Active Air Monitoring for Understanding the Ventilation and Infection Risks of SARS-CoV-2 Transmission in Public Indoor Spaces
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of the Sampling Area
2.2. Measurement Instrumentation
2.3. Data Collection and Analysis
2.4. Estimation of Ventilation
2.5. Evaluation of the Infection Risk
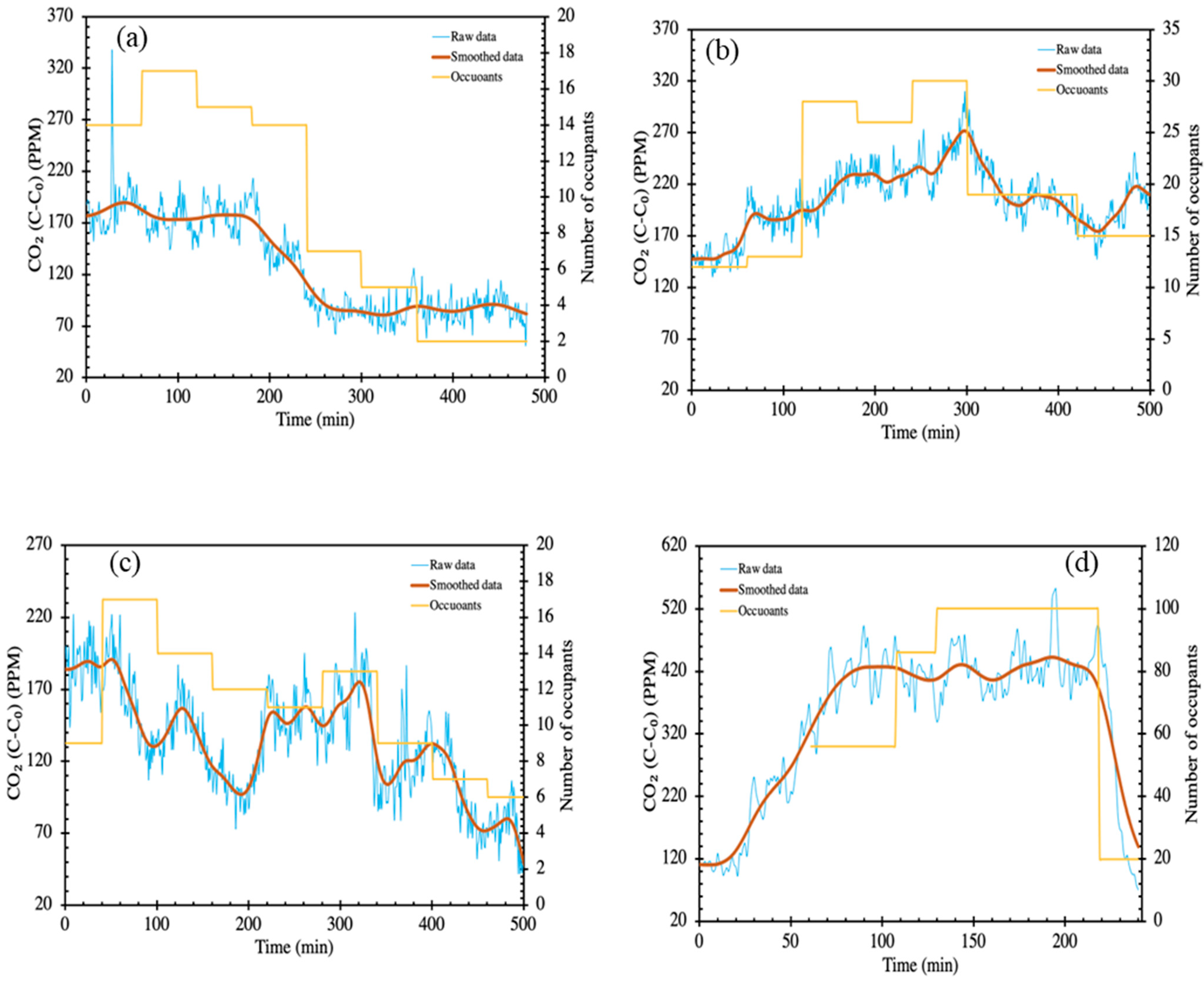
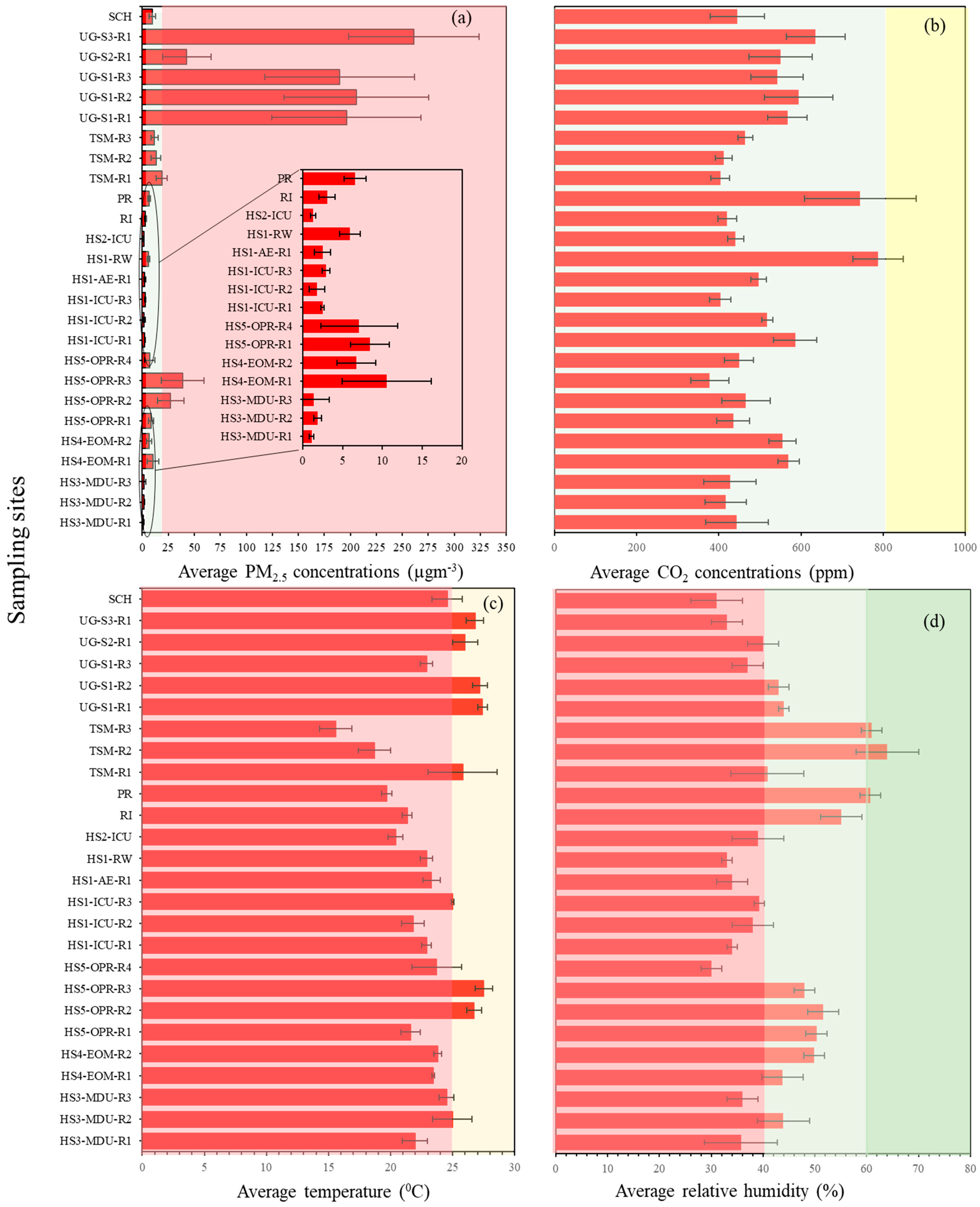
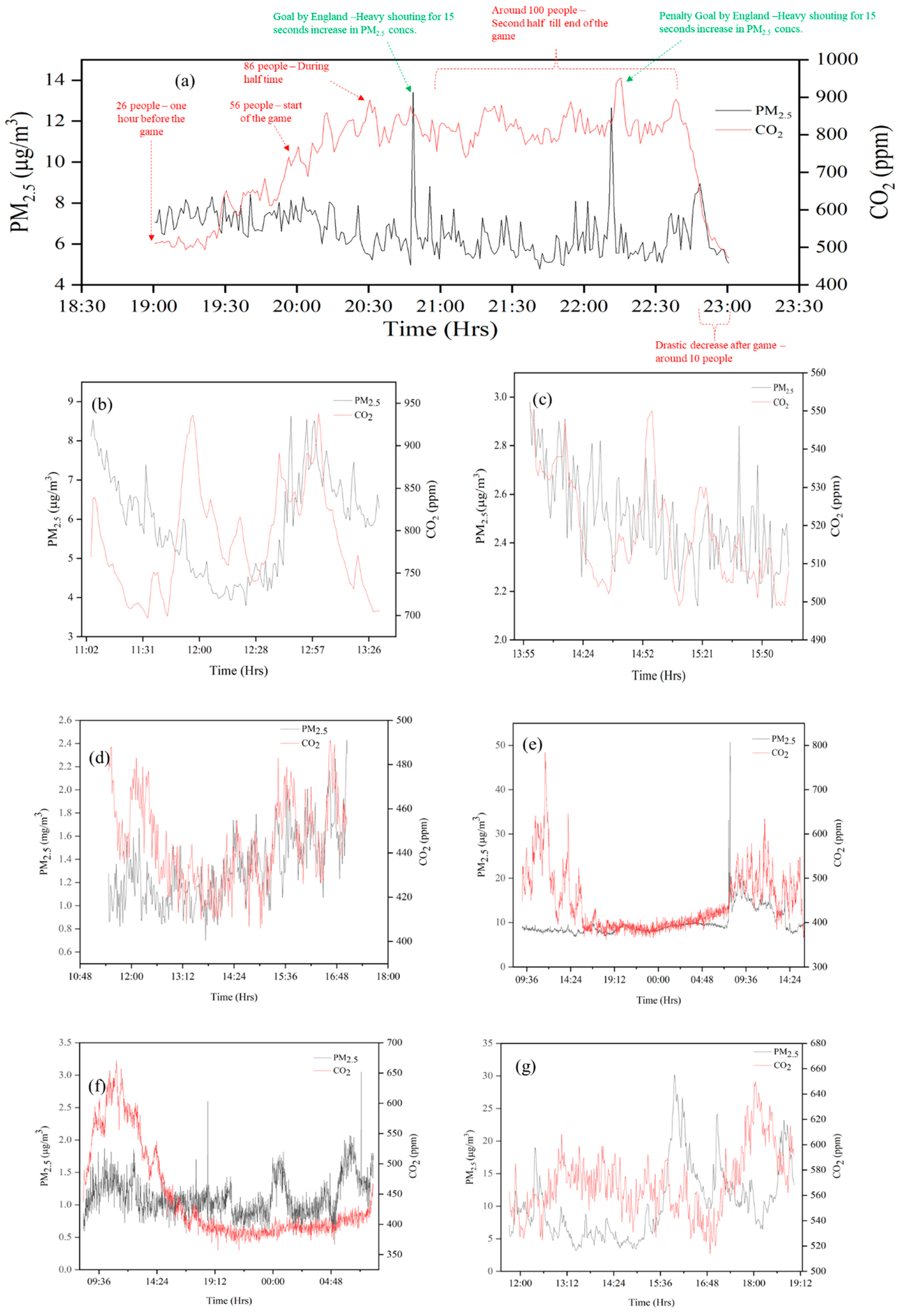
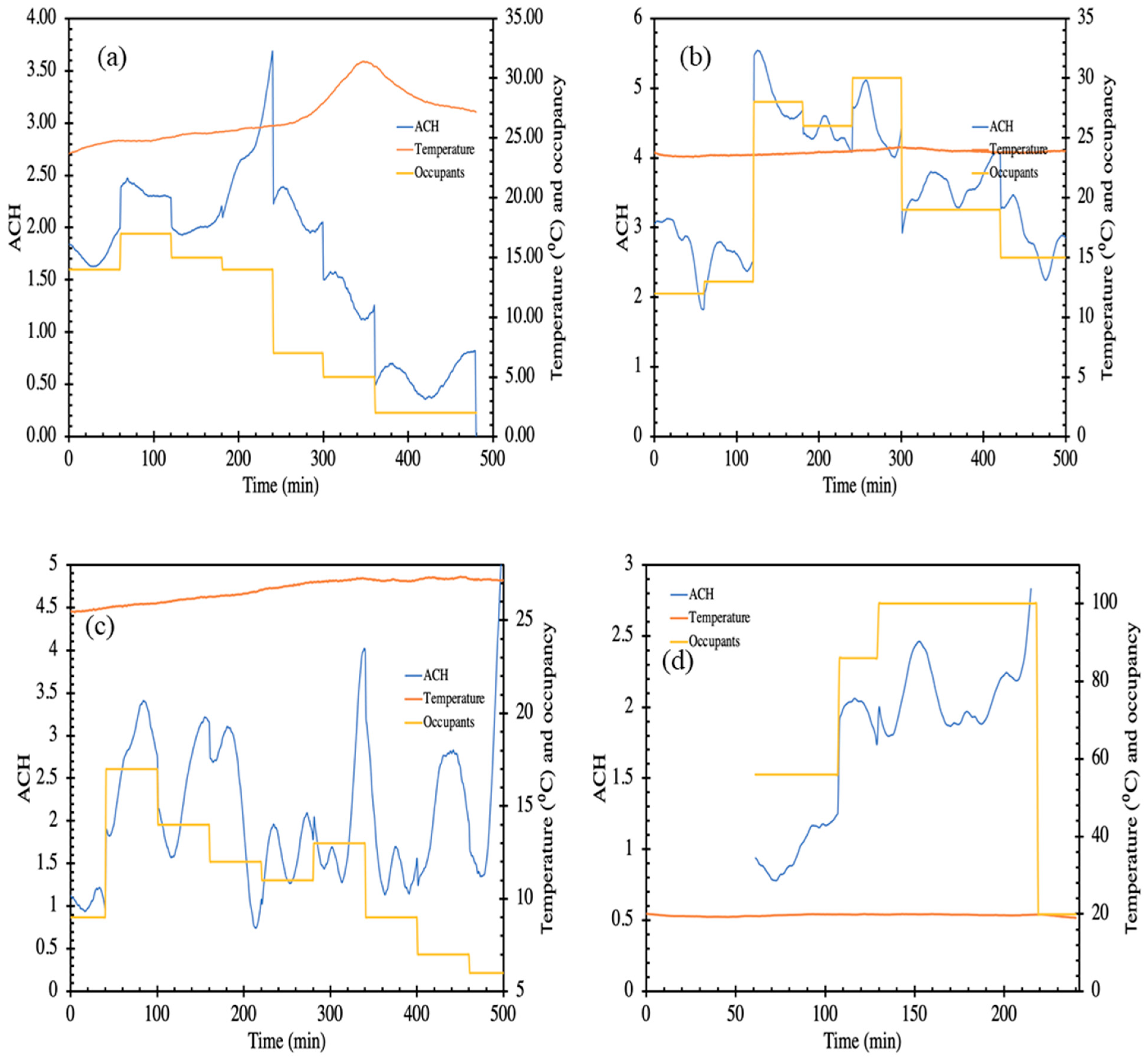
3. Results and Discussion
3.1. PM2.5 and CO2 Concentrations in Different Microenvironments
3.2. Variations in Indoor Temperature and Relative Humidity
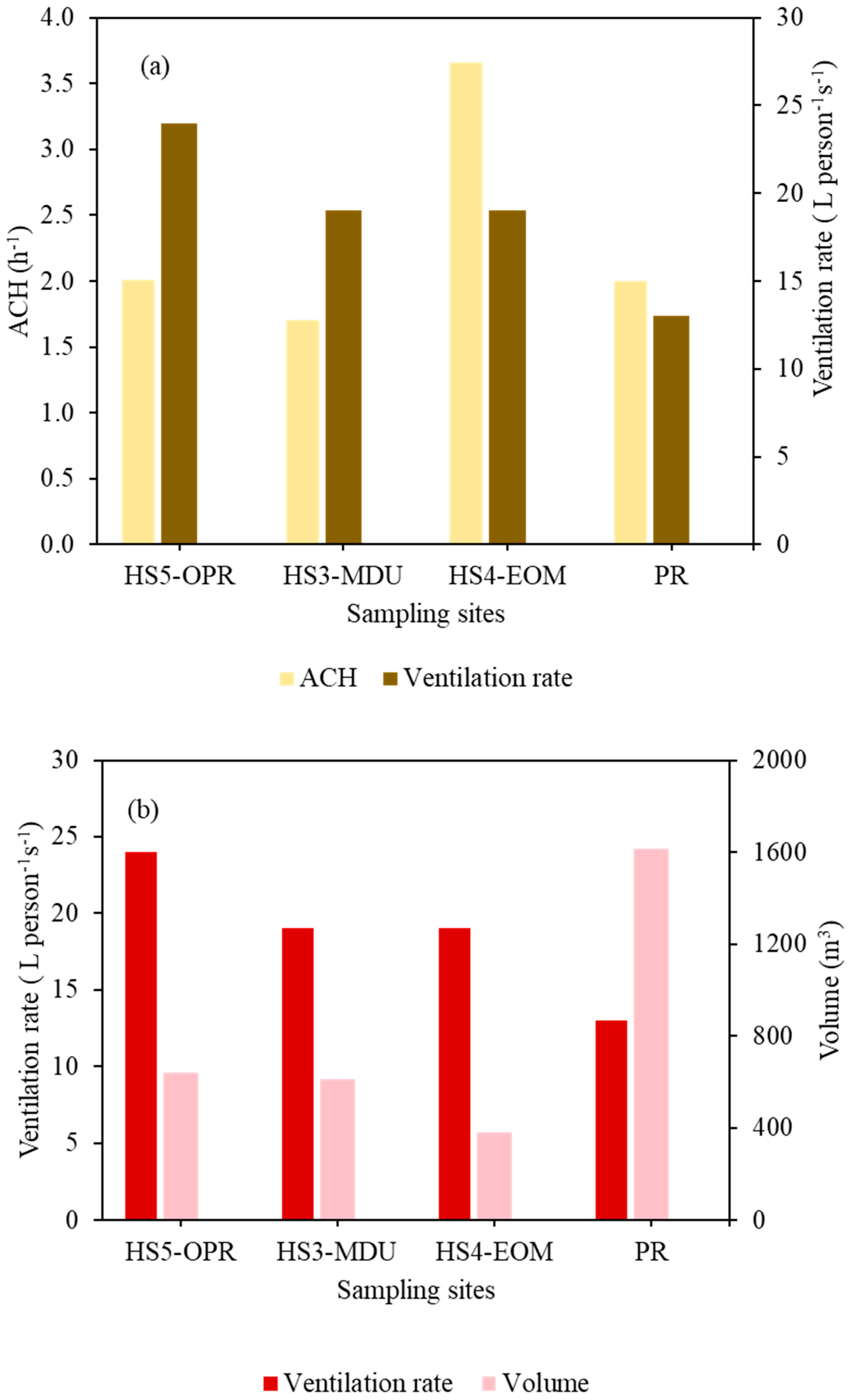
3.3. Ventilation Conditions
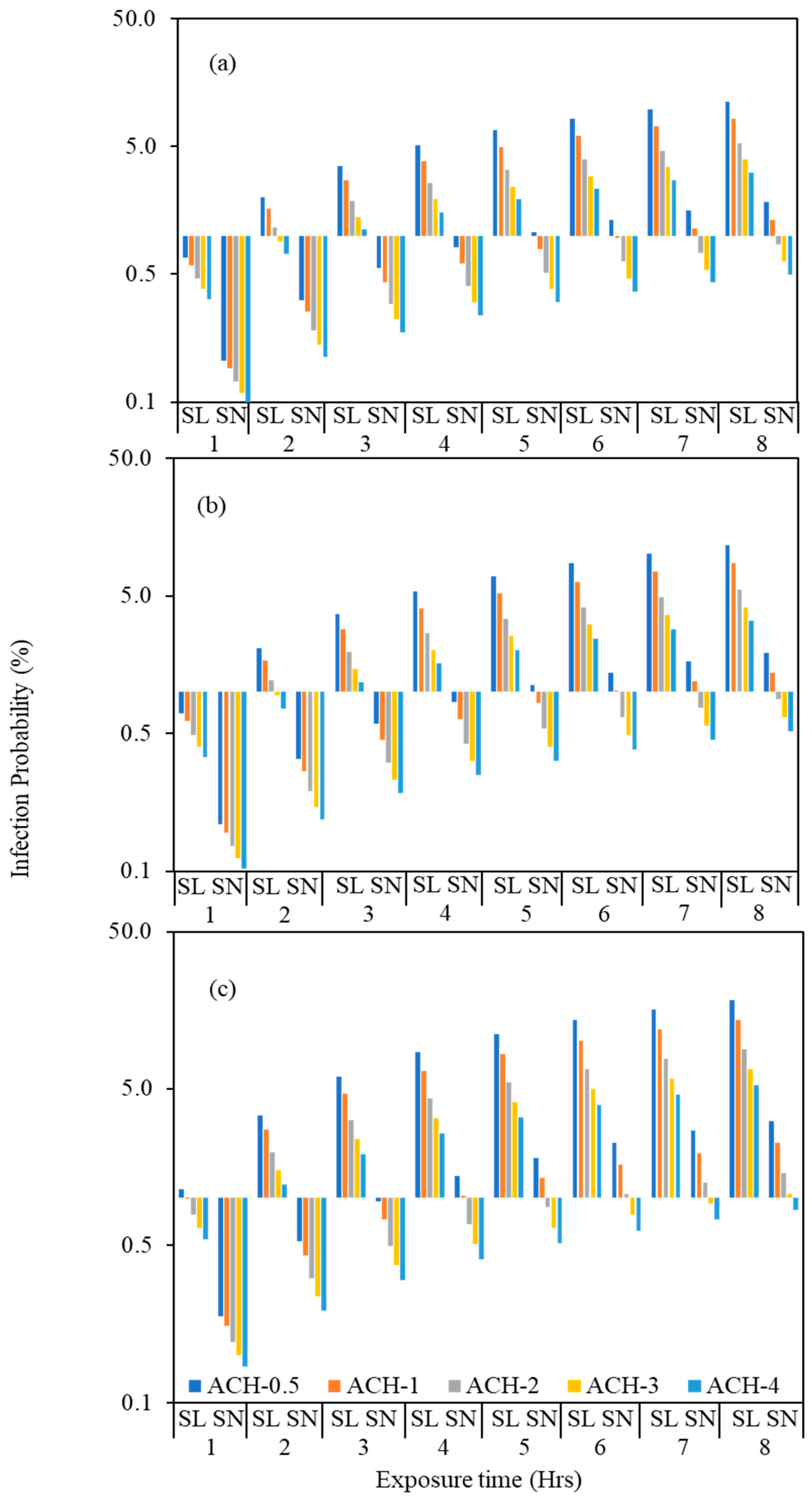
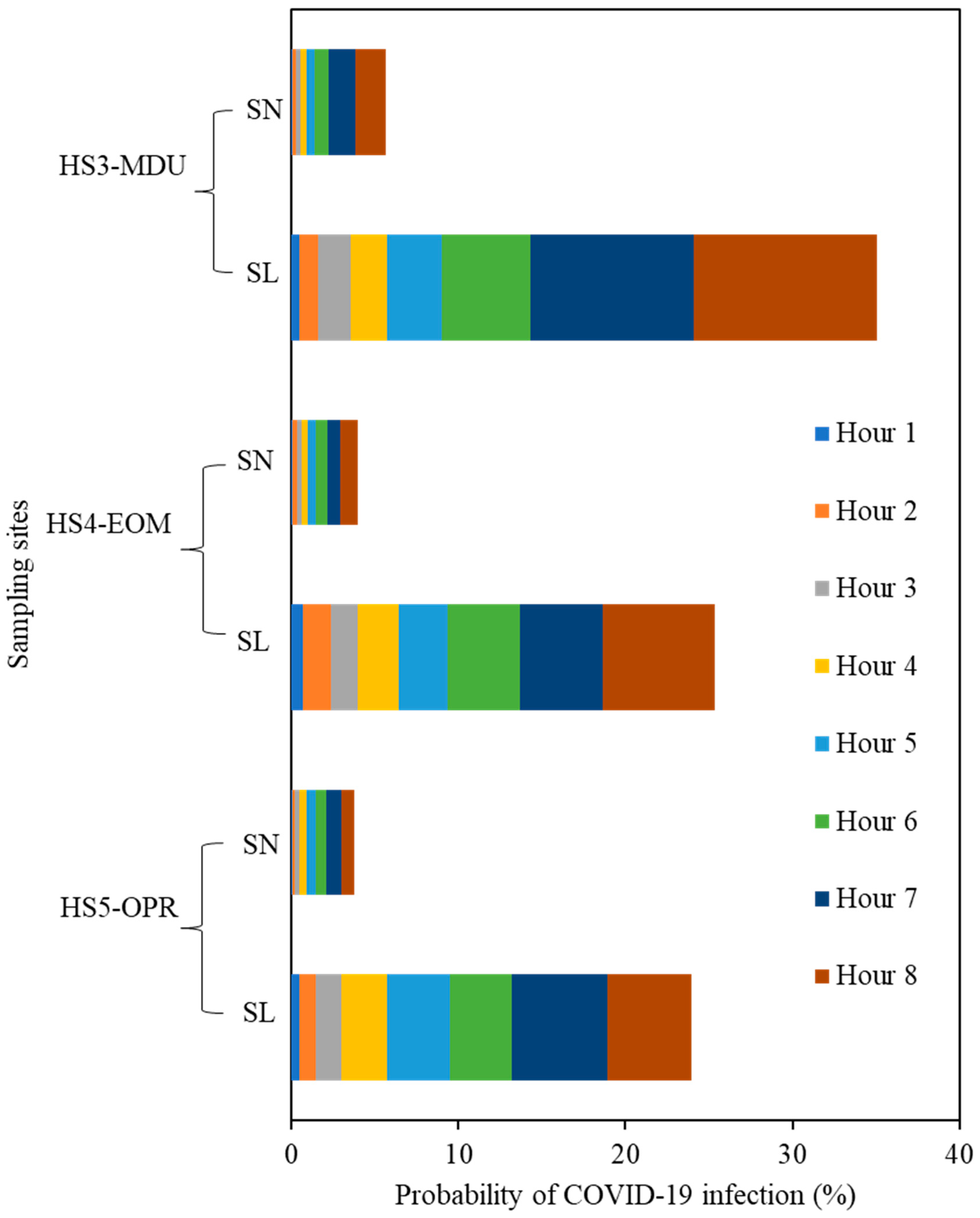
3.4. Estimation of COVID Infection Probability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lednicky, J.A.; Lauzard, M.; Fan, Z.H.; Jutla, A.; Tilly, T.B.; Gangwar, M.; Usmani, M.; Shankar, S.N.; Mohamed, K.; Eiguren-Fernandez, A.; et al. Viable SARS-CoV-2 in the air of a hospital room with COVID-19 patients. Int. J. Infect. Dis. 2020, 100, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus–Infected Pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Tang, H. Comparison of COVID-19 infection risks through aerosol transmission in supermarkets and small shops. Sustain. Cities Soc. 2022, 76, 103424. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.T.; Romero, P.; Valero-Amaro, V.; Arranz, J.I.; Montero, I. Ventilation conditions and their influence on thermal comfort in examination classrooms in times of COVID-19. A case study in a Spanish area with Mediterranean climate. Int. J. Hyg. Environ. Health 2022, 240, 113910. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Omidvarborna, H.; Tiwari, A.; Morawska, L. The nexus between in-car aerosol concentrations, ventilation and the risk of respiratory infection. Environ. Int. 2021, 157, 106814. [Google Scholar] [CrossRef]
- Stabile, L.; Buonanno, G.; Frattolillo, A.; Dell’Isola, M. The effect of the ventilation retrofit in a school on CO2, airborne particles, and energy consumptions. Build. Environ. 2019, 156, 1–11. [Google Scholar] [CrossRef]
- Di Gilio, A.; Palmisani, J.; Pulimeno, M.; Cerino, F.; Cacace, M.; Miani, A.; de Gennaro, G. CO2 concentration monitoring inside educational buildings as a strategic tool to reduce the risk of SARS-CoV-2 airborne transmission. Environ. Res. 2021, 202, 111560. [Google Scholar] [CrossRef]
- Villanueva, F.; Notario, A.; Cabañas, B.; Martín, P.; Salgado, S.; Gabriel, M.F. Assessment of CO2 and aerosol (PM2.5, PM10, UFP) concentrations during the reopening of schools in the COVID-19 pandemic: The case of a metropolitan area in Central-Southern Spain. Environ. Res. 2021, 197, 111092. [Google Scholar] [CrossRef]
- Schade, W.; Reimer, V.; Seipenbusch, M.; Willer, U. Experimental investigation of aerosol and CO2 dispersion for evaluation of COVID-19 infection risk in a concert hall. Int. J. Environ. Res. Public Health 2021, 18, 3037. [Google Scholar] [CrossRef]
- Bazant, M.Z.; Kodio, O.; Cohen, A.E.; Khan, K.; Gu, Z.; Bush, J.W.M. Monitoring carbon dioxide to quantify the risk of indoor airborne transmission of COVID-19. Flow 2021, 1, 1–18. [Google Scholar] [CrossRef]
- Deol, A.K.; Scarponi, D.; Beckwith, P.; Yates, T.A.; Karat, A.S.; Yan, A.W.C.; Baisley, K.S.; Grant, A.D.; White, R.G.; McCreesh, N. Estimating ventilation rates in rooms with varying occupancy levels: Relevance for reducing transmission risk of airborne pathogens. PLoS ONE 2021, 16, e0253096. [Google Scholar] [CrossRef] [PubMed]
- Pavilonis, B.; Ierardi, A.M.; Levine, L.; Mirer, F.; Kelvin, E.A. Estimating aerosol transmission risk of SARS-CoV-2 in New York City public schools during reopening. Environ. Res. 2021, 195, 110805. [Google Scholar] [CrossRef] [PubMed]
- Holshue, M.L.; DeBolt, C.; Lindquist, S.; Lofy, K.H.; Wiesman, J.; Bruce, H.; Spitters, C.; Ericson, K.; Wilkerson, S.; Tural, A.; et al. First Case of 2019 Novel Coronavirus in the United States. N. Engl. J. Med. 2020, 382, 929–936. [Google Scholar] [CrossRef]
- Khanna, C.R.; Cicinelli, M.V.; Gilbert, S.S.; Honavar, G.S.; Gudlavalleti, V.S.M. COVID-19 pandemic: Lessons learned and future directions. Indian J. Ophthalmol. 2020, 68, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Al Huraimel, K.; Alhosani, M.; Kunhabdulla, S.; Stietiya, M.H. SARS-CoV-2 in the environment: Modes of transmission, early detection and potential role of pollutions. Sci. Total Environ. 2020, 744, 140946. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Hama, S.; Omidvarborna, H.; Sharma, A.; Sahani, J.; Abhijith, K.V.; Debele, S.E.; Zavala-Reyes, J.C.; Barwise, Y.; Tiwari, A. Temporary reduction in fine particulate matter due to ‘anthropogenic emissions switch-off’ during COVID-19 lockdown in Indian cities. Sustain. Cities Soc. 2020, 62, 102382. [Google Scholar] [CrossRef]
- Enyoh, C.E.; Verla, A.W.; Qingyue, W.; Yadav, D.K.; Hossain Chowdhury, M.; Isiuku, B.O.; Chowdhury, T.; Ibe, F.C.; Verla, E.N.; Maduka, T.O. Indirect Exposure to Novel Coronavirus (SARS-CoV-2): An Overview of Current Knowledge. J. Teknol. Lab. 2020, 9, 67–77. [Google Scholar] [CrossRef]
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmann, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020, 104, 246–251. [Google Scholar] [CrossRef]
- Setti, L.; Passarini, F.; De Gennaro, G.; Barbieri, P.; Perrone, M.G.; Borelli, M.; Palmisani, J.; Di Gilio, A.; Piscitelli, P.; Miani, A. Airborne transmission route of COVID-19: Why 2 meters/6 feet of interpersonal distance could not be enough. Int. J. Environ. Res. Public Health 2020, 17, 2932. [Google Scholar] [CrossRef]
- Jayaweera, M.; Perera, H.; Gunawardana, B.; Manatunge, J. Transmission of COVID-19 virus by droplets and aerosols: A critical review on the unresolved dichotomy. Environ. Res. 2020, 188, 109819. [Google Scholar] [CrossRef]
- Morawska, L.; Cao, J. Airborne transmission of SARS-CoV-2: The world should face the reality. Environ. Int. 2020, 139, 105730. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. J. Am. Med. Assoc. 2020, 323, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Chia, P.Y.; Coleman, K.K.; Tan, Y.K.; Ong, S.W.X.; Gum, M.; Lau, S.K.; Lim, X.F.; Lim, A.S.; Sutjipto, S.; Lee, P.H.; et al. Detection of air and surface contamination by SARS-CoV-2 in hospital rooms of infected patients. Nat. Commun. 2020, 11, 2800. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.J.; Otter, J.A.; Price, J.R.; Cimpeanu, C.; Garcia, M.; Kinross, J.; Boshier, P.R.; Mason, S.; Bolt, F.; Alison, H.; et al. Investigating SARS-CoV-2 surface and air contamination in an acute healthcare setting during the peak of the COVID-19 pandemic in London. medRxiv 2020. [Google Scholar] [CrossRef]
- Bulfone, T.C.; Malekinejad, M.; Rutherford, G.W.; Razani, N. Outdoor Transmission of SARS-CoV-2 and Other Respiratory Viruses: A Systematic Review. J. Infect. Dis. 2021, 223, 550–561. [Google Scholar] [CrossRef]
- Stabile, L.; Pacitto, A.; Mikszewski, A.; Morawska, L.; Buonanno, G. Ventilation procedures to minimize the airborne transmission of viruses in classrooms. Build. Environ. 2021, 202, 108042. [Google Scholar] [CrossRef]
- Rodríguez, M.; Llanos Palop, M.; Seseña, S.; Rodríguez, A. Are the Portable Air Cleaners (PAC) really effective to terminate airborne SARS-CoV-2? Sci. Total Environ. 2021, 785, 147300. [Google Scholar] [CrossRef]
- Zhu, S.; Jenkins, S.; Addo, K.; Heidarinejad, M.; Romo, S.A.; Layne, A.; Ehizibolo, J.; Dalgo, D.; Mattise, N.W.; Hong, F.; et al. Ventilation and laboratory confirmed acute respiratory infection (ARI) rates in college residence halls in College Park, Maryland. Environ. Int. 2020, 137, 105537. [Google Scholar] [CrossRef]
- Kumar, P.; Rawat, N.; Tiwari, A. Micro-characteristics of a naturally ventilated classroom air quality under varying air purifier placements. Environ. Res. 2022, in press. [Google Scholar] [CrossRef]
- Nor, N.S.M.; Yip, C.W.; Ibrahim, N.; Jaafar, M.H.; Rashid, Z.Z.; Mustafa, N.; Hamid, A.H.H.; Chandru, K.; Latif, M.T.; Saw, P.E.; et al. Particulate matter (PM2.5) as a potential SARS-CoV-2 carrier. Sci. Rep. 2021, 11, 2508. [Google Scholar] [CrossRef]
- Ahlawat, A.; Wiedensohler, A.; Mishra, S.K. An overview on the role of relative humidity in airborne transmission of SARS-CoV-2 in indoor environments. Aerosol Air Qual. Res. 2020, 20, 1856–1861. [Google Scholar] [CrossRef]
- Bhagat, R.; Davies Wykes, M.; Dalziel, S.; Linden, P. Effects of ventilation on the indoor spread of COVID-19. J. Fluid Mech. 2020, 903, F1. [Google Scholar] [CrossRef] [PubMed]
- Woodward, H.; de Kreij, R.J.B.; Kruger, E.S.; Fan, S.; Tiwari, A.; Hama, S.; Noel, S.; Wykes, M.S.D.; Kumar, P.; Linden, P.F. An evaluation of the risk of airborne transmission of COVID-19 on an inter-city train carriage. Indoor Air 2022, 32, e13121. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Stabile, L.; Morawska, L. Estimation of airborne viral emission: Quanta emission rate of SARS-CoV-2 for infection risk assessment. Environ. Int. 2020, 141, 105794. [Google Scholar] [CrossRef]
- Miller, S.L.; Nazaroff, W.W.; Jimenez, J.L.; Boerstra, A.; Buonanno, G.; Dancer, S.J.; Kurnitski, J.; Marr, L.C.; Morawska, L.; Noakes, C. Transmission of SARS-CoV-2 by inhalation of respiratory aerosol in the Skagit Valley Chorale superspreading event. Indoor Air 2021, 31, 314–323. [Google Scholar] [CrossRef]
- Batterman, S. Review and extension of CO2-based methods to determine ventilation rates with application to school classrooms. Int. J. Environ. Res. Public Health 2017, 14, 145. [Google Scholar] [CrossRef]
- Kappelt, N.; Russell, H.S.; Kwiatkowski, S.; Afshari, A.; Johnson, M.S. Correlation of Respiratory Aerosols with Metabolic Carbon Dioxide. 2021. Available online: https://www.researchsquare.com/article/rs-490702/v1 (accessed on 20 July 2021).
- Pawar, S.; Stanam, A.; Chaudhari, M.; Rayudu, D. Effects of temperature on COVID-19 transmission. medRxiv 2020. [Google Scholar] [CrossRef]
- Smith, J.D.; Barratt, B.M.; Fuller, G.W.; Kelly, F.J.; Loxham, M.; Nicolosi, E.; Priestman, M.; Tremper, A.H.; Green, D.C. PM2.5 on the London Underground. Environ. Int. 2020, 134, 105188. [Google Scholar] [CrossRef] [PubMed]
- Mohammadyan, M.; Keyvani, S.; Bahrami, A.; Yetilmezsoy, K.; Heibati, B.; Pollitt, K.J.G. Assessment of indoor air pollution exposure in urban hospital microenvironments. Air Qual. Atmos. Health 2019, 12, 151–159. [Google Scholar] [CrossRef]
- Morawska, L.; Jamriska, M.; Guo, H.; Jayaratne, E.R.; Cao, M.; Summerville, S. Variation in indoor particle number and PM2.5 concentrations in a radio station surrounded by busy roads before and after an upgrade of the HVAC system. Build. Environ. 2009, 44, 76–84. [Google Scholar] [CrossRef]
- Zhao, X.; Zhou, W.; Han, L.; Locke, D. Spatiotemporal variation in PM2.5 concentrations and their relationship with socioeconomic factors in China’s major cities. Environ. Int. 2019, 133, 105145. [Google Scholar] [CrossRef] [PubMed]
- Noti, J.D.; Blachere, F.M.; McMillen, C.M.; Lindsley, W.G.; Kashon, M.L.; Slaughter, D.R.; Beezhold, D.H. High Humidity Leads to Loss of Infectious Influenza Virus from Simulated Coughs. PLoS ONE 2013, 8, e57485. [Google Scholar] [CrossRef] [PubMed]
- Nottmeyer, L.; Armstrong, B.; Lowe, R.; Abbott, S.; Meakin, S.; O’Reilly, K. The association of COVID-19 incidence with temperature, humidity, and UV radiation-A global multi-city analysis. Sci. Total Environ. 2022, 854, 158636. [Google Scholar] [CrossRef] [PubMed]
- Smither, S.J.; Eastaugh, L.S.; Findlay, J.S.; Lever, M.S. Experimental aerosol survival of SARS-CoV-2 in artificial saliva and tissue culture media at medium and high humidity. Emerg. Microbes Infect. 2020, 9, 1415–1417. [Google Scholar] [CrossRef]
- Thornton, G.M.; Fleck, B.A.; Dandnayak, D.; Kroeker, E.; Zhong, L.; Hartling, L. The impact of heating, ventilation, and air conditioning (HVAC) design features on the transmission of viruses, including the 2019 novel coronavirus (COVID-19): A systematic review of humidity. PLoS ONE 2022, 17, e0275654. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, L.; Murayama, H.; Hashizume, M. The impact of temperature on the transmissibility and virulence of COVID-19 in Tokyo, Japan. Sci. Rep. 2021, 11, 24477. [Google Scholar] [CrossRef]
- Mao, N.; Zhang, D.; Li, Y.; Li, Y.; Li, J.; Zhao, L. How do temperature, humidity, and air saturation state affect the COVID-19 transmission risk? Environ. Sci. Pollut. Res. 2022, 1–15. [Google Scholar] [CrossRef]
- Guo, L.; Yang, Z.; Zhang, L.; Wang, S.; Bai, T.; Xiang, Y.; Long, E. Systematic review of the effects of environmental factors on virus inactivation: Implications for coronavirus disease 2019. Int. J. Environ. Sci. Technol. 2021, 18, 2865–2878. [Google Scholar] [CrossRef]
- Aganovic, A.; Bi, Y.; Cao, G.; Drangsholt, F.; Kurnitski, J. Estimating the impact of indoor relative humidity on SARS-CoV-2 airborne transmission risk using a new modification of the Wells-Riley model. Build. Environ. 2021, 205, 108278. [Google Scholar] [CrossRef]
- Zivelonghi, A.; Lai, M. Mitigating aerosol infection risk in school buildings: The role of natural ventilation, volume, occupancy, and CO2 monitoring. Build. Environ. 2021, 204, 108139. [Google Scholar] [CrossRef]


| Type of Environment | Pollutants | Description | Reference |
|---|---|---|---|
| Supermarkets and small shops | CO2 | The average infection risk in supermarkets is higher than small shops (p-value < 0.001). Infection risks are higher for staff working with infected staff compared to customers. | [3] |
| Education centers | CO2 | The maximum CO2 concentration value recorded in one of the tests was 808 ppm, which is the limit for category IDA 2 buildings (including educational establishments), which was not exceeded. The report recommended that windows should be open when outside temperatures are mild. | [4] |
| In car | PM2.5 and CO2 | The probability of transmission from an infected person was found to be lower when the windows were open, the risk of infection increased 2-fold in a windows closed-Air conditioning scenario, and 10-fold in a windows closed-recirculation scenario. | [5] |
| Classrooms | CO2 | A mass balance approach was used to quantify the ability of both mechanical ventilation and ad hoc airing to mitigate airborne transmission. The mechanically ventilated classrooms required a control unit in order to check the air exchange rate and set the corresponding constant fresh flow rate. Whereas naturally ventilated classrooms needed to have manual airing cycles to increase the air exchange rate, which, in turn, reduced the probability of SARS-CoV-2 infection transmission. | [6] |
| Classrooms | CO2 | The CO2 monitoring in real-time helps to formulate tailored ventilation protocol to devise effective air exchanges and prevent SARS-CoV-2 transmission. | [7] |
| Educational buildings | CO2, PM2.5, PM10 and UFP | The preschool rooms registered better ventilation conditions, while secondary classrooms exhibited the highest peak and average CO2 concentrations. | [8] |
| Concert hall | di-ethylhexyl-sebacate aerosols (0.3 µm) (DEHS) and CO2 | The results show that a performer who is a potential emitter of aerosols with 0.3 µm diameter at 1.5 m distance would be carried up to the ceiling by the fresh air ventilation system with a vertical air flow of 0.05 m/s. Under these conditions, aerosol and CO2 concentrations did increase significantly in the concert hall. Audiences can wear facemasks to protect against longer range transport of small and large particles. | [9] |
| Lecture Halls | CO2 | CO2 in a well-mixed space acts as a passive scalar by tracking the ambient flow conditions and is removed only through exchange with outdoor air. The use of face masks reduces the ratio of aerosol-borne pathogen to CO2 concentration dramatically, and therefore reduces the risk of indoor transmission. | [10] |
| Primary health clinic | CO2 | Improved ventilation not only potentially reduces COVID-19 deaths, but also reduces the high numbers of deaths that occur from other airborne infectious diseases such as tuberculosis. | [11] |
| Classrooms | CO2 | Transmission probabilities are lower in older school buildings and lower-income neighborhoods due to the greater outdoor airflow associated with older, non-renovated buildings that are poorly insulated. | [12] |
| Sampling Site (Code) | Number of Samples (n) | Sampling Duration | Total Hours Sampled (h) | Type of Ventilation | Occupants | |
|---|---|---|---|---|---|---|
| Start Time | End Time | |||||
| Hospital number 1 Respiratory ward (HS1-RW) | 149 | 11:03:45 | 13:31:45 | 2.28 | NPV | 4 patients occupying in a ward with 6 beds |
| Hospital number 1 Intensive Care Unit (HS1-ICU) | 126 | 13:58:17 | 16:03:17 | 2.05 | NPV | NA |
| 1473 | 13:04:44 | 13:37:44 | 24.33 | NPV | NA | |
| 367 | 11:45:40 | 17:52:40 | 6:07 | NPV | NA | |
| Hospital number 1 Accident and Emergency ward (HS1-AER) | 401 | 09:30:57 | 16:11:57 | 6:41 | MV | Around 5–14 during (09:30 to 16:30 h) |
| Hospital number 2 Intensive Care Unit (HS2-ICU) | 335 | 11:28 | 17:03:01 | 5.35 | NPV | NA |
| School (SCH) | 1858 | 08:58:51 | 15:56:51 | 30.57 | MV | NA |
| Hospital 3 Medical Day Unit (HS3-MDU) | 1454 | 08:04:09 | 08:18:09 | 24.14 | MV | Around 12–15 during (08:30 to 12:30 h) |
| 1406 | 08:48:34 | 08:14:34 | 23:36 | MV | Around 12–15 during (08:30 to 12:30 h) | |
| 1452 | 09:30:06 | 09:42:06 | 24:12 | MV | Around 08–16 during (09:30 to 13:00 h) | |
| Hospital 4 Emergency/Outpatient Room (HS4-EOM) | 439 | 11:42:43 | 19:01:43 | 7.19 | MV | Around 15–20 during (09:00 to 18:00 h) |
| 520 | 09:42:09 | 18:22:09 | 8.40 | MV | Around 15–20 during (09:00 to 18:00 h) | |
| Research Institute (RI) | 4357 | 13:15:39 | 13:52:39 | 72:37 | MV | Around 10 during (09:00 to 17:00 h) |
| Hospital 5 Outpatient Ward (HS5-OPR) | 1397 | 08:42:43 | 07:59:43 | 23.20 | MV | Around 10–12 during (08:30 to 16:00 h) |
| 1402 | 09:05:51 | 08:27:51 | 23.38 | MV | Around 10–12 during (08:30 to 16:00 h) | |
| 1375 | 09:30:43 | 08:25:43 | 22:55 | MV | Around 3–13 during (09:30 to 17:00 h) | |
| 1441 | 09:35:48 | 09:36:48 | 24:01 | MV | Around 5–14 during (09:35 to 17:00 h) | |
| Pub/Restaurant (PR) | 254 | 18:46:33 | 23:00:33 | 4.14 | MV | Around 80 to 100 during (18:00 to 23:00 h) |
| Train station main concourse (TSM) | 497 | 09:31:45 | 17:49:45 | 8.18 | NV | Commuters to national rail and tube |
| 546 | 09:20:22 | 18:26:22 | 9:06 | NV | Commuters to national rail and tube | |
| 424 | 09:15:39 | 16:19:39 | 7:04 | NV | Commuters to national rail and tube | |
| Underground Site1 (UG-S1) | 453 | 10:15:51 | 17:48:51 | 7:33 | MV | Commuters to national rail and other tube stations |
| 556 | 09:15:10 | 18:31:10 | 9:16 | MV | Commuters to national rail and other tube stations | |
| Underground Site1 (UG-S2) | 440 | 09:50:07 | 17:10:07 | 7:20 | MV | Commuters to national rail and other tube station |
| 541 | 09:30:26 | 18:31:26 | 9:01 | NV | Commuters to national rail and other tube station | |
| Underground Site3 (UG-S3) | 603 | 08:00:08 | 18:03:08 | 10:03 | MV | Commuters to national rail and other tube station |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, P.; Kalaiarasan, G.; Bhagat, R.K.; Mumby, S.; Adcock, I.M.; Porter, A.E.; Ransome, E.; Abubakar-Waziri, H.; Bhavsar, P.; Shishodia, S.; et al. Active Air Monitoring for Understanding the Ventilation and Infection Risks of SARS-CoV-2 Transmission in Public Indoor Spaces. Atmosphere 2022, 13, 2067. https://doi.org/10.3390/atmos13122067
Kumar P, Kalaiarasan G, Bhagat RK, Mumby S, Adcock IM, Porter AE, Ransome E, Abubakar-Waziri H, Bhavsar P, Shishodia S, et al. Active Air Monitoring for Understanding the Ventilation and Infection Risks of SARS-CoV-2 Transmission in Public Indoor Spaces. Atmosphere. 2022; 13(12):2067. https://doi.org/10.3390/atmos13122067
Chicago/Turabian StyleKumar, Prashant, Gopinath Kalaiarasan, Rajesh K. Bhagat, Sharon Mumby, Ian M. Adcock, Alexandra E. Porter, Emma Ransome, Hisham Abubakar-Waziri, Pankaj Bhavsar, Swasti Shishodia, and et al. 2022. "Active Air Monitoring for Understanding the Ventilation and Infection Risks of SARS-CoV-2 Transmission in Public Indoor Spaces" Atmosphere 13, no. 12: 2067. https://doi.org/10.3390/atmos13122067
APA StyleKumar, P., Kalaiarasan, G., Bhagat, R. K., Mumby, S., Adcock, I. M., Porter, A. E., Ransome, E., Abubakar-Waziri, H., Bhavsar, P., Shishodia, S., Dilliway, C., Fang, F., Pain, C. C., & Chung, K. F. (2022). Active Air Monitoring for Understanding the Ventilation and Infection Risks of SARS-CoV-2 Transmission in Public Indoor Spaces. Atmosphere, 13(12), 2067. https://doi.org/10.3390/atmos13122067









