Acceptor Stem Differences Contribute to Species-Specific Use of Yeast and Human tRNASer
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strains
2.2. Yeast Transformation
2.3. Plasmid Constructs
2.4. Western Blot Assay
2.5. Fluorescence Heat Shock Reporter
2.6. Mutual Information Analysis
3. Results and Discussion
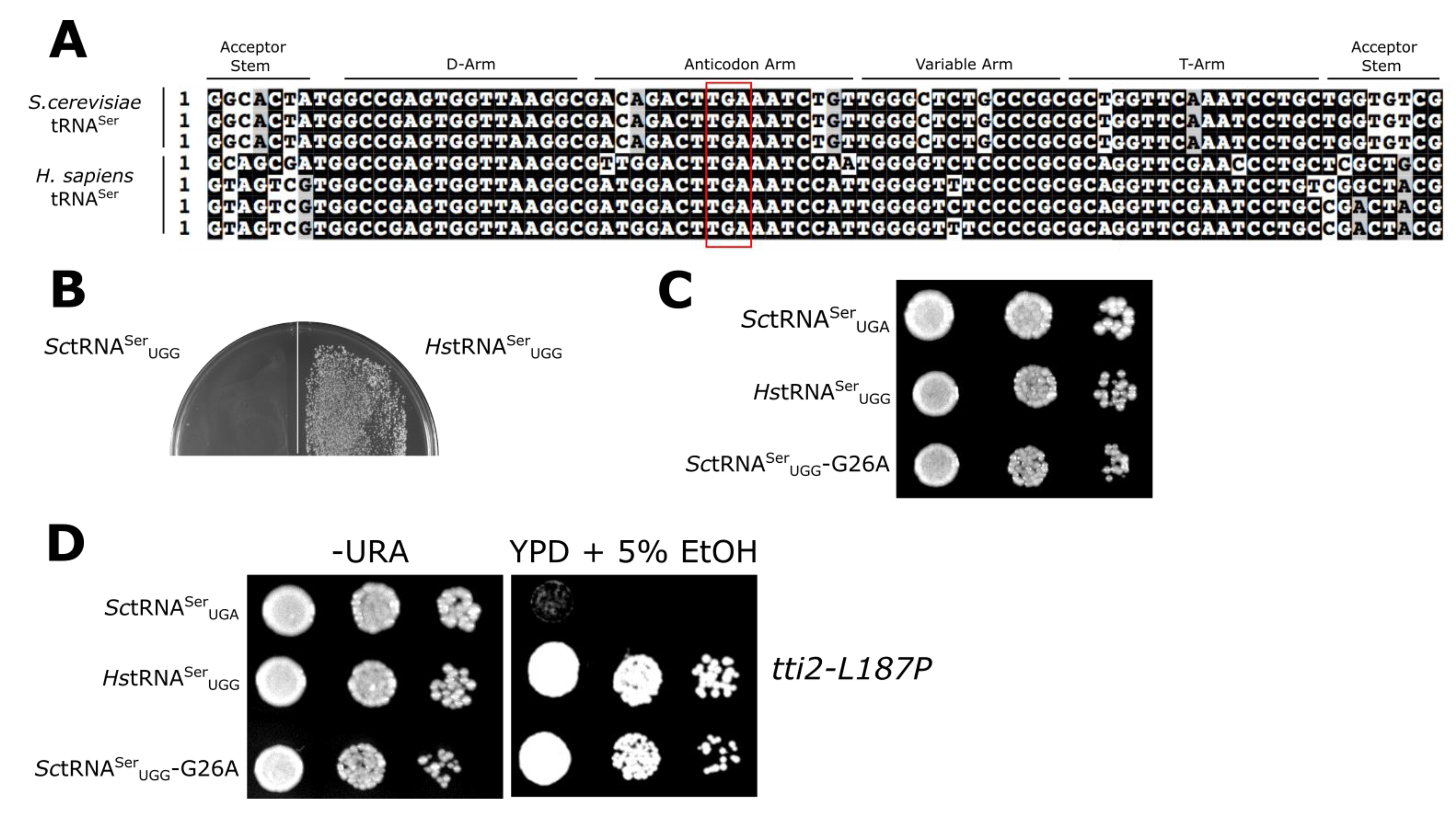
3.1. Mistranslation Assay for tRNASer Function
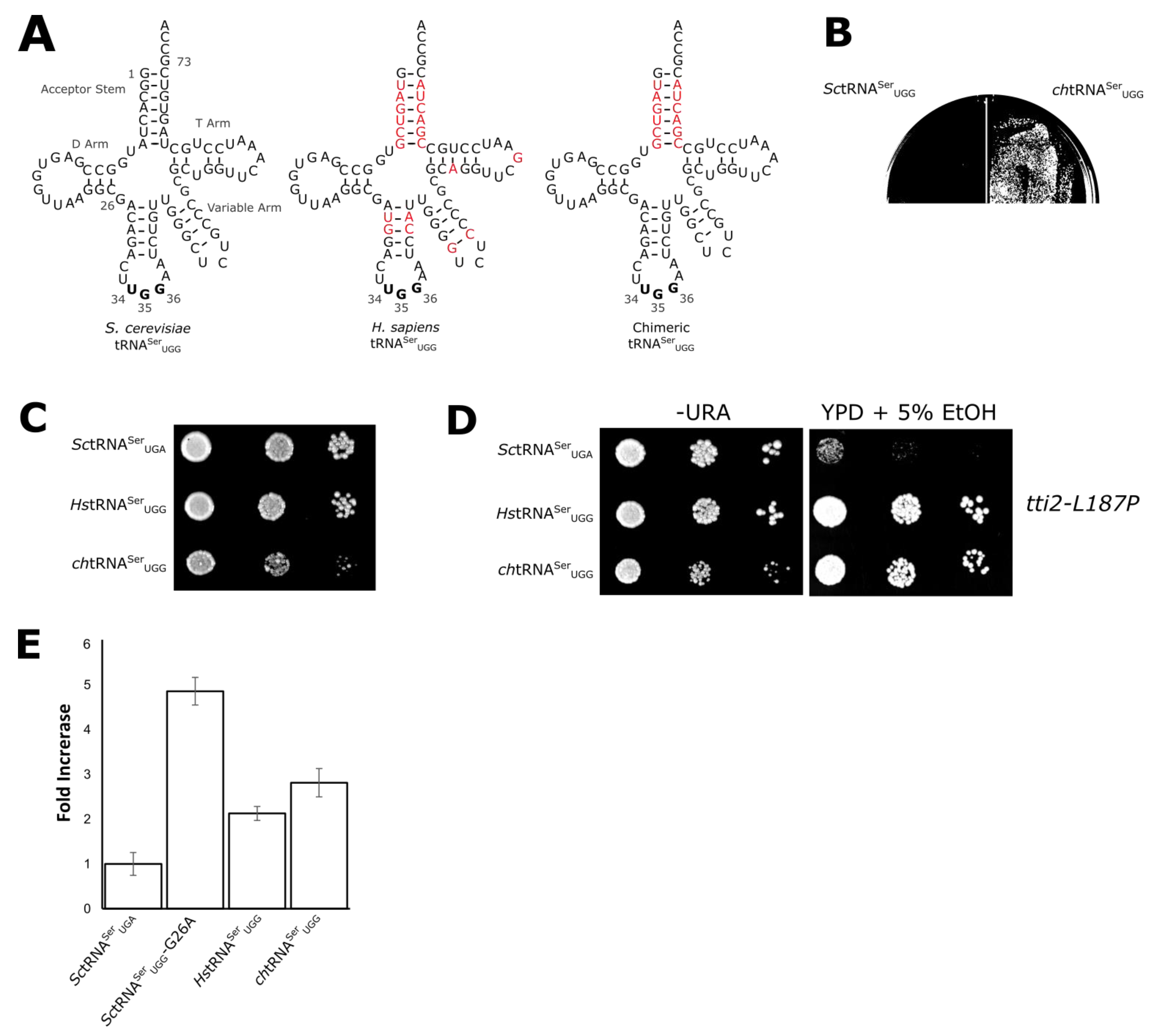
3.2. Evaluating Mechanisms for the Reduced Functionality of Human tRNASer
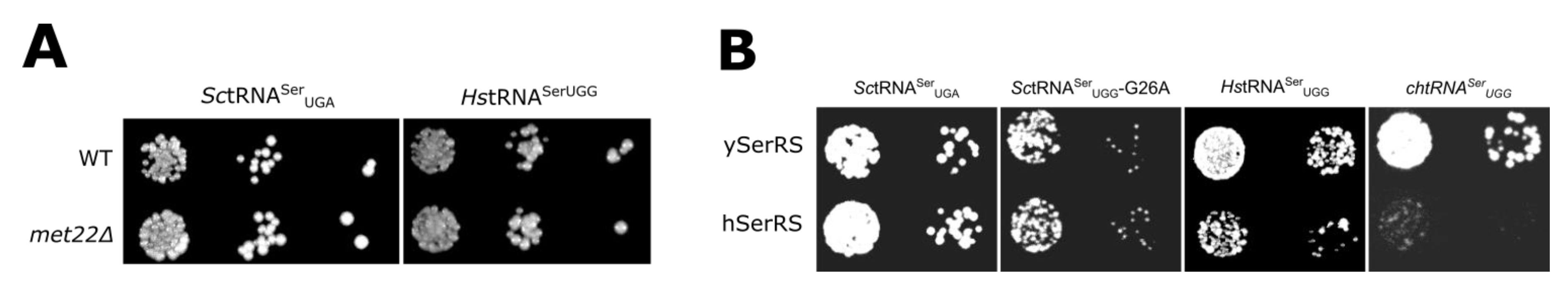
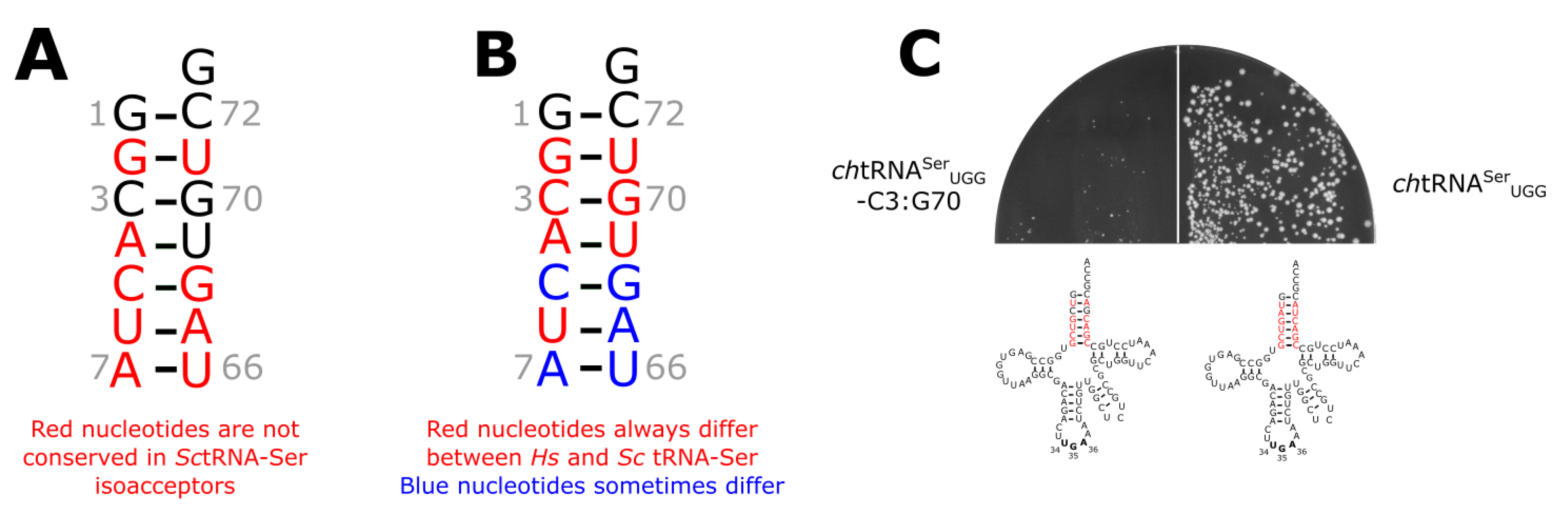
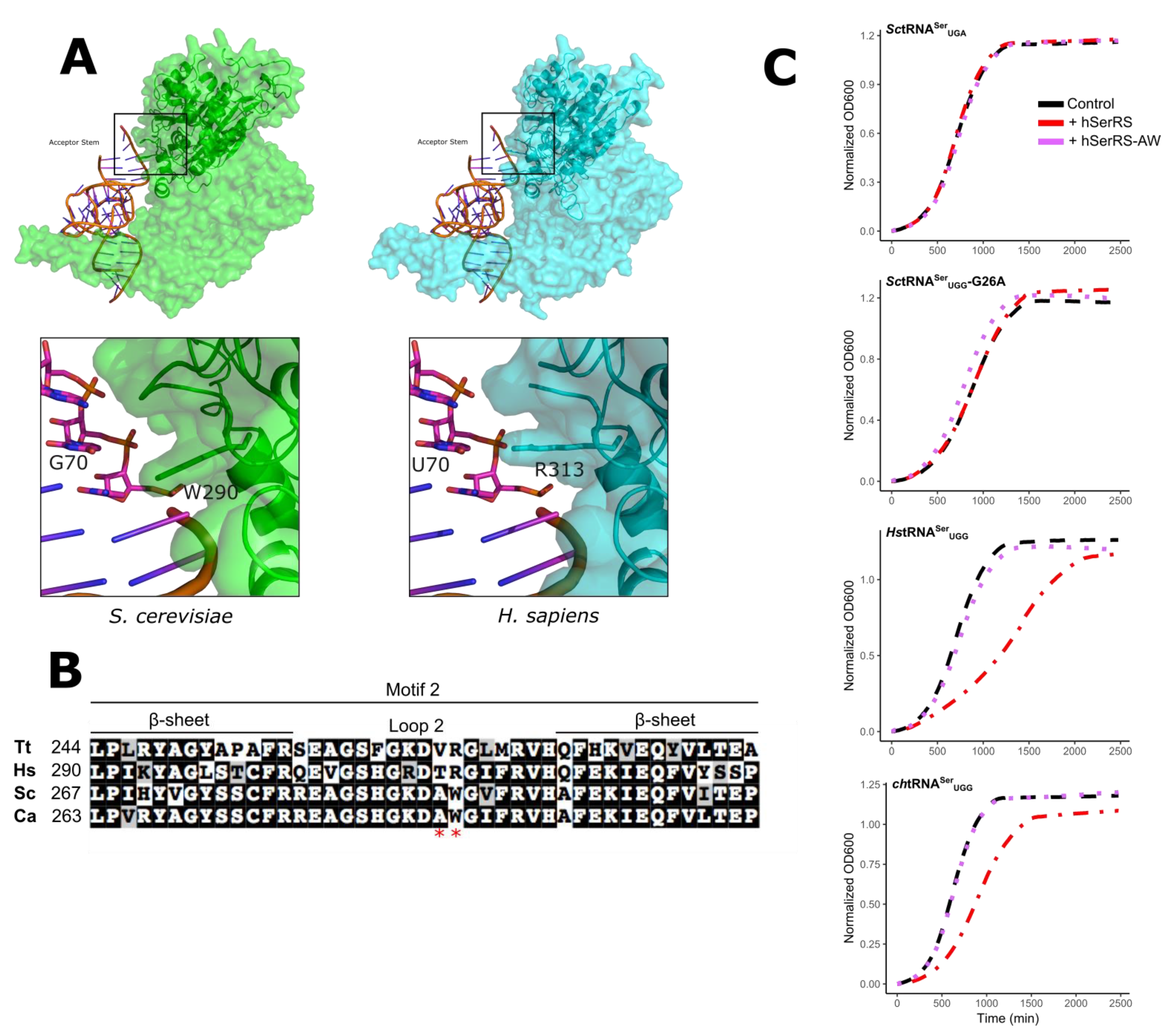
3.3. The tRNASer 3:70 Base Pair Contributes to Species Specificity
3.4. Unique Aspects of the SerRS Motif 2
3.5. Applications for Mistranslation
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoffman, K.S.; O’Donoghue, P.; Brandl, C.J. Mistranslation: From adaptations to applications. Biochim. Biophys. Acta 2017. [Google Scholar] [CrossRef] [PubMed]
- Giegé, R.; Jühling, F.; Pütz, J.; Stadler, P.; Sauter, C.; Florentz, C. Structure of transfer RNAs: Similarity and variability. Wiley Interdiscip. Rev. RNA 2012, 3, 37–61. [Google Scholar] [CrossRef] [PubMed]
- Rich, A.; RajBhandary, U.L. Transfer RNA: Molecular structure, sequence, and properties. Annu. Rev. Biochem. 1976, 45, 805–860. [Google Scholar] [CrossRef] [PubMed]
- De Duve, C. Transfer RNAs: The second genetic code. Nature 1988, 333, 117–118. [Google Scholar] [CrossRef] [PubMed]
- Commans, S.; Lazard, M.; Delort, F.; Blanquet, S.; Plateau, P. tRNA anticodon recognition and specification within subclass IIb aminoacyl-tRNA synthetases. J. Mol. Biol. 1998, 278, 801–813. [Google Scholar] [CrossRef] [PubMed]
- Giegé, R.; Sissler, M.; Florentz, C. Universal rules and idiosyncratic features in tRNA identity. Nucleic Acids Res. 1998, 26, 5017–5035. [Google Scholar] [CrossRef] [PubMed]
- Imura, N.; Weiss, G.B.; Chambers, R.W. Reconstitution of alanine acceptor activity from fragments of yeast tRNA-Ala II. Nature 1969, 222, 1147–1148. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.M.; Schimmel, P. Evidence that a major determinant for the identity of a transfer RNA is conserved in evolution. Biochemistry 1989, 28, 6800–6804. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.M.; Schimmel, P. A Simple structural feature is a major determinant of the identity of a transfer RNA. Nature 1988, 333, 140–145. [Google Scholar] [CrossRef]
- Hoffman, K.S.; Berg, M.D.; Shilton, B.H.; Brandl, C.J.; O’Donoghue, P. Genetic Selection for mistranslation rescues a defective co-chaperone in yeast. Nucleic Acids Res. 2017, 45, 3407–3421. [Google Scholar] [CrossRef]
- Asahara, H.; Himeno, H.; Tamura, K.; Nameki, N.; Hasegawa, T.; Shimizu, M. Escherichia coli seryl-tRNA synthetase recognizes tRNASer by its characteristics tertiary structure. J. Mol. Biol. 1994, 738–748. [Google Scholar] [CrossRef]
- Biou, V.; Yaremchuk, A.; Tukalo, M.; Cusack, S. The 2.9 Å crystal structure of T. thermophilus seryl-tRNA synthetase complexed with tRNASer. Science 1994, 263, 1404–1410. [Google Scholar] [CrossRef] [PubMed]
- Himeno, H.; Yoshida, S.; Soma, A.; Nishikawa, K. Only one nucleotide insertion to the long variable arm confers an efficient serine acceptor activity upon Saccharomyces cerevisiae tRNALeu in vitro. J. Mol. Biol. 1997, 268, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.L.J.; Poruri, K.; Martinis, S.A. tRNA synthetase: tRNA aminoacylation and beyond. Wiley Interdiscip. Rev. RNA 2014, 5, 461–480. [Google Scholar] [CrossRef] [PubMed]
- Eriani, G.; Delarue, M.; Poch, O.; Gangloff, J.; Moras, D. Partition of tRNA synthetases into two classes based on mutually exclusive sets of sequence motifs. Nature 1990, 347, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Ibba, M.; Soll, D. Aminoacyl-tRNA synthesis. Annu. Rev. Biochem. 2000, 69, 617–650. [Google Scholar] [CrossRef] [PubMed]
- Eriani, G.; Dirheimer, G.; Gangloff, J. Cysteinyl-tRNA synthetase: Determination of the last E. Coli aminoacyl-tRNA synthetase primary structure. Nucleic Acids Res. 1991, 19, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, I.; Nureki, O.; Ugaji-Yoshikawa, Y.; Kuwabara, S.; Shimada, A.; Tateno, M.; Lorber, B.; Giegé, R.; Moras, D.; Yokoyama, S.; et al. The 2.0 Å crystal structure of Thermus thermophilus methionyl-tRNA synthetase reveals two RNA-binding modules. Structure 2000, 8, 197–208. [Google Scholar] [CrossRef]
- Artymiuk, P.J.; Rice, D.W.; Poirrette, A.R.; Willet, P. A tale of two synthetases. Nat. Struct. Biol. 1994, 1, 758–760. [Google Scholar] [CrossRef] [PubMed]
- Cusack, S.; Berthet-Colominas, C.; Härtlein, M.; Nassar, N.; Leberman, R. A Second class of synthetase structure revealed by X-ray analysis of Escherichia coli seryl-tRNA synthetase at 2.5 A. Nature 1990, 347, 249–255. [Google Scholar] [CrossRef]
- Cusack, S.; Härtlein, M.; Leberman, R. Sequence, structural and evolutionary relationships between class 2 aminoacyl-tRNA synthetases. Nucleic Acids Res. 1991, 19, 3489–3498. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.F.; Hartman, H. The evolution of class II aminoacyl-tRNA synthetases and the first code. FEBS Lett. 2015, 589, 3499–3507. [Google Scholar] [CrossRef] [PubMed]
- Ruff, M.; Krishnaswamy, S.; Boeglin, M.; Poterszman, A.; Mitschler, A.; Podjarny, A.; Rees, B.; Thierry, J.C.; Moras, D. Class II aminoacyl transfer RNA synthetases: Crystal structure of yeast aspartyl-tRNA synthetase complexed with tRNAAsp. Science 1991, 252, 1682–1689. [Google Scholar] [CrossRef] [PubMed]
- Cavarelli, J.; Moras, D. Recognition of tRNAs by aminoacyl-tRNA synthetases. FASEB J. 1993, 7, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Eichert, A.; Oberthuer, D.; Betzel, C.; Geßner, R.; Erdmann, V.A.; Fürste, J.P.; Förster, C. The seryl-tRNA synthetase/tRNA ser acceptor stem interface is mediated via a specific network of water molecules. Biochem. Biophys. Res. Commun. 2011, 412, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Bilokapic, S.; Maier, T.; Ahel, D.; Gruic-Sovulj, I.; Söll, D.; Weygand-Durasevic, I.; Ban, N. Structure of the unusual seryl-tRNA synthetase reveals a distinct zinc-dependent mode of substrate recognition. EMBO J. 2006, 25, 2498–2509. [Google Scholar] [CrossRef] [PubMed]
- Vincent, C.; Borel, F.; Willison, J.C.; Laberman, R.; Härtlein, M. Seryl-tRNA synthetase from Escherichia coli: Functional evidence for cross-dimer tRNA binding during aminoacylation. Nucleic Acids Res. 1995, 23, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Guo, Y.; Tian, Q.; Jia, Q.; Gao, Y.; Zhang, Q.; Zhou, C.; Xie, W. SerRS-tRNASec complex Structures reveal mechanism of the first step in selenocysteine biosynthesis. Nucleic Acids Res. 2015, 43, 10534–10545. [Google Scholar] [CrossRef]
- Normanly, J.; Ogden, R.C.; Horvath, S.J.; Abelson, J. Changing the identity of a transfer RNA. Nature 1986, 321, 213–219. [Google Scholar] [CrossRef]
- Normanly, J.; Ollick, T.; Abelson, J. Eight base changes are sufficient to convert a leucine-inserting tRNA into a serine-inserting tRNA. Proc. Natl. Acad. Sci. USA 1992, 89, 5680–5684. [Google Scholar] [CrossRef]
- Shiba, K.; Stello, T.; Motegi, H.; Noda, T.; Musier-Forsyth, K.; Schimmel, P. Human lysyl-tRNA synthetase accepts nucleotide 73 variants and rescues Escherichia coli double-defective mutant. J. Biol. Chem. 1997, 272, 22809–22816. [Google Scholar] [CrossRef] [PubMed]
- Ripmaster, T.L.; Shiba, K.; Schimmel, P. Wide cross-species aminoacyl-tRNA synthetase replacement in vivo: Yeast cytoplasmic alanine enzyme replaced by human polymyositis serum antigen. Proc. Natl. Acad. Sci. USA 1995, 92, 4932–4936. [Google Scholar] [CrossRef] [PubMed]
- Rips, J.; Meyer-Schuman, R.; Breuer, O.; Tsabari, R.; Shaag, A.; Revel-Vilk, S.; Reif, S.; Elpeleg, O.; Antonellis, A.; Harel, T. MARS variant associated with both recessive interstitial lung and liver disease and dominant Charcot-Marie-Tooth disease. Eur. J. Med. Genet. 2018. [Google Scholar] [CrossRef] [PubMed]
- Burke, B.; Yang, F.; Chen, F.; Stehlin, C.; Chan, B.; Musier-Forsyth, K. Evolutionary coadaptation of the motif 2−acceptor stem interaction in the class II prolyl-tRNA synthetase system. Biochemistry 2000, 39, 15540–15547. [Google Scholar] [CrossRef] [PubMed]
- Edwards, H.; Trézéguet, V.; Schimmel, P. An Escherichia coli tyrosine transfer RNA is a leucine-specific transfer RNA in the yeast Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1991, 88, 1153–1156. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.; Chin, J.W. Designer proteins: Applications of genetic code expansion in cell biology. Nat. Rev. Mol. Cell Biol. 2012, 13, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Winzeler, E.A.; Davis, R.W. Functional analysis of the yeast genome. Curr. Opin. Genet. Dev. 1997, 7, 771–776. [Google Scholar] [CrossRef]
- Tong, A.H.; Evangelista, M.; Parsons, A.B.; Xu, H.; Bader, G.D.; Pagé, N.; Robinson, M.; Raghibizadeh, S.; Hogue, C.W.; Bussey, H.; et al. Systematic genetic analysis with ordered arrays of yeast deletion mutants. Science 2001, 294, 2364–2368. [Google Scholar] [CrossRef]
- Berg, M.D.; Hoffman, K.S.; Genereaux, J.; Mian, S.; Trussler, R.S.; Haniford, D.B.; O’Donoghue, P.; Brandl, C.J. Evolving mistranslating tRNAs through a phenotypically ambivalent intermediate in Saccharomyces cerevisiae. Genetics 2017, 206, 1865–1879. [Google Scholar] [CrossRef]
- Hoffman, K.S.; Duennwald, M.L.; Karagiannis, J.; Genereaux, J.; McCarton, A.S.; Brandl, C.J. Saccharomyces cerevisiae Tti2 regulates PIKK proteins and stress response. G3 2016, 6, 1649–1659. [Google Scholar] [CrossRef]
- Brandman, O.; Stewart-Ornstein, J.; Wong, D.; Larson, A.; Williams, C.C.; Li, G.-W.; Zhou, S.; King, D.; Shen, P.S.; Weibezahn, J.; et al. A Ribosome-bound quality control complex triggers degradation of nascent peptides and signals translation stress. Cell 2012, 151, 1042–1054. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A.; Lang, V.; Cook, R.; Brandl, C.J. Identification of native complexes containing the yeast coactivator/repressor proteins NGG1/ADA3 and ADA2. J. Biol. Chem. 1997, 272, 5571–5578. [Google Scholar] [CrossRef] [PubMed]
- Dickson, R.J.; Gloor, G.B. Bioinformatics Identification of Coevolving Residues. In Homing Endonucleases: Methods and Protocols; Edgell, D.R., Ed.; Humana Press: Totowa, NJ, USA, 2014; pp. 223–243. [Google Scholar]
- Rocha, R.; Pereira, P.J.B.; Santos, M.A.S.; Macedo-Ribeiro, S. Unveiling the structural basis for translational ambiguity tolerance in a human fungal pathogen. Proc. Natl. Acad. Sci. USA 2011, 108, 14091–14096. [Google Scholar] [CrossRef] [PubMed]
- Itoh, Y.; Sekine, S. ichi; Kuroishi, C.; Terada, T.; Shirouzu, M.; Kuramitsu, S.; Yokoyama, S. Crystallographic and mutational studies of seryl-tRNA synthetase from the archaeon Pyrococcus horikoshii. RNA Biol. 2008, 5, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Shi, Y.; Yang, X.-L. Crystal structure of human seryl-tRNA synthetase and ser-sa complex reveals a molecular lever specific to higher eukaryotes. Structure 2013, 21, 2078–2086. [Google Scholar] [CrossRef]
- Belrhali, H.; Yaremchuk, A.; Tukalo, M.; Larsen, K.; Berthet-Colominas, C.; Leberman, R.; Beijer, B.; Sproat, B.; Als-Nielsen, J.; Grübel, G. Crystal structures at 2.5 angstrom resolution of seryl-tRNA synthetase complexed with two analogs of seryl adenylate. Science 1994, 263, 1432–1436. [Google Scholar] [CrossRef]
- Wang, Y.; Geer, L.Y.; Chappey, C.; Kans, J.A.; Bryant, S.H. Cn3D: Sequence and Structure views for entrez. Trends Biochem. Sci. 2000, 25, 300–302. [Google Scholar] [CrossRef]
- Dickson, R.J.; Gloor, G.B. The MIp toolset: An efficient algorithm for calculating mutual information in protein alignments. arXiv, 2013; arXiv:1304.4573. [Google Scholar]
- Thompson, J.; Dahlberg, A.E. Testing the conservation of the translational machinery over evolution in diverse environments: Assaying Thermus thermophilus ribosomes and initiation factors in a coupled transcription—translation system from Escherichia coli. Nucleic Acids Res. 2004, 32, 5954–5961. [Google Scholar] [CrossRef] [PubMed]
- Ganoza, M.C.; Kiel, M.C.; Aoki, H. Evolutionary conservation of reactions in translation. Microbiol. Mol. Biol. Rev. 2002, 66, 460–485. [Google Scholar] [CrossRef]
- Chan, P.P.; Lowe, T.M. GtRNAdb 2.0: An Expanded database of transfer RNA genes identified in complete and draft genomes. Nucleic Acids Res. 2016, 44, D184–D189. [Google Scholar] [CrossRef] [PubMed]
- Dewe, J.M.; Whipple, J.M.; Chernyakov, I.; Jaramillo, L.N.; Phizicky, E.M. The yeast rapid tRNA decay pathway competes with elongation factor 1A for substrate tRNAs and acts on tRNAs lacking one or more of several modifications. RNA 2012, 18, 1886–1896. [Google Scholar] [CrossRef]
- Chernyakov, I.; Whipple, J.M.; Kotelawala, L.; Grayhack, E.J.; Phizicky, E.M. Degradation of several hypomodified mature tRNA species in Saccharomyces cerevisiae is mediated by Met22 and the 5’-3’ exonucleases Rat1 and Xrn1. Genes Dev. 2008, 22, 1369–1380. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef] [PubMed]
- Cavarelli, J.; Rees, B.; Ruff, M.; Thierry, J.C.; Moras, D. Yeast tRNAAsp recognition by its cognate class II aminoacyl-tRNA synthetase. Nature 1993, 362, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Goldgur, Y.; Mosyak, L.; Reshetnikova, L.; Ankilova, V.; Lavrik, O.; Khodyreva, S.; Safro, M. The Crystal structure of phenylalanyl-tRNA synthetase from Thermus thermophilus complexed with cognate tRNA (Phe). Structure 1997, 5, 59–68. [Google Scholar] [CrossRef]
- Eiler, S.; Dock-Bregeon, A.-C.; Moulinier, L.; Thierry, J.C.; Moras, D. Synthesis of aspartyl-tRNAAsp in Escherichia coli--A snapshot of the second step. EMBO J. 1999, 18, 6532–6541. [Google Scholar] [CrossRef] [PubMed]
- Achsel, T.; Gross, H.J. Identity determinants of human tRNASer: Sequence elements necessary for serylation and maturation of a tRNA with a long extra arm. EMBO J. 1993, 12, 3333–3338. [Google Scholar] [CrossRef]
- Lenhard, B.; Filipic, S.; Landeka, I.; Ivan, S.; So, D. Defining the active site of yeast seryl-tRNA synthetase. J. Biol. Chem. 1997, 272, 1136–1141. [Google Scholar] [CrossRef]
- Luscombe, N.M.; Laskowski, R.A.; Thornton, J.M. Amino acid—base interactions: A three-dimensional analysis of protein—DNA interactions at an atomic level. Nucleic Acids Res. 2001, 29, 2860–2874. [Google Scholar] [CrossRef]
- Davies, D.R.; Goryshin, I.Y.; Reznikoff, W.S.; Rayment, I. Three-dimensional structure of the Tn5 synaptic complex transposition intermediate. Science 2000, 289, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.J.; Cheng, X. Base flipping. Annu. Rev. Biochem. 1998, 67, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Berg, M.D. Western University, London, Canada. in preparation.
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berg, M.D.; Genereaux, J.; Zhu, Y.; Mian, S.; Gloor, G.B.; Brandl, C.J. Acceptor Stem Differences Contribute to Species-Specific Use of Yeast and Human tRNASer. Genes 2018, 9, 612. https://doi.org/10.3390/genes9120612
Berg MD, Genereaux J, Zhu Y, Mian S, Gloor GB, Brandl CJ. Acceptor Stem Differences Contribute to Species-Specific Use of Yeast and Human tRNASer. Genes. 2018; 9(12):612. https://doi.org/10.3390/genes9120612
Chicago/Turabian StyleBerg, Matthew D., Julie Genereaux, Yanrui Zhu, Safee Mian, Gregory B. Gloor, and Christopher J. Brandl. 2018. "Acceptor Stem Differences Contribute to Species-Specific Use of Yeast and Human tRNASer" Genes 9, no. 12: 612. https://doi.org/10.3390/genes9120612
APA StyleBerg, M. D., Genereaux, J., Zhu, Y., Mian, S., Gloor, G. B., & Brandl, C. J. (2018). Acceptor Stem Differences Contribute to Species-Specific Use of Yeast and Human tRNASer. Genes, 9(12), 612. https://doi.org/10.3390/genes9120612




