Listeria monocytogenes Isolated from Illegally Imported Food Products into the European Union Harbor Different Virulence Factor Variants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Listeria monocytogenes Isolates
2.2. Multilocus Sequence Typing and Pulsed-Field Gel Electrophoresis Typing
2.3. Sequencing and Analysis of Virulence Genes
2.4. Nucleotide Sequence Accession Number
3. Results and Discussion
3.1. Strain Characteristics and Genotype Diversity
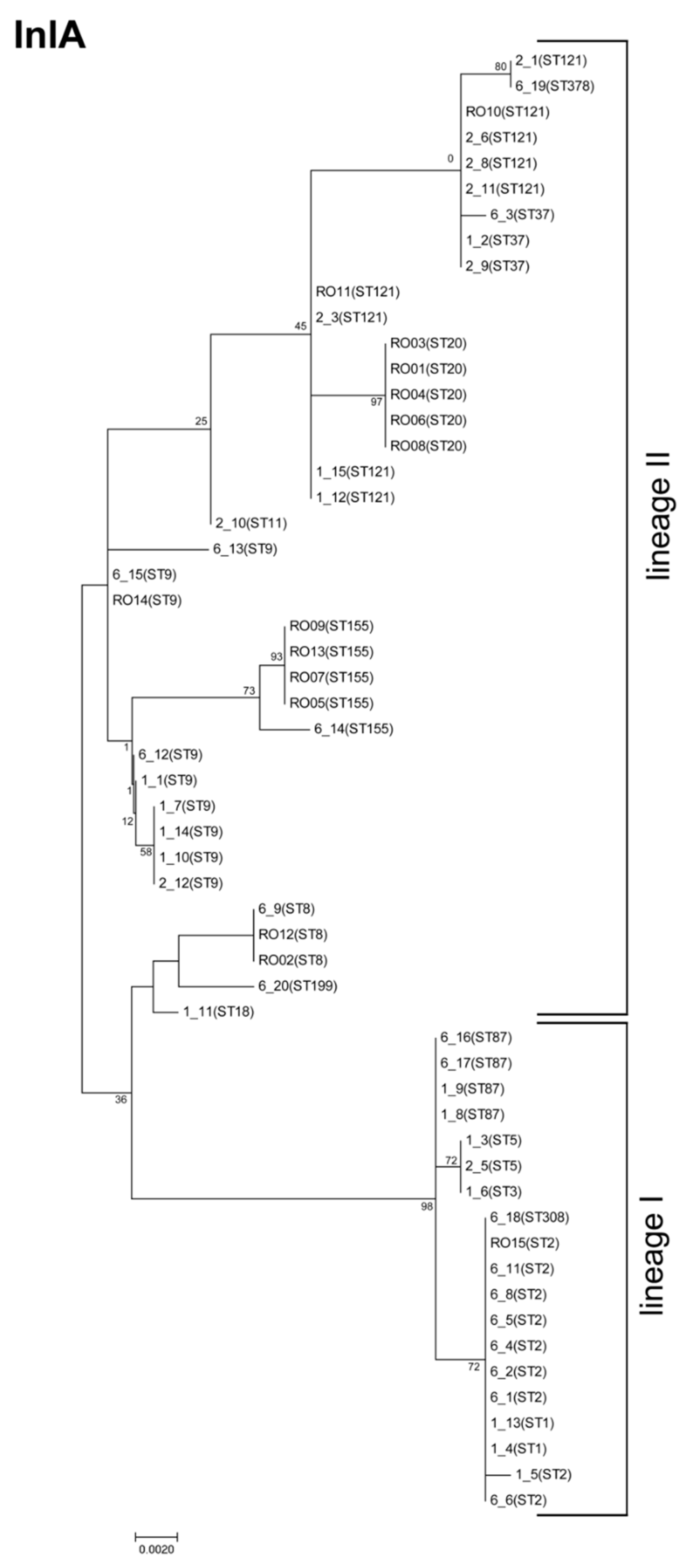
3.2. InlA Variants
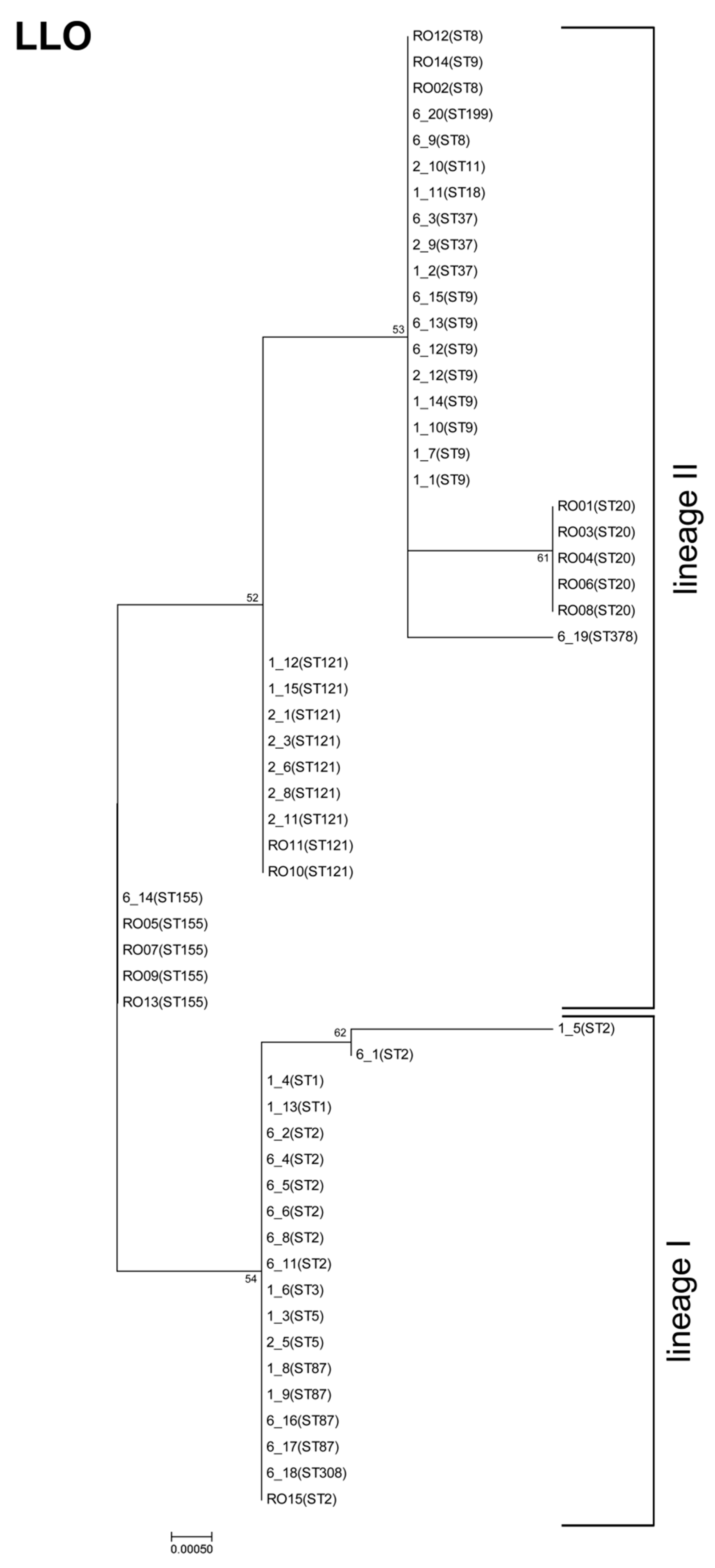
3.3. Listeriolysin O Variants
3.4. ActA Variants
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mangili, A.; Gendreau, M.A. Transmission of infectious diseases during commercial air travel. Lancet 2005, 365, 989–996. [Google Scholar] [CrossRef]
- Noordhuizen, J.; Surborg, H.; Smulders, F.J. On the efficacy of current biosecurity measures at EU borders to prevent the transfer of zoonotic and livestock diseases by travellers. Vet. Q. 2013, 33, 161–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kapperud, G.; Lassen, J.; Hasseltvedt, V. Salmonella infections in Norway: Descriptive epidemiology and a case-control study. Epidemiol. Infect. 1998, 121, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Nockler, K.; Wichmann-Schauer, H.; Hiller, P.; Muller, A.; Bogner, K. Trichinellosis outbreak in Bavaria caused by cured sausage from Romania, January 2007. Euro Surveill. 2007, 12, E070823. [Google Scholar] [CrossRef] [PubMed]
- Beutlich, J.; Hammerl, J.A.; Appel, B.; Nockler, K.; Helmuth, R.; Jost, K.; Ludwig, M.L.; Hanke, C.; Bechtold, D.; Mayer-Scholl, A. Characterization of illegal food items and identification of foodborne pathogens brought into the European Union via two major German airports. Int. J. Food Microbiol. 2015, 209, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Allerberger, F.; Wagner, M. Listeriosis: A resurgent foodborne infection. Clin. Microbiol. Infect. 2010, 16, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Cossart, P.; Toledo-Arana, A. Listeria monocytogenes, a unique model in infection biology: An overview. Microbes Infect. 2008, 10, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Boland, J.A.; Kuhn, M.; Berche, P.; Chakraborty, T.; Dominguez-Bernal, G.; Goebel, W.; Gonzalez-Zorn, B.; Wehland, J.; Kreft, J. Listeria pathogenesis and molecular virulence determinants. Clin. Microbiol. Rev. 2001, 14, 584–640. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority; European Centre for Disease Prevention and Control. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2015. EFSA J. 2016, 14, e04634. [Google Scholar] [CrossRef]
- Gandhi, M.; Chikindas, M.L. Listeria: A foodborne pathogen that knows how to survive. Int. J. Food Microbiol. 2007, 113, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Chaturongakul, S.; Raengpradub, S.; Wiedmann, M.; Boor, K.J. Modulation of stress and virulence in Listeria monocytogenes. Trends Microbiol. 2008, 16, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Schoder, D.; Strauss, A.; Szakmary-Brandle, K.; Stessl, B.; Schlager, S.; Wagner, M. Prevalence of major foodborne pathogens in food confiscated from air passenger luggage. Int. J. Food Microbiol. 2015, 209, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Lazaro, D.; Ariza-Miguel, J.; Diez-Valcarce, M.; Stessl, B.; Beutlich, J.; Fernandez-Natal, I.; Hernandez, M.; Wagner, M.; Rovira, J. Identification and molecular characterization of pathogenic bacteria in foods confiscated from non-EU flights passengers at one Spanish airport. Int. J. Food Microbiol. 2015, 209, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Ciolacu, L.; Stessl, B.; Bolocan, A.S.; Oniciuc, E.A.; Wagner, M.; Rychli, K.; Nicolau, A.I. Tracking foodborne pathogenic bacteria in raw and ready-to-eat food illegally sold at the eastern EU border. Foodborne Pathog. Dis. 2016, 13, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Bonazzi, M.; Lecuit, M.; Cossart, P. Listeria monocytogenes internalin and E-cadherin: From structure to pathogenesis. Cell. Microbiol. 2009, 11, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Schnupf, P.; Portnoy, D.A. Listeriolysin O: A phagosome-specific lysin. Microbes Infect. 2007, 9, 1176–1187. [Google Scholar] [CrossRef] [PubMed]
- Camejo, A.; Carvalho, F.; Reis, O.; Leitao, E.; Sousa, S.; Cabanes, D. The arsenal of virulence factors deployed by Listeria monocytogenes to promote its cell infection cycle. Virulence 2011, 2, 379–394. [Google Scholar] [CrossRef] [PubMed]
- Travier, L.; Guadagnini, S.; Gouin, E.; Dufour, A.; Chenal-Francisque, V.; Cossart, P.; Olivo-Marin, J.C.; Ghigo, J.M.; Disson, O.; Lecuit, M. ActA promotes Listeria monocytogenes aggregation, intestinal colonization and carriage. PLoS Pathog. 2013, 9, e1003131. [Google Scholar] [CrossRef] [PubMed]
- Ciolacu, L.; Nicolau, A.I.; Wagner, M.; Rychli, K. Listeria monocytogenes isolated from food samples from a Romanian black market show distinct virulence profiles. Int. J. Food Microbiol. 2015, 209, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Ragon, M.; Wirth, T.; Hollandt, F.; Lavenir, R.; Lecuit, M.; Le Monnier, A.; Brisse, S. A new perspective on Listeria monocytogenes evolution. PLoS Pathog. 2008, 4, e1000146. [Google Scholar] [CrossRef] [PubMed]
- Cantinelli, T.; Chenal-Francisque, V.; Diancourt, L.; Frezal, L.; Leclercq, A.; Wirth, T.; Lecuit, M.; Brisse, S. “Epidemic clones” of Listeria monocytogenes are widespread and ancient clonal groups. J. Clin. Microbiol. 2013, 51, 3770–3779. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Pulsenet International. 2013. Available online: http://www.pulsenetinternational.org/assets/PulseNet/uploads/pfge/PNL04_ListeriaPFGEProtocol.pdf (accessed on 15 July 2018).
- Tenover, F.C.; Arbeit, R.D.; Goering, R.V.; Mickelsen, P.A.; Murray, B.E.; Persing, D.H.; Swaminathan, B. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: Criteria for bacterial strain typing. J. Clin. Microbiol. 1995, 33, 2233–2239. [Google Scholar] [PubMed]
- Katoh, K.; Toh, H. Recent developments in the MAFFT multiple sequence alignment program. Brief. Bioinform. 2008, 9, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.T.; Taylor, W.R.; Thornton, J.M. The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci. 1992, 8, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, E.M.; Björkman, J.T.; Kiil, K.; Grant, K.; Dallman, T.; Painset, A.; Amar, C.; Roussel, S.; Guillier, L.; Félix, B.; et al. Closing gaps for performing a risk assessment on Listeria monocytogenes in ready-to-eat (RTE) foods: Activity 3, the comparison of isolates from different compartments along the food chain, and from humans using whole genome sequencing (WGS) analysis. EFSA Support. Publ. 2017, 14, 1151E. [Google Scholar] [CrossRef]
- Chenal-Francisque, V.; Lopez, J.; Cantinelli, T.; Caro, V.; Tran, C.; Leclercq, A.; Lecuit, M.; Brisse, S. Worldwide distribution of major clones of Listeria monocytogenes. Emerg. Infect. Dis. 2011, 17, 1110–1112. [Google Scholar] [CrossRef] [PubMed]
- Maury, M.M.; Tsai, Y.-H.; Charlier, C.; Touchon, M.; Chenal-Francisque, V.; Leclercq, A.; Criscuolo, A.; Gaultier, C.; Roussel, S.; Brisabois, A.; et al. Uncovering Listeria monocytogenes hypervirulence by harnessing its biodiversity. Nat. Genet. 2016, 48, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Moura, A.; Criscuolo, A.; Pouseele, H.; Maury, M.M.; Leclercq, A.; Tarr, C.; Bjorkman, J.T.; Dallman, T.; Reimer, A.; Enouf, V.; et al. Whole genome-based population biology and epidemiological surveillance of Listeria monocytogenes. Nat. Microbiol. 2016, 2, 16185. [Google Scholar] [CrossRef] [PubMed]
- Jensen, A.K.; Björkman, J.T.; Ethelberg, S.; Kiil, K.; Kemp, M.; Nielsen, E.M. Molecular typing and epidemiology of human listeriosis cases, Denmark, 2002–2012. Emerg. Infect. Dis. 2016, 22, 625–633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linke, K.; Ruckerl, I.; Brugger, K.; Karpiskova, R.; Walland, J.; Muri-Klinger, S.; Tichy, A.; Wagner, M.; Stessl, B. Reservoirs of Listeria species in three environmental ecosystems. Appl. Environ. Microbiol. 2014, 80, 5583–5592. [Google Scholar] [CrossRef] [PubMed]
- Amajoud, N.; Leclercq, A.; Soriano, J.M.; Bracq-Dieye, H.; El Maadoudi, M.; Senhaji, N.S.; Kounnoun, A.; Moura, A.; Lecuit, M.; Abrini, J. Prevalence of Listeria spp. and characterization of Listeria monocytogenes isolated from food products in Tetouan, Morocco. Food Control 2018, 84, 436–441. [Google Scholar] [CrossRef]
- Martin, B.; Perich, A.; Gomez, D.; Yanguela, J.; Rodriguez, A.; Garriga, M.; Aymerich, T. Diversity and distribution of Listeria monocytogenes in meat processing plants. Food Microbiol. 2014, 44, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Fagerlund, A.; Langsrud, S.; Schirmer, B.C.; Moretro, T.; Heir, E. Genome analysis of Listeria monocytogenes sequence type 8 strains persisting in salmon and poultry processing environments and comparison with related strains. PLoS ONE 2016, 11, e0151117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muhterem-Uyar, M.; Ciolacu, L.; Wagner, K.H.; Wagner, M.; Schmitz-Esser, S.; Stessl, B. New aspects on Listeria monocytogenes ST5-ECVI predominance in a heavily contaminated cheese processing environment. Front. Microbiol. 2018, 9, 64. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiao, Y.; Lan, R.; Xu, X.; Liu, G.; Wang, X.; Zhang, L.; Pang, H.; Jin, D.; Dai, H.; et al. Characterization of Listeria monocytogenes isolated from human listeriosis cases in China. Emerg. Microbes Infect. 2015, 4, e50. [Google Scholar] [CrossRef] [PubMed]
- Halbedel, S.; Prager, R.; Fuchs, S.; Trost, E.; Werner, G.; Flieger, A. Whole genome sequencing of recent Listeria monocytogenes isolates from Germany reveals population structure and disease clusters. J. Clin. Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Ross, W.H.; Whiting, R.C.; Van Stelten, A.; Nightingale, K.K.; Wiedmann, M.; Scott, V.N. Variation in Listeria monocytogenes dose responses in relation to subtypes encoding a full-length or truncated internalin A. Appl. Environ. Microbiol. 2011, 77, 1171–1180. [Google Scholar] [CrossRef] [PubMed]
- Nightingale, K.K.; Ivy, R.A.; Ho, A.J.; Fortes, E.D.; Njaa, B.L.; Peters, R.M.; Wiedmann, M. inlA premature stop codons are common among Listeria monocytogenes isolates from foods and yield virulence-attenuated strains that confer protection against fully virulent strains. Appl. Environ. Microbiol. 2008, 74, 6570–6583. [Google Scholar] [CrossRef] [PubMed]
- Nightingale, K.K.; Windham, K.; Martin, K.E.; Yeung, M.; Wiedmann, M. Select Listeria monocytogenes subtypes commonly found in foods carry distinct nonsense mutations in inlA, leading to expression of truncated and secreted internalin A, and are associated with a reduced invasion phenotype for human intestinal epithelial cells. Appl. Environ. Microbiol. 2005, 71, 8764–8772. [Google Scholar] [CrossRef] [PubMed]
- Olier, M.; Pierre, F.; Rousseaux, S.; Lemaitre, J.P.; Rousset, A.; Piveteau, P.; Guzzo, J. Expression of truncated Internalin A is involved in impaired internalization of some Listeria monocytogenes isolates carried asymptomatically by humans. Infect. Immun. 2003, 71, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Rychli, K.; Wagner, E.M.; Ciolacu, L.; Zaiser, A.; Tasara, T.; Wagner, M.; Schmitz-Esser, S. Comparative genomics of human and non-human Listeria monocytogenes sequence type 121 strains. PLoS ONE 2017, 12, e0176857. [Google Scholar] [CrossRef] [PubMed]
- Schmitz-Esser, S.; Müller, A.; Stessl, B.; Wagner, M. Genomes of sequence type 121 Listeria monocytogenes strains harbor highly conserved plasmids and prophages. Front. Microbiol. 2015, 6, 380. [Google Scholar] [CrossRef] [PubMed]
- Ebner, R.; Stephan, R.; Althaus, D.; Brisse, S.; Maury, M.; Tasara, T. Phenotypic and genotypic characteristics of Listeria monocytogenes strains isolated during 2011–2014 from different food matrices in Switzerland. Food Control 2015, 57, 321–326. [Google Scholar] [CrossRef] [Green Version]
- Gelbicova, T.; Kolackova, I.; Pantucek, R.; Karpiskova, R. A novel mutation leading to a premature stop codon in inlA of Listeria monocytogenes isolated from neonatal listeriosis. New Microbiol. 2015, 38, 293–296. [Google Scholar] [PubMed]
- Rousseaux, S.; Olier, M.; Lemaitre, J.P.; Piveteau, P.; Guzzo, J. Use of PCR-restriction fragment length polymorphism of inlA for rapid screening of Listeria monocytogenes strains deficient in the ability to invade Caco-2 cells. Appl. Environ. Microbiol. 2004, 70, 2180–2185. [Google Scholar] [CrossRef] [PubMed]
- Lecuit, M.; Ohayon, H.; Braun, L.; Mengaud, J.; Cossart, P. Internalin of Listeria monocytogenes with an intact leucine-rich repeat region is sufficient to promote internalization. Infect. Immun. 1997, 65, 5309–5319. [Google Scholar] [PubMed]
- Bierne, H.; Sabet, C.; Personnic, N.; Cossart, P. Internalins: A complex family of leucine-rich repeat-containing proteins in Listeria monocytogenes. Microbes Infect. 2007, 9, 1156–1166. [Google Scholar] [CrossRef] [PubMed]
- Gray, M.J.; Zadoks, R.N.; Fortes, E.D.; Dogan, B.; Cai, S.; Chen, Y.; Scott, V.N.; Gombas, D.E.; Boor, K.J.; Wiedmann, M. Listeria monocytogenes isolates from foods and humans form distinct but overlapping populations. Appl. Environ. Microbiol. 2004, 70, 5833–5841. [Google Scholar] [CrossRef] [PubMed]
- Lety, M.A.; Frehel, C.; Berche, P.; Charbit, A. Critical role of the N-terminal residues of listeriolysin O in phagosomal escape and virulence of Listeria monocytogenes. Mol. Microbiol. 2002, 46, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Lety, M.-A.; Frehel, C.; Dubail, I.; Beretti, J.-L.; Kayal, S.; Berche, P.; Charbit, A. Identification of a PEST-like motif in listeriolysin O required for phagosomal escape and for virulence in Listeria monocytogenes. Mol. Microbiol. 2001, 39, 1124–1139. [Google Scholar] [CrossRef] [PubMed]
- Schnupf, P.; Hofmann, J.; Norseen, J.; Glomski, I.J.; Schwartzstein, H.; Decatur, A.L. Regulated translation of listeriolysin O controls virulence of Listeria monocytogenes. Mol. Microbiol. 2006, 61, 999–1012. [Google Scholar] [CrossRef] [PubMed]
- Glomski, I.J.; Gedde, M.M.; Tsang, A.W.; Swanson, J.A.; Portnoy, D.A. The Listeria monocytogenes hemolysin has an acidic pH optimum to compartmentalize activity and prevent damage to infected host cells. J. Cell Biol. 2002, 156, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- Michel, E.; Reich, K.A.; Favier, R.; Berche, P.; Cossart, P. Attenuated mutants of the intracellular bacterium Listeria monocytogenes obtained by single amino-acid substitutions in Listeriolysin-O. Mol. Microbiol. 1990, 4, 2167–2178. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.; Rosenshine, I.; Cossart, P.; Finlay, B.B. Listeriolysin O activates mitogen-activated protein kinase in eucaryotic cells. Infect. Immun. 1996, 64, 2359–2361. [Google Scholar] [PubMed]
- Melton-Witt, J.A.; McKay, S.L.; Portnoy, D.A. Development of a single-gene, signature-tag-based approach in combination with alanine mutagenesis to identify listeriolysin O residues critical for the in vivo survival of Listeria monocytogenes. Infect. Immun. 2012, 80, 2221–2230. [Google Scholar] [CrossRef] [PubMed]
- Skoble, J.; Portnoy, D.A.; Welch, M.D. Three regions within ActA promote Arp2/3 complex-mediated actin nucleation and Listeria monocytogenes motility. J. Cell Biol. 2000, 150, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.A.; Theriot, J.A.; Portnoy, D.A. The tandem repeat domain in the Listeria monocytogenes ActA protein controls the rate of actin-based motility, the percentage of moving bacteria, and the localization of vasodilator-stimulated phosphoprotein and profilin. J. Cell Biol. 1996, 135, 647–660. [Google Scholar] [CrossRef] [PubMed]
- Lasa, I.; David, V.; Gouin, E.; Marchand, J.B.; Cossart, P. The amino-terminal part of ActA is critical for the actin-based motility of Listeria monocytogenes; the central proline-rich region acts as a stimulator. Mol. Microbiol. 1995, 18, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Pistor, S.; Chakraborty, T.; Walter, U.; Wehland, J. The bacterial actin nucleator protein ActA of Listeria monocytogenes contains multiple binding sites for host microfilament proteins. Curr. Biol. 1995, 5, 517–525. [Google Scholar] [CrossRef]
- Roberts, A.J.; Wiedmann, M. Allelic exchange and site-directed mutagenesis probe the contribution of ActA amino-acid variability to phosphorylation and virulence-associated phenotypes among Listeria monocytogenes strains. FEMS Microbiol. Lett. 2006, 254, 300–307. [Google Scholar] [CrossRef] [PubMed]

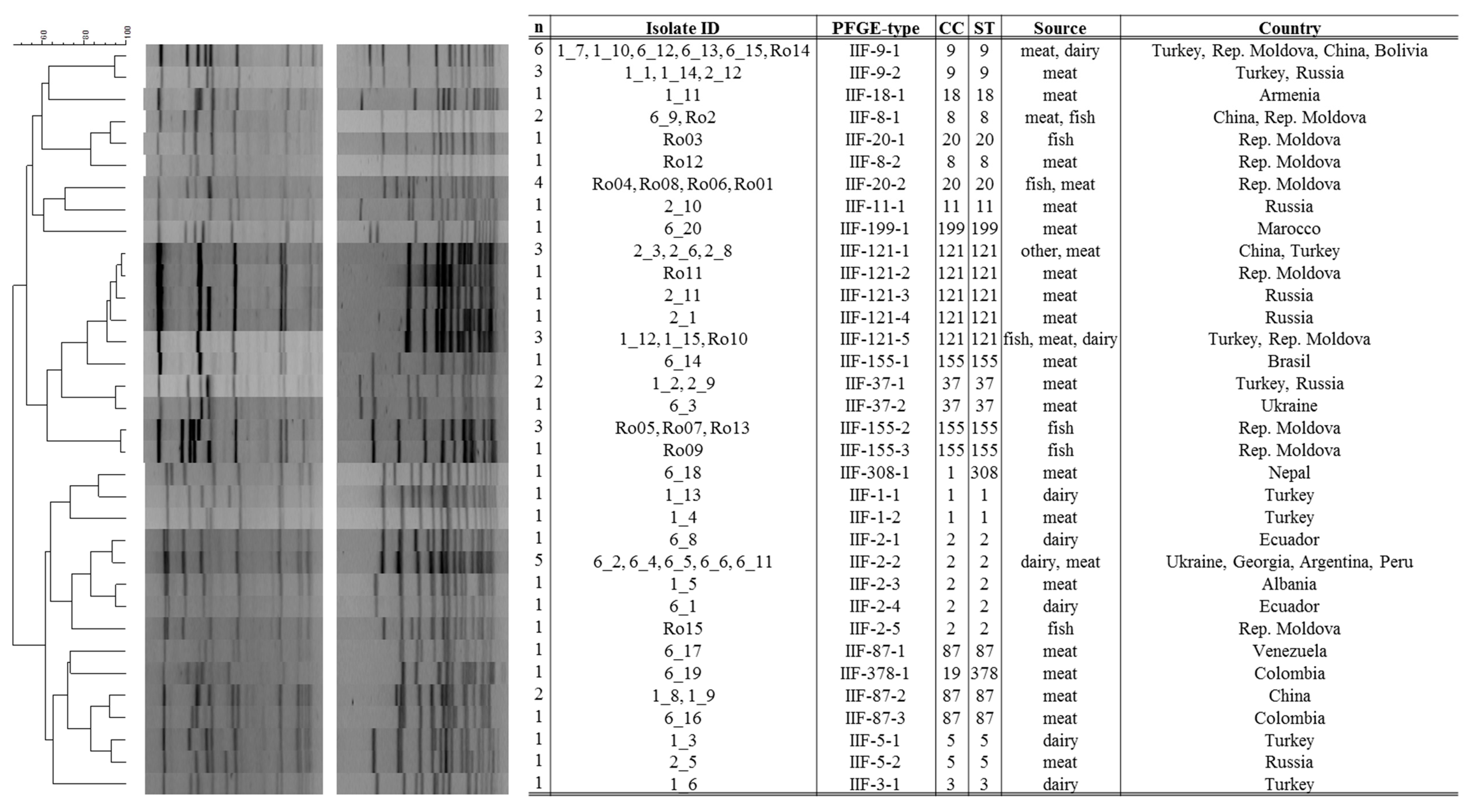



| Continent | Country | STs |
|---|---|---|
| Africa (n = 1) | Morocco (n = 1) | 199 (n = 1) |
| Asia (n = 28) | Armenia (n = 1) | 18 (n = 1) |
| China (n = 5) | 8, 9, 121 (n = 1); 87 (n = 2) | |
| Georgia (n = 1) | 2 (n = 1) | |
| Nepal (n = 1) | 308 (n = 1) | |
| Russia (n = 6) | 5, 9, 11, 37 (n = 1); 121 (n = 2) | |
| Turkey (n = 14) | 3, 5, 37 (n = 1); 1 (n = 2); 121 (n = 4); 9 (n = 5) | |
| Europe (n = 18) | Albania (n = 1) | 2 (n = 1) |
| Republic of Moldova (n = 15) | 2, 9 (n = 1); 8, 21 (n = 2); 155 (n = 4); 20 (n = 5) | |
| Ukraine (n = 2) | 2, 37 (n = 1) | |
| South America (n = 10) | Argentina (n = 1) | 2 (n = 1) |
| Bolivia (n = 1) | 9 (n = 1) | |
| Brazil (n = 1) | 155 (n = 1) | |
| Colombia (n = 2) | 87, 378 (n = 1) | |
| Ecuador (n = 3) | 2 (n = 3) | |
| Peru (n = 1) | 2 (n = 1) | |
| Venezuela (n = 1) | 87 (n = 1) |
| Food Source | STs |
|---|---|
| Dairy (n = 9) | 1, 3, 5, 9, 121 (n = 1) |
| 2 (n = 4) | |
| Fish (n = 11) | 2, 8, 121 (n = 1) |
| 20, 155 (n = 4) | |
| Meat (n = 36) | 1, 5, 11, 18, 20, 155, 199, 308, 378 (n = 1) |
| 8 (n = 2) | |
| 37 (n = 3) | |
| 2, 87 (n = 4) | |
| 121 (n = 6) | |
| 9 (n = 8) | |
| Other (n = 1) | 121 (n = 1) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rychli, K.; Stessl, B.; Szakmary-Brändle, K.; Strauß, A.; Wagner, M.; Schoder, D. Listeria monocytogenes Isolated from Illegally Imported Food Products into the European Union Harbor Different Virulence Factor Variants. Genes 2018, 9, 428. https://doi.org/10.3390/genes9090428
Rychli K, Stessl B, Szakmary-Brändle K, Strauß A, Wagner M, Schoder D. Listeria monocytogenes Isolated from Illegally Imported Food Products into the European Union Harbor Different Virulence Factor Variants. Genes. 2018; 9(9):428. https://doi.org/10.3390/genes9090428
Chicago/Turabian StyleRychli, Kathrin, Beatrix Stessl, Kati Szakmary-Brändle, Anja Strauß, Martin Wagner, and Dagmar Schoder. 2018. "Listeria monocytogenes Isolated from Illegally Imported Food Products into the European Union Harbor Different Virulence Factor Variants" Genes 9, no. 9: 428. https://doi.org/10.3390/genes9090428
APA StyleRychli, K., Stessl, B., Szakmary-Brändle, K., Strauß, A., Wagner, M., & Schoder, D. (2018). Listeria monocytogenes Isolated from Illegally Imported Food Products into the European Union Harbor Different Virulence Factor Variants. Genes, 9(9), 428. https://doi.org/10.3390/genes9090428







