Whole Genome Analyses Suggests that Burkholderia sensu lato Contains Two Additional Novel Genera (Mycetohabitans gen. nov., and Trinickia gen. nov.): Implications for the Evolution of Diazotrophy and Nodulation in the Burkholderiaceae
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Genomes
2.2. Phylogenetic Analysis
2.3. Average Nucleotide Identity and Average Amino Acid Identity
2.4. Genome-Informed Differential Characteristics
2.5. Analysis of nif and nod Genes
2.6. Plant Growth Promotion Analysis
2.7. Pathogenicity Tests
2.8. Bioinformatics Analysis of the T3SS
3. Results and Discussion
3.1. Whole-Genome Sequences
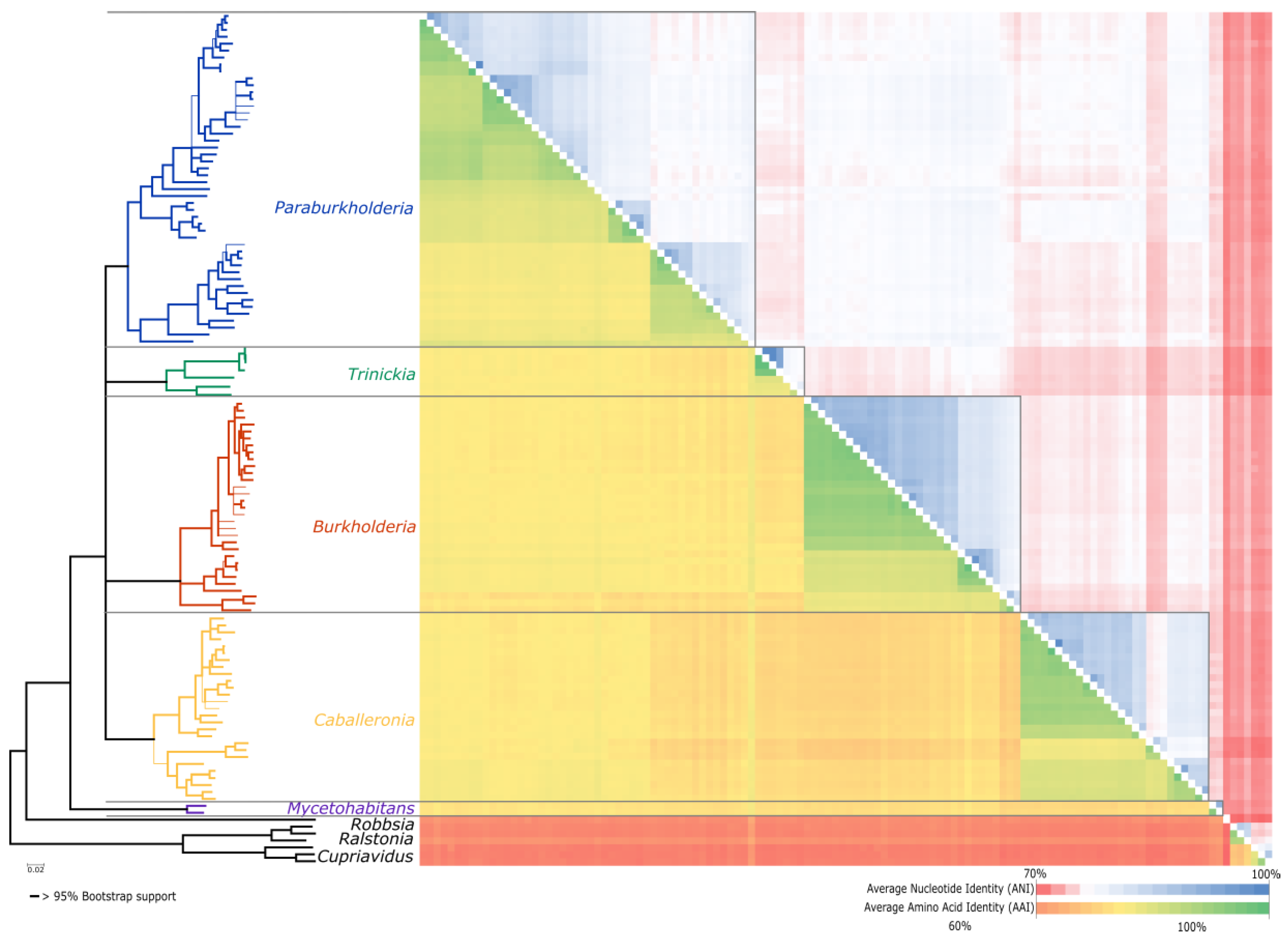
3.2. Phylogenetic Analysis
3.3. Average Nucleotide and Average Amino Acid Identity
3.4. Genome-Informed Differential Characteristics
3.5. nif and nod Gene Analysis
3.6. Nodulation and Plant Growth Promotion Features
3.7. Virulence Tests
3.8. Description of New Genera
3.8.1. Description of Mycetohabitans gen. nov.
- ○
- Mycetohabitans (My.ce.to.ha’bi.tans. Gr. n. mykês, etos, fungus; L. pres. part. habitans inhabiting; N.L. fem. n. Mycetohabitans inhabitant of fungi).
- Description of Mycetohabitans rhizoxinica
- Description of Mycetohabitans endofungorum
3.8.2. Description of Trinickia gen. nov.
- ○
- Trinickia (Tri.nick’i.a. N.L. fem. n. Trinickia formed after M.J. Trinick, an Australian microbiologist who was the first to isolate β-rhizobia from Mimosa).
- Description of Trinickia symbiotica comb. nov.
- Description of Trinickia caryophylli comb. nov.
- Description of Trinickia dabaoshanensis comb. nov.
- Description of Trinickia soli comb. nov.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Euzéby, J.P. List of bacterial names with standing in nomenclature: A folder available on the internet. Int. J. Syst. Evol. Microbiol. 1997, 47, 590–592. [Google Scholar] [CrossRef] [PubMed]
- Yabuuchi, E.; Kosako, Y.; Oyaizu, H.; Yano, I.; Hotta, H.; Hashimoto, Y.; Ezaki, T.; Arakawa, M. Proposal of Burkholderia gen. nov. and transfer of seven species of the genus Pseudomonas homology group ii to the new genus, with the type species Burkholderia cepacia (Palleroni and Holmes 1981) comb. nov. Microbiol. Immunol. 1992, 36, 1251–1275. [Google Scholar] [CrossRef] [PubMed]
- Estrada-de los Santos, P.; Vinuesa, P.; Martínez-Aguilar, L.; Hirsch, A.M.; Caballero-Mellado, J. Phylogenetic analysis of Burkholderia species by multilocus sequence analysis. Curr. Microbiol. 2013, 67, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Estrada-de los Santos, P.; Rojas-Rojas, F.U.; Tapia-García, E.Y.; Vásquez-Murrieta, M.S.; Hirsch, A.M. To split or not to split: An opinion on dividing the genus Burkholderia. Ann. Microbiol. 2016, 66, 1303–1314. [Google Scholar] [CrossRef]
- Gyaneshwar, P.; Hirsch, A.M.; Moulin, L.; Chen, W.-M.; Elliott, G.N.; Bontemps, C.; Estrada-de los Santos, P.; Gross, E.; dos Reis, F.B., Jr.; Sprent, J.I. Legume-nodulating β-proteobacteria: Diversity, host range, and future prospects. Mol. Plant Microbe Int. 2011, 24, 1276–1288. [Google Scholar] [CrossRef] [PubMed]
- Eberl, L.; Vandamme, P. Members of the genus Burkholderia: Good and bad guys. F1000Research 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Sawana, A.; Adeolu, M.; Gupta, R.S. Molecular signatures and phylogenomic analysis of the genus Burkholderia: Proposal for division of this genus into the emended genus Burkholderia containing pathogenic organisms and a new genus Paraburkholderia gen. nov. harboring environmental species. Front. Genet. 2014, 5, 429. [Google Scholar] [CrossRef] [PubMed]
- Dobritsa, A.P.; Samadpour, M. Transfer of eleven Burkholderia species to the genus Paraburkholderia and proposal of Caballeronia gen. nov., a new genus to accommodate twelve species of Burkholderia and Paraburkholderia. Int. J. Syst. Evol. Microbiol. 2016, 66, 2836–2846. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Santos, L.; Castro, D.B.A.; Ferreira-Tonin, M.; Corrêa, D.B.A.; Weir, B.S.; Park, D.; Ottoboni, L.M.M.; Neto, J.R.; Destéfano, S.A.L. Reassessment of the taxonomic position of Burkholderia andropogonis and description of Robbsia andropogonis gen. nov., comb. nov. Anton. Leeuw. Int. J. Gen. 2017, 110, 727–736. [Google Scholar] [CrossRef] [PubMed]
- De Lajudie, P.M.; Young, J.P.W. International committee on systematics of prokaryotes subcommittee for the taxonomy of Rhizobium and Agrobacterium minutes of the meeting, Budapest, 25 August 2016. Int. J. Syst. Evol. Microbiol. 2017, 67, 2485–2494. [Google Scholar] [CrossRef] [PubMed]
- Beukes, C.; Palmer, M.; Manyaka, P.; Chan, W.Y.; Avontuur, J.; van Zyl, E.; Huntemann, M.; Clum, A.; Pillay, M.; Palaniappan, K. Genome data provides high support for generic boundaries in Burkholderia sensu lato. Front. Microbiol. 2017, 8, 1154. [Google Scholar] [CrossRef] [PubMed]
- Partida-Martinez, L.P.; Groth, I.; Schmitt, I.; Richter, W.; Roth, M.; Hertweck, C. Burkholderia rhizoxinica sp. nov. and Burkholderia endofungorum sp. nov., bacterial endosymbionts of the plant-pathogenic fungus Rhizopus microsporus. Int. J. Syst. Evol. Microbiol. 2007, 57, 2583–2590. [Google Scholar] [CrossRef] [PubMed]
- Sheu, S.Y.; Chou, J.H.; Bontemps, C.; Elliott, G.N.; Gross, E.; James, E.K.; Sprent, J.I.; Young, J.P.W.; Chen, W.M. Burkholderia symbiotica sp. nov., isolated from root nodules of Mimosa spp. native to north-east Brazil. Int. J. Syst. Evol. Microbiol. 2012, 62, 2272–2278. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Guo, J.; Chen, M.; Feng, G.; Yao, Q. Burkholderia dabaoshanensis sp. nov., a heavy-metal-tolerant bacteria isolated from Dabaoshan mining area soil in China. PLoS ONE 2012, 7, e50225. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.-H.; Kim, B.-Y.; Weon, H.-Y.; Kwon, S.-W.; Go, S.-J.; Stackebrandt, E. Burkholderia soli sp. nov., isolated from soil cultivated with Korean ginseng. Int. J. Syst. Evol. Microbiol. 2007, 57, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Goris, J.; Konstantinidis, K.T.; Klappenbach, J.A.; Coenye, T.; Vandamme, P.; Tiedje, J.M. DNA–DNA hybridization values and their relationship to whole-genome sequence similarities. Int. J. Syst. Evol. Microbiol. 2007, 57, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidis, K.T.; Tiedje, J.M. Genomic insights that advance the species definition for prokaryotes. Proc. Natl. Am. Sci. USA 2005, 102, 2567–2572. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.; Rosselló-Móra, R. Shifting the genomic gold standard for the prokaryotic species definition. Proc. Natl. Am. Sci. USA 2009, 106, 19126–19131. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidis, K.T.; Tiedje, J.M. Towards a genome-based taxonomy for prokaryotes. J. Bacteriol. 2005, 187, 6258–6264. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidis, K.T.; Tiedje, J.M. Prokaryotic taxonomy and phylogeny in the genomic era: Advancements and challenges ahead. Curr. Opin Microbiol. 2007, 10, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-R, L.M.; Konstantinidis, K.T. Bypassing cultivation to identify bacterial species. Microbe Mag. 2014, 9, 111–118. [Google Scholar] [CrossRef]
- Suárez-Moreno, Z.R.; Caballero-Mellado, J.; Coutinho, B.G.; Mendonça-Previato, L.; James, E.K.; Venturi, V. Common features of environmental and potentially beneficial plant-associated Burkholderia. Microb. Ecol. 2012, 63, 249–266. [Google Scholar] [CrossRef] [PubMed]
- Bennett, S. Solexa Ltd. Pharmacogenomics 2004, 5, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Bushnell, B. Bbtools Software Package. Available online: http://sourceforge.net/projects/bbmap (accessed on 23 May 2018).
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D. Spades: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Seemann, T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef] [PubMed]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The rast server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Blom, J.; Kreis, J.; Spänig, S.; Juhre, T.; Bertelli, C.; Ernst, C.; Goesmann, A. Edgar 2.0: An enhanced software platform for comparative gene content analyses. Nucleic Acids Res. 2016, 44, W22–W28. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Muscle: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Abascal, F.; Zardoya, R.; Posada, D. Prottest: Selection of best-fit models of protein evolution. Bioinformatics 2005, 21, 2104–2105. [Google Scholar] [CrossRef] [PubMed]
- Kück, P.; Longo, G.C. FASconCAT-G: Extensive functions for multiple sequence alignment preparations concerning phylogenetic studies. Front. Zool. 2014, 11, 81. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Gevers, D.; Cohan, F.M.; Lawrence, J.G.; Spratt, B.G.; Coenye, T.; Feil, E.J.; Stackebrandt, E.; Van de Peer, Y.; Vandamme, P.; Thompson, F.L. Opinion: Re-evaluating prokaryotic species. Nat. Rev. Microbiol. 2005, 3, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S.; Kawashima, S.; Nakaya, A. The KEGG databases at GenomeNet. Nucleic Acids Res. 2002, 30, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.; Steenkamp, E.T.; Coetzee, M.P.A.; Blom, J.; Venter, S.N. Genome based characterization of biological processes that differentiate closely related bacteria. Front. Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar] [CrossRef]
- Edgar, R.C. Muscle: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinformatics 2004, 5, 113. [Google Scholar] [CrossRef] [PubMed]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. Modelfinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2014, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Paradis, E.; Claude, J.; Strimmer, K. Ape: Analyses of phylogenetics and evolution in R language. Bioinformatics 2004, 20, 289–290. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Smith, D.K.; Zhu, H.; Guan, Y.; Lam, T.T.Y. Ggtree: An R package for visualization and annotation of phylogenetic trees with their covariates and other associated data. Methods Ecol. Evol. 2017, 8, 28–36. [Google Scholar] [CrossRef]
- Caballero-Mellado, J.; Onofre-Lemus, J.; Estrada-de Los Santos, P.; Martínez-Aguilar, L. The tomato rhizosphere, an environment rich in nitrogen-fixing Burkholderia species with capabilities of interest for agriculture and bioremediation. Appl. Enviorn. Microbiol. 2007, 73, 5308–5319. [Google Scholar] [CrossRef] [PubMed]
- Estrada-de los Santos, P.; Bustillos-Cristales, R.; Caballero-Mellado, J. Burkholderia, a genus rich in plant-associated nitrogen fixers with wide environmental and geographic distribution. Appl. Enviorn. Microbiol. 2001, 67, 2790–2798. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.K.; Patriquin, D.G. Characterization of a substance produced by Azospirillum which causes branching of wheat root hairs. Can. J. Microbiol. 1985, 31, 206–210. [Google Scholar] [CrossRef]
- Elliott, G.N.; Chen, W.M.; Chou, J.H.; Wang, H.C.; Sheu, S.Y.; Perin, L.; Reis, V.M.; Moulin, L.; Simon, M.F.; Bontemps, C. Burkholderia phymatum is a highly effective nitrogen-fixing symbiont of Mimosa spp. and fixes nitrogen ex planta. New Phytol. 2007, 173, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.L.; Fasi, A.C.; Ramette, A.; Smith, J.J.; Hammerschmidt, R.; Sundin, G.W. Identification and onion pathogenicity of Burkholderia cepacia complex isolates from the onion rhizosphere and onion field soil. Appl. Environ. Microbiol. 2008, 74, 3121–3129. [Google Scholar] [CrossRef] [PubMed]
- Vílchez, J.I.; Navas, A.; González-López, J.; Arcos, S.C.; Manzanera, M. Biosafety test for plant growth-promoting bacteria: Proposed environmental and human safety index (EHSI) protocol. Front. Microbiol. 2016, 6, 1514. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.A. Accelerated evolution and Muller's rachet in endosymbiotic bacteria. Proc. Natl. Am. Sci. USA 1996, 93, 2873–2878. [Google Scholar] [CrossRef]
- Cardona, S.T.; Wopperer, J.; Eberl, L.; Valvano, M.A. Diverse pathogenicity of Burkholderia cepacia complex strains in the Caenorhabditis elegans host model. FEMS Microbiol. Lett. 2005, 250, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Mirarab, S.; Warnow, T. ASTRAL-II: Coalescent-based species tree estimation with many hundreds of taxa and thousands of genes. Bioinformatics 2015, 31, i44–i52. [Google Scholar] [CrossRef] [PubMed]
- Sayyari, E.; Mirarab, S. Fast coalescent-based computation of local branch support from quartet frequencies. Mol. Biol. Evol. 2016, 33, 1654–1668. [Google Scholar] [CrossRef] [PubMed]
- McCutcheon, J.P.; Moran, N.A. Extreme genome reduction in symbiotic bacteria. Nat. Rev. Microbiol. 2012, 10, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.A.; Wernegreen, J.J. Lifestyle evolution in symbiotic bacteria: Insights from genomics. Trends Ecol. Evol. 2000, 15, 321–326. [Google Scholar] [CrossRef]
- Gillis, M.; Van, T.V.; Bardin, R.; Goor, M.; Hebbar, P.; Willems, A.; Segers, P.; Kersters, K.; Heulin, T.; Fernandez, M.P. Polyphasic taxonomy in the genus Burkholderia leading to an emended description of the genus and proposition of Burkholderia vietnamiensis sp. nov. for N2-fixing isolates from rice in Vietnam. Int. J. Syst. Evolut. Microbiol. 1995, 45, 274–289. [Google Scholar] [CrossRef]
- Da Silva, K.; de Souza Cassetari, A.; Lima, A.S.; De Brandt, E.; Pinnock, E.; Vandamme, P.; de Souza Moreira, F.M. Diazotrophic Burkholderia species isolated from the amazon region exhibit phenotypical, functional and genetic diversity. Syst. Appl. Microbiol. 2012, 35, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Faoro, H.; Rene Menegazzo, R.; Battistoni, F.; Gyaneshwar, P.; do Amaral, F.P.; Taulé, C.; Rausch, S.; Gonçalves Galvão, P.; de los Santos, C.; Mitra, S. The oil-contaminated soil diazotroph Azoarcus olearius DQS-4T is genetically and phenotypically similar to the model grass endophyte Azoarcus sp. BH72. Environ. Microbiol. Rep. 2017, 9, 223–238. [Google Scholar] [CrossRef] [PubMed]
- De Meyer, S.E.; Briscoe, L.; Martínez-Hidalgo, P.; Agapakis, C.M.; Estrada de-los Santos, P.; Seshadri, R.; Reeve, W.; Weinstock, G.; O’Hara, G.; Howieson, J.G. Symbiotic Burkholderia species show diverse arrangements of nif/fix and nod genes and lack typical high-affinity cytochrome cbb3 oxidase genes. Mol. Plant Microbe Int. 2016, 29, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Sheu, S.-Y.; Chou, J.-H.; Bontemps, C.; Elliott, G.N.; Gross, E.; dos Reis Junior, F.B.; Melkonian, R.; Moulin, L.; James, E.K.; Sprent, J.I. Burkholderia diazotrophica sp. nov., isolated from root nodules of Mimosa spp. Int. J. Syst. Evol. Microbiol. 2013, 63, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Bournaud, C.; de Faria, S.M.; dos Santos, J.M.F.; Tisseyre, P.; Silva, M.; Chaintreuil, C.; Gross, E.; James, E.K.; Prin, Y.; Moulin, L. Burkholderia species are the most common and preferred nodulating symbionts of the piptadenia group (Tribe mimoseae). PLoS ONE 2013, 8, e63478. [Google Scholar] [CrossRef] [PubMed]
- Bournaud, C.; Moulin, L.; Cnockaert, M.; de Faria, S.; Prin, Y.; Severac, D.; Vandamme, P. Paraburkholderia piptadeniae sp. nov. and Paraburkholderia ribeironis sp. nov., two root-nodulating symbiotic species of Piptadenia gonoacantha in Brazil. Int. J. Syst. Evol. Microbiol. 2017, 67, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Dos Reis, F.B., Jr.; Simon, M.F.; Gross, E.; Boddey, R.M.; Elliott, G.N.; Neto, N.E.; de Fatima Loureiro, M.; de Queiroz, L.P.; Scotti, M.R.; Chen, W.M. Nodulation and nitrogen fixation by Mimosa spp. in the Cerrado and Caatinga biomes of Brazil. New Phytol. 2010, 186, 934–946. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.C.; Alves, P.C.; Rhem, M.F.K.; dos Santos, J.M.F.; James, E.K.; Gross, E. Brazilian species of Calliandra Benth. (tribe ingeae) are nodulated by diverse strains of Paraburkholderia. Syst. Appl. Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lemaire, B.; Chimphango, S.B.; Stirton, C.; Rafudeen, S.; Honnay, O.; Smets, E.; Chen, W.M.; Sprent, J.; James, E.K.; Muasya, A.M. Biogeographical patterns of legume-nodulating Burkholderia spp.: From African fynbos to continental scales. Appl. Environ. Microbiol. 2016, 82, 5099–5115. [Google Scholar] [CrossRef] [PubMed]
- Talbi, C.; Delgado, M.; Girard, L.; Ramirez-Trujillo, A.; Caballero-Mellado, J.; Bedmar, E. Burkholderia phymatum strains capable of nodulating Phaseolus vulgaris are present in Moroccan soils. Appl. Environ. Microbiol. 2010, 76, 4587–4591. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.Y.; Ridgway, H.J.; James, T.K.; James, E.K.; Chen, W.-M.; Sprent, J.I.; Young, J.P.W.; Andrews, M. Burkholderia sp. induces functional nodules on the south african invasive legume Dipogon lignosus (Phaseoleae) in New Zealand soils. Microb. Ecol. 2014, 68, 542–555. [Google Scholar] [CrossRef] [PubMed]
- Dall’Agnol, R.F.; Plotegher, F.; Souza, R.C.; Mendes, I.C.; dos Reis Junior, F.B.; Béna, G.; Moulin, L.; Hungria, M. Paraburkholderia nodosa is the main N2-fixing species trapped by promiscuous common bean (Phaseolus vulgaris L.) in the Brazilian ‘cerradão’. FEMS Microbiol. Ecol. 2016, 92, fiw108. [Google Scholar] [CrossRef] [PubMed]
- Lardi, M.; de Campos, S.B.; Purtschert, G.; Eberl, L.; Pessi, G. Competition experiments for legume infection identify Burkholderia phymatum as a highly competitive β-rhizobium. Front. Microbiol. 2017, 8, 1527. [Google Scholar] [CrossRef] [PubMed]
- Bontemps, C.; Elliott, G.N.; Simon, M.F.; Reis Júnior, F.B.; Gross, E.; Lawton, R.C.; Neto, N.E.; Fatima Loureiro, M.; Faria, S.M.; Sprent, J.I.; et al. Burkholderia species are ancient symbionts of legumes. Mol. Ecol. 2010, 19, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Moulin, L.; Klonowska, A.; Caroline, B.; Booth, K.; Vriezen, J.A.; Melkonian, R.; James, E.K.; Young, J.P.W.; Bena, G.; Hauser, L. Complete genome sequence of Burkholderia phymatum STM815T, a broad host range and efficient nitrogen-fixing symbiont of Mimosa species. Stand. Genom. Sci. 2014, 9, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Partida-Martinez, L.P.; Hertweck, C. Pathogenic fungus harbours endosymbiotic bacteria for toxin production. Nature 2005, 437, 884–888. [Google Scholar] [CrossRef] [PubMed]
- Angus, A.A.; Agapakis, C.M.; Fong, S.; Yerrapragada, S.; Estrada-de Los Santos, P.; Yang, P.; Song, N.; Kano, S.; Caballero-Mellado, J.; De Faria, S.M. Plant-associated symbiotic Burkholderia species lack hallmark strategies required in mammalian pathogenesis. PLoS ONE 2014, 9, e83779. [Google Scholar] [CrossRef] [PubMed]
- Hueck, C.J. Type III protein secretion systems in bacterial pathogens of animals and plants. Microbiol. Mol. Biol. Rev. 1998, 62, 379–433. [Google Scholar] [PubMed]
- Lackner, G.; Moebius, N.; Partida-Martínez, L.P.; Boland, S.; Hertweck, C. Evolution of an endofungal lyfestyle: Deductions from the Burkholderia rhizoxinica genome. BMC Genom. 2011, 12, 210. [Google Scholar] [CrossRef] [PubMed]



| Statistic | Paraburkholderia eburnea | Paraburkholderia rhynchosiae | Mycetohabitans endofungorum | Trinickia caryophylli | Trinickia caryophylli | Trinickia dabaoshanensis | Trinickia soli | Trinickia symbiotica | Trinickia symbiotica | Trinickia symbiotica | Paraburkholderia caribensis |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Strain | JCM 18070T | WSM3937T | HKI 456T | LMG 2155T = Ballard 720T | Ballard 720T = LMG 2155T | GIMN1.004T | GP25-8T | JPY 345T | JPY 581 | JPY 366 | TJ182 |
| Sequencing Centre | DOE Joint Genome Institute | MicrobesNG | DOE Joint Genome Institute | DOE Joint Genome Institute | MicrobesNG | MicrobesNG | MicrobesNG | DOE Joint Genome Institute | MicrobesNG | MicrobesNG | MicrobesNG |
| Sequencing Platform | Illumina HiSeq-2000 1TB | Illumina HiSeq 2500 | Illumina HiSeq-2000 1TB | Illumina HiSeq-2000 1TB | Illumina HiSeq 2500 | Illumina HiSeq 2500 | Illumina HiSeq 2500 | Illumina HiSeq-2000 1TB | Illumina HiSeq 2500 | Illumina HiSeq 2500 | Illumina HiSeq 2500 |
| NCBI taxonomy ID | 1,189,126 | 487,049 | 417,203 | 28,094 | 28,094 | 564,714 | 380,675 | 863,227 | 863,227 | 863,227 | 75,105 |
| NCBI BioProject ID | PRJNA369942 | PRJNA427925 | PRJNA370785 | PRJNA369920 | PRJNA427926 | PRJNA427927 | PRJNA427928 | PRJNA369937 | PRJNA427929 | PRJNA445642 | PRJNA445638 |
| Number of reads | 6,886,312 | 1,204,873 | 7,561,076 | 7,357,578 | 962,962 | 828,393 | 918,663 | 6,294,534 | 2,076,457 | 1,180,541 | 809,533 |
| Assembly method | SPAdes | SPAdes | SPAdes | SPAdes | SPAdes | SPAdes | SPAdes | SPAdes | SPAdes | SPAdes | SPAdes |
| Sequencing coverage | 149.1X | 58.8X | 348.7X | 169.1X | 56.4X | 47.6X | 61.6X | 149.1X | 131.3X | 66.2X | 35.9X |
| N50 | 294,829 | 226,289 | 213,816 | 480,986 | 187,187 | 186,667 | 231,363 | 252,951 | 255,942 | 387,494 | 89,490 |
| L50 | 7 | 12 | 6 | 6 | 13 | 13 | 10 | 9 | 9 | 7 | 31 |
| Largest contig [bp] | 983,800 | 527,307 | 365,500 | 792,225 | 401,224 | 433,345 | 514,473 | 819,300 | 663,178 | 786,277 | 294,652 |
| Number of contigs | 58 | 181 | 76 | 49 | 161 | 104 | 105 | 61 | 121 | 57 | 242 |
| Genome size [bp] | 6,947,977 | 8,032,361 | 3,288,408 | 6,543,652 | 6,581,896 | 7,093,755 | 6,096,514 | 6,714,023 | 6,753,015 | 7,005,740 | 9,206,228 |
| G+C content | 64.09% | 61.74% | 61.27% | 64.72% | 64.72% | 63.28% | 62.98% | 63.00% | 63.01% | 63.00% | 62.49% |
| Assembly Accession Number | GCA_002917095.1 | GCA_002879865.1 | GCA_002927045.1 | GCA_900177465.1 | GCA_002879875.1 | GCA_002879885.1 | GCA_002879855.1 | GCA_002934455.1 | GCA_002879935.1 | GCA_003028655.1 | GCA_003028645.1 |
| Feature | Plant Pathogen | N-Fixation | Chitinolytic Activity * | Predator Bacterium | Cell Type | Fungus Endosymbiont | Legume Nodulation | Obligately Endosymbiont | NO3 to NO2 | Growth at >60 °C | OL-1 | OL-2 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species | |||||||||||||
| Burkholderia cepacia J2315T | + | − ** | − | − | Rods | − | − | − | − | − | + | + | |
| Caballeronia glathei ATCC 29195T | − | + | − | − | Rods | − | − | − | nf | − | nd | nd | |
| Chitinimonas taiwanensis cfT | nd | − | + | − | Rods | − | − | − | + | − | nd | nd | |
| Cupriavidus necator N-1T | − | − ** | − | + | Short rods | − | − ** | − | + | − | − | − | |
| Lautropia mirabilis AB2188T | nd | − | − | − | Coccoid | − | − | − | + | − | nd | nd | |
| Limnobacter thiooxidans CS-K2T | nd | − | − | − | Rods | − | − | − | − | − | nd | nd | |
| Mycetohabitans rhizoxinica HKI 454T | + | − | − | − | Coccoid rods | + | − | − | nd | − | nd | nd | |
| Pandoraea apista LMG 16407T | nd | − | − | − | Rods | − | − | − | − | − | nd | nd | |
| Paraburkholderia graminis C4D1MT | − | − | − | − | Rods | − | − | − | + | − | nd | nd | |
| Paucimonas lemoignei A62T | nd | + | − | − | Rods | − | − | − | − | − | nd | nd | |
| Polynucleobacter necessarius ATCC 30859T | nd | − | − | − | Rods | − | − | + | nd | − | nd | nd | |
| Ralstonia pickettii ATCC 27511T | + | − | − | − | Rods | − | − | − | + | − | − | − | |
| Robbsia andropogonis LMG 2129T | + | − | − | − | Rods | − | − | − | − | − | − | − | |
| Thermothrix thiopara ATCC 29244T | nd | nd | − | nd | Rods | nd | nd | − | + | + | nd | nd | |
| Trinickia symbiotica JPY345T | − | + | − | − | Rods | − | + | − | + | − | − | − | |
| Strains | Rating for the Degree of Tissue Maceration after 72 h |
|---|---|
| Control | 0 |
| B. cepacia 68P128 | 3 (67–100% macerated tissue area) |
| M. rhizoxinica HKI 454T | 0 |
| T. symbiotica JPY 581 | 0 |
| T. symbiotica JPY 366 | 0 |
| T. symbiotica JPY 347 | 0 |
| T. caryophylli Ballard 720T | 2 (34–66% macerated tissue area) |
| T. soli GP25-8T | 0 |
| T. dabaoshanensis GIMN1004T | 0 |
| P. caballeronis LMG 26416T | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estrada-de los Santos, P.; Palmer, M.; Chávez-Ramírez, B.; Beukes, C.; Steenkamp, E.T.; Briscoe, L.; Khan, N.; Maluk, M.; Lafos, M.; Humm, E.; et al. Whole Genome Analyses Suggests that Burkholderia sensu lato Contains Two Additional Novel Genera (Mycetohabitans gen. nov., and Trinickia gen. nov.): Implications for the Evolution of Diazotrophy and Nodulation in the Burkholderiaceae. Genes 2018, 9, 389. https://doi.org/10.3390/genes9080389
Estrada-de los Santos P, Palmer M, Chávez-Ramírez B, Beukes C, Steenkamp ET, Briscoe L, Khan N, Maluk M, Lafos M, Humm E, et al. Whole Genome Analyses Suggests that Burkholderia sensu lato Contains Two Additional Novel Genera (Mycetohabitans gen. nov., and Trinickia gen. nov.): Implications for the Evolution of Diazotrophy and Nodulation in the Burkholderiaceae. Genes. 2018; 9(8):389. https://doi.org/10.3390/genes9080389
Chicago/Turabian StyleEstrada-de los Santos, Paulina, Marike Palmer, Belén Chávez-Ramírez, Chrizelle Beukes, Emma T. Steenkamp, Leah Briscoe, Noor Khan, Marta Maluk, Marcel Lafos, Ethan Humm, and et al. 2018. "Whole Genome Analyses Suggests that Burkholderia sensu lato Contains Two Additional Novel Genera (Mycetohabitans gen. nov., and Trinickia gen. nov.): Implications for the Evolution of Diazotrophy and Nodulation in the Burkholderiaceae" Genes 9, no. 8: 389. https://doi.org/10.3390/genes9080389
APA StyleEstrada-de los Santos, P., Palmer, M., Chávez-Ramírez, B., Beukes, C., Steenkamp, E. T., Briscoe, L., Khan, N., Maluk, M., Lafos, M., Humm, E., Arrabit, M., Crook, M., Gross, E., Simon, M. F., Dos Reis Junior, F. B., Whitman, W. B., Shapiro, N., Poole, P. S., Hirsch, A. M., ... James, E. K. (2018). Whole Genome Analyses Suggests that Burkholderia sensu lato Contains Two Additional Novel Genera (Mycetohabitans gen. nov., and Trinickia gen. nov.): Implications for the Evolution of Diazotrophy and Nodulation in the Burkholderiaceae. Genes, 9(8), 389. https://doi.org/10.3390/genes9080389







