1. Introduction
Rod-cone dystrophy (RCD), also known as retinitis pigmentosa, is a heterogeneous group of inherited retinal disorders affecting rod photoreceptors in the majority of cases with secondary cone degeneration [
1]. Population-based studies showed that RCD affects one in 4000 individuals around the world [
1]. Subjects diagnosed with RCD initially complain of night blindness followed by progressive visual field constriction, abnormal color vision and eventually loss of central vision [
1]. These visual symptoms indicate the gradual loss of the two photoreceptor types: rods, which mediate achromatic vision in dim-lit environments and cones, which are important for daylight color vision and fine acuity [
1]. Photoreceptors are post-mitotic sensory neurons that display a polarized structure including a biosynthetically-active inner segment (IS) and a photosensitive outer segment (OS) [
2]. The two segments are connected via a specialized bridge-like structure, the connecting cilium (CC), which represents the transitional zone of a prototypical primary cilium [
2]. The CC is a region with increasing relevance for ciliary homeostasis due to its implication in ciliogenesis [
3] and protein trafficking [
4].
RCD is inherited as a Mendelian trait in most cases [
1]. On the basis of its mode of inheritance and prevalence, RCD can be divided into three groups: autosomal dominant (ad) (15–25%), autosomal recessive (ar) (35–50%) and X-linked (xl) (7–15%) [
5]. To date, mutations in more than 83 genes are implicated in non-syndromic RCD [
6,
7] Of those, at least 15 encode proteins that localize to the CC and/or basal body (BB) of the cell [
8]. Most of the RCD mutant genes account for only a small proportion of cases, with the exception of rhodopsin (
RHO, Mendelian Iheritance in Man (MIM): 180380), usherin (
USH2A, MIM: 608400) and retinitis pigmentosa GTPase regulator (
RPGR, MIM: 312610) in which mutations together account for ~30% of all RCD cases [
1,
6].
Our group has previously identified mutations in a novel gene
KIZ (MIM: 615757), encoding a ciliary centrosomal protein, kizuna. The precise function of this protein, whose defect accounts for 1% of arRCD in the European population [
9], is still unknown. The purpose of the current study was to report additional mutations in
KIZ and further characterize its protein localization.
2. Materials and Methods
2.1. Ethics Statement
The study protocol was conducted in accordance with the declaration of Helsinki, the national guidelines and the regional ethics committee. Informed written consent was obtained from every participant. Human DNA collection for genetic research was project No. EUDRACT 2006-A00347-44, n°06693 and was approved by CPP Ile de France V (regional ethics committee) on the 24th of October 2006. Human skin biopsies to derive cells for research was project n°10820 and was approved by the CPP Ile de France V on the 8th of November 2010 and expanded on the 2nd of October 2012.
All animal procedures were performed according to the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Visual Research and were approved by the French Minister of Agriculture (authorization A-75-1863 delivered on 9 November 2011). All efforts were made to minimize suffering.
2.2. Targeted Next-Generation Sequencing and Validation
The next generation sequencing (NGS) panel was selected from the SureSelect Human All Exon Kits Version 4 (Agilent, Massy, France) and covers 198 IRD genes in total. The eArray web-based probe design tool that was used for this purpose can be found at
https://earray.chem.agilent.com/earray. All probes were designed and synthesized by Agilent Technologies (Santa Clara, CA, USA). Sequence capture, enrichment and elution were performed according to Agilent’s instructions. Identified variants in
KIZ (NM_018474.4) were validated by direct Sanger sequencing, and co-segregation analyses was performed in the DNA of available family members as previously described [
9]. Similarly, the presence of the two mutations c.52G>T, p.Glu18* and c.119_122del, p.Lys40Ilefs*14 in the fibroblast cell line of patient CIC01225 was confirmed by direct Sanger sequencing using genomic primers and conditions as previously described [
9].
2.3. Pathogenic Predictions
To assess the pathogenicity of the novel
KIZ variant, we studied amino acid conservation across 30 species in the UCSC Genome Browser (University of California, Santa Cruz, CA, USA), for which a coding sequence was present [
10]. Pathogenicity was also evaluated on the basis of bio-informatic predictions as Polyphen (polymorphism phenotyping) [
11] and SIFT (sorting intolerant from tolerant) [
12]. In addition, we determined the exact minor allele frequency of the herein identified variants in
KIZ with the Genome Aggregation Database (gnomAD) [
13] or a software (Alamut v2.7.1, Interactive Biosoftware, Rouen, France).
2.4. RNA Extraction and Real Time PCR Analysis
In total, we investigated four whole blood samples, among which three were controls and one corresponding to the affected individual: CIC01225 (compound heterozygous for the c.52G>T (p.Glu18*) and c.119_122del (p.Lys40Ilefs*14) mutations in KIZ). Two samples of human fibroblasts (unaffected vs. CIC01225) were also included. Total RNA was isolated from whole blood cells and fibroblasts of CIC01225, Family C (737), using a kit (RNeasy Mini Kit, Qiagen, Hilden, Germany), and 500 ng were used to synthesize cDNA with a reverse transcriptase (SuperScript®II, Invitrogen, Carlsbad, Czech Republic), according to the manufacturer’s protocol. Quantitative real-time polymerase chain reaction (PCR) was performed using a PCR system (Applied Biosystems 7500 Real-Time, Life technologies) with a kit (Power SYBR Green kit, Applied Biosystems, Life technologies) for KIZ and 18S transcripts. Specific primers were designed in exons 6 and 7 of KIZ (6F: 5′-CAGAACCACAGCCAAATCCA-3′; and 7R: 5′-TCTGAGTCTGGTGTTTGTCC-3′) spanning intron 6. All experiments were carried out in triplicates in a total reaction volume of 25 µL containing 2.5 ng of cDNA. KIZ mRNA levels were normalized to 18S rRNA. To test for the expression of the different exons of KIZ in blood, fibroblasts and retina, PCR-experiments on cDNA were performed with primers located in exons 1–4 (1F: 5′-GTCTCCTTCGGCAACCCC-3′; and 4R: 5′-TGTCTTCATCTGTCAGTTCCTC -3′), in exons 4–6 (4F: (5′-GAGGAACTGACAGATGAAGAC-3′; and 6R: 5′-CCTGGATTTGGCTGTGGTTC-3′) and in exons 6–13 (6F: (5′-CAGAACCACAGCCAAATCCA-3′; and 13R: 5′-AGTTACTGTCATCAGACTCATCC-3′). All experiments were carried out in a total reaction volume of 12.5 µL using 3 µL of 1:10 diluted cDNA. The specificity of all PCR products was first verified by electrophoresis on 2% agarose gel and subsequently by Sanger sequencing.
2.5. Statistical Analysis
Statistical analyses were conducted using SPSS software Version 20 (SPSS, Inc., Chicago, IL, USA). Since normalized KIZ expression and cilium length data are arbitrarily distributed, we used a Mann–Whitney U test, a non-parametric test that compares the medians. This is the reason behind representing the data in a box plot format where the median (bar), the maximum and minimum values and the quartiles are shown. To investigate whether the compound mutation in KIZ decreased the percentage of ciliated cells, we performed a chi-square test using two categorical variables: ciliated cell (yes and no) vs. individual (control and CIC01225) in ~60 fibroblast cells equally divided as control and affected. The threshold for statistical significance was set at a p-value of <0.05.
2.6. Antibodies and Microscopy
Anti-acetylated α-tubulin mouse monoclonal antibody (T7451), anti-γ-tubulin mouse monoclonal antibody (T6557) and anti-c-myc mouse monoclonal antibody (11667149001) were purchased from companies (Sigma-Aldrich, St. Quentin Fallavier, France for the first two; Roche, Basel, Switzerland, for the last). Mouse monoclonal anti-rhodopsin antibody (MAB5316) and Lectin PNA conjugate 594 (L-32460) were purchased from two other companies (Millipore, Guyancourt, France and Invitrogen, respectively). Secondary donkey antibodies anti-rabbit and anti-mouse were conjugated with Alexa Fluor 488 (711-545-152, Jackson Immuno Research Laboratories, Baltimore, MD, USA) and Cy3 (715-165-150, Jackson Immuno Research) or with Alexa Fluor 647 (715-605-150, Jackson Immuno Research) fluorophores, respectively. The 40,6-diamidino-2-phenylindole (DAPI) was purchased from a company (Euromedex, Souffelweyersheim, France). In addition, anti-mouse and anti-rabbit horse radish peroxidase (HRP)-antibodies (715-035-150 and 711-545-152, respectively) (Jackson Immuno Research Baltimore, MD) were used for Western blot analysis. Confocal image stacks were performed on an Olympus FV1200 laser-scanning confocal microscope (Olympus, Rungis, France). DAPI counterstaining, AlexaFluor-594 and AlexaFluor-488 immunolabeling were excited by using 405- and 559-nm laser diodes lines and 488–515-nm argon ion laser lines, respectively. The objectives used was an Olympus 40x NA1.30-WD 0.20 or PLAPON 60x SC NA 1.40 WD 0.12. Control of the microscope and image acquisition was conducted by using Olympus Fluoview software Version 4.2. Image acquisition was conducted at a resolution of 1024 × 1024 pixels, with a scan rate of 8–10 μs·pixel−1 and with or without zoom. Images were acquired sequentially, line by line, in order to reduce excitation and emission crosstalk, and step size was defined according to the Nyquist–Shannon sampling theorem. Exposure settings that minimized oversaturated pixels in the final images were used. Twelve-bit images were processed with ImageJ or FIJI (National Institutes of Health, Bethesda, MD, USA) and finally converted in 24-bit RGB color mode. Figures were then assembled by using the Adobe Photoshop CC and Adobe Illustrator CC softwares (Adobe Systems, Mountain View, CA, USA).
2.7. Antibody Tests
The human KIZ and its mouse ortholog PLK1S1 are 66% identical. Therefore, we used the following species-specific antibodies. The anti-PLK1S1 antibody, which was a gift of Blanche Capel (Duke University Medical Center, Durham, NC, USA) [
14], is a rabbit polyclonal antibody produced against the 3’ end of mouse PLK1S1. The ability of this antibody to detect PLK1S1 was demonstrated in the work of Tang et al. [
14]. At that time, murine
Plk1s1 was called
Gm114. Using the Western blot experiment on adult wild-type mouse testis lysates, anti-PLK1S1 recognized a 76-kDa protein, the predicted size of the full-length protein [
14], which was absent in homozygous mutants. In addition, the GM114 protein was also not detected by immunocytochemistry in the mutant adult testis, while it shows a nice immunolocalization in wild-type mice [
14]. Therefore, we considered this antibody to be able to detect PLK1S1.
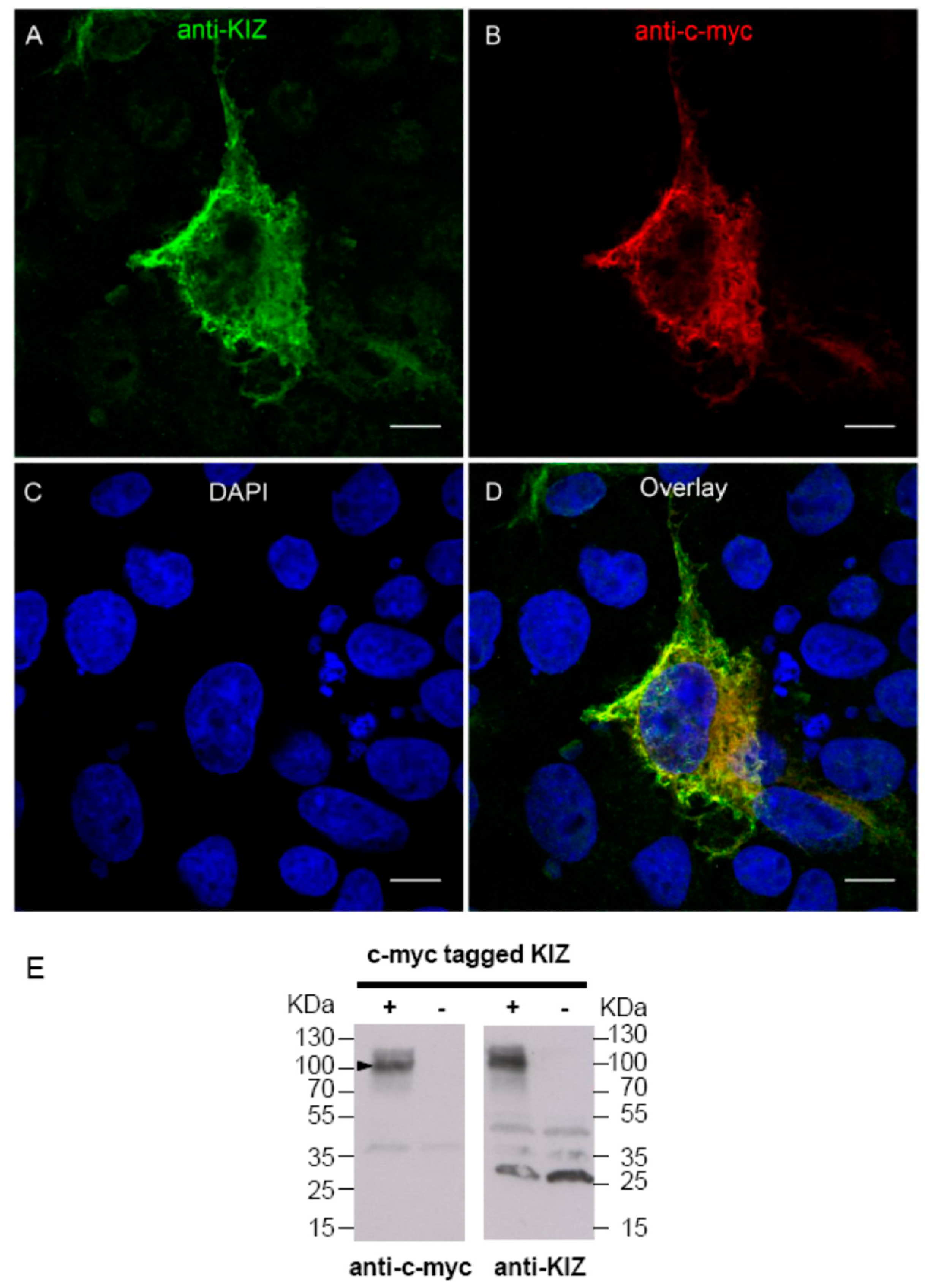
The anti-KIZ antibody is a commercially available rabbit polyclonal antibody (SAB2700541, Sigma-Aldrich) produced with a recombinant human KIZ fragment corresponding to the region between amino acids 187 and 438 of the human protein. To check if it is indeed able to detect the human protein, we overexpressed a c-myc tagged KIZ construct in COS-1 cells. In detail: a pBudCE4.1 plasmid (GeneCust, Dubelange, Luxembourg) containing coding exons of the human KIZ sequence (reference NM_018474.4) tagged with the c-myc epitope was synthesized. To test if the anti-KIZ antibody is indeed able to detect the respective protein by immunolocalization studies, the following procedures were performed:
In 24-well plates, 175,000 COS-1 cells per well were seeded on coated coverslips and transfected after 6 h with this plasmid, applying the calcium chloride method [
15]: a solution of 750 ng of plasmid diluted in a solution of 0.1 M calcium chloride in 2 ×
N-2-hydroxyethylpiperazine-
N'-2-ethanesulfonic acid (HEPES) buffered saline were deposited in each well. To test anti-KIZ antibody, 48 h after transfection, COS-1 cells were fixed in 4% paraformaldehyde (PFA) for 5 min, permeabilized with 1× phosphate buffered saline (PBS) , 0.2% Triton X100 solution for 15 min, and then, intracellular staining was performed applying anti-KIZ rabbit (1:50) and anti-c-myc mouse (1:500) (11667149001, Roche, Basel, Switzerland) antibodies in 1 × PBS, 0.1% gelatine, 0.05% Tween 20, 0.1% bovine serum albumin (BSA). We pre-incubated the primary antibody solution with crude COS-1 cells (1:100) for 15 min at 37 °C and deposited this antibody solution on transfected cells for 1 h at room temperature (RT). Secondary antibody solution, donkey anti-rabbit conjugated with Alexa Fluor 488 and donkey anti-mouse with Cy3 (1:1000) in 1 × PBS, 0.1% gelatin, 0.05% Tween 20, 0.1% BSA, was subsequently incubated 30 min at RT. After fixation with 1% PFA for 5 min, nuclei were stained with DAPI (1:1000). Cells were then mounted with a mounting medium (Fluoromount-G, SouthernBiotech, Birmingham, AL, USA) using coverslips.
To test if the anti-KIZ antibody is indeed able to detect the respective protein by immunoblotting and to investigate if KIZ shows a different protein profile in fibroblasts of patients compared to control upon Western blot analysis, the following procedures were performed: confluent transfected and untransfected COS-1 cells and fibroblasts of the patient CIC01225 and control were washed in 1 × PBS and collected by scrapping. After a short centrifugation (800 rpm for 5 min), the pellet cells were resuspended in lysis buffer (50 mM Tris pH 7.5, 150 mM NaCl, 1% Triton-X100), incubated at 4 °C for 30 min with intermittent vortexing and sonication three times for ten-seconds bursts. After centrifugation at 14,000 × g for 10 min, 20 μg of the clarified lysates were loaded on an SDS-gel and visualized with the anti-c-myc-mouse antibody (1:500) (11667149001, Roche) or the anti-KIZ polyclonal rabbit antibody (1:1000) (SAB2700541, Sigma-Aldrich) and respective secondary anti- mouse and anti-rabbit HRP-antibodies (1:10,000) (Jackson Immuno Research) using standard Western blot protocols.
While the mouse anti-PLK1S1 antibody recognized a 76-kDa protein, the predicted size of the full-length protein [
14], the human anti-KIZ antibody recognized a protein of approximately 100 KDa. It was argued that this discrepancy is probably due to the amino acid composition of KIZ rather than any posttranslational modification [
16].
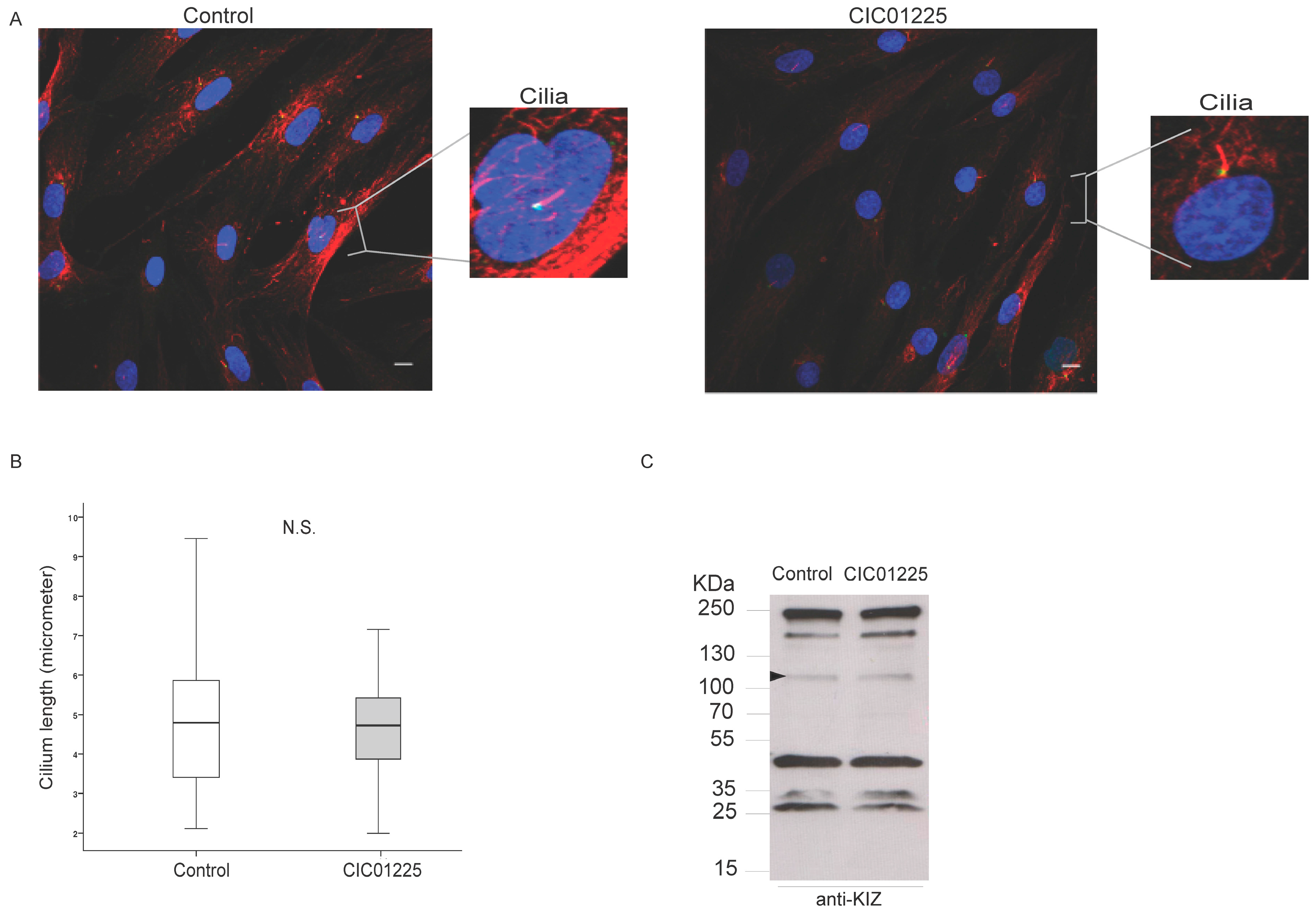
2.8. Human Fibroblasts Staining and Cilia Length Measurements
Fibroblasts of CIC01225, Family C (737) and a non-mutated control were derived from human skin biopsies and were cultured in a specific medium (DMEM, 10% FBS, 2% sodium pyruvate and 1% penicillin streptomycin; Life technologies, Carlsbad, CA, USA) or in the same medium without antibiotics, in a humidified 37 °C, 5% CO
2 incubator. After two passages, we induced monocilia formation in these fibroblasts; cell cultures were deprived of fetal bovine serum for 24 h prior to fixation; and immunostaining experiments were carried out in triplicates and performed as described previously [
9]. Briefly, anti-human KIZ (1:250) and anti-acetylated-α-tubulin (1:1000) antibodies were applied to human fibroblasts with subsequent standard secondary antibodies in addition to DAPI (1:1000) counterstaining. Cells were then mounted with a mounting medium (Mowiol preparation, Sigma) using coverslips.
Cilium length was measured using a software (ImageJ software, Bethesda, MD, USA) [
17]. The mounted preparations were used to capture 10 fields of cells on the confocal microscope giving data for approximately 6 cilia per field of view.
2.9. Preparation of Mouse Retinas
Six-week-old C57BL/6JRj (Janvier, Genest Saint Isle, France) mice were euthanized by CO
2 administration and cervical dislocation. Eyes were removed and prepared as follows: a hole just behind the ora serrata was made, and the eyeballs were placed in 4% (
w/v) PFA in 0.12 M phosphate buffer, pH 7.2 for 1 h at RT. The lens was removed, and the eyecups were fixed again for 20 min in 4% PFA and cryoprotected with increasing concentrations of ice-cold sucrose in 0.12 M phosphate buffer, pH 7.2 (10% for 1 h and 30% overnight). Subsequently, the eyecups were embedded in 7.5% gelatin, 10% sucrose 1 × PBS and frozen in a dry ice-cooled isopentane bath. Subsequently, 10-μm retinal sections were made with a cryostat (model HM560; Thermo Fisher; Microm, Walldorf, Germany) [
18], mounted with a mounting medium (Mowiol preparation, Sigma) on glass slides (Super-Frost, Thermo Fisher Scientific, Waltham, MA, USA) and kept at −80 °C.
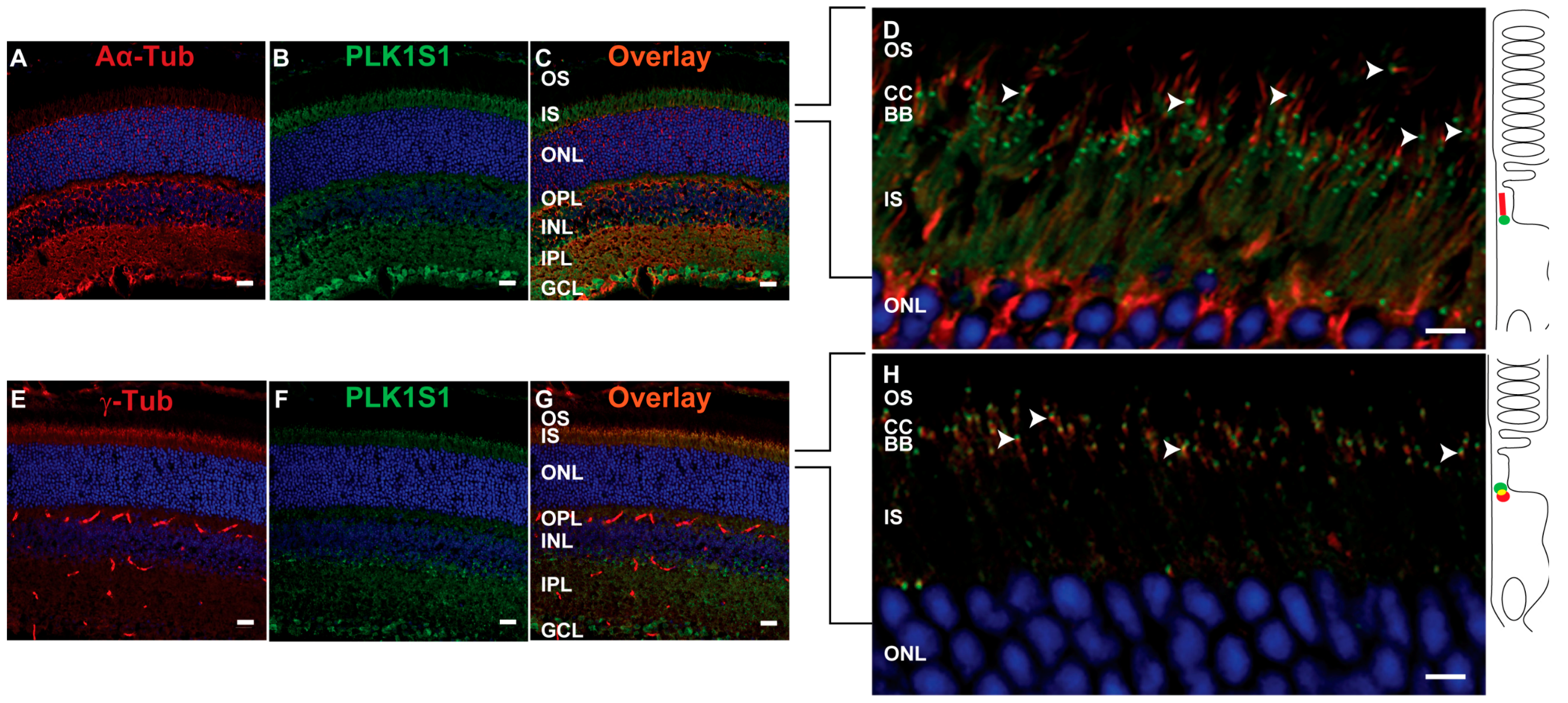
2.10. PLKLS1 Localization in Mouse Retina
Mouse cryosections were post-fixed in methanol at −20 °C for 5 min and then blocked with 5% BSA, 0.1% Triton X-100, 0.1% heat-inactivated goat serum and 1 × PBS for 1 h at RT. An overnight incubation at 4 °C with anti-mouse PLK1S1 (1:500), anti-α-acetylated and anti-γ-tubulin (1:1000 was performed prior to washing 3 times for 30 min each in PBTrit (PBS and 0.1% Triton X-100). Donkey secondary antibodies anti-rabbit Alexa Fluor 488 (1:1000) and donkey anti-mouse Cy3 (1:1000) or donkey anti-mouse Alexa Fluor 647 (1:1000), DAPI (1:1000) and lectin PNA conjugate 594 (1:1000) were used during an overnight incubation at 4 °C [
14]. Negative controls were performed with the use of secondary antibodies alone. Sections were washed 3 times for 30 min in PBTrit and then mounted with a mounting medium (Mowiol preparation, Sigma) using coverslips.
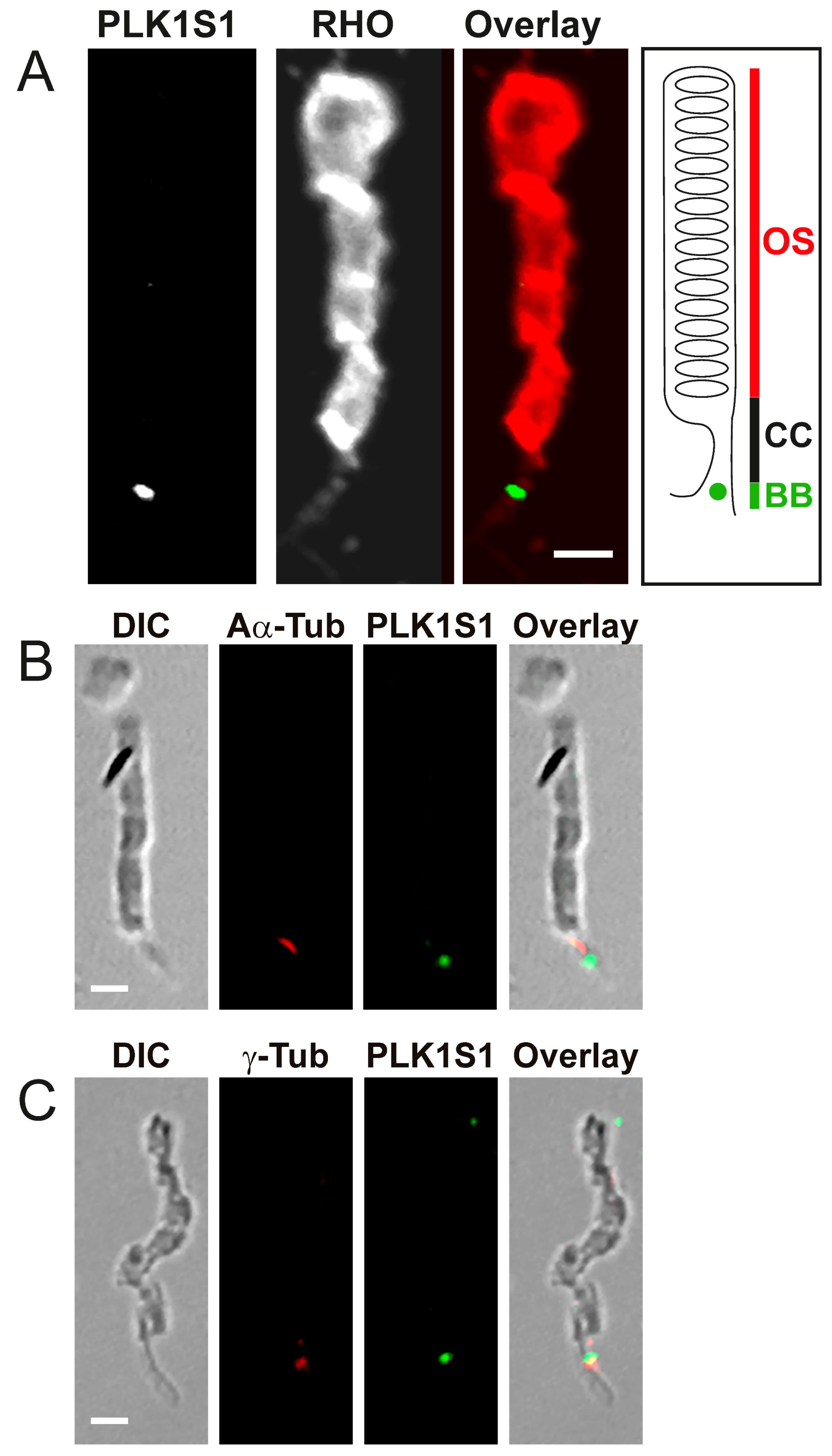
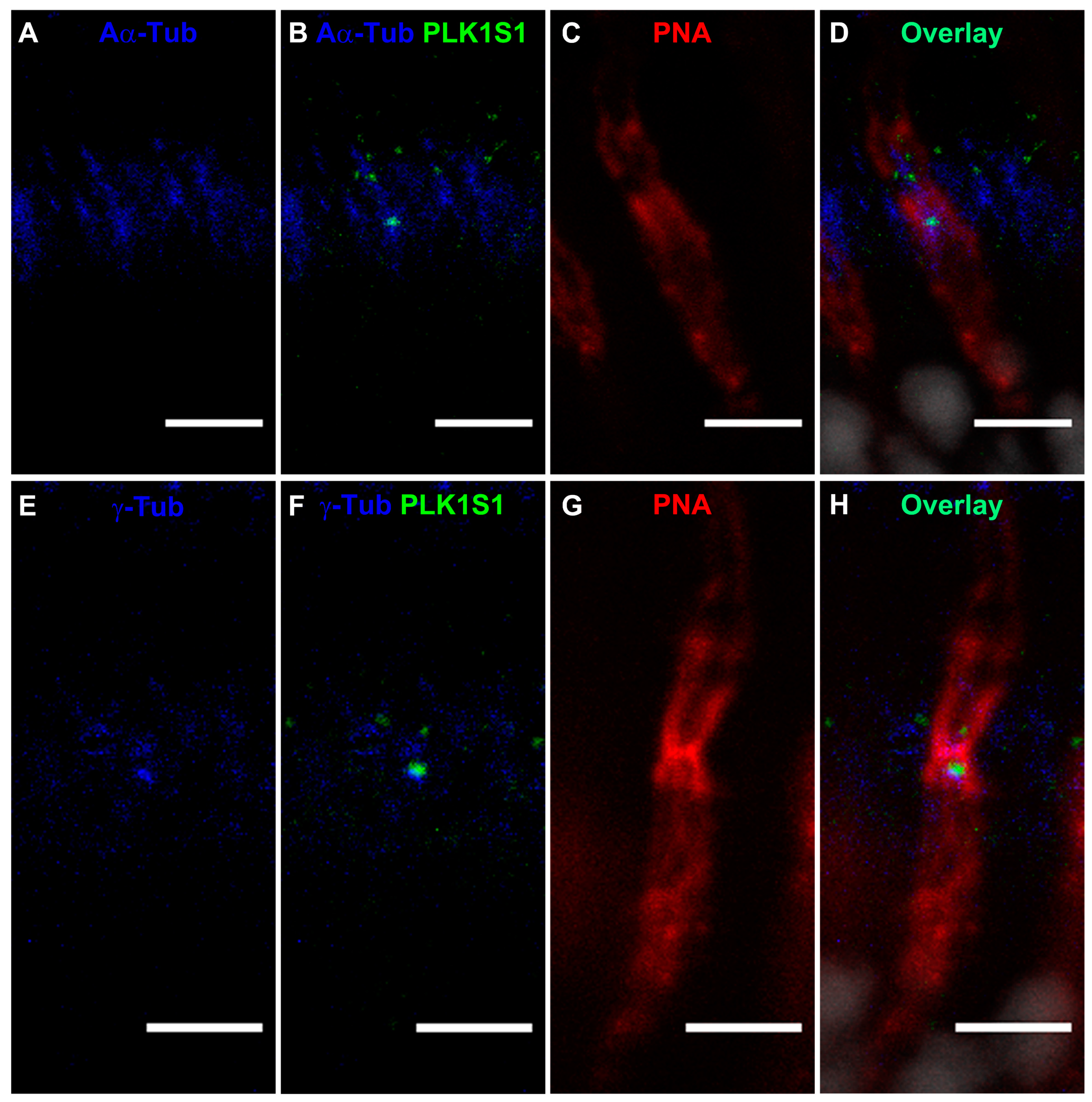
2.11. PLK1S1 Localization in Isolated Mouse Photoreceptors
Fresh mouse retinas were gently vortexed for 30 s in 400 μL of 1 × PBS with calcium; this step allows a separation of the photoreceptor sensory cilium (PSC) complexes from the remainder of the retina. The PSC complexes were collected from the upper fraction of the supernatant using a wide-open pipette transferred on a glass slide (Super-Frost, Thermo Fisher Scientific) and fixed with methanol at −20 °C for 5 min. After fixation, PSC were blocked in 0.2% gelatin, 0.1% Triton X-100 and in 1 × PBS for 1 h at RT. Staining was achieved following overnight incubation at 4 °C with anti-mouse PLK1S1 (1:250) and anti-rhodopsin (1:250), anti-mouse PLK1S1 (1:250) and anti-α-acetylated (1:500) or anti-γ-tubulin (1:1000). After incubation, PSC were washed twice in PBT (1 × PBS and 0.1% Tween 20) and stained with anti-rabbit Alexa Fluor 488 (1:1000) and anti-mouse Cy3 (1:1000) for 1 h at RT and then mounted with a mounting medium (Mowiol preparation, Sigma) on coverslips.
4. Discussion
We recently identified three different mutations in
KIZ; c.226C>T (p.Arg76*), c.119_122del (p.Lys40Ilefs*14) and c.52G>T (p.Glu18*) in three patients with arRCD [
9].
KIZ codes for a 673-amino acid protein representing a centrosomal substrate of Polo-like kinase-1 that mediates mitotic chromosomal stabilization [
16]. In 2008, Tang and co-workers [
14] reported that the mouse ortholog PLK1S1 (called Gm114 at this time) is expressed in male germ cells at early stages of embryogenesis, in spermatocytes and in spermatids of adult mouse testis, suggesting an important role in mammalian germ line stem cell self-renewal and differentiation [
14]. The role of KIZ or its mouse ortholog PLK1S1 in retina function was less well understood. Quantitative real-time PCR in human tissues showed that the
KIZ transcript was most abundant in the retina, followed by the retinal pigment epithelium, whole blood cells and fibroblasts. Immunofluorescent staining of serum-starved human fibroblasts showed KIZ immunolocalization to the basal body of monocilia [
9]. Additional in silico transcriptomic analyses in mice revealed that the mouse ortholog
Plk1s1 is expressed in rod photoreceptors and ex in vivo studies showed its expression in the outer nuclear layer of the mouse retina [
9]. Herein, we show that PLK1S1 protein is also localized in the mouse retina and more precisely at the BB of the CC in rod photoreceptors, but most likely also in cone photoreceptors. Due to their presence on a wide variety of mammalian cells (including human), defects in cilia metabolism are associated with an increased number of Mendelian and complex diseases [
22]. These comprise a heterogeneous group of pathologies including inherited retinal diseases [
4], cystic kidney disease [
23], infertility [
24], chronic respiratory problems, hypertension [
25] and obesity [
25]. It is remarkable that half of the implicated genes encoding ciliary proteins are localized in the BB of photoreceptors. This observation underlies the necessity of maintaining an adequate bridge-like structure required for a correct trafficking process and serving as a gate for proteins destined for the ciliary transport [
26]. Since the CC, of which the core of the cilium extends from the BB, is the only intracellular passage between the IS and OS of the photoreceptors [
26], all molecules have to traffic from the synthesis location within the cell body and the IS through the CC to reach the OS. This trafficking (import/export) is thought to be regulated by proteins arranged in networks at the BB [
2]. The localization of PLK1S1 at the BB suggests its putative involvement in the regulation of this complex transport network.
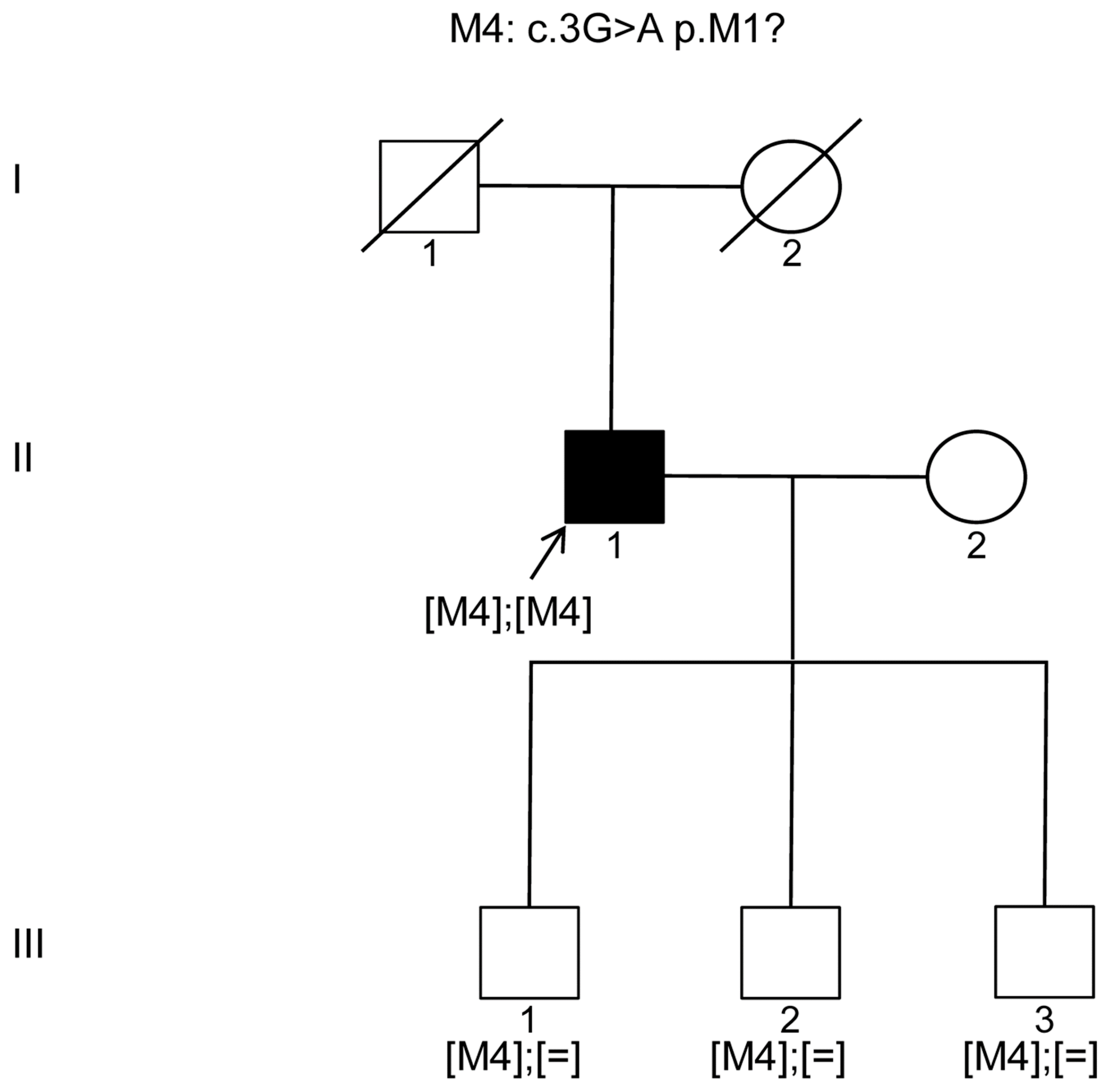
Our studies herein revealed two additional patients with most likely homozygous mutations in
KIZ; one patient harbored the previously identified mutation c.226C>T (p.Arg76*) [
9] and the other one the c.3G>A (p.Met1?) affecting presumably the start codon of
KIZ. Recently, the same c.226C>T (p.Arg76*) mutation was detected in two sisters with RCD from an Ashkenazi Jewish pedigree [
19]. To our knowledge, our work on
KIZ mutations is only the third report documenting mutations in arRCD patients. This might be due to the fact that the prevalence of this gene defect seems to be rare and perhaps only a few research groups have added this gene on their targeted NGS panel. However, our study highlights the importance of adding this gene to such high throughput screening panels especially when screening Ashkenazi Jewish and Latino populations.
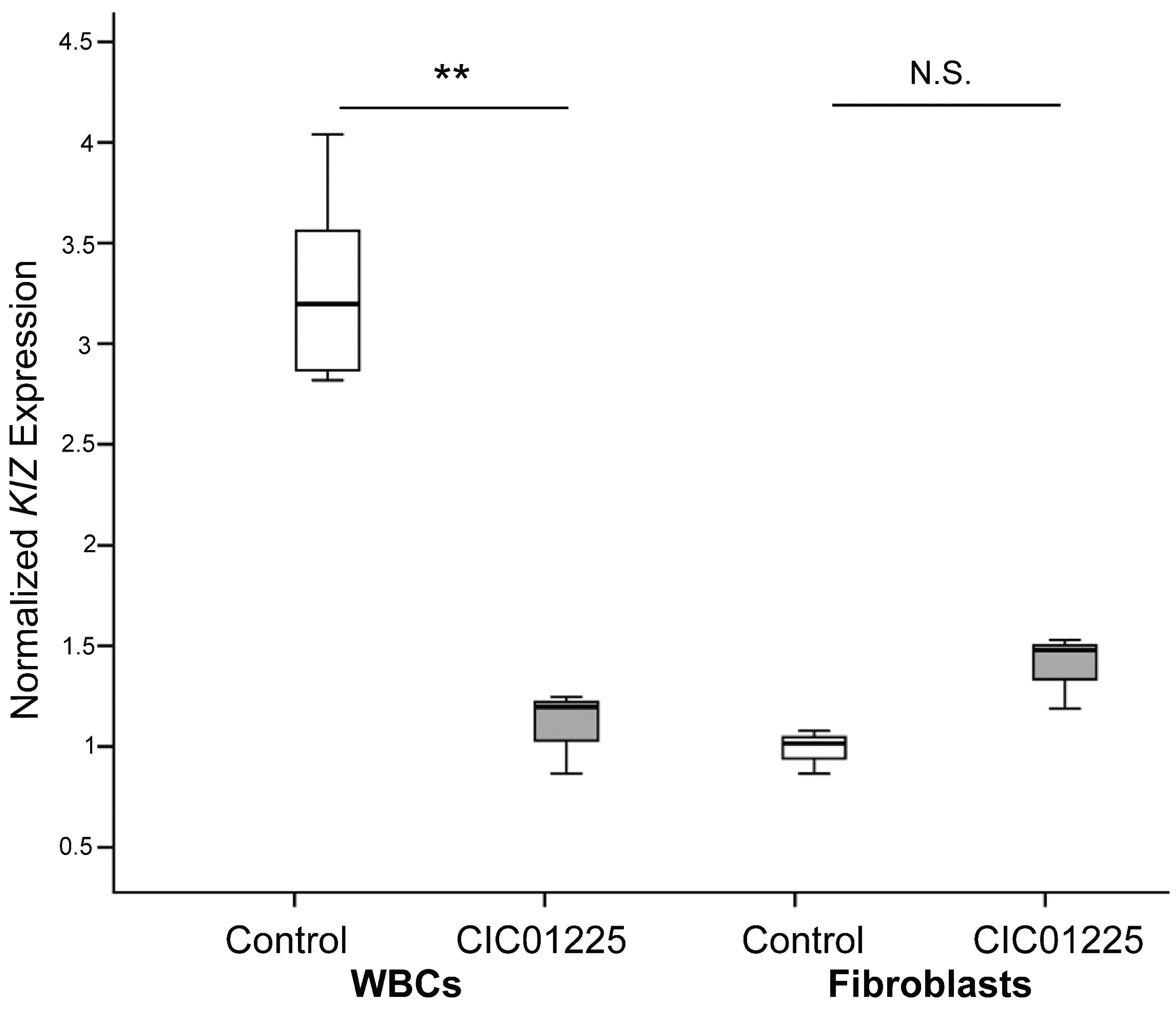
Furthermore, our studies showed that human fibroblasts derived from patients carrying truncating mutations in
KIZ are most likely not a good model to study the pathogenic mechanism of this gene defect. This holds true for expression, protein localization and functional analyses. In most cases, mutations leading to a premature termination codon tend to trigger nonsense-mediated mRNA decay (NMD); however, in other cases, NMD remains inactive, resulting in a truncated protein with a premature termination codon. Albeit we showed that
KIZ expression in whole blood cells was three-times lower in a patient with truncating mutations compared to controls, no significant difference of
KIZ expression was detected in mutant
KIZ patient-derived fibroblasts compared to controls. Since
KIZ mRNA levels in fibroblasts did not significantly differ between the affected individual and the control, we speculate that the phenomenon of NMD is not active in this tissue under the in vitro condition we used. Interestingly, our previous expression studies revealed already that
KIZ expression is quite low in fibroblasts < blood < RPE < retina with the highest expression in retina [
9], further confirming the possibility that fibroblasts are most likely not the best in vitro system to study the pathogenic mechanism of mutations in
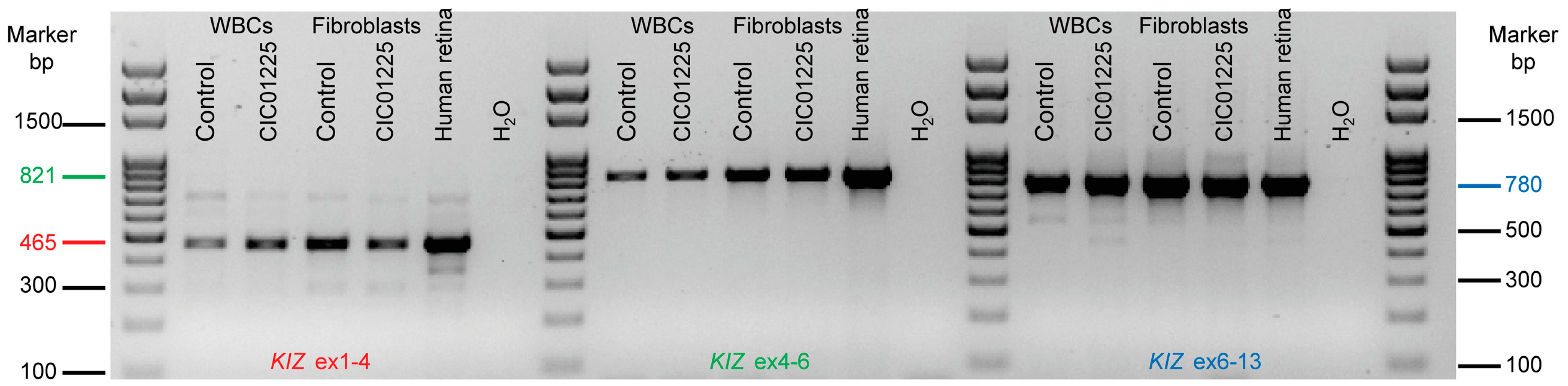
KIZ. This might be due to tissue-specific transcripts. For
KIZ, seven different transcripts have been described of which only transcript 1 (NM 018474.4) and transcript 5 (NM_001352434), of which part of exon 7 is missing, are predicted to be affected by the mutations found in exons 1 and 2 in the studied patient. To investigate, if tissue specific transcripts may account for the unaltered
KIZ expression observed in patient fibroblasts compared to control, the presence of transcript 1 was analyzed in blood, fibroblasts and retina. All tissues revealed indeed transcript 1 at high expression levels as shown by RT-PCR experiments, arguing against the hypothesis that in fibroblasts transcript 1 is not present and, thus, the mutations do not affect the expression. However, we cannot exclude that other transcripts are also present albeit at lower expression levels, rescuing the phenotype in fibroblasts. At the same time, in order to investigate the function and pathogenic mechanism on protein level in human fibroblasts, immunolocalization studies, cilia length measurements and immunoblot analyses were performed at the same time with a commercially available antibody. This antibody directed against the human KIZ protein revealed immunolabeling and a band at ~100 KDa upon Western blot analyses in cells overexpressing a tagged protein, which was absent in untransfected cells documenting that this antibody can indeed detect KIZ in this system. Immunolabeling and a band at ~100 KDa upon Western blot in HeLa cells, which could be blocked by small interfering RNA-treated cells, had also previously been reported for a different KIZ antibody [
16]. However, when we applied the antibody that we checked in overexpressing cells, we could detect KIZ not only in control’s, but also in patient's fibroblasts, localizing it at the basal body of monocilia. In addition, cilia length did not differ between case and control samples (
p > 0.05). Similarly, the same protein profile in patient- and control-derived fibroblast cells upon immunoblotting was detected showing among others a band at ~100 KDa. Mass spectrometry investigation of the band needs to be done to confirm if this band represents indeed human KIZ.
Therefore, the question remains, why KIZ would be still present in patient fibroblasts albeit the truncating mutations. NMD was first described in 1979 in human cells and in yeast as implicated in eliminating truncated proteins [
27]. In mammalian cells, its main function is to reduce errors in gene expression by eliminating mRNA transcripts that contain premature termination codons [
28] at more than 50–55 bp upstream of an exon-exon junction. Mechanistic studies reported that NMD efficiency can vary between cell types [
29], tissue types [
30] and individuals [
31]. Furthermore, it was also shown that nonsense mutations in the first exon of certain genes (such as
Beta Globin) do not activate NMD [
32,
33] since essential NMD components cannot be recruited. According to the 50–55-bp rule, both c.52G>T, p.Glu18* and c.119_122del, p.Lys40Ilefs*14 in KIZ may escape the NMD process as they are less than 50 bp from the exon-exon junction. Interestingly, c.52G>T, p.Glu18* mutation is located in the first exon of KIZ. However, if these mutants escape NMD, this would result in very short proteins, most likely not detectable with the anti-KIZ antibody, directed against the C-terminal region of the protein.
On the other hand, a premature termination codon read-through could occur in fibroblasts from patient and control, subsequently restoring a full-length protein in patient’s cells, especially since our fibroblast cells were initially cultured in a media containing 1% of aminoglycosides antibiotics (see Materials and Methods, e.g., streptomycin). This hypothesis is supported by the work of Schwarz and co-workers [
34] on induced pluripotent stem cells (iPSC)-derived retinal pigment epithelium cells from an Xl RCD patient, which showed that aminoglycosides (G418) and other molecules (PTC124) can promote read-through of the RP2 p.Arg120* mutation and thus restore
RP2 function [
34]. However, when we performed these studies using aminoglycoside-free media, we could not see any difference with respect to protein localization, cilia length and protein expression between mutant and control cells.
An alternative explanation would be that other isoforms, not affected by the two mutations found in the studied patient, but also detected by the antibody are still formed in fibroblasts, and thus, the phenotype is not observed in this system. Indeed the anti-KIZ antibody is a rabbit polyclonal antibody produced with a recombinant human KIZ fragment corresponding to the region between amino acids 187 and 438 of the human protein. These amino acids are present in all seven isoforms. Again, if indeed other transcripts are present in addition to transcript 1, albeit at lower levels and the respective mRNA translated, the respective proteins could be detected by this antibody. This finding urges for a specific antibody directed against isoform 1.
Although, we did not detect any physical effect on ciliogenesis in fibroblasts, we cannot exclude that mutations in
KIZ affect the cilia structure or function in the human retina itself. Despite the presence of cilia in a variety of tissues [
35], mutant ciliary proteins may have various impacts on their stability or their consequences may be variable in maintaining cellular homeostasis among these different tissues [
36,
37]. This variability may explain the restricted retinal phenotype found in our RCD patients carrying
KIZ mutations [
9] and other cases of isolated retinopathies linked to mutations on other ciliary proteins [
38].
Future directions would be to generate human pluripotent stem cells from fibroblasts with subsequent differentiation in retina-like structure in vitro [
39]. Such a step may provide a more relevant in vitro model of the disease and unravel the molecular and cellular mechanisms associated with mutations in
KIZ and other ciliary proteins underlying RCD.
While the present study provides new insights into the molecular properties of KIZ and its mouse ortholog, future in vivo (animal models) and in vitro (human pluripotent stem cells) studies will help to understand its precise role in the retina and the associated pathogenic mechanism(s) leading to this form of RCD.