Willing to Be Involved in Cancer
Abstract
:1. History
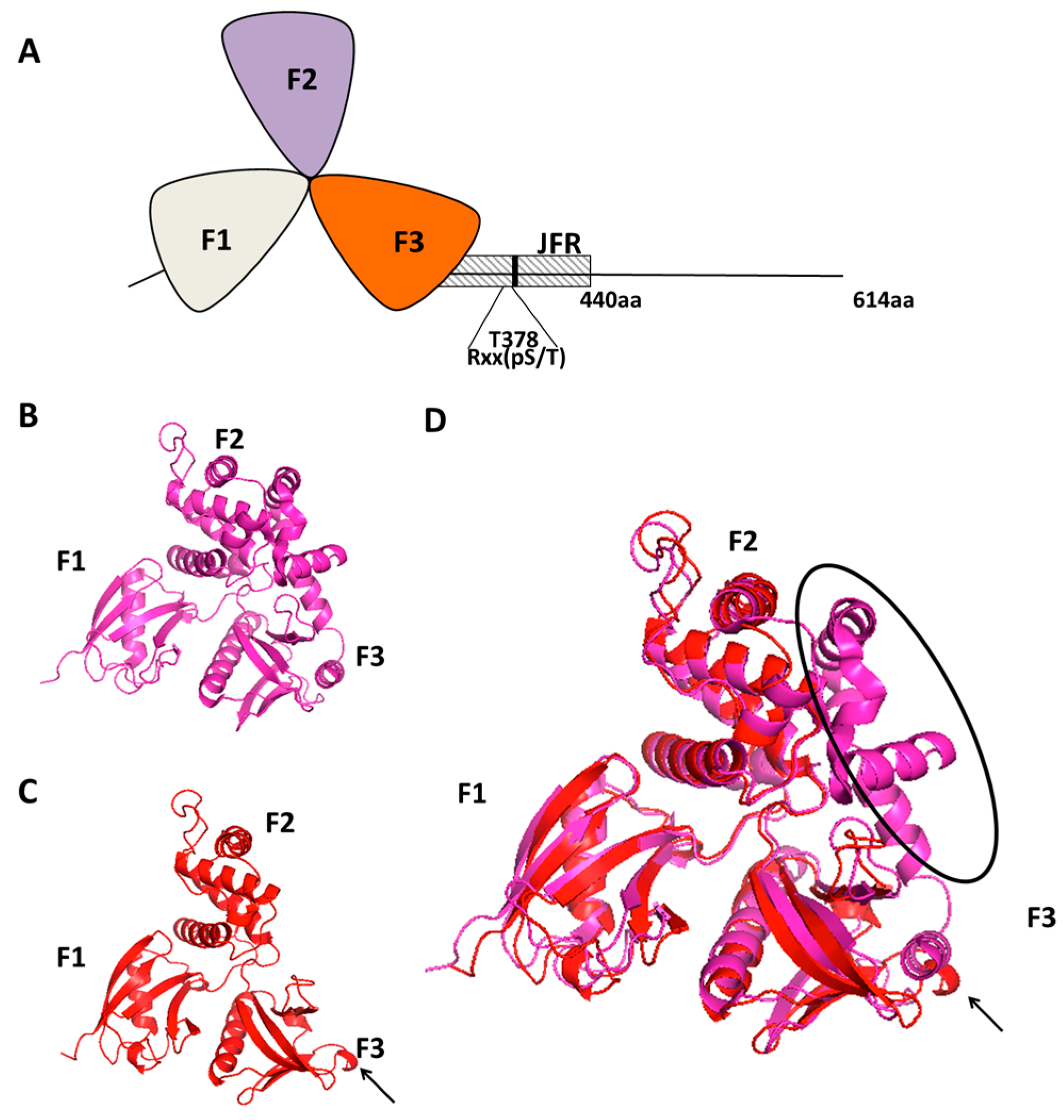
2. Willin, the Protein
3. Willin and Signalling Pathways
3.1. Hippo Pathway
3.2. Ex Is Not Fully Conserved in Mammalian Willin/FRMD6
3.3. Willin Associates with Junctional Components
3.4. Connections to the Actin Cytoskeleton
4. Willin and Cancer
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 3D-SIM | three-dimensional structured illumination microscopy |
| aPKC | atypical Protein Kinase C |
| AJ | adherens junction |
| CASPR | contactin associated protein |
| Ed | Echinoid |
| Ex | Expanded |
| EMT | epithelial-mesenchymal transition |
| FAK | focal adhesion kinase |
| FERM | Four point one, ezrin, radixin, moesin |
| HEK | human embryonic kidney |
| HM | Hippo pathway-interacting motif |
| TJ | tight junction |
| Y2H | yeast two hybrid |
References
- Charles, P.; Tait, S.; Faivre-Sarrailh, C.; Barbin, G.; Gunn-Moore, F.; Denisenko-Nehrbass, N.; Guennoc, A.M.; Girault, J.A.; Brophy, P.J.; Lubetzki, C. Neurofascin is a glial receptor for the paranodin/caspr-contactin axonal complex at the axoglial junction. Curr. Biol. 2002, 12, 217–220. [Google Scholar] [CrossRef]
- Tait, S.; Gunn-Moore, F.; Collinson, J.M.; Huang, J.; Lubetzki, C.; Pedraza, L.; Sherman, D.L.; Colman, D.R.; Brophy, P.J. An oligodendrocyte cell adhesion molecule at the site of assembly of the paranodal axo-glial junction. J. Cell Biol. 2000, 150, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Garver, T.D.; Ren, Q.; Tuvia, S.; Bennett, V. Tyrosine phosphorylation at a site highly conserved in the l1 family of cell adhesion molecules abolishes ankyrin binding and increases lateral mobility of neurofascin. J. Cell Biol. 1997, 137, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Gunn-Moore, F.J.; Hill, M.; Davey, F.; Herron, L.R.; Tait, S.; Sherman, D.; Brophy, P.J. A functional ferm domain binding motif in neurofascin. Mol. Cell. Neurosci. 2006, 33, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Bretscher, A. Purification of the intestinal microvillus cytoskeletal proteins villin, fimbrin, and ezrin. Methods Enzymol. 1986, 134, 24–37. [Google Scholar] [PubMed]
- Gould, K.L.; Bretscher, A.; Esch, F.S.; Hunter, T. Cdna cloning and sequencing of the protein-tyrosine kinase substrate, ezrin, reveals homology to band 4.1. EMBO J. 1989, 8, 4133–4142. [Google Scholar] [PubMed]
- Gould, K.L.; Cooper, J.A.; Bretscher, A.; Hunter, T. The protein-tyrosine kinase substrate, p81, is homologous to a chicken microvillar core protein. J. Cell Biol. 1986, 102, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Gunn-Moore, F.J.; Welsh, G.I.; Herron, L.R.; Brannigan, F.; Venkateswarlu, K.; Gillespie, S.; Brandwein-Gensler, M.; Madan, R.; Tavare, J.M.; Brophy, P.J.; et al. A novel 4.1 ezrin radixin moesin (ferm)-containing protein, ‘willin’. FEBS Lett. 2005, 579, 5089–5094. [Google Scholar] [CrossRef] [PubMed]
- Moleirinho, S.; Tilston-Lunel, A.; Angus, L.; Gunn-Moore, F.; Reynolds, P.A. The expanding family of ferm proteins. Biochem. J. 2013, 452, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Venkateswarlu, K.; Gunn-Moore, F.; Oatey, P.B.; Tavare, J.M.; Cullen, P.J. Nerve growth factor- and epidermal growth factor-stimulated translocation of the adp-ribosylation factor-exchange factor grp1 to the plasma membrane of pc12 cells requires activation of phosphatidylinositol 3-kinase and the grp1 pleckstrin homology domain. Biochem. J. 1998, 335 Pt 1, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Venkateswarlu, K.; Gunn-Moore, F.; Tavare, J.M.; Cullen, P.J. Egf-and ngf-stimulated translocation of cytohesin-1 to the plasma membrane of pc12 cells requires pi 3-kinase activation and a functional cytohesin-1 ph domain. J. Cell Sci. 1999, 112 Pt 12, 1957–1965. [Google Scholar] [PubMed]
- Sato, N.; Yonemura, S.; Obinata, T.; Tsukita, S.; Tsukita, S. Radixin, a barbed end-capping actin-modulating protein, is concentrated at the cleavage furrow during cytokinesis. J. Cell Biol. 1991, 113, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Brannigan, F. Characterisation of “Willin”, a Sciatic Nerve-Derived Novel Member of the Ferm Domain-Containing Family of Proteins. Ph.D. Thesis, University of St Andrews, St Andrews, UK, 2006. [Google Scholar]
- Madan, R.; Brandwein-Gensler, M.; Schlecht, N.F.; Elias, K.; Gorbovitsky, E.; Belbin, T.J.; Mahmood, R.; Breining, D.; Qian, H.; Childs, G.; et al. Differential tissue and subcellular expressionof erm proteins in normal and malignant tissues: Cytoplasmic ezrin expression has prognostic signficance for head and neck squamous cell carcinoma. Head Neck 2006, 28, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Schlecht, N.F.; Brandwein-Gensler, M.; Smith, R.V.; Kawachi, N.; Broughel, D.; Lin, J.; Keller, C.E.; Reynolds, P.A.; Gunn-Moore, F.J.; Harris, T.; et al. Cytoplasmic ezrin and moesin correlate with poor survival in head and neck squamous cell carcinoma. Head Neck Pathol. 2012, 6, 232–243. [Google Scholar] [CrossRef] [PubMed]
- Hamaratoglu, F.; Willecke, M.; Kango-Singh, M.; Nolo, R.; Hyun, E.; Tao, C.; Jafar-Nejad, H.; Halder, G. The tumour-suppressor genes nf2/merlin and expanded act through hippo signalling to regulate cell proliferation and apoptosis. Nat. Cell Biol. 2006, 8, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, P.A.; Smolen, G.A.; Palmer, R.E.; Sgroi, D.; Yajnik, V.; Gerald, W.L.; Haber, D.A. Identification of a DNA-binding site and transcriptional target for the ews-wt1(+kts) oncoprotein. Genes Dev. 2003, 17, 2094–2107. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, P.A.; Sigaroudinia, M.; Zardo, G.; Wilson, M.B.; Benton, G.M.; Miller, C.J.; Hong, C.; Fridlyand, J.; Costello, J.F.; Tlsty, T.D. Tumor suppressor p16ink4a regulates polycomb-mediated DNA hypermethylation in human mammary epithelial cells. J. Biol. Chem. 2006, 281, 24790–24802. [Google Scholar] [CrossRef] [PubMed]
- Harvey, K.; Tapon, N. The salvador-warts-hippo pathway—An emerging tumour-suppressor network. Nat. Rev. Cancer 2007, 7, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Harvey, K.F.; Zhang, X.; Thomas, D.M. The hippo pathway and human cancer. Nat. Rev. Cancer 2013, 13, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Pan, D. Hippo signaling in organ size control. Genes Dev. 2007, 21, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Genevet, A.; Wehr, M.C.; Brain, R.; Thompson, B.J.; Tapon, N. Kibra is a regulator of the salvador/warts/hippo signaling network. Dev. Cell 2010, 18, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zheng, Y.; Dong, J.; Klusza, S.; Deng, W.M.; Pan, D. Kibra functions as a tumor suppressor protein that regulates hippo signaling in conjunction with merlin and expanded. Dev. Cell 2010, 18, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Badouel, C.; Gardano, L.; Amin, N.; Garg, A.; Rosenfeld, R.; Le Bihan, T.; McNeill, H. The ferm-domain protein expanded regulates hippo pathway activity via direct interactions with the transcriptional activator yorkie. Dev. Cell 2009, 16, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Boedigheimer, M.; Laughon, A. Expanded: A gene involved in the control of cell proliferation in imaginal discs. Development 1993, 118, 1291–1301. [Google Scholar] [PubMed]
- McCartney, B.M.; Kulikauskas, R.M.; LaJeunesse, D.R.; Fehon, R.G. The neurofibromatosis-2 homologue, merlin, and the tumor suppressor expanded function together in drosophila to regulate cell proliferation and differentiation. Development 2000, 127, 1315–1324. [Google Scholar] [PubMed]
- Nguyen, R.; Reczek, D.; Bretscher, A. Hierarchy of merlin and ezrin N- and C-terminal domain interactions in homo- and heterotypic associations and their relationship to binding of scaffolding proteins ebp50 and e3karp. J. Biol. Chem. 2001, 276, 7621–7629. [Google Scholar] [CrossRef] [PubMed]
- Pellock, B.J.; Buff, E.; White, K.; Hariharan, I.K. The drosophila tumor suppressors expanded and merlin differentially regulate cell cycle exit, apoptosis, and wingless signaling. Dev. Biol. 2007, 304, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Pan, D. The hippo signaling pathway in development and cancer. Dev. Cell 2010, 19, 491–505. [Google Scholar] [CrossRef] [PubMed]
- Angus, L.; Moleirinho, S.; Herron, L.; Sinha, A.; Zhang, X.; Niestrata, M.; Dholakia, K.; Prystowsky, M.B.; Harvey, K.F.; Reynolds, P.A.; et al. Willin/FRMD6 expression activates the hippo signaling pathway kinases in mammals and antagonizes oncogenic yap. Oncogene 2012, 31, 238–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumgartner, R.; Poernbacher, I.; Buser, N.; Hafen, E.; Stocker, H. The ww domain protein kibra acts upstream of hippo in drosophila. Dev. Cell 2010, 18, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Genevet, A.; Tapon, N. The hippo pathway and apico-basal cell polarity. Biochem. J. 2011, 436, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Moleirinho, S.; Chang, N.; Sims, A.H.; Tilston-Lunel, A.M.; Angus, L.; Steele, A.; Boswell, V.; Barnett, S.C.; Ormandy, C.; Faratian, D.; et al. Kibra exhibits mst-independent functional regulation of the hippo signaling pathway in mammals. Oncogene 2013, 32, 1821–1830. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Chen, Y.; Ji, M.; Dong, J. Kibra regulates hippo signaling activity via interactions with large tumor suppressor kinases. J. Biol. Chem. 2011, 286, 7788–7796. [Google Scholar] [CrossRef] [PubMed]
- Yue, T.; Tian, A.; Jiang, J. The cell adhesion molecule echinoid functions as a tumor suppressor and upstream regulator of the hippo signaling pathway. Dev. Cell 2012, 22, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Bossuyt, W.; Chen, C.L.; Chen, Q.; Sudol, M.; McNeill, H.; Pan, D.; Kopp, A.; Halder, G. An evolutionary shift in the regulation of the hippo pathway between mice and flies. Oncogene 2014, 33, 1218–1228. [Google Scholar] [CrossRef] [PubMed]
- Moleirinho, S.; Patrick, C.; Tilston-Lunel, A.M.; Higginson, J.R.; Angus, L.; Antkowiak, M.; Barnett, S.C.; Prystowsky, M.B.; Reynolds, P.A.; Gunn-Moore, F.J. Willin, an upstream component of the hippo signaling pathway, orchestrates mammalian peripheral nerve fibroblasts. PLoS ONE 2013, 8, e60028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Visser-Grieve, S.; Hao, Y.; Yang, X. Human homolog of drosophila expanded, hex, functions as a putative tumor suppressor in human cancer cell lines independently of the hippo pathway. Oncogene 2012, 31, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Ishiuchi, T.; Takeichi, M. Willin and par3 cooperatively regulate epithelial apical constriction through apkc-mediated rock phosphorylation. Nat. Cell Biol. 2011, 13, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Ishiuchi, T.; Takeichi, M. Nectins localize willin to cell-cell junctions. Genes Cells 2012, 17, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Huttlin, E.L.; Ting, L.; Bruckner, R.J.; Gebreab, F.; Gygi, M.P.; Szpyt, J.; Tam, S.; Zarraga, G.; Colby, G.; Baltier, K.; et al. The bioplex network: A systematic exploration of the human interactome. Cell 2015, 162, 425–440. [Google Scholar] [CrossRef] [PubMed]
- Rual, J.F.; Venkatesan, K.; Hao, T.; Hirozane-Kishikawa, T.; Dricot, A.; Li, N.; Berriz, G.F.; Gibbons, F.D.; Dreze, M.; Ayivi-Guedehoussou, N.; et al. Towards a proteome-scale map of the human protein-protein interaction network. Nature 2005, 437, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.L.; Gajewski, K.M.; Hamaratoglu, F.; Bossuyt, W.; Sansores-Garcia, L.; Tao, C.; Halder, G. The apical-basal cell polarity determinant crumbs regulates hippo signaling in drosophila. Proc. Nat. Acad. Sci. USA 2010, 107, 15810–15815. [Google Scholar] [CrossRef] [PubMed]
- Ling, C.; Zheng, Y.; Yin, F.; Yu, J.; Huang, J.; Hong, Y.; Wu, S.; Pan, D. The apical transmembrane protein crumbs functions as a tumor suppressor that regulates hippo signaling by binding to expanded. Proc. Nat. Acad. Sci. USA 2010, 107, 10532–10537. [Google Scholar] [CrossRef] [PubMed]
- Robinson, B.S.; Huang, J.; Hong, Y.; Moberg, K.H. Crumbs regulates salvador/warts/hippo signaling in drosophila via the ferm-domain protein expanded. Curr. Biol. 2010, 20, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Bennett, F.C.; Harvey, K.F. Fat cadherin modulates organ size in drosophila via the salvador/warts/hippo signaling pathway. Curr. Biol. 2006, 16, 2101–2110. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Feng, Y.; Rauskolb, C.; Maitra, S.; Fehon, R.; Irvine, K.D. Delineation of a fat tumor suppressor pathway. Nat. Genet. 2006, 38, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.; Tsatskis, Y.; Gardano, L.; Tapon, N.; McNeill, H. The tumor-suppressor gene fat controls tissue growth upstream of expanded in the hippo signaling pathway. Curr. Biol. 2006, 16, 2081–2089. [Google Scholar] [CrossRef] [PubMed]
- Willecke, M.; Hamaratoglu, F.; Kango-Singh, M.; Udan, R.; Chen, C.L.; Tao, C.; Zhang, X.; Halder, G. The fat cadherin acts through the hippo tumor-suppressor pathway to regulate tissue size. Curr. Biol. 2006, 16, 2090–2100. [Google Scholar] [CrossRef] [PubMed]
- Tilston-Lünel, A.M.; Haley, K.; Schlecht, N.; Wang, Y.; Chatterton, A.L.D.; Moleirinho, S.; Watson, A.; Hundal, H.S.; Prystowsky, M.B.; Gunn-Moore, F.J.; et al. Crumbs3b promotes tight junctions in an ezrin-dependent manner in mammalian cells. J. Mol. Cell Biol. 2016. [Google Scholar] [CrossRef]
- Gaspar, P.; Tapon, N. Sensing the local environment: Actin architecture and hippo signalling. Curr. Opin. Cell Biol. 2014, 31, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Solon, J.; Levental, I.; Sengupta, K.; Georges, P.C.; Janmey, P.A. Fibroblast adaptation and stiffness matching to soft elastic substrates. Biophys. J. 2007, 93, 4453–4461. [Google Scholar] [CrossRef] [PubMed]
- Densham, R.M.; O’Neill, E.; Munro, J.; Konig, I.; Anderson, K.; Kolch, W.; Olson, M.F. Mst kinases monitor actin cytoskeletal integrity and signal via c-jun N-terminal kinase stress-activated kinase to regulate p21waf1/cip1 stability. Mol. Cell. Biol. 2009, 29, 6380–6390. [Google Scholar] [CrossRef] [PubMed]
- Romano, D.; Nguyen, L.K.; Matallanas, D.; Halasz, M.; Doherty, C.; Kholodenko, B.N.; Kolch, W. Protein interaction switches coordinate raf-1 and mst2/hippo signalling. Nat. Cell Biol. 2014, 16, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, B.G.; Gaspar, P.; Bras-Pereira, C.; Jezowska, B.; Rebelo, S.R.; Janody, F. Actin-capping protein and the hippo pathway regulate f-actin and tissue growth in drosophila. Development 2011, 138, 2337–2346. [Google Scholar] [CrossRef] [PubMed]
- Sansores-Garcia, L.; Bossuyt, W.; Wada, K.; Yonemura, S.; Tao, C.; Sasaki, H.; Halder, G. Modulating f-actin organization induces organ growth by affecting the hippo pathway. EMBO J. 2011, 30, 2325–2335. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Gonzalez, R.; Zallen, J.A. Cell mechanics and feedback regulation of actomyosin networks. Sci. Signal. 2009. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of yap/taz in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Wada, K.; Itoga, K.; Okano, T.; Yonemura, S.; Sasaki, H. Hippo pathway regulation by cell morphology and stress fibers. Development 2011, 138, 3907–3914. [Google Scholar] [CrossRef] [PubMed]
- Varelas, X.; Samavarchi-Tehrani, P.; Narimatsu, M.; Weiss, A.; Cockburn, K.; Larsen, B.G.; Rossant, J.; Wrana, J.L. The crumbs complex couples cell density sensing to hippo-dependent control of the tgf-beta-smad pathway. Dev. Cell 2010, 19, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Wei, X.; Li, W.; Udan, R.S.; Yang, Q.; Kim, J.; Xie, J.; Ikenoue, T.; Yu, J.; Li, L.; et al. Inactivation of yap oncoprotein by the hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 2007, 21, 2747–2761. [Google Scholar] [CrossRef] [PubMed]
- Aragona, M.; Panciera, T.; Manfrin, A.; Giulitti, S.; Michielin, F.; Elvassore, N.; Dupont, S.; Piccolo, S. A mechanical checkpoint controls multicellular growth through yap/taz regulation by actin-processing factors. Cell 2013, 154, 1047–1059. [Google Scholar] [CrossRef] [PubMed]
- Moleirinho, S. Mammalian Upstream Hippo Signalling Pathway Proteins Activate Core Pathway Kinases and Functionally Antagonize Oncogenic YAP. Ph.D. Thesis, University of St Andrews, St Andrews, UK, 2013. [Google Scholar]
- Knights, A.J.; Funnell, A.P.; Crossley, M.; Pearson, R.C. Holding tight: Cell junctions and cancer spread. Trends Cancer Res. 2012, 8, 61–69. [Google Scholar] [PubMed]
- Forster, C. Tight junctions and the modulation of barrier function in disease. Histochem. Cell Biol. 2008, 130, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Runkle, E.A.; Mu, D. Tight junction proteins: From barrier to tumorigenesis. Cancer Lett. 2013, 337, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Turksen, K.; Troy, T.C. Junctions gone bad: Claudins and loss of the barrier in cancer. Biochim. Biophys. Acta 2011, 1816, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Acloque, H.; Adams, M.S.; Fishwick, K.; Bronner-Fraser, M.; Nieto, M.A. Epithelial-mesenchymal transitions: The importance of changing cell state in development and disease. J. Clin. Investig. 2009, 119, 1438–1449. [Google Scholar] [CrossRef] [PubMed]
- Nieto, M.A. Epithelial-mesenchymal transitions in development and disease: Old views and new perspectives. Int. J. Dev. Biol. 2009, 53, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, R.P.; Veltman, D.; Machesky, L.M. Actin-bundling proteins in cancer progression at a glance. J. Cell Sci. 2012, 125, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, R.D.; Angelo, L.S.; Kurzrock, R. Nf2/merlin in hereditary neurofibromatosis 2 versus cancer: Biologic mechanisms and clinical associations. Oncotarget 2014, 5, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.T.; Chen, X.L.; Lim, Y.; Hanson, D.A.; Vo, T.T.; Howerton, K.; Larocque, N.; Fisher, S.J.; Schlaepfer, D.D.; Ilic, D. Nuclear fak promotes cell proliferation and survival through ferm-enhanced p53 degradation. Mol. Cell 2008, 29, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Clucas, J.; Valderrama, F. Erm proteins in cancer progression. J. Cell Sci. 2014, 127, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Kong, J.; Li, Y.; Liu, S.; Jin, H.; Shang, Y.; Quan, C.; Li, Y.; Lin, Z. High expression of ezrin predicts poor prognosis in uterine cervical cancer. BMC Cancer 2013. [Google Scholar] [CrossRef] [PubMed]
- Elzagheid, A.; Korkeila, E.; Bendardaf, R.; Buhmeida, A.; Heikkila, S.; Vaheri, A.; Syrjanen, K.; Pyrhonen, S.; Carpen, O. Intense cytoplasmic ezrin immunoreactivity predicts poor survival in colorectal cancer. Hum. Pathol. 2008, 39, 1737–1743. [Google Scholar] [CrossRef] [PubMed]
- Park, H.R.; Min, S.K.; Min, K.; Jun, S.Y.; Seo, J.; Kim, K.H.; Choi, J. Differential expression of ezrin in epithelial skin tumors: Cytoplasmic ezrin immunoreactivity in squamous cell carcinoma. Int. J. Dermatol. 2010, 49, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Hoover, K.B.; Bryant, P.J. The genetics of the protein 4.1 family: Organizers of the membrane and cytoskeleton. Curr. Opin. Cell Biol. 2000, 12, 229–234. [Google Scholar] [CrossRef]
- Herron, L.R. A Study of the Behaviour and Interactions of the Novel Ferm Protein Willin. Ph.D. Thesis, University of St Andrews, St Andrews, UK, 2008. [Google Scholar]


© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gunn-Moore, F.J.; Tilston-Lünel, A.M.; Reynolds, P.A. Willing to Be Involved in Cancer. Genes 2016, 7, 37. https://doi.org/10.3390/genes7070037
Gunn-Moore FJ, Tilston-Lünel AM, Reynolds PA. Willing to Be Involved in Cancer. Genes. 2016; 7(7):37. https://doi.org/10.3390/genes7070037
Chicago/Turabian StyleGunn-Moore, Frank J., Andrew M. Tilston-Lünel, and Paul A. Reynolds. 2016. "Willing to Be Involved in Cancer" Genes 7, no. 7: 37. https://doi.org/10.3390/genes7070037
APA StyleGunn-Moore, F. J., Tilston-Lünel, A. M., & Reynolds, P. A. (2016). Willing to Be Involved in Cancer. Genes, 7(7), 37. https://doi.org/10.3390/genes7070037





