Exceptional Chromosomal Evolution and Cryptic Speciation of Blind Mole Rats Nannospalax leucodon (Spalacinae, Rodentia) from South-Eastern Europe
Abstract
1. Introduction
1.1. Phylogeny and Systematics
1.2. Speciation
1.2.1. The Greater BMR Genus Spalax
1.2.2. The Lesser BMR Genus Nannospalax
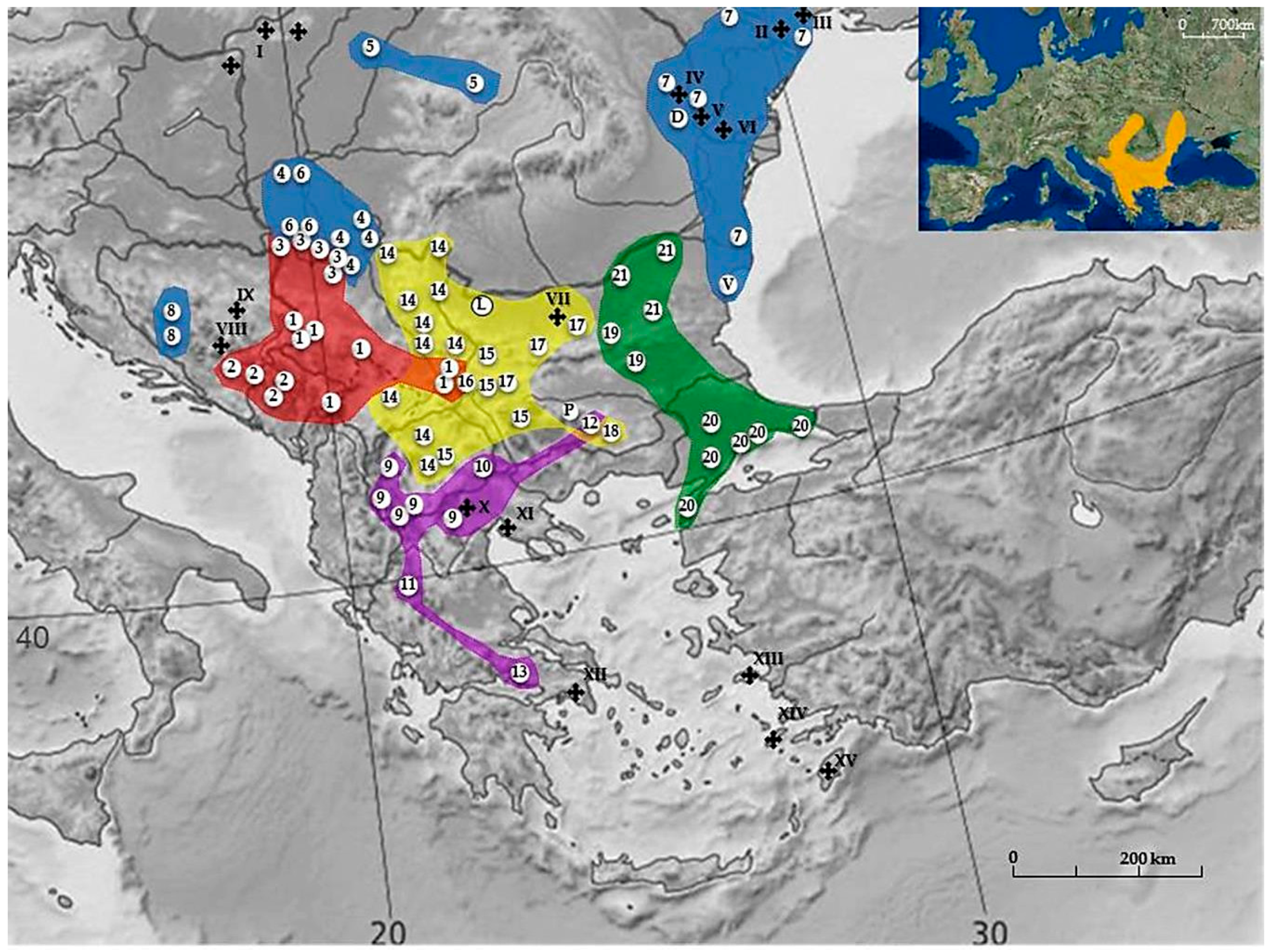
2. Mediterranean BMR N. leucodon Cryptospecies
2.1. Phylogenetic Dendrogram
- I
- The Yugoslav Branch (NF = 82–90) is characterized by the presence of a very small pair of metacentric autosomes.
- montanoserbicus (Savić and Soldatović, 1974) 2n = 56; NF = 82; with 15–16 pairs of small acrocentrics is probably a relic of an ancestor originally settled in the Central Balkan region. Today its populations occupy a wide discontinuous area of island-type isolates of preglacial Mid-Balkan Mountains (the Rhodope Mt.—Dinaride in Hercegovina and Montenegro), over 700 m a.s.l. It is possible that the area of this CF spreads to the territory of the Rhodopes in neighboring Bulgaria as well. The forms hercegovinensis and syrmiensis probably originate from it [29]. They share an identical 2n and NF, but with significant dissimilarities in the morphology of some autosomal groups. There are no data regarding reproductive isolation between them. Nevertheless, considering the morpho and craniometric discrepancies, the geographical distance between their distributional areas and the biogeographic differences among their habitats, they could be considered as a biologically separate species—cryptospecies.
- hercegovinensis (Mehely, 1909) 2n = 54; NF = 90 was described as a separate form (species) [68] recorded in the Dinaric Mountains in Montenegro and Hercegovina.
- syrmiensis (Mehely, 1909) 2n = 54; NF = 90 is a group of populations with different chromosomal morphology from hercegovinensis. They occupy the plains of Srem and the right bank of the rivers Sava and Danube from the entry of the river Drina into the Sava and the Great Morava into the Danube. A slight discrepancy in karyotype structure (see Table S1) has been recognized in one population (Banovo Brdo, Belgrade).
- II
- The Central Balkan Branch is characterized by very similar NF values, divided into two subsidiaries: the North Balkan branch (hungaricus, transsylvanicus, leucodon, montanosyrmiensis, and monticola) and the South Balkan branch (hellenicus, thracius, strumiciensis, makedonicus, and epiroticus). This group diverged very early with 2n almost 60 for hellenicus, epiroticus, and leucodon, all characterized with fourteen pairs of acrocentric autosomes. The CF leucodon drifted to the northern part of the Balkan Peninsula and generated the northern branch—CFs hungaricus, transsylvanicus, montanosyrmiensis, and monticola with variable 2n = 48–54, but almost identical NF, namely 84 or 86. The Makedonian branch diverged from the epirus CF, while the CFs strumiciensis and thracius separated from the initial branch somewhat later.
- IIa
- North Balkan subsidiary branch. The existence of reproductive isolation between some CFs has been proved.
- 4.
- hungaricus (Nehring, 1898) 2n = 48; NF = 84 inhabits a large area of Pannonian lowland in Bačka, Banat, and a narrow belt in northern Serbia, where it overlaps with syrmiensis on the slopes of Mt. Avala.
- 5.
- 6.
- montanosyrmiensis (Savić and Soldatović, 1974) 2n = 54; NF = 86 is a relict species present on Fruška Gora, where two populations with a difference in the Y chromosome exist, and additionally in Kelebia on the Serbian/Hungarian border [42].
- 7.
- leucodon (Nordmann, 1840) 2n = 56; NF = 84 is distributed in Moldavia, Dobrudzha, Odessa and South-western Ukraine [11]. There is a certain chromosomal similarity with dobrudzha CF, so a recent hypothetical dispersal of N. leucodon into Moldova and Southern Ukraine from the southwest was suggested. Nearby on the Black Sea coast, Eastern Bulgaria, varna CF (2n = 52, NFa = 76, NF = 80) was reported but only from description of the locality [72].
- 8.
- monticola (Nehring, 1898) is a relict species 2n = 54; NF = 84 that occupies the western border of the Mediterranean BMR range, i.e., Kupreško Polje, Bosnia and Herzegovina. The area probably spreads to the central parts of the Dinaric massif. Although the karyotype is similar to that of montanosyrmiensis, morpho- and craniometrics diverge on a greater scale.
- IIb
- South Balkan subsidiary branch (2n = 54, 56 and 58; NF = 84 and 88). They inhabit the South-eastern and far Southern parts of the Balkan Peninsula. According to karyotype similarities and morpho- and craniometric characters, several forms could belong to this CF:
- 9.
- makedonicus (Savić and Soldatović, 1974) 2n = 52; NF = 86 inhabits the South-western Balkan Peninsula including Western Makedonia, North-western Greece, and probably spreading into neighboring Albania.
- 10.
- strumiciensis (Savić and Soldatović, 1974) 2n = 54; NF = 88 occurs in Dabilja, Strumica Valley, Macedonia (FYROM).
- 11.
- epiroticus (Savić, 1982) 2n = 56; NF = 84 is found in Lefkothea, Epirus, Greece.
- 12.
- thracius (Savić, 1982) 2n = 56; NF = 88 inhabits Novo Selo on the Thracian plain, Bulgaria.
- 13.
- hellenicus (Mehely, 1909) 2n = 58; NF = 88 was recorded in Parnas, Greece.
According to their morphological characters several more CFs were described as thermacius (Hinton, 1920) (= strumiciensis), insularis (Thomas, 1917), thessalicus (Ondrias, 1966), and peloponnesiacus (Ondrias, 1966) [29]. - III
- The Serbicus Branch has a very high NF of 90–98. The first CF to develop was the Sofia-East population with 11 pairs of acrocentric autosomes. CFs ovchepolensis and serbicus were formed later, as well as the tranensis CF, represented by only one population and possibly rhodopiensis CF, also represented by a single population in Dobrostan, Bulgaria, although this form digresses slightly from other CFs of this branch.
- 14.
- serbicus (Mehely) 2n = 54; NF = 98 includes several CFs that are reproductively isolated from neighboring CFs. They inhabit valleys of Eastern Serbia, Northern Makedonia, Kosovo, and South-western Bulgaria. The suggestion that morpho- and craniometric characters are similar to monticola, hungaricus, and montanosyrmiensis, because of a common ancestor, should be further explored. Some authors recognize the highly similar CF lom in two isolated areas in North-western and South-western Bulgaria [72].
- 15.
- ovchepolensis (Savić and Soldatović, 1974) 2n = 54; NF = 94 is found at Ovče Polje, Makedonia (FYROM).
- 16.
- tranensis (Peshev, 1981) 2n = 54; NF = 96 was recorded in Tran, Bulgaria.
- 17.
- sofiensis (Peshev, 1983) 2n = 56; NF = 90 was registerd in Cherven Briag, Bulgaria.
- 18.
- rhodopiensis (Peshev, 1981) 2n = 54; NF = 92 occurs in Dobrostan, Bulgaria and should be analyzed in greater detail regarding karyotype structure [72].
- IV
- East Balkan Branch. Contrary to pronounced 2n differences, the NF values vary within narrow limits of 74 to 78 and therefore it was suggested that Robertsonian fusions of acrocentric autosomes were mostly responsible for the karyotype transformations. No crossbreeding experiments have been done in this group.
- 19.
- turcicus (Mehely, 1909) 2n = 56; NF = 78 has seventeen pairs of acrocentric autosomes and was the first to diverge from the basic branch. Now it probably represents the oldest existing form and inhabits the Lower Thrace lowlands.
Out of the same group, two CFs were formed in Bulgaria — srebarna and kozarevets, with nine and eight pairs of acrocentric autosomes, respectively.- 20.
- bulgaricus (Peshev, 1981) 2n = 46 and 48; NF = 76 consists of two populations with different karyotypes.
- 21.
- srebarnensis (Peshev, 1981) 2n = 48; NF = 78 is found in Russe, Targoviste, and Silistra regions in North-east Bulgaria.
2.2. Natural Hybrids
2.3. Experimental Crossbreeding
3. Chromosomal Rearrangements in Nannospalax leucodon
The Fusion or Fission Hypothesis?
4. Molecular Research in the Subfamily Spalacinae
5. Cryptic Speciation in N. leucodon
5.1. Reproductive Isolation
5.2. Sympatric/Parapatric Distribution
5.3. Morphological/Physiological Modifications
5.4. Genetic Distances
5.5. Further Perspectives
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Musser, G.G.; Carleton, M.D. Order Rodentia. In Mammal Species of the World: A Taxonomic and Geographic Reference; Wilson, D.E., Reeder, D.M., Eds.; Johns Hopkins University Press: Baltimore, MD, USA, 2005; pp. 745–1601. [Google Scholar]
- Baker, R.J.; Bradley, R.D. Speciation in Mammals and the genetic species concept. J. Mammal. 2006, 87, 643–662. [Google Scholar] [CrossRef] [PubMed]
- Castiglia, R. Sympatric sister species in rodents are more chromosomally differentiated than allopatric ones: Implications for the role of chromosomal rearrangements in speciation. Mammal Rev. 2013. [Google Scholar] [CrossRef]
- Soldatović, B.; Živković, B.; Savić, I.; Milošević, M. Comparative karyotype analysis of two populations of Spalax leucodon Nordmann, 1840. Arch. Biol. Nauka 1966, 18, 15–16. [Google Scholar]
- Savić, I.; Soldatović, B. Die Verbreitung der Karyotypen der Blindmaus Spalax (Mesospalax) in Jugoslawien. Arch. Biol. Nauka 1974, 26, 115–122. [Google Scholar]
- Zima, J. Comparative cytogenetics of Palaearctic mammals. Folia Zool. 1993, 42, 97–104. [Google Scholar]
- Zima, J. Chromosomal evolution in small mammals (Insectivora, Chiroptera, Rodentia). Hystrix 2000, 11, 5–15. [Google Scholar]
- Ferguson-Smith, M.A.; Trifonov, V. Mammalian karyotype evolution. Nat. Rev. Genet. 2007, 8, 950–962. [Google Scholar] [CrossRef] [PubMed]
- Romanenko, S.A.; Perelman, P.L.; Trifonov, V.A.; Graphodatsky, A.S. Chromosomal evolution in Rodentia. Heredity 2012, 108, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Topachevski, V.A. The fauna of the USSR: Mammals, mole rats, Spalacidae. Nauka Leningr. 1969, 3, 1–308. (In Russian) [Google Scholar]
- Lyapunova, E.A.; Vorontsov, N.N.; Martynova, L. Cytological differentiation of burrowing mammals in the Palaearctic. In Symposium Theriologicum II; Kratochvíl, J., Obrtel, R., Eds.; Academia: Prague, Czech Republic, 1974; pp. 203–215. [Google Scholar]
- Gromov, I.M.; Baranova, G.I. Catalogue of Mammals in USSR. Nauka Leningr. 1981, 1–455. [Google Scholar]
- Savić, I.R. Familie Spalacidae Gray, 1821—Blindmäuse. In Handbuch der Säugetiere Europas 2/I; Niethammer, J., Krapp, F., Eds.; Akademische Verlagsgesellschaft: Wiesbaden, Germany, 1982; pp. 539–584. [Google Scholar]
- Kryštufek, B.; Ivanitskaya, E.; Arslan, A.; Arslan, E.; Bužan, E.V. Evolutionary history of mole rats (genus Nannospalax) inferred from mitochondrial cytochrome b sequences. Biol. J. Linn. Soc. 2012, 105, 446–455. [Google Scholar] [CrossRef]
- Chişamera, G.; Bužan, E.V.; Sahlean, T.; Murairu, D.; Zupan, S.; Kryštufek, B. Bukovina blind mole rat Spalax graecus revisited: Phylogenetics, morphology, taxonomy, habitat associations and conservation. Mammal Rev. 2014, 44, 19–29. [Google Scholar] [CrossRef]
- Avivi, A.; Ashur-Fabian, O.; Amariglio, N.; Nevo, E.; Rechavi, G. p53—A Key Player in Tumoral and Evolutionary Adaptation, A Lesson from the Israeli Blind Subterranean Mole Rat. Cell Cycle 2005, 4, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Gorbunova, V.; Hine, C.; Tian, X.; Ablaeva, J.; Gudkov, A.V.; Nevo, E.; Seluanov, A. Cancer resistance in the blind mole rat is mediated by concerted necrotic cell death mechanism. Proc. Natl. Acad. Sci. USA 2012, 109, 19392–19396. [Google Scholar] [CrossRef] [PubMed]
- Azpurua, J.; Seluanov, A. Long-lived cancer resistant rodents as new model species for cancer research. Front. Genet. 2013. [Google Scholar] [CrossRef] [PubMed]
- Manov, I.; Hirsh, M.; Iancu, T.C.; Malik, A.; Sotnichenko, N.; Band, M.; Avivi, A.; Shams, I. Pronounced cancer resistance in a subterranean rodent, the blind mole-rat, Spalax: In vivo and in vitro evidence. BMC Biol. 2013, 11. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.D.; Nevo, E.; Han, L.J.; Levanon, E.Y.; Zhao, J.; Avivi, A.; Larkin, D.; Jiang, X.; Feranchuk, S.; Zhu, Y.; et al. Genome-wide adaptive complexes to underground stresses in blind mole rats. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Nevo, E. Mosaic Evolution of Subterranean Mammals: Regression, Progression and Global Convergence; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Avivi, A.; Gerlach, F.; Joel, A.; Reuss, S.; Burmester, T.; Nevo, E.; Hankeln, T. Neuroglobin, cytoglobin, and myoglobin contribute to hypoxia adaptation of the subterranean mole rat Spalax. Proc. Natl. Acad. Sci. USA 2010, 107, 21570–21575. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.; Korol, A.; Hübner, S.; Hernandez, A.G.; Thimmapuram, J.; Ali, S.; Glasser, F.; Paz, A.; Avivi, A.; Band, M. Transcriptome Sequencing of the Blind Subterranean Mole Rat, Spalax galili: Utility and Potential for the Discovery of Novel Evolutionary Patterns. PLoS ONE 2011, 6, e21227. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.; Hangmann, J.; Shams, I.; Avivi, A.; Hankeln, T. Molecular evolution of antioxidant and hypoxia response in long-lived, cancer-resistant blind mole rats: The Nrf2-Keap1 pathway. Gene 2015. [Google Scholar] [CrossRef] [PubMed]
- Nasser, N.J.; Avivi, A.; Shafat, I.; Edovitsky, E.; Zcharia, E.; Ilan, N.; Vlodavsky, I.; Nevo, E. Alternatively spliced Spalax heparanase inhibits extracellular matrix degradation, tumor growth, and metastasis. Proc. Natl. Acad. Sci. USA 2009, 106, 2253–2258. [Google Scholar] [CrossRef] [PubMed]
- Ashur-Fabian, O.; Avivi, A.; Trakhtenbrot, L.; Adamsky, K.; Cohen, M.; Kajakaro, G.; Joel, A.; Amariglio, N.; Nevo, E.; Rechavi, G. Evolution of p53 in hypoxia-stressed Spalax mimics human tumor mutation. Proc. Natl. Acad. Sci. USA 2004, 101, 12236–12241. [Google Scholar] [CrossRef] [PubMed]
- Shams, I.; Malik, A.; Manov, I.; Joel, A.; Band, M.; Avivi, A. Transcription Pattern of p53-targeted DNA Repair Genes in the Hypoxia-Tolerant Subterranean Mole Rat Spalax. J. Mol. Biol. 2013, 425, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Avivi, A.; Oster, H.; Joel, A.; Beiles, A.; Albrecht, U.; Nevo, E. Circadian genes in a blind subterranean mammal. III. Molecular cloning and circadian regulation of cryptochrome genes in the blind subterranean mole rat, Spalax ehrenbergi superspecies. J. Biol. Rhythm. 2004, 19, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Savić, I.; Soldatović, B. Karyotype evolution and taxonomy of the genus Nannospalax Palmer, 1903, Mammalia, in Europe. Serbian Acad. Sci. Arts Sep. Ed. 1984, 59, 1–104. [Google Scholar]
- Nevo, E. Stress, adaptation, and speciation in the evolution of the blind mole rat, Spalax, in Israel. Mol. Phylogenet. Evol. 2013, 66, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Arslan, A.; Kryštufek, B.; Matur, F.; Zima, J. Review of chromosome races in blind mole rats (Spalax and Nannospalax). Folia Zool. 2016, 65, 249–301. [Google Scholar]
- Kryštufek, B.; Amori, G. Nannospalax leucodon. The IUCN Red List of Threatened Species 2017, e.T14328A113301882. Available online: http://dx.doi.org/10.2305/IUCN.UK.2017-1.RLTS.T14328A113301882.en (accessed on 20 October 2017).
- Németh, A.; Révay, T.; Hegyeli, Z.; Farkas, J.; Czabán, D.; Rózsás, A.; Csorba, G. Chromosomal forms and risk assessment of Nannospalax (superspecies leucodon) (Mammalia: Rodentia) in the Carpathian Basin. Folia Zool. 2009, 58, 349–361. [Google Scholar]
- Csorba, G.; Krivek, G.; Sendula, T.; Homonnay, Z.G.; Hegyeli, Z.; Sugár, S.; Farkas, J.; Stojnić, N.; Németh, A. How can scientific researches change conservation priorities? A review of decade-long research on blind mole-rats (Rodentia: Spalacinae) in the Carpathian Basin. Therya 2015, 6, 103–121. [Google Scholar] [CrossRef]
- Savić, I.; Nevo, E. The Spalacidae: Evolutionary history, speciation, and population biology. In Evolution of Subterranean Mammals at the Organismal and Molecular Levels; Nevo, E., Reig, O.A., Eds.; Alan R. Liss, Inc.: New York, NY, USA, 1990; pp. 129–153. [Google Scholar]
- Nevo, E.; Ivanitskaya, E.; Beiles, A. Adaptive Radiation of Blind Subterranean Mole Rats: Naming and Revisiting the Four Siblingspecies of the Spalax ehrenbergi Superspecies in Israel: Spalax galili (2n = 52), S. golani (2n = 54), S. carmeli (2n = 58) and S.judaei (2n = 60); Bachkhuys Publishers: Leiden, The Netherlands, 2001. [Google Scholar]
- Savić, I.; Soldatović, B. Contribution to the study of ecogeographic distribution and evolution of chromosomal forms of the Spalacidae from the Balkan Peninsula. Arch. Biol. Nauka 1977, 29, 141–156. (In Serbian) [Google Scholar]
- Nevo, E. Mode, tempo, and pattern of evolution in subterranean mole rats of the Spalax ehrenbergi superspecies in the Quaternary of Israel. Quat. Int. 1993, 19, 13–19. [Google Scholar] [CrossRef]
- Sözen, M.; Matur, F.; Çolak, E. Some karyological records and a new chromosomal form for Spalax (Mammalia: Rodentia) in Turkey. Folia Zool. 2006, 55, 247–256. [Google Scholar]
- Kryštufek, B.; Vohralík, V. Mammals of Turkey and Cyprus. Rodentia II. Cricetinae, Muridae, Spalacidae, Calomyscidae, Capromyidae, Hystricidae, Castoridae; Knjižnica Annales Majora: Koper, Slovenia, 2009. [Google Scholar]
- De Bruijn, H. Remains of the mole rat Microspalax odessanus Topachevski from Karaburun (Greece, Makedonia) and the family Spalacidae. Proc. Koninkl. Ned. Akad. Wet. Ser. B 1984, 87, 417–425. [Google Scholar]
- Németh, A.; Krnács, G.; Krizsik, V.; Révay, T.; Czabán, D.; Stojnić, N.; Farkas, J.; Csorba, G. European rodents on the edge: Status and distribution of the Vojvodina blind mole rat. Springerplus 2013, 2. [Google Scholar] [CrossRef] [PubMed]
- Ünay, E. On fossil Spalacidae (Rodentia). In The evolution of Western Eurasian Neogene Mammal Faunas; Bernor, R.L., Fahlbush, V., Mittmann, H.W., Eds.; Columbia University Press: New York, NY, USA, 1996; pp. 246–252. [Google Scholar]
- De Bruijn, H.; Bosma, A.A.; Wessels, W. Are the Rhizomyinae and the Spalacinae closely related? Contradistinctive conclusions between genetics and palaeontology. Palaeobio Palaeoenv. 2015, 95, 257–269. [Google Scholar] [CrossRef]
- Bolliger, T. Family Anomalomyidae. In The Miocene Land Mammals of Europe; Rössner, G.E., Heissig, K., Eds.; Verlag Dr. Friederich Pfeil: München, Germany, 1999; pp. 389–394. [Google Scholar]
- Şen, Ş.; Sarica, N. Middle-Late Miocene Spalacidae (Mammalia) from Western Anatolia, and the Phylogeny of the Family. Yerbilimleri 2011, 32, 21–50. [Google Scholar]
- de Bruijn, H.; Marković, Z.; Wilma, W. Late Oligocene rodents from Banovići (Bosnia and Herzegovina). Palaeodiversity 2013, 6, 63–105. [Google Scholar]
- De Bruijn, H. A new stage in the evolution of the mole rats (Rodentia, Spalacinae) from the Early Miocene of Northern Greece. Hist. Biol. 2017, 29, 571–575. [Google Scholar] [CrossRef]
- Hofmeijer, K.G.; de Bruijn, H. The mammals from the Lower Miocene of Aliveri (Island of Evia, Greece). Part 4, The Spalacidae and Anomalomyidae. Proc. Koninkl. Ned. Akad. Wet.Ser. B. 1985, 88, 185–198. [Google Scholar]
- Hadid, Y.; Németh, A.; Snir, S.; Pavlíček, T.; Csorba, G.; Kázmér, M.; Major, A.; Mezhzherin, S.; Rusin, M.; Coşkun, Y.; et al. Is evolution of blind mole rats determined by climate oscillations? PLoS ONE 2012, 7, e30043. [Google Scholar] [CrossRef] [PubMed]
- Vorontsov, N.N.; Martynova, L.N.; Fomicheva, I.I. An electrophoretic comparison of the blood proteins in mole rats of the fauna of the USSR (Spalacinae, Rodentia). Zool. Zh. 1977, 56, 1207–1215. (In Russian) [Google Scholar]
- Németh, A.; Homonnay, Z.G.; Krizsik, V.; Csorba, M.; Pavlíček, T.; Hegyeli, Z.; Hadid, Y.; Sugár, S.; Farkas, J.; Csorba, G. Old views and new insights: Taxonomic revision of the Bukovina blind mole rat, Spalax graecus (Rodentia: Spalacinae). Zool. J. Linn. Soc. 2013, 169, 903–914. [Google Scholar] [CrossRef]
- Wahrman, J.; Goitein, R.; Nevo, E. Mole rat Spalax: Evolutionary significance of chromosome variation. Science 1969, 164, 82–84. [Google Scholar] [CrossRef] [PubMed]
- Matthey, R. Formules chromosomiques de Muridae et de Spalacidae. La question du polymorphisme chromosomique chez les Mammifères. Rev. Suisse Zool. 1959, 66, 175–209. [Google Scholar] [CrossRef]
- Walknowska, J. Les chromosomes chez Spalax leucodon Nordm. Folia Biol. (Kraków) 1963, 11, 30–48. [Google Scholar]
- Harrison, D.L.; Bates, P.J.J. The Mammals of Arabia; Harrison Zoological Museum: Kent, UK, 1991. [Google Scholar]
- Nevo, E.; Filippucci, M.G.; Redi, C.; Simson, S.; Heth, G.; Beiles, A. Karyotype and genetic evolution in speciation of subterranean mole rats of the genus Spalax in Turkey. Biol. J. Linn. Soc. 1995, 54, 203–229. [Google Scholar] [CrossRef]
- Nevo, E.; Filippucci, M.G.; Beiles, A. Genetic polymorphisms in subterranean mammals (Spalax ehrenbergi superspecies) in the Near East revisited: Patterns and theory. Heredity 1994, 72, 465–487. [Google Scholar] [CrossRef] [PubMed]
- Arslan, A.; Zima, J. Karyotypes of the mammals of Turkey and neighboring regions: A review. Folia Zool. 2014, 63, 1–62. [Google Scholar] [CrossRef]
- Savić, I.; Soldatović, B. Distribution range and evolution of chromosomal forms in the Spalacidae of the Balkan Peninsula and bordering regions. J. Biogeogr. 1979, 6, 363–374. [Google Scholar] [CrossRef]
- Arslan, A.; Zima, J. Heterochromatin distribution and localization of NORs in the 2n = 48 cytotypes of Nannospalax xanthodon and N. ehrenbergi. Turk. J. Zool. 2017, 41, 390–396. [Google Scholar] [CrossRef]
- Raicu, P.; Bratosin, S.; Hamar, M. Study on the karyotype of Spalax leucodon Nordm. and S. microphthalmus Güld. Caryologia 1968, 21, 127–135. [Google Scholar] [CrossRef]
- Raicu, P.; Duma, D. Cytogenetical study in Sp. leucodon in Moldavia (Romania). Genet. Res. 1969, 13, 99–104. [Google Scholar] [CrossRef]
- Peshev, D.T.; Vorontsov, N.N. Chromosomal variability in the mole rats Nannospalax leucodon Nordmann complex in Bulgaria. In Proceedings of the Third Intern. Theriological Congress Abstract Book, Helsinki, Finland, 1982; Myllymaki, A., Pulliainen, P., Eds.; p. 190. [Google Scholar]
- Kandemir, I.; Sözen, M.; Matur, F.; Kankılıç, T.; Martínková, N.; Çolak, F.; Özkurt, S.Ö.; Çolak, E. Phylogeny of species and cytotypes of mole rats (Spalacidae) in Turkey inferred from mitochondrial cytochrome b sequences. Folia Zool. 2012, 61, 25–33. [Google Scholar]
- Hausser, J.; Fedyk, S.; Fredga, K.; Searle, J.B.; Volobouev, V.; Woicik, J.M.; Zima, J. Definition and nomenclature of the chromosome races of Sorex araneus. Folia Zool. 1994, 43 (Suppl. I), 1–9. [Google Scholar]
- Petrova, T.V.; Tesakov, A.S.; Kowalskaya, Y.M.; Abramson, N.I. Cryptic speciation in the narrow-headed vole, Lasiopodomys (Stenocranius) gregalis, (Rodentia: Cricetidae). Zool. Scr. 2016, 45, 618–629. [Google Scholar] [CrossRef]
- Soldatović, B. Cytogenetic Study of the Speciation of the Genus Spalax in Yugoslavia. Ph.D. Thesis, University of Beograd, Belgrade, Serbia, 1971. (In Serbian). [Google Scholar]
- Soldatović, B. Karyotype analysis and cytogenetic aspects of speciation in the genus Spalax. Zbornik Prir. Nauk. Mat. Srp. 1977, 52, 5–58. (In Serbian) [Google Scholar]
- Soldatović, B.; Savić, I. New karyotype form of the mole rat (Spalax Güld.). Arch. Biol. Nauka 1973, 25, 13–14. [Google Scholar]
- Savić, I.R.; Rempe, U. Vergleichende kraniometrische Untersuchungen über Vertrerer der Gattung Spalax (Microspalax) auf der Balkan-Halbinsel. Vortrag zur 51. In Proceedings of the Hauptversammlung der Deutschen Gesellschaft für Säugetierkunde, Bamberg, Germany, 18–23 September 1977. [Google Scholar]
- Peshev, D. On the caryotypes in some populations of the mole rat (Spalax leucodon Nordmann) in Bulgaria. Zool. Anz. 1981, 206, 129–131. [Google Scholar]
- Sözen, M. A karyological study on subterranean mole rats of the Spalax leucodon Nordmann, 1840 superspecies in Turkey. Mamm. Biol. 2004, 64, 420–429. [Google Scholar] [CrossRef]
- Sözen, M.; Çolak, F.; Sevindik, M.; Matur, F. Cytotypes of Nannospalax xanthodon (Satunin, 1898) (Rodentia, Spalacidae) from western Anatolia. Turk. J. Zool. 2013, 37, 462–469. [Google Scholar] [CrossRef]
- Kankılıç, T.; Kankılıç, T.; Sözen, M.; Çolak, E. Genetic Diversity and Geographic Variation of Chromosomal races of Nannospalax xanthodon (Nordmann, 1840) and Nannospalax ehrenbergi (Nehring, 1898) from Turkey, Revealed by RAPD Analysis. Acta Zool. Bulg. 2013, 65, 45–58. [Google Scholar]
- Coşkun, Y.; Ulutürk, S.; Kaya, A. Karyotypes of Nannospalax (Palmer 1903) populations (Rodentia: Spalacidae) from centraleastern Anatolia, Turkey. Hystrix 2010, 21, 89–96. [Google Scholar]
- Ivanitskaya, E.; Rashkovetsky, L.; Nevo, E. Chromosomes in a hybrid zone of Israeli mole rats (Spalax, Rodentia). Russ. J. Genet. 2010, 46, 1149–1151. [Google Scholar] [CrossRef]
- Nevo, E.; Bar-El, H. Hybridization and speciation in fossorial mole rats. Evolution 1976, 30, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Ivanitskaya, E.; Coskun, Y.; Nevo, E. Banded karyotypes of mole rats (Spalax, Spalacidae, Rodentia) from Turkey. J. Zool. Syst. Evol. Res. 1997, 35, 171–177. [Google Scholar] [CrossRef]
- Ivanitskaya, E.; Sözen, M.; Rashkovetsky, L.; Matur, F.; Nevo, F. Discrimination of 2n = 60 Spalax leucodon cytotypes (Spalacidae, Rodentia) in Turkey by means of classical and molecular cytogenetic techniques. Cytogenet. Genome Res. 2008, 122, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Matur, F.; Çolak, F.; Ceylan, T.; Sevindik, M.; Sözen, M. Chromosomal evolution of the genus Nannospalax (Palmer 1903) (Rodentia, Muridae) from western Turkey. Turk. J. Zool. 2013, 37, 470–487. [Google Scholar] [CrossRef]
- Arslan, A.; Akan, Ş.; Zima, J. Variation in C-heterochromatin and NORs distribution among chromosomal races of mole rats (Spalacidae) from Central Anatolia, Turkey. Mamm. Biol. 2011, 76, 28–35. [Google Scholar] [CrossRef]
- Nevo, E.; Ivanitskaya, E.; Filippucci, M.G.; Beiles, A. Speciation and adaptive radiation of subterranean mole rats, Spalax ehrenbergi superspecies, in Jordan. Biol. J. Linn. Soc. 2000, 69, 263–281. [Google Scholar] [CrossRef]
- Matur, F.; Çolak, F.; Sevindik, M.; Sözen, M. Chromosome differentiation of four 2n = 50 chromosomal forms of Turkish mole rat. Nannospalax nehringi. Zool. Sci. 2011, 28, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Ivanitskaya, E.; Belyayev, A.; Nevo, E. Heterochromatin differentiation shows the pathways of karyotypic evolution in Israeli mole rats (Spalax, Spalacidae, Rodentia). Cytogenet. Genome Res. 2005, 111, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Zima, J.; Macholán, M.; Kryštufek, B.; Petkovski, S. Karyotypes of certain small mammals (Insectivora, Rodentia) from Makedonia. Scopolia 1997, 38, 1–15. [Google Scholar]
- Savic, I.R. Diversification of the Balkan fauna: Its origin, historical development and present status. In Advances in Arachnology and Developmental Biology Papers dedicated to Prof. Dr. Božidar Ćurčić; Makarov, S.E., Dimitrijević, R.N., Eds.; Inst. Zool.: Belgrade, Serbia; BAS: Sofia, Belgrade; Fac. Life Sci.: Vienna, Austria; SASA: Belgrade, Serbia; UNESCO MAB: Belgrade, Serbia, 2008; Volume 12, pp. 57–78. [Google Scholar]
- Arslan, E.; Gülbahçe, E.; Arıkoğlu, H.; Arslan, A.; Bužan, E.; Kryštufek, B. Mitochondrial divergence between three cytotypes of the Anatolian mole rat, Nannospalax xanthodon (Nordmann, 1849). Zool. Middle East 2010, 50, 27–34. [Google Scholar] [CrossRef]
- Reyes, A.; Nevo, E.; Saccone, C. DNA sequence variation in the mitochondrial control region of subterranean mole rats, Spalax ehrenbergi superspecies, in Israel. Mol. Biol. Evol. 2003, 20, 622–632. [Google Scholar] [CrossRef] [PubMed]
- Yüksel, E.; Gülkaç, M.D. On the karyotypes of some populations of the subterranean mole rats in the lower Euphrates basin, Turkey. Caryologia 1992, 45, 175–190. [Google Scholar] [CrossRef]
- Catzeflis, F.M.; Nevo, E.; Ahlquist, J.E.; Sibley, C. Relationships of the chromosomal species in the Eurasian mole rats of the Spalax ehrenbergi group as determined by DNA–DNA hybridization and an estimate of the spalacid-murid divergence time. J.Mol. Evol. 1989, 29, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Nižetić, D.; Stevanović, M.; Soldatović, B.; Savić, I.; Crkvenjakov, R. Limited polymorphism of both classes of MHC genes in four different species of the Balkan mole rat. Immunogenetics 1988, 28, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Nevo, E. Molecular evolution and environmental stress. In Gene Families: Studies of DNA, RNA, Enzymes and Proteins; Xue, G., Xue, Y., Xu, Z., Holmes, R., Hammond, L., Lim, H.A., Eds.; World Scientific Pub. Co.: Singapore, 2001; pp. 73–87. [Google Scholar]
- Avise, J.C. Phylogeography: The History and Formation of Species; Harvard University Press: Cambridge, UK, 2000. [Google Scholar]
- Karanth, K.P.; Avivi, A.; Beharav, A.; Nevo, E. Microsatellite diversity in populations of blind subterranean mole rats (Spalax ehrenbergi superspecies) in Israel: Speciation and adaptation. Biol. J. Linn. Soc. 2004, 83, 229–241. [Google Scholar] [CrossRef]
- Nevo, E.; Beiles, A.; Spradling, T. Molecular evolution of cytochrome b of subterranean mole rats, Spalax ehrenbergi superspecies, in Israel. J. Mol. Evol. 1999, 49, 215–226. [Google Scholar] [CrossRef] [PubMed]
- White, T.A.; Bordewich, M.; Searl, J.B. A network approach to study karyotypic evolution: The chromosomal races of the common shrew (Sorex araneus) and house mouse (Mus musculus) as model systems. Syst. Biol. 2010, 59, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Wójcik, J.M.; Borodin, P.M.; Fedyk, S.; Fredga, K.; Hausser, J.; Mishta, A.; Orlov, V.N.; Searle, J.B.; Volobouev, V.T.; Zima, J. The list of the chromosome races of the common shrew Sorex araneus (updated 2002): The International Sorex araneus Cytogenetics Committee (ISACC). Mammalia 2003, 67, 169–178. [Google Scholar] [CrossRef]
- Piálek, J.; Hauffe, H.C.; Searle, J.B. Chromosomal variation in the house mouse. Biol. J. Linn. Soc. 2005, 84, 535–563. [Google Scholar] [CrossRef]
- Nevo, E.; Beiles, A. Ribosomal DNA nontranscribed spacer polymorphism in subterranean mole rats: Genetic differentiation, environmental correlates, and phylogenetic relationships. Evol. Ecol. 1988, 2, 139–156. [Google Scholar] [CrossRef]
- Kankılıç, T.; Gürpınar, C. Revised classification design of the Anatolian species of Nannospalax (Rodentia: Spalacidae) using RFLP analysis. Turk. J. Zool. 2014, 38, 68–78. [Google Scholar] [CrossRef]
- Kankılıç, T.; Kankılıç, T.; Şeker, P.S.O.; Kıvanç, E. Morphological and biometrical comparisons of the baculum in the genus Nannospalax Palmer, 1903 (Rodentia: Spalacidae) from Turkey with consideration of its taxonomic importance. Turk. J. Zool. 2014, 38, 144–157. [Google Scholar] [CrossRef]
- Dobzhansky, T.; Epling, C. Contribution to the Genetics, Taxonomy and Ecology of Drosophila pseudoobscura and Its Relatives; Carnegie Institution of Washington: Washington, DC, USA, 1944; Volume 544, pp. 1–46. [Google Scholar]
- Mayr, E. Artbegriff und Evolution; Verlag Paul Parey: Hamburg/Berlin, Germany, 1967; p. 617. [Google Scholar]
- Orr, H.A. Dobzhansky, Bateson and the genetics of speciation. Genetics 1996, 144, 1331–1335. [Google Scholar] [PubMed]
- White, M.J.D. Modes of Speciation; WH Freeman: San Francisco, CA, USA, 1978. [Google Scholar]
- Navarro, A.; Barton, N.H. Chromosomal speciation and molecular divergence-accelerated evolution in rearranged chromosomes. Science 2003, 300, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Dobzhansky, T. The Y chromosome of Drosophila pseudoobscura. Genetics 1935, 20, 366–376. [Google Scholar] [PubMed]
- Bush, G.L.; Case, S.M.; Wilson, A.C.; Patton, J.L. Rapid speciation and chromosomal evolution in mammals. Proc. Nat. Acad. Sci. USA 1977, 74, 3942–3946. [Google Scholar] [CrossRef] [PubMed]
- Searle, J.B. Chromosomal hybrid zones in eutherian mammals. In Hybrid Zones and the Evolutionary Process; Harrison, R.G., Ed.; Oxford University Press: Oxford, UK, 1993; pp. 309–353. [Google Scholar]
- Sözen, M.; Çolak, E.; Yiğit, N.; Ozkurt, Ş.; Verimli, R. Contributions to the karyology and taxonomy of the genus Spalax Güldenstaedt, 1770 (Mammalia: Rodentia) in Turkey. Z. Säugetierkd. 1999, 64, 210–219. [Google Scholar]
- Golenishchev, F.N.; Meyer, M.N.; Bulatova, N.S. The hybrid zone between two karyomorphs of Microtus arvalis (Rodentia, Arvicolinae). Proc. Zool. Inst. Russ. Acad. Sci. 2001, 289, 89–94. [Google Scholar]
- Kovalskaya, Y.M.; Savinetskaya, L.E.; Aksenova, T.G. Experimental Hybridization of Voles of the Genus Microtus s.l. M. socialis with Species of the Group arvalis (Mammalia, Rodentia). Biol. Bull. 2014, 41, 559–563. [Google Scholar] [CrossRef]
- Bickford, D.; Lohman, D.J.; Sodhi, N.S.; Peter, K.L.N.; Meier, R.; Winker, K.; Ingram, K.K.; Das, I. Cryptic species as a window on diversity and conservation. Trends Ecol. Evol. 2006, 22. [Google Scholar] [CrossRef] [PubMed]
- Faria, R.; Navarro, A. Chromosomal speciation revisited: Rearranging theory with pieces of evidence. Trends Ecol. Evol. 2010, 25. [Google Scholar] [CrossRef] [PubMed]
- Cvijić, J. Die Tektonik der Balkanhalbinsel; Comptes Rendus, IX Congrès International de Géographie de la France: Paris, France, 1904; Volume 5, pp. 247–254. [Google Scholar]
- Yang, F.; Carter, N.P.; Shi, L.; Ferguson-Smith, M.A. A comparative study of karyotypes of muntjacs by chromosome painting. Chromosoma 1995, 103, 642–652. [Google Scholar] [CrossRef] [PubMed]
- Peshev, D. Craniological study of the species of the genus Spalax (Spalacidae, Mammalia) I. Sex Dimophism. Zool. Anz. 1989, 222, 83–91. [Google Scholar]
- Nevo, E.; Shaw, C.R. Genetic variation in a subterranean mammal, Spalax ehrenbergi. Biochem. Genet. 1972, 7, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; O’Brien, P.C.; Weinbnerg, J.; Ferguson-Smith, M.A. A reappraisal of the tandem fusion theory of karyotype evolution in the Indian muntjac using chromosome painting. Chromosome Res. 1997, 5, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Qiu, L.; Mrasek, K.; Zhang, J.; Liehr, T.; Quintana, L.G.; Li, Z. Common Fragile Sites: Genomic Hotspots of DNA Damage and Carcinogenesis. Int. J. Mol. Sci. 2012, 13, 11974–11999. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Herrera, A.; Castresana, J.; Robinson, T.J. Is mammalian chromosomal evolution driven by regions of genome fragility? Genome Biol. 2006, 7, R115. [Google Scholar] [CrossRef] [PubMed]
- Ferguson-Smith, M.A.; Yang, F.; O’Brien, P.C. Comparative mapping using chromosome sorting and painting. ILAR J. 1998, 39, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Stanyon, R.; Yang, F.; Cavagne, P.; O’Brien, P.C.M.; Bagga, M.; Ferguson-Smith, M.A.; Wienberg, J. Reciprocal chromosome painting shows that genomic rearrangement between rat and mouse proceeds ten times faster than between humans and cats. Cytogenet. Cell Genet. 1999, 84, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; Nishida-Umehara, C.; Kuroiwa, A.; Tsuchiya, K.; Matsuda, Y. Identification of chromosome rearrangements between the laboratory mouse (Mus musculus) and the Indian spiny mouse (Mus platythrix) by comparative FISH analysis. Chromosome Res. 2003, 11, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Deuve, J.L.; Bennett, N.C.; Britton-Davidian, J.; Robinson, T.J. Chromosomal phylogeny and evolution of the African mole-rats (Bathyergidae). Chromosome Res. 2008, 16, 57–74. [Google Scholar] [CrossRef] [PubMed]
- Liehr, T.; Starke, H.; Heller, A.; Kosyakova, N.; Mrasek, K.; Gross, M.; Karst, C.; Steinhaeuser, U.; Hunstig, F.; Fickelscher, I.; et al. Multicolor fluorescence in situ hybridization (FISH) applied to FISH-banding. Cytogenet. Genome Res. 2006, 114, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Liehr, T.; Heller, A.; Starke, H.; Claussen, U. FISH banding methods: Applications in research and diagnostics. Expert. Rev. Mol. Diagn. 2002, 2, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Weise, A.; Mrasek, K.; Fickelscher, I.; Claussen, U.; Cheung, S.W.; Cai, W.W.; Liehr, T.; Kosyakova, N. Molecular Definition of High-resolution Multicolor Banding Probes: First Within the Human DNA Sequence Anchored FISH Banding Probe Set. J. Histochem. Cytochem. 2008, 56, 487–493. [Google Scholar] [CrossRef] [PubMed]
| CF | 2n | NF | Sampling Localities | Branches [29] | |
|---|---|---|---|---|---|
| 1 | montanoserbicus | 56 | 82 | Tara, Zlatibor, Čajetina, Čigota, Kopaonik, Vlasina-Klisura, Vlasina-Sokolovo, Čakor. | I |
| 2 | hercegovinensis | 54 | 90 | Durmitor, Zelengora, Maglić, Čemerno, Neretva, Gvozd, Njegovuđa. | |
| 3 | syrmiensis | 54 | 90 | Višnjica, B. brdo, Košutnjak, Avala, Jajinci, Smederevo-Udovice, Bogatić (Mačva), St. Pazova. | |
| 4 | hungaricus | 48 | 84 | Hajdukovo, Šušara, Dolovo, Jajinci, Avala. | IIa |
| 5 | transsylvanicus | 50 | 84 | Jucu, Cluj-Napoca region, Transylvania. | |
| 6 | montanosyrmiensis | 54 | 86 | Stražilovo, Čortanovci, Kelebia. | |
| 7 | leucodon | 56 | 84 | Odessa, Orgeev, Bacau, Perieni, Constanta. | |
| 8 | monticola | 54 | 84 | Kupreško Polje, Šuica, Ljubuša Mt. | |
| 9 | makedonicus | 52 | 86 | Jakupica Mt., Karadžica, Pelagonia, Prilep, Bitolj, Ohrid, lake Vegoritis, Arnissa. | IIb |
| 10 | strumiciensis | 54 | 88 | Strumičko Polje (Strumica Valley). | |
| 11 | epiroticus | 56 | 84 | Lefkothea (Epirus, NW Greece). | |
| 12 | thracius | 56 | 88 | Novo Selo, Plovdiv (Bulgaria). | |
| 13 | hellenicus | 58 | 88 | Levadia (Southern Greece). | |
| 14 | serbicus | 54 | 98 | Biskuplje, Rogljevo, Resavska Cave, Ram (Đerdap), Resava, Kladovo, Rtanj Mt., Niš, Pirot, Priština, Katlanovo, T.Veles. | III |
| 15 | ovchepolensis | 54 | 94 | Ovče Polje (Eastern Makedonia). | |
| 16 | tranensis | 54 | 96 | Tran (Western Bulgaria). | |
| 17 | sofiensis | 56 | 90 | Sofia-East, Cherven Briag (Bulgaria). | |
| 18 | rhodopiensis | 54 | 92 | Dobrostan near Asenovgrad (Bulgaria). | |
| 19 | turcicus | 56 | 76–78 | European Turkey, Lower Thrace. | IV |
| 20 | bulgaricus | 46 | 76 | Kozarevets near Veliko Tarnovo, Sliven region, 370 m a.s.l. (Bulgaria). | |
| 21 | srebarnensis | 48 | 78 | Srebarna, right bank of the River Danube, 80 m a.s.l. (Bulgaria). | |
| D | dobrudzha | 54–56 | 78–84 | Dobrudzha (Romania). | |
| L | lom | 54 | 98 | Lom (Bulgaria). | |
| P | pazardzhik | 54 | 86 | Pazardzhik (Bulgaria). | |
| V | varna | 52 | 80 | Varna (Bulgaria). |
| Fossil Findings | Locality | |
|---|---|---|
| I | N. cf. leucodon | West and North Hungary |
| II | N. macoveii | Grebenniki |
| III | N. odessanus | Odessa |
| IV | N. macoveii | Berešti |
| V | N. macoveii | Gavonosy (Gabanoasa) |
| VI | N. macoveii | Malušteni |
| VII | N. cf. leucodon | Golema Lisza peschtera |
| VIII | N. cf. leucodon | Marinova Cave |
| IX | Vetuspalax progressus | Banovići (BandH) |
| X | Debruijinia kostakii | Karydia (Greece) |
| XI | N. macoveii | Serrai |
| XII | Pliospalax tourkobouniensis | Tourkobonnia Hill |
| XIII | N.cf. nehringi | Chios |
| XIV | N.cf. nehringi | Kalymnos |
| XV | Pliospalax sotirisi | Maritsa |
| Female CF | Male CF | Mating | Embryos |
|---|---|---|---|
| hungaricus * | hungaricus * | + | + |
| hungaricus | syrmiensis | + | − |
| hungaricus | montanoserbicus | − | − |
| hungaricus | montanosyrmiensis | + | − |
| syrmiensis | montanosyrmiensis | + | − |
| syrmiensis | montanoserbicus | − | − |
| makedonicus | montanoserbicus | + | − |
| montanoserbicus | serbicus | + | − |
| syrmiensis | serbicus | + | − |
| monticola | hungaricus | + | − |
| monticola | montanosyrmiensis | + | − |
| montanosyrmiensis | syrmiensis | + | − |
| syrmiensis | hungaricus | + | − |
| montanoserbicus * | montanoserbicus * | + | + |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savić, I.; Ćirović, D.; Bugarski-Stanojević, V. Exceptional Chromosomal Evolution and Cryptic Speciation of Blind Mole Rats Nannospalax leucodon (Spalacinae, Rodentia) from South-Eastern Europe. Genes 2017, 8, 292. https://doi.org/10.3390/genes8110292
Savić I, Ćirović D, Bugarski-Stanojević V. Exceptional Chromosomal Evolution and Cryptic Speciation of Blind Mole Rats Nannospalax leucodon (Spalacinae, Rodentia) from South-Eastern Europe. Genes. 2017; 8(11):292. https://doi.org/10.3390/genes8110292
Chicago/Turabian StyleSavić, Ivo, Duško Ćirović, and Vanja Bugarski-Stanojević. 2017. "Exceptional Chromosomal Evolution and Cryptic Speciation of Blind Mole Rats Nannospalax leucodon (Spalacinae, Rodentia) from South-Eastern Europe" Genes 8, no. 11: 292. https://doi.org/10.3390/genes8110292
APA StyleSavić, I., Ćirović, D., & Bugarski-Stanojević, V. (2017). Exceptional Chromosomal Evolution and Cryptic Speciation of Blind Mole Rats Nannospalax leucodon (Spalacinae, Rodentia) from South-Eastern Europe. Genes, 8(11), 292. https://doi.org/10.3390/genes8110292





