MafA Expression Preserves Immune Homeostasis in Human and Mouse Islets
Abstract
1. Introduction
2. Materials and Methods
2.1. MAFA Co-Expression Correlations in RNA-Sequencing Data from Human Pancreatic Islets
2.2. Statistical Analysis on MAF Co-Expression Correlations
2.3. Animals
2.4. Mouse Islet Isolation
2.5. Coxsackievirus B3 Virus Islet Infection Assay
2.6. Assessment of Islet Cell Viability
2.7. RNA Extraction from Mouse Islets
2.8. Complementary DNA Synthesis and Quantitative PCR
2.9. Cell Culture, Small Interfering RNA Transfection, and Protein Analysis
2.10. Immunohistochemistry
3. Results
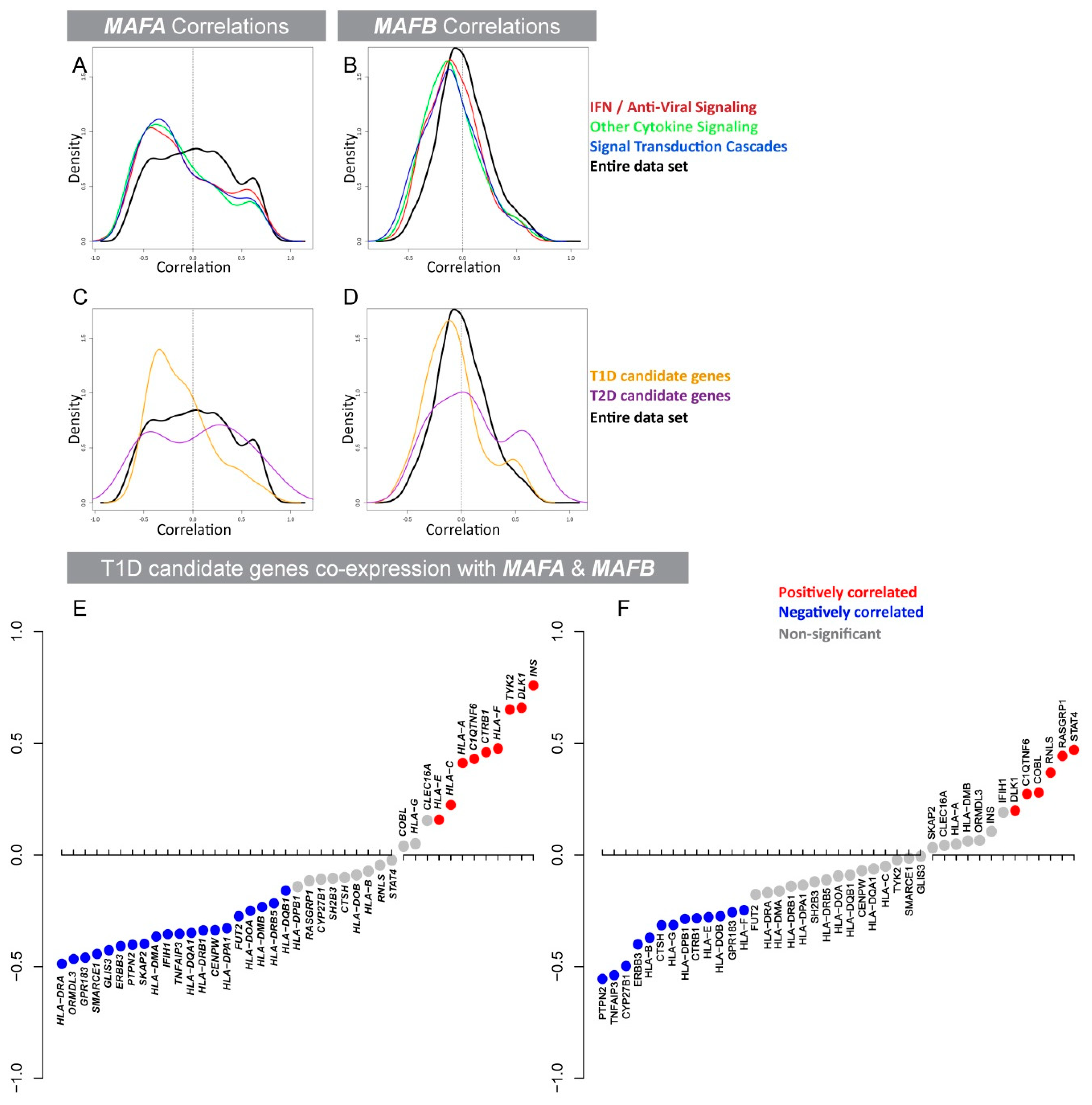
3.1. Expression of the β-Cell-Specific Transcription Factor MAFA Is Strongly Negatively Correlated with Pro-Inflammatory Cytokine-Induced Signaling Networks and T1D Susceptibility Genes
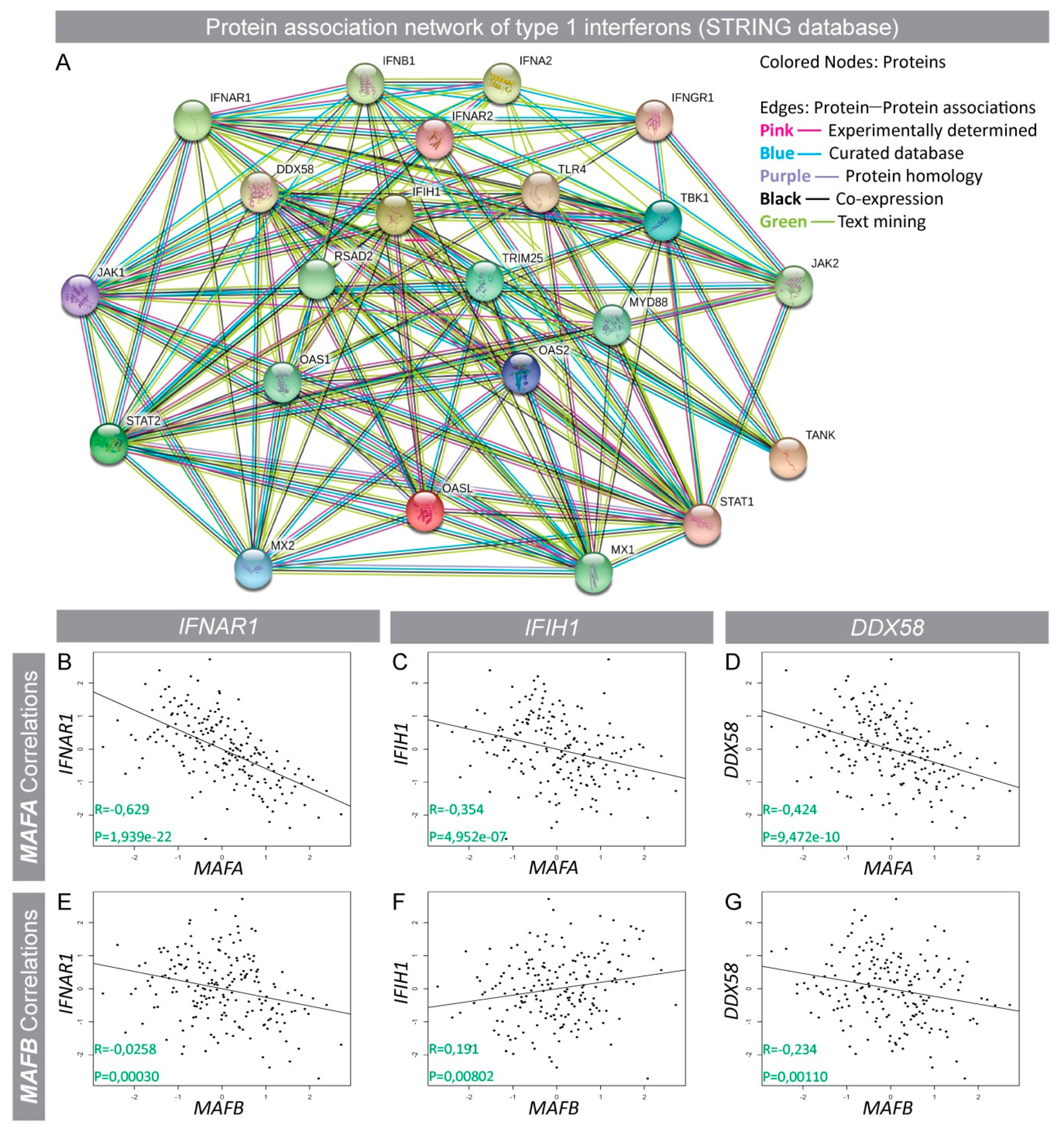
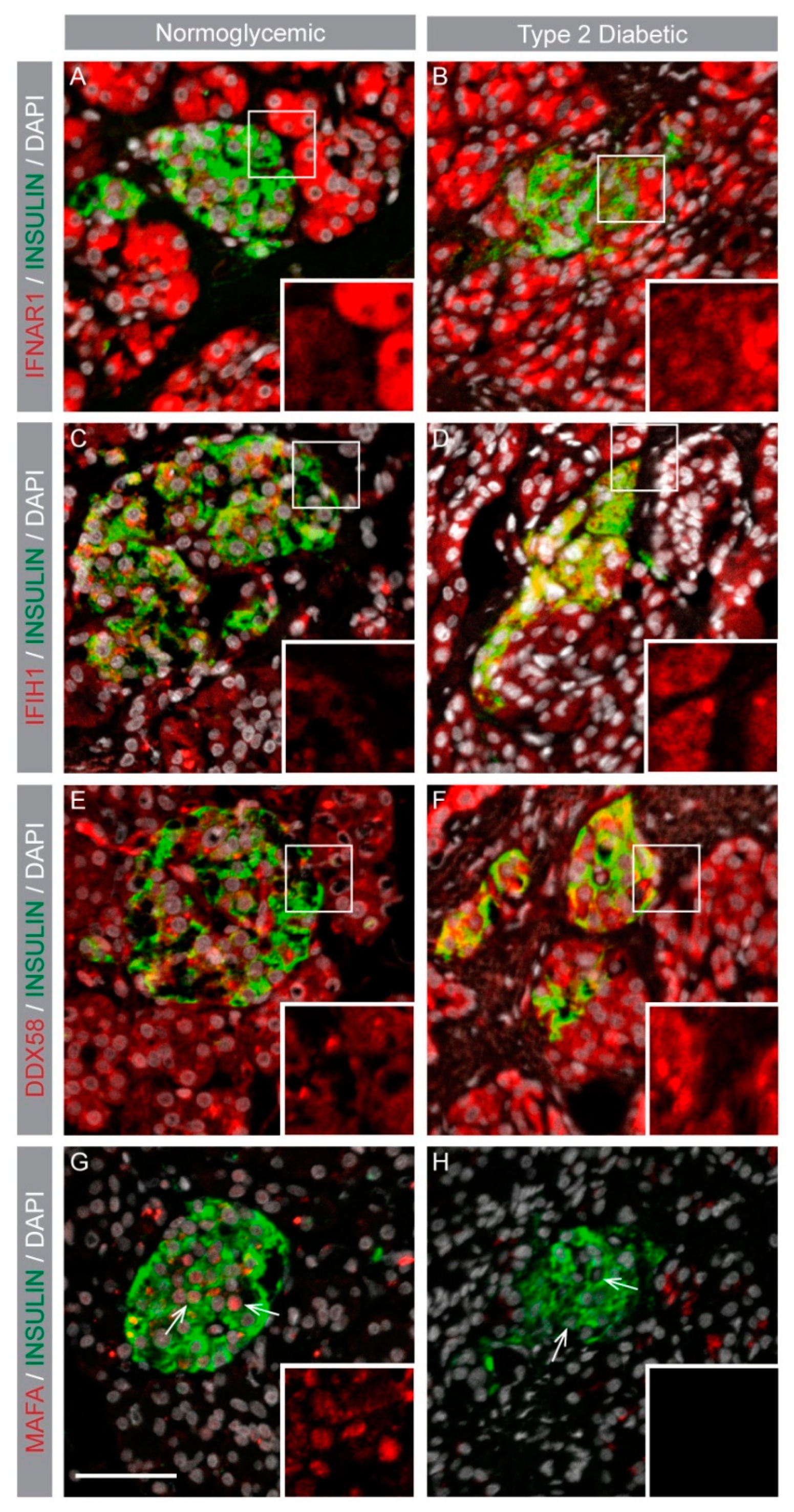
3.2. Interferon-Induced (IFI) Genes IFIH1 (Mda5) and DDX58 (Rig1) Were Negatively Correlated with MAFA Expression in Human Islets and Protein Expression Was Enhanced in Type 2 Diabetic Islets
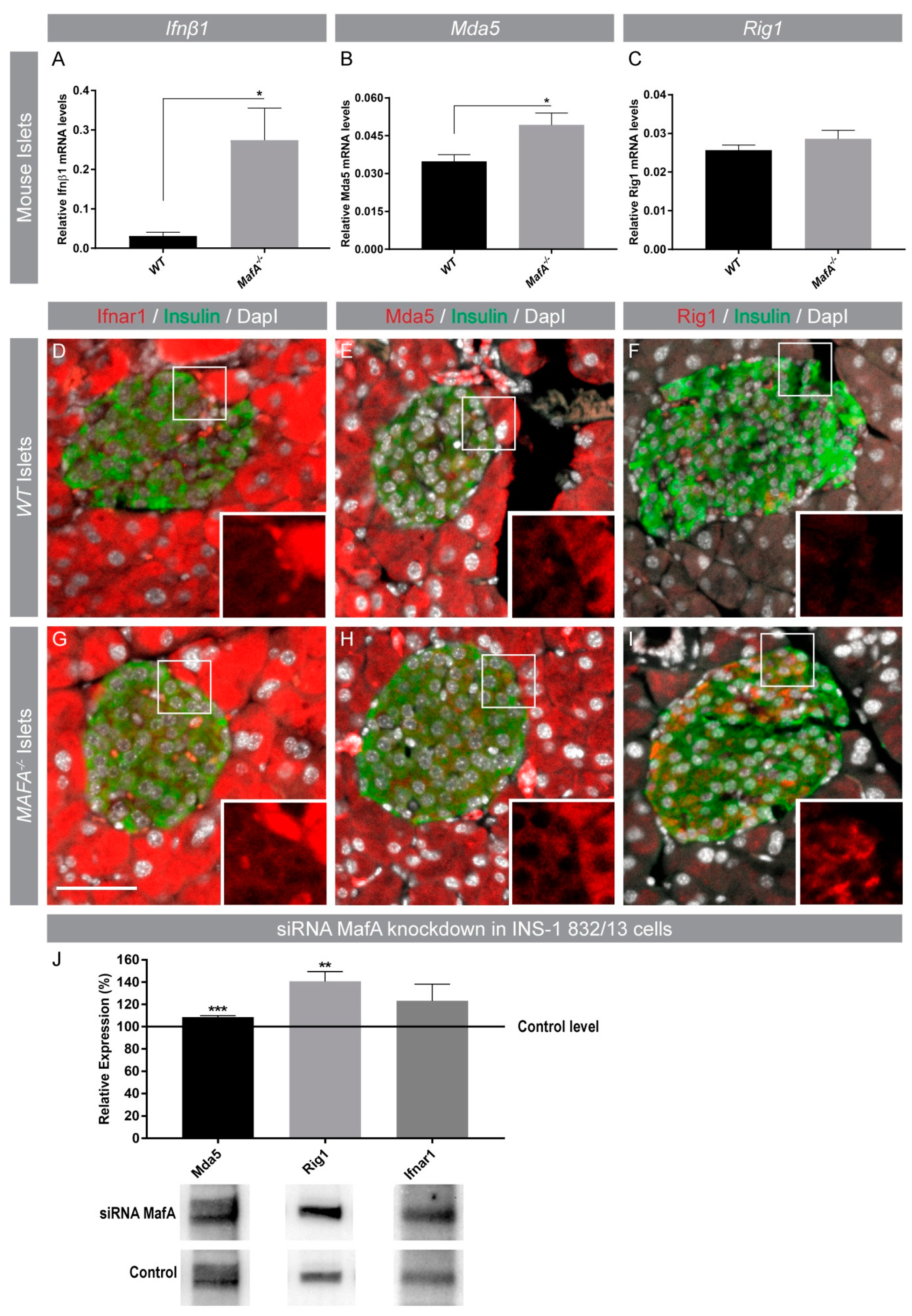
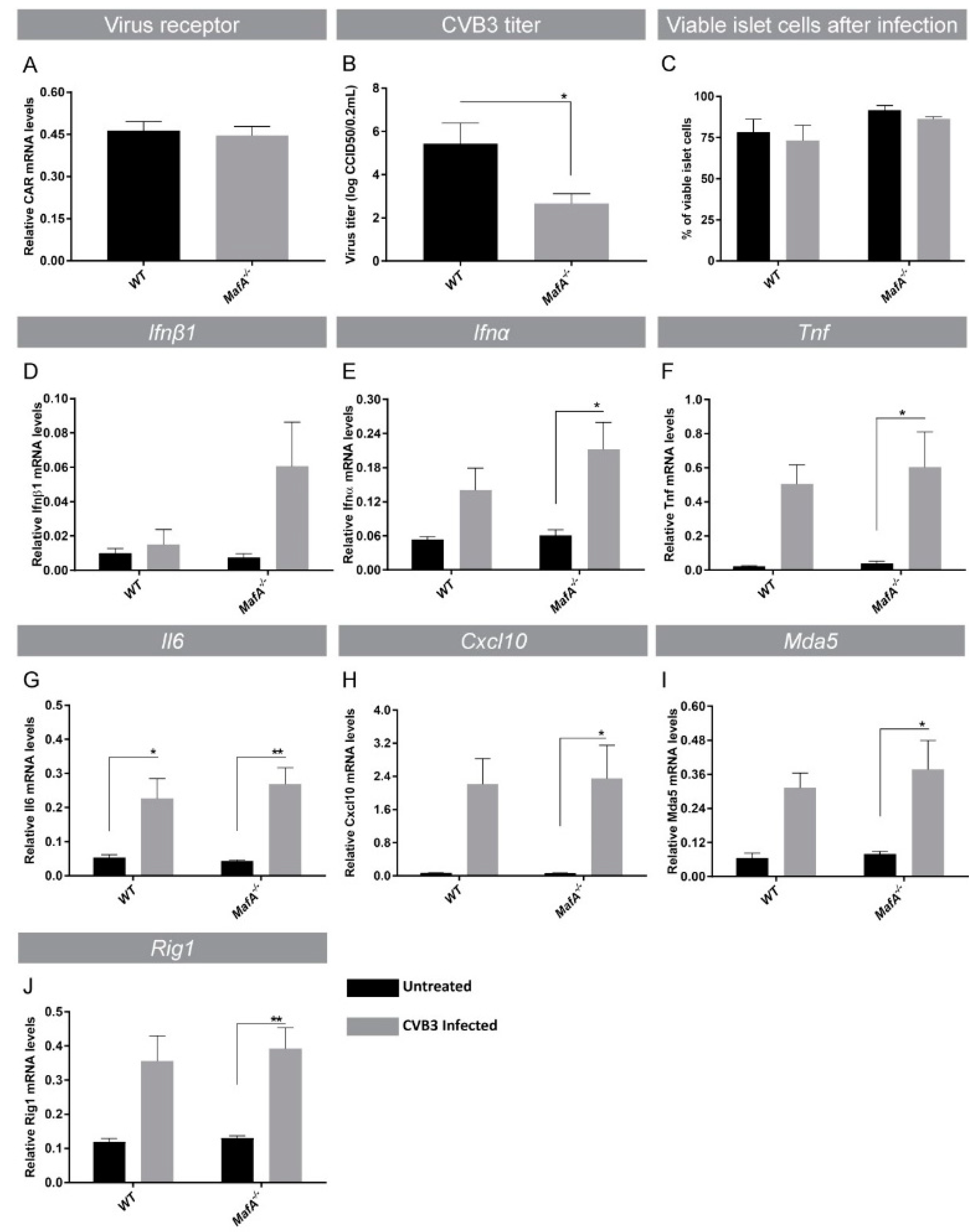
3.3. Acute and Long-Term Loss of MafA Enhances Mda5 and Rig1 Expression
3.4. MafA Deficient Mouse Islets Have Reduced Virus Propagation after Coxsackievirus B3 Infection
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cheon, H.; Borden, E.C.; Stark, G.R. Interferons and their stimulated genes in the tumor microenvironment. Semin. Oncol. 2014, 41, 156–173. [Google Scholar] [CrossRef] [PubMed]
- Newby, B.N.; Mathews, C.E. Type I interferon is a catastrophic feature of the diabetic islet microenvironment. Front. Endocrinol. (Lausanne) 2017, 8, 232. [Google Scholar] [CrossRef] [PubMed]
- Eizirik, D.L.; Sammeth, M.; Bouckenooghe, T.; Bottu, G.; Sisino, G.; Igoillo-Esteve, M.; Ortis, F.; Santin, I.; Colli, M.L.; Barthson, J.; et al. The human pancreatic islet transcriptome: expression of candidate genes for type 1 diabetes and the impact of pro-inflammatory cytokines. PLoS Genet. 2012, 8, e1002552. [Google Scholar] [CrossRef] [PubMed]
- Akbar, A.N.; Lord, J.M.; Salmon, M. IFN-alpha and IFN-beta: A link between immune memory and chronic inflammation. Immunol. Today 2000, 21, 337–342. [Google Scholar] [CrossRef]
- Alba, A.; Puertas, M.C.; Carrillo, J.; Planas, R.; Ampudia, R.; Pastor, X.; Bosch, F.; Pujol-Borrell, R.; Verdaguer, J.; Vives-Pi, M. IFN beta accelerates autoimmune type 1 diabetes in nonobese diabetic mice and breaks the tolerance to beta cells in nondiabetes-prone mice. J. Immunol. 2004, 173, 6667–6675. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.C.; Guo, H.; Coulson, R.M.; Smyth, D.J.; Pekalski, M.L.; Burren, O.S.; Cutler, A.J.; Doecke, J.D.; Flint, S.; McKinney, E.F.; et al. A type I interferon transcriptional signature precedes autoimmunity in children genetically at risk for type 1 diabetes. Diabetes 2014, 63, 2538–2550. [Google Scholar] [CrossRef]
- Huang, X.; Yuang, J.; Goddard, A.; Foulis, A.; James, R.F.; Lernmark, A.; Pujol-Borrell, R.; Rabinovitch, A.; Somoza, N.; Stewart, T.A. Interferon expression in the pancreases of patients with type I diabetes. Diabetes 1995, 44, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Kado, S.; Miyamoto, J.; Komatsu, N.; Iwaki, Y.; Ozaki, H.; Taguchi, H.; Kure, M.; Sarashina, G.; Watanabe, T.; Katsura, Y.; et al. Type 1 diabetes mellitus caused by treatment with interferon-beta. Intern. Med. 2000, 39, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Schneider, W.M.; Chevillotte, M.D.; Rice, C.M. Interferon-stimulated genes: A complex web of host defenses. Annu. Rev. Immunol. 2014, 32, 513–545. [Google Scholar] [CrossRef] [PubMed]
- Schoggins, J.W.; Rice, C.M. Interferon-stimulated genes and their antiviral effector functions. Curr. Opin. Virol. 2011, 1, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, A.; Yanai, H. Interferon signalling network in innate defence. Cell. Microbiol. 2006, 8, 907–922. [Google Scholar] [CrossRef] [PubMed]
- Belardelli, F. Role of interferons and other cytokines in the regulation of the immune response. APMIS 1995, 103, 161–179. [Google Scholar] [CrossRef] [PubMed]
- Bocci, V. The physiological interferon response. Immunol. Today 1985, 6, 7–9. [Google Scholar] [CrossRef]
- Taniguchi, T.; Takaoka, A. A weak signal for strong responses: interferon-alpha/beta revisited. Nat. Rev. Mol. Cell Biol. 2001, 2, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, T.; Takaoka, A. The interferon-alpha/beta system in antiviral responses: a multimodal machinery of gene regulation by the IRF family of transcription factors. Curr. Opin. Immunol. 2002, 14, 111–116. [Google Scholar] [CrossRef]
- Qaisar, N.; Jurczyk, A.; Wang, J.P. Potential role of type I interferon in the pathogenic process leading to type 1 diabetes. Curr. Opin. Endocrinol. Diabetes Obes. 2018, 25, 94–100. [Google Scholar] [CrossRef]
- Kim, H.; Seed, B. The transcription factor MafB antagonizes antiviral responses by blocking recruitment of coactivators to the transcription factor IRF3. Nat. Immunol. 2010, 11, 743–750. [Google Scholar] [CrossRef]
- Motohashi, H.; Igarashi, K. MafB as a type I interferon rheostat. Nat. Immunol. 2010, 11, 695–696. [Google Scholar] [CrossRef]
- Dai, C.; Brissova, M.; Hang, Y.; Thompson, C.; Poffenberger, G.; Shostak, A.; Chen, Z.; Stein, R.; Powers, A.C. Islet-enriched gene expression and glucose-induced insulin secretion in human and mouse islets. Diabetologia 2012, 55, 707–718. [Google Scholar] [CrossRef]
- Guo, S.; Dai, C.; Guo, M.; Taylor, B.; Harmon, J.S.; Sander, M.; Robertson, R.P.; Powers, A.C.; Stein, R. Inactivation of specific beta cell transcription factors in type 2 diabetes. J. Clin. Investig. 2013, 123, 3305–3316. [Google Scholar] [CrossRef]
- Artner, I.; Bianchi, B.; Raum, J.C.; Guo, M.; Kaneko, T.; Cordes, S.; Sieweke, M.; Stein, R. MafB is required for islet beta cell maturation. Proc. Natl. Acad. Sci. USA 2007, 104, 3853–3858. [Google Scholar] [CrossRef] [PubMed]
- Artner, I.; Hang, Y.; Mazur, M.; Yamamoto, T.; Guo, M.; Lindner, J.; Magnuson, M.A.; Stein, R. MafA and MafB regulate genes critical to beta-cells in a unique temporal manner. Diabetes 2010, 59, 2530–2539. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, W.; Takahashi, S.; Yasuda, K. MafA is critical for maintenance of the mature beta cell phenotype in mice. Diabetologia 2015, 58, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Moriguchi, T.; Kajihara, M.; Esaki, R.; Harada, A.; Shimohata, H.; Oishi, H.; Hamada, M.; Morito, N.; Hasegawa, K.; et al. MafA is a key regulator of glucose-stimulated insulin secretion. Mol. Cell. Biol. 2005, 25, 4969–4976. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liu, Q.; Zhou, Z.; Ikeda, Y. PDX1, Neurogenin-3, and MAFA: Critical transcription regulators for beta cell development and regeneration. Stem Cell Res. Ther. 2017, 8, 240. [Google Scholar] [CrossRef] [PubMed]
- Noso, S.; Kawabata, Y.; Babaya, N.; Hiromine, Y.; Kawasaki, E. Association study of MAFA and MAFB, genes related to organ-specific autoimmunity, with susceptibility to type 1 diabetes in japanese and caucasian populations. J. Genet. Syndr. Gene Ther. 2013, 4, 1–6. [Google Scholar] [CrossRef]
- Fadista, J.; Vikman, P.; Laakso, E.O.; Mollet, I.G.; Esguerra, J.L.; Taneera, J.; Storm, P.; Osmark, P.; Ladenvall, C.; Prasad, R.B.; et al. Global genomic and transcriptomic analysis of human pancreatic islets reveals novel genes influencing glucose metabolism. Proc. Natl. Acad. Sci. USA 2014, 111, 13924–13929. [Google Scholar] [CrossRef]
- Ottosson-Laakso, E.; Krus, U.; Storm, P.; Prasad, R.B.; Oskolkov, N.; Ahlqvist, E.; Fadista, J.; Hansson, O.; Groop, L.; Vikman, P. Glucose-induced changes in gene expression in human pancreatic islets: Causes or consequences of chronic hyperglycemia. Diabetes 2017, 66, 3013–3028. [Google Scholar] [CrossRef]
- Belinky, F.; Nativ, N.; Stelzer, G.; Zimmerman, S.; Iny Stein, T.; Safran, M.; Lancet, D. PathCards: multi-source consolidation of human biological pathways. Database 2015, 2015. [Google Scholar] [CrossRef]
- Hayashi, S.; Tenzen, T.; McMahon, A.P. Maternal inheritance of Cre activity in a Sox2Cre deleter strain. Genesis 2003, 37, 51–53. [Google Scholar] [CrossRef]
- Sarmiento, L. Enteroviral Meningitis and Emergence of Rare Enterovirus Types: Cuban Experience; Nova Science Publishers: New York, NY, USA, 2004. [Google Scholar]
- Lennette, E.H. General Principles Underlying Laboratory Diagnosis of Viral and Rickettsial Infections; American Public Health Association: New York, NY, USA, 1969. [Google Scholar]
- Ganic, E.; Singh, T.; Luan, C.; Fadista, J.; Johansson, J.K.; Cyphert, Holly A.; Bennet, H.; Storm, P.; Prost, G.; Ahlenius, H.; et al. MafA-controlled nicotinic receptor expression is essential for insulin secretion and is impaired in patients with type 2 diabetes. Cell Rep. 2002, 14, 1991–2002. [Google Scholar] [CrossRef]
- Artner, I.; Le Lay, J.; Hang, Y.; Elghazi, L.; Schisler, J.C.; Henderson, E.; Sosa-Pineda, B.; Stein, R. MafB: an activator of the glucagon gene expressed in developing islet alpha- and beta-cells. Diabetes 2006, 55, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, W.; Kondo, T.; Salameh, T.; El Khattabi, I.; Dodge, R.; Bonner-Weir, S.; Sharma, A. A switch from MafB to MafA expression accompanies differentiation to pancreatic beta-cells. Dev. Biol. 2006, 293, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Hang, Y.; Yamamoto, T.; Benninger, R.K.; Brissova, M.; Guo, M.; Bush, W.; Piston, D.W.; Powers, A.C.; Magnuson, M.; Thurmond, D.C.; et al. The MafA transcription factor becomes essential to islet beta-cells soon after birth. Diabetes 2014, 63, 1994–2005. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.E.; Robertson, R.P.; Hernandez, R.; Matveyenko, A.V.; Gurlo, T.; Butler, P.C. Beta cell nuclear musculoaponeurotic fibrosarcoma oncogene family A (MafA) is deficient in type 2 diabetes. Diabetologia 2012, 55, 2985–2988. [Google Scholar] [CrossRef] [PubMed]
- Stewart, T.A.; Hultgren, B.; Huang, X.; Pitts-Meek, S.; Hully, J.; MacLachlan, N.J. Induction of type I diabetes by interferon-alpha in transgenic mice. Science 1993, 260, 1942–1946. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Franceschini, A.; Wyder, S.; Forslund, K.; Heller, D.; Huerta-Cepas, J.; Simonovic, M.; Roth, A.; Santos, A.; Tsafou, K.P.; et al. STRING v10: Protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 2015, 43, D447–452. [Google Scholar] [CrossRef]
- Crampton, S.P.; Deane, J.A.; Feigenbaum, L.; Bolland, S. Ifih1 gene dose effect reveals MDA5-mediated chronic type I IFN gene signature, viral resistance, and accelerated autoimmunity. J. Immunol. 2012, 188, 1451–1459. [Google Scholar] [CrossRef]
- Errett, J.S.; Suthar, M.S.; McMillan, A.; Diamond, M.S.; Gale, M., Jr. The essential, nonredundant roles of RIG-I and MDA5 in detecting and controlling West Nile virus infection. J. Virol. 2013, 87, 11416–11425. [Google Scholar] [CrossRef]
- Kato, H.; Takeuchi, O.; Sato, S.; Yoneyama, M.; Yamamoto, M.; Matsui, K.; Uematsu, S.; Jung, A.; Kawai, T.; Ishii, K.J.; et al. Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 2006, 441, 101–105. [Google Scholar] [CrossRef]
- Gorman, J.A.; Hundhausen, C.; Errett, J.S.; Stone, A.E.; Allenspach, E.J.; Ge, Y.; Arkatkar, T.; Clough, C.; Dai, X.; Khim, S.; et al. The A946T variant of the RNA sensor IFIH1 mediates an interferon program that limits viral infection but increases the risk for autoimmunity. Nat. Immunol. 2017, 18, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Hultcrantz, M.; Huhn, M.H.; Wolf, M.; Olsson, A.; Jacobson, S.; Williams, B.R.; Korsgren, O.; Flodstrom-Tullberg, M. Interferons induce an antiviral state in human pancreatic islet cells. Virology 2007, 367, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Drescher, K.M.; Kono, K.; Bopegamage, S.; Carson, S.D.; Tracy, S. Coxsackievirus B3 infection and type 1 diabetes development in NOD mice: Insulitis determines susceptibility of pancreatic islets to virus infection. Virology 2004, 329, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Kanno, T.; Kim, K.; Kono, K.; Drescher, K.M.; Chapman, N.M.; Tracy, S. Group B coxsackievirus diabetogenic phenotype correlates with replication efficiency. J. Virol. 2006, 80, 5637–5643. [Google Scholar] [CrossRef] [PubMed]
- Foulis, A.K.; Farquharson, M.A.; Meager, A. Immunoreactive alpha-interferon in insulin-secreting beta cells in type 1 diabetes mellitus. Lancet 1987, 2, 1423–1427. [Google Scholar] [CrossRef]
- Kallionpaa, H.; Elo, L.L.; Laajala, E.; Mykkanen, J.; Ricano-Ponce, I.; Vaarma, M.; Laajala, T.D.; Hyoty, H.; Ilonen, J.; Veijola, R.; et al. Innate immune activity is detected prior to seroconversion in children with HLA-conferred type 1 diabetes susceptibility. Diabetes 2014, 63, 2402–2414. [Google Scholar] [CrossRef]
- Guerci, A.P.; Guerci, B.; Levy-Marchal, C.; Ongagna, J.; Ziegler, O.; Candiloros, H.; Guerci, O.; Drouin, P. Onset of insulin-dependent diabetes mellitus after interferon-alfa therapy for hairy cell leukaemia. Lancet 1994, 343, 1167–1168. [Google Scholar] [CrossRef]
- Oka, R.; Hiroi, N.; Shigemitsu, R.; Sue, M.; Oshima, Y.; Yoshida-Hiroi, M. Type 1 diabetes mellitus associated with pegylated interferon-alpha plus ribavirin treatment for chronic hepatitis c: case report and literature review. Clin. Med. Insights Endocrinol. Diabetes 2011, 4, 39–45. [Google Scholar] [CrossRef]
- Uonaga, T.; Yoshida, K.; Harada, T.; Shimodahira, M.; Nakamura, Y. Case of type 1 diabetes mellitus following interferon beta-1a treatment for multiple sclerosis. Intern. Med. 2012, 51, 1875–1877. [Google Scholar] [CrossRef]
- Concannon, P.; Rich, S.S.; Nepom, G.T. Genetics of type 1A diabetes. N. Engl. J. Med. 2009, 360, 1646–1654. [Google Scholar] [CrossRef]
- Todd, J.A. Etiology of type 1 diabetes. Immunity 2010, 32, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Santin, I.; Eizirik, D.L. Candidate genes for type 1 diabetes modulate pancreatic islet inflammation and beta-cell apoptosis. Diabetes. Obes. Metab. 2013, 15 (Suppl. 3), 71–81. [Google Scholar] [CrossRef] [PubMed]
- Kameswaran, V.; Bramswig, N.C.; McKenna, L.B.; Penn, M.; Schug, J.; Hand, N.J.; Chen, Y.; Choi, I.; Vourekas, A.; Won, K.J.; et al. Epigenetic regulation of the DLK1-MEG3 microRNA cluster in human type 2 diabetic islets. Cell Metab. 2014, 19, 135–145. [Google Scholar] [CrossRef]
- Wallace, C.; Smyth, D.J.; Maisuria-Armer, M.; Walker, N.M.; Todd, J.A.; Clayton, D.G. The imprinted DLK1-MEG3 gene region on chromosome 14q32.2 alters susceptibility to type 1 diabetes. Nat. Genet. 2010, 42, 68–71. [Google Scholar] [CrossRef]
- Yoneyama, M.; Onomoto, K.; Jogi, M.; Akaboshi, T.; Fujita, T. Viral RNA detection by RIG-I-like receptors. Curr. Opin. Immunol. 2015, 32, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Rice, G.I.; Del Toro Duany, Y.; Jenkinson, E.M.; Forte, G.M.; Anderson, B.H.; Ariaudo, G.; Bader-Meunier, B.; Baildam, E.M.; Battini, R.; Beresford, M.W.; et al. Gain-of-function mutations in IFIH1 cause a spectrum of human disease phenotypes associated with upregulated type I interferon signaling. Nat. Genet. 2014, 46, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, H.; Jin, Y.; Podolsky, R.; Reddy, M.V.; Pedersen, J.; Bode, B.; Reed, J.; Steed, D.; Anderson, S. IFIH1 polymorphisms are significantly associated with type 1 diabetes and IFIH1 gene expression in peripheral blood mononuclear cells. Hum. Mol. Genet. 2009, 18, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Zurawek, M.; Fichna, M.; Fichna, P.; Skowronska, B.; Dzikiewicz-Krawczyk, A.; Januszkiewicz, D.; Nowak, J. Cumulative effect of IFIH1 variants and increased gene expression associated with type 1 diabetes. Diabetes Res. Clin. Pract. 2015, 107, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Winkler, C.; Lauber, C.; Adler, K.; Grallert, H.; Illig, T.; Ziegler, A.G.; Bonifacio, E. An interferon-induced helicase (IFIH1) gene polymorphism associates with different rates of progression from autoimmunity to type 1 diabetes. Diabetes 2011, 60, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.; Kariuki, S.N.; Franek, B.S.; Kumabe, M.; Kumar, A.A.; Badaracco, M.; Mikolaitis, R.A.; Guerrero, G.; Utset, T.O.; Drevlow, B.E.; et al. Autoimmune disease risk variant of IFIH1 is associated with increased sensitivity to IFN-alpha and serologic autoimmunity in lupus patients. J. Immunol. 2011, 187, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Jermendy, A.; Szatmari, I.; Laine, A.P.; Lukacs, K.; Horvath, K.H.; Korner, A.; Madacsy, L.; Veijola, R.; Simell, O.; Knip, M.; et al. The interferon-induced helicase IFIH1 Ala946Thr polymorphism is associated with type 1 diabetes in both the high-incidence Finnish and the medium-incidence Hungarian populations. Diabetologia 2010, 53, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Aminkeng, F.; Van Autreve, J.E.; Weets, I.; Quartier, E.; Van Schravendijk, C.; Gorus, F.K.; Van der Auwera, B.J.; Belgian Diabetes, R. IFIH1 gene polymorphisms in type 1 diabetes: genetic association analysis and genotype-phenotype correlation in the Belgian population. Hum. Immunol. 2009, 70, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Smyth, D.J.; Cooper, J.D.; Bailey, R.; Field, S.; Burren, O.; Smink, L.J.; Guja, C.; Ionescu-Tirgoviste, C.; Widmer, B.; Dunger, D.B.; et al. A genome-wide association study of nonsynonymous SNPs identifies a type 1 diabetes locus in the interferon-induced helicase (IFIH1) region. Nat. Genet. 2006, 38, 617–619. [Google Scholar] [CrossRef] [PubMed]
- Lincez, P.J.; Shanina, I.; Horwitz, M.S. Reduced expression of the MDA5 Gene IFIH1 prevents autoimmune diabetes. Diabetes 2015, 64, 2184–2193. [Google Scholar] [CrossRef] [PubMed]
- Conrad, E.; Dai, C.; Spaeth, J.; Guo, M.; Cyphert, H.A.; Scoville, D.; Carroll, J.; Yu, W.M.; Goodrich, L.V.; Harlan, D.M.; et al. The MAFB transcription factor impacts islet alpha-cell function in rodents and represents a unique signature of primate islet beta-cells. Am. J. Physiol. Endocrinol. Metab. 2016, 310, E91–E102. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, T.A.; Zhao, L.; Artner, I.; Jarrett, H.W.; Friedman, D.; Means, A.; Stein, R. Members of the large Maf transcription family regulate insulin gene transcription in islet beta cells. Mol. Cell. Biol. 2003, 23, 6049–6062. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, T.; Sarmiento, L.; Luan, C.; Prasad, R.B.; Johansson, J.; Cataldo, L.R.; Renström, E.; Soneji, S.; Cilio, C.; Artner, I. MafA Expression Preserves Immune Homeostasis in Human and Mouse Islets. Genes 2018, 9, 644. https://doi.org/10.3390/genes9120644
Singh T, Sarmiento L, Luan C, Prasad RB, Johansson J, Cataldo LR, Renström E, Soneji S, Cilio C, Artner I. MafA Expression Preserves Immune Homeostasis in Human and Mouse Islets. Genes. 2018; 9(12):644. https://doi.org/10.3390/genes9120644
Chicago/Turabian StyleSingh, Tania, Luis Sarmiento, Cheng Luan, Rashmi B. Prasad, Jenny Johansson, Luis R. Cataldo, Erik Renström, Shamit Soneji, Corrado Cilio, and Isabella Artner. 2018. "MafA Expression Preserves Immune Homeostasis in Human and Mouse Islets" Genes 9, no. 12: 644. https://doi.org/10.3390/genes9120644
APA StyleSingh, T., Sarmiento, L., Luan, C., Prasad, R. B., Johansson, J., Cataldo, L. R., Renström, E., Soneji, S., Cilio, C., & Artner, I. (2018). MafA Expression Preserves Immune Homeostasis in Human and Mouse Islets. Genes, 9(12), 644. https://doi.org/10.3390/genes9120644




