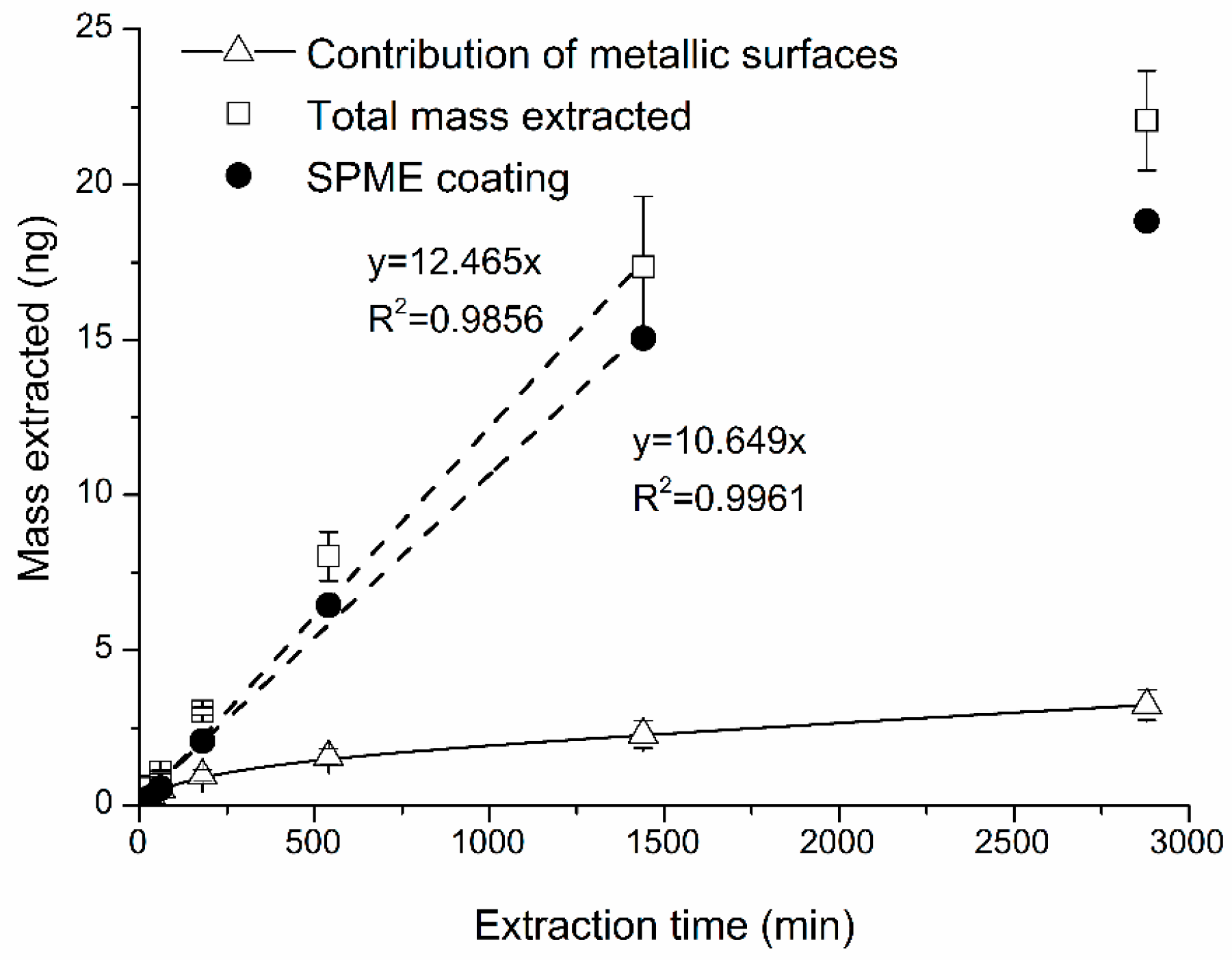
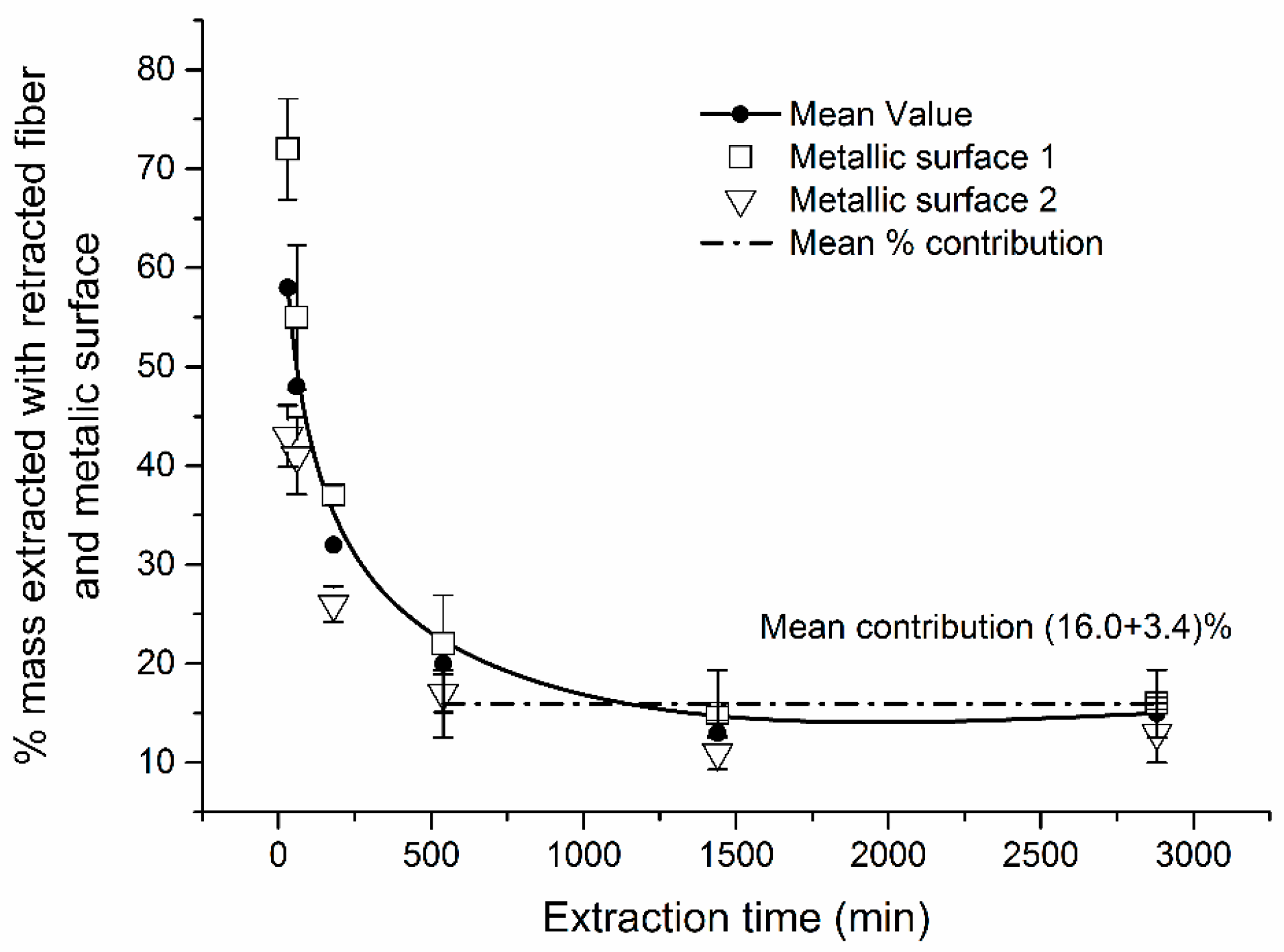
2.2. Contribution of Adsorption by Metallic Parts of SPME Needle Assembly
3. Experiments
3.1. Chemicals
3.2. SPME Conditions
3.3. GC-MS Conditions
3.4. MS Detector Calibration with Naphthalene Standard Solution
3.5. Method Detection Limits (MDLs)
- t = the student’s value at 95% confidence level and SD estimate with n − 1 degrees of freedom;
- SD = the standard deviation of 10 replicates;
- MDL for p = 0.95 was then estimated at 11.5 ppb or 0.06 mg·m−3.
3.6. Standard Gas Generation System
3.7. Variability Associated with the Same SPME Fiber Type and Fiber Coating Type
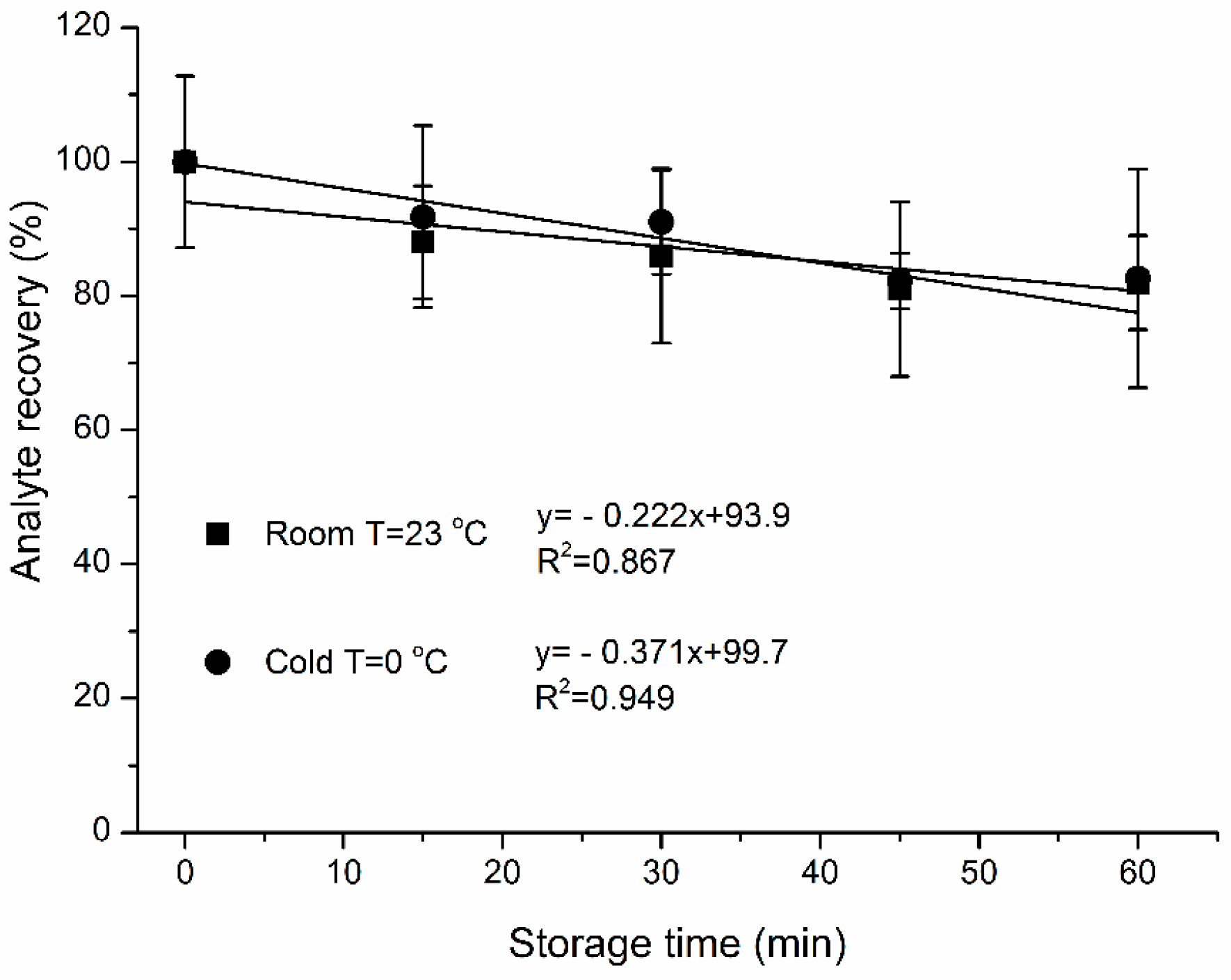
3.8. Effects of Storage Time and Temperature on Analyte Mass Loss
3.9. Effects of t, Cg, Z and Adsorption by Metallic SPME Assembly: Verification of Fick’s Law Model for TWA-SPME Gas Sampling of Naphthalene
3.10. Verification of TWA-SPME Method for Naphthalene Quantification in Vehicle Exhaust Gas
4. Results and Discussion
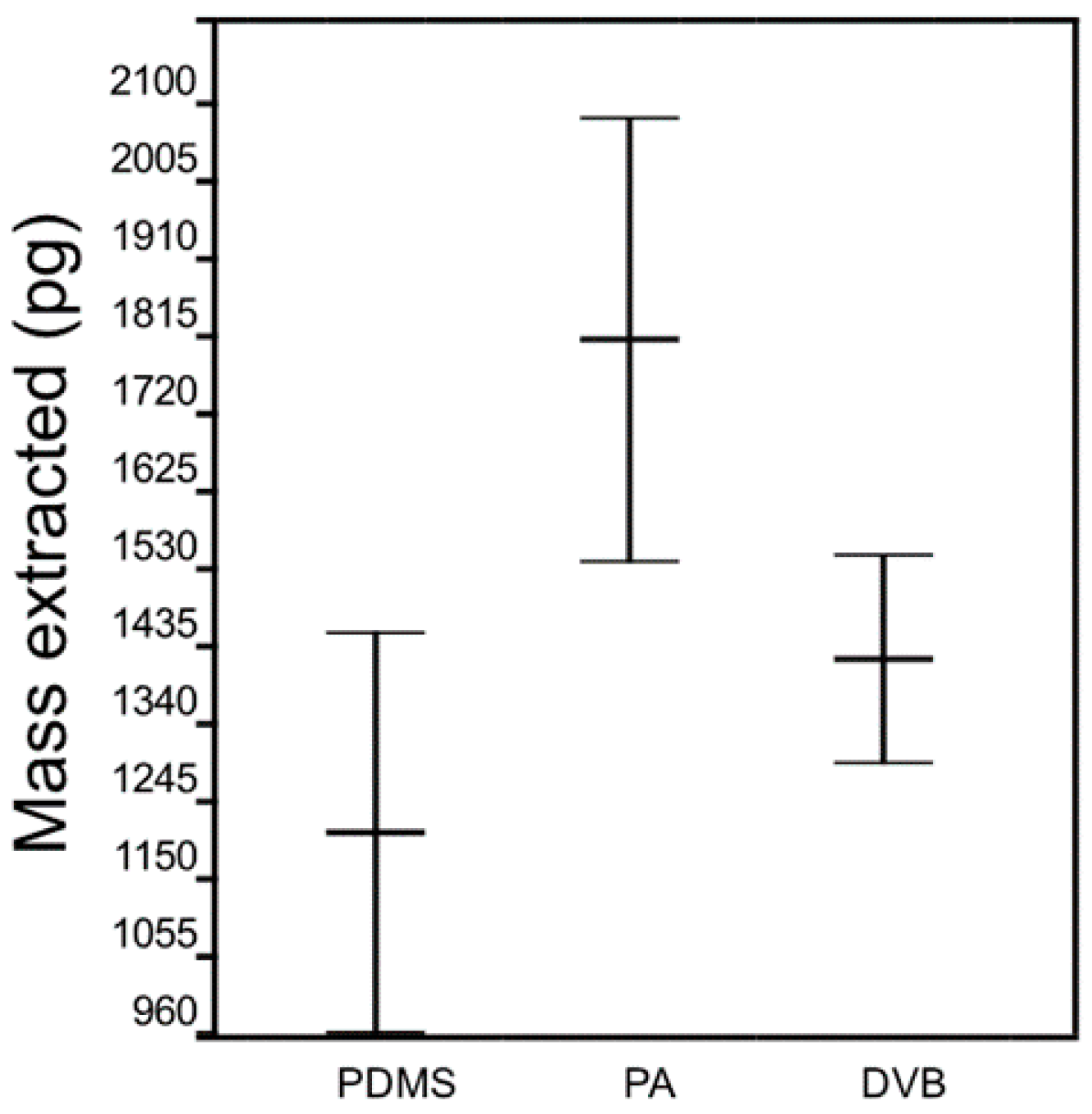
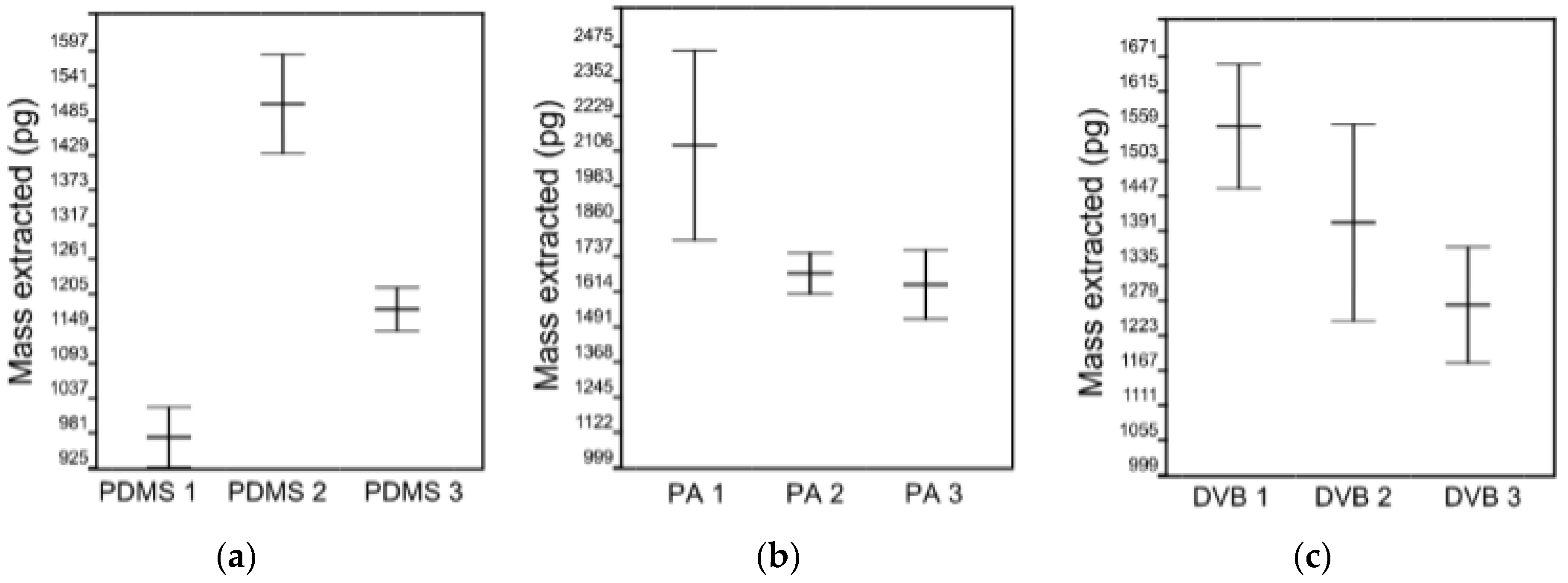
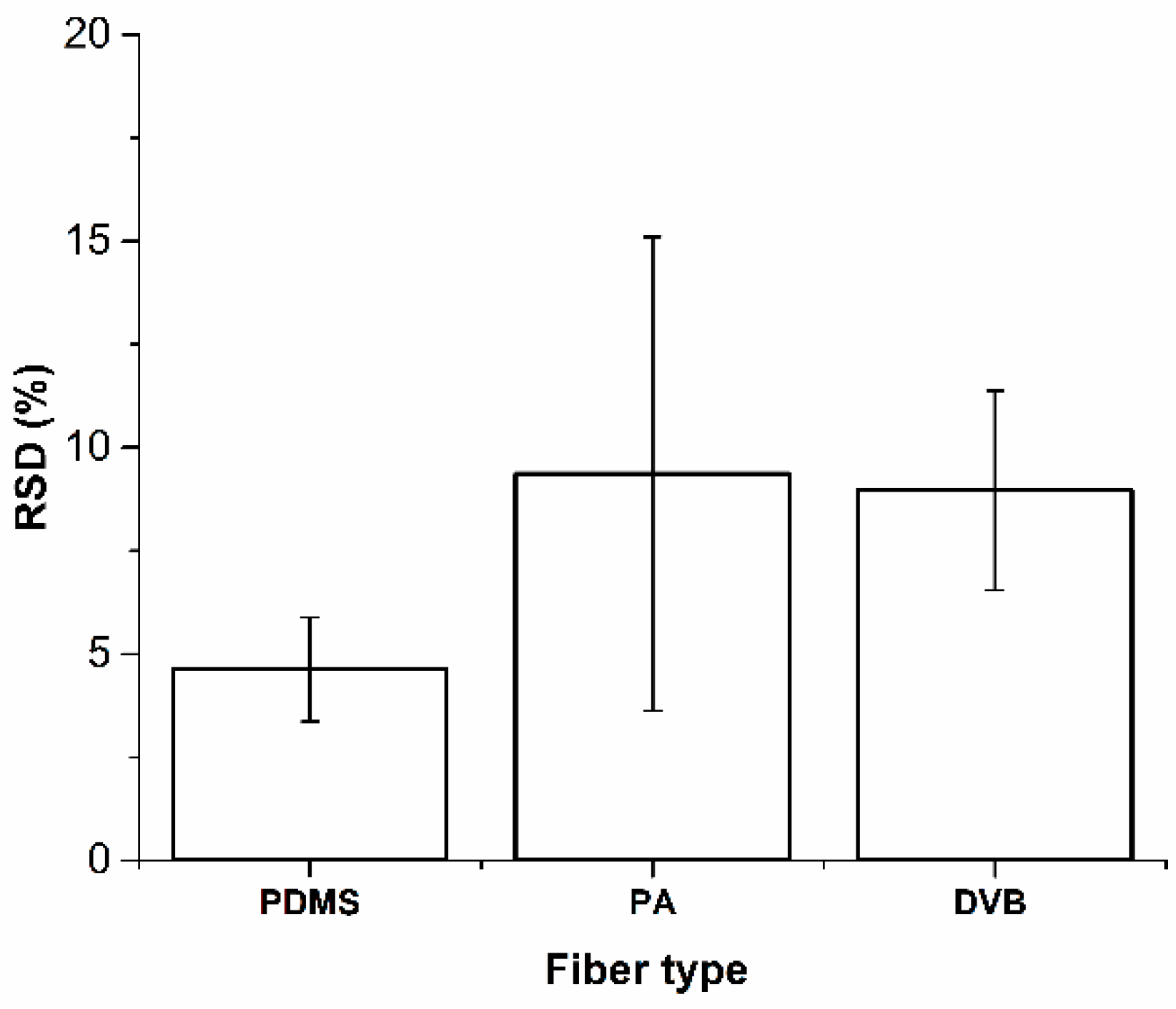
4.1. Variability Associated with the Same SPME Fiber Type and Fiber Coating Type
4.2. Effects of Storage Time and Temperature on Sample Mass Loss
4.3. Effect of Sampling Time (t)
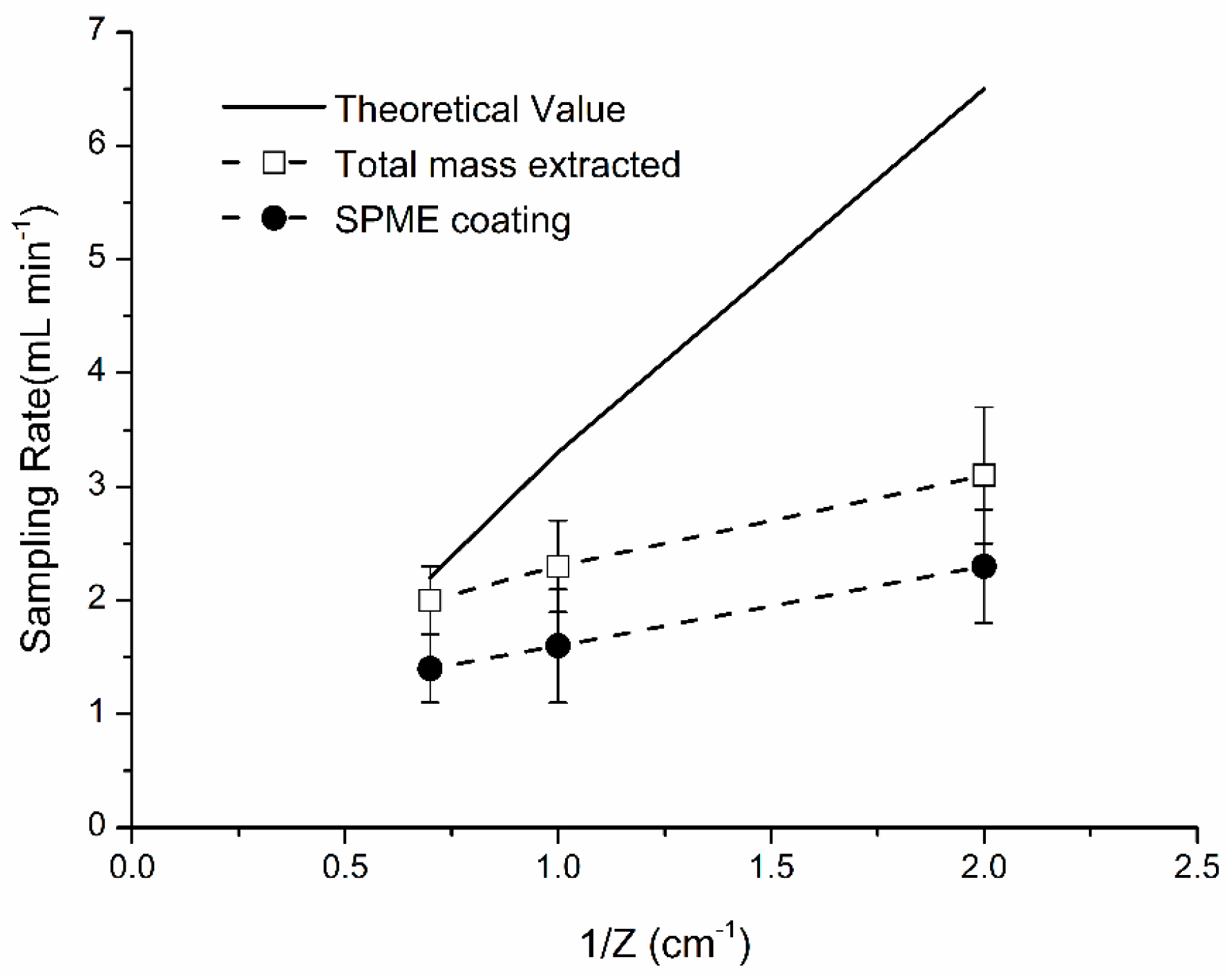
4.4. Effect of Retraction Depth Z
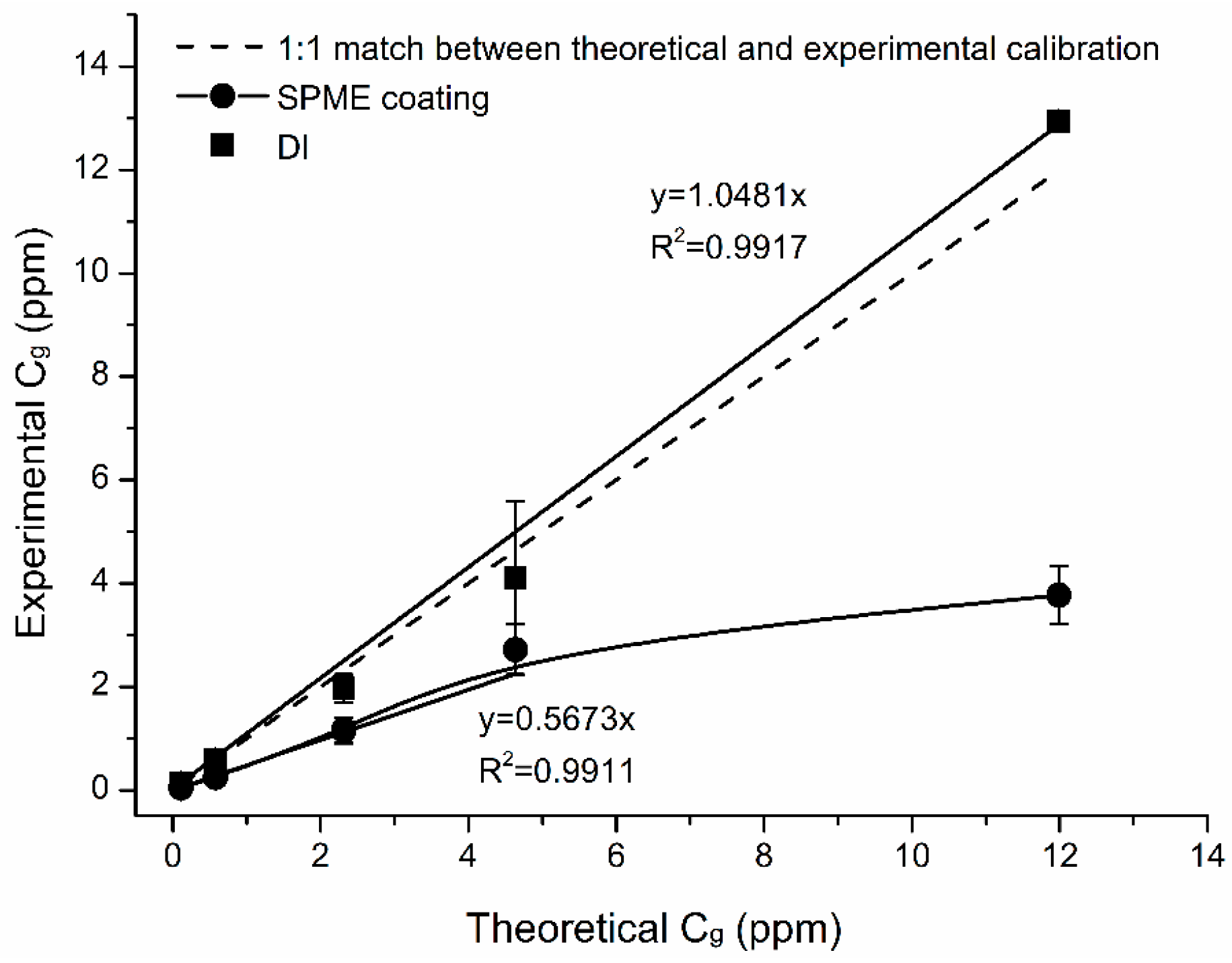
4.5. Effect of Naphthalene Concentration (Cg)
4.6. Validation of TWA-SPME Method for Naphthalene Quantification in Vehicle Exhaust Gas
5. Conclusions
- The effects of variability of SPME fibers quality on mass extracted are significant. ANOVA showed no effect on mass extracted by randomly selected PA and DVB fiber coating, while randomly selected PDMS fibers extract significantly different masses of analytes. However, reproducible results with lower than 5% RSD were obtained with PDMS coating, which was chosen as an optimal fiber for further experiments.
- Losses during sample storage time ranged from ~0.2 ± 0.07%/min and 0.4 ± 0.07%/min sample losses for 23 °C and 0 °C, respectively. Storage temperature had no influence on sample recovery from SPME fiber coating. Sample storage in cool temperatures does not appear to have practical benefits.
- SPME fiber coating was saturated by using a sampling time of longer than 1440 min.
- Effects of gas concentrations on naphthalene mass extracted with TWA-SPME were linear in the whole studied concentration range (0.1–12 ppm) with R2 > 0.99. Mean masses extracted by SPME were lower than predicted by theory by 54%. It was likely due to a saturation of SPME fiber when subjected to Cg = 4 ppm. The mean % difference for (lower range) Cg < 4 ppm was 49% for naphthalene. The mean % difference for (higher range) Cg > 4 ppm was 60%.
- Effects of fiber retraction depth % of naphthalene mass were calculated by dividing mass extracted by “broken” fiber to the total mass extracted by SPME assembly (coating + metallic parts). Percent difference between experimental and theoretical mass was 39%. It is recommended using a shorter Z to minimize the % contribution by metallic parts of SPME fiber.
- The interfering contribution of extraction by (adsorption to) metallic parts of SPME needle assembly itself was reproducible and ranged from 26% to 43% for shorter sampling time (<540 min) and was virtually constant (16%) for sampling time longer than 540 min.
- The new method was verified on exhaust gases from idling pickup truck and a tractor and compared side-by-side with a direct injection of sampled exhaust gas method. DI analysis was below method detection limits.
- The TWA-SPME approach can be considered for adoption to a wide range of measurements involving volatile organic compounds (VOCs) and semivolatile organics, a condition that careful method development focuses on the variability of SPME fibers, fiber coatings, and quantifying the contribution of extraction (adsorption) to SPME metallic parts.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- U.S. Environmental Protection Agency. Compendium of Methods for the Determination of Toxic Organic Compounds in Ambient Air. Available online: https://www3.epa.gov/ttnamti1/files/ambient/airtox/tocomp99.pdf (accessed on 3 September 2014).
- U.S. Environmental Protection Agency. Naphthalene (CASRN 91-20-3). Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0436_summary.pdf (accessed on 15 September 2014).
- U.S. Environmental Protection Agency. Health and Environmental Effects Profile for Naphthalene. Available online: https://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=49301 (accessed on 28 January 2015).
- U.S. Environmental Protection Agency. Health Effects Assessment for Naphthalene. Available online: https://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=40598 (accessed on 15 October 2014).
- U.S. Environmental Protection Agency. Interim Methods for Development of Inhalation Reference Doses. Available online: https://cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=35707&CFID=80546352&CFTOKEN=63397154 (accessed on 28 October 2014).
- U.S. Environmental Protection Agency. Methods for Derivation of Inhalation Reference Concentrations and Application of Inhalation Dosimetry. Available online: https://www.epa.gov/sites/production/files/2014-11/documents/rfc_methodology.pdf (accessed on 28 November 2014).
- Shaltout, A.A.; Boman, J.; Welz, B.; Castilho, I.N.B.; Al Ashkar, E.A.; Gaita, S.M. Method development for the determination of Cd, Cu, Ni and Pb in PM2.5 particles sampled in industrial and urban areas of Greater Cairo, Egypt, using high-resolution continuum source graphite furnace atomic absorption spectrometry. Microchem. J. 2014, 113, 4–9. [Google Scholar] [CrossRef]
- De Abrantes, R.; de Assuncao, J.; Pesquero, C. Emission of polycyclic aromatic hydrocarbons from light-duty diesel vehicles exhaust. Atmos. Environ. 2004, 38, 1631–1640. [Google Scholar] [CrossRef]
- Moreira dos Santos, C.Y.; de Almeida Azevedo, D.; de Aquino Neto, F.R. Atmospheric distribution of organic compounds from urban areas near a coal-fired power station. Atmos. Environ. 2004, 38, 1247–1257. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, C.; Cheng, Y.; Liu, C.; Zhang, H.; Zhang, G.; Sun, X.; Mu, Y. Serious BTEX pollution in rural area of the North China Plain during winter season. J. Environ. Sci. (China) 2015, 30, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Marr, L.C.; Kirchstetter, T.W.; Harley, R.A. Characterization of polycyclic aromatic hydrocarbons in motor vehicle fuels and exhaust emissions. Environ. Sci. Technol. 1999, 33, 3091–3099. [Google Scholar] [CrossRef]
- Heeb, N.V.; Schmid, P.; Kohler, M.; Gujer, E.; Zennegg, M.; Wenger, D.; Wichser, A.; Ulrich, A.; Gfeller, U.; Honegger, P.; et al. Secondary effects of catalytic diesel particulate filters: Conversion of PAHs versus formation of nitro-PAHs. Environ Sci. Technol. 2008, 42, 3773–3779. [Google Scholar] [CrossRef] [PubMed]
- Westerholm, R.; Li, H. A multivariate statistical analysis of fuel-related polycyclic aromatic hydrocarbon emissions from heavy-duty diesel vehicles. Environ. Sci. Technol. 1994, 28, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Westerholm, R.N.; Alsberg, T.E.; Frommelin, A.B.; Strandell, E. Effect of fuel polycyclic aromatic hydrocarbon content on the emissions of polycyclic aromatic hydrocarbons and other mutagenic substances from a gasoline-fueled automobile. Environ. Sci. Technol. 1988, 22, 925–930. [Google Scholar] [CrossRef] [PubMed]
- EN 590. Automotive Fuels-Diesel-Requirements and Test Methods. Available online: http://www.envirochem.hu/www.envirochem.hu/documents/EN_590_2009_hhV05.pdf (accessed on 25 February 2016).
- Hill, N. EU Fuel Quality Monitoring—2002 Summary Report Final report produced for the European Commission, DG Environment. A E A Technol. Environ. 2004, 33–46. Available online: https://circabc.europa.eu/sd/a/ae36bf6d-2a23-46e7-b14b-5c15f70efc6d/fqm_summary_2003.pdf (accessed on 25 February 2016).
- Aakko, P. PAH Content of Diesel Fuels. Available online: http://www.vtt.fi/inf/julkaisut/muut/2006/VTT-1155-06-AROM.pdf (accessed on 3 March 2015).
- Laurikko, J.; Nylund, N.O. Regulated and Unregulated Emissions from catalyst vehicles at Low Ambient Temperatures. SAE Tech. Pap. 1993, 13. [Google Scholar] [CrossRef]
- Westerholm, R.; Christensen, A. Regulated and unregulated exhaust emissions from two three-way catalyst equipped gasoline fuelled vehicles. Atmos. Environ. 1996, 30, 3529–3536. [Google Scholar] [CrossRef]
- Clairotte, M.; Adam, T.W.; Zardini, A.A.; Manfredi, U.; Martini, G.; Krasenbrink, A.; Vicet, A.; Tournié, E.; Astorga, C. Effects of low temperature on the cold start gaseous emissions from light duty vehicles fuelled by ethanol-blended gasoline. Appl. Energy 2013, 102, 44–54. [Google Scholar] [CrossRef]
- Tsapakis, M.; Stephanou, E.G. Occurrence of gaseous and particulate polycyclic aromatic hydrocarbons in the urban atmosphere: Study of sources and ambient temperature effect on the gas/particle concentration and distribution. Environ. Pollut. 2005, 133, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Mandalakis, M.; Tsapakis, M.; Tsoga, A.; Stephanou, E.G. Gas-particle concentrations and distribution of aliphatic hydrocarbons, PAHs, PCBs and PCDD/Fs in the atmosphere of Athens (Greece). Atmos. Environ. 2002, 36, 4023–4035. [Google Scholar] [CrossRef]
- Possanzini, M.; di Palo, V.; Gigliucci, P.; Tomasi Scianò, M.C.; Cecinato, A. Determination of phase-distributed PAH in Rome ambient air by denuder/GC-MS method. Atmos. Environ. 2004, 38, 1727–1734. [Google Scholar] [CrossRef]
- Bezabeh, D.Z. Screening of aerosol filter samples for PAHs and Nitro-PAHs by laser desorption ionization TOF Mass Spectrometry. Aerosol Sci. Technol. 1999, 30, 288–299. [Google Scholar] [CrossRef]
- Bertoni, G.; Tappa, R.; Cecinato, A. Environmental monitoring of semi-volatile polyciclic aromatic hydrocarbons by means of diffusive sampling devices and GC-MS analysis. Chromatographia 2001, 53, 312–316. [Google Scholar] [CrossRef]
- Wei, M.C.; Chang, W.T.; Jen, J.F. Monitoring of PAHs in air by collection on XAD-2 adsorbent then microwave-assisted thermal desorption coupled with headspace solid-phase microextraction and gas chromatography with mass spectrometric detection. Anal. Bioanal. Chem. 2007, 387, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, R.; Hernández, J.J.; Lyons, L.L. Determination of PAHs in diesel particulate matter using thermal extraction and solid phase micro-extraction. Atmos. Environ. 2009, 43, 655–662. [Google Scholar] [CrossRef]
- Pacenti, M.; Lofrumento, C.; Dugheri, S.; Zoppi, A.; Borsi, I.; Speranza, A.; Boccalon, P.; Arcangeli, G.; Antoniucci, A.; Castellucci, E.M. Physicochemical characterization of exhaust particulates from gasoline and diesel engines by solid-phase micro extraction sampling and combined raman microspectroscopic/fast gas-chromotography mass spectrometry analysis. Eur. J. Inflamm. 2009, 7, 25–37. [Google Scholar]
- Koziel, J.A.; Odziemkowski, M.; Pawliszyn, J. Sampling and analysis of airborne particulate matter and aerosols using in-needle trap and SPME fiber devices. Anal. Chem. 2001, 73, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Baimatova, N.; Koziel, J.A.; Kenessov, B. Quantification of benzene, toluene, ethylbenzene and o-xylene in internal combustion engine exhaust with time-weighted average solid phase microextraction and gas chromatography mass spectrometry. Anal. Chim. Acta 2015, 873, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Lesellier, E. Extraction and analysis of polycyclic aromatic hydrocarbons (PAHs) by solid phase micro-extraction/supercritical fluid chromatography (SPME/SFC). Analusis 1999, 27, 363–368. [Google Scholar] [CrossRef]
- Tuncel, S.G.; Topal, T. Multifactorial optimization approach for determination of polycyclic aromatic hydrocarbons in sea sediments of Turkish Mediterranean coast. Am. J. Anal. Chem. 2011, 02, 783–794. [Google Scholar] [CrossRef][Green Version]
- Cam, D.; Gagni, S.; Meldolesi, L.; Galletti, G. Determination of polycyclic aromatic hydrocarbons in sediment using solid-phase microextraction with gas chromatography-mass spectrometry. J. Chromatogr. Sci. 2000, 38, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Doong, R.; Chang, S.; Sun, Y. Solid-phase microextraction and headspace solid-phase microextraction for the determination of high molecular-weight polycyclic aromatic hydrocarbons in water and soil samples. J. Chromatogr. Sci. 2000, 38, 528–534. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Y.; Zhang, J.; Ding, Y.; Zhou, J.; Ni, L.; Sun, C. Quantitative determination of 16 polycyclic aromatic hydrocarbons in soil samples using solid-phase microextraction. J. Sep. Sci. 2009, 32, 3951–3957. [Google Scholar] [CrossRef] [PubMed]
- Odziemkowski, M.; Koziel, J. A.; Irish, D.E.; Pawliszyn, J. Sampling and raman confocal microspectroscopic analysis of airborne particulate matter using poly(dimethylsiloxane) solid-phase microextraction fibers. Anal. Chem. 2001, 73, 3131–3139. [Google Scholar] [CrossRef] [PubMed]
- Woolcock, P.J.; Koziel, J.A.; Cai, L.; Johnston, P.A.; Brown, R.C. Analysis of trace contaminants in hot gas streams using time-weighted average solid-phase microextraction: Proof of concept. J. Chromatogr. A 2013, 1281, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Woolcock, P.J.; Koziel, J.A.; Johnston, P.A.; Brown, R.C.; Broer, K.M. Analysis of trace contaminants in hot gas streams using time-weighted average solid-phase microextraction: Pilot-scale validation. Fuel 2015, 153, 552–558. [Google Scholar] [CrossRef]
- Koziel, J.A.; Nguyen, L.T.; Glanville, T.D.; Ahn, H.K.; Frana, T.S.; van Leeuwen, J.H. Method for sampling and analysis of volatile biomarkers in process gas from aerobic digestion of poultry carcass using time-weighted average SPME and GC-MS. Food Chem. 2017, 232, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Koziel, J.A.; Pawliszyn, J. Air sampling and analysis of volatile organic compounds with solid phase microextraction. J. Air Waste Manag. Assoc. 2001, 51, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Koziel, J.A.; Spinhirne, J.P. Generation and calibration of standard gas mixtures for volatile fatty acids using permeation tubes and solid-phase microextraction. Am. Soc. Agric. Eng. 2003, 46, 1639–1646. [Google Scholar]
- Koziel, J.A.; Martos, P.A.; Pawliszyn, J. System for the generation of standard gas mixtures of volatile and semi-volatile organic compounds for calibrations of solid-phase microextraction and other sampling devices. J. Chromatogr. A 2004, 1025, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Pawliszyn, J. Handbook of Solid Phase Microextraction, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2012; ISBN 9780124160170. [Google Scholar]
- Rhead, M.M.; Pemberton, R.D. Sources of naphthalene in diesel exhaust emissions. Energy Fuels 1996, 10, 837–843. [Google Scholar] [CrossRef]
- Khalili, N.R.; Scheff, P.A.; Holsen, T. PAH source fingerprints for coke ovens, diesel and gasoline engines, highway tunnels, and wood combustion emissions. Atmos. Environ. 1995, 29, 533–542. [Google Scholar] [CrossRef]
- Mi, H.; Lee, W.; Tsai, P.; Chen, C. A comparison on the emission of polycyclic aromatic hydrocarbons and their corresponding carcinogenic potencies from a vehicle engine using leaded and lead-free gasoline. Environ. Health Perspect. 2001, 109, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
| Theoretical Cg, ppm | Experimental Cg, ppm | |||||
|---|---|---|---|---|---|---|
| SPME | DI | |||||
| Mean | STDEV | RSD | Mean | STDEV | RSD | |
| 0.11 | 0.052 | 0.007 | 13 | 0.15 | 0.02 | 16 |
| 0.58 | 0.25 | 0.04 | 15 | 0.60 | 0.11 | 18 |
| 2.3 | 1.2 | 0.3 | 21 | 2.0 | 0.3 | 14 |
| 4.6 | 2.7 | 0.5 | 18 | 4.1 | 1.5 | 36 |
| 12 | 3.8 | 0.6 | 15 | 13.0 | 0.2 | 1.5 |
| Vehicle | Z, mm | t, h | T, °C * | Cg, mg·m−3 | |||
|---|---|---|---|---|---|---|---|
| TWA SPME Mean | RSD (%) | DI Mean | RSD | ||||
| Full-size pickup | 10 | 8 | 27–95 | (0.08 ± 0.02) | 21 | n/d | n/d |
| Full-size pickup | 3 | 1 | 27–75 | (0.3 ± 0.05) | 16 | n/d | n/d |
| Tractor | 10 | 1 | 27–122 | (0.2 ± 0.05) | 27 | n/d | n/d |
| Vehicle Characteristics | Type of Sampling | Country of Study | Concentration Range, mg·m−3 | References | ||
|---|---|---|---|---|---|---|
| Model | Year | Mileage, km | ||||
| Diesel exhaust | ||||||
| Ford F-250 XL Super Duty pickup truck | 2005 | ~139,200 | TWA SPME 8 h | USA | (0.08 ± 0.02) | This study |
| Ford F-250 XL Super Duty pickup truck | 2005 | ~139,200 | TWA SPME 1 h | (0.30 ± 0.05) | ||
| John Deere 5830 forage harvester | ~30 years | - | (0.20 ± 0.05) | |||
| Perkins Prima diesel engine | - | - | Exhaust gas sampling was performed using the total exhaust solvent stripping apparatus (TESSA) sampling system. The TES was dissolved in hexane (1 mL) and transferred quantitatively to the top of a slurry-packed silica column. The silica was cleaned by Soxhlet extraction with DCM for 24 h before being fully activated at 185 °C for 12 h. | UK | 0.026 | [44] |
| Mazda E5 with Premium leaded gasoline | - | 100,00 | Collected with XAD-2 adsorbent, extracted by Soxhlet for 24 h, with a mixed solvent (n-hexane and dichloromethane, v:v = 1:1, 500 mL each). The extract was concentrated by purging with ultrapure nitrogen (flow rate 1.0 L/min) to 2 mL | Taiwan | 0.0615–0.120 * | [46] |
| Mazda E5 with 92-Lead-free gasoline | 0.0882–0.146 * | |||||
| Mazda E5 with 95-Lead-free gasoline | 0.0875–0.147 | |||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baimatova, N.; Koziel, J.A.; Kenessov, B. Passive Sampling and Analysis of Naphthalene in Internal Combustion Engine Exhaust with Retracted SPME Device and GC-MS. Atmosphere 2017, 8, 130. https://doi.org/10.3390/atmos8070130
Baimatova N, Koziel JA, Kenessov B. Passive Sampling and Analysis of Naphthalene in Internal Combustion Engine Exhaust with Retracted SPME Device and GC-MS. Atmosphere. 2017; 8(7):130. https://doi.org/10.3390/atmos8070130
Chicago/Turabian StyleBaimatova, Nassiba, Jacek A. Koziel, and Bulat Kenessov. 2017. "Passive Sampling and Analysis of Naphthalene in Internal Combustion Engine Exhaust with Retracted SPME Device and GC-MS" Atmosphere 8, no. 7: 130. https://doi.org/10.3390/atmos8070130
APA StyleBaimatova, N., Koziel, J. A., & Kenessov, B. (2017). Passive Sampling and Analysis of Naphthalene in Internal Combustion Engine Exhaust with Retracted SPME Device and GC-MS. Atmosphere, 8(7), 130. https://doi.org/10.3390/atmos8070130